The Relationships between Phenolic Content, Pollen Diversity, Physicochemical Information and Radical Scavenging Activity in Honey
Abstract
:1. Introduction
2. Results and Discussion
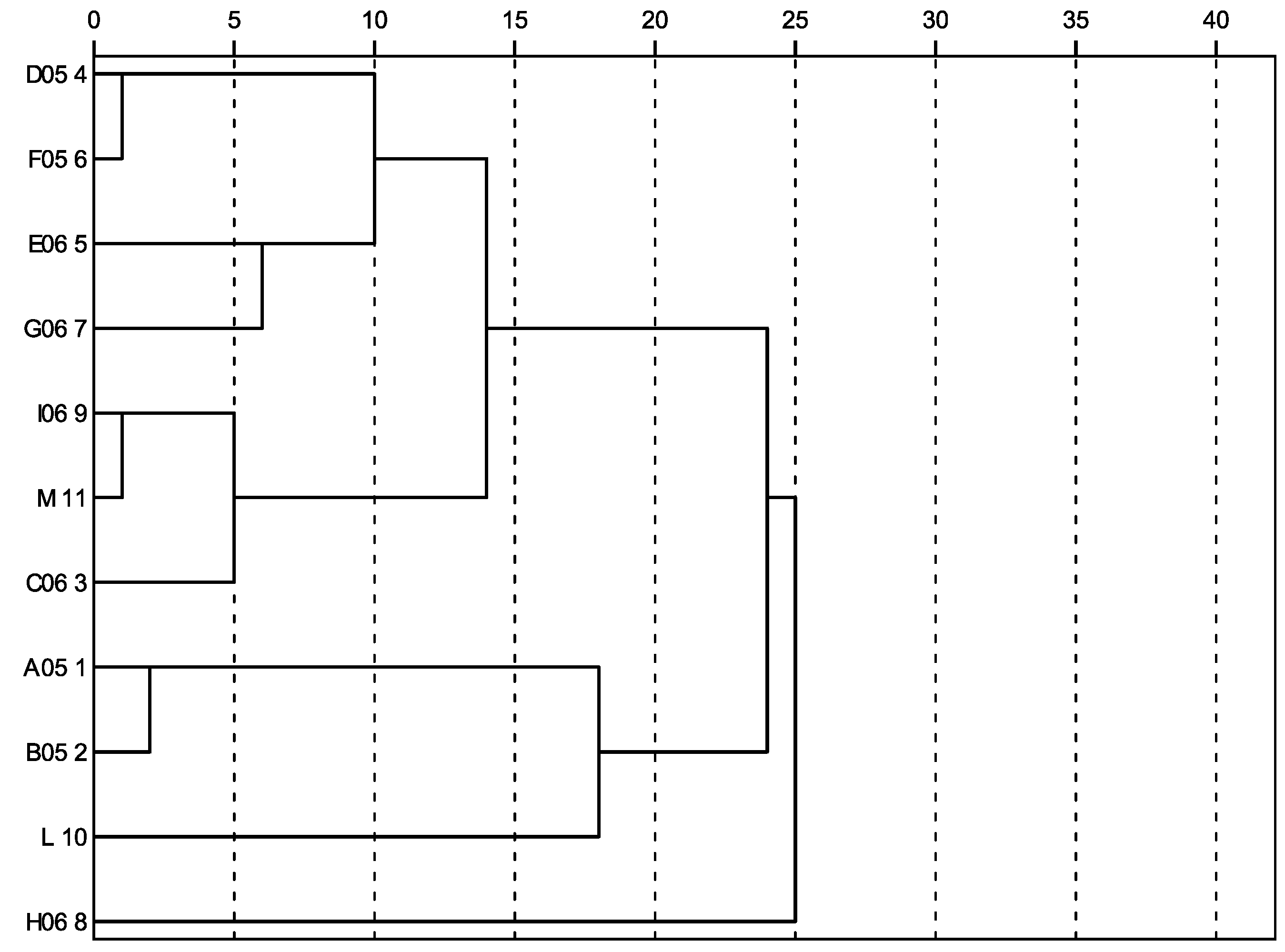
2.1. Variability assessment through melissopalynological analyses
| Sample code | Origin | Location | Data of collection | Pollen supplying plants (%) |
|---|---|---|---|---|
| A05 | Lombardia region | Monno (1066 m a.s.l.) | June 2005 | Castanea (78.2), Ericaceae (12.7), Rubus (4.4), Achillea (1.8), Pyrus (1.7), Trifolium (0.4) |
| B05 | Lombardia region | Monno (1066 m a.s.l.) | July 2005 | Castanea (75.2),Ericaceae (10.7), Rubus (3.9), Achillea (0.7), Tilia (0.4), Salix (0.3) |
| C06 | Lombardia region | Monno (1066 m a.s.l.) | August 2006 | Rhododendron (42), Rubus (30), Cruciferae (5.4), Trifolium (4.6), Salix (2.5),Robinia (1.8), Tilia (1.4), Umbelliferae (0.7) |
| D05 | Lombardia region | Saviore dell’Adamello (1210 m a.s.l.) | July2005 | Castanea (65.3), Rhododendron (16.5), Rubus (12.7), Trifolium (1.3), Salix (1.0), Myosotis (0.6), Knautia (0.4) |
| E06 | Lombardia region | Saviore dell’Adamello (1210 m a.s.l.) | August 2006 | Rubus (46.1), Rhododendron (38,4), Trifolium (3.1), Myosotis (2.9), Compositae (1.4), Achillea (1.9), Silene (1.0) |
| F05 | Lombardia region | Vezza d’Oglio(1080 m a.s.l.) | July 2005 | Rhododendron (80.0), Cruciferae (7.4), Rubus (2.9), Trifolium (1.3), Castanea (1.3), Umbelliferae (1.1), Astragalus (0.8) |
| G06 | Lombardia region | Vezza d’Oglio (1080 m a.s.l.) | May 2006 | Myosotis (70), Taraxacum (31.8), Prunus (21.7), Lotus (14.3), Salix (10.4), Acer (6.4), Rubus (3.3), Sorbus (3.3) |
| H06 | Lombardia region | Vezza d’Oglio (1080 m a.s.l.) | June 2006 | Trifolium (30.0), Robinia (18.2), Rhododendron (14.0), Rubus (13.6), Myosotis (7.9), Sorbus (5), Prunus (1.1) |
| I06 | Lombardia region | Vezza d’Oglio (1080m a.s.l.) | July 2006 | Trifolium (30.0), Myosotis (28.9), Umbelliferae (11.8), Rubus (5.1), Salix (4.2), Acer (3.2), Salvia (2.4), Lotus (2.4) |
| L | Veneto region | not available | 2006 | Castanea (92), Rubus (2,4), Trifolium (2,2), Eucalyptus (2.0) |
| M | Veneto region | not available | 2006 | Salix (28.5), Acer (27), Prunus (13.7), Sorbus (9.8), Robinia (4.6), Aesculus (4.3), Trifolium (3.1), Amorpha (3.0), Rubus (1.5) |
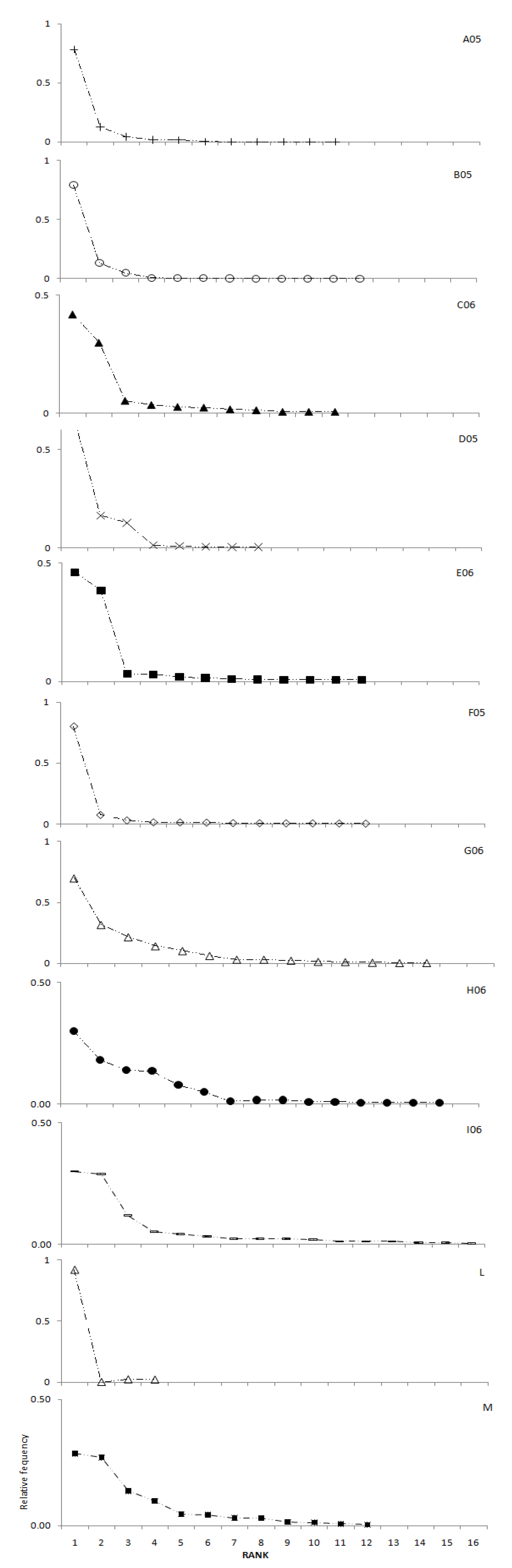
2.2. Variability assessment through physicochemical measurements, total phenolic content and radical scavenging activity
| Sample | pH | Free acidity (meq/kg) | Conductivity (10-4 S cm-1) | Moisture (%) | Total phenolic content (mg GAE/100g ± SD) | Radical Scavenging Activity (RSA) IC50 (mg/mL ± SD) |
|---|---|---|---|---|---|---|
| A05 | 4.66 | 26 | 6.77 | 16.5 | 62.80 ± 2.09 | 16.14 ± 0.73 |
| B05 | 4.60 | 26 | 7.38 | 18.5 | 57.79 ± 4.89 | 12.03 ± 0.35 |
| C06 | 4.04 | 29 | 4.70 | 17.3 | 25.31 ± 0.61 | 63.23 ± 1.24 |
| D05 | 4.05 | 14 | 2.30 | 17.3 | 43.12 ± 3.32 | 52.28 ± 0.31 |
| E06 | 4.51 | 23 | 6.10 | 16.3 | 34.13 ± 2.98 | 21.30 ± 1.39 |
| F05 | 3.85 | 9 | 2.59 | 16.8 | 36.85 ± 4.74 | 44.06 ± 0.08 |
| G06 | 4.41 | 18 | 5.40 | 18.1 | 32.22 ± 4.53 | 20.62 ± 1.87 |
| H06 | 3.85 | 13 | 1.50 | 16.4 | 15.13 ± 3.93 | 11.39 ± 2.11 |
| I06 | 3.88 | 23 | 3.30 | 16.9 | 29.06 ± 7.32 | 56.36 ± 3.90 |
| L | 5.21 | 21 | 16.80 | 17.0 | 82.49 ± 4.05 | 19.00 ± 1.84 |
| M | 4.34 | 23 | 7.84 | 16.7 | 24.21 ± 1.43 | 64.33 ± 2.76 |
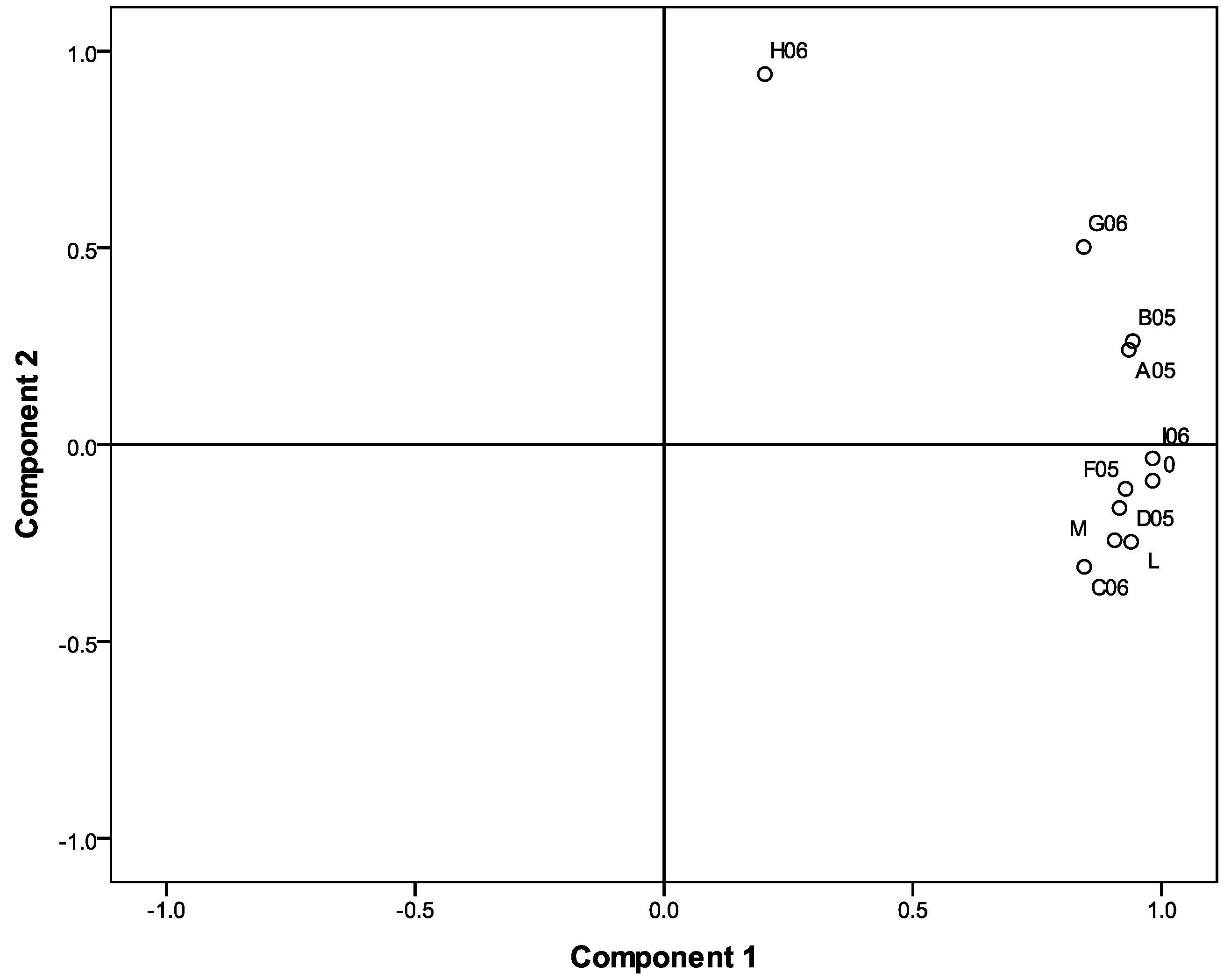
2.3. The relationship between radical scavenging activity (RSA) and total phenolic content
3. Experimental
3.1. Honey samples
3.2. Melissopalynological analysis and physicochemical variables
3.3. Assessment of total phenolic content
3.4. Radical scavenging activity (RSA)
3.5. Statistical analysis
4. Conclusions
References and Notes
- Downey, G.; Hussey, K.; Kelly, J. D.; Walshe, T. F.; Martin, P. G. Preliminary Contribution to the Characterisation of Artisanal Honey Produced on the Island of Ireland by Palynological and Physico-Chemical Data. Food Chem. 2005, 91, 347–354. [Google Scholar] [CrossRef]
- Ferreira, I. C. F. R.; Aires, E.; Barreira, J. C. M.; Estevinho, L. M. Antioxidant Activity of Portuguese Honey Samples: Different Contributions of the Entire Honey and Phenolic Extract. Food Chem. 2009, 114, 1438–1443. [Google Scholar] [CrossRef]
- Meda, A.; Lamien, C. E.; Romito, M.; Millogo, J.; Nacoulma, O. G. Determination of the Total Phenolic, Flavonoid and Proline Contents in Burkina Fasan Honey, as well as Their Radical Scavenging Activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Buratti, S.; Benedetti, S.; Cosio, M. S. Evaluation of The Antioxidant Power of Honey, Propolis and Royal Jelly by Amperometric Flow Injection Analysis. Talanta 2007, 71, 1387–1392. [Google Scholar] [CrossRef]
- Fiorani, M.; Accorsi, A.; Blasa, M.; Diamantini, G.; Piatti, E. Flavonoids from Italian Multifloral Honeys Reduce the Extracellular Ferricyanide in Human Red Blood Cells. J. Agr. Food Chem. 2006, 54, 8328–8334. [Google Scholar] [CrossRef]
- Vela, L.; De Lorenzo, C.; Pérez, R. A. Antioxidant Capacity of Spanish Honeys and Its Correlation with Polyphenol Content and other Physicochemical Properties. J. Sci. Food Agric. 2007, 87, 1069–1075. [Google Scholar] [CrossRef]
- Baltrušaityté, V.; Venskutonis, P.R.; Ceksteryte, V. Radical Scavenging Activity of Different Floral Origin Honey and Beebread Phenolic Extracts. Food Chem. 2007, 101, 502–514. [Google Scholar]
- Küçük, M.; Kolayli, S.; Karaoğlu, Ş.; Ulusoy, E.; Baltaci, C.; Candan, F. Biological Activities and Chimica Composition of Three Honeys of Different Types from Anatolia. Food Chem. 2007, 100, 526–534. [Google Scholar] [CrossRef]
- Gheldof, N.; Wang, X. H.; Engeseth, N. J. Identification and Quantification of Antioxidant components of honey from various floral sources. J. Agr. Food Chem. 2002, 50, 5870–5877. [Google Scholar]
- Goossens, A.; Haäkkinen, S. T.; Laakso, I.; Seppänen-Laakso, T.; Biondi, S.; De Sutter, V.; Lammertyn, F.; Nuutila, A. M.; Söderlund, H.; Zabeau, M.; Inze´, D.; Oksman-Caldentey, K. M. A Functional Genomics Approach Toward the Understanding of Secondary Metabolism in Plant Cells. Proc. Natl. Acad. Sci. USA. 2003, 100, 8595–8600. [Google Scholar]
- Ramachandra Rao, S.; Ravishankar, G.A. Plant Cell Cultures: Chemical Factories of Secondary Metabolites. Biotechnol. Adv. 2002, 20, 101–153. [Google Scholar] [CrossRef]
- Bertoncelj, J.; Doberšeka, U.; Jamnika, M.; Goloba, T. Evaluation of the Phenolic Content, Antioxidant activity and Colour of Slovenian Honey. Food Chem. 2007, 105, 822–828. [Google Scholar] [CrossRef]
- Beretta, G.; Granata, P.; Ferrero, M.; Orioli, M.; Maffei Facino, R. Standardization of Antioxidant Properties of Honey by a Combination of Spectrophotometric/Fluorimetric Assays and Chemometrics. Anal. Chim. Acta 2005, 533, 185–191. [Google Scholar] [CrossRef]
- Corbella, E.; Cozzolino, D. Classification of the Floral Origin of Uruguayan Honeys by Chemical and Physical Characteristics Combined with Chemometrics. LWT-Food Sci. Technol. 2006, 39, 534–539. [Google Scholar] [CrossRef]
- European Commission (2002) Council Directive 2001/110/CE concerning honey. Offic. J. Eur. Commu. 2002, L10: 47–L10:52.
- Chudzinska, M.; Baralkiewicz, D. Estimation of Honey Authenticity by Multielements Characteristics Using Inductively Coupled Plasma-Mass Spectrometry (ICP-MS) Combined with Chemometrics. Food Chem. Toxicol. 2010, 48, 284–290. [Google Scholar] [CrossRef]
- Louveaux, J.; Maurizio, A.; Vorwhol, G. Methods of melissopalynology. Bee World 1978, 59, 139–157. [Google Scholar]
- Borgdanov, S.; Martin, P.; Lüllmann, C. Harmonised Methods of the European Honey Commission. Apidologie 1997, extra issue, 1–59. [Google Scholar]
- Peréz, R. A.; Iglesias, M. T.; Pueyo, E.; Gonzáles, M.; De Lorenzo, C. Amino Acid Composition and Antioxidant Capacity of Spanish Honeys. J. Agric. Food Chem. 2007, 55, 360–365. [Google Scholar] [CrossRef]
- Singleton, V. L.; Rossi, J. A. Colorimetry of Total Phenolics with Phosphomolybdic-phosphotungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Myers, J. A.; Harms, K. E. Local Immigration, Competition from Dominant Guilds, and the Ecological Assembly of High-Diversity Pine Savannas. Ecol 2009, 90, 2745–2754. [Google Scholar] [CrossRef]
- Smith, R. L.; Smith, T. M. Ecology and Field Biology: Hands-On Field Package, 6rd ed; Benjamin Cummings: San Francisco, CA, USA, 2008. [Google Scholar]
- Gauch, H. G., Jr. Multivariate Analysis in Community Ecology; Cambridge University Press: Cambridge, UK, 1982. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Giorgi, A.; Madeo, M.; Baumgartner, J.; Lozzia, G.C. The Relationships between Phenolic Content, Pollen Diversity, Physicochemical Information and Radical Scavenging Activity in Honey. Molecules 2011, 16, 336-347. https://doi.org/10.3390/molecules16010336
Giorgi A, Madeo M, Baumgartner J, Lozzia GC. The Relationships between Phenolic Content, Pollen Diversity, Physicochemical Information and Radical Scavenging Activity in Honey. Molecules. 2011; 16(1):336-347. https://doi.org/10.3390/molecules16010336
Chicago/Turabian StyleGiorgi, Annamaria, Moira Madeo, Johann Baumgartner, and Giuseppe Carlo Lozzia. 2011. "The Relationships between Phenolic Content, Pollen Diversity, Physicochemical Information and Radical Scavenging Activity in Honey" Molecules 16, no. 1: 336-347. https://doi.org/10.3390/molecules16010336
APA StyleGiorgi, A., Madeo, M., Baumgartner, J., & Lozzia, G. C. (2011). The Relationships between Phenolic Content, Pollen Diversity, Physicochemical Information and Radical Scavenging Activity in Honey. Molecules, 16(1), 336-347. https://doi.org/10.3390/molecules16010336





