Preparation and Characterization of Novel Gellan Gum Hydrogels Suitable for Modified Drug Release
Abstract
:1. Introduction
2. Results and Discussion
- - physical gel of Gellan (cooling of hot solutions)
- - physical gel of Gellan in the presence of different amounts of Lys (cooling of hot solutions)
- - chemical gel of Gellan in the presence of different amounts of Lys and EDC/NHS
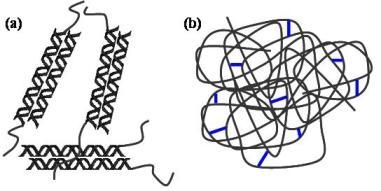
2.1. Structural differences between physical and chemical hydrogels
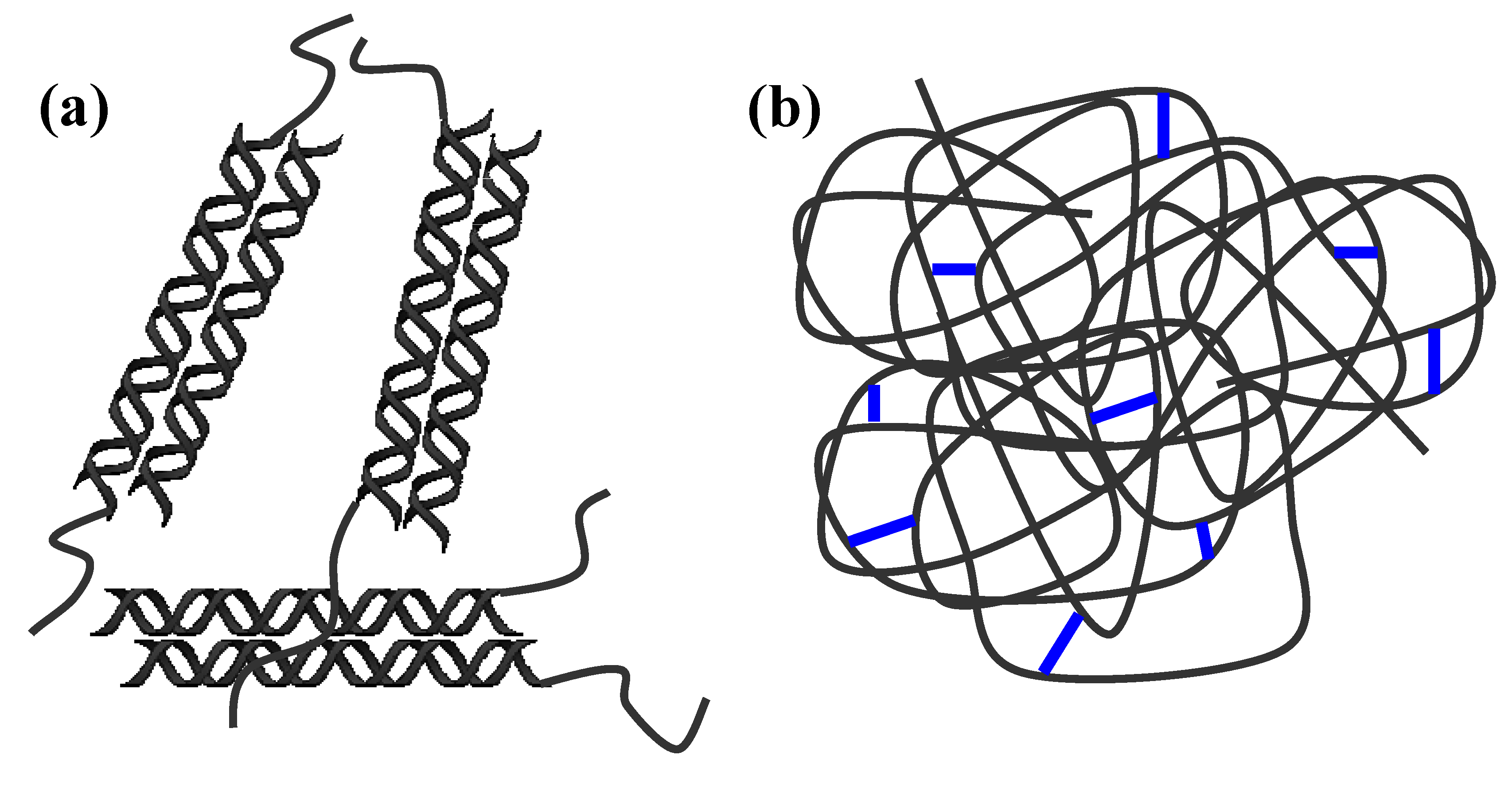
2.2. Water uptake
| H2O | NaCl 0.9% w/v | SIF | SGF | |
|---|---|---|---|---|
| [Gellan 2%]Phys | 0.96 ± 0.03 | 0.99 ± 0.04 | 0.97 ± 0.02 | 0.77 ± 0.02 |
| [Gellan 2% + Lys0.1]Phys | 1.03 ± 0.04 | 1.04 ± 0.03 | 1.02 ± 0.04 | 0.93 ± 0.02 |
| [Gellan 2% + Lys 0.2]Phys | 1.02 ± 0.02 | 1.02 ± 0.04 | 1.03 ± 0.03 | 1.00 ± 0.04 |
| [Gellan 2% + Lys 0.3]Phys | 0.99 ± 0.02 | 1.02 ± 0.02 | 1.02 ± 0.04 | 0.98 ± 0.02 |
| [Gellan 2% + Lys 0.1]Chem | 21.95 ± 0.20 | 1.48 ± 0.03 | 1.54 ± 0.03 | 1.34 ± 0.04 |
| [Gellan 2% + Lys 0.2]Chem | 12.98 ± 0.15 | 1.04 ± 0.02 | 1.02 ± 0.03 | 0.75 ± 0.03 |
| [Gellan 2% + Lys 0.3]Chem | 7.06 ± 0.15 | 0.99 ± 0.04 | 0.98 ± 0.02 | 0.70 ± 0.02 |
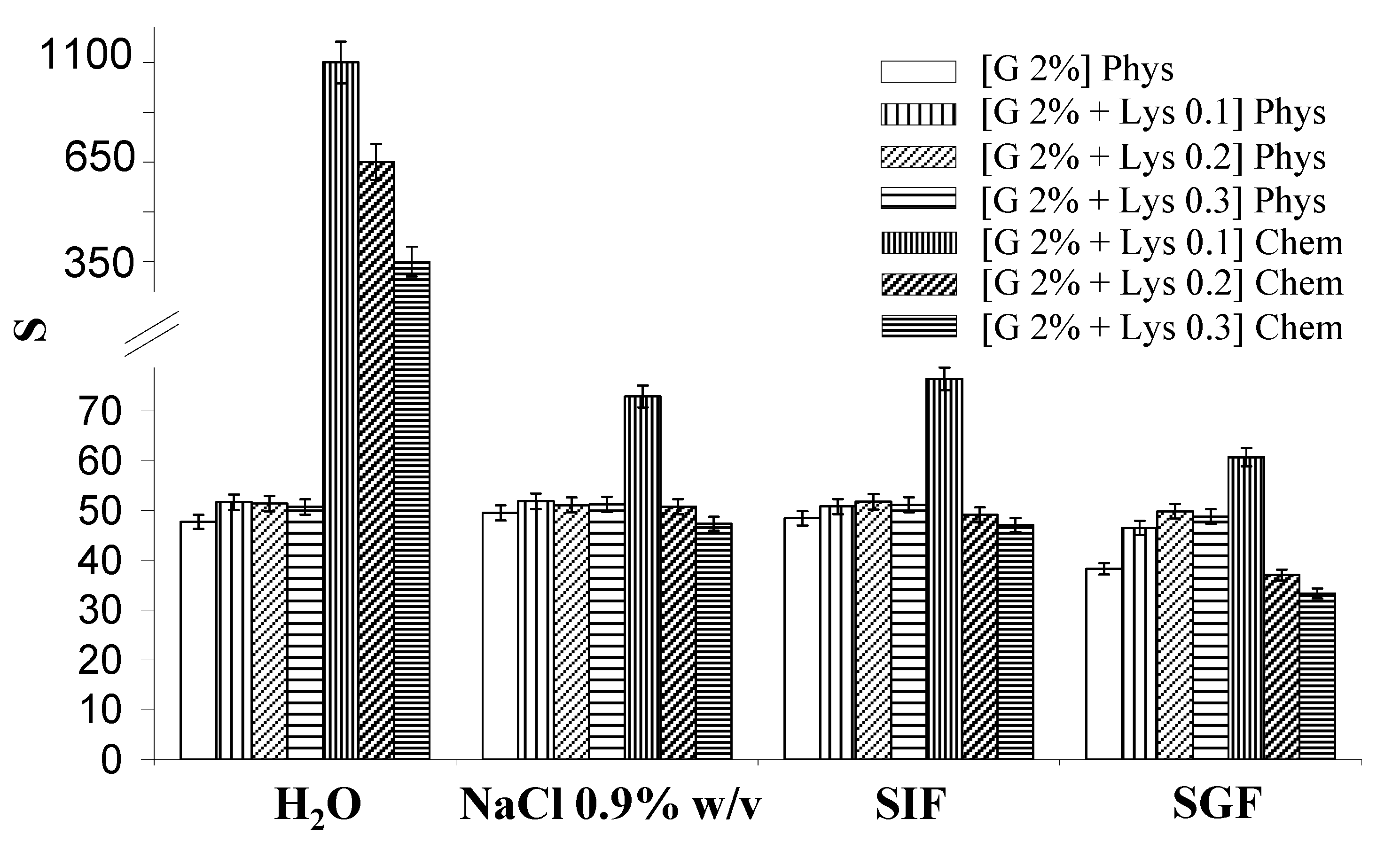
2.3. Rheological measurements
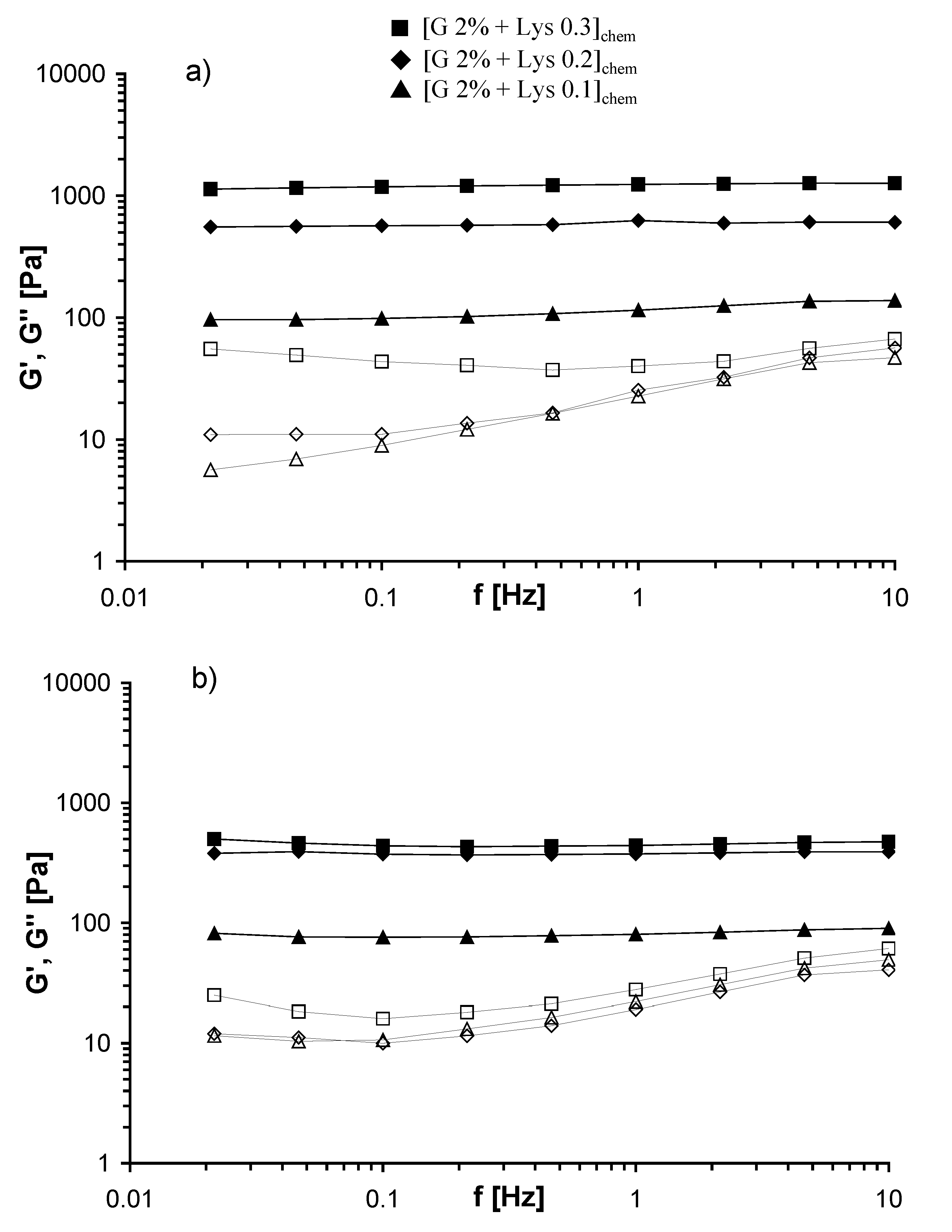
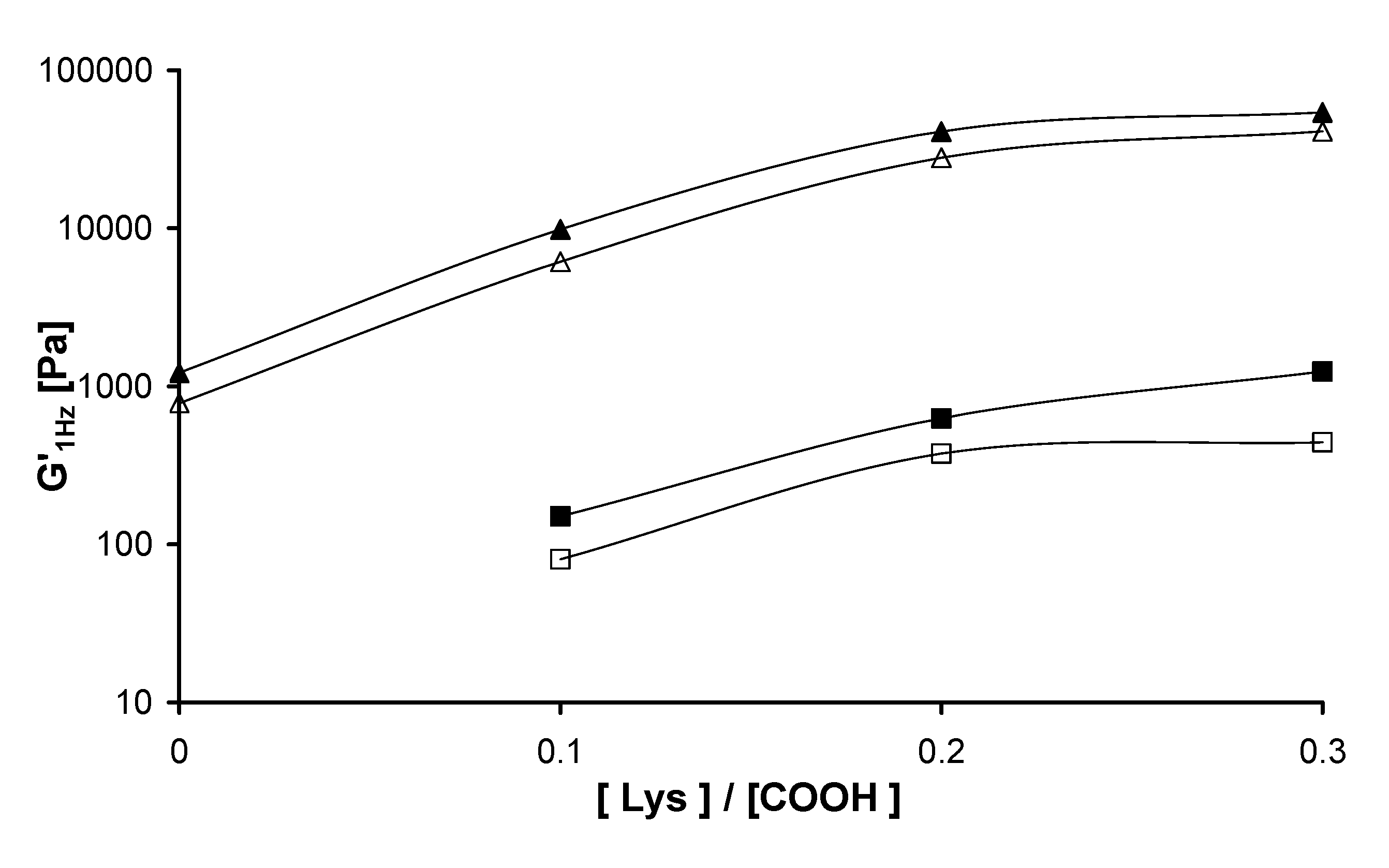
| T = 25 °C | T = 37 °C | |||
|---|---|---|---|---|
| σYield (Pa) | γYield | σYield (Pa) | γYield | |
| [Gellan 2% + Lys 0.1]Phys | 2226 ± 509 | 0.335 ± 0.218 | 1646 ± 110 | 0.324 ± 0.003 |
| [Gellan 2% + Lys 0.2]Phys | 2457 ± 1034 | 0.094 ± 0.040 | 2587 ± 698 | 0.203 ± 0.061 |
| [Gellan 2% + Lys 0.3]Phys | 3296 ± 146 | 0.095 ± 0.003 | 3235 ± 76 | 0.145 ± 0.011 |
| [Gellan 2% + Lys 0.1]Chem | 1130 ± 34 | 11.585 ± 0.417 | 701 ± 88 | 2.705 ± 0.358 |
| [Gellan 2% + Lys 0.2]Chem | 1210 ± 314 | 2.160 ± 0.452 | 675 ± 50 | 1.782 ± 0.075 |
| [Gellan 2% + Lys 0.3]Chem | 1321 ± 160 | 1.425 ± 0.219 | 770 ± 290 | 0.729 ± 0.080 |
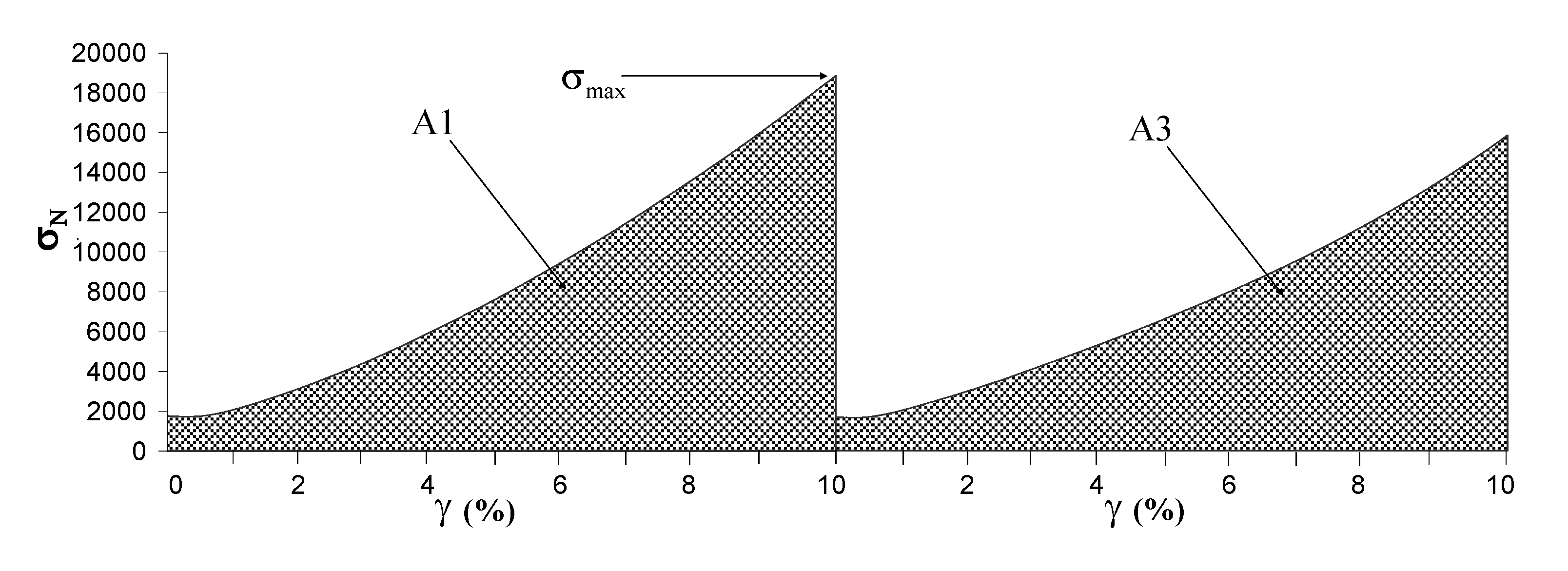
2.4. Compressibility tests
| A1 (N/m2) | A3/A1 (%) | E (N/m2) | |
|---|---|---|---|
| [Gellan 2% + Lys 0.1]Phys | 38950 ± 1530 | 65.6 ± 0.5 | 73830 ± 3740 |
| [Gellan 2% + Lys 0.2]Phys | 72890 ± 17800 | 72.8 ± 3.7 | 154690 ± 38000 |
| [Gellan 2% + Lys 0.3]Phys | 74100 ± 19400 | 87.4 ± 0.5 | 168090 ± 38290 |
| [Gellan 2% + Lys 0.1]Chem | 10990 ± 1660 | 99.1 ± 0.6 | 13440 ± 1470 |
| [Gellan 2% + Lys 0.2]Chem | 14100 ± 2130 | 94.2 ± 0.8 | 18470 ± 4750 |
| [Gellan 2% + Lys 0.3]Chem | 17920 ± 3140 | 88.1 ± 1.2 | 24990 ± 5700 |
2.5. In vitro release studies
2.5.1. Release of vitamin B12
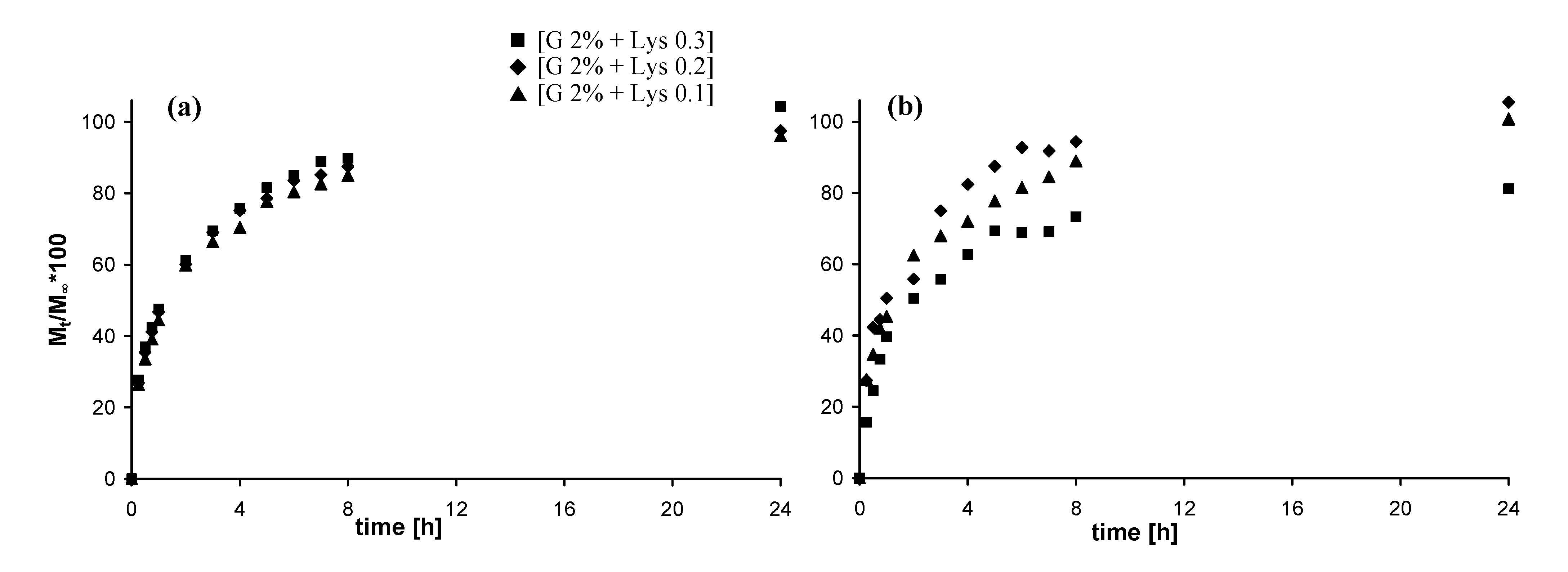
2.5.2. Release of fluorescein isothiocyanate-dextran
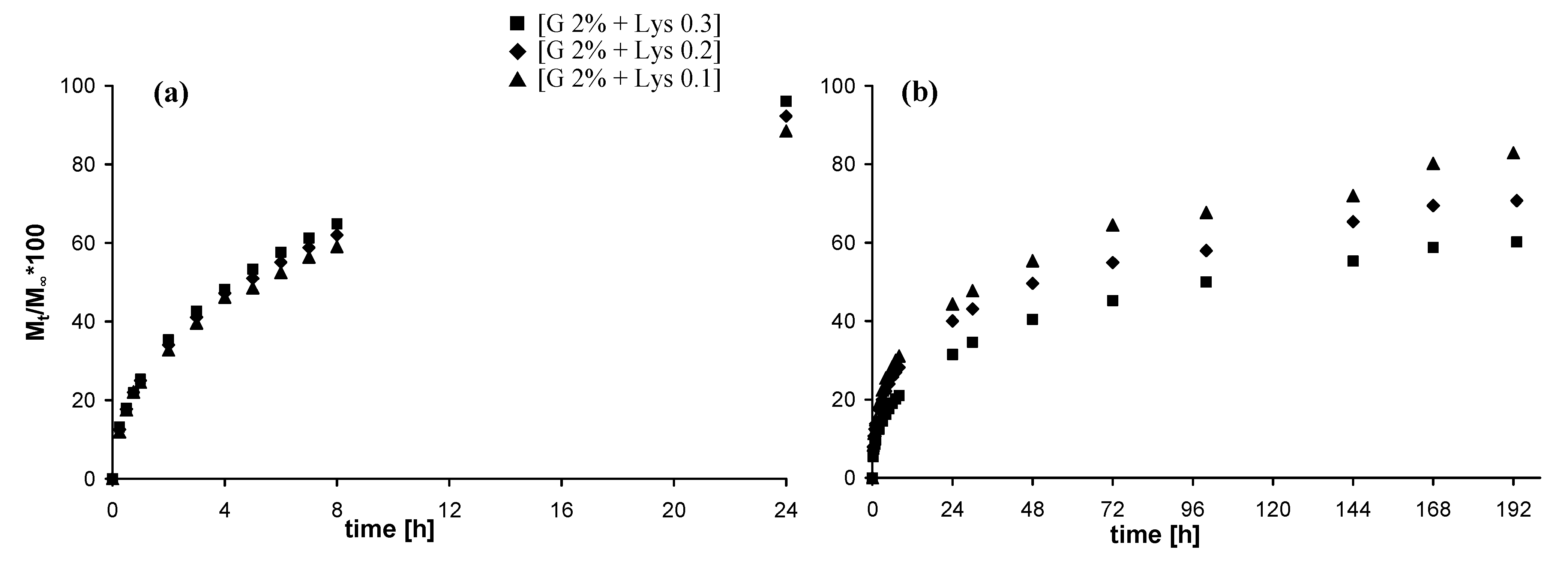
3. Experimental Section
3.1. Materials
3.2. Physical hydrogel preparation
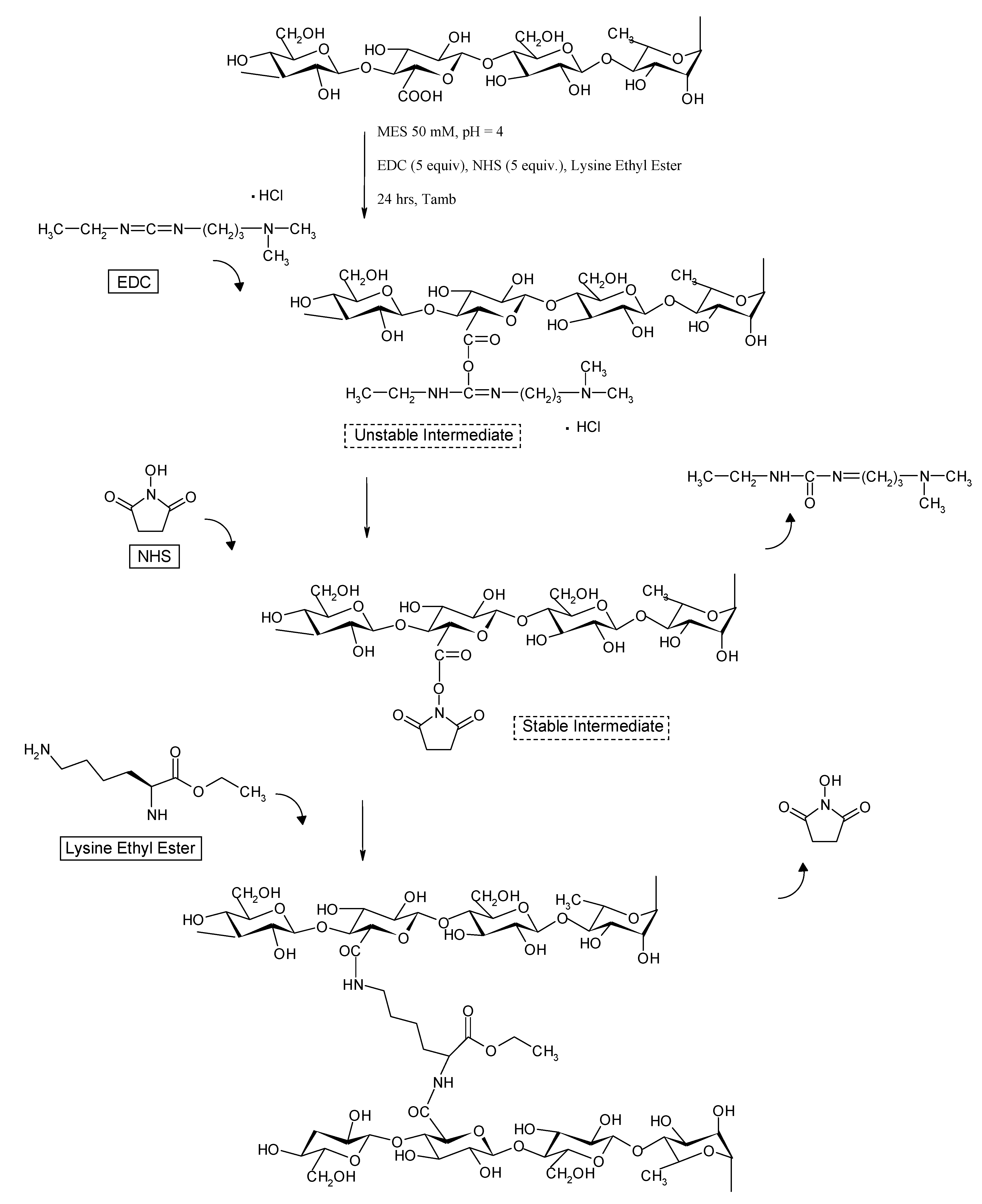
3.3. Chemical hydrogel preparation
- cP = 20 mg/mL
- EDC] / [COOH] = 5
- EDC] / [NHS] = 1
- Lys] / [COOH] = 0.1, 0.2, 0.3
3.4. Water uptake
3.5. Rheological measurements

3.6. Compressibility measurements
3.7. In vitro release
4. Conclusions
Acknowledgements
- Samples Availability: Samples of the physical and chemical gels [Gellan 2% + Lys 0.1], [Gellan 2% + Lys 0.2] and [Gellan 2% + Lys 0.3] are available from the authors.
References and Notes
- Van Tomme, S.R.; Storm, G.; Hennink, W.E. In situ gelling hydrogels for pharmaceutical and biomedical applications. Int. J. Pharm. 2008, 355, 1–18. [Google Scholar] [CrossRef]
- Coviello, T.; Matricardi, P.; Marianecci, C.; Alhaique, F. Polysaccharide hydrogels for modified release formulations. J. Control. Release 2007, 119, 5–24. [Google Scholar] [CrossRef]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichikawa, H. Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Pollock, T.J. Sphingas Groups of Exopolysaccharides (EPS). In Biopolymers – Polysaccharides I; Vandamme, E.J., De Baets, S., Steinbüchel, A., Eds.; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2002; pp. 239–253. [Google Scholar]
- Miyoshi, E.; Takaya, T.; Nishinari, K. Rheological and thermal studies of gel-sol transition in Gellan gum aqueous solutions. Carbohydr. Polym. 1996, 30, 109–119. [Google Scholar] [CrossRef]
- Grasdalen, H.; Smidsroed, O. Gelation of Gellan gum. Carbohydr. Polym. 1987, 7, 371–393. [Google Scholar] [CrossRef]
- Crescenzi, V.; Dentini, M.; Coviello, T.; Rizzo, R. Comparative analysis of the behavior of Gellan gum (S-60) and welan gum (S-130) in dilute aqueous solution. Carbohydr. Res. 1986, 149, 425–432. [Google Scholar] [CrossRef]
- Horinaka, J.; Kani, K.; Hori, Y.; Maeda, S. Effect of pH on the conformation of Gellanchains in aqueous systems. Biophys. Chem. 2004, 111, 223–227. [Google Scholar] [CrossRef]
- Carlfors, J.; Edsman, K.; Petersson, R.; Jörnving, K. Rheological evaluation of Gelrite in situ gels for ophthalmic use. Eur. J. Pharm. Sci. 1998, 6, 113–119. [Google Scholar]
- Miyazaki, S.; Aoyama, H.; Kawasaki, N.; Kubo, W.; Attwood, D. In situ gelling Gellanformulations as vehicles for oral drug delivery. J. Control. Release 1998, 55, 57–66. [Google Scholar] [CrossRef]
- Agnihotri, S.A.; Jawalkar, S.S.; Aminabhavi, T.M. Controlled release of cephalexin through Gellan gum beads: Effect of formulation parameters on entrapment efficiency, size, and drug release. Eur. J. Pharm. Biopharm. 2006, 63, 249–261. [Google Scholar] [CrossRef]
- Agnihotri, S.A.; Aminabhavi, T.M. Development of novel interpenetrating network Gellan gum-poly(vinyl alcohol) hydrogel microspheres for the controlled release of carvedilol. Drug Dev. Ind. Pharm. 2005, 31, 491–503. [Google Scholar] [CrossRef]
- Coviello, T.; Dentini, M.; Rambone, G.; Desideri, P.; Carafa, M; Murtas, E; Riccieri, F.M.; Alhaique, F. A novel co-crosslinked polysaccharide: studies for a controlled delivery matrix. J. Control. Release 1999, 60, 287–295. [Google Scholar] [CrossRef]
- Bhakoo, M.; Woerly, S.; Duncan, R. Release of antibiotics and antitumor agents from alginate and Gellan gum gels. Proc. Int. Symp. Cont. Rel. Bio. Mater. 1991, 18, 441–442. [Google Scholar]
- Crescenzi, V.; Francescangeli, A.; Taglienti, A. New Gelatin-Based Hydrogels via Enzymatic Networking. Biomacromolecules 2002, 3, 1384–1391. [Google Scholar] [CrossRef]
- Kuijpers, A.J.; Engbers, G.H.M.; Feijen, J.; De Smedt, S.C.; Meyvis, T.K.L.; Demeester, J.; Krijgsveld, J.; Zaat, S.A.J.; Dankert, J. Characterization of the Network Structure of Carbodiimide Cross-Linked Gelatin Gels. Biomacromolecules 1999, 32, 3325–3333. [Google Scholar] [CrossRef]
- Ambrosio, L.; Nicolais, L.; Sannino, A. Biodegradable, super absorbent polymer hydrogels and a method for their preparation. Eur. Pat. Appl. EP183 6227, 2007. [Google Scholar]
- Matricardi, P..; Pontoriero, M.; Coviello, T.; Casadei, M.A.; Alhaique, F. In situ cross-linkable novel alginate-dextran methacrylate IPN hydrogels for biomedical applications: Mechanical characterization and drug delivery properties. Biomacromolecules 2008, 9, 2014–2020. [Google Scholar] [CrossRef]
- Coviello, T.; Grassi, M.; Palleschi, A.; Bocchinfuso, G.; Coluzzi, G.; Banishoeib, F.; Alhaique, F. A new scleroglucan/borax hydrogel: Swelling and drug release studies. Int. J. Pharm. 2005, 289, 97–107. [Google Scholar] [CrossRef]
- Lapasin, R.; Pricl, S. Rheology. In Rheology of Industrial Polysaccharides: Theory and Applications; Blackie Academic & Professional, Chapman & Hall: London, UK, 1995; pp. 162–249. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Matricardi, P.; Cencetti, C.; Ria, R.; Alhaique, F.; Coviello, T. Preparation and Characterization of Novel Gellan Gum Hydrogels Suitable for Modified Drug Release. Molecules 2009, 14, 3376-3391. https://doi.org/10.3390/molecules14093376
Matricardi P, Cencetti C, Ria R, Alhaique F, Coviello T. Preparation and Characterization of Novel Gellan Gum Hydrogels Suitable for Modified Drug Release. Molecules. 2009; 14(9):3376-3391. https://doi.org/10.3390/molecules14093376
Chicago/Turabian StyleMatricardi, Pietro, Claudia Cencetti, Roberto Ria, Franco Alhaique, and Tommasina Coviello. 2009. "Preparation and Characterization of Novel Gellan Gum Hydrogels Suitable for Modified Drug Release" Molecules 14, no. 9: 3376-3391. https://doi.org/10.3390/molecules14093376
APA StyleMatricardi, P., Cencetti, C., Ria, R., Alhaique, F., & Coviello, T. (2009). Preparation and Characterization of Novel Gellan Gum Hydrogels Suitable for Modified Drug Release. Molecules, 14(9), 3376-3391. https://doi.org/10.3390/molecules14093376