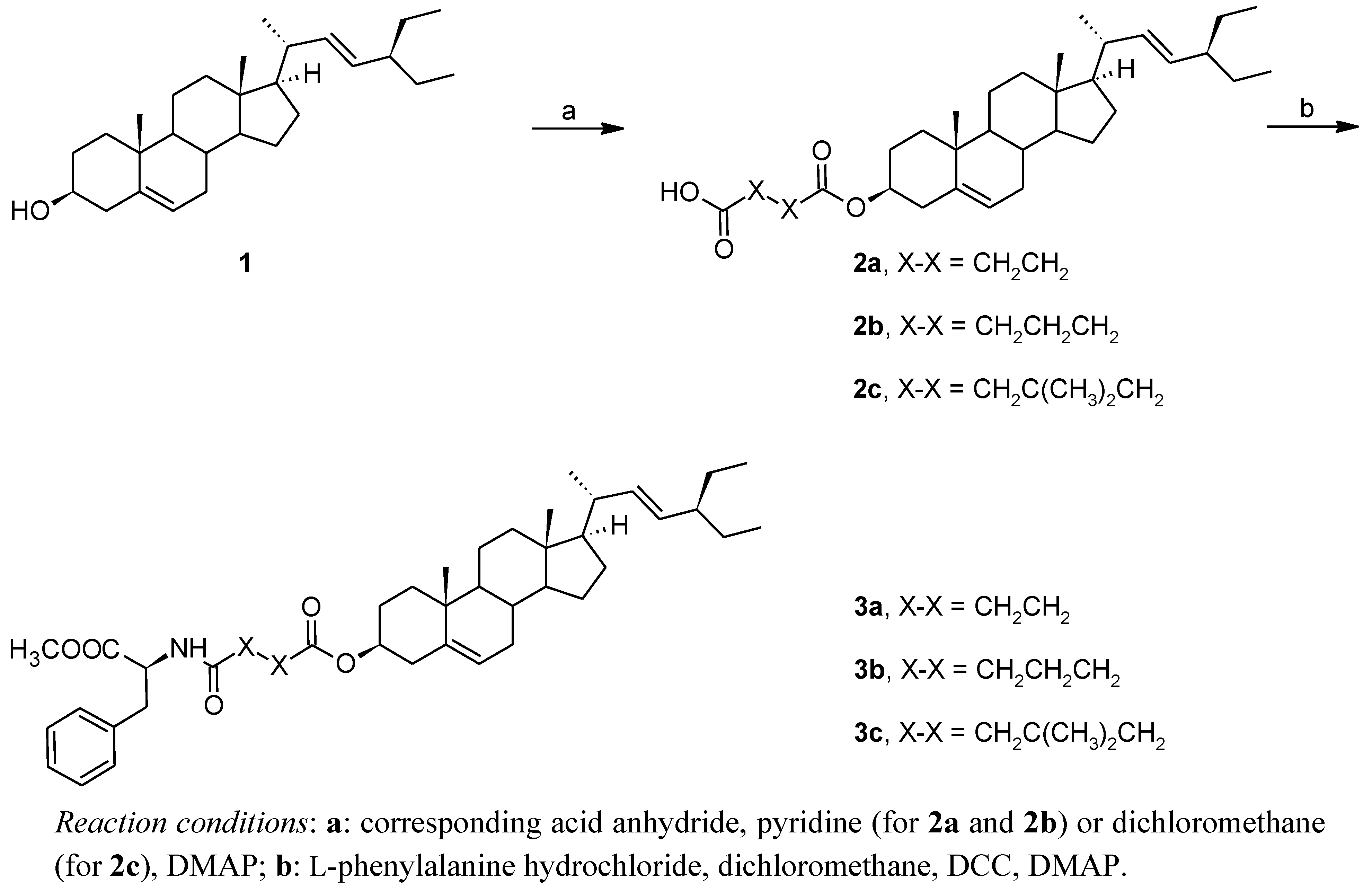
3. Experimental
3.1. General
The 1H-NMR and the 13C-NMR spectra were recorded on a Bruker AVANCE 600 MHz spectrometer at 600.13 MHz and 150.90 MHz in deuteriochloroform using tetramethylsilane (δ = 0.0) as internal reference. 1H-NMR data are presented in the following order: chemical shift (δ) expressed in ppm, multiplicity (s, singlet; d, doublet; t, triplet; q, quartet; m, multiplet), coupling constants in Hertz, number of protons. Infrared spectra were measured with a Nicolet 205 FT-IR spectrometer. Mass spectra were measured with a Waters ZMD mass spectrometer in a positive ESI mode. TLC was carried out on silica gel plates (Merck 60F254) and the visualization was performed both, by UV detection and by spraying with the methanolic solution of phosphomolybdic acid (5%) followed by heating. Elemental analyses were performed on a Perkin Elmer 2400, series II CHNS/O analyzer (USA). Melting points were determined on a Kofler MHK melting point apparatus (Franz Küstner Nacht, KG, Dresden, Germany) and are uncorrected. All chemicals and solvents were purchased from regular commercial sources in analytical grade and the solvents were purified by general methods before use. For column chromatography, silica gel 60 (0.063–0.200 mm) from Merck was used. ACD/Labs software, ACD/logD DB, version 12.01, was used for calculation of solubility, partition coefficient (log P) and distribution coefficient (log D) of the prepared compounds.
3.2. 4-Oxo-4-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]butanoic Acid (2a), 5-Oxo-5-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]pentanoic Acid (2b) and 5-Oxo-5-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]-3,3-dimethylpentanoic Acid (2c)
A catalytic amount of 4-dimethylaminopyridine (DMAP; 0.2 g, 1.637 mmol) was added to a solution of stigmasterol (
1, 4.0 g, 9.693 mmol) and the anhydride of the corresponding dicarboxylic acid (15.188 mmol) in either dry pyridine (17.5 mL; in the preparation of
2a and
2b) or dry dichloromethane (10 mL; in the preparation of
2c); path
a in
Scheme 1. The mixture was stirred at room temperature for 7 days and then poured onto a mixture of ice (30 mL)/hydrochloric acid (15 mL). The organic layer was extracted with chloroform (5 × 40 mL), the combined extracts were dried over sodium sulfate, and the solvent was finally evaporated to dryness. The residue was purified by column chromatography using a chloroform/methanol gradient (100:0 to 60:1) mixture as mobile phase. Yields:
2a: 4.62 g (93%),
2b: 3.41 g (67%),
2c: 3.07 g (57%).
4-Oxo-4-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]butanoic acid (2a): 1H-NMR: 5.37 (ddt, 1H, J = 1.3; 1.3; 1.8; 5.0 Hz, H-6´), 5.15 (dd, 1H, J = 8.7; 15.2 Hz, H-22´), 5.01 (dd, 1H, J = 8.9; 15.2Hz, H-23´), 4.63 (dddd, 1H, J = 4.2; 7.1; 9.5; 11.6 Hz, H-3´), 2.61 (m, 2H, H-31´), 2.68 (m, 2H, H-32´), 2.30–2.33 (m, 2H, H-4´), 2.00–2.06 (m, 1H, H-20´), 1.55–1.60 (m) + 1.80–1.86 (m, 2H, H-2´), 1.52–1.56 (m, 1H, H-24´), 1.51–1.55 (m, 1H, H-25´), 1.46–1.51 (m, 2H, H-11´), 1.46–1.51 (m, 1H, H-8´), 1.37–1.41 (m) + 1.93–2.01 (m, 2H, H-7´), 1.14–1.19 (m) + 1.41–1.46 (m, 2H, H-28´), 1.21–1.28 (m) + 1.70 (ddd, 2H, J = 6.1; 9.8; 13.0 Hz, H-16´), 1.12–1.17 (m, 1H, H-17´), 1.09–1.12 (m) + 1.83–1.88 (m, 2H, H-1´), 1.07 (ddd, 1H, J = 6.2; 11.3; 12.6 Hz, H-14´), 1.06–1.11 (m) + 1.96-2.00 (m, 2H, H-12´), 1.021 (d, 3H, J = 6.6 Hz, H-21´), 1.020 (s, 3H, H-19´), 1.01–1.06 (m) + 1.50–1.56 (m, 2H, H-15´), 0.95 (ddd, J = 5.1; 10.9; 12.1 Hz, H-9´), 0.846 (d, 3H, J = 6.5 Hz, H-26´), 0.805 (t, 3H, J = 7.4 Hz, H-29´), 0.795 (d, 3H, J = 6.6 Hz, H-27´), 0.695 (s, 3H, H-18´). 13C-NMR: 177.54 (s, C-33´), 171.53 (s, C-30´), 139.49 (s, C 5´), 138.31 (d, C-22´), 129.24 (d, C-23´), 122.72 (d, C-6´), 74.53 (d, C-3´), 56.75 (d, C 14´), 55.88 (d, C-17´), 51.21 (d, C-24´), 49.98 (d, C-9´), 42.17 (s, C-13´), 40.51 (d, C-20´), 39.59 (t, C 12´), 37.98 (t, C-4´), 36.56 (s, C-10´), 36.92 (t, C-1´), 31.87 (d, C-25´), 31.87 (t, C-8´), 31.81 (t, C-7´), 29.20 (t, C-31´), 28.91 (t, C-32´), 28.89 (t, C-16´), 27.66 (t, C-2´), 25.40 (t, C-28´), 24.34 (t, C-15´), 21.22 (q, C-21´), 21.09 (q, C-26´), 20.99 (t, C-11´), 19.30 (q, C-19´), 18.97 (q, C-27´), 12.25 (q, C-29´), 12.03 (q, C-18´). IR (KBr): [cm−1] 1732 vs and 1715 s (C=O), 1381 w (CH3), 1178 s (C-O). For C33H52O4 (512.75) calcd.: 77.29% C, 10.22% H; found: 77.15% C, 10.41% H. MS (ESI, 20 eV): [M+Na]+ 535.
5-Oxo-5-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]pentanoic acid (2b): 1H-NMR: 5.37 (ddt, 1H, J = 1.2; 1.2; 2.0, 5.8 Hz, H-6´), 5.15 (dd, 1H, J = 8.7; 15.2 Hz, H-22´), 5.02 (ddd, 1H, J = 0.6; 9.0; 15.2 Hz, H-23´), 4.62 (dddd, 1H, J = 4.3; 7.5; 9.4; 11.3 Hz, H-3´), 2.44 (t, 2H, J = 7.3 Hz, H-31´), 2.38 (t, 2H, J = 7.3 Hz, H-33´), 2.39–2.33 (m, 2H, H-4´), 2.00–2.07 (m, 1H, H-20´), 1.96 (m, 2H, H-32´), 1.55–1.62 (m) + 1.83–1.86 (m, 2H, H-2´), 1.53–1.57 (m, 1H, H-24´), 1.51–1.54 (m, 1H, H-25´), 1.48–1.53 (m, 2H, H-11´), 1.48–1.53 (m, 1H, H-8´), 1.39–1.44 (m) + 1.94–2.00 (m, 2H, H-7´), 1.14–1.19 (m) + 1.41–1.45 (m, 2H, H-28´), 1.22–1.28 (m) + 1.70 (ddd, 2H, J = 6.1; 9.8; 13.0 Hz, H-16´), 1.12–1.16 (m, 1H, H-17´), 1.09–1.12 (m) + 1.83–1.88 (m, 2H, H-1´), 1.07 (ddd, 1H, J = 6.1; 11.3; 12.6 Hz, H-14´), 1.08–1.13 (m) + 1.97–2.01 (m, 2H, H-12´), 1.022 (d, 3H, J = 6.6 Hz, H-21´), 1.020 (s, 3H, H-19´), 1.01–1.05 (m) + 1.51–1.57 (m, 2H, H-15´), 0.96 (ddd, J = 5.0; 10.9; 12.2 Hz, H-9´), 0.846 (d, 3H, J = 6.5 Hz, H-26´), 0.799 (t, 3H, J = 7,4 Hz, H-29´), 0.796 (d, 3H, J = 6.7 Hz, H-27´), 0.696 (s, 3H, H-18´). 13C-NMR: 178.06 (s, C-34´), 172.29 (s, C-29´), 139.55 (s, C 5´), 138.31 (d, C-22´), 129.23 (d, C-23´), 122.68 (d, C-6´), 74.09 (d, C-3´), 56.75 (d, C 14´), 55.88 (d, C-17´), 51.21 (d, C-24´), 49.99 (d, C-9´), 42.17 (s, C-13´), 40.51 (d, C-20´), 39.59 (t, C 12´), 38.09 (t, C-4´), 36.94 (t, C-1´), 36.57 (s, C-10´), 33.50 (t, C-31´), 32.83 (t, C 33´), 31.87 (d, C-25´), 31.87 (t, C-8´), 31.82 (t, C-7´), 28.91 (t, C-16´), 27.76 (t, C-2´), 25.41 (t, C-28´), 24.34 (t, C-15´), 21.21 (q, C-21´), 21.09 (q, C-26´), 20.99 (t, C-11´), 19.88 (t, C-32´), 19.30 (q, C-19´), 18.96 (q, C-27´), 12.25 (q, C-29´), 12.03 (q, C-18´). IR (KBr): [cm−1] 1739 vs and 1712 s (C=O), 1380 w (CH3), 1180 s (C-O). For C34H54O4 (526.77) calcd.: 77.52% C, 10.33% H, found: 77.34% C, 10.31% H. MS (ESI, 20 eV): [M+Na]+ 549.
5-Oxo-5-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]-3,3-dimethylpentanoic acid (2c): 1H-NMR: 5.38 (ddt, 1H, J = 1.3; 1.3; 2.0; 5.0 Hz, H-6´), 5.15 (dd, 1H, J = 8.6; 15.1 Hz, H-22´), 5.02 (dd, 1H, J = 8.9; 15.1 Hz, H-23´), 4.65 (dddd, 1H, J = 4.1; 7.7; 9.0; 11.3 Hz, H-3´), 2.46 (s, 2H, H-33´), 2.41 (s, 2H, H-31´), 2.31–2.33 (m, 2H, H-4´), 2.02–2.08 (m, 1H, H-20´), 1.57–1.62 (m) + 1.84–1.89 (m, 2H, H-2´), 1.52–1.56 (m, 1H, H-25´), 1.52–1.56 (m, 1H, H-24´), 1.49–1.55 (m, 1H, H-8´), 1.47–1.53 (m, 2H, H-11´), 1.43–1.49 (m) + 1.95–2.00 (m, 2H, H-7´), 1.23–1.28 (m) + 1.68–1.73 (m, 2H, H-16´), 1.17–1.21 (m) + 1.97–2.02 (m, 2H, H-12´), 1.14–1.18 (m) + 1.41–1.44 (m, 2H, H-28´), 1.14 (s, 3H, H-35´), 1.13–1.18 (m, 1H, H-17´), 1.12–1.16 (m) + 1.84–1.89 (m, 2H, H-1´), 1.03–1.07 (m) + 1.53–1.57 (m, 2H, H-15´), 1.02 (d, 3H, J = 6.6 Hz, H-21´), 1.02 (s, 1H, H-19´), 0.99–1.05 (m, 1H, H-14´), 0.94–0.99 (m, 1H, H-9´), 0.85 (d, 3H, J = 6.5 Hz, H-26´), 0.81 (t, 3H, J = 7.3 Hz, H-29´), 0.80 (d, 3H, J = 6.5 Hz, H-27´), 0.70 (s, 3H, H-18´). 13C-NMR: 175.57 (s, C-34´), 172.17 (s, C-30´), 139.44 (s, C 5´), 138.30 (d, C-22´), 129.28 (d, C-23´), 122.80 (d, C-6´), 74.38 (d, C-3´), 56.76 (d, C 14´), 55.92 (d, C-17´), 51.22 (d, C-24´), 50.00 (d, C-9´), 45.42 (t, C-31´), 45.21 (t, C-33´), 42.19 (s, C-13´), 40.50 (d, C-20´), 39.30 (t, C 12´), 38.11 (t, C-4´), 36.95 (t, C-1´), 36.60 (s, C-10´), 33.50 (t, C-31´), 32.83 (t, C-33´), 31.87 (d, C-25´), 31.87 (t, C-8´), 31.83 (t, C-7´), 28.90 (t, C-16´), 27.98 (q, C-35´), 27.81 (t, C-2´), 25.40 (t, C-28´), 21.22 (q, C-21´), 21.08 (q, C-26´), 21.00 (t, C-11´), 19.31 (q, C-19´), 18.97 (q, C-27´), 12.24 (q, C-29´), 12.04 (q, C-18´). IR (KBr): [cm−1] 1733 vs and 1714 s (C=O), 1390 w (CH3), 1178 s (C-O). For C36H58O4 (554.82) calcd.: 77.93% C, 10.54% H; found: 77.77% C, 10.51% H. MS (ESI, 20 eV): [M+Na]+ 577.
3.3. Methyl N-{4-oxo-4-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]butanoyl}-L-phenylalanine (3a), Methyl N-{5-oxo-5-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]pentanoyl}-L-phenylalanine (3b) and Methyl N-{5-oxo-5-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]-3,3-dimethylpentanoyl}-L-phenylalanine (3c)
A solution of stigmasteryl hemiester (
2a–
2c, 0.195 mmol), L-phenylalanine hydrochloride (0.195 mmol), DCC (0.234 mmol) and DMAP (0.058 mmol) in dry dichlomethane (10 mL), containing several drops of dry pyridine (to neutralize the hydrogen chloride formed), was stirred at room temperature for 2 days; path
b in
Scheme 1. The solvent was then evaporated, and the residue was purified by column chromatography on silica gel, using a chloroform/methanol gradient (100:0 to 80:1) mixture as mobile phase. Yields:
3a: 97%,
3b: 96%,
3c: 89%.
Methyl N-{4-oxo-4-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]butanoyl}-L-phenylalanine (3a): 1H-NMR: 7.27–7.31 (m, 1H, H-38´), 7.23–7.26 (m, 1H, H-39´), 7.09–7.12 (m, 1H, H-37´), 5.35 (m, 1H, H-6´), 5.15 (dd, 1H, J = 8.8; 15.2 Hz, H-22´), 5.02 (dd, 1H, J = 8.8; 15.2 Hz, H-23´), 4.88 (dt, 1H, J = 5.8; 5.8; 7.7 Hz, H-34´), 4.58–4.64 (m, 1H, H-3´), 3.72 (s, 3H, H-41´), 3.09 (dd, 2H, J = 5.6; 13.9 Hz, H-35´)+3.14 (dd, 2H, J = 5.8; 13.9 Hz, H 35´), 2.59 (ddd, 2H, J = 6.0; 7.0; 17.1 Hz, H-31´)+2.63 (dt, 1H, J = 7.2; 7.2; 17.1 Hz, H-31´), 2.45–2.51 (m, 2H, H-4´), 2.28–2.32 (m, 2H, H-32´), 1.02 (d, 3H, J = 6.7 Hz, H-21´), 1.01 (s, 3H, H-19´), 0.85 (d, 3H, J = 6.5 Hz, H-26´), 0.80 (t, 3H, J = 7.4 Hz, H-29´), 0.79 (d, 3H, J = 6.5 Hz, H-27´), 0.69 (s, 3H, H-18´). 13C-NMR: 172.14 (s, C-33´), 171.92 (s, C-40´), 170.99 (s, C-30´), 139.58 (s, C-5´), 138.31 (d, C-22´), 135.78 (s, C-36´), 129.27 (d, C-37´), 129.23 (d, C-23´), 128.56 (t, C-38´), 127.11 (d, C-39´), 122.66 (d, C-6´), 74.37 (d, C-3´), 56.75 (d, C-14´), 55.88 (d, C-17´), 53.13 (d, C-34´), 52.32 (q, C-41´), 51.21 (d, C-24´), 50.00 (d, C-9´), 42.17 (s, C-13´), 40.50 (d, C-20´), 39.59 (t, C 12´), 38.02 (t, C-4´), 37.86 (t, C-35´), 36.94 (t, C-1´), 36.57 (s, C-10´), 31.87 (d, C-25´), 31.87 (t, C-8´), 31.81 (t, C-7´), 30.89 (t, C-32´), 29.66 (t, C-31´), 28.91 (t, C-16´), 27.68 (t, C-2´), 25.40 (t, C-28´), 24.33 (t, C-15´), 21.20 (q, C-21´), 21.09 (q, C-26´), 20.98 (t, C-11´), 19.30 (q, C-19´), 18.96 (q, C-27´), 12.25 (q, C-29´), 12.02 (q, C-18´). IR (KBr): [cm−1] 3282m (-CO-NH-), 2956 m and 2947 m (-CH2), 2890 m and 2869 m (-CH3), 1743 vs and 1644 s (C=O), 1216 s (Ar), 1177 s (C-O). For C43H63NO5 (673.97) calcd.: 76.74% C, 9.36% H, found: 76.64% C, 9.34% H. MS (ESI, 20 eV): [M+Na]+ 696. Calculated partition coefficient: neutral form logP = 11.22 ± 0.51, form charged on nitrogen logP = 9.22 ± 1.00. Calculated diffusion coefficient for neutral pH: logD = 11.22. Solubility in pure water at pH = 7: 2.9606 × 10−7 mg·mL−1.
Methyl N-{5-oxo-5-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]pentanoyl}-L-phenylalanine (3b): 1H-NMR: 7.27–7.31 (m, 1H, H-39´), 7.23–7.26 (m, 1H, H-40´), 7.08–7.11 (m, 1H, H-38´), 5.36–5.38(m, 1H, H-6´), 5.15 (dd, 1H, J = 8.7; 15.1 Hz, H-22´), 5.02 (dd, 1H, J = 8.7; 15.1 Hz, H-23´), 4.90 (dt, 1H, J = 5.9; 5.9; 7.9 Hz, H-35´), 4.58–4.64 (m, 1H, H-3´), 3.73 (m, 3H, H-42´), 3.16 (dd, 2H, J = 5.8; 13.9 Hz, H-36´) + 3.08 (dd, 2H, J = 6.1; 13.9 Hz, H-36´), 2.29–2.31 (m, 2H, H-4´), 2.26–2.34 (m, 2H, H-31´), 2.20–2.27 (m, 2H, H-33´), 1.91 (m, 2H, H-32´), 1.02 (d, 3H, J = 6.6 Hz, H-21´), 1.02 (s, 3H, H-19´), 0.85 (d, 3H, J = 6.6 Hz, H-26´), 0.81 (t, 3H, J = 7.3 Hz, H-29´), 0.79 (d, 3H, J = 6.7 Hz, H-27´), 0.70 (s, 3H, H-18´). 13C-NMR: 172.51 (s, C-34´), 172.06 (s, C-41´), 171.72 (s, C-30´), 139.50 (s, C-5´), 138.31 (d, C-22´), 135.80 (s, C-37´), 129.23 (d, C-23´), 129.21 (d, C-38´), 128.59 (d, C-39´), 127.14 (d, C-40´), 122.67 (d, C-6´), 74.00 (d, C-3´), 56.75 (d, C-14´), 55.88 (d, C-17´), 52.97 (d, C-35´), 52.35 (q, C-42´), 51.21 (d, C-24´), 50.00 (d, C-9´), 42.17 (s, C-13´), 40.51 (d, C-20´), 40.00 (d, C-9´), 39.59 (t, C-12´), 38.10 (t, C-4´), 37.89 (t, C-36´), 36.95 (t, C-1´), 36.58 (s, C-10´), 35.25 (t, C-33´), 33.51 (t, C-31´), 31.87 (d, C-25´), 31.87 (t, C-8´), 31.82 (t, C-7´), 28.91 (t, C-16´), 27.78 (t, C-2´), 25.40 (t, C-28´), 24.33 (t, C-15´), 21.21 (q, C 21´), 21.10 (q, C-26´), 20.99 (t, C-11´), 20.78 (t, C-32´), 19.31 (q, C-19´), 18.96 (q, C-27´), 12.25 (q, C-29´), 12.03 (q, C-18´). IR (KBr): [cm−1] 3072 m (-CO-NH-), 2960 m and 2934 m (-CH2), 2892 m and 2880 m (-CH3), 1738 vs and 1653 s (C=O), 1218 s (Ar), 1181 s (C-O). For C44H65NO5(687.99) calcd.: 76.93% C, 9.46% H; found: 76.89% C, 9.46% H. MS (ESI, 20 eV): [M+Na]+ 710. Calculated partition coefficient: neutral form logP = 11.49 ± 0.50; form charged on nitrogen logP = 9.49 ± 1.00. Calculated diffusion coefficient for neutral pH: logD = 11.49. Solubility in pure water at pH = 7: 2.0952 × 10−7 mg·mL−1.
Methyl N-{5-oxo-5-[(3β,22E)-stigmasta-5,22-dien-3-yloxy]-3,3-dimethylpentanoyl}-L-phenylalanine (3c): 1H-NMR: 7.27–7.30 (m, 1H, H-42´), 7.22–7.25 (m, 1H, H-43´), 7.15–7.18 (m, 1H, H-41´), 5.40–5.42 (m, 3H, H-6´), 5.16 (dd, 1H, J = 8.7; 15.2 Hz, H-22´), 5.02 (dd, 1H, J = 8.6; 15.2 Hz, H-23´), 4.87 (dt, 1H, J = 5.4; 8.1; 8.1 Hz, H-38´), 4.60–4.66 (m, 1H, H-3´), 3.72 (s, 3H, H-44´), 3.19 (dd, 1H, J = 5.4; 14.1 Hz, H-39´), 3.00 (dd, 1H, J = 8.1; 14.1 Hz, H-39´), 2.39 (d, 1H, J = 12.5, H-31´), 2.30 (d, 1H, J = 12.7, H-33´), 2.18 (d, 1H, J = 12.7 Hz, H-33´), 2.05 (d, 2H, J = 12.5 Hz, H-31´), 1.03 (d, 3H, J = 6.6 Hz, H-21´), 1.03 (s, 3H, H-36´), 1.03 (s, 3H, H-19´), 0.99 (s, 3H, H-35´), 0.85 (d, 3H, J = 6.7 Hz, H-26´), 0.81 (t, 3H, J = 7.4 Hz, H-29´), 0.81 (d, 3H, J = 6.6 Hz, H-27´), 0.71 (s, 3H, H-18´). 13C-NMR: 172.67 (s, C-37´), 172.49 (s, C-34´), 171.102(s, C-30´), 139.46 (s, C-5´), 138.29 (d, C-22´), 136.28 (s, C-40´), 129.26 (d, C-23´), 129.12 (d, C-42´), 128.52 (d, C-41´), 126.97 (d, C-43´), 122.80 (d, C-6´), 74.25 (d, C-3´), 56.76 (d, C-14´), 55.89 (d, C-17´), 53.18 (d, C-38´), 52.21 (q, C-44´), 51.21 (d, C-24´), 50.02 (d, C-9´), 46.83 (t, C-33´), 44.62 (t, C-31´), 42.18 (s, C-13´), 40.51 (d, C-20´), 39.59 (t, C 12´), 38.16 (t, C-4´), 37.80 (t, C-39´), 36.96 (t, C-1´), 36.60 (s, C-10´), 33.65 (s, C-32´), 31.88 (d, C-25´), 31.88 (t, C-7´), 31.81 (d, C-8´), 28.91 (t, C-16´), 28.72 (q, C-36´), 28.68 (q, C-35´), 27.78 (t, C-2´), 25.40 (t, C-28´), 24.34 (t, C-15´), 21.21 (q, C-21´), 21.10 (q, C-26´), 20.99 (t, C-11´), 19.30 (q, C-19´), 18.96 (q, C-27´), 12.25 (q, C-29´), 12.03 (q, C-18´). IR (KBr): [cm−1] 3318 m (-CO-NH-), 2956 m and 2937 m (-CH2), 2890 m and 2869 s (-CH3), 1731 vs and 1653 s (C=O), 1220 s (Ar), 1177 s (C-O). For C46H69NO5 (716.05) calcd.: 77.27% C, 9.65% H; found: 77.23% C, 9.64% H. MS (ESI, 20 eV): [M+Na]+ 738. Calculated partition coefficient: neutral form logP = 12.26 ± 0.51, form charged on nitrogen logP = 10.26 ± 1.00. Calculated diffusion coefficient for neutral pH: logD = 12.26. Solubility in pure water at pH = 7: 9.4142 × 10−8 mg·mL−1.