Synthesis, Structures and Properties of Cu(II) and Mn(II) Complexes with 1,10-Phenanthroline-2-carboxylic acid and 2,2’-Bipyridine Ligands
Abstract
:1. Introduction
2. Results and Discussion
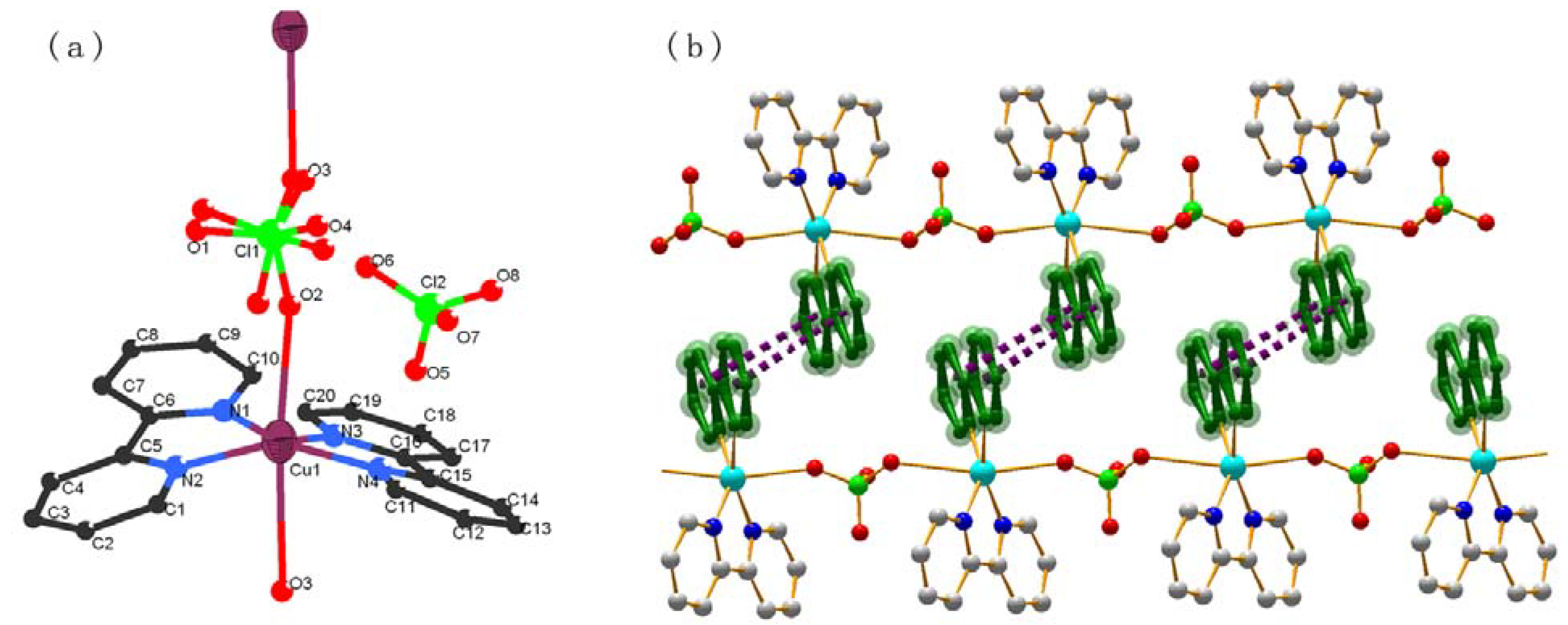
2.1. Crystal structures of compounds 1-4
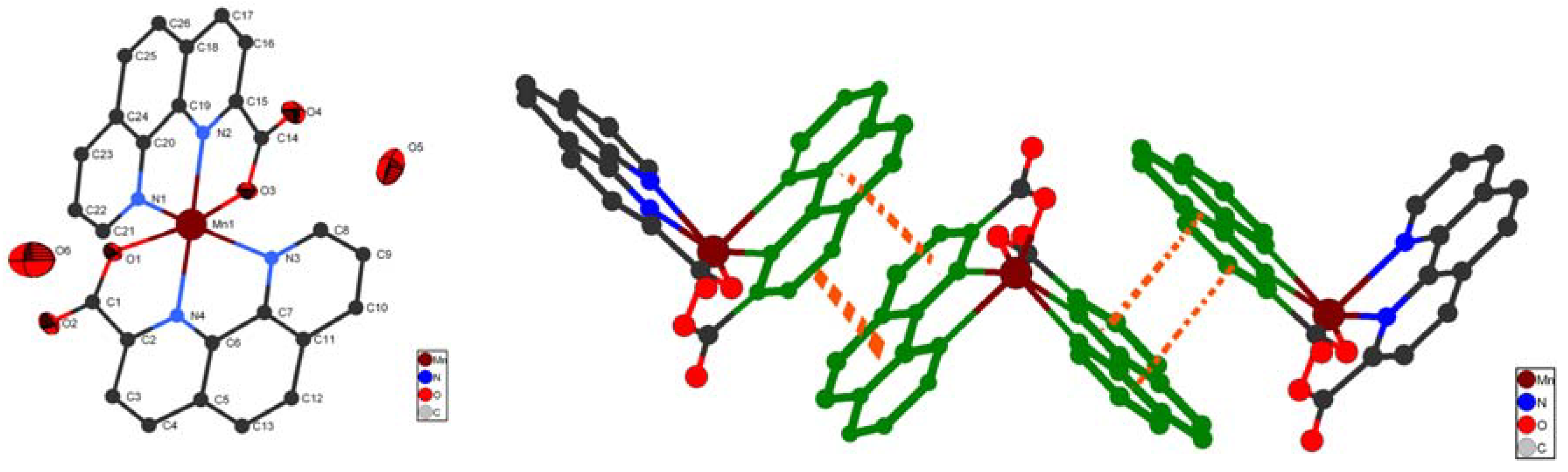
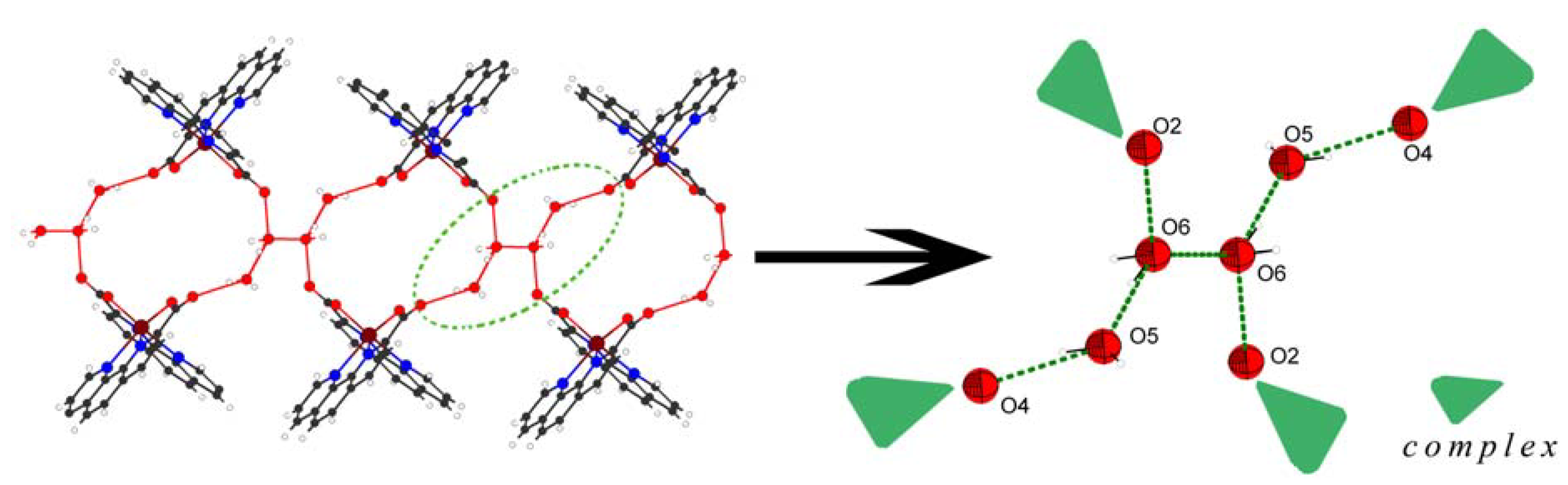
| Complexes | 1 | 2 | 3 | 4 | |
|---|---|---|---|---|---|
| Empirical formula | C26H18MnN4O6 | C60H72Cl4Cu4N12O28 | C22H20Cu2N6O12 | C20H16Cl2CuN4O8 | |
| Formula mass | 537.38 | 1801.22 | 687.52 | 574.81 | |
| Temperature(K) | 296(2) K | 296(2) K | 296(2) K | 296(2) K | |
| Crystal system | Triclinic | Triclinic | Monoclinic | Triclinic | |
| Space group | p-1 | p-1 | P2(1)/n | P-1 | |
| a(Å) | 10.0889(9) | 10.1983(18) | 7.7035(2) | 7.4088(19) | |
| b(Å) | 10.5636(9) | 14.267(2) | 10.2045(3) | 11.227(3) | |
| c(Å) | 12.1510(10) | 15.162(4) | 16.5347(5) | 14.877(4) | |
| α(°) | 71.456(3) | 114.261(12) | 90 | 110.164(8) | |
| β(°) | 68.256(3) | 92.692(11) | 100.132(2) | 96.489(9) | |
| γ(°) | 82.130(4) | 110.063(8) | 90 | 99.608(8) | |
| V(Å3) | 1140.13(17) | 1843.2(6) | 1279.53(6) | 1125.7(5) | |
| Z | 2 | 1 | 2 | 2 | |
| Dcalc(gcm-3) | 1.565 | 1.623 | 1.784 | 1.696 | |
| μ(mm−1) | 0.632 | 1.373 | 1.740 | 1.264 | |
| F[000] | 550 | 696 | 582 | ||
| θ(°) | 1.89 to 25.00 | 1.51 to 25.01 | 2.36 to 25.01 | 1.98 to 25.01 | |
| Data/restraints/parameters | 3943 /0 / 334 | 920 | 2259 / 0 / 190 | 3915 / 0 / 308 | |
| Goodness-of-fit on F2 | 1.029 | 1.000 | 1.096 | 1.080 | |
| Final Ra indices[I>2σ(I)] | R1 = 0.0624, wR2 = 0.1658 | R1 = 0.0627, wR2 = 0.1734 | R1 = 0.0267, wR2 = 0.0752 | R1 = 0.0777, wR2 = 0.2403 | |
| R(int) | 0.0457 | 0.0931 | 0.0266 | 0.0272 | |
| R indices (all data) | R1 = 0.1232, wR2 = 0.1963 | R1 = 0.1358, wR2 = 0.1979 | R1 = 0.0325, wR2 = 0.0786 | R1 = 0.0883, wR2 = 0.2552 | |
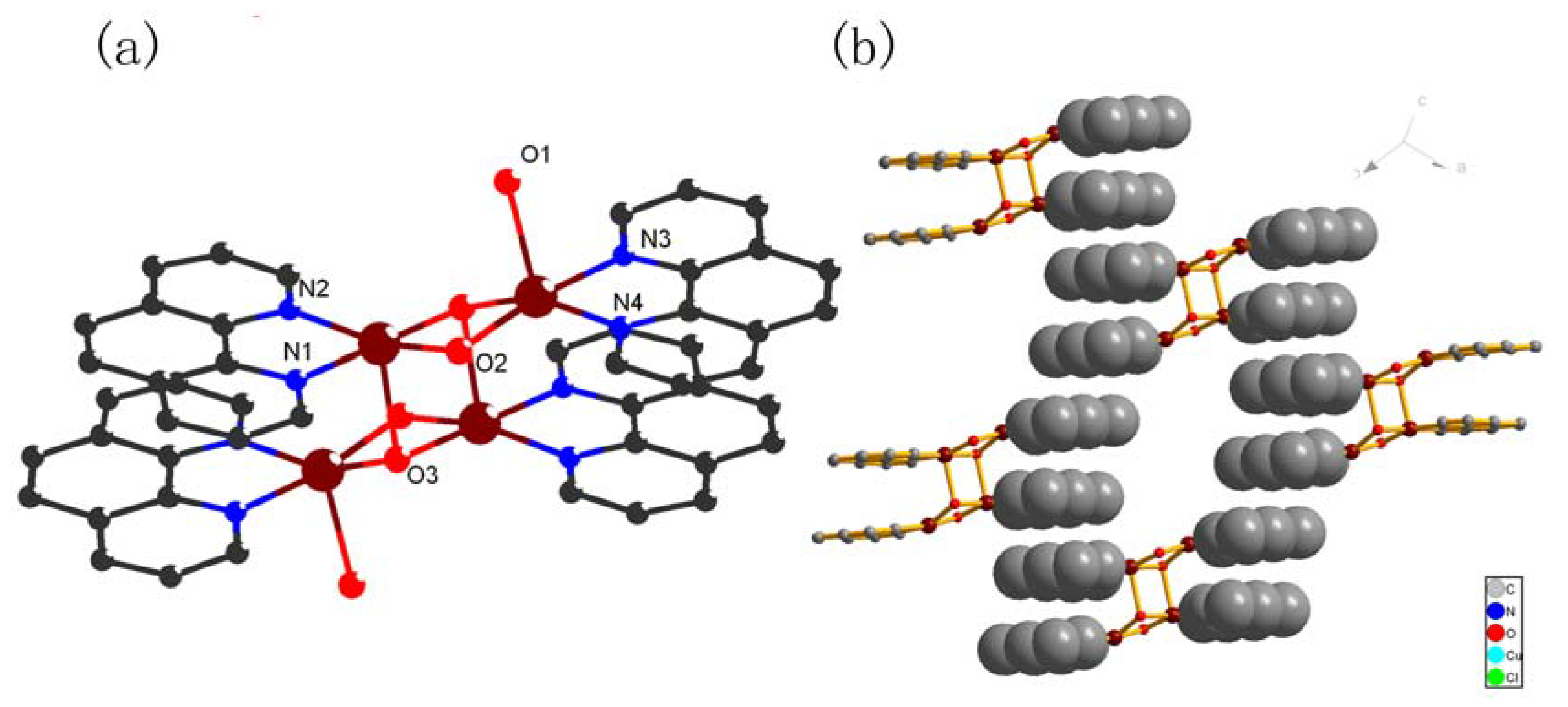

| Complex 1 | |||
|---|---|---|---|
| Mn(1)-O(3) | 2.156(4) | Mn(1)-N(2) | 2.177(4) |
| Mn(1)-O(1) | 2.160(4) | Mn(1)-N(4) | 2.183(4) |
| Mn(1)-N(3) | 2.370(5) | Mn(1)-N(1) | 2.440(4) |
| O(3)-Mn(1)-O(1) | 105.32(16) | O(3)-Mn(1)-N(2) | 73.80(15) |
| O(1)-Mn(1)-N(2) | 118.99(16) | O(3)-Mn(1)-N(4) | 127.70(15) |
| O(1)-Mn(1)-N(4) | 73.27(17) | N(2)-Mn(1)-N(4) | 153.89(17) |
| O(3)-Mn(1)-N(3) | 92.17(15) | O(1)-Mn(1)-N(3) | 143.85(16) |
| N(2)-Mn(1)-N(3) | 96.02(16) | N(4)-Mn(1)-N(3) | 71.02(16) |
| O(3)-Mn(1)-N(1) | 143.97(15) | O(1)-Mn(1)-N(1) | 92.31(15) |
| N(2)-Mn(1)-N(1) | 70.16(16) | N(4)-Mn(1)-N(1) | 87.16(16) |
| N(3)-Mn(1)-N(1) | 91.40(15) | ||
| Complex 2 | |||
| Cu(1)-O(2) | 1.919(4) | Cu(1)-O(3)#1 | 1.971(4) |
| Cu(1)-N(1) | 2.023(5) | Cu(1)-N(2) | 2.035(4) |
| Cu(1)-O(3) | 2.335(4) | Cu(1)-Cu(2) | 2.9347(9) |
| Cu(2)-O(2) | 1.937(4) | Cu(2)-O(3)#1 | 1.973(3) |
| Cu(2)-N(4) | 2.016(5) | Cu(2)-N(3) | 2.030(5) |
| Cu(2)-O(1) | 2.250(5) | O(3)-Cu(1)#1 | 1.971(4) |
| O(3)-Cu(2)#1 | 1.973(3) | O(2)-Cu(1)-O(3)#1 | 81.55(15) |
| O(2)-Cu(1)-N(1) | 96.60(17) | O(3)#1-Cu(1)-N(1) | 178.04(16) |
| O(2)-Cu(1)-N(2) | 164.89(18) | O(3)#1-Cu(1)-N(2) | 100.31(17) |
| N(1)-Cu(1)-N(2) | 81.30(18) | O(2)-Cu(1)-O(3) | 97.81(16) |
| O(3)#1-Cu(1)-O(3) | 86.15(15) | N(1)-Cu(1)-O(3) | 94.79(17) |
| N(2)-Cu(1)-O(3) | 97.28(15) | O(2)-Cu(1)-Cu(2) | 40.68(11) |
| O(2)-Cu(2)-O(3)#1 | 81.05(15) | O(2)-Cu(2)-N(4) | 95.43(19) |
| O(3)#1-Cu(2)-N(4) | 156.91(19) | O(2)-Cu(2)-N(3) | 171.42(19) |
| O(3)#1-Cu(2)-N(3) | 98.89(18) | N(4)-Cu(2)-N(3) | 81.2(2) |
| O(2)-Cu(2)-O(1) | 96.55(18) | O(3)#1-Cu(2)-O(1) | 101.24(17) |
| N(4)-Cu(2)-O(1) | 101.83(19) | N(3)-Cu(2)-O(1) | 91.88(19) |
| Symmetry transformations used to generate equivalent atoms: #1 -x+1,-y+1,-z+1 | |||
| Complex 3 | |||
| Cu(1)-O(1) | 2.0135(18) | N(1)-Cu(1)-N(4) | 97.27(10) |
| Cu(1)-N(3) | 2.018(2) | O(1)-Cu(1)-N(2) | 93.09(8) |
| Cu(1)-N(1) | 2.036(2) | N(3)-Cu(1)-N(2) | 98.90(9) |
| Cu(1)-N(4) | 2.041(2) | N(1)-Cu(1)-N(2) | 77.45(9) |
| Cu(1)-O(1) | 2.0135(18) | N(4)-Cu(1)-N(2) | 99.83(9) |
| Cu(1)-O(2)#1 | 2.325(2) | O(1)-Cu(1)-O(2)#1 | 76.85(7) |
| O(1)-Cu(1)-N(3) | 92.97(9) | N(3)-Cu(1)-O(2)#1 | 88.32(9) |
| O(1)-Cu(1)-N(1) | 90.37(8) | N(1)-Cu(1)-O(2)#1 | 95.82(9) |
| N(3)-Cu(1)-N(1) | 175.19(8) | N(4)-Cu(1)-O(2)#1 | 90.83(8) |
| O(1)-Cu(1)-N(4) | 166.14(9) | N(2)-Cu(1)-O(2)#1 | 167.98(8) |
| N(3)-Cu(1)-N(4) | 80.18(10) | ||
| Symmetry transformations used to generate equivalent atoms: #1 -x+1,-y+2,-z+1 | |||
| Complex 4 | |||
| Cu(1)-N(3) | 1.973(6) | N(1)-Cu(1)-N(4) | 103.5(2) |
| Cu(1)-N(1) | 1.984(5) | N(2)-Cu(1)-N(4) | 151.9(2) |
| Cu(1)-N(2) | 1.994(5) | N(3)-Cu(1)-N(2) | 102.2(2) |
| Cu(1)-N(4) | 1.995(6) | N(1)-Cu(1)-N(2) | 81.7(2) |
| N(3)-Cu(1)-N(1) | 160.9(2) | N(3)-Cu(1)-N(4) | 81.9(3) |
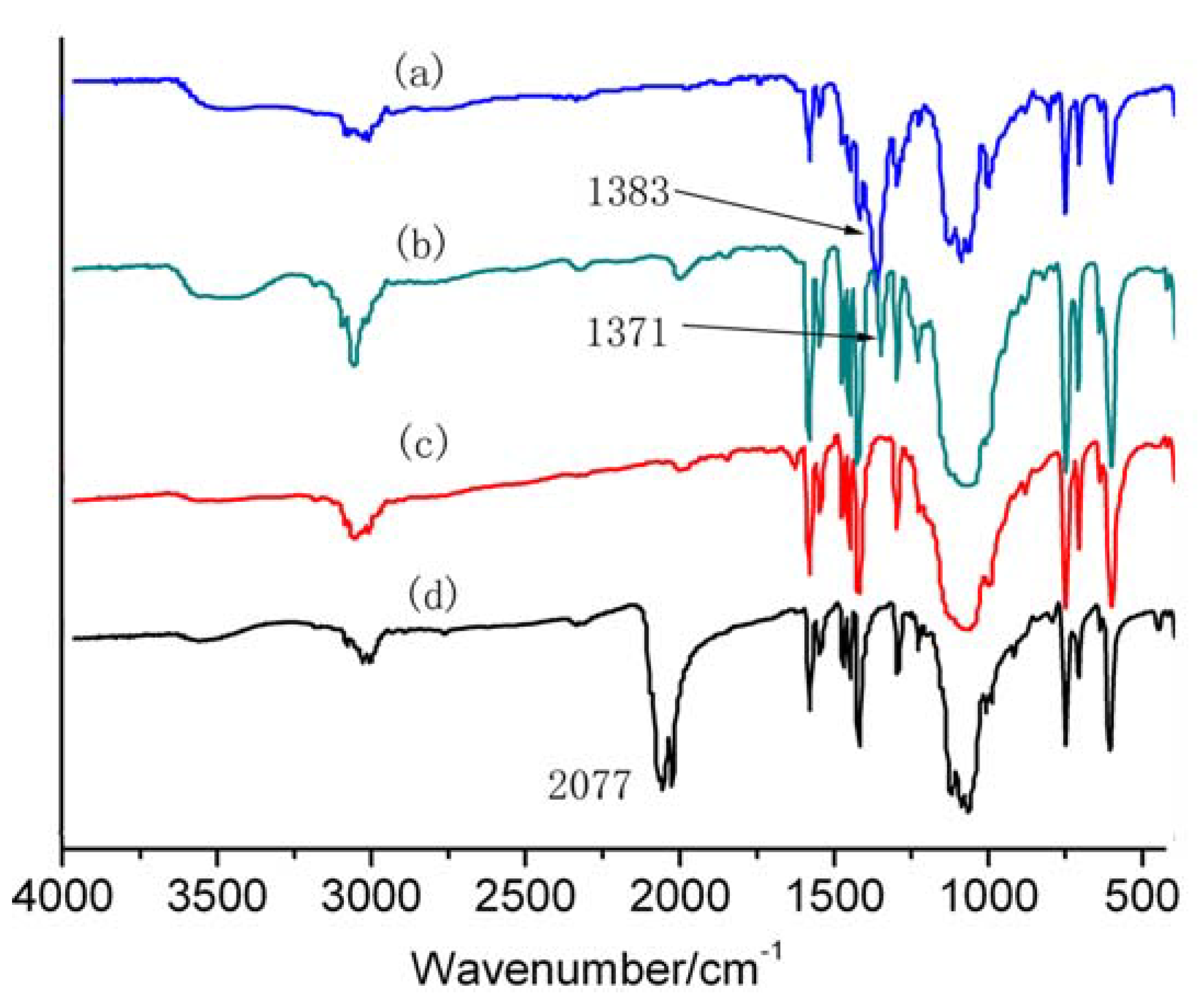
3. Experimental
3.1. Materials and Physical Measurements
3.2. Synthesis of 1-4
3.2.1. Synthesis of [Mn(phenca)2](H2O)2 (1)
3.2.2. Synthesis of [Cu4(phen)4(OH-)4(H2O)2](DMF)4(ClO4-)4(H2O) (2)
3.2.3. Synthesis of [Cu2(2,2-bipy)2(C2O42-)(H2O)2(NO3)2] (3)
3.2.4. Synthesis of [Cu (2,2-bipy)2(ClO4-)](ClO4-) (4)
3.3. Single-crystal Structure Determination
4. Conclusions
Supplementary Materials
Acknowledgements
- Samples Availability: Contact the authors.
References and Notes
- Chui, S.S.; Lo, S.M.F.; Charmant, J.P.H.; Orpen, A.G.; I.D., Williams. A Chemically Functionalizable Nanoporous Material [Cu3(TMA)2(H2O)3]n. Science 1999, 283, 1148–1150. [Google Scholar] [CrossRef]
- Leininger, S.; Olenyuk, B.; Stang, P.J. Self-Assembly of Discrete Cyclic Nanostructures Mediated by Transition Metals. Chem. Rev. 2000, 100, 853–908. [Google Scholar] [CrossRef]
- Biradha, K.; Zaworotko, M.J. A Supramolecular Analogue of Cyclohexane Sustained by Aromatic C−H···π Interactions: Complexes of 1,3,5-Trihydroxybenzene with Substituted Pyridines. J. Am. Chem. Soc. 1998, 120, 6431–6432. [Google Scholar] [CrossRef]
- Cockroft, S.L.; Hunter, C.A.; Lawson, K.R.; Perkins, J.; Urch, C.J. Electrostatic Control of Aromatic Stacking Interactions. J. Am. Chem. Soc. 2005, 127, 8594–8595. [Google Scholar]
- Cheruzel, L.E.; Mashuta, M.S.; Buchanan, R.M. A supramolecular assembly of side-by-side polyimidazole tripod coils stabilized by π–π stacking and unique boric acid templated hydrogen bonding interactions. Chem. Commun. 2005, 2223–2225. [Google Scholar]
- Chipot, C.; Jaffe, R.; Maigret, B.; Pearlman, D.A.; Kollman, P.A. Benzene Dimer: A Good Model for π−π Interactions in Proteins? A Comparison between the Benzene and the Toluene Dimers in the Gas Phase and in an Aqueous Solution. J. Am. Chem. Soc. 1996, 118, 11217–11224. [Google Scholar] [CrossRef]
- Pletneva, E.V.; Laederach, A.T.; Fulton, D.B.; Kostić, N.M. The Role of Cation−π Interactions in Biomolecular Association. Design of Peptides Favoring Interactions between Cationic and Aromatic Amino Acid Side Chains. J. Am. Chem. Soc. 2001, 123, 6232–6245. [Google Scholar] [CrossRef]
- Meyer, E.A.; Castellano, R.K.; Diederich, F. Interactions with Aromatic Rings in Chemical and Biological Recognition. Angew. Chem. Int. Ed. 2003, 42, 1210–1250. [Google Scholar]
- Alcock, N.W.; Barker, P.R.; Haider, J.M.; Hannon, M.J.; Painting, C.L.; Plummer, E.A.; Rissanen, K.; Saarenketo, P. Red and blue luminescent metallo-supramolecular coordination polymers assembled through π–π interactions. J. Chem. Soc. Dalton Trans. 2000, 1447–1462. [Google Scholar]
- Saenger, W. Principles of Nucleic Acid Structure; Springer-Verlag: NewYork, NY, USA, 1984; pp. 132–135. [Google Scholar]
- Nohra, B.; Yao, Y.; Lescop, C.; Réau, R. Coordination Polymers with π-Stacked Metalloparacyclophane Motifs: F-Shaped Mixed-Coordination Dinuclear Connectors. Angew. Chem. Int. Ed. 2007, 46, 8242–8245. [Google Scholar] [CrossRef]
- Sauvage, J.P.; Collin, J.P.; Chambron, J.C.; Guillerez, S.; Coudret, C. Ruthenium(II) and Osmium(II) Bis(terpyridine) Complexes in Covalently-Linked Multicomponent Systems: Synthesis, Electrochemical Behavior, Absorption Spectra, and Photochemical and Photophysical Properties. Chem.Rev. 1994, 94, 993–1019. [Google Scholar] [CrossRef]
- Egli, M.; Sarkhel, S. Lone Pair−Aromatic Interactions: To Stabilize or Not to Stabilize. Acc. Chem. Res. 2007, 40, 197–205. [Google Scholar] [CrossRef]
- Mooibroek, T.J.; Teat, S.J.; Massera, C.; Gamez, P.; Reedijk, J. Crystallographic and Theoretical Evidence of Acetonitrile−π Interactions with the Electron-Deficient 1,3,5-Triazine Ring. Cryst. Growth Des. 2006, 6, 1569–1574. [Google Scholar] [CrossRef]
- Chas, M.; Blanco, V.; Peinador, C.; Quintela, J.M. Synthesis of [3]Catenanes Based on Metal-Directed Self-Assembly and π-Donor/π-Acceptor Interactions. Org. Lett. 2007, 9, 675–678. [Google Scholar] [CrossRef]
- Munakata, M.; Wu, L.P.; Kuroda-Sowa, T.; Maekawa, M.; Suenaga, Y.; Ning, G.L.; Kojima, T. Supramolecular Silver(I) Complexes with Highly Strained Polycyclic Aromatic Compounds. J. Am. Chem. Soc. 1998, 120, 8610–8618. [Google Scholar]
- Paul, R.L.; Couchman, S.M.; Jeffery, J.C.; McCleverty, J.A.; Reeves, Z.R.; Ward, M.D. Effects of metal co-ordination geometry on self-assembly: A dinuclear double helicate complex and a tetranuclear cage complex of a new bis-bidentate bridging ligand. J. Chem. Soc. Dalton Trans. 2000, 845–851. [Google Scholar]
- Liu, Y.Y.; Ma, J.F.; Yang, J.; Ma, J.C.; Su, Z.M. Versatile frameworks constructed from divalent metals and 1,2,3,4-butanetetracarboxylate anion: Syntheses, crystal structures, luminescence and magnetic properties. Cryst. Eng. Comm. 2008, 10, 894–904. [Google Scholar]
- Bondar, O.A.; Lukashuk, L.V.; Lysenko, A.B.; Krautscheid, H.; Rusanov, E.B.; Chernega, A.N.; Domasevitch, K.V. New microporous copper(II) coordination polymers based upon bifunctional 1,2,4-triazole/tetrazolate bridges . Cryst. Eng. Comm. 2008, 10, 1216–1226. [Google Scholar]
- Gregory, J.K.; Clary, D.C.; Liu, K.; Brown, M.G.; Saykally, R.J. The Water Dipole Moment in Water Clusters. Science 1997, 275, 814–817. [Google Scholar] [CrossRef]
- Su, Z.; Bai, Z.S.; Xu, J.; Okamura, T.; Liu, G.X.; Chu, Q.; Wang, X.F.; Sun, W.Y.; Ueyama, N. Synthesis, structure and property of cobalt(II) complexes with 3,5-di(1H-imidazol-1-yl)benzoic acid. Cryst. Eng. Comm. 2009, 11, 873–880. [Google Scholar]
- Sun, J.; Tong, X.; Xu, H. Synthesis, structures and properties of Cu and Cd complexes with 1,10-phenanthroline. Inorg. Chem. Commun. 2010, 13, 645–648. [Google Scholar] [CrossRef]
- Sun, J.; Tong, X. Aqua(6,6'-oxydipicolinato-κ2O,N,N',O')copper(II). Acta Cryst. 2010, E66, m70. [Google Scholar]
- Siemens, SAINT: Area Detector Control and Integration Software.; Siemens Analytical X-ray Instruments Inc.: Madison, WI, USA, 1996.
- Sheldrick, G.M. SHELXL97 and SHELXTL Software Reference Manual, 5.1; Bruker AXS Inc.: Madison, WI, USA, 1997. [Google Scholar]
- Crystallographic data (excluding structure factors) for the structures in this paper have been deposited with the Cambridge Crystallographic Date Centre as supplementary publications CCDC 776482, 776483, 770153 and 770155 for 1-4, respectively. Copies of the data can be obtained free of charge on application to CCDC, 12 Union Road, Cambridge CB2 1EZ, UK [Fax: 441223-336-033; Email: deposit@ccdc.cam.ac.uk.].
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sun, J.; Xu, H. Synthesis, Structures and Properties of Cu(II) and Mn(II) Complexes with 1,10-Phenanthroline-2-carboxylic acid and 2,2’-Bipyridine Ligands. Molecules 2010, 15, 8349-8359. https://doi.org/10.3390/molecules15118349
Sun J, Xu H. Synthesis, Structures and Properties of Cu(II) and Mn(II) Complexes with 1,10-Phenanthroline-2-carboxylic acid and 2,2’-Bipyridine Ligands. Molecules. 2010; 15(11):8349-8359. https://doi.org/10.3390/molecules15118349
Chicago/Turabian StyleSun, Jingya, and Huanzhi Xu. 2010. "Synthesis, Structures and Properties of Cu(II) and Mn(II) Complexes with 1,10-Phenanthroline-2-carboxylic acid and 2,2’-Bipyridine Ligands" Molecules 15, no. 11: 8349-8359. https://doi.org/10.3390/molecules15118349
APA StyleSun, J., & Xu, H. (2010). Synthesis, Structures and Properties of Cu(II) and Mn(II) Complexes with 1,10-Phenanthroline-2-carboxylic acid and 2,2’-Bipyridine Ligands. Molecules, 15(11), 8349-8359. https://doi.org/10.3390/molecules15118349




