Antioxidant Phenolic Compounds of Dracaena Cambodiana
Abstract
:1. Introduction
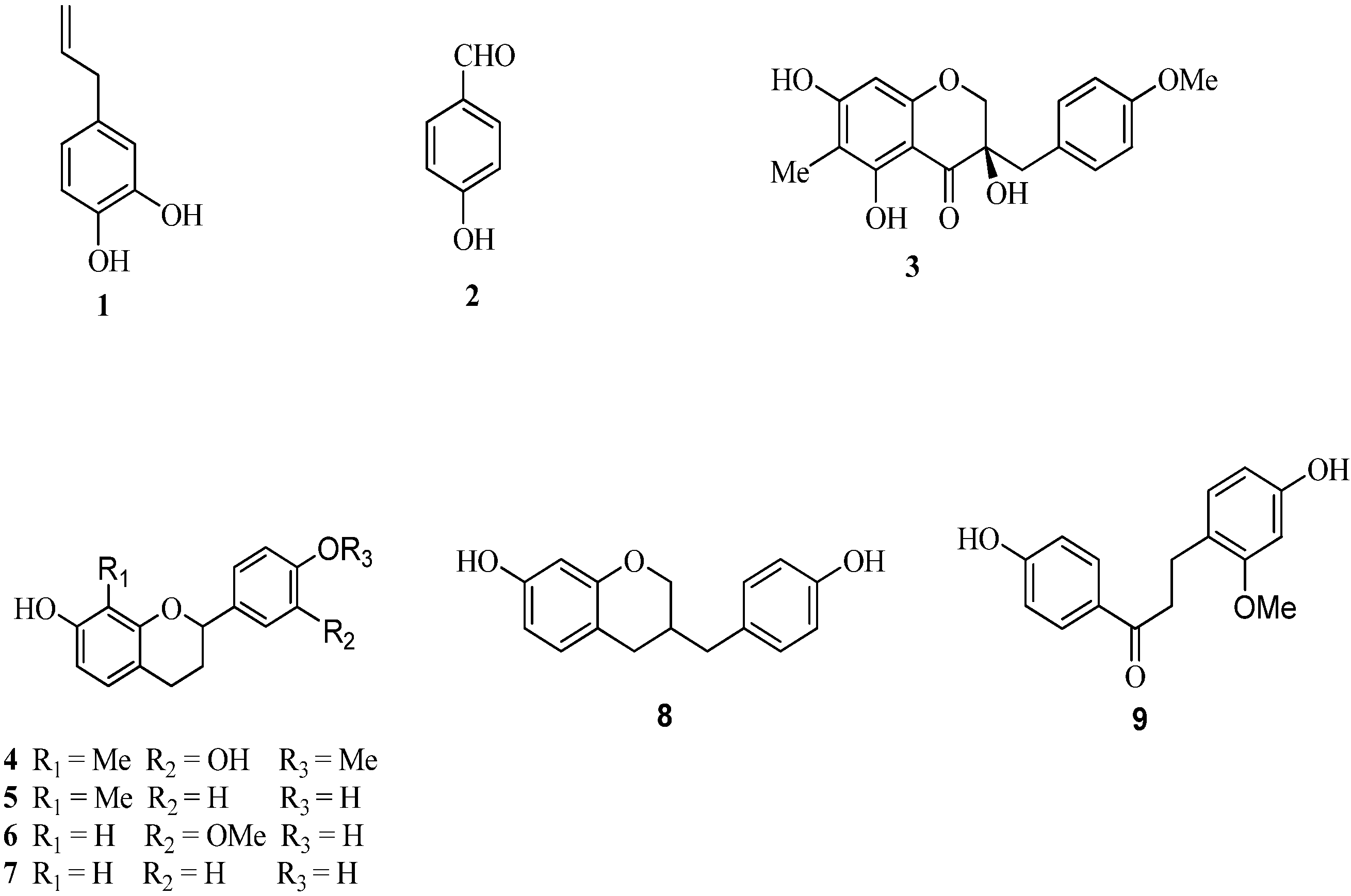
2. Results and Discussion
2.1. Fractions
| Concentration μg/mL | DPPH radical-scavenging capacity (%) | ABTS free radical-scavenging capacity (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| PEF | EAF | BUF | AF | Vc | PEF | EAF | BUF | AF | Trolox® | |
| 800 | 11.90 | 29.49 | 18.22 | 12.45 | ND | 28.79 | 69.10 | 42.42 | 35.96 | ND |
| 400 | - | 16.02 | - | - | ND | 17.56 | 39.47 | 25.43 | 23.46 | ND |
| 200 | 2.93 | - | 6.04 | 7.33 | ND | 12.08 | 23.03 | 15.73 | 17.98 | 99.58 |
| 100 | 1.60 | 4.30 | 3.30 | 2.66 | 73.22 | 7.86 | 14.61 | 10.39 | 8.43 | 65.45 |
| 50 | 0.64 | 3.94 | 2.84 | 1.74 | 35.61 | 5.62 | 10.39 | 7.44 | 7.16 | 38.34 |
| 25 | 0.18 | 2.66 | 0.73 | 1.65 | 19.56 | 4.78 | 9.41 | 6.60 | 5.06 | 16.43 |
| 12.5 | ND | ND | ND | ND | 5.41 | ND | ND | ND | ND | 10.39 |
| 6.25 | ND | ND | ND | ND | 4.71 | ND | ND | ND | ND | 6.60 |
| Test materials | DPPH EC50 | ABTS IC50 | Superoxideradical (mol α-Tocopherol/g DW) | Total phenolics (GAE g/g extract) | Total flavonoids (RE mg/g extract) |
|---|---|---|---|---|---|
| PEF | 5.007 | 1.510 | 0.221 ± 0.12 | 4.88 ± 1.35b | 0 |
| EAF | 1.613 | 0.535 | 1.028 ±0.53 | 27.82 ± 1.02a | 65.70 ± 2.44a |
| BUF | 2.441 | 0.884 | 0.939 ± 0.46 | 9.06 ± 0.26b | 43.59 ± 2.45a |
| AF | 4.874 | 1.100 | 0.419 ± 0.32 | 5.02 ± 0.18b | 15.68 ± 3.14b |
2.2. Sub-Fractions
| Test materials | DPPH EC50 | ABTS IC50 | Superoxide radical (mol α-tocopherol/g DW) |
|---|---|---|---|
| Compound 1 | 0.087 | 0.010 | 4.348 ± 1.74 |
| Compound 2 | 0.108 | 0.012 | 1.362 ± 0.58 |
| Compound 3 | – | 0.138 | 0.272 ± 0.15 |
| Compound 4 | 0.262 | 0.275 | 0.087 ± 0.02 |
| Compound 5 | 0.535 | 0.033 | – |
| Compound 6 | 0.413 | 0.040 | 0.551 ± 0.46 |
| Compound 7 | – | 0.021 | 0.125 ± 0.02 |
| Compound 8 | – | 0.022 | – |
| Compound 9 | – | 0.046 | – |
| Ascorbic acid | 0.069 | ND | ND |
| Trolox® | ND | 0.089 | ND |
2.3. The Structure-Activity Relationships of Compounds Derived from D. Cambodiana
3. Experimental
3.1. General
3.2. Plant Materials
3.3. Extraction and Isolation of Antioxidant Compounds
3.4. Assays
3.4.1. DPPH scavenging capacity
3.4.2. ABTS free radical scavenging assay
3.4.3. Superoxide radical-scavenging capacity
3.4.4. Determination of the amount of total phenolics
3.4.5. Determination of the amount of total flavonoids
3.5. Statistical Analysis
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds 1–9 are available from the authors.
References
- Aruoma, O.I. Free radicals, oxidative stress, and antioxidants in human health and disease. J. Am. Oil Chem. Soc. 1998, 75, 199–212. [Google Scholar] [CrossRef]
- Block, G.; Langseth, L. Antioxidant vitamins and disease prevention. Food Technol. 1994, 48, 80–84. [Google Scholar]
- Frankel, E.N.; Huang, S.W.; Aeschbach, R.; Prior, E. Anti-oxidant activity of a rosemary extract and its constituents, carnosic acid, carnosol, and rosmarinic acid, in bulk oil and oil-in-water emul-sion. J. Agric. Food Chem. 1996, 44, 131–135. [Google Scholar] [CrossRef]
- Madsen, H.L.; Bertelsen, G. Spices as antioxidants. Trends Food Sci. Tech. 1995, 6, 271–277. [Google Scholar] [CrossRef]
- Shahidi, F.; Wanasundara, P.K.J.D. Phenolic anti-oxidants. Crit. Rev. Food. Sci. Nutr. 1992, 32, 67–103. [Google Scholar] [CrossRef]
- Lachman, J.; Pivec, V.; Hubáček, J.; Řeháková, V. Flavonoid substances in the fruits of sea buckthorn (Hippophaë rhamnoides L.). Sci. Agric. Bohem. 1985, 17, 169–183. [Google Scholar]
- Lachman, J.; Fernández, E.C.; Orsák, M. Yacon [Smallanthus sonchifolia (Poepp. Et Endl.) H. Robinson] chemical composition and use – a review. Plant Soil Environ. 2003, 49, 283–290. [Google Scholar]
- Nsimba, R.Y.; Kikuzaki, H.; Konishi, Y. Antioxidant activity of various extracts and fractions of Chenopodium quinoa and Amaranthus spp. seeds. Food Chem. 2008, 106, 760–766. [Google Scholar] [CrossRef]
- Chang, S.K.; Sung, P.M. Antioxidant activities of ethanol extracts from seeds in fresh Bokbunja (Rubus coreanus Miq.) and wine processing waste. Bioresource Technol. 2008, 99, 4503–4509. [Google Scholar] [CrossRef]
- Sousa, M.M.; Melo, M.J.; Parola, A.J.; Seixas de Melo, J.S.; Catarino, F.; Pina, F.; Cook, F.E.; Simmonds, M.S.; Lopes, J.A. Flavylium chromophores as species markers for dragon’s blood resins from Dracaena and Daemonorops trees. J. Chromatogr. A. 2008, 1209, 153–161. [Google Scholar] [CrossRef]
- Cai, X.T.; Xu, Z.F. Studies on the plant origin of Chinese dragon’s blood. Acta Bot. Yunnan. 1979, 1, 1–9. [Google Scholar]
- Guang dong Institute of Botany, Flora Hainanica; Science Press: Beijing, China, 1977; p. 155.
- He, L.; Wang, Z.H.; Tu, P.F.; Hou, H. Advances in study on chemical constituents and pharmacological activities in plants of Dracaena Vand. ex L. Chin. Tradit. Herb Drugs 2004, 35, 221–228. [Google Scholar]
- Liu, J.; Dai, H.F.; Wu, J.; Zeng, Y.B.; Mei, W.L. Flavanes from Dracaena cambodiana. Z. Naturforsch. 2008, 63b, 1407–1410. [Google Scholar]
- Liu, J.; Mei, W.L.; Wu, J.; Zhao, Y.X.; Peng, M.; Dai, H.F. A new cytotoxic homoisoflavonoid from Dracaena cambodiana. J. Asian Nat. Prod. Res. 2009, 11, 192–195. [Google Scholar] [CrossRef]
- Liu, J.; Mei, W.L.; Dai, H.F. Study on bioactive chemical constituents from Dracaena cambodiana. In the first symposium on Li folk in Hainan province, Haikou, China, 2008.
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Šulc, M.; Lachman, J.; Hamouz, K.; Orsák, M.; Dvořák, P.; Horáčková, V. Selection and evaluation of methods for determination of antioxidant activity of purple- and red-fleshed potato varieties. Chem. Listy 2007, 101, 584–591. [Google Scholar]
- Stief, T.W. The physiology and pharmacology of single oxygen. Med. Hypotheses 2003, 60, 567–572. [Google Scholar] [CrossRef]
- Li, H.Y.; Hao, Z.B.; Wang, X.L. Antioxidant activities of extracts and fractions from Lysimachia foenum-graecum Hance. Bioresource Technol. 2009, 100, 970–974. [Google Scholar] [CrossRef]
- Negro, C.; Tommasi, L.; Miceli, A. Phenolic compounds and antioxidant activity from red grape marc extracts. Bioresource Technol. 2003, 87, 41–44. [Google Scholar] [CrossRef]
- Choi, J.S.; Chung, H.Y.; Kang, S.S.; Jung, M.J.; Kim, J.W.; No, J.K.; et al. The structure-activity relationship of flavonoids as scavengers of peroxynitrite. Phytother. Res. 2002, 16, 232–235. [Google Scholar] [CrossRef]
- Han, J.; Weng, X.; Bi, K. Antioxidants from a Chinese medicinal herb-Lithospermumerythrorhizon. Food Chem. 2008, 106, 2–10. [Google Scholar] [CrossRef]
- Zhang, Z.; Liao, L.; Moore, J.; Wu, T.; Wang, Z. Antioxidant phenolic compounds from walnut kernels (Juglans regia L.). Food Chem. 2009, 113, 160–165. [Google Scholar] [CrossRef]
- Lu, Y.; Yeap Foo, L. Antioxidant and radical scavenging capacities of polyphenols from apple pomace. Food Chem. 2000, 68, 81–85. [Google Scholar] [CrossRef]
- Fang, Z.; Zhang, Y.; Lü, Y.; Ma, G.; Chen, J.; Liu, D.; Ye, X. Phenolic compounds and antioxidant capacities of bayberry juices. Food Chem. 2009, 113, 884–888. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Sakanaka, S.; Ishihara, Y. Comparison of antioxidant properties of persimmon vinegar and some other commercialvinegars in radicalscavenging assays and on lipid oxidation in tuna homogenates. Food Chem. 2008, 107, 739–744. [Google Scholar] [CrossRef]
- Chang, S.T.; Wu, J.H.; Wang, S.Y.; Kang, P.L.; Yang, N.S.; Shyur, L.F. Antioxidant activity of extracts fromAcaciaconfusa bark and heartwood. J. Agric. Food Chem. 2001b, 49, 3420–3424. [Google Scholar]
- Jia, Z.S.; Tang, M.C.; Wu, J.M. The determination of flavonoid content in mulberry and their scavengingeffectsonsuper-oxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Luo, Y.; Wang, H.; Xu, X.; Mei, W.; Dai, H. Antioxidant Phenolic Compounds of Dracaena Cambodiana. Molecules 2010, 15, 8904-8914. https://doi.org/10.3390/molecules15128904
Luo Y, Wang H, Xu X, Mei W, Dai H. Antioxidant Phenolic Compounds of Dracaena Cambodiana. Molecules. 2010; 15(12):8904-8914. https://doi.org/10.3390/molecules15128904
Chicago/Turabian StyleLuo, Ying, Hui Wang, Xuerong Xu, Wenli Mei, and Haofu Dai. 2010. "Antioxidant Phenolic Compounds of Dracaena Cambodiana" Molecules 15, no. 12: 8904-8914. https://doi.org/10.3390/molecules15128904
APA StyleLuo, Y., Wang, H., Xu, X., Mei, W., & Dai, H. (2010). Antioxidant Phenolic Compounds of Dracaena Cambodiana. Molecules, 15(12), 8904-8914. https://doi.org/10.3390/molecules15128904




