Assessment of the Content of Phenolics and Antioxidant Action of Inflorescences and Leaves of Selected Species from the Genus Sorbus Sensu Stricto
Abstract
:1. Introduction
| Sample No. | Plant sources | Antioxidant activity b | |||
|---|---|---|---|---|---|
| Scientific name | Plant part tested a | DPPH EC50 | LA peroxidation (% inhibition) e | ||
| (µg/mL) c | TEAA (mmol/g) d | ||||
| 1. | Sorbus aucuparia L. | I | 16.69 ± 0.26 A, B | 0.78 | 68.34 ± 0.59 A |
| 2. | Sorbus aucuparia L. | L | 24.10 ± 0.29 C | 0.54 | 58.69 ± 0.46 B, C |
| 3. | Sorbus pohuashanensis (Hance) Hedl. | I | 17.89 ± 0.55 B, D | 0.73 | 68.69 ± 1.24 A |
| 4. | Sorbus pohuashanensis (Hance) Hedl. | L | 43.86 ± 1.51 E | 0.30 | 50.21 ± 0.96 D |
| 5. | Sorbus scalaris Koehne | I | 27.65 ± 1.09 F | 0.47 | 55.23 ± 1.46 E |
| 6. | Sorbus scalaris Koehne | L | 57.86 ± 1.63 G | 0.23 | 41.70 ± 1.05 F |
| 7. | Sorbus prattii Koehne var. prattii | L | 24.26 ± 0.45 C | 0.54 | 57.51 ± 0.87 B |
| 8. | Sorbus americana Marsh. | L | 38.76 ± 0.84 H | 0.34 | 54.29 ± 0.85 E |
| 9. | Sorbus commixta Hedl. | I | 23.22 ± 0.33 C | 0.56 | 78.21 ± 1.61 G |
| 10. | Sorbus commixta Hedl. | L | 28.56 ± 0.64 F | 0.46 | 58.65 ± 1.28 B, C |
| 11. | Sorbus decora (Sarg.) C.K. Schneid. | I | 16.20 ± 0.27 A | 0.81 | 70.99 ± 1.39 H |
| 12. | Sorbus decora (Sarg.) C.K. Schneid. | L | 27.21 ± 0.72 F | 0.48 | 59.99 ± 0.97 C |
| 13. | Sorbus wilfordii Koehne | L | 15.23 ± 0.54 A | 0.86 | 86.92 ± 1.15 I |
| 14. | Sorbus sambucifolia (Cham. & Schltdl.) M. Roem. | I | 28.03 ± 0.80 F | 0.47 | 58.12 ± 1.50 B, C |
| 15. | Sorbus sambucifolia (Cham. & Schltdl.) M. Roem. | L | 52.63 ± 1.25 I | 0.25 | 54.03 ± 1.05 E |
| 16. | Sorbus gracilis (Sieb. & Zucc.) K. Koch | I | 19.09 ± 0.69 D, J | 0.68 | 73.01 ± 1.09 J |
| 17. | Sorbus gracilis (Sieb. & Zucc.) K. Koch | L | 20.71 ± 0.66 J | 0.63 | 70.72 ± 1.00 H |
| 18. | Sorbus sitchensis M. Roem. | I | 20.75 ± 0.40 J | 0.63 | 68.26 ± 0.78 A |
| 19. | Sorbus sitchensis M. Roem. | L | 54.23 ± 1.08 K | 0.24 | 53.13 ± 0.86 E |
| 20. | Sorbus cashmiriana Hedl. | L | 48.59 ± 1.05 L | 0.27 | 53.59 ± 0.78 E |
| 21. | Sorbus koehneana C.K. Schneid. | I | 16.20 ± 0.52 A | 0.81 | 73.34 ± 1.07 J |
| 22. | Sorbus koehneana C.K. Schneid. | L | 24.74 ± 0.34 C | 0.53 | 54.15 ± 0.94 E |
| 23. | Sorbus pogonopetala Koehne | L | 19.87 ± 0.48 J | 0.66 | 74.73 ± 1.13 J |
| 24. | Sorbus setschwanensis (C.K. Schneid.) Koehne | L | 23.30 ± 0.56 C | 0.56 | 63.77 ± 0.70 K |
| Quercetin | 1.44 ± 0.05 | 88.38 ± 1.15 | |||
| Trolox | 3.27 ± 0.10 | 92.74 ± 1.10 | |||
2. Results and Discussion
2.1. Phenolic profile of the analysed Sorbus tissues
| Sample | No. | Total phenols (GAE, %) b | Caffeoylquinic acids c (%) | Proanthocyanidins (CyE, %) d | |
|---|---|---|---|---|---|
| NChA | ChA | ||||
| CL1: | 1. | 10.02 ± 0.22 A | 0.74 ± 0.01 A | 2.27 ± 0.01 A | 5.94 ± 0.12 A |
| 3. | 11.32 ± 0.03 B | 0.70 ± 0.01 B | 2.48 ± 0.02 B, C | 7.67 ± 0.05 B | |
| 17. | 10.72 ± 0.16 C | 0.03 ± 0.01 C | 0.93 ± 0.01 D, E | 6.56 ± 0.07 C | |
| 18. | 10.08 ± 0.11 A, D | 0.45 ± 0.02 D | 3.13 ± 0.06 F | 7.14 ± 0.11 D | |
| 11. | 11.67 ± 0.16 E | 1.26 ± 0.02 E | 3.85 ± 0.06 G | 6.40 ± 0.12 C | |
| 21. | 11.67 ± 0.05 E | 1.98 ± 0.01 F | 2.05 ± 0.02 H | 6.86 ± 0.12 E | |
| 16. | 11.06 ± 0.16 F | 0.19 ± 0.01 G | 3.31 ± 0.03 I | 6.54 ± 0.16 C | |
| 23. | 10.90 ± 0.26 C, F | 0.22 ± 0.01 H | 1.63 ± 0.02 J | 5.89 ± 0.04 A | |
| 9. | 9.29 ± 0.14 G | 0.76 ± 0.01 A | 3.92 ± 0.02 G | 5.98 ± 0.21 A | |
| 13. | 12.31 ± 0.32 H | 0.13 ± 0.01 I | 2.58 ± 0.02 B | 5.31 ± 0.04 F | |
| CL2: | 2. | 8.23 ± 0.06 I, J | 0.51 ± 0.01 J | 1.90 ± 0.08 K | 3.59 ± 0.09 G, H |
| 7. | 8.44 ± 0.08 J | 1.10 ± 0.04 K | 2.81 ± 0.09 L | 2.90 ± 0.06 I | |
| 10. | 8.08 ± 0.25 I | 0.05 ± 0.01 C | 0.79 ± 0.01 E, M | 3.58 ± 0.09 G, H | |
| 12. | 8.10 ± 0.09 I | 0.19 ± 0.01 G | 2.10 ± 0.01 H | 4.03 ± 0.09 J | |
| 14. | 8.22 ± 0.40 I, J | 0.42 ± 0.01 L | 4.17 ± 0.12 N | 3.79 ± 0.16 H, K | |
| 5. | 8.47 ± 0.21 J | 0.60 ± 0.01 M | 2.36 ± 0.02 A, C | 5.68 ± 0.10 L, M | |
| 22. | 9.87 ± 0.11 A | 0.53 ± 0.01 J | 1.97 ± 0.02 H, K | 5.81 ± 0.09 A, M | |
| 24. | 10.18 ± 0.17 D | 0.22 ± 0.01 H | 2.61 ± 0.02 B | 5.56 ± 0.08 L | |
| CL3: | 4. | 6.26 ± 0.19 K | 0.12 ± 0.01 I, O | 0.67 ± 0.01 M, O | 3.93 ± 0.03 J, K |
| 20. | 5.78 ± 0.12 L | 0.37 ± 0.01 N | 1.25 ± 0.01 P | 4.02 ± 0.05 J | |
| 8. | 6.47 ± 0.09 K | 0.04 ± 0.01 C | 1.85 ± 0.01 K | 3.66 ± 0.09 G | |
| 6. | 4.23 ± 0.15 M | 0.36 ± 0.01 N | 1.24 ± 0.01 P | 1.47 ± 0.04 N | |
| 15. | 5.07 ± 0.02 N | 0.10 ± 0.02 O | 1.02 ± 0.01 D | 1.96 ± 0.09 O | |
| 19. | 4.89 ± 0.07 N | 0.05 ± 0.01 C | 0.56 ± 0.01 O | 1.48 ± 0.05 N | |
| Sample | No. | Flavonoid aglycones d (%) | ||
|---|---|---|---|---|
| QU | KA | SX | ||
| CL1: | 1. | 1.048 ± 0.034 A | 0.084 ± 0.002 A | 0.190 ± 0.002 A |
| 3. | 0.400 ± 0.014 B | 0.039 ± 0.001 B | 0.017 ± 0.001 B | |
| 17. | 0.113 ± 0.001 C | 0.008 ± 0.001 C | nd | |
| 18. | 0.384 ± 0.028 B, K | 0.021 ± 0.001 D | 0.048 ± 0.001 C | |
| 11. | 0.839 ± 0.008 D, E | 0.059 ± 0.001 E, F | 0.070 ± 0.002 D | |
| 21. | 0.266 ± 0.014 F | 0.024 ± 0.003 D | 0.050 ± 0.005 C | |
| 16. | 0.194 ± 0.006 G, H | 0.012 ± 0.001 C | 0.072 ± 0.003 D | |
| 23. | 0.382 ± 0.023 B, K | 0.263 ± 0.006 G | nd | |
| 9. | 0.422 ± 0.008 B, J | 0.050 ± 0.003 F | 0.045 ± 0.003 C | |
| 13. | 0.878 ± 0.015 E, I | 0.046 ± 0.001 B, F | nd | |
| CL2: | 2. | 0.903 ± 0.003 I | 0.157 ± 0.005 H | nd |
| 7. | 0.790 ± 0.020 D | 0.117 ± 0.001 I, J | nd | |
| 10. | 0.470 ± 0.004 J | 0.011 ± 0.001 C | nd | |
| 12. | 0.474 ± 0.012 J | 0.035 ± 0.001 B | nd | |
| 14. | 0.805 ± 0.017 D | 0.058 ± 0.001 E, F | 0.127 ± 0.003 E | |
| 5. | 0.341 ± 0.005 K | 0.062 ± 0.001 E | 0.145 ± 0.002 F | |
| 22. | 0.247 ± 0.007 F, G | 0.106 ± 0.002 K | nd | |
| 24. | 0.566 ± 0.031 L | 0.306 ± 0.007 L | nd | |
| CL3: | 4. | 0.123 ± 0.004 C, H | 0.030 ± 0.001 B, D | nd |
| 20. | 0.525 ± 0.006 L | 0.113 ± 0.002 J, K | nd | |
| 8. | 0.460 ± 0.019 J | 0.039 ± 0.001 B | nd | |
| 6. | 0.217 ± 0.009 G | 0.125 ± 0.009 I | nd | |
| 15. | 0.163 ± 0.006 C, H | 0.011 ± 0.001 C | nd | |
| 19. | 0.270 ± 0.012 F | 0.023 ± 0.001 D | nd | |
2.2. Antioxidant activity of the extracts from Sorbus tissues
| r (R2) | DPPH EC50 (µg/mL) c | LA peroxidation (% inhibition) c |
|---|---|---|
| TPC (GAE, %) a | -0.9535 (0.9092)* | 0.8664 (0.7506)* |
| TPh (%)b | -0.8525 (0.7267)* | 0.7377 (0.5442)* |
| Proanthocyanidins (CyE, %) | -0.8007 (0.6411)* | 0.7456 (0.5559)* |
| Caffeoylquinic acids (ChA+NChA, %) | -0.6238 (0.3891)** | 0.4998 (0.2498)*** |
| Flavonoid aglycones (%) | -0.4248 (0.1804)*** | 0.2297 (0.0528) |
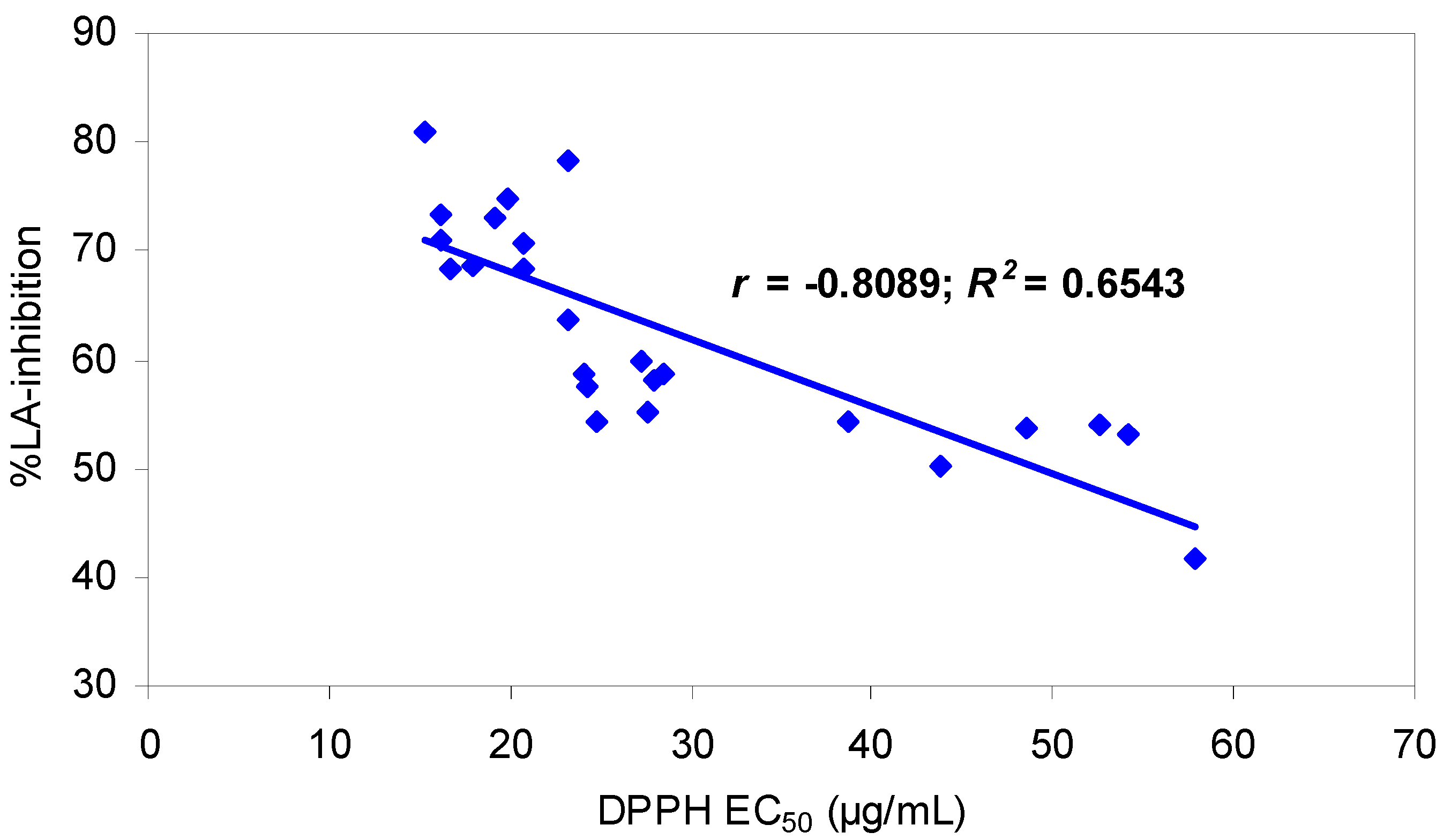
2.3. Relationships among the estimates of antioxidant capacities obtained from the DPPH and LA-peroxidation assays, and phenolic contents
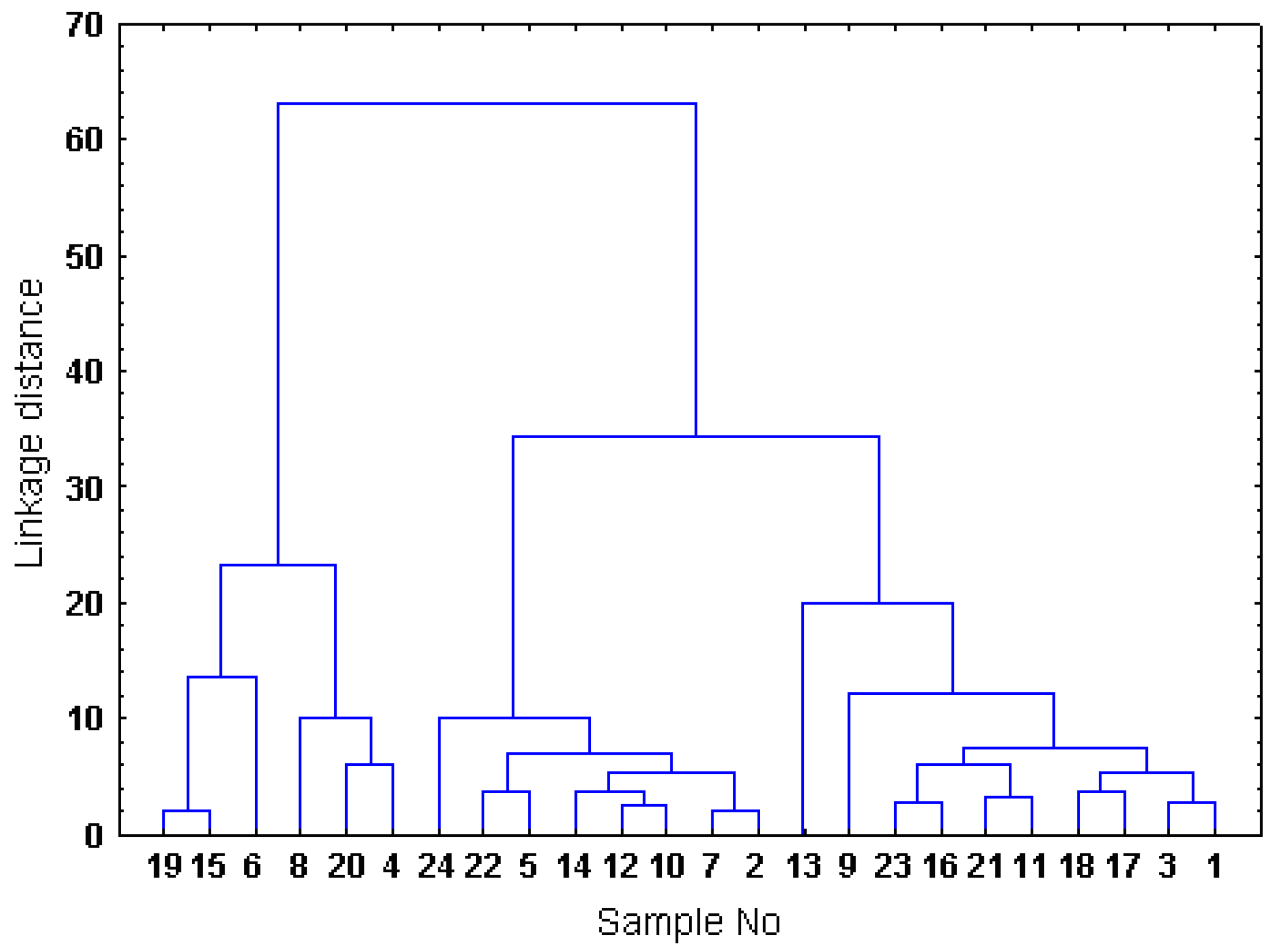
2.4. Hierarchical cluster analysis of the Sorbus samples
3. Experimental
3.1. Plant material
| Scientific name | Voucher specimen no. | Scientific name | Voucher specimen no. |
|---|---|---|---|
| Sorbus aucuparia L. | KFG/HB/07001-SAUC | Sorbus sambucifolia (Cham. & Schltdl.) M. Roem. | KFG/HB/08008-SSAM |
| Sorbus pohuashanensis (Hance) Hedl. | KFG/HB/07002-SPOH | ||
| Sorbus scalaris Koehne | KFG/HB/08005-SSCA | Sorbus gracilis (Sieb. & Zucc.) K. Koch | KFG/HB/07006-SGRA |
| Sorbus prattii Koehne var. prattii | KFG/HB/08006-SPRA | Sorbus sitchensis M. Roem. | KFG/HB/08009-SSIT |
| Sorbus americana Marsh. | KFG/HB/07003-SAME | Sorbus cashmiriana Hedl. | KFG/HB/07007-SCAS |
| Sorbus commixta Hedl. | KFG/HB/07004-SCOM | Sorbus koehneana C.K. Schneid. | KFG/HB/07008-SKOE |
| Sorbus decora (Sarg.) C.K. Schneid. | KFG/HB/08007-SDEC | Sorbus pogonopetala Koehne | KFG/HB/07009-SPOG |
| Sorbus wilfordii Koehne | KFG/HB/07005-SWIL | Sorbus setschwanensis (C.K. Schneid.) Koehne | KFG/HB/08010-SSET |
3.2. Chemicals and instrumentation
3.3. Preparation of plant extracts for testing antioxidant activity and phenolic profile
3.4. DPPH free radical-scavenging test
3.5. Linoleic acid (LA) peroxidation test
3.6. Determination of total phenolic content
3.7. Determination of total proanthocyanidin content
3.8. Total flavonoid content
3.9. The content of chlorogenic acid isomers
3.10. Statistical analysis
4. Conclusions
Acknowledgements
- Sample Availability: Plant samples are available from the authors.
References and Notes
- McAllister, H. The Genus Sorbus: Mountain Ash and Other Rowans; Royal Botanic Gardens, Kew: Surrey, UK, 2005. [Google Scholar]
- Hukkanen, A.T.; Pölönen, S.S.; Kärenlampi, S.O.; Kokko, H.I. Antioxidant capacity and phenolic content of sweet rowanberries. J. Agr. Food Chem. 2006, 54, 112–119. [Google Scholar] [CrossRef]
- Olszewska, M.A.; Michel, P. Antioxidant activity of inflorescences, leaves and fruits of three Sorbus species in relation to their polyphenolic composition. Nat. Prod. Res. 2009, 23, 1507–1521. [Google Scholar] [CrossRef]
- McCune, L.M.; Johns, T. Antioxidant activity in medicinal plants associated with the symptoms of diabetes mellitus used by the Indigenous Peoples of the North American boreal forest. J. Ethnopharmacol. 2002, 82, 197–205. [Google Scholar] [CrossRef]
- Bae, J.T.; Sim, G.S.; Kim, J.H.; Pyo, H.B.; Yun, J.W.; Lee, B.C. Antioxidative activity of the hydrolytic enzyme treated Sorbus commixta Hedl. and its inhibitory effect on matrix metalloproteinase-1 in UV irradiated human dermal fibroblasts. Arch. Pharm. Res. 2007, 30, 1116–1123. [Google Scholar] [CrossRef]
- Sohn, E.J.; Kang, D.G.; Mun, Y.J.; Woo, W.H.; Lee, H.S. Anti-atherogenic effects of the methanol extract of Sorbus cortex in atherogenic-diet rats. Biol. Pharm. Bull. 2005, 28, 1444–1449. [Google Scholar] [CrossRef]
- Kang, D.G.; Sohn, E.J.; Lee, A.S.; Kim, J.S.; Lee, D.H.; Lee, H.S. Methanol extract of Sorbus commixta cortex prevents vascular inflammation in rats with a high fructose-induced metabolic syndrome. Am. J. Chin. Med. 2007, 35, 265–277. [Google Scholar] [CrossRef]
- Kang, D.G.; Lee, J.K.; Choi, D.H.; Sohn, E.J.; Moon, M.K.; Lee, H.S. Vascular relaxation by the methanol extract of Sorbus cortex via NO-cGMP pathway. Biol. Pharm. Bull. 2005, 28, 860–864. [Google Scholar] [CrossRef]
- Termentzi, A.; Kefalas, P.; Kokkalou, E. Antioxidant activities of various extracts and fractions of Sorbus domestica fruit at different maturity stages. Food Chem. 2006, 98, 599–608. [Google Scholar] [CrossRef]
- Olszewska, M.A.; Michel, P. Activity-guided isolation and identification of free radical-scavenging components from various leaf extracts of Sorbus aria (Ehrh.) Pers. Nat. Prod. Res. 2011, in press. [Google Scholar]
- Ndhlala, A.R.; Moyo, M.; Van Staden, J. Natural antioxidants: fascinating or mythical biomolecules? Molecules 2010, 15, 6905–6930. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationship of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Cai, Y.; Luo, Q.; Sun, M.; Corke, H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 2004, 74, 2157–2184. [Google Scholar] [CrossRef]
- Surveswaran, S.; Cai, Y.; Corke, H.; Sun, M. Systematic evaluation of natural phenolic antioxidants from 133 Indian medicinal plants. Food Chem. 2007, 102, 938–953. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agr. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Sultana, B.; Anwar, F.; Przybylski, R. Antioxidant activity of phenolic components present in barks of Azadirachta indica, Terminalia arjuna, Acacia nilotica, and Eugenia jambolana Lam. Trees. Food Chem. 2007, 104, 1106–1114. [Google Scholar] [CrossRef]
- Yokozawa, T.; Chen, C.P.; Dong, E.; Tanaka, T.; Nonaka, G.I.; Nishioka, I. Study on the inhibitory effect of tannins and flavonoids against the 1,1-diphenyl-2-picrylhydrazyl radical. Biochem. Pharmacol. 1998, 56, 231–222. [Google Scholar]
- Zimmerman, H.J. Hepatotoxicity: the Adverse Effects of Drugs and Other Chemicals on the Liver; Lippincott, Williams & Wilkins: Philadelphia, PA, USA, 1999; pp. 274–275. [Google Scholar]
- Aron, P.M.; Kennedy, J.A. Flavan-3-ols: Nature, occurrence and biological activity. Mol. Nutr. Food Res. 2008, 52, 79–104. [Google Scholar] [CrossRef]
- Galati, G.; O’Brien, P.J. Potential toxicity of flavonoids and other dietary phenolics: significance for their chemopreventive and anticancer properties. Free Radic. Biol. Med. 2004, 37, 287–303. [Google Scholar] [CrossRef]
- Watanabe, T.; Arai, Y.; Mitsui, Y.; Kusaura, T.; Okawa, W.; Kajihara, Y.; Saito, I. The blood pressure-lowering effect and safety of chlorogenic acid from green coffee bean extract in essential hypertension. Clin. Exp. Hypertens. 2006, 28, 439–449. [Google Scholar] [CrossRef]
- Burda, S.; Oleszek, W. Antioxidant and antiradical activities of flavonoids. J. Agr. Food Chem. 2001, 49, 2774–2779. [Google Scholar] [CrossRef]
- Meyer, A.S.; Donovan, J.L.; Pearson, D.A.; Waterhouse, A.L.; Frankel, E.N. Fruit hydroxycinnamic acids inhibit human low-density lipoprotein oxidation in vitro. J. Agr. Food Chem. 1998, 46, 1783–1787. [Google Scholar] [CrossRef]
- Borneo, R.; Leon, A.E.; Aguirre, A.; Ribotta, P.; Cantero, J.J. Antioxidant capacity of medicinal plants from the Province of Cordoba (Argentina) and their in vitro testing in a model food system. Food Chem. 2009, 112, 664–670. [Google Scholar] [CrossRef]
- Scherer, R.; Godoy, H.T. Antioxidant activity index (AAI) by the 2,2-diphenyl-1-picrylhydrazyl method. Food Chem. 2009, 112, 654–658. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Lebensm. Wiss. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Azuma, K.; Nakayama, M.; Koshioka, M.; Ippoushi, K.; Yamaguchi, Y.; Kohata, K.; Yamauchi, Y.; Ito, H.; Higashio, H. Phenolic antioxidants from the leaves of Corchorus olitorius L. J. Agr. Food Chem. 1999, 47, 3963–3966. [Google Scholar] [CrossRef]
- Haraguchi, H.; Ishikawa, H.; Sanchez, Y.; Ogura, T.; Kubo, Y.; Kubo, I. Antioxidative constituents in Heterotheca inoloides. Bioorg. Med. Chem. 1997, 5, 865–871. [Google Scholar] [CrossRef]
- Chen, H.-M.; Muramoto, K.; Yamauchi, F.; Nokihara, K. Antioxidant activity of designed peptides based on the antioxidative peptide isolated from digests of a soybean protein. J. Agr. Food Chem. 1996, 44, 2619–2623. [Google Scholar] [CrossRef]
- Porter, L.J.; Hrstich, L.N.; Chana, B.G. The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochemistry 1986, 25, 223–230. [Google Scholar]
- Olszewska, M. Separation of quercetin, sexangularetin, kaempferol and isorhamnetin for simultaneous HPLC determination of flavonoid aglycones in inflorescences, leaves and fruits of three Sorbus species. J. Pharm. Biomed. Anal. 2008, 48, 629–635. [Google Scholar] [CrossRef]
- Nagels, L.; van Dongen, W.; de Brucker, J.; de Pooter, H. High-performance liquid chromatographic separation of naturally occuring esters of phenolic acids. J. Chromatogr. 1980, 187, 181–187. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Olszewska, M.A.; Nowak, S.; Michel, P.; Banaszczak, P.; Kicel, A. Assessment of the Content of Phenolics and Antioxidant Action of Inflorescences and Leaves of Selected Species from the Genus Sorbus Sensu Stricto. Molecules 2010, 15, 8769-8783. https://doi.org/10.3390/molecules15128769
Olszewska MA, Nowak S, Michel P, Banaszczak P, Kicel A. Assessment of the Content of Phenolics and Antioxidant Action of Inflorescences and Leaves of Selected Species from the Genus Sorbus Sensu Stricto. Molecules. 2010; 15(12):8769-8783. https://doi.org/10.3390/molecules15128769
Chicago/Turabian StyleOlszewska, Monika A., Sławomira Nowak, Piotr Michel, Piotr Banaszczak, and Agnieszka Kicel. 2010. "Assessment of the Content of Phenolics and Antioxidant Action of Inflorescences and Leaves of Selected Species from the Genus Sorbus Sensu Stricto" Molecules 15, no. 12: 8769-8783. https://doi.org/10.3390/molecules15128769
APA StyleOlszewska, M. A., Nowak, S., Michel, P., Banaszczak, P., & Kicel, A. (2010). Assessment of the Content of Phenolics and Antioxidant Action of Inflorescences and Leaves of Selected Species from the Genus Sorbus Sensu Stricto. Molecules, 15(12), 8769-8783. https://doi.org/10.3390/molecules15128769





