Novel Oxidative Ring Opening Reaction of 1H-Isotelluro-chromenes to Bis(o-formylstyryl) Ditellurides
Abstract
:1. Introduction
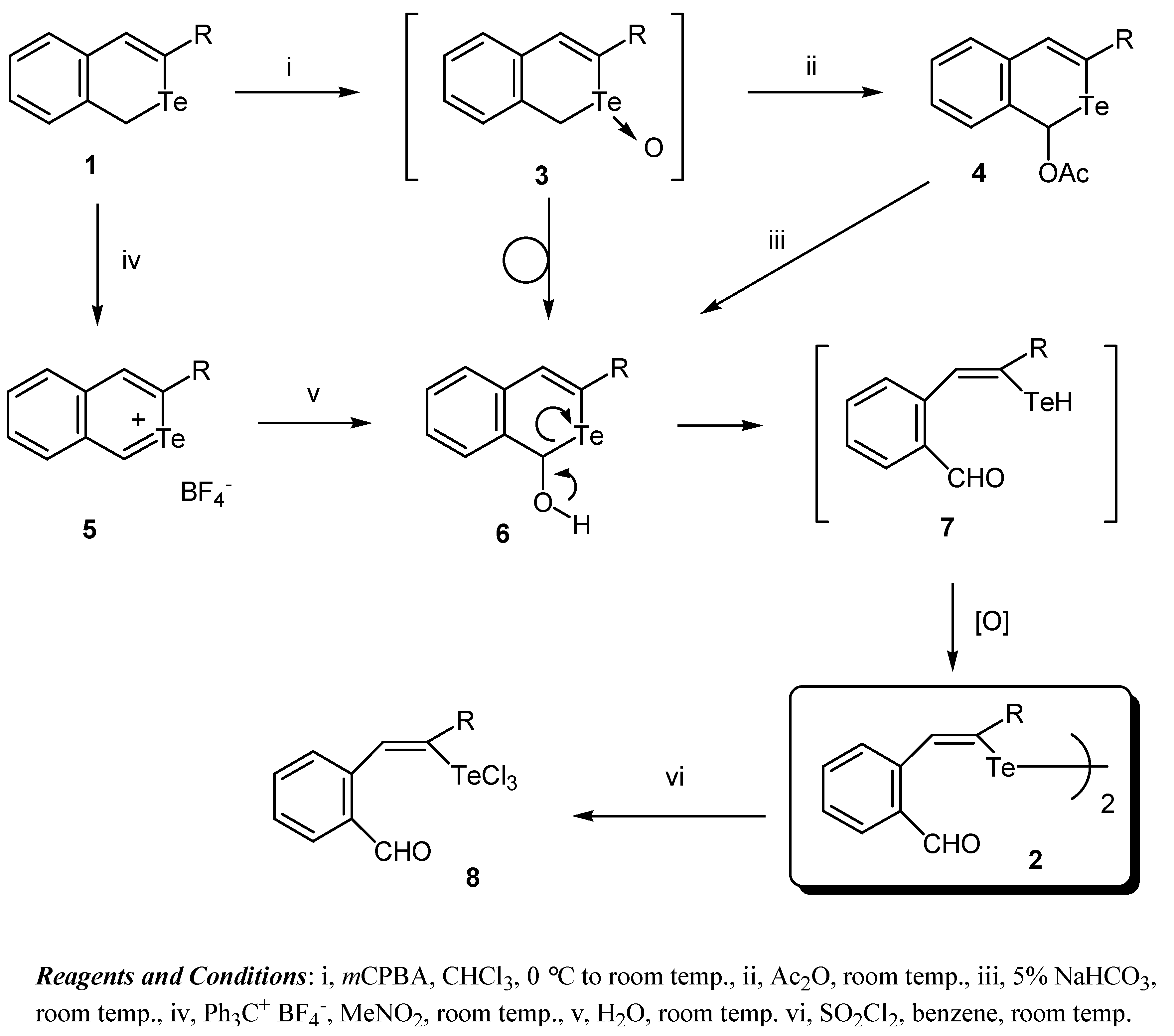
2. Results and Discussion
| Entry | Product | R1 | R2 | Yield (%) | Appearance (mp, °C) |
|---|---|---|---|---|---|
| 1 | 2a | t-Bu | H | 83 | orange prisms, 140–143 |
| 2 | 2b | H | H | 43 | orange prisms, 95–97 |
| 3 | 2c | Me | H | 57 | orange oil |
| 4 | 2d | n-Pro | H | 60 | orange oil |
| 5 | 2e | n-Bu | H | 70 | orange oil |
| 6 | 2f | 1-cyclohexenyl | H | 83 | orange oil |
| 7 | 2g | n-Oct | H | 62 | orange oil |
| 8 | 2h | Ph | H | 67 | orange oil |
| 9 | 2i | t-Bu | Ph | 80 | orange oil |
| 10 | 2j | t-Bu | Me | 0 | --- |
3. Experimental
Acknowledgements
- Sample Availability: Samples of the compounds 1,2 are available from the authors.
References and Notes
- Kuthan, J.; Sebek, P.; Bohm, S. Advances in Heterocyclic Chemistry: Developments in the Chemistry of Thiopyrans, Selenopyrans, and Telluropyrans, 1st ed.; Katritzky, A.R., Ed.; Academic Press: London, UK, 1994; Volume 59, pp. 179–244. [Google Scholar]
- Sadekov, I.D.; Minkin, V.I. Advances in Heterocyclic Chemistry: Six-Membered Heterocycles with a Tellurium Atom, 1st ed.; Katritzky, A.R., Ed.; Academic Press: London, UK, 1995; Volume 63, pp. 1–60. [Google Scholar]
- Detty, M.R.; O’Regan, M.B. The Chemistry of Heterocyclic Compounds: Tellurium-Containing Heterocycles; Taylor, E.C., Ed.; John Wiley & Sons: New York, USA, 1994; Volume 53, pp. 1–490. [Google Scholar]
- Irgolic, K.Y. The Organic Chemistry of Tellurium; Gordon and Breach: New York, USA, 1974. [Google Scholar]
- Engman, L. Synthetic applications of organotellurium chemistry. Acc. Chem. Res. 1985, 18, 274–279. [Google Scholar] [CrossRef]
- Patai, S.; Rappoport, Z. The Chemistry of Organic Selenium and Tellurium Compounds; John Wiley & Sons: New York, USA, 1986; Volume I, pp. 1–939. [Google Scholar]
- Patai, S.; Rappoport, Z. The Chemistry of Organic Selenium and Tellurium Compounds; John Wiley & Sons: New York, USA, 1987; Volume II. [Google Scholar]
- Petragnani, N. Tellurium in Organic Synthesis; Academic Press: London, UK, 1994. [Google Scholar]
- Sashida, H. Synthesis of tellurium-containing heterocycles by intramolecular hydrotelluration of tellurols to a triple bond. Rev. Heteroatom. Chem. 2000, 22, 59–78. [Google Scholar]
- Sashida, H. Preparations of heterocycles by intramolecular cyclization of tellurols and related compounds to a triple bond. J. Syn. Org. Chem. Jpn. 2001, 59, 355–362. [Google Scholar] [CrossRef]
- Sashida, H.; Ohyanagi, K. Studies on tellurium-containing heterocycles. 2-Benzotelluropyrylium salts: First preparation and reactions with Grignard reagents. J. Chem. Soc. Perkin Trans. 1 1998, 2123–2124. [Google Scholar]
- Sashida, H.; Ohyanagi, K.; Minoura, M.; Akiba, K.Y. Studies on tellurium-containing heterocycles. Synthesis and structure of 2-benzotelluropyrylium salts and 2-benzoselenopyrylium salts. J. Chem. Soc. Perkin Trans. I 2002, 606–612. [Google Scholar]
- Sashida, H. 1,5-Dilithiated synthetic building block, (E)-o-(2'-lithiovinyl)benzyllithium by tellurium-lithium exchange. Synthesis 1999, 1866–1868. [Google Scholar] [CrossRef]
- Drevko, B.I.; Fomenko, L.A.; Kharchenko, V.G. Oxidation of 2,4,6-triaryl-4H-thio(selenol)pyrans with selenium dioxide. Khim. Geterotsikl. Soedin. 1989, 767–769. [Google Scholar]
- Kataoka, T.; Ohe, Y.; Umeda, A.; Iwamura, T.; Yoshimatsu, M.; Shimizu, H. Novel oxidative ring contraction of dihydroselenopyrans to selenophenes. J. Chem. Soc. Chem. Commun. 1993, 577–579. [Google Scholar]
- Ruwet, A.; Messen, J.; Renson, M. Selective oxidation of 1-thio- and 1-selenochromenes. Formation of 1-thio- and selenocoumarins, 2-formylbenzo[b]thiophenes, 2-formylbenzo[b] selenophenes, and other isomers. Bul. Soc. Chim. Belg. 1969, 78, 459–464. [Google Scholar]
- Dereu, N.; Renson, M. Oxydation de telluro-1 chromenes. J. Organomet. Chem. 1983, 258, 163–169. [Google Scholar] [CrossRef]
- Sadekov, I.D.; Nivorozhkin, V.L.; Ladatko, A.A.; Minkin, V.I. Reactions of 2H-tellurochromene. Khim. Geterotsikl. Soedin. 1988, 1050–1054. [Google Scholar]
- Hori, M.; Kataoka, T.; Shimizu, H.; Hongo, J.; Kido, M. Reaction of 1H-2-benzothiopyran 2-oxides with active methylene compounds: A novel ring contraction of 1-aryl derivatives to benzo[c]thiophenes. X-ray molecular structure of 1-(2,2-diacetylvinyl)-3-phenylbenzo[c]thio-phenes. J. Chem. Soc, Perkin Trans. 1 1989, 1611–1618. [Google Scholar]
- Okuma, K. Kojima, K.; Y. Koga, Y.; Shioji, K. Oxidation of 1H-benzoselenopyrans. Generation of benzoselenophenes, benzaldehydes, and benzophenones. Heterocycles 2000, 52, 1021–1024. [Google Scholar]
- Sashida, H.; Satoh, H.; Ohyanagi, K. Reaction of 2-benzotelluropyrylium salts with organocopper reagents: Introduction of a carbon functional group at the C-1 position of the tellurium cation ring. Heterocycles 2004, 63, 309–317. [Google Scholar] [CrossRef]
Appendix
Appendix 1
Appendix 2
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sashida, H.; Satoh, H.; Ohyanagi, K.; Kaname, M. Novel Oxidative Ring Opening Reaction of 1H-Isotelluro-chromenes to Bis(o-formylstyryl) Ditellurides. Molecules 2010, 15, 1466-1472. https://doi.org/10.3390/molecules15031466
Sashida H, Satoh H, Ohyanagi K, Kaname M. Novel Oxidative Ring Opening Reaction of 1H-Isotelluro-chromenes to Bis(o-formylstyryl) Ditellurides. Molecules. 2010; 15(3):1466-1472. https://doi.org/10.3390/molecules15031466
Chicago/Turabian StyleSashida, Haruki, Hirohito Satoh, Kazuo Ohyanagi, and Mamoru Kaname. 2010. "Novel Oxidative Ring Opening Reaction of 1H-Isotelluro-chromenes to Bis(o-formylstyryl) Ditellurides" Molecules 15, no. 3: 1466-1472. https://doi.org/10.3390/molecules15031466
APA StyleSashida, H., Satoh, H., Ohyanagi, K., & Kaname, M. (2010). Novel Oxidative Ring Opening Reaction of 1H-Isotelluro-chromenes to Bis(o-formylstyryl) Ditellurides. Molecules, 15(3), 1466-1472. https://doi.org/10.3390/molecules15031466





