Convenient Synthesis and Biological Evaluation of Modafinil Derivatives: Benzhydrylsulfanyl and Benzhydrylsulfinyl [1,2,3]triazol-4-yl-methyl Esters
Abstract
:1. Introduction
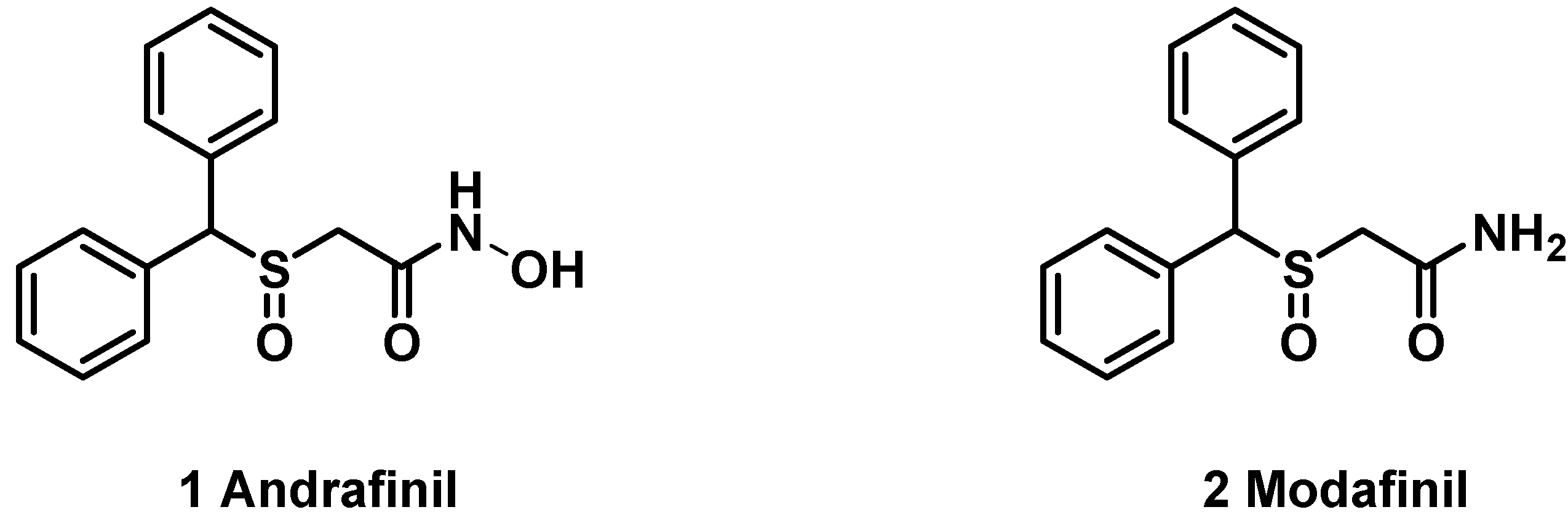
2. Results and Discussion
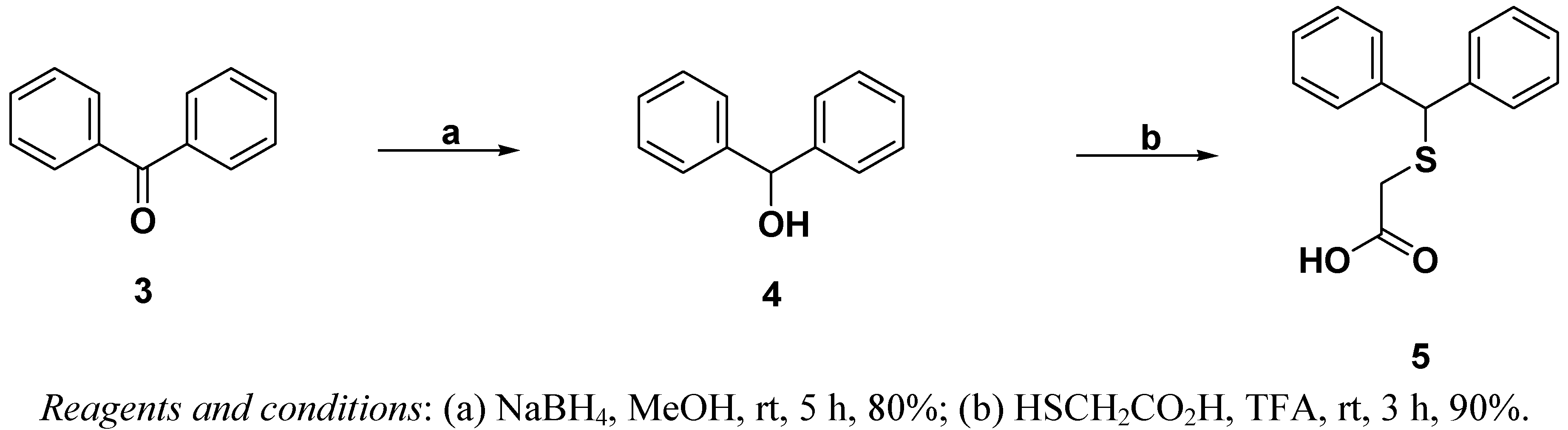
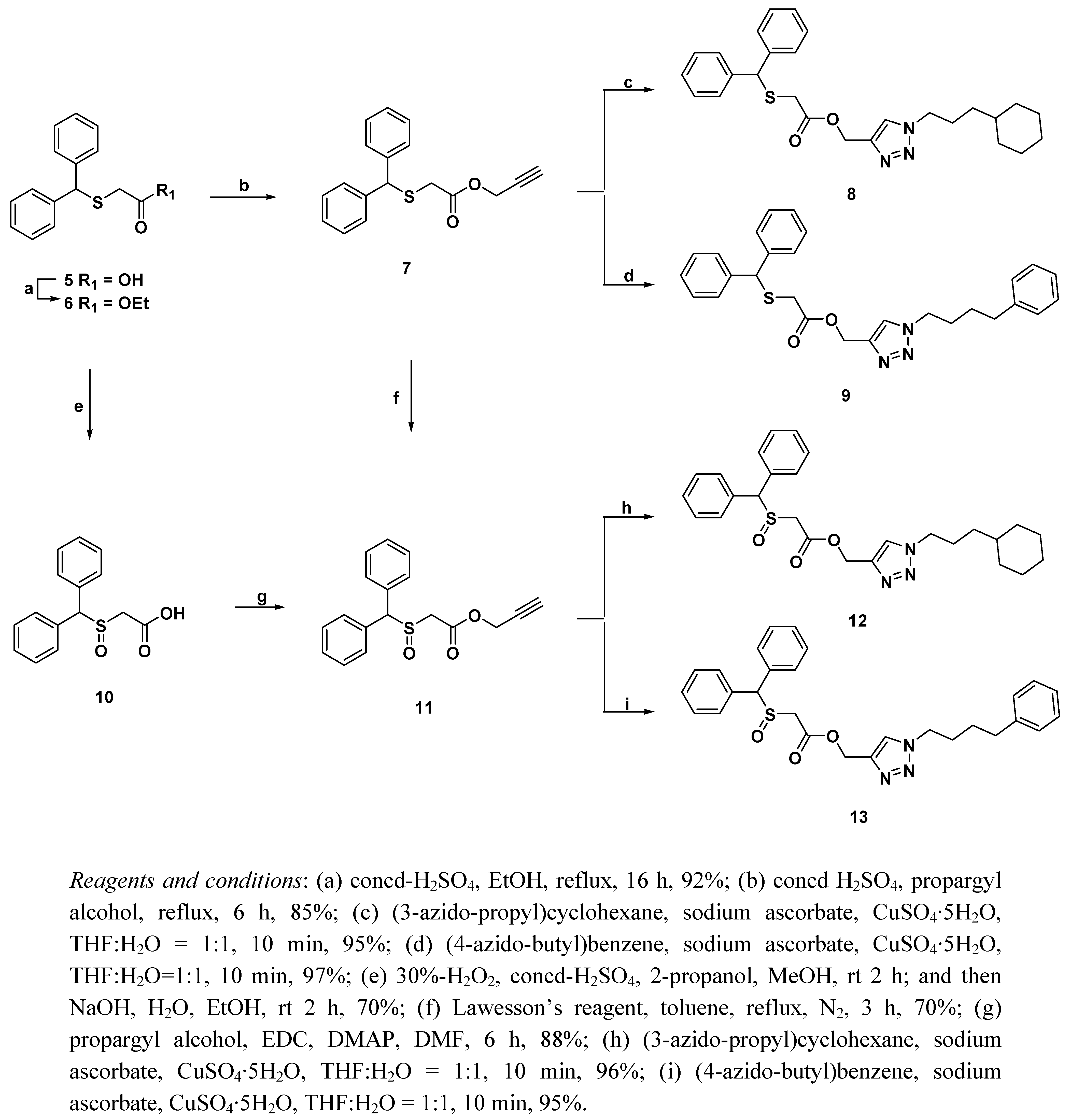
2.1. Chemistry
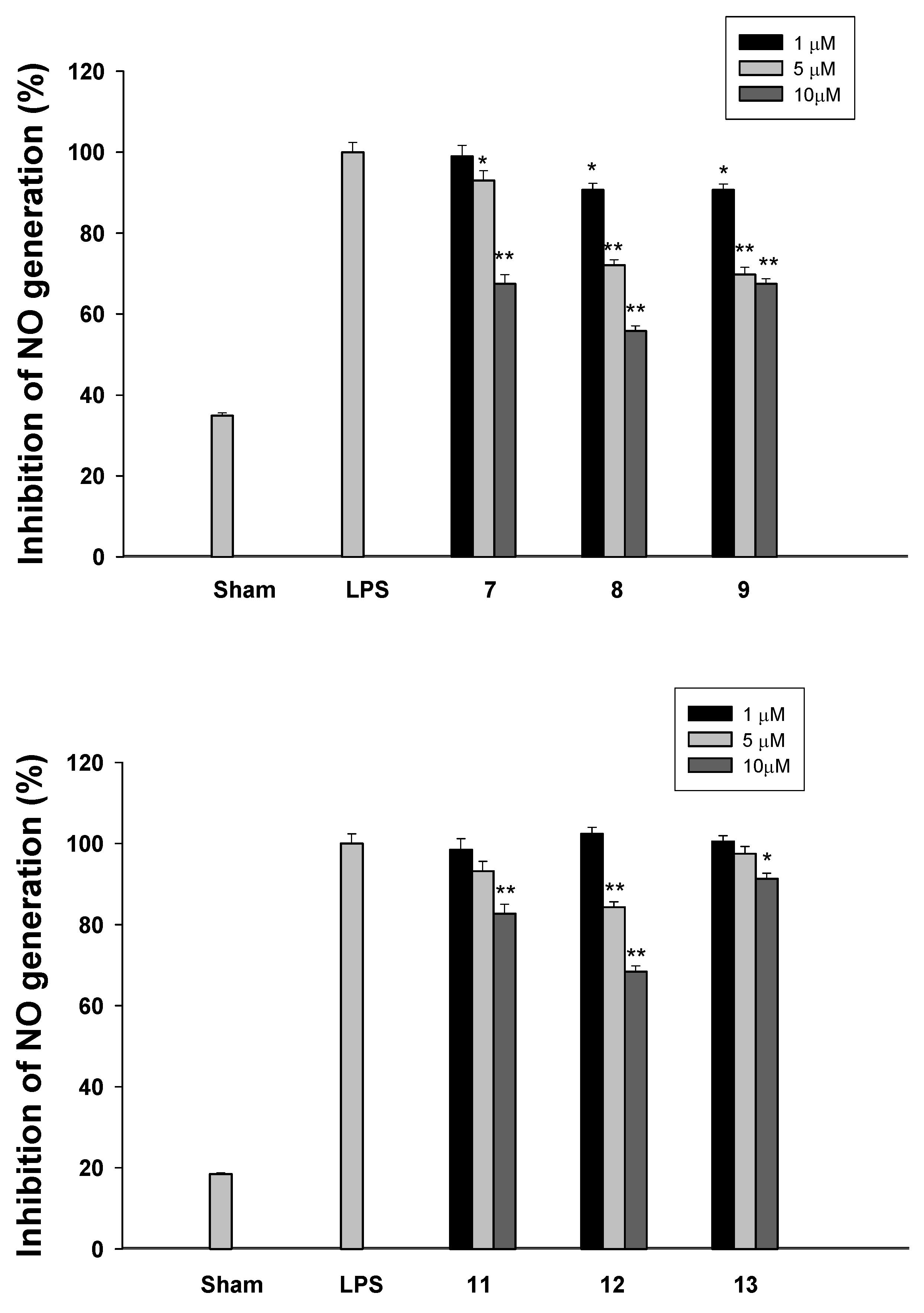
2.2. Biology
3. Experimental Section
3.1. General Experimental Procedure for Preparation of Compounds 7 and 11
3.2. General Experimental Procedure for Preparation of Compounds 8, 9, 12, and 13
3.3. BV-2 Microglia Culture
3.4. Nitric Oxide Generation Assay
4. Conclusions
Acknowledgments
References and Notes
- Robertson, P.; DeCory, H.H.; Madan, A.; Parkinson, A. In vitro inhibition and induction of human hepatic cytochrome P450 enzymes by modafinil. Drug Metab. Dispos. 2000, 28, 664–671. [Google Scholar]
- van Vliet, S.A.; Blezer, E.L.; Jongsma, M.J.; Vanwersch, R.A.; Olivier, B.; Philippens, I.H. Exploring the neuroprotective effects of modafinil in a marmoset Parkinson model with immunohistochemistry, magnetic resonance imaging and spectroscopy. Brain Res. 2008, 1189, 219–228. [Google Scholar] [CrossRef]
- van Vliet, S.A.; Vanwersch, R.A.; Jongsma, M.J.; van der Gugten, J.; Olivier, B.; Philippens, I.H. Neuroprotective effects of modafinil in a marmoset Parkinson model: Behavioral and neurochemical aspects. Behav. Pharmacol. 2006, 17, 453–462. [Google Scholar] [CrossRef]
- Chatterjie, N.; Stables, J.P.; Wang, H.; Alexander, G.J. Anti-narcoleptic agent modafinil and its sulfone: A novel facile synthesis and potential anti-epileptic activity. Neurochem. Res. 2004, 29, 1481–1486. [Google Scholar] [CrossRef]
- De Risi, C.; Ferraro, L.; Pollini, G.P.; Tanganelli, S.; Valente, F.; Veronese, A.C. Efficient synthesis and biological evaluation of two modafinil analogues. Bioorg. Med. Chem. 2008, 16, 9904–9910. [Google Scholar] [CrossRef]
- Ballon, J.S.; Feifel, D. A systematic review of modafinil: Potential clinical uses and mechanisms of action. J. Clin. Psychiat. 2006, 67, 554–566. [Google Scholar] [CrossRef]
- Swanson, J.M.; Greenhill, L.L.; Lopez, F.A.; Sedillo, A.; Earl, G.Q.; Jiang, J.G.; Biederman, J. Modafinil film-coated tablets in children and adolescents with attention-deficit/hyperactivity disorder: results or randomized, double-blind, placebo-controlled, fixed-dose astudy followed by abrupt discontinuation. J. Clin. Psychiat. 2006, 67, 137–147. [Google Scholar] [CrossRef]
- Campos, M.P.; Hassan, B.J.; Riechelmann, R.; Del Giglio, A. Cancer-related fatigue: A review. Rev. Assoc. Med. Bras. 2011, 57, 211–219. [Google Scholar] [CrossRef]
- Pelissier-Alicot, A.-L.; Piercecchi-Marti, M.-D.; Bartoli, C.; Kuhlmann, E.; Coiffait, P.-E.; Sanvoisin, A.; Giocanti, D.; Leonetti, G. Abusive prescription of psychostimulants: a study of two cases. J. Forensic Sci. 2006, 51, 407–410. [Google Scholar] [CrossRef]
- Chatterjie, N.; Stables, J.P.; Wang, H.; Alexander, G.J. Anti-narcoleptic agent modafinil and its sulfone: A novel facile synthesis and potential anti-epileptic activity. Neurochem. Res. 2004, 29, 1481–1486. [Google Scholar] [CrossRef]
- De Risi, C.; Ferraro, L.; Pollini, G.P.; Tanganelli, S.; Valente, F.; Veronese, A.C. Efficient synthesis and biological evaluation of two modafinil analogues. Bioorg. Med. Chem. 2008, 6, 9904–9910. [Google Scholar]
- Zhou, J.; He, R.; Johnson, K.M.; Ye, Y.P.; Kozikowski, A.P. Piperidine-based nocaine/modafinil hybrid ligands as highly potent monoamine transporter inhibitors: Efficient drug discovery by rational lead hybridization. J. Med. Chem. 2004, 47, 5821–5824. [Google Scholar] [CrossRef]
- Kraft, G.H.; Brown, J. Modafinil for fatigue in MS: A randomized placebo-controlled double-blind study. Neurology 2005, 65, 1995–1997. [Google Scholar] [CrossRef]
- Nieves, A.V.; Lang, A.E. Treatment of excessive daytime sleepiness in patients with Parkinson’s disease with modafinil. Clin. Neuropharmacol. 2002, 25, 111–114. [Google Scholar] [CrossRef]
- Swanson, J.M.; Greenhill, L.L.; Lopez, F.A.; Sedillo, A.; Earl, G.Q.; Jiang, J.G.; Biederman, J. Modafinil film-coated tablets in children and adolescents with attention-deficit/hyperactivity disorder: results or randomized, double-blind, placebo-controlled, fixed-dose astudy followed by abrupt discontinuation. J. Clin. Psychiat. 2006, 67, 137–147. [Google Scholar] [CrossRef]
- Vocci, F.J.; Elkashef, A. Pharmacotherapy and other treatments for cocaine abuse and dependence. Curr. Opin. Psychiat. 2005, 18, 265–270. [Google Scholar] [CrossRef]
- Olivo, H.F.; Osorio-Lozada, A.; Peeples, T.L. Microbial oxidation/amidation of benzhydrylsulfanyl acetic acid. Synthesis of (+)-modafinil. Tetrahedron: Asymmetry 2005, 16, 3507–3511. [Google Scholar] [CrossRef]
- Osorio-Lozada, A.; Prisinzano, T.; Olivo, H.F. Synthesis and determination of the absolute stereochemistry of the enantiomers of adrafinil and modafinil. Tetrahedron: Asymmetry 2004, 15, 3811–3815. [Google Scholar] [CrossRef]
- Minzenberg, M.J.; Carter, C.S. Modafinil: A review of neurochemical actions and effects on cognition. Neuropsychopharmacology 2008, 33, 1477–1502. [Google Scholar] [CrossRef]
- Prisinzano, T.; Podobinski, J.; Tidgewell, K.; Luo, M.; Swenson, D. Synthesis and determination of the absolute configuration of the enantiomers modafinil. Tetrahedron: Asymmetry 2004, 15, 1053–1058. [Google Scholar] [CrossRef]
- Salama, Z.B. Water soluble esters of [N-(4-amino-2-butynyl)] with anticancer activity. WO 2005095369 A1 2005.
- Rostovtsev, V.V.; Green, L.G.; Fokin, V.V.; Sharpless, K.B. A stepwise Huisgen cycloaddition process: Copper(I)-catalyzed regioselective "ligation" of azides and terminal alkynes. Angew. Chem. Int. Ed. 2002, 41, 2596–2599. [Google Scholar] [CrossRef]
- Amblard, F.; Cho, J.H.; Schinazi, R.F. Cu(I)-catalyzed Huisgen azide-alkyne 1,3-dipolar cycloaddition reaction in nucleoside, nucleotide, and oligonucleotide chemistry. Chem. Rev. 2009, 109, 4207–4220. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnock, J.S.; Tannenbaum, P.S.R. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar]
- Perrin, D.D.; Armarego, W.L.F. Purification of Laboratory Chemicals, 3rd ed; Pergamon Press: Oxford, UK, 1988. [Google Scholar]
- Sample Availability: Samples of the compounds 8-9 and 12-13 are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jung, J.-C.; Lee, Y.; Son, J.-Y.; Lim, E.; Jung, M.; Oh, S. Convenient Synthesis and Biological Evaluation of Modafinil Derivatives: Benzhydrylsulfanyl and Benzhydrylsulfinyl [1,2,3]triazol-4-yl-methyl Esters. Molecules 2011, 16, 10409-10419. https://doi.org/10.3390/molecules161210409
Jung J-C, Lee Y, Son J-Y, Lim E, Jung M, Oh S. Convenient Synthesis and Biological Evaluation of Modafinil Derivatives: Benzhydrylsulfanyl and Benzhydrylsulfinyl [1,2,3]triazol-4-yl-methyl Esters. Molecules. 2011; 16(12):10409-10419. https://doi.org/10.3390/molecules161210409
Chicago/Turabian StyleJung, Jae-Chul, Yeonju Lee, Jee-Young Son, Eunyoung Lim, Mankil Jung, and Seikwan Oh. 2011. "Convenient Synthesis and Biological Evaluation of Modafinil Derivatives: Benzhydrylsulfanyl and Benzhydrylsulfinyl [1,2,3]triazol-4-yl-methyl Esters" Molecules 16, no. 12: 10409-10419. https://doi.org/10.3390/molecules161210409
APA StyleJung, J.-C., Lee, Y., Son, J.-Y., Lim, E., Jung, M., & Oh, S. (2011). Convenient Synthesis and Biological Evaluation of Modafinil Derivatives: Benzhydrylsulfanyl and Benzhydrylsulfinyl [1,2,3]triazol-4-yl-methyl Esters. Molecules, 16(12), 10409-10419. https://doi.org/10.3390/molecules161210409



