Antioxidant Properties of Cap and Stipe from Coprinus comatus
Abstract
:1. Introduction
2. Results and Discussion
2.1. Extraction Yield
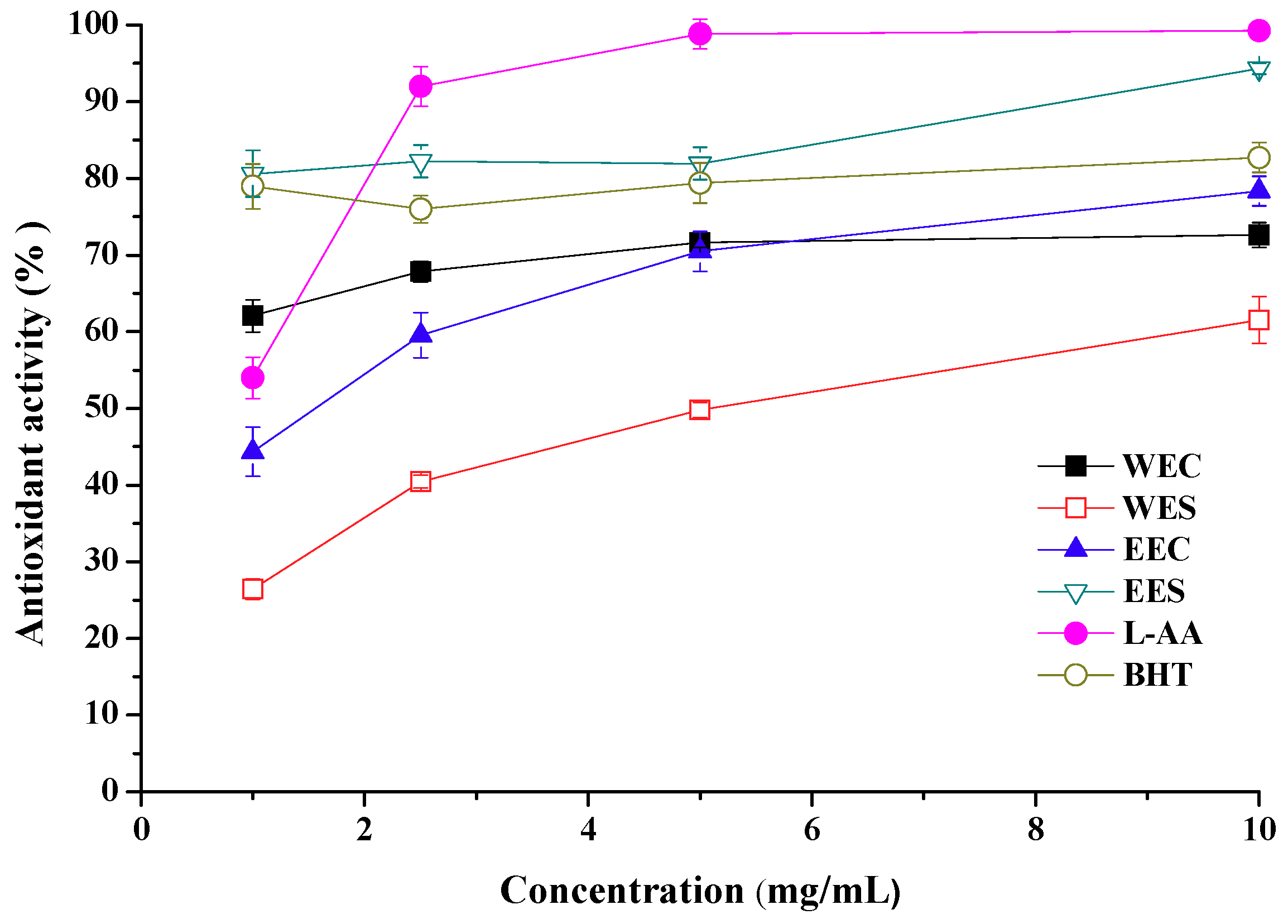
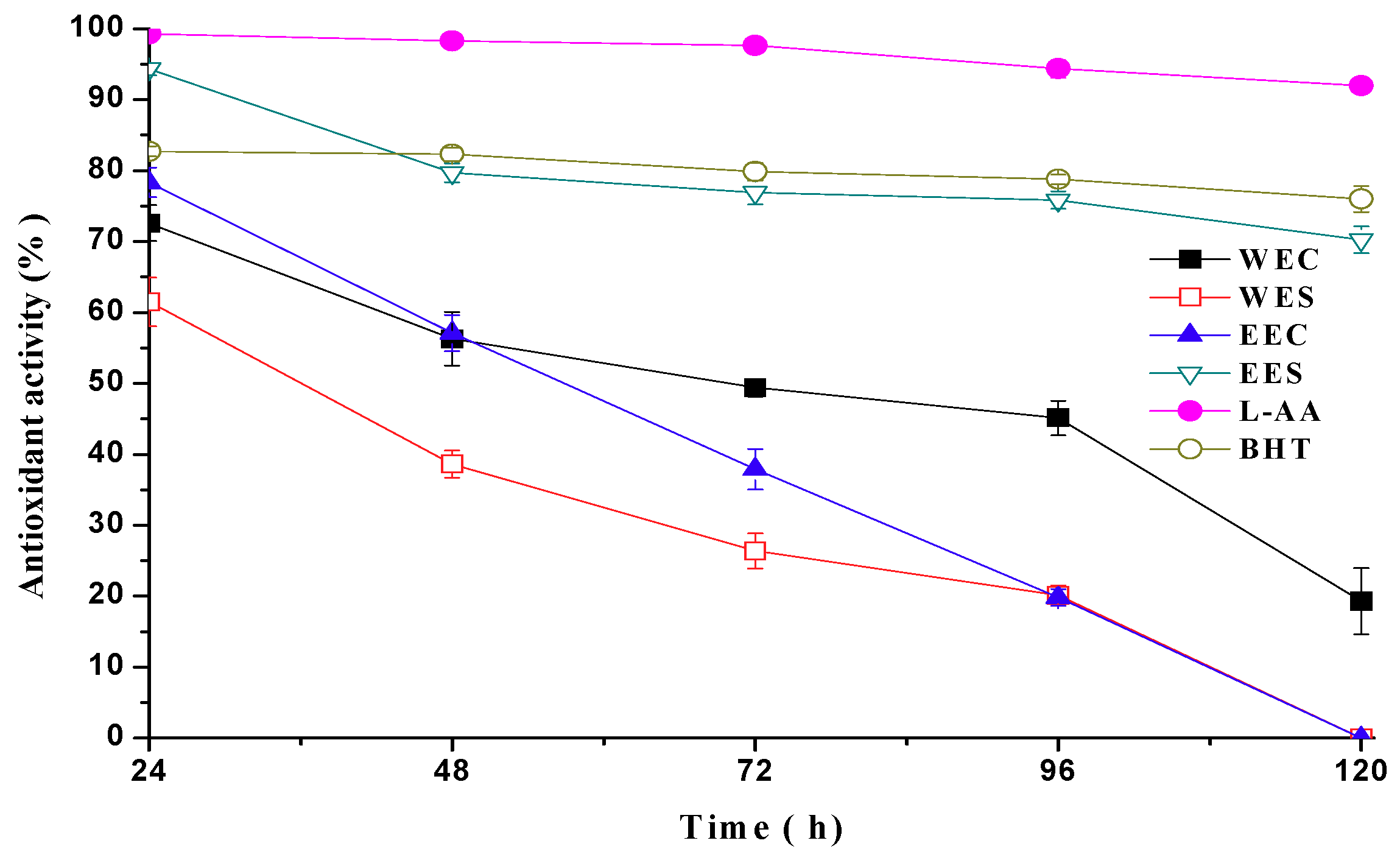
2.2. Antioxidant Activity
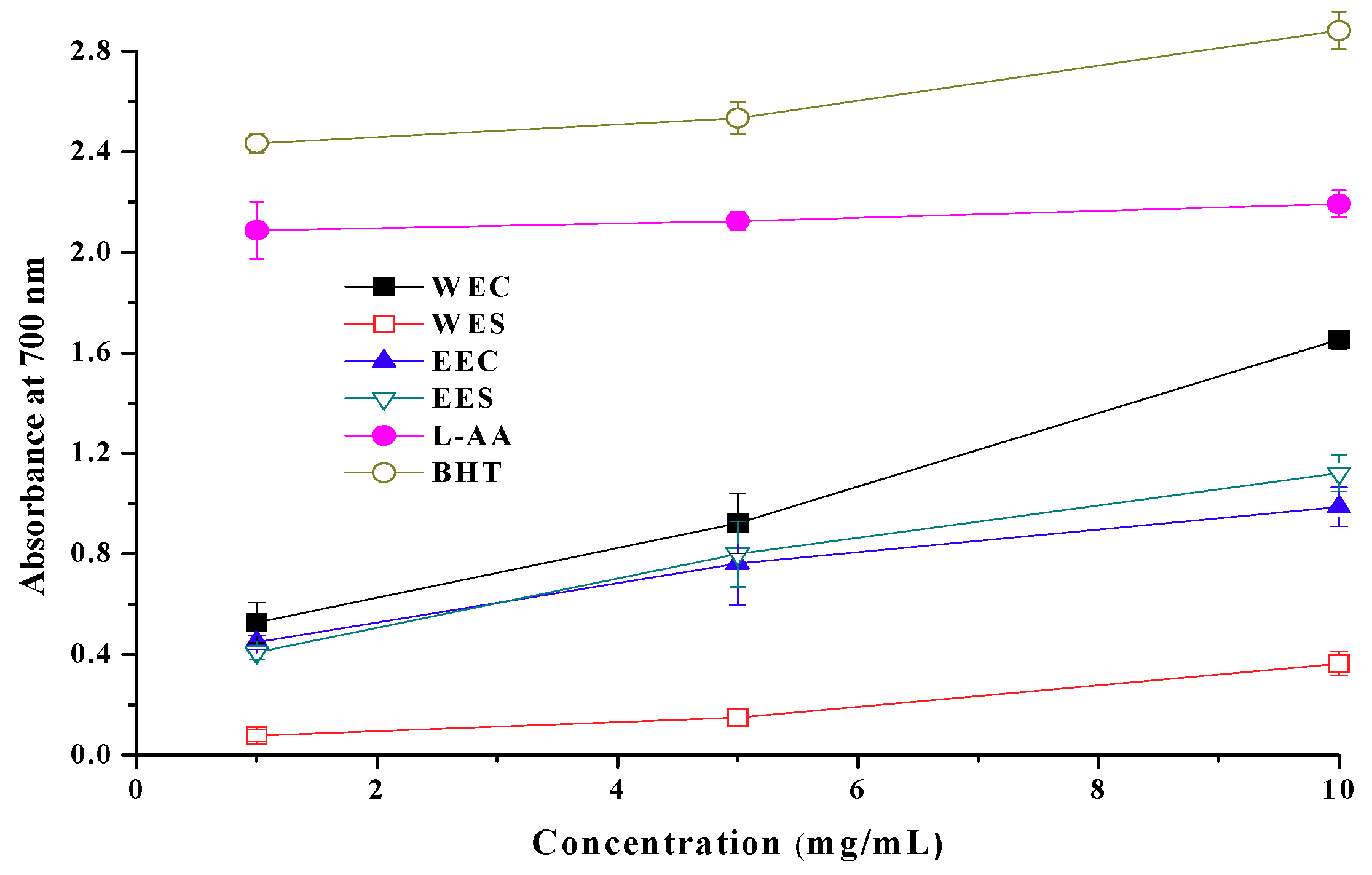
2.3. Reducing Power
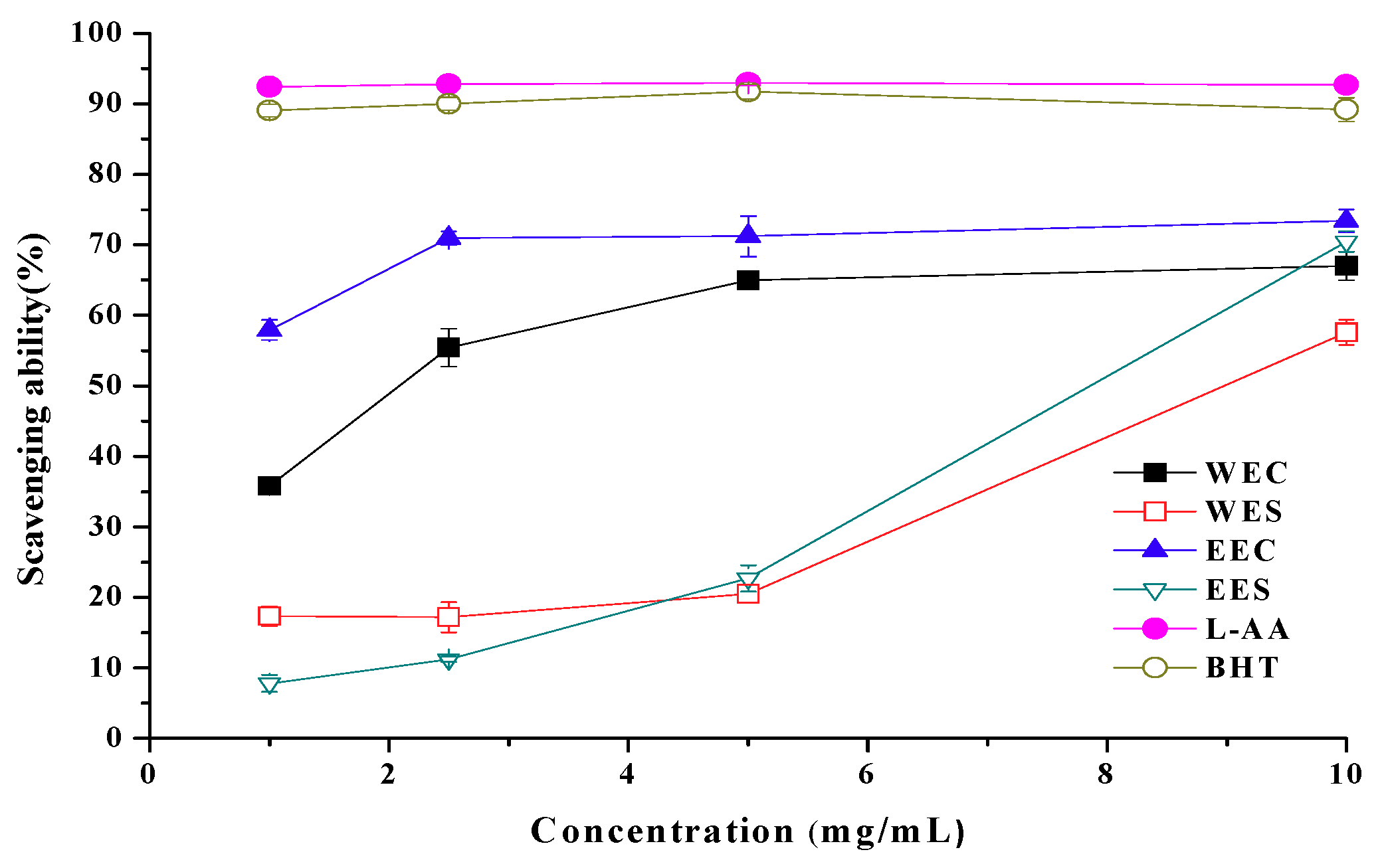
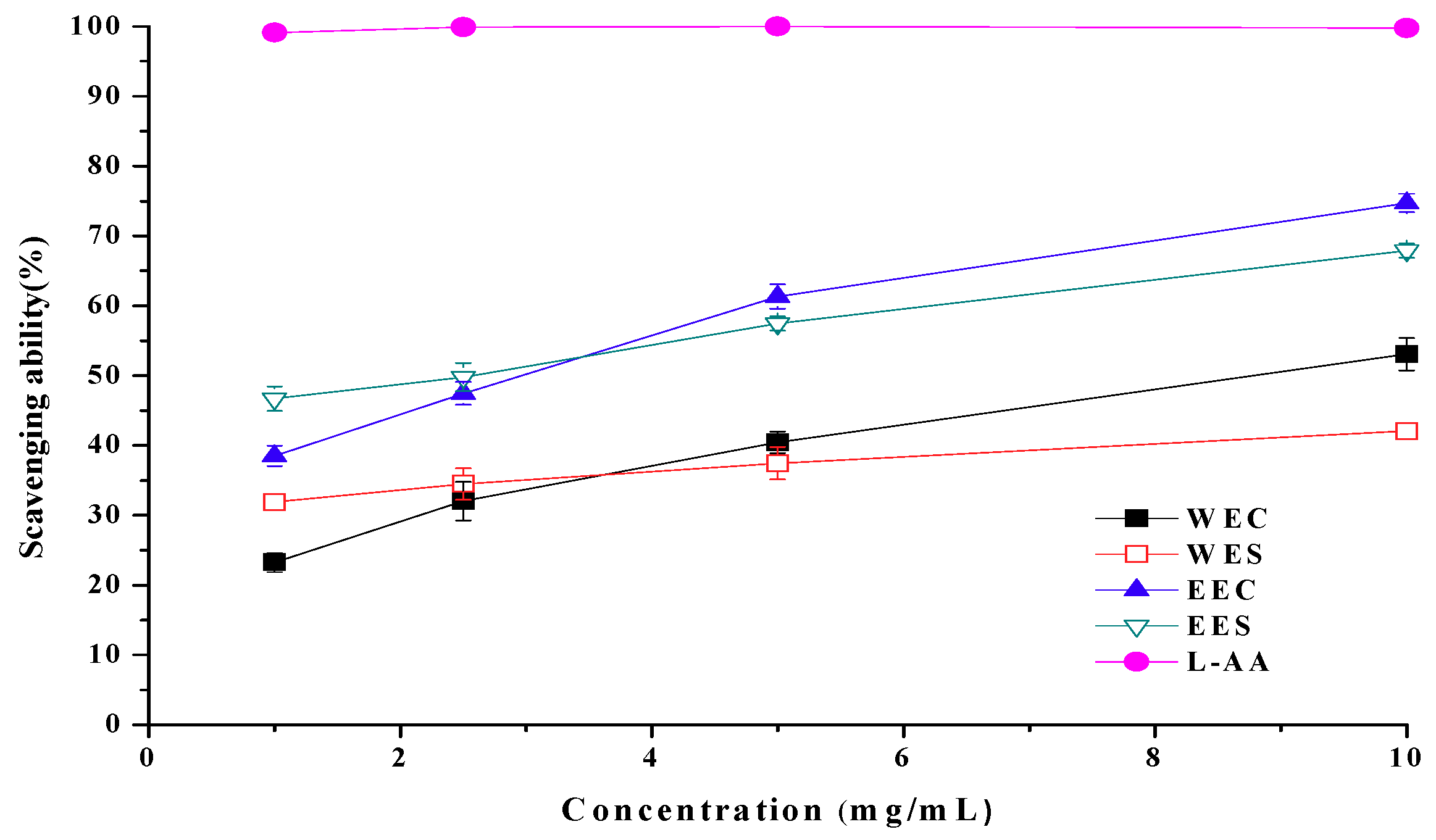
2.4. Scavenging Ability on 1,1-Diphenyl-2-picrylhydrazyl (DPPH) Radicals
2.5. Scavenging Ability on Hydroxyl Radicals
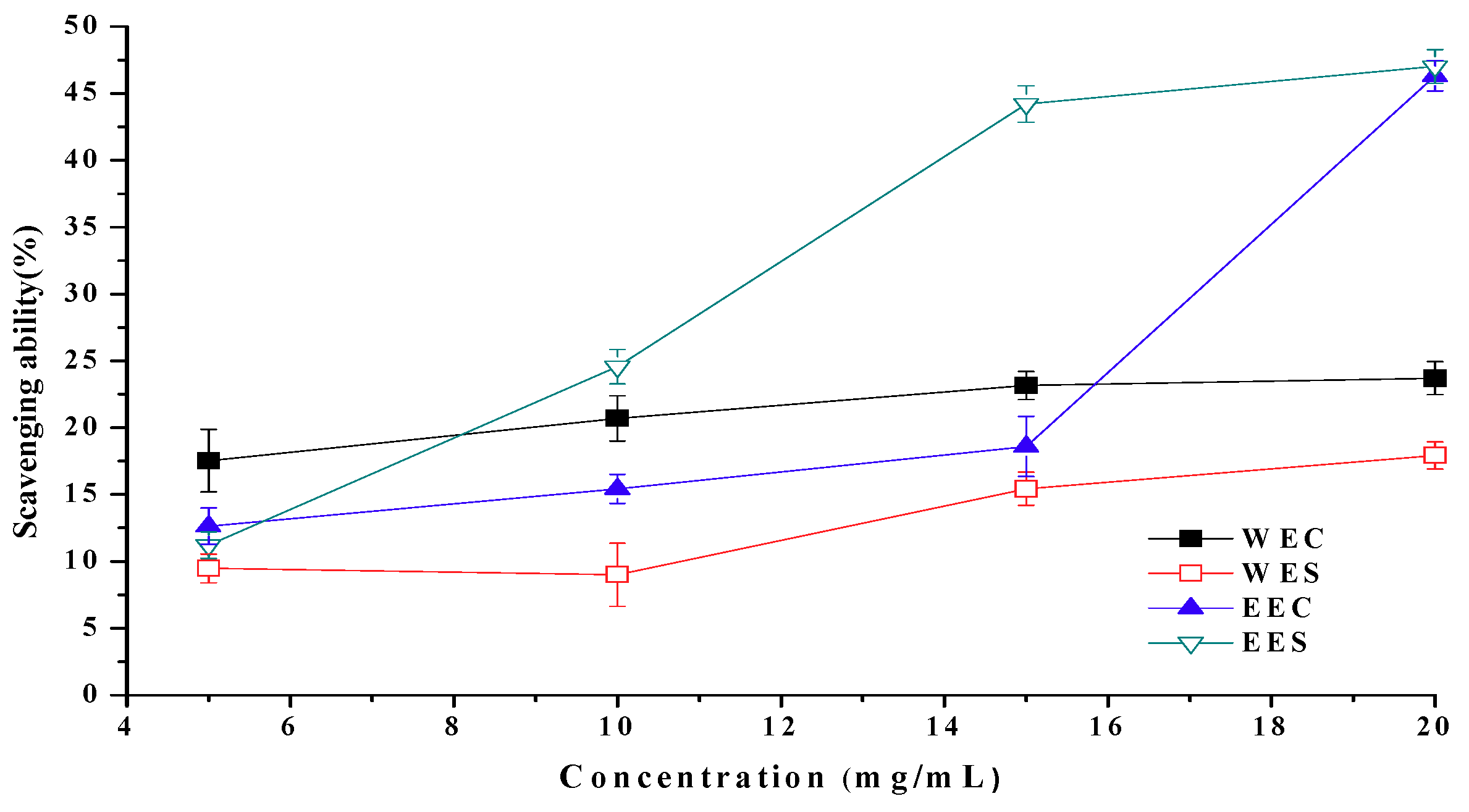
2.6. Scavenging Ability on Superoxide Radicals
2.7. EC50 Values in Antioxidant Properties
2.8. Antioxidant Components
3. Experimental
3.1. Materials
3.2. Extraction
3.3. Antioxidant Activity
3.4. Reducing Power
3.5. Scavenging Ability on DPPH Radicals
3.6. Scavenging Ability on Hydroxyl Radicals
3.7. Scavenging Ability on Superoxide Radicals
3.8. Determination of Antioxidant Components
3.9. Statistical Analysis
4. Conclusions
Acknowledgements
References and Notes
- Bailey, C.J.; Turner, S.L.; Jakeman, K.J.; Hayes, W.A. Effect of Coprinus comatus on plasma glucose concentrations in mice. Planta Med. 1984, 50, 525–526. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.M.; Zhang, J.S.; Tang, Q.J.; Liu, Y.F.; Zhang, A.Q.; Pan, Y.J. Structural elucidation of a neutral fucogalactan from the mycelium of Coprinus comatus. Carbohydr. Res. 2006, 341, 1130–1134. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.H.; Lin, H.C.; Mau, J.L. Antioxidant properties of several commercial mushrooms. Food Chem. 2002, 77, 229–235. [Google Scholar] [CrossRef]
- Mau, J.L.; Chang, C.N.; Huang, S.J.; Chen, C.C. Antioxidant properties of methanolic extracts from Grifola frondosa, Morchella esculenta and Termitomyces albuminosus mycelia. Food Chem. 2004, 87, 111–118. [Google Scholar] [CrossRef]
- Lee, Y.L.; Jian, S.Y.; Lian, P.Y.; Mau, J.L. Antioxidant properties of extracts from a white mutant of the mushroom Hypsizigus marmoreus. J. Food Compos. Anal. 2008, 21, 116–124. [Google Scholar] [CrossRef]
- List, P.H. Occurrence of ergothioneine in shaggy-mane, Coprinus comatus. Arch. Pharm. Ber. Dtsch. Pharm. Ges. 1957, 62, 517–520. [Google Scholar] [CrossRef]
- Badalyan, C.M.; Gasparyan, A.V.; Garibyan, N.G. Investigation of the antioxidant activity of some basidial macromycetes. Mikol. Fitopatol. 2003, 37, 63–68. [Google Scholar]
- Tsai, S.Y.; Tsai, H.L.; Mau, J.L. Antioxidant properties of Coprinus comatus. J. Food Biochem. 2009, 33, 368–389. [Google Scholar] [CrossRef]
- Yu, J.; Cui, P.J.; Zeng, W.L.; Xie, X.L.; Liang, W.J.; Lin, G.B. Protective effect of selenium-polysaccharides from the mycelia of Coprinus comatus on alloxan-induced oxidative stress in mice. Food Chem. 2009, 117, 42–47. [Google Scholar] [CrossRef]
- Suo, X.; Li, B.; Nie, L.; Li, B. Nutritional Components of stalk and cap of Coprinus comatus fruit body. Edible Fungi 2009, 29, 74–75. [Google Scholar]
- Tanaka, M.; Kuie, C.W.; Nagashima, Y.; Taguchi, T. Application of antioxidative Maillard reaction products from histidine and glucose to sadine products. Nippon Suisan Gakk. 1988, 54, 1409–1414. [Google Scholar] [CrossRef]
- Lee, S.E.; Hwang, H.J.; Ha, J.S.; Jeong, H.S.; Kim, J.H. Screening of medicinal plant extracts for antioxidant activity. Life Sci. 2003, 73, 167–179. [Google Scholar] [CrossRef]
- Fridovich, I. Biological effects of superoxide radical. Arch. Biochem. Biophys. 1986, 247, 1–11. [Google Scholar] [CrossRef]
- Boskou, D. Sources of natural phenolic antioxidants. Trends Food Sci. Tech. 2006, 17, 505–512. [Google Scholar] [CrossRef]
- Catherine, R.E.; Nicholas, M.; George, P. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar]
- Catherine, R.E.; Nicholas, M.; George, P. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radical Biol. Med. 1996, 20, 933–956. [Google Scholar]
- Tourino, S.; Selga, A.; Jimenez, A.; Julia, L.; Lozano, C.; Lizarraga, D. Procyanidin fraction from pine (Pinus pinaster) bark: Radical scavenging power in solution, s ntioxidant activity in emulsion, and antiproliferative effect in melanoma cells. J. Agric. Food Chem. 2005, 53, 4728–4735. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ooi, V.E.C.; Chang, S.T. Free radical scavenging activities of mushroom polysaccharide extracts. Life Sci. 1997, 60, 763–771. [Google Scholar] [CrossRef]
- Tseng, Y.H.; Yang, J.H.; Mau, J.L. Antioxidant properties of polysaccharides from Ganoderma tsugae. Food Chem. 2008, 107, 732–738. [Google Scholar] [CrossRef]
- Mitsuda, H.; Yasumodo, K.; Iwami, F. Antioxidant action of indole compounds during the autoxidantion of linoleic acid. Eiyo to Shokuryo 1966, 19, 210–214. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on products of browning reactions: Antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Shimada, K.; Fujikawa, K.; Yahara, K.; Nakamura, T. Antioxidative properties of xanthan on the autoxidantion of soybean oil in cyclodextrin emulsion. J. Agric. Food Chem. 1992, 40, 945–948. [Google Scholar] [CrossRef]
- Smirnoff, N.; Cumbers, Q.J. Hydroxyl radical scavenging activity of compatible solutes. Phytochem. 1989, 28, 1057–1060. [Google Scholar] [CrossRef]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol as a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Yu, Y.; Cao, S. Investigation on the effects of quercetin self-oxidation on its super oxygen-anion free radical scavenging ability. Sci. Tech. Food Industry 2006, 27, 75–78. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic- phosphotungstic acid reagents. Am. J. Enol. Viticult. 1965, 16, 144–158. [Google Scholar]
- Jia, Z.; Tang, M.; Wu, J. The determination of flavonoid content in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–599. [Google Scholar]
- Boyer, R.F. Modern Experimental Biochemistry; Addison-Wesley: New York, NY, USA, 1986. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
| Extract | Extraction yield a (g/100 g dry weight) | |
|---|---|---|
| Ethanolic | Hot water | |
| Stipe | 11.7 ± 0.7 | 46.5 ± 2.3 |
| Cap | 14.5 ± 1.1 | 49.2 ± 0.6 |
| Extract | Antioxidant attribute | EC50 a (mg/mL) | |
|---|---|---|---|
| Cap | Stipe | ||
| Ethanolic | Antioxidant activity Reducing power Scavenging ability on DPPH radicals Scavenging ability on OH radicals Scavenging ability on superoxide radicals | 1.56 ± 0.24 b 1.67 ± 0.13 0.86 ± 0.06 3.23 ± 0.28 25.3 ± 0.21 | 0.62 ± 0.42 1.93 ± 0.23 7.86 ± 0.16 3.35 ± 0.17 20.7 ± 0.38 |
| Hot water | Antioxidant activity Reducing power Scavenging ability on DPPH radicals Scavenging ability on OH radicals Scavenging ability on superoxide radicals | 0.81 ± 0.03 0.95 ± 0.05 2.09 ± 0.26 8.66 ± 0.35 -- c | 5.08 ± 0.17 14.8 ± 0.22 8.98 ± 0.19 16.9 ± 0.41 -- |
| Antioxidant components | Content (mg/g) | ||
|---|---|---|---|
| Ethanolic | Hot water | ||
| Stipe | Total phenols | 9.71 ± 0.26 a | 3.60 ± 0.64 |
| Total flavonoid | 3.52 ± 0.20 | 0.19 ± 0.02 | |
| α-Tocopherol | 11.1 ± 0.14 | 0.52 ± 0.02 | |
| γ-Tocopherol | 0.20 ± 0.01 | 0.05 ± 0.01 | |
| δ-Tocopherol | 0.45 ± 0.03 | 0.01 ± 0.01 | |
| Ascorbic acid | Nd b | nd | |
| Polysaccharide | 64.4 ± 2.56 | 547 ± 4.63 | |
| Cap | Total phenols | 13.5 ± 1.18 | 20.0 ± 0.44 |
| Total flavonoid | 2.13 ± 0.31 | 1.88 ± 0.39 | |
| α-Tocopherol | 11.0 ± 0.18 | 0.64 ± 0.03 | |
| γ-Tocopherol | 0.29 ± 0.01 | 0.04 ± 0.01 | |
| δ-Tocopherol | 0.62 ± 0.04 | 0.03 ± 0.01 | |
| Ascorbic acid | nd | nd | |
| Polysaccharide | 58.5 ± 3.12 | 269.7 ± 6.64 | |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, B.; Lu, F.; Suo, X.; Nan, H.; Li, B. Antioxidant Properties of Cap and Stipe from Coprinus comatus. Molecules 2010, 15, 1473-1486. https://doi.org/10.3390/molecules15031473
Li B, Lu F, Suo X, Nan H, Li B. Antioxidant Properties of Cap and Stipe from Coprinus comatus. Molecules. 2010; 15(3):1473-1486. https://doi.org/10.3390/molecules15031473
Chicago/Turabian StyleLi, Bo, Fei Lu, Xiaomin Suo, Haijuan Nan, and Bin Li. 2010. "Antioxidant Properties of Cap and Stipe from Coprinus comatus" Molecules 15, no. 3: 1473-1486. https://doi.org/10.3390/molecules15031473
APA StyleLi, B., Lu, F., Suo, X., Nan, H., & Li, B. (2010). Antioxidant Properties of Cap and Stipe from Coprinus comatus. Molecules, 15(3), 1473-1486. https://doi.org/10.3390/molecules15031473




