Steroidal Lactones from Withania somnifera, an Ancient Plant for Novel Medicine
Abstract
:Introduction
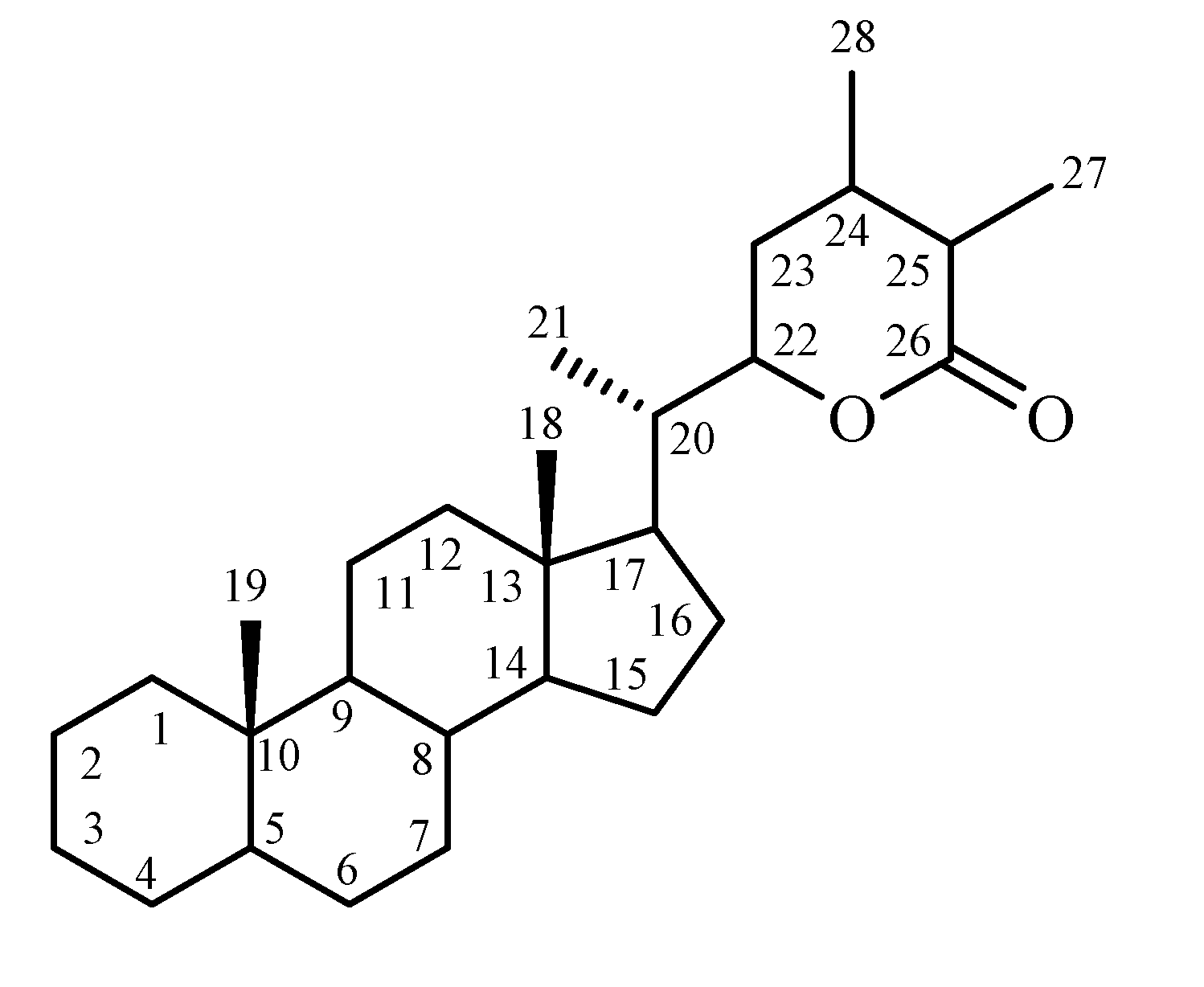
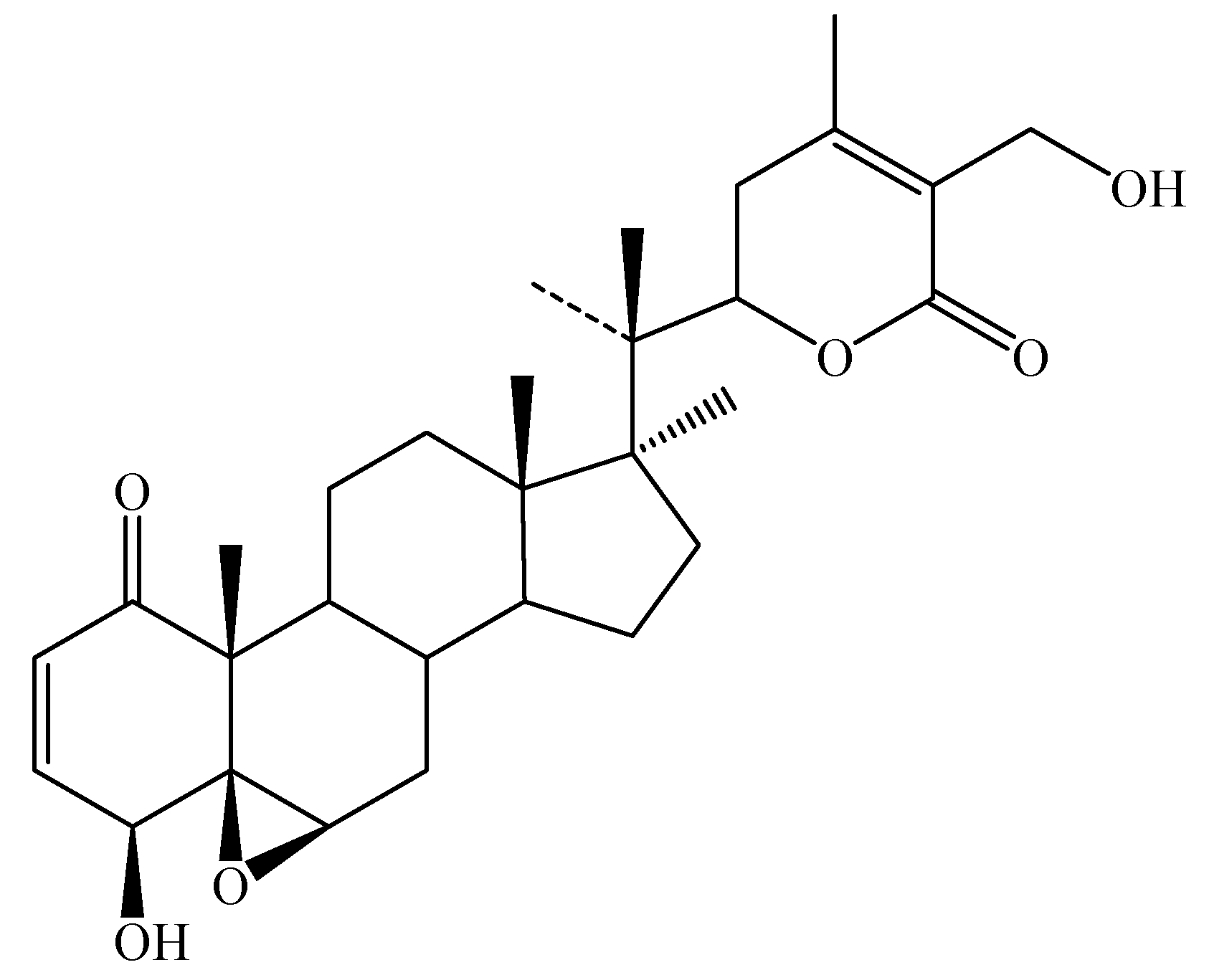
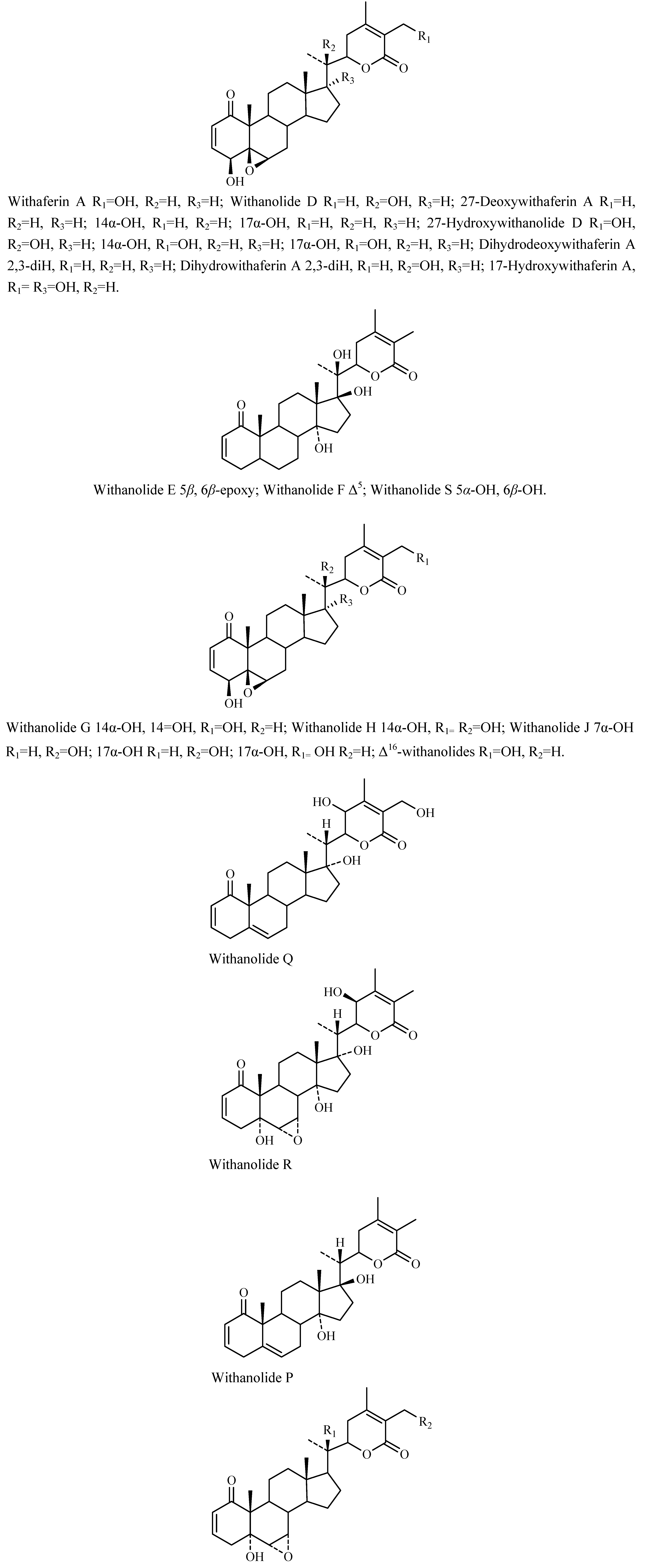
Chemical constituents
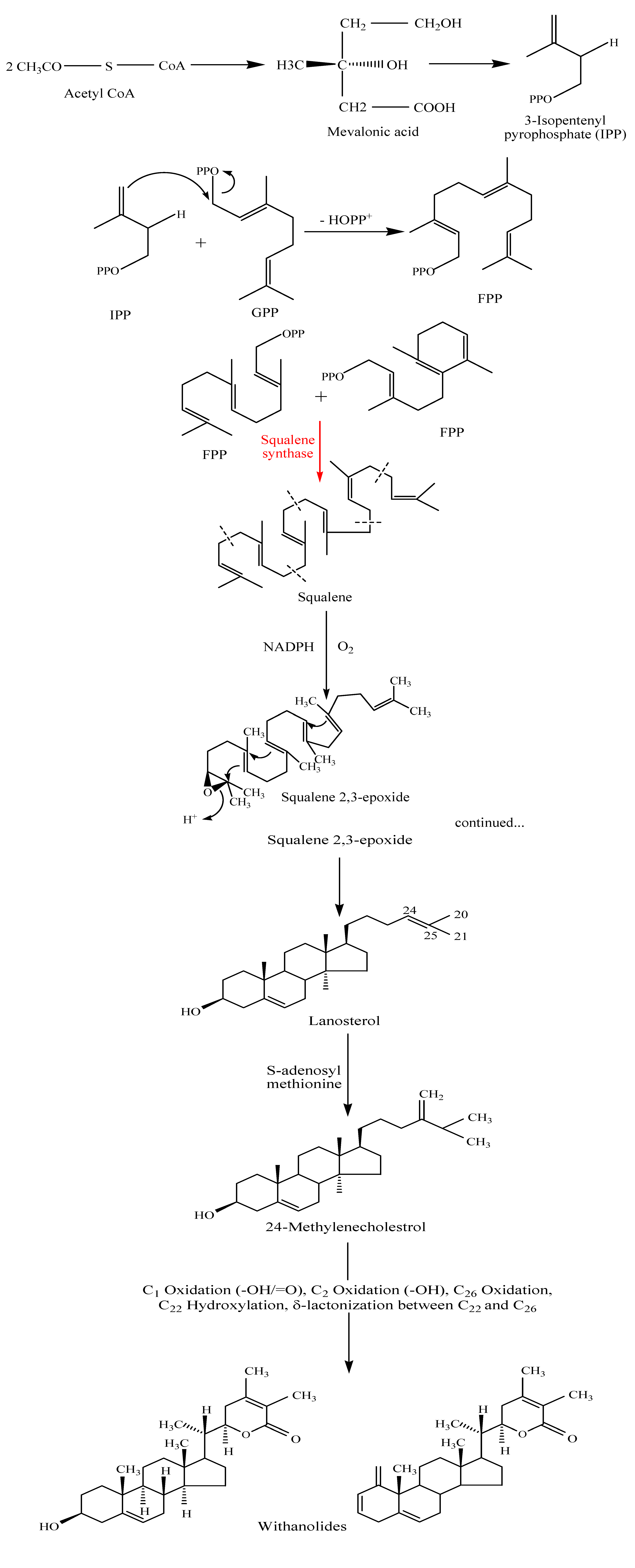
Biosynthesis of withanolides
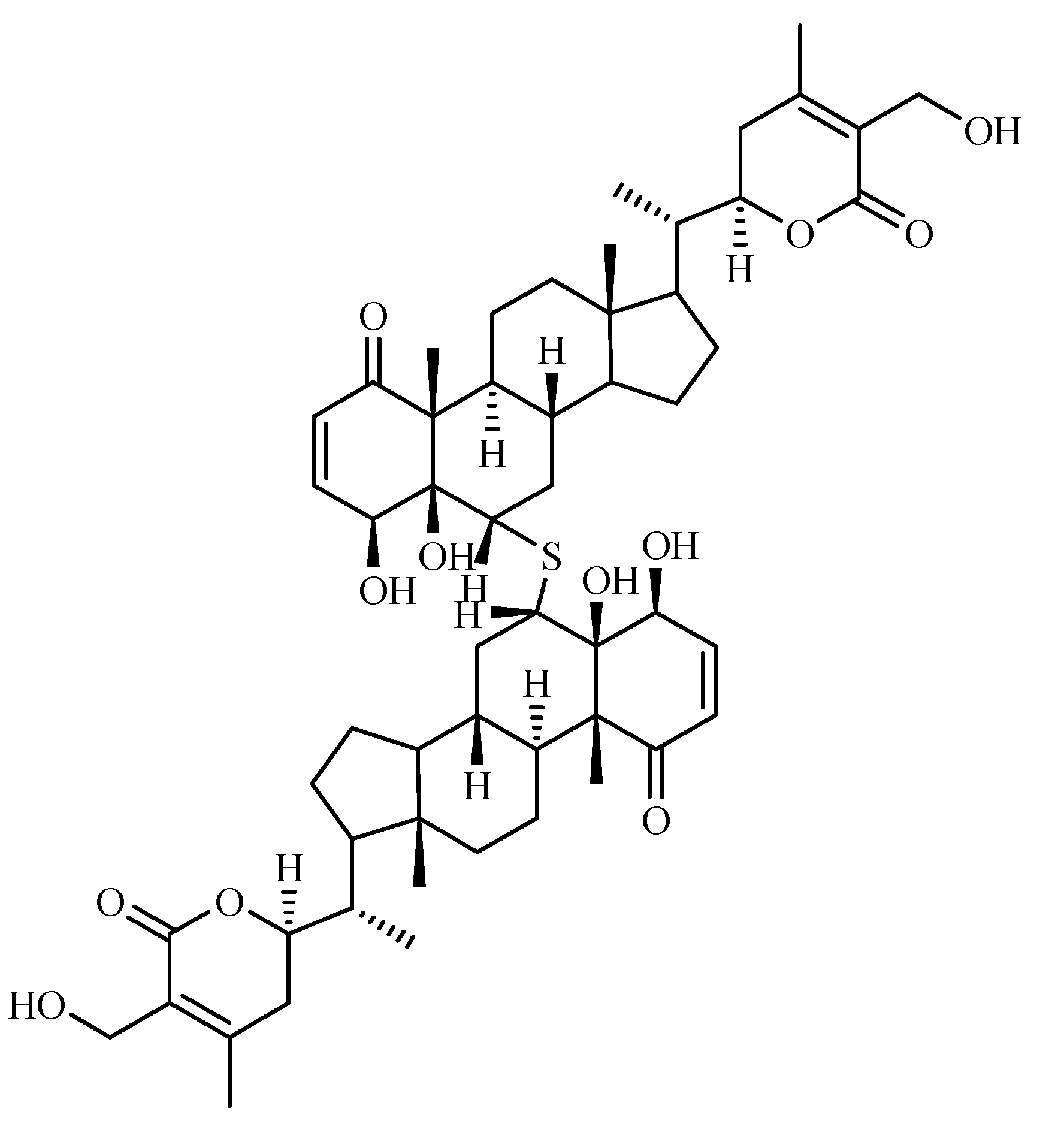
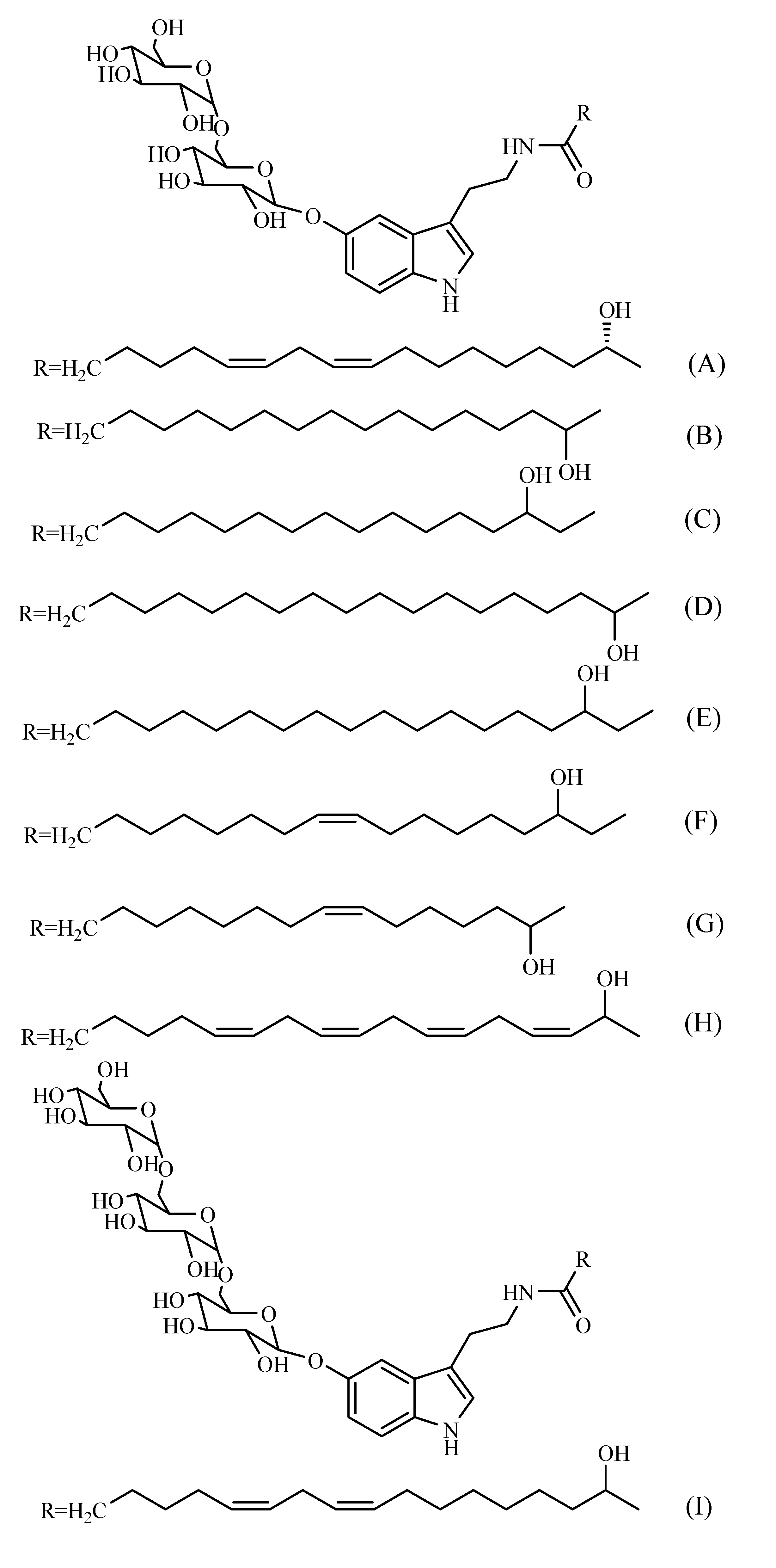
Other compounds
Pharmacological activities of withanolides
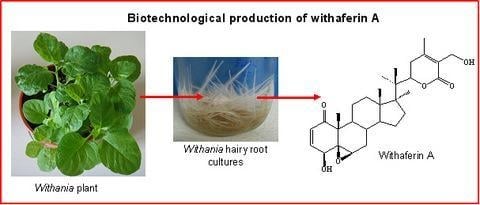
A biotechnological approach to withanolide production
Hairy root cultures as a source of withanolides
Conclusions
Acknowledgements
References and Notes
- Schonbeck-Temesy, E. In Flora Iranica; Rechinger, K.H., Ed.; Akademische Druck-u. Verlagsanstalt: Graz, Austria, 1972; No. 100; pp. 29–26. [Google Scholar]
- Hepper, F.N. In Solanaceae III: taxonomy, chemistry, evolution; Hawkes, J.G., Lester, R.N., Nee, M., Estrada, E., Eds.; Royal Botanic Gardens, Kew: UK, 1991; pp. 211–227. [Google Scholar]
- Warrier, P.K.; Nambiar, V.P.K.; Ramankutty, C. Indian Medicinal Plants: A Compendium of 500 species; Orient Longman: Hyderabad, India, 1996; Vol. 5, p. 409. [Google Scholar]
- Hunziker, A.T. Genera Solanacearum: the genera of the Solanaceae illustrated, arranged according to a new system; Gantner Verlag: Ruggell, Liechtenstein, 2001. [Google Scholar]
- Javanshir, K. Vegetation of Bashagerd. University of Tehran Publication: Tehran, Iran, 2000; pp. 156–162. [Google Scholar]
- Sharma, R. Agro-Techniques of Medicinal Plants; Daya Publishing House: New Delhi, India, 2004; pp. 31–33. [Google Scholar]
- Panwar, J.; Tarafdar, J.C. Distribution of three endangered medicinal plant species and their colonization with arbuscular mycorrhizal fungi. J. Arid Environ. 2006, 65, 337–350. [Google Scholar] [CrossRef]
- Anonymous. The Wealth of India, (Raw Materials); CSIR: New Delhi, India., 1976; Volume 10, pp. 580–585. [Google Scholar]
- Asthana, R.; Raina, M.K. Pharmacology of Withania somnifera (L.) Dunal-a review. Indian Drugs 1989, 26, 199–205. [Google Scholar]
- Singh, S.; Kumar, S. Withania somnifera: The Indian Ginseng Ashwagandha; Central Institute of Medicinal and Aromatic Plants: Lucknow, India, 1998. [Google Scholar]
- Marderosion, A.D. The Review of Natural Products, Facts and Comparisons; St. Louis, MI, USA, 2001; pp. 630–632. [Google Scholar]
- Nadkarni, K.M. Indian Materia Medica; Popular Prakshan Limited: Bombay, India, 1976; p. 1291. [Google Scholar]
- Williamson, E.M. Major Herbs of Ayurveda; Churchill Livingstone: London, UK, 2002; pp. 322–323. [Google Scholar]
- Kapoor, L.D. Handbook of Ayurvedic Medicinal Plants; CRC Press: London, UK, 2001; pp. 337–338. [Google Scholar]
- Watt, G.A. Dictionary of The Economic Products of India; Cosmo Publication: Delhi, India, 1972; Vol. 6, p. 309. [Google Scholar]
- Atal, C.K.; Sethi, P.D. A preliminary chemical examination of Withania coagulans. Indian J. Pharm. 1963, 25, 163–164. [Google Scholar]
- Sheridan Lea, M.A. A rennet ferment contained in the seeds of Withania coagulans. Am. J. Pharm. 1884, 56, 12–14. [Google Scholar]
- Kirtikar, K.R.; Basu, B.D. Indian Medicinal Plants; Shiva Publishers: Dehradun, India, 1991; Vol. 3, p. 1783. [Google Scholar]
- Khan, M.T.J.; Ashraf, M.; Tehniyat, S.; Bukhtair, M.K.; Ashraf, S.; Ahmad, W. Antibacterial activity of Withania coagulans. Fitoterapia 1993, 64, 367–370. [Google Scholar]
- Mozaffarian, V. Trees and shrubs of Iran; Farhange Moaser: Tehran, Iran, 2003; pp. 874–877. [Google Scholar]
- Atta-ur-Rahman; Jamal, A.S.; Choudary, M.I.; Asif, I. Two withanolides from Withania somnifera. Phytochemistry 1991, 30, 3824–3825. [Google Scholar] [CrossRef]
- Atta-ur-Rahman; Abbas, S.; Dur-e-Shawar; Jamal, A.S.; Choudhary, M.I. New withanolides from Withania spp. J. Nat. Prod. 1993, 56, 1000–1006. [Google Scholar] [CrossRef]
- Choudary, M.I.; Abbas, S.; Jamal, A.S.; Atta-ur-Rahman. Withania somnifera- A source of exotic withanolides. Heterocycles 1996, 42, 555–563. [Google Scholar] [CrossRef]
- Rastogi, R.P.; Mehrotra, B.N. Compendium of Indian Medicinal Plants; Central Drug Research Institute: New Delhi, India, 1998. [Google Scholar]
- Bandyopadhyay, M.; Jha, S.; Tepfer, D. Changes in morphological phenotypes and withanolide composition of Ri-transformed roots of Withania somnifera. Plant cell Rep. 2007, 36, 599–609. [Google Scholar] [CrossRef]
- Atal, C.K.; Gupta, O.P.; Ranghunathan, K.; Dhar, K.L.; Central Council for Research in Indian Medicine and Homeopathy, New Delhi, India. 1975.
- Anonymous. Monograph: Withania somnifera. Altern. Med. Rev. 2004, 9, 211–214. [Google Scholar]
- Tursunova, R.N.; Maslennikova, V.A.; Abubakirov, N.K. Withanolides in the vegetable kingdom. Chem. Nat. Comp. 1977, 13, 131–138. [Google Scholar] [CrossRef]
- Christen, P. Les withanolides, steroids vegetaux a structure originale. Pharm. Acta Helv. 1986, 61, 242–246. [Google Scholar]
- Glotter, E. Withanolides and related ergostane-type steroids. Nat. Prod. Rep. 1991, 8, 415–440. [Google Scholar] [CrossRef]
- Alfonso, D.; Bernardinelli, G.; Kapetanidis, I. Withanolides from Iochroma coccineum. Phytochemistry 1993, 34, 517–521. [Google Scholar] [CrossRef]
- Alfonso, D.; Kapetanidis, I. Withanolides from Iochroma gesnerioides. Phytochemistry 1994, 36, 179–183. [Google Scholar] [CrossRef]
- Kirson, I.; Glotter, E.; Lavie, D.; Abraham, A. Constituents of Withania somnifera Dun. XII. The withanolides of an Indian chemotype. J. Chem. Soc. 1971, C, 2032–2044. [Google Scholar]
- Uvais, M.; Sultanabawa, S.; Sotheeswraan, S.; Balasubramanian, S.; El-Kawi, M.A.; Slatkin, D.J.; Schiff, P. L. Pachyovatamine, a bisbenzylisoquinoline alkaloid and other alkaloids from Pachygone ovate. Phytochemistry 1985, 24, 589–592. [Google Scholar] [CrossRef]
- Hussain, S.F.; Khan, L.; Guinaudeau, H.; Leet, J.E.; Freyer, A. J.; Shamma, M. The alkaloidal profile of Cocculus pendulud. Tetrahedron 1984, 40, 2513–2517. [Google Scholar] [CrossRef]
- Leet, J.E.; Hussain, S.F.; Minard, R.D.; Shamma, M. Heterocycles 1982, 19, 2355–2360. [CrossRef]
- Khan, P. M.; Ahmad, S.; Rubnawaz, H.; Malik, A. The first of withanolide from the family Labiatae. Phytochemistry 1999, 51, 669–671. [Google Scholar] [CrossRef]
- Kirson, I.; Abraham, A.; Lavie, D. Analysis of hybrids of Withania somnifera L. (Dun.) Chemotypes III (Israel) by Indian I (Delhi). Israel J. Chem. 1977, 16, 20. [Google Scholar] [CrossRef]
- Nittala, S.S.; Lavie, D. Chemistry and genetics of withanolides in Withania somnifera hybrids. Phytochemistry 1981, 20, 2741–2748. [Google Scholar] [CrossRef]
- Ray, A.B.; Gupta, M. Progress in the Chemistry of Organic Natural Products; Herz, W., Kirby, G.W., Moore, R.E., Steglich, W., Tamm, C., Eds.; Springer Verlag: New York, NY, USA, 1994; Vol. 63, pp. 1–106. [Google Scholar]
- Manitto, P. Biosynthesis of Natural Products; Ellis Howard Limited: Chichester, UK, 1981; pp. 267–279. [Google Scholar]
- Danishefsky, I. Biochemistry for Medical Sciences; Little, Brown and Company: Boston, MA, USA, 1980; p. 232. [Google Scholar]
- Velde, V.V.; Lavie, D. New withanolides of biogenetic interest from Withania somnifera. Phytochemistry 1981, 20, 1359–1363. [Google Scholar] [CrossRef]
- Lavie, D.; Glotter, E.; Shvo, Y. Constituents of Withania somnifera Dun. Part IV The structure of withaferin-A. J. Am. Chem. Soc. 1965, 30, 7517–7531. [Google Scholar]
- Abraham, A.; Kirson, I.; Glotter, E.; Lavie, D. A chemotaxonomic study of Withania somnifera (L.) Dun. Phytochemistry 1968, 7, 957–962. [Google Scholar] [CrossRef]
- Gupta, A.P.; Verma, R.K.; Misra, H.O.; Gupta, M.M. Quantitative determination of withaferin A in different plant parts of Withania somnifera by TLC densitometry. J. Med. Arom. Plant Sci. 1996, 18, 788–790. [Google Scholar]
- Subaraju, G.V.; Vanisree, M.; Rao, C.V.; Sivaramakrishna, C.; Sridhar, P.; Jayaprakasam, B.; Nair, M.G. Ashwagandhanolide, a bioactive dimeric thiowithanolide isolated from the roots of Withania somnifera. J. Nat. Prod. 2006, 69, 1790–1792. [Google Scholar] [CrossRef]
- Jayaprakasam, B.; Strasburg, G.A.; Nair, M.G. Potent lipid peroxidation inhibitors from Withania somnifera fruits. Tetrahedron 2004, 60, 3109–3121. [Google Scholar] [CrossRef]
- Gupta, G.L.; Rana, A.C. Withania somnifera (Ashwagandha): A Review. Pharmacog. Rev. 2007, 1, 129–136. [Google Scholar]
- Johri, S.; Jamwal, U.; Rasool, S.; Kumar, A.; Verma, V.; Qazi, G.N. Purification and characterization of peroxidases from Withania somnifera (AGB 002) and their ability to oxidize IAA. Plant Sci. 2005, 169, 1014–1021. [Google Scholar] [CrossRef]
- Bhattacharya, S.K.; Satyan, K.S.; Chakrabarti, A. Effect of Trasina, an Ayurvedic herbal formulation, on pancreatic islet superoxide dismutase activity in hyperglycaemic rats. Indian J. Exp. Biol. 1997, 35, 297–299. [Google Scholar]
- Dhuley, J.N. Effect of ashwagandha on lipid peroxidation in stress-induced animals. J. Ethnopharmacol. 1998, 60, 173–178. [Google Scholar] [CrossRef]
- Bhattacharya, S.K.; Bhattacharya, A.; Sairam, K.; Ghosal, S. Anxiolytic-antidepressant activity of Withania somnifera glycowithanolides: an experimental study. Phytomedicine 2000, 7, 463–469. [Google Scholar] [CrossRef]
- Bhattacharya, S.K.; Muruganandam, A.V. Adaptogenic activity of Withania somnifera: an experimental study using a rat model of chronic stress. Pharmacol. Biochem. Behav. 2003, 75, 547–555. [Google Scholar] [CrossRef]
- Bhatnagar, M.; Sisodia, S.S.; Bhatnagar, R. Antiulcer and Antioxidant Activity of Asparagus racemosus WILLD and Withania somnifera DUNAL in Rats. Ann. N. Y. Acad. Sci. 2005, 1056, 261–278. [Google Scholar] [CrossRef]
- Schliebs, R.; Liebmann, A.; Bhattacharya, S.K.; Kumar, A.; Ghosal, S.; Bigl, V. Systemic administration of defined extracts from Withania somnifera (Indian Ginseng) and Shilajit differentially affects cholinergic but not glutamatergic and GABAergic markers in rat brain. Neurochem. Int. 1997, 30, 181–190. [Google Scholar] [CrossRef]
- Dhuley, J.N. Nootropic-like effect of Ashawagandha (Withania somnifera L.) in micc. Phytoter. Res. 15, 524–528. [Google Scholar]
- Tohda, C.; Kuboyama, T.; Komatsu, K. Dentrite extension by methanol extracts of Ashwagandha (roots of Withania somnifera) in SK-N-SH cells. NeuroReport 2000, 11, 1981–1985. [Google Scholar] [CrossRef]
- Zhao, J.; Nakamura, N.; Hattori, M.; Kuboyama, T.; Tohda, C.; Komatsu, K. Withanolide derivatives from the roots of Withania somnifera and their neurite outgrowth activities. Chem. Pharm. Bull. 2002, 50, 760–765. [Google Scholar] [CrossRef]
- Tohda, C.; Kuboyama, T.; Komatsu, K. Search for natural products related to regeneration of the neuronal network. Neurosignals 2005, 14, 34–45. [Google Scholar] [CrossRef]
- Kumar, A.; Kulkarni, S.K. Effect of BR-16A (Mentat), a polyherbal formulation on drug-induced catalepsy in mice. Indian J. Exp. Biol. 2006, 44, 45–48. [Google Scholar]
- Ahmad, M.; Saleem, S.; Ahmad, A.S.; Ansari, M.A.; Yousuf, S.; Hoda, M.N.; Islam, F. Neuroprotective effects of Withania somnifera on 6-hydroxydopamine induced Parkinsonism in rats. Hum. Exp. Toxicol. 2005, 24, 137–147. [Google Scholar] [CrossRef]
- Anbalagan, K.; Sadique, J. Influence of an Indian medicine (Ashwagandha) on acutephase reactants in inflammation. Indian J. Exp. Biol. 1981, 19, 245–249. [Google Scholar]
- Anbalagan, K.; Sadique, J. Role of prostaglandins in acute phase proteins in inflammation. Biochem. Med. 1984, 31, 236–245. [Google Scholar] [CrossRef]
- Begum, V.H.; Sadique, J. Long term effect of herbal drug Withania somnifera on adjuvant induced arthritis in rats. Indian J. Exp. Biol. 1988, 26, 877–882. [Google Scholar]
- Somasundaram, S.; Sadique, J.; Subramoniam, A. Influence of extra-intestinal inflammation on the in vitro absorption of 14C-glucose and the effects of anti-inflammatory drugs in the jejunum of rats. Clin. Exp. Pharmacol. Physiol. 1983, 10, 147–152. [Google Scholar] [CrossRef]
- Maurya, R.; Akanksha; Jayendra; Sinh, A.B.; Srivastava, A.K. Coagulanolide, a withanolide from Withania coagulans fruits and antihyperglycemic activity. Bioorg. Med. Chem. Lett. 2008, 18, 6534–6537. [Google Scholar] [CrossRef]
- Prakash, J.; Gupta, S.K.; Dinda, A.K. Withania somnifera root extract prevents DMBA-induced quamous cell carcinoma of skin in Swiss albino mice. Nutr. Cancer 2002, 42, 91–97. [Google Scholar] [CrossRef]
- Falsey, R.R.; Marron, M.T.; Gunaherath, G.M.; Shirahatti, N.; Mahadevan, D.; Gunatilaka, A.A.; Whitesell, L. Actin microfilament aggregation induced by withaferin A is mediated by annexin II. Nat. Chem. Biol. 2006, 2, 33–38. [Google Scholar] [CrossRef]
- Yang, H.; Shi, G.; Dou, Q.P. The tumor proteasome is a primary target for the natural anticancer compound withaferin A isolated from “Indian winter cherry”. Mol. Pharmacol. 2007, 71, 426–437. [Google Scholar] [CrossRef]
- Sen, N.; Banerjee, B.; Das, B.B.; Ganguly, A.; Sen, T.; Pramanik, S.; Mukhopadhyay, S.; Majumder, H.K. Apoptosis is induced in leishmanial cells by a novel protein kinase inhibitor withaferin A and is facilitated by apoptotic topoisomerase I-DNA complex. Cell Death Differ. 2007, 14, 358–367. [Google Scholar] [CrossRef]
- Oh, J.H.; Lee, T.J.; Kim, S.H.; Choi, Y.H.; Lee, S.H.; Lee, J.M.; Kim, Y.H.; Park, J.W.; Know, T.K. Induction of apoptosis by withaferin A in human leukaemia U937 cells through down-regulation of Akt phosphorylation. Apoptosis 2008, 13, 1494–1504. [Google Scholar] [CrossRef]
- Malhotra, C.L.; Das, P.K.; Dhalla, N.S.; Prasad, K. Studies on Withania ashwagandha, Kaul. III. The effect of total alkaloids on the cardiovascular system and respiration. Indian J. Med. Res. 1981, 49, 448–460. [Google Scholar]
- Roja, G.; Heble, M.R.; Sipahimalani, A.T. Tissue cultures of Withania somnifera: morphogenesis and withanolide synthesis. Phytother. Res. 1991, 5, 185–187. [Google Scholar] [CrossRef]
- Ray, S.; Jha, S. Production of withaferin A in shoot cultures of Withania somnifera. Planta med. 2001, 67, 432–436. [Google Scholar] [CrossRef]
- Wadegaonkar, P.A.; Bhagwat, K.A.; Rai, M.K. Direct rhizogenesis and establishment of fast growing normal root organ culture of Withania somnifera Dunal. Plant cell Tissue Org. Cult. 2006, 84, 202–204. [Google Scholar]
- Sangwan, R.S.; Chaurasiya, N.D.; Lal, P.; Misra, L.; Uniyal, G.C.; Tuli, R.; Sangwan, N.S. Whitanolide A bioregeneration in in vitro shoot cultures of ashwagandha (Withania somnifera Dunal), a main medicinal plant in Ayurveda. Chem. Pharm. Bull. 2007, 55, 1371–1375. [Google Scholar] [CrossRef]
- Toivonen, L. Utilization of hairy root cultures for production of secondary metabolites. Biotechnol. Prog. 1993, 9, 12–20. [Google Scholar] [CrossRef]
- Palazón, J.; Cusidó, R.M.; Roig, C.; Piñol, M.T. Effect of rol genes from Agrobacterium rhizogenes TL-DNA on nicotine production in tobacco root cultures. Plant Physiol. Biochem. 1997, 35, 155–162. [Google Scholar]
- Sevón, N.; Oksman-Caldentey, K.M. Agrobacterium rhizogenes-mediates transformation: Root cultures as a source of alkaloids. Planta Med. 2002, 68, 859–868. [Google Scholar] [CrossRef]
- Banerjee, S.; Naqvi, A.A.; Mandal, S.; Ahuja, P.S. Transformation of Withania somnifera (L.) Dunal by Agrobacterium rhizogenes: Infectivity and phytochemical studies. Phytother. Res. 1994, 8, 452–455. [Google Scholar] [CrossRef]
- White, F.F.; Nester, E.W. Hairy root: plasmid encodes virulence in Agrobacterium rhizogenes. J. Bacteriol. 1980, 141, 1134–1141. [Google Scholar]
- Jouanin, L. Restriction map of an agropine-type Ri-plasmid and its homologies with Ti-plasmids. Plasmid 1984, 12, 91–102. [Google Scholar] [CrossRef]
- Binns, A.N.; Tomashow, J.V. Cell biology of Agrobacterium infection and transformation of plants. Annu. Rev. Microbiol. 1988, 42, 575–606. [Google Scholar] [CrossRef]
- Morris, R.O. Genes specifying auxin and cytokinin biosynthesis in phytopathogens. Annu. Rev. Plant Physiol. 1986, 37, 509–538. [Google Scholar] [CrossRef]
- Chriqui, D.; Guivarch, A.; Dewitte, W.; Prinsen, E.; Van Onkelen, H. Rol genes and root initiation and development. Plant Soil 1996, 187, 47–55. [Google Scholar] [CrossRef]
- Nilsson, O.; Olsson, O. Getting to the root: the role of the Agrobacterium rhizogenes rol genes in the formation of hairy roots. Physiol. Plant. 1997, 100, 463–473. [Google Scholar] [CrossRef]
- Piñol, M.T.; Palazón, J.; Cusidó, R.; Serrano, M. Effects of Ri T-DNA from Agrobacterium rhizogenes on growth and hyoscyamine production in Datura stramonium root cultures. Bot. Acta 1996, 109, 133–138. [Google Scholar]
- Bonhomme, V.; Laurain-Mattar, D.; Fliniaux, M.A. Effects of the rol C gene on hairy root: Induction development and tropane alkaloid production by Atropa belladonna. J. Nat. Prod. 2000, 63, 1249–1252. [Google Scholar] [CrossRef]
- Bonhomme, V.; Laurain-Mattar, D.; Lacoux, J.; Fliniaux, M.A.; Jacquin-Dubreil, A. Tropane alkaloid production by hairy roots of Atropa belladonna obtained after transformation with Agrobacterium rhizogenes 15834 and Agrobacterium tumefaciens containing rol A, B, C genes only. J. Biotechnol. 2000, 81, 151–158. [Google Scholar] [CrossRef]
- Palazón, J.; Cusidó, R.M.; Gonzalo, J.; Bonill, M.; Morales, C.; Piñol, M.T. Relation between the amount of rol C gene product and indole alkaloid accumulation in Catharanthus roseus transformed root cultures. J. Plant Physiol. 1998, 153, 712–718. [Google Scholar] [CrossRef]
- Bulgakov, V.P.; Khodakovskaya, M.V.; Labetskaya, N.V.; Chernoded, G.K.; Zhuravlev, Y.N. The impact of plant rol C oncogene on ginsenoside production by ginseng hairy root cultures. Phytochemistry 1998, 49, 1929–1934. [Google Scholar] [CrossRef]
- Moyano, E.; Fornalé, S.; Palazón, J.; Cusidó, R.M.; Bonfill, M.; Morales, C.; Piñol, M.T. Effect of Agrobacterium rhizogenes T-DNA on alkaloid production in Solanaceae plants. Phytochemistry 1999, 52, 1287–1292. [Google Scholar] [CrossRef]
- Mallol, A.; Cusidó, R.M.; Palazón, J.; Bonfill, M.; Morales, C.; Piñol, M.T. Ginsenoside production in different phenotypes of Panax ginseng transformed roots. Phytochemistry 2001, 57, 365–371. [Google Scholar] [CrossRef]
- Mirjalili, M.H.; Fakhr-Tabatabaei, S.M.; Bonfill, M.; Alizadeh, H.; Cusido, R.M.; Ghassempour, A.; Palazón, J. Morphology and withanolide production of Withania coagulans hairy root cultures. Eng. Life Sci. 2008, 9, 1–8. [Google Scholar]
- Vitali, G.; Conte, L.; Nicoletti, M. Withanolide composition and in vitro culture of Italian Withania somnifera. Planta Med. 1996, 62, 287–288. [Google Scholar] [CrossRef]
- Ray, S.; Ghosh, B.; Sen, S.; Jha, S. Withanolide production by root cultures of Withania somnifera transformed with Agrobacterium rhizogenes. Planta Med. 1996, 62, 571–573. [Google Scholar] [CrossRef]
- Sample Availability: Samples of withaferin A and withanolide A are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mirjalili, M.H.; Moyano, E.; Bonfill, M.; Cusido, R.M.; Palazón, J. Steroidal Lactones from Withania somnifera, an Ancient Plant for Novel Medicine. Molecules 2009, 14, 2373-2393. https://doi.org/10.3390/molecules14072373
Mirjalili MH, Moyano E, Bonfill M, Cusido RM, Palazón J. Steroidal Lactones from Withania somnifera, an Ancient Plant for Novel Medicine. Molecules. 2009; 14(7):2373-2393. https://doi.org/10.3390/molecules14072373
Chicago/Turabian StyleMirjalili, Mohammad Hossein, Elisabeth Moyano, Mercedes Bonfill, Rosa M. Cusido, and Javier Palazón. 2009. "Steroidal Lactones from Withania somnifera, an Ancient Plant for Novel Medicine" Molecules 14, no. 7: 2373-2393. https://doi.org/10.3390/molecules14072373
APA StyleMirjalili, M. H., Moyano, E., Bonfill, M., Cusido, R. M., & Palazón, J. (2009). Steroidal Lactones from Withania somnifera, an Ancient Plant for Novel Medicine. Molecules, 14(7), 2373-2393. https://doi.org/10.3390/molecules14072373