Antioxidant, Anti-Glycation and Anti-Inflammatory Activities of Phenolic Constituents from Cordia sinensis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Compounds and Their Biological Activities
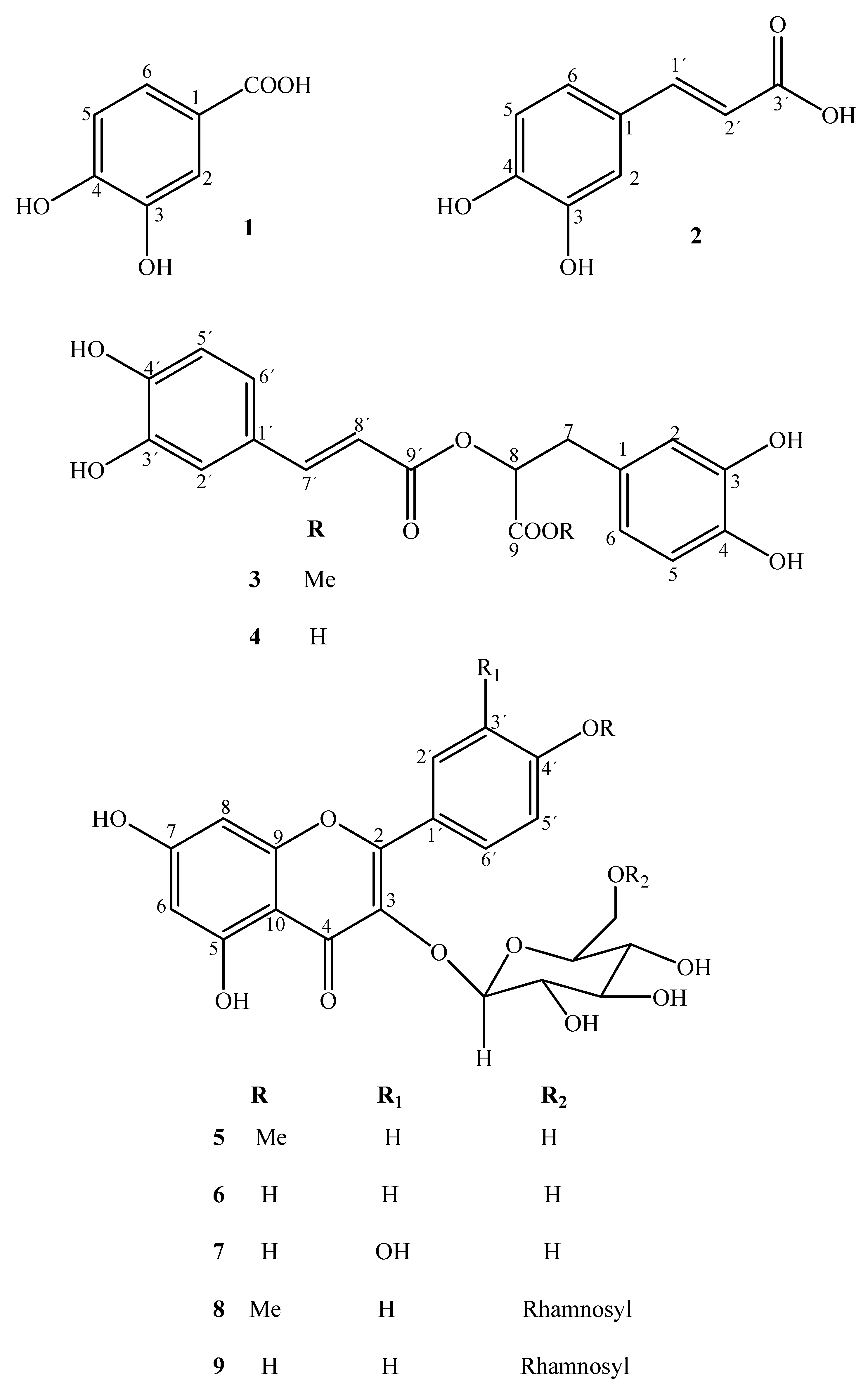
| Compounds | DPPH Scavenging Activity IC50a [μM] |
|---|---|
| 1 | 16.3 ± 0.19 |
| 2 | 14.1 ± 0.14 |
| 3 | 22.7 ± 0.17 |
| 4 | 13.5 ± 0.21 |
| 5 | 55.5 ± 0.12 |
| 6 | 53.4 ± 0.88 |
| 7 | 19.1 ± 0.90 |
| 8 | 42.4 ± 0.91 |
| 9 | 39.5 ± 0.85 |
| BHA b | 44.3 ± 0.09 |
| Compounds | % Inhibition |
|---|---|
| 1 | 68.0 |
| 2 | 69.2 |
| 3 | 88.4 |
| 4 | 87.3 |
| 5 | 76.5 |
| 6 | 74.0 |
| 7 | 71.2 |
| 8 | 80.7 |
| 9 | 79.0 |
| Rutin a | 86.0 |
| Group (3 rats in each) | Treatment 100 mg/kg | Edema Volume (Vc = Vf − V0) | Percent Inhibition (%) |
|---|---|---|---|
| 1 | Cage-1 control | 0 | |
| 2 | Diclofenac Sodium | 0.22 ± 0.05 | 57.6 |
| 3 | 1 | 0.23 ± 0.04 | 55.0 |
| 4 | 2 | 0.26 ± 0.19 | 50.0 |
| 5 | 3 | - | - |
| 6 | 4 | - | - |
| 7 | 5 | 0.24 ± 0.13 | 62.4 |
| 8 | 6 | 0.21 ± 0.11 | 59.6 |
| 9 | 7 | 0.25 ± 0.08 | 51.2 |
| 10 | 8 | 0.35 ± 0.21 | 43.5 |
| 11 | 9 | 0.32 ± 0.07 | 38.4 |
2.2. Chemotaxonomic Significance
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. HPLC Analysis of the Purity of Compounds 2–5, 8 and 9
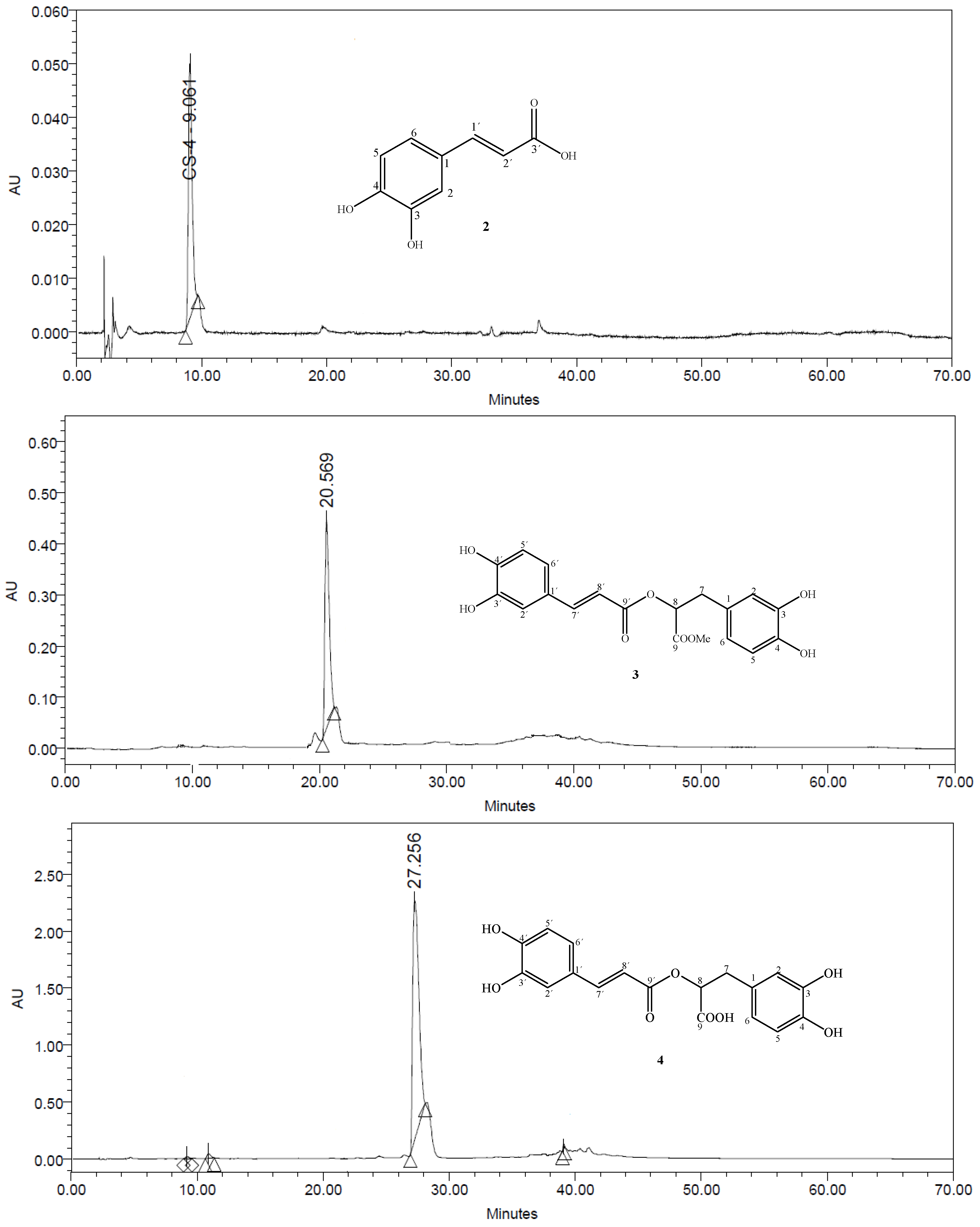

3.5. Spectral Data
4. Conclusions
Conflict of Interest
Acknowledgements
References and Notes
- Thirupathi, K.; Kumar, S.S.; Raju, V.S.; Ravikumar, B.; Krishna, D.R.; Mohan, G.K. A review of medicinal plants of the genus Cordia: Their chemistry and pharmacological uses. J. Nat. Remed. 2008, 8, 1–10. [Google Scholar] [CrossRef]
- Ioset, J.R.; Marston, A.; Gupta, M.P.; Hostettmann, K. Antifungal and larvicidal compounds from the root bark of Cordia alliodora. J. Nat. Prod. 2000, 63, 424–426. [Google Scholar] [CrossRef]
- Menezes, J.A.; Lemos, T.; Pessoa, O.; Braz-Filho, R.; Montenegro, R.; Wilke, D.; Costa-Lotufo, L.; Pessoa, C.; Moraes, M.; Silveira, E. A cytotoxic meroterpenoid benzoquinone from roots of Cordia globosa. Planta Med. 2005, 71, 54–58. [Google Scholar] [CrossRef]
- Jean-Robert, I.; Andrew, M.; Mahabir, P.G.; Kurt, H. Antifungal and larvicidal cordiaquinones from the roots of Cordia curassavica. Phytochemistry 2000, 53, 613–617. [Google Scholar] [CrossRef]
- Medeiros, R.; Passos, G.F.; Vitor, C.E.; Koepp, J.; Mazzuco, T.L.; Pianowski, L.F.; Campos, M.M.; Calixto, J.B. Effect of two active compounds obtained from the essential oil of Cordia verbenacea on the acute inflammatory responses elicited by LPS in the rat paw. Br. J. Pharmacol. 2007, 151, 618–627. [Google Scholar] [CrossRef]
- Ioset, J.R.; Marston, A.; Gupta, M.P.; Hostettmann, K. Antifungal and larvicidal meroterpenoid naphthoquinones and a naphthoxirene from the roots of Cordia linnaei. Phytochemistry 1998, 47, 729–734. [Google Scholar] [CrossRef]
- Ahmed, M.W. Taxonomy and distribution of Cordia sinensis and C. nevillii (Boraginaceae), a widespread species pair in Africa and Asia. Nord. J. Bot. 1990, 9, 649–656. [Google Scholar] [CrossRef]
- Richard, M.M.; Callistus, K.P.O.; Nick, O.O.; Paul, O.O. Antitubercular and phytochemical investigation of methanol extracts of medicinal plants used by the Samburu community in Kenya. Trop. J. Pharm. Res. 2010, 9, 379–385. [Google Scholar]
- Buniyamin, A.A.; Doris, N.O.; Eric, K.O. Isolation and characterization of two phenolics compounds from stem bark of Musanga cecropiodes R. brown (Moraceae). Acta Pol. Pharm. 2007, 64, 183–185. [Google Scholar]
- Nedime, D.; Seckin, O.; Esra, U.; Yasar, D.; Mustafa, K. The Isolation of Carboxylic Acids from the Flowers of Delphinium formosum. Turk. J. Chem. 2001, 25, 93–97. [Google Scholar]
- Eun-Rhan, W.; Mei, S.P. Antioxidative constituents from Lycopus lucidus. Arch. Pharm. Res. 2004, 27, 173–176. [Google Scholar] [CrossRef]
- Lin, L.; Feng-Rui, S.; Rong, T.; Yong-Ri, J.; Zhi-Qiang, L.; Shu-Ying, L. Studies on the homolytic and heterolytic cleavage of kaempferol and kaempferide glycosides using electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Sp. 2010, 24, 169–172. [Google Scholar] [CrossRef]
- Amal, M.Y.M.; Ahmed, I.K.; Mahmoud, A.S. Isolation, structural elucidation of flavonoid constituents from Leptadenia pyrotechnica and evaluation of their toxicity and antitumor activity. Pharm. Biol. 2009, 47, 539–552. [Google Scholar] [CrossRef]
- Choi, W.H.; Park, W.Y.; Hwang, B.Y.; Oh, G.J.; Kang, S.J.; Lee, K.S.; Ro, J.S. Phenolic compounds from the stem bark of Cornus walteri Wagner. Kor. J. Pharmacog. 1998, 29, 217–224. [Google Scholar]
- Kelly, E.H.; Anthony, R.T.; Dennis, J.B. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Saskia, A.B.E.A.; Marcel, J.D.G.; Dirk-Jan, B.; Michel, N.J.L.T.; Gabrielle, D.O.K.; Wim, J.F.V.; Aalt, B. A quantum chemical explanation of the antioxidant activity of flavonoids. Chem. Res. Toxicol. 1996, 9, 1305–1312. [Google Scholar] [CrossRef]
- Gulcin, I.; Alici, H.A.; Cesur, M. Determination of in vitro antioxidant and radical scavenging activities of protocol. Chem. Pharm. Bull. 2005, 53, 281–285. [Google Scholar] [CrossRef]
- Rahbar, S.; Figarola, J.L. Novel inhibitors of advanced glycation endproducts. Arch. Biochem. Biophys. 2003, 419, 63–79. [Google Scholar] [CrossRef]
- Yamaguchi, F.; Ariga, T.; Yoshimura, Y.; Nakazawa, H. Antioxidative and antiglycation activity of garcinol from Garcinia indica fruit rind. J. Agric. Food Chem. 2000, 48, 180–185. [Google Scholar] [CrossRef]
- Kim, H.Y.; Kim, K. Protein glycation inhibitory and antioxidative activities of some plant extracts in vitro. J. Agric. Food Chem. 2003, 51, 1586–1591. [Google Scholar] [CrossRef]
- Christopher, J.M. Inflammation Protocols, carrageenan-induced paw oedema in the rats and mouse. Methods Mol. Biol. 2003, 225, 115–121. [Google Scholar]
- Jane, E.S.A.M.; Telma, L.G.L.; Edilberto, R.S.; Raimundo, B.F.; Otília, D.L.P. Trichotomol, a New Cadinenediol from Cordia trichotoma. J. Braz. Chem. Soc. 2001, 12, 787–790. [Google Scholar] [CrossRef]
- Bina, S.S.; Sobiya, P.; Sabira, B. Two new abietane diterpenes from Cordia latifolia. Tetrahedron 2006, 62, 10087–10090. [Google Scholar] [CrossRef]
- Nakamura, N.; Kojima, S.; Lim, Y.A.; Meselhy, M.R.; Hattori, M.; Gupta, M.P.; Correa, M. Dammarane-type triterpenes from Cordia spinescens. Phytochemistry 1997, 46, 1139–1141. [Google Scholar] [CrossRef]
- Vincent, V.V.; David, L.; Raymond, Z.; Amabile, K.M.; Sylvio, P. Cordialin A and B, two new triterpenes from Cordia verbenacea DC. J. Chem. Soc. Perkin Trans. 1 1982, 2697–2700. [Google Scholar]
- Kuroyanagi, M.; Kawahara, N.; Sekita, S.; Satake, M.; Hayashi, T.; Takase, Y.; Masuda, K. Dammarane-type triterpenes from the Brazilian medicinal plant Cordia multispicata. J. Nat. Prod. 2003, 66, 1307–12. [Google Scholar] [CrossRef]
- Renata, P.S.; Edilberto, R.S.; Daniel, E.A.U.; Otilia, D.L.P.; Francisco, A.V.; Raimundo, B.F. 1H and 13C NMR spectral data of new saponins from Cordia piauhiensis. Magn. Reson. Chem. 2007, 45, 692–694. [Google Scholar] [CrossRef]
- Renata, P.S.; Francisco, A.V.; Telma, L.G.L.; Edilberto, R.S.; Raimundo, B.F.; Otilia, D.L.P. Structure elucidation and total assignment of 1H and 13C NMR data for a new bisdesmoside saponin from Cordia piauhiensis. Magn. Reson. Chem. 2003, 41, 735–738. [Google Scholar] [CrossRef]
- Renata, P.S.; Telma, L.G.L.; Otilia, D.L.P.; Raimundo, B.F.; Edson, R.F.; Francisco, A.V.; Edilberto, R.S. Chemical constituents of Cordia piauhiensis—Boraginaceae. J. Braz. Chem. Soc. 2005, 16, 662–665. [Google Scholar] [CrossRef]
- Fun, C.; Svendsen, A.B. The essential oil of Cordia cylindrostachya Roem. & Schult. grown on Aruba. J. Essen. Oil Res. 1990, 2, 209–210. [Google Scholar] [CrossRef]
- Kuppast, I.J.; Nayak, P.V. Wound healing activity of Cordia dichotoma Forst. f. fruits. Nat. Prod. Rad. 2006, 5, 103–107. [Google Scholar]
- Agnihotri, V.K.; Srivastava, S.D.; Srivastava, S.K.; Pitre, S.; Rusia, K. Constiuents of Cordia obliqua as potential anti-inflammatory agents. Indian J. Pharm. Sci. 1987, 49, 66–69. [Google Scholar]
- Wang, Y.; Ohtani, K.; Kasai, R.; Yamasaki, K. Flavonol glycosides and henolics from leaves of Cordia dichotoma. Nat. Med. 1996, 50, 367. [Google Scholar]
- Sâmia, A.S.S.; Maria, F.A.; Josean, F.T.; Emídio, V.L.C.; Jose, M.B.F.; Marcelo, S.S. Flavanones from aerial parts of Cordia globosa (Jacq.) Kunth, Boraginaceae. Rev. Bras. Farmacogn. 2010, 20, 682–68. [Google Scholar] [CrossRef]
- Afzal, M.; Obuekwe, C.; Khan, A.R.; Barakat, H. Antioxidant activity of Cordia myxa L. and its hepatoprotective potential. Electron. J. Environ. Agric. Food Chem. 2007, 6, 2236–2242. [Google Scholar]
- Parks, J.; Gyeltshen, T.; Prachyawarakorn, V.; Mahidol, C.; Ruchirawat, S.; Kittakoop, P. Glutarimide alkaloids and a terpenoid benzoquinone from Cordia globifera. J. Nat. Prod. 2010, 73, 992–994. [Google Scholar] [CrossRef]
- Samia, A.S.S.; Augusto, L.S.; Maria, F.A.; Emidio, V.L.C.; Jose, M.B.F.; Marcelo, S.S.; Raimundo, B.F. A new arylnaphthalene type lignan from Cordia rufescens A. DC. (Boraginaceae). ARKIVOC 2004, vi, 54–58. [Google Scholar]
- Geller, F.; Schmidt, C.; Gottert, M.; Fronza, M.; Schattel, V.; Heinzmann, B.; Werz, O.; Flores, E.M.; Merfort, I.; Laufer, S. Identification of rosmarinic acid as the major active constituent in Cordia americana. J. Ethnopharmacol. 2010, 128, 561–566. [Google Scholar] [CrossRef]
- Ferrari, F.; Monache, F.D.; Compagnone, R.; Oliveri, M.C. Chemical constituents of Cordia dentata flowers. Fitoterapia 1997, 68, 88. [Google Scholar]
- Sabira, B.; Sobiya, P.; Bina, S.S.; Shazia, K.; Shahina, F.; Musarrat, R. Chemical constituents of Cordia latifolia and their nematicidal activity. Chem. Biodiv. 2011, 8, 850–861. [Google Scholar] [CrossRef]
- Samples Availability: Not available.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Al-Musayeib, N.; Perveen, S.; Fatima, I.; Nasir, M.; Hussain, A. Antioxidant, Anti-Glycation and Anti-Inflammatory Activities of Phenolic Constituents from Cordia sinensis. Molecules 2011, 16, 10214-10226. https://doi.org/10.3390/molecules161210214
Al-Musayeib N, Perveen S, Fatima I, Nasir M, Hussain A. Antioxidant, Anti-Glycation and Anti-Inflammatory Activities of Phenolic Constituents from Cordia sinensis. Molecules. 2011; 16(12):10214-10226. https://doi.org/10.3390/molecules161210214
Chicago/Turabian StyleAl-Musayeib, Nawal, Shagufta Perveen, Itrat Fatima, Muhammad Nasir, and Ajaz Hussain. 2011. "Antioxidant, Anti-Glycation and Anti-Inflammatory Activities of Phenolic Constituents from Cordia sinensis" Molecules 16, no. 12: 10214-10226. https://doi.org/10.3390/molecules161210214
APA StyleAl-Musayeib, N., Perveen, S., Fatima, I., Nasir, M., & Hussain, A. (2011). Antioxidant, Anti-Glycation and Anti-Inflammatory Activities of Phenolic Constituents from Cordia sinensis. Molecules, 16(12), 10214-10226. https://doi.org/10.3390/molecules161210214




