Identification of Major Phenolic Compounds from Nephelium lappaceum L. and Their Antioxidant Activities
Abstract
:1. Introduction
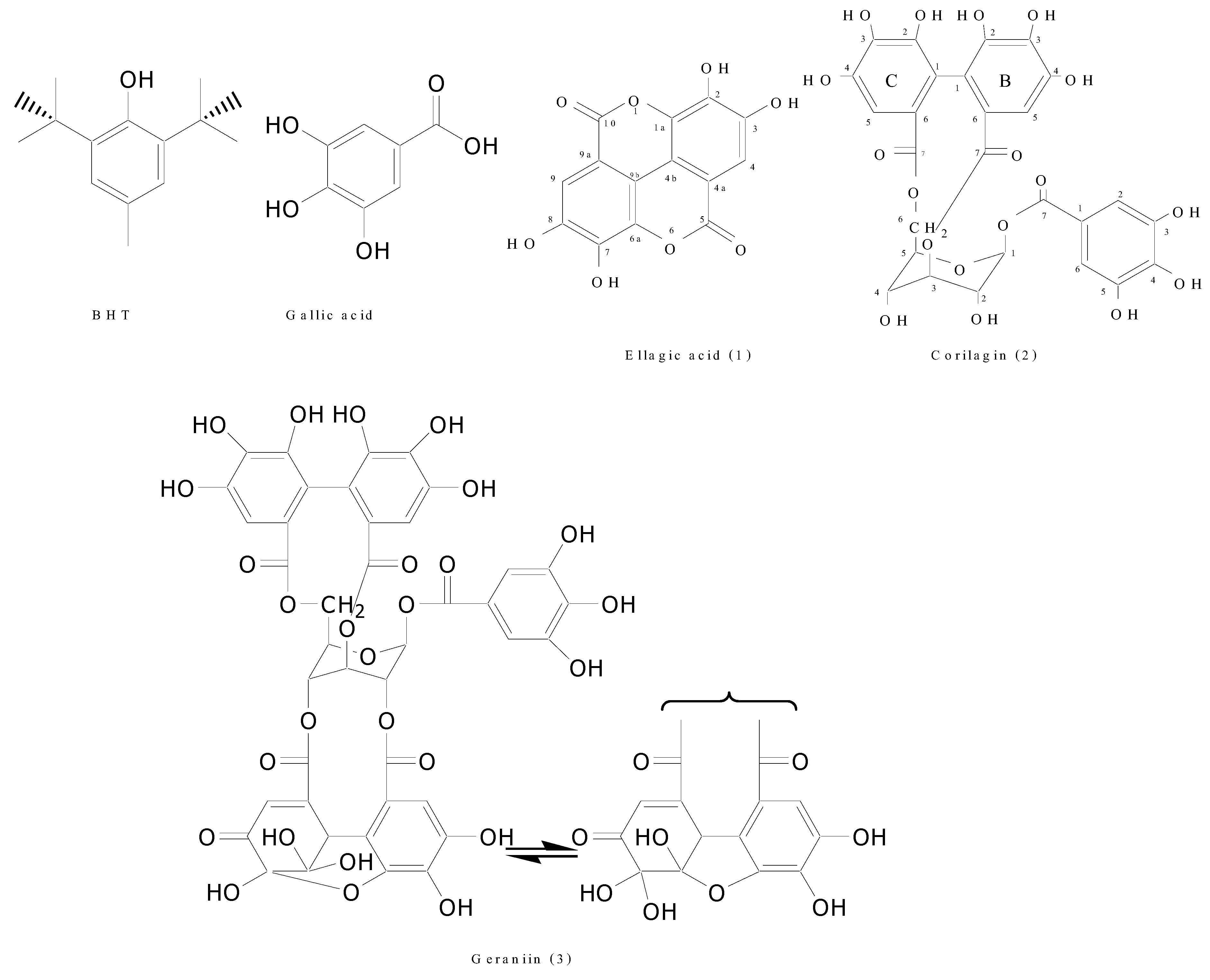
2. Results and Discussion
2.1. Isolation and identification of bioactive compounds from N. lappaceum peel
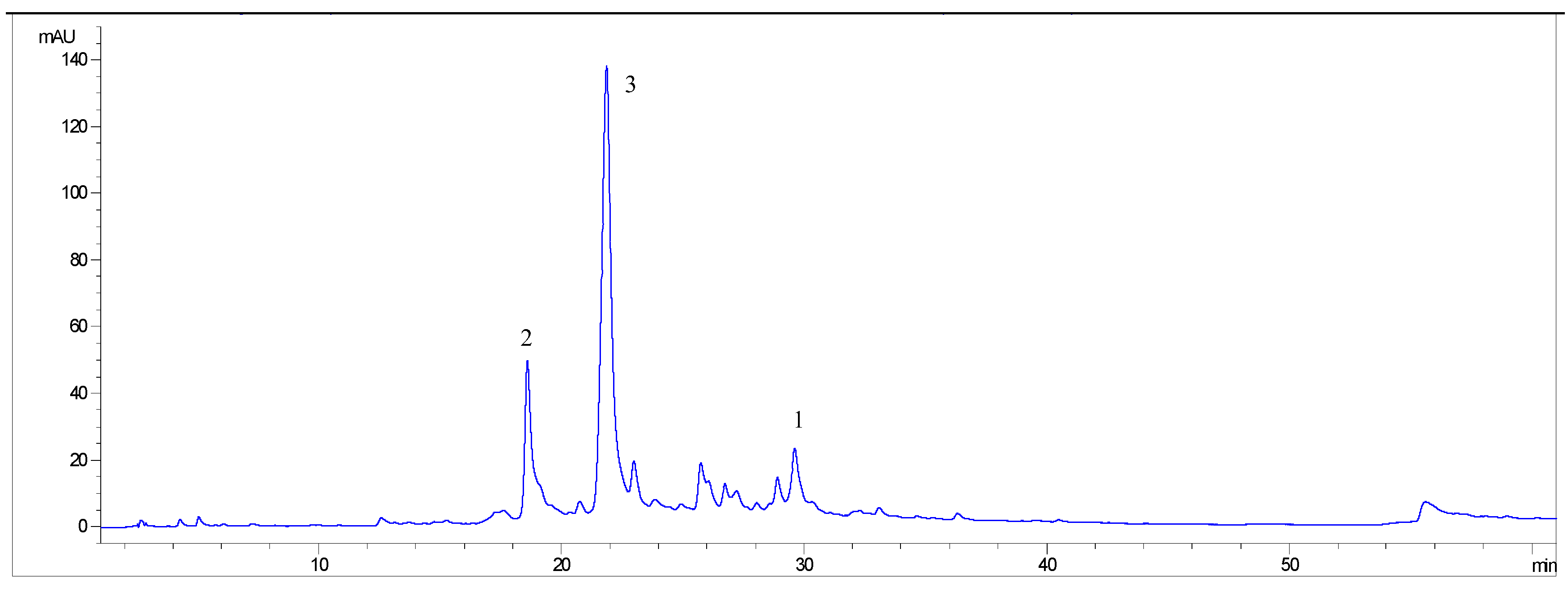
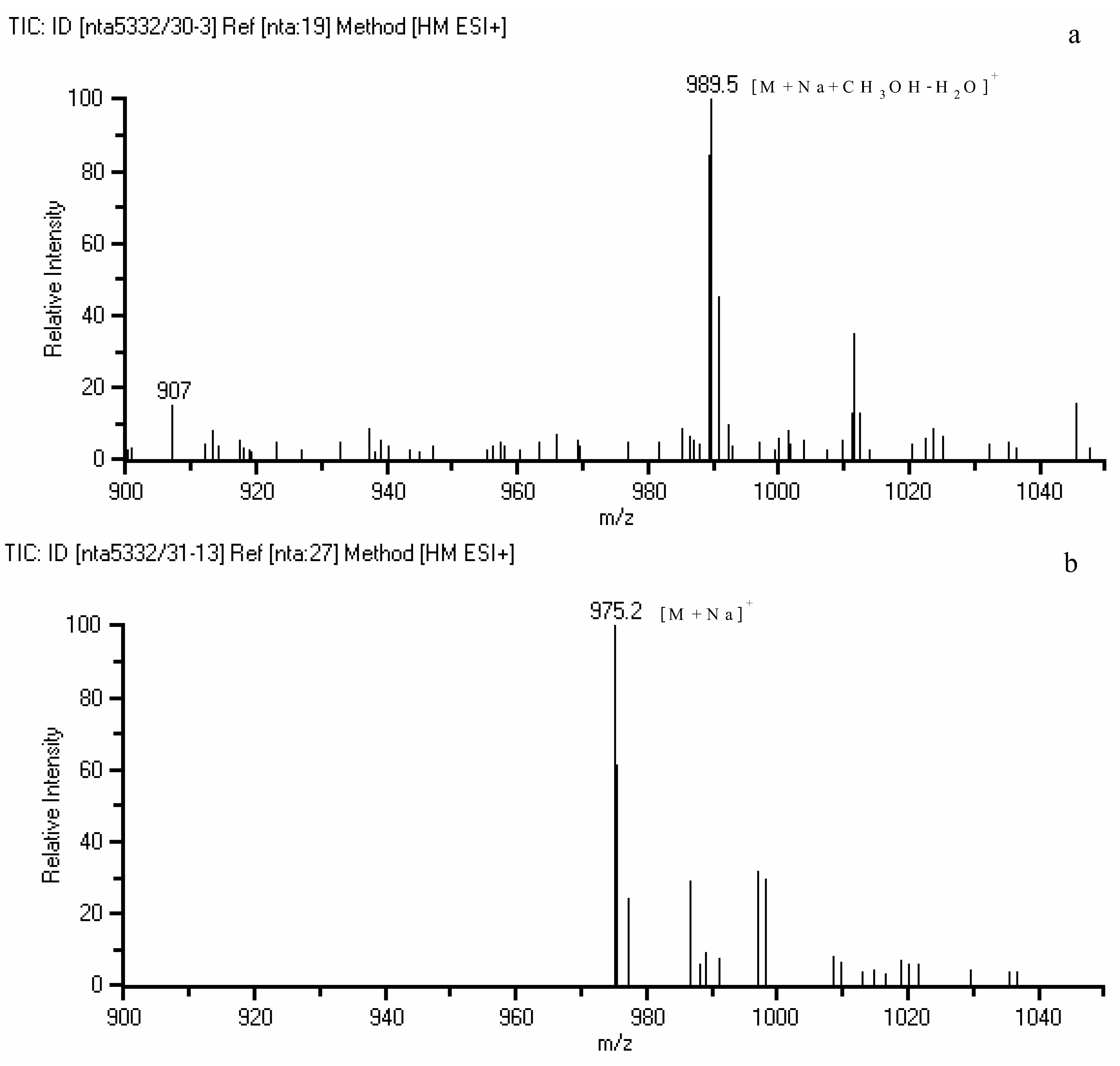
2.2. Quantification of bioactive identified components
2.3. Antioxidant activity
| Compounds | Lipid peroxidation inhibition capacity | DPPH• scavenging activity | ||
|---|---|---|---|---|
| IC50 (µM) | Mol BHT/mol phenolics | IC50 (µM) | Mol BHT/mol phenolics | |
| Geraniin | 0.38 ± 0.01 a | 186 ± 3.00 | 0.79 ± 0.05 a | 87.1 ± 5.76 |
| Corilagin | 0.70 ± 0.03 b | 103 ± 3.72 | 1.42 ± 0.10 b | 48.6 ± 3.44 |
| Ellagic acid | 0.94 ± 0.10 c | 76.9 ± 7.63 | 1.64 ± 0.15 b | 42.4 ± 3.85 |
| Gallic acid | 1.70 ± 0.13 d | 42.2 ± 3.10 | 2.49 ± 0.11 c | 27.7 ± 1.15 |
| BHT | 71.3 ± 0.86 | - | 68.8 ± 2.10 | - |
| Reference | IC50 (µM) | |||
|---|---|---|---|---|
| gallic acid | ellagic acid | corilagin | geraniin | |
| Zheng et al., 2009 [30] | 4.71 | 3.17 | ||
| Latté et al., 2004 [31] | 32.9 | 2.70 | ||
| Xu et al., 2007 [32] | 27.4 | 18.7 | ||
| Lin et al., 2008 [24] | 1.27 (pH 7.9), 0.92 (pH 4.5) | |||
| Yokozawa et al., 1999 [33] | 8.14 | 4.60 | 2.89 | 2.50 |
| This work | 2.49 | 1.64 | 1.42 | 0.79 |
3. Experimental
3.1. General
3.2. Extraction, isolation and identification of bioactive compounds from N. lappaceum peel
3.3. Phenazine derivative preparation
3.4. Analytical HPLC and quantification of bioactive components
3.5. Determination of antioxidant activities
3.5.1. Linoleic peroxidation method
3.5.2. DPPH• scavenging activity
3.6. Statistical analysis
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds are available from the authors.
References
- Croft, K.D. Antioxidant effects of plant phenolic compounds. In Antioxidants in Human Health and Disease; Basu, T.K., Temple, N.J., Garg, M.L., Eds.; CABI Publishing: New York, NY, USA, 1999; pp. 109–112. [Google Scholar]
- Halliwell, B. Antioxidants in human health and disease. Annu. Rev. Nutr. 1996, 16, 33–50. [Google Scholar] [CrossRef]
- Barlow, S.M. Toxicological aspects of antioxidants used as food additives. In Food Antioxidants; Hudson, B.J.F., Ed.; Elsevier Applied Science: New York, NY, USA, 1990; pp. 253–307. [Google Scholar]
- Chanwitheesuk, A.; Teerawutgulrag, A.; Rakariyatham, N. Screening of antioxidant activity and antioxidant compounds of some edible plants of Thailand. Food Chem. 2005, 92, 491–497. [Google Scholar] [CrossRef]
- Pratt, D.E. Natural antioxidants from plant material. In Phenolic Compounds in Food and Their Effects on Health; Huang, I.M.T., Ho, C.T., Lee, C.Y., Eds.; American Chemical Society: Washington, DC, USA, 1992; pp. 54–72. [Google Scholar]
- Jayaprakasha, G.K.; Singh, R.P.; Sakariah, K.K. Antioxidant activity of grape seed (Vitis vinifera) extracts on peroxidation models in vitro. Food Chem. 2001, 73, 285–290. [Google Scholar] [CrossRef]
- Negro, C.; Tommasi, L.; Miceli, A. Phenolic compounds and antioxidant activity from red grape marc extracts. Bioresour. Technol. 2003, 87, 41–44. [Google Scholar] [CrossRef]
- Singh, R.P.; Murthy, K.N.C.; Jayaprakasha, G.K. Studies on the antioxidant activity of pomegranate (Punica granatum) peel and seed extracts using in vitro models. J. Agric. Food Chem. 2002, 50, 81–86. [Google Scholar] [CrossRef]
- Luo, W.; Zhao, M.; Yang, B.; Shen, G.; Rao, G. Identification of bioactive compounds in Phyllenthus emblica L. fruit and their free radical scavenging activities. Food Chem. 2009, 114, 499–504. [Google Scholar] [CrossRef]
- Chung, S.-K.; Nam, J.-A.; Jeon, S.-Y.; Kim, S.-I.; Lee, H.-J.; Chung, T.H.; Song, K.-S. A prolyl endopeptidase-inhibiting antioxidant from Phyllanthus ussurensis. Arch. Pharm. Res. 2003, 26, 1024–1028. [Google Scholar] [CrossRef]
- Foo, L.Y.; Wong, H. Phyllanthusiin D, an unusual hydrolysable tannin from Phyllanthus amarus. Phytochem. 1992, 31, 711–713. [Google Scholar]
- Thitilertdecha, N.; Teerawutgulrag, A.; Rakariyatham, N. Antioxidant and antibacterial activities of Nephelium lappaceum L. extracts. LWT Food Sci. Technol. 2008, 41, 2029–2035. [Google Scholar] [CrossRef]
- Foo, L.Y. Amariin, a di-dehydrohexahydroxydiphenoyl hydrolysable tannin from Phyllanthus amarus. Phytochemisty 1993, 33, 487–491. [Google Scholar] [CrossRef]
- Grundhöfer, P.; Niemetz, R.; Schilling, G.; Gross, G.G. Biosynthesis and subcellular distribution of hydrolyzable tannins. Phytochemistry 2001, 57, 915–927. [Google Scholar] [CrossRef]
- Luger, P.; Weber, M.; Kashino, S.; Amakura, Y.; Yoshida, T.; Okuda, T.; Beurskens, G.; Dauter, Z. Structure of the tannin geraniin based on conventional X-ray data at 295 K and on Synchrotron data at 293 and 120 K. Acta Cryst. 1998, B54, 687–694. [Google Scholar]
- Ito, H.; Iguchi, A.; Hatano, T. Identification of urinary and intestinal bacterial metabolites of ellagitannin geraniin in rats. J. Agric. Food Chem. 2008, 56, 393–400. [Google Scholar]
- Notka, F.; Meier, G.; Wagner, R. Concerted inhibitory activities of Phyllanthus amarus on HIV replication in vitro and ex vivo. Antiviral Res. 2004, 64, 93–102. [Google Scholar]
- Yang, C.-M.; Cheng, H.-Y.; Lin, T.-C.; Chiang, L.-C.; Lin, C.-C. The in vitro activity of geraniin and 1,3,4,6-tetra-O-galloyl-β-D-glucose isolated from Phyllanthus urinaria against herpes simplex virus type 1 and type 2 infection. J. Ethnopharmacol. 2007, 110, 555–558. [Google Scholar] [CrossRef]
- Rogerio, A.P.; Fontanari, C.; Borducchi, É.; Keller, A.C.; Rusco, M.; Soares, E.G.; Albuquerque, D.A.; Faccioli, L.H. Anti-inflammatory effects of Lafoensia pacari and ellagic acid in a murine model of asthma. Eur. J. Pharmacol. 2008, 580, 262–270. [Google Scholar]
- Kashiwada, Y. Antitumor agent, 129. Tannins and related compounds as selective cytotoxic agents. J. Nat. Prod. 1992, 55, 1033–1043. [Google Scholar] [CrossRef]
- Larrosa, M.; Tomás-Barberán, F.A.; Espín, J.C. The dietary hydrolysable tannin punicalagin releases ellagic acid that induces apoptosis in human colon adenocarcinoma Caco-2 cells by using the mitochondrial pathway. J. Nutr. Biochem. 2006, 17, 611–625. [Google Scholar] [CrossRef]
- Srivastava, A.; Rao, L.J.M.; Shivanandappa, T. Isolation of ellagic acid from the aqueous extract of the roots of Decalepis hamiltonii: Antioxidant activity and cytoprotective effect. Food Chem. 2007, 103, 224–233. [Google Scholar] [CrossRef]
- Reddy, M.K.; Gupta, S.K.; Jacob, M.R.; Khan, S.I.; Ferreira, D. Antioxidant, antimalarial and antimicrobial activities of tannin-rich fractions, ellagitannins and phenolic acids from Punica granatum L. Planta Medica 2007, 73, 461–467. [Google Scholar] [CrossRef]
- Lin, S.-Y.; Wang, C.-C.; Lu, Y.-L.; Wu, W.-C.; Hou, W.-C. Anitoxidant, anti-semicarbazide-sensitive amine oxidase, and anti-hypertensive activities of geraniin isolated from Phyllanthus urinaria. Food Chem. Toxicol. 2008, 46, 2485–2492. [Google Scholar] [CrossRef]
- Okonogi, S.; Duangrat, C.; Anuchpreeda, S.; Tachakittirungrod, S.; Chowwanapoonpohn, S. Comparison of antioxidant capacities and cytotoxicities of certain fruit peels. Food Chem. 2007, 103, 839–846. [Google Scholar] [CrossRef]
- Palanisamy, U.; Cheng, H.M.; Masilamani, T.; Subramaniam, T.; Ling, L.T.; Radhakrishnan, A.K. Rind of rambutan, Nephelium lappaceum, a potential source of natural antioxidants. Food Chem. 2008, 109, 54–63. [Google Scholar] [CrossRef]
- Box, H.C.; Maccubbin, A.E. Lipid peroxidation and DNA damage. Nutrition 1997, 13, 920–921. [Google Scholar] [CrossRef]
- Priyadarsini, K.I.; Khopde, S.M.; Kumar, S.S.; Mohan, H. Free radical studies of ellagic acid, a natural phenolic antioxidant. J. Agric. Food Chem. 2002, 50, 2200–2206. [Google Scholar] [CrossRef]
- Yokozawa, T.; Chen, C.P.; Dong, E.; Tanaka, T.; Nonaka, G.-I.; Nishioka, I. Study on the inhibitory effect of tannins and flavonoids against the 1,1-diphenyl-2-picrylhydrazyl radical. Biochem. Pharmacol. 1998, 56, 213–222. [Google Scholar]
- Zheng, G.; Xu, L.; Wu, P.; Xie, H.; Jiang, Y.; Chen, F.; Wei, X. Polyphenols from longan seeds and their radical-scavenging activity. Food Chem. 2009, 433–436. [Google Scholar]
- Latté, K.P.; Kolodziej, H. Antioxidant properties of phenolic compounds from Pelargonium reniforme. J. Agric. Food Chem. 2004, 52, 4899–4902. [Google Scholar] [CrossRef]
- Xu, M.; Zha, Z.-J.; Qin, X.-L.; Zhang, X.-L.; Yang, C.-R.; Zhang, Y.-J. Phenolic antioxidants from the whole plant of Phyllanthus urinaria. Chem. Biodiver. 2007, 2246–2252. [Google Scholar]
- Yokozawa, T.; Chen, C.P.; Dong, E.; Tanaka, T.; Nonaka, G.-I.; Nishioka, I. Study on the inhibitory effect of tannins and flavonoids against the 1,1-diphenyl-2-picrylhydrazyl radical. Biochem. Pharmacol. 1998, 213–222. [Google Scholar]
- Cai, Y.-Z.; Sun, M.; Xing, J.; Luo, Q.; Corke, H. Structure-radical scavenging activity relationships of phenolic compounds from traditional Chinese medicinal plants. Life Sci. 2006, 78, 2872–2888. [Google Scholar] [CrossRef]
- Kinoshita, S.; Inoue, Y.; Nakama, S.; Ichiba, T.; Aniya, Y. Antioxidant and hepatoprotective actions of medicinal herb, Terminalia catappa L. from Okinawa Island and its tannin corilagin. Phytomedicine 2007, 14, 755–762. [Google Scholar] [CrossRef]
- Montealegre, R.R.; Peces, R.R.; Vozmediano, J.L.C.; Gascueňa, J.M.; Romero, E.G. Phenolic compounds in skins and seeds of ten grape Vitis vinifera varieties grown in a warm climate. J. Food Compos. Anal. 2006, 19, 687–693. [Google Scholar] [CrossRef]
- Latté, K.P.; Kolodziej, H. Pelargoniins, new ellagitannins from Pelargonium reniforme. Phytochemisty 2000, 54, 701–708. [Google Scholar] [CrossRef]
- Gohar, A.A.; Lahloub, M.F.; Niwa, M. Antibacterial polyphenol from Erodium glaucophyllum. Z. Naturforsch. 2003, 58c, 670–674. [Google Scholar]
- Yen, G.-C.; Hsieh, C.-L. Antioxidant activity of extracts from Du-zhong (Eucommia ulmoides) toward various lipid peroxidation models in vitro. J. Agric. Food Chem. 1998, 46, 3952–3957. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 26, 1199–1200. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Thitilertdecha, N.; Teerawutgulrag, A.; Kilburn, J.D.; Rakariyatham, N. Identification of Major Phenolic Compounds from Nephelium lappaceum L. and Their Antioxidant Activities. Molecules 2010, 15, 1453-1465. https://doi.org/10.3390/molecules15031453
Thitilertdecha N, Teerawutgulrag A, Kilburn JD, Rakariyatham N. Identification of Major Phenolic Compounds from Nephelium lappaceum L. and Their Antioxidant Activities. Molecules. 2010; 15(3):1453-1465. https://doi.org/10.3390/molecules15031453
Chicago/Turabian StyleThitilertdecha, Nont, Aphiwat Teerawutgulrag, Jeremy D. Kilburn, and Nuansri Rakariyatham. 2010. "Identification of Major Phenolic Compounds from Nephelium lappaceum L. and Their Antioxidant Activities" Molecules 15, no. 3: 1453-1465. https://doi.org/10.3390/molecules15031453
APA StyleThitilertdecha, N., Teerawutgulrag, A., Kilburn, J. D., & Rakariyatham, N. (2010). Identification of Major Phenolic Compounds from Nephelium lappaceum L. and Their Antioxidant Activities. Molecules, 15(3), 1453-1465. https://doi.org/10.3390/molecules15031453




