Tris(2,4,6-trimethoxyphenyl)phosphine (TTMPP): Efficient Catalysts for the Cyanosilylation and Cyanocarbonation of Aldehydes and Ketones
Abstract
:Introduction
Results and Discussion
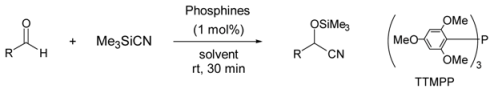 | ||||
|---|---|---|---|---|
| Entry | Phosphines | Aldehyde | solvent | Yield (%) |
| 1 | TTMPP | C6H5CHO | DMF | 98 |
| 2 | 4-CH3OC6H4CHO | 99 | ||
| 3 | 4-ClC6H4CHO | 95 | ||
| 4 | 4-NO2C6H4CHO | 98 | ||
| 5 | α-Naphthaldehyde | 92 | ||
| 6 | β-Naphthaldehyde | 90 | ||
| 7 | C8H17CHO | 95 | ||
| 8 | C6H5CH2CH2CHO | 92 | ||
| 9 | cyclo-C6H11CHO | 93 | ||
| 10 | TMPP[b] | C6H5CHO | 45 | |
| 11 | Ph3P | 30 | ||
| 12 | nBu3P | 68 | ||
| 13 | TTMPP | THF | 95 | |
| 14 | CH3CN | 55 | ||
| 15 | toluene | 30 | ||
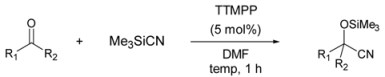 | |||
|---|---|---|---|
| Entry | Ketones (3) | Temp (°C) | Yield (%) |
| 1 |  | r.t. | 97 |
| 2[b], [c] | 99 | ||
| 3 | 95 | ||
| 4 | 93 | ||
| 5 | 98 | ||
| 6 | 90 | ||
| 7 | 93 | ||
| 8 | 94 | ||
| 9 | 76 | ||
| 10[d] | r.t. | trace | |
| 11 | 50 °C | 97 | |
| 12 | 96 | ||
| 13 | 95 | ||
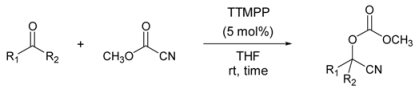 | |||
|---|---|---|---|
| Entry | Carbonyl Compound (3) | Time (h) | Yield (%) |
| C6H5CHO | 1 h | 98 | |
| 2 | 4-CH3OC6H4CHO | 4 h | 85 |
| 3 | 4-ClC6H4CHO | 95 | |
| 4 | 4-BrC6H4CHO | 98 | |
| 5 | 4-NO2C6H4CHO | 88 | |
| 6 | α-Naphthaldehyde | 1 h | 98 |
| 7 | β-Naphthaldehyde | 85 | |
| 8 | C8H17CHO | 4 h | 87 |
| 9 | C6H5CH2CH2CHO | 92 | |
| 10 | cyclo-C6H11CHO | 88 | |
| 11[b] | C6H13COCH3 | 20 h | 75 |
| 12[b] | C6H5CH2CH2COCH3 | 85 | |
| 13[b] | C6H5COCH3 | 24 h | 5 |
| 14[b], [c] | 48 h | 10 | |
Experimental
General
General Procedure for the TTMPP-Catalyzed Cyanosilylation of Aldehydes and Ketones
General Procedure for the TTMPP-Catalyzed Cyanocarbonation of Aldehydes
Conclusions
References
- Evans, D.A.; Truesdale, L.K. Carbonyl insertion reactions of silicon pseudohalides: catalysis. Tetrahedron Lett. 1973, 4929–4932. [Google Scholar]
- Evans, D.A.; Carroll, G.L.; Truesdale, L.K. Synthetic applications of trimethylsilyl cyanide. An efficient synthesis of aminomethyl alcohols. J. Org. Chem. 1974, 39, 914–917. [Google Scholar] [CrossRef]
- North, M. Catalytic, asymmetric cyanohydrin formation. Synlett 1993, 807–820. [Google Scholar] [CrossRef]
- Gregory, J.H. Cyanohydrins in nature and the laboratory: Biology, preparations, and synthetic applications. Chem. Rev. 1999, 99, 3649–3682. [Google Scholar] [CrossRef]
- North, M. Synthesis and applications of non-racemic cyanohydrins. Tetrahedron: Asymmetry 2003, 14, 147–176. [Google Scholar] [CrossRef]
- Brunel, J.-M.; Holmes, I.P. Chemically catalyzed asymmetric cyanohydrin syntheses. Angew. Chem. Int. Ed. 2004, 43, 2752–2778. [Google Scholar] [CrossRef]
- Khan, N.H.; Kureshy, R.I.; Abdi, S.H.R.; Agrawal, S.; Jasra, R. Metal catalyzed asymmetric cyanation reactions. Cood. Chem. Rev. 2008, 252, 593–623. [Google Scholar] [CrossRef]
- Kobayashi, S.; Tsuchiya, Y.; Mukaiyama, T. A facile sythesis of cyanohydrin trimethylsilyl ethers by the addition reaction of trimethylsilyl cyanide with aldehydes under basic condition. Chem. Lett. 1991, 537–540. [Google Scholar]
- Córdoba, R.; Plumet, J. Methyltriphenylphosphonium iodide catalyzes the addition of trimethylsilyl cyanide to aldehydes. Tetrahedron Lett. 2003, 44, 6157–6159. [Google Scholar] [CrossRef]
- Amurrio, I.; Córdoba, R.; Csákÿ, A.G.; Plumet, J. Tetrabutylammonium cyanide catalyzes the addition of TMSCN to aldehydes and ketones. Tetrahedron 2004, 60, 10521–10524. [Google Scholar] [CrossRef]
- Wang, X.; Tian, S.-K. Catalytic cyanosilylation of ketones with simple phosphonium salt. Tetrahedron Lett. 2007, 48, 6010–6013. [Google Scholar] [CrossRef]
- Fetterly, B.; Verkade, J.G. P(RNCH2CH2)N: Efficient catalysis for the cyanosilylation of aldehydes and ketones. Tetrahedron Lett. 2005, 46, 8061–8066. [Google Scholar] [CrossRef]
- Wada, M.; Higashizaki, S. A highly basic triphenylphosphine, [2,4,6-(MeO)3C6H2]3P. J. Chem. Soc. Chem. Commun. 1984, 482–483. [Google Scholar] [CrossRef]
- Yoshimoto, K.; Kawabata, H.; Nakamichi, N.; Hayashi, M. Tris(2,4,6-trimethoxyphenyl)-phosphine (TTMPP): A novel catalyst for selective deacetylation. Chem. Lett. 2001, 30, 934–935. [Google Scholar]
- Kawabata, H.; Hayashi, M. Lewis base-catalyzed transformation of α,β-unsaturated aldehydes to saturated carboxylic acids, esters, and amides. Tetrahedron Lett. 2002, 43, 5645–5647. [Google Scholar] [CrossRef]
- Weeden, J.A.; Chisfolm, J.D. Phosphine-catalyzed nitroaldol reactions. Tetrahedron Lett. 2006, 47, 9313–9316. [Google Scholar] [CrossRef]
- Hirata, N.; Hayashi, M. Nitroaldol reaction catalyzed by tris(2,4,6-trimethoxyphenyl)phosphine (TTMPP). Synth. Commun. 2007, 37, 1653–1657. [Google Scholar] [CrossRef]
- Chuprakov, S.; Malyshev, D.A.; Trofimov, A.; Gevorgyan, V. Sila Morita-Baylis-Hillman reaction of cyclopropenes. J. Am. Chem. Soc. 2007, 129, 14868–14869. [Google Scholar] [CrossRef]
- Trofimov, A.; Gevorgyan, V. Sila-Morita-Baylis-Hillman reaction of arylvinyl ketones: overcoming the dimerization problem. Org. Lett. 2009, 11, 253–255. [Google Scholar] [CrossRef]
- Matsukawa, S.; Okano, N.; Imamoto, T. Phosphine catalyzed aldol reaction between ketene silyl acetals and aldehydes: nucleophilic O–Si and C–Si bond cleavage by phosphines. Tetrahedron Lett. 2000, 41, 103–107. [Google Scholar] [CrossRef]
- Matsukawa, S.; Obu, K. TTMPP catalyzed one-pot silyl ketene acetal-imine condensation route to β-lactams. Chem. Lett. 2004, 33, 1626–1627. [Google Scholar] [CrossRef]
- Matsukawa, S.; Kitazaki, E. Catalytic cyanomethylation of carbonyl compounds and imines with highly basic phosphine Tetrahedron Lett. 2008, 49, 2982–2984. [Google Scholar]
- Matsukawa, S.; Saijo, M. TTMPP-catalyzed trifluoromethylation of carbonyl compounds and imines with trifluoromethylsilane. Tetrahedron Lett. 2008, 49, 4855–4857. [Google Scholar]
- Matsukawa, S.; Sekine, I. TTMPP-catalyzed addition of alkynes using trimethylsilylacetylenes. Synth. Commun. 2009, 39, 1718–1721. [Google Scholar] [CrossRef]
- Matsukawa, S.; Tsukamoto, K. TTMPP: An efficient organocatalyst in the ring-opening of aziridines with silylated nucleophiles. Org. Biomol. Chem. 2009, 7, 3792–3796. [Google Scholar] [CrossRef]
- Belokon’, Y.N.; Clegg, W.; Harrington, R.W.; Ishibashi, E.; Nomura, H.; North, M. Enantioselective and Diastereoselective Syntheses of Cyanohydrin Carbonates. Tetrahedron 2007, 63, 9724–9740. [Google Scholar]
- Tian, S.-K.; Deng, Li. Enantioselective cyanocarbonation of ketones with chiral base. Tetrahedron 2006, 62, 11320–11330. [Google Scholar] [CrossRef]
- Scholl, M.; Lim, C.-K.; Fu, G.C. Convenient and efficient conversion of aldehydes to acylated cyanohydrins using tributyltin cyanide as a catalyst. J. Org. Chem. 1995, 60, 6229–6231. [Google Scholar] [CrossRef]
- Okimoto, M.; Chiba, T. A convenient and improved method for the preparation of cyanohydrin esters from acyl cyanides and aldehydes. Synthesis 1996, 1188–1190. [Google Scholar] [CrossRef]
- Tian, S.-K.; Deng, Li. A highly enantioselective chiral Lewis base-catalyzed asymmetric cyanation of ketones. J. Am. Chem. Soc. 2001, 123, 6195–6196. [Google Scholar] [CrossRef]
- Tian, J.; Yamagiwa, N.; Matsunaga, S.; Shibasaki, M. An asymmetric cyanation reaction and sequential asymmetric cyanation-nitroaldol reaction using a [YLi3{tris(binaphthoxide)}] single catalyst component: Catalyst tuning with achiral additives. Angew. Chem. Int. Ed. 2002, 41, 3636–3638. [Google Scholar] [CrossRef]
- Iwanami, K.; Hinakubo, Y.; Oriyama, T. Catalyst-free DMSO-promoted synthesis of cyanohydrin carbonates from aldehydes. Tetrahedron Lett. 2005, 46, 5881–5883. [Google Scholar] [CrossRef]
- Chinchilla, R.; Nájera, R.; Ortega, F.J. Enantioselective cyanoformylation of aldehydes using a recyclable dimeric cinchonidine ammonium salt as an organocatalyst. Tetrahedron: Asymmetry 2008, 19, 265–268. [Google Scholar] [CrossRef]
- Prakash, G.K.S.; Vaghoo, H.; Panja, C.; Surampudi, V.; Kultyshev, R.; Mathew, T.; Olah, G. A. Effect of carbonates/phosphates as nucleophilic catalysts in dimethylformamide for efficient cyanosilylation of aldehydes and ketones. Proc. Natl. Acad. Sci. USA 2007, 104, 3026–3030. [Google Scholar]
- Sample Availability: Samples of the compounds of interest are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Matsukawa, S.; Sekine, I.; Iitsuka, A. Tris(2,4,6-trimethoxyphenyl)phosphine (TTMPP): Efficient Catalysts for the Cyanosilylation and Cyanocarbonation of Aldehydes and Ketones. Molecules 2009, 14, 3353-3359. https://doi.org/10.3390/molecules14093353
Matsukawa S, Sekine I, Iitsuka A. Tris(2,4,6-trimethoxyphenyl)phosphine (TTMPP): Efficient Catalysts for the Cyanosilylation and Cyanocarbonation of Aldehydes and Ketones. Molecules. 2009; 14(9):3353-3359. https://doi.org/10.3390/molecules14093353
Chicago/Turabian StyleMatsukawa, Satoru, Izumi Sekine, and Ayumi Iitsuka. 2009. "Tris(2,4,6-trimethoxyphenyl)phosphine (TTMPP): Efficient Catalysts for the Cyanosilylation and Cyanocarbonation of Aldehydes and Ketones" Molecules 14, no. 9: 3353-3359. https://doi.org/10.3390/molecules14093353
APA StyleMatsukawa, S., Sekine, I., & Iitsuka, A. (2009). Tris(2,4,6-trimethoxyphenyl)phosphine (TTMPP): Efficient Catalysts for the Cyanosilylation and Cyanocarbonation of Aldehydes and Ketones. Molecules, 14(9), 3353-3359. https://doi.org/10.3390/molecules14093353




