Rubiacordone A: A New Anthraquinone Glycoside from the Roots of Rubia cordifolia
Abstract
:Introduction
Results and Discussion
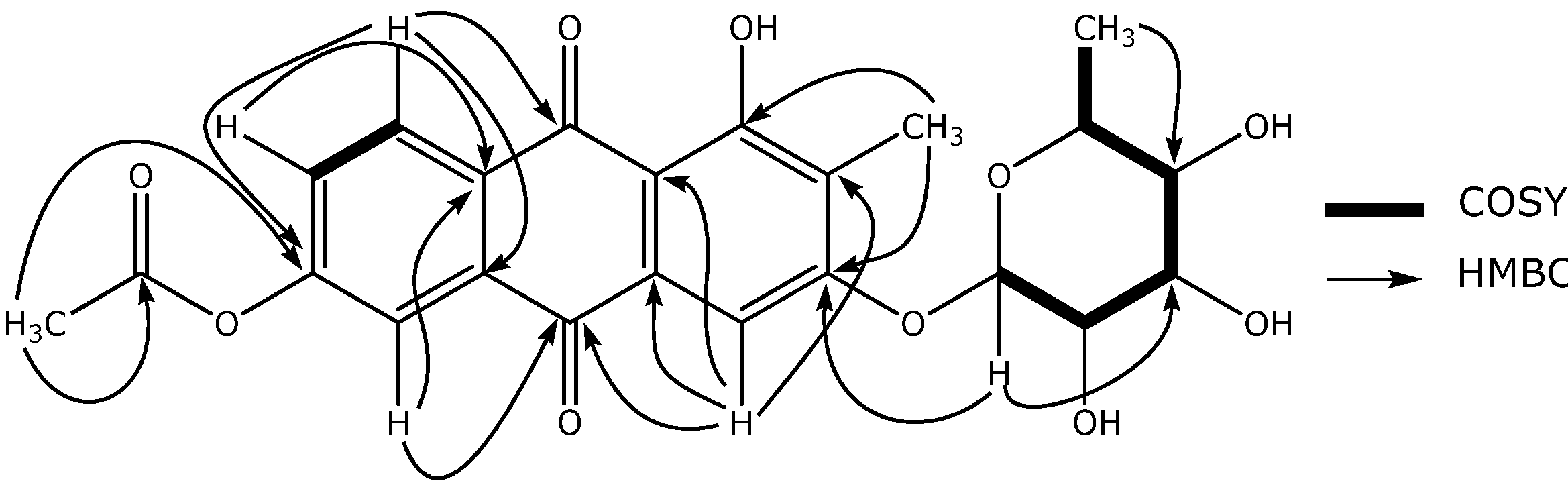
Characterization of compound 1
| Position | 1H-NMR | J (Hz) | 13C-NMR | DEPT | COSY | HMBC |
|---|---|---|---|---|---|---|
| 1 | - | - | 162.0 | C | - | - |
| 2 | - | - | 123.4 | C | - | - |
| 3 | - | - | 156.8 | C | - | - |
| 4 | 7.35, s | - | 104.6 | CH | - | C-2, C-4a, C-9a, C-10 |
| 4a | - | - | 132.7 | C | - | - |
| 5 | 7.45, d | 2.4 | 113.1 | CH | H-7 | C-7, C-8a, C-10 |
| 6 | - | - | 158.7 | C | - | - |
| 7 | 7.21, dd | 8.0, 2.4 | 120.5 | CH | H-5, H-8 | C-5, C-8a |
| 8 | 7.95, d | 8.0 | 129.1 | CH | H-7 | C-6, C-9, C-10a |
| 8a | - | - | 124.5 | C | - | - |
| 9 | - | - | N.D. c | - | - | - |
| 9a | - | - | 108.6 | C | - | - |
| 10 | - | - | N.D. c | - | - | - |
| 10a | - | - | 131.7 | C | - | - |
| Ac-C=O | - | - | 169.2 | C | - | - |
| Ac-Me | 2.10, s | - | 24.3 | CH3 | - | Ac-C=O, C-6 |
| 2-CH3 | 1.05, s | - | 8.7 | CH3 | - | C-1, C-2, C-3 |
| Rha | ||||||
| 1’ | 5.48, d | 1.5 | 103.3 | CH | H-2’ | C-5’, C-3’ |
| 2’ | 4.22, dd | 1.5, 3.4 | 71.7 | CH | H-1’, H-3’ | C-3’, C-4’ |
| 3’ | 3.79, dd | 3.4, 10.0 | 72.9 | CH | H-2’, H-4’ | C-1’, C-5’ |
| 4’ | 3.36, dd | 9.5, 10.0 | 72.0 | CH | H-3’, H-5’ | C-2’, C-5’ C-6’ |
| 5’ | 3.45, m | - | 73.2 | CH | H-4’, H-6’ | C-1’, C-3’ |
| 6’ | 0.88, d | 6.0 | 18.6 | CH3 | H-5’ | C-4’, C-5’ |
Bioactivity Results
| Inhibition zone diameter (mm) | |||||
|---|---|---|---|---|---|
| CEb | 1 | 2 | Amracin | ||
| 200 μg/disc | 20 μg/disc | 20 μg/disc | 10 μg/disc | ||
| S. aureus | 6 ± 0.5 | 13 ± 0.3 | 19 ± 0.6 | 32 ± 0.5 | |
| B. subtilis | 16 ± 0.6 | 26 ± 0.5 | 27 ± 0.3 | 41 ± 0.3 | |
| S. epidermidis | 7 ± 0.3 | 14 ± 0.4 | 11 ± 0.5 | 36 ± 0.6 | |
| S. faecalis | 14 ± 0.6 | 29 ± 0.2 | 22 ± 0.5 | 35 ± 0.5 | |
| B. cereus | 14 ± 0.3 | 24 ± 0.6 | 26 ± 0.5 | 35 ± 0.5 | |
| V. parahaemolyticus | 10 ± 0.6 | 8 ± 0.5 | 11 ± 0.3 | 24 ± 0.6 | |
| P. aeruginosa | 7 ± 0.5 | 9 ± 0.5 | 8 ± 0.6 | 21 ± 0.4 | |
| S. typhimurium | 6 ± 0.4 | 12 ± 0.2 | 8 ± 0.3 | 32 ± 0.5 | |
| E. coli | 13 ± 0.4 | 10 ± 0.4 | 11 ± 0.5 | 28 ± 0.4 | |
Conclusions
Experimental
General
Extraction and isolation
Acid hydrolysis of compound 1
Absolute configuration of rhamnose
Antimicrobial assay
Acknowledgements
References and Notes
- Chen, S.; Fen, R.; Chen, B.; Jing, L.; Dong, X.; Gu, Z.; Wang, S. A study on the medicinal plants of genus Rubia. I. Botanical origins and resource of Chinese traditional drug “Qian Cao” (Madder). Tian Ran Chan Wu Yan Jiu Yu Kai Fa (in Chinese). 1991, 4, 10–18. [Google Scholar]
- Li, S. Ben Cao Gang Mu; (in Chinese). The People’s Health Press: Bei Jing, P.R. China, 1982; p. 1621. [Google Scholar]
- Jiangsu New Medical College. Dictionary of Traditional Chinese Medicine; (in Chinese). Jiangsu Technology Press: Nan Jing, P.R. China, 1977; p. 376. [Google Scholar]
- Itokawa, H.; Qiao, Y.; Takeya, K.; Iitaka, Y. New triterpenoids from Rubia cordifolia var. pratensis. (Rubiaceae). Chem. Pharm. Bull. 1989, 37, 1670–1672. [Google Scholar] [CrossRef]
- Tao, J.; Morikawa, T.; Ando, S.; Matsuda, H.; Yoshikawa, M. Bioactive constituents from Chinese natural medicines. XI. Inhibitors on NO production and degranulation in RBL-2H3 from Rubia yunnanensis: Structures of Rubianosides II, III, and IV, Rubianol-g, and Rubianthraquinone. Chem. Pharm. Bull. 2003, 51, 654–662. [Google Scholar] [CrossRef]
- Singh, R.; Geetanjali. Isoaltion and synthesis of anthraquinones and related compounds of Rubia cordifolia. J. Serb. Chem. Soc. 2005, 70, 937–942. [Google Scholar] [CrossRef]
- Cai, Y.; Sun, M.; Xing, J.; Corke, H. Antioxidant phenolic constituents in roots of Rheum officeinale and Rubia cordifolia: structure-radical scavenging activeity relationships. J. Agric. Food Chem. 2004, 52, 7884–7890. [Google Scholar]
- Itokawa, H.; Mihara, K.; Takeya, K. Studies on a novel anthraquinone and its glycosides isolated from Rubia cordifolia and R. akane. Chem. Pharm. Bull. 1983, 31, 2353–2358. [Google Scholar] [CrossRef]
- Mischenko, N.; Fedoreyev, S.; Glazunov, V.; Chernoded, G.; Bulgakov, V.; Zhuravlev, Y. Anthraquinone production by callus cultures of Rubia cordifolia. Fitoterapia 1999, 70, 552–557. [Google Scholar] [CrossRef]
- Lee, J.; Hitosuyanagi, Y.; Takeya, K. Structure of cytotoxic bicycleic hexapeptides, RA-XIX, -XX, -XXI, and -XXII, from Rubia cordifolia L. Tetrahedron 2008, 64, 4117–4125. [Google Scholar] [CrossRef]
- Lee, J.; Hitosuyanagi, Y.; Kim, I.; Hasuda, T.; Takeya, K. A novel bicyclic hexapeptide, RA-XVIII, from Rubia cordifolia: structure, semi-synthesis, and cytotoxicy. Bioorg. Med. Chem. Letters 2008, 18, 808–811. [Google Scholar] [CrossRef]
- Son, J.; Jung, S.; Jung, J.; Fang, Z.; Lee, C.; Seo, C.; Moon, D.; Kim, B.; Kin, M.; Woo, M. Anticancer constituents from the roots of Rubia cordifolia L. Chem. Pharm. Bull. 2008, 56, 213–216. [Google Scholar] [CrossRef]
- Berghe, V.; Vlietinck, A. Screening methods for antibacterial and antiviral agents from higher plants. Methods Plant Biochem. 1991, 6, 47–68. [Google Scholar]
- Sample Availability: Samples are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, X.; Liu, Z.; Chen, Y.; Wang, L.-J.; Zheng, Y.-N.; Sun, G.-Z.; Ruan, C.-C. Rubiacordone A: A New Anthraquinone Glycoside from the Roots of Rubia cordifolia. Molecules 2009, 14, 566-572. https://doi.org/10.3390/molecules14010566
Li X, Liu Z, Chen Y, Wang L-J, Zheng Y-N, Sun G-Z, Ruan C-C. Rubiacordone A: A New Anthraquinone Glycoside from the Roots of Rubia cordifolia. Molecules. 2009; 14(1):566-572. https://doi.org/10.3390/molecules14010566
Chicago/Turabian StyleLi, Xiang, Zhi Liu, Yun Chen, Li-Juan Wang, Yi-Nan Zheng, Guang-Zhi Sun, and Chang-Chun Ruan. 2009. "Rubiacordone A: A New Anthraquinone Glycoside from the Roots of Rubia cordifolia" Molecules 14, no. 1: 566-572. https://doi.org/10.3390/molecules14010566
APA StyleLi, X., Liu, Z., Chen, Y., Wang, L.-J., Zheng, Y.-N., Sun, G.-Z., & Ruan, C.-C. (2009). Rubiacordone A: A New Anthraquinone Glycoside from the Roots of Rubia cordifolia. Molecules, 14(1), 566-572. https://doi.org/10.3390/molecules14010566





