CuO and Ag2O/CuO Catalyzed Oxidation of Aldehydes to the Corresponding Carboxylic Acids by Molecular Oxygen
Abstract
:Introduction
Results and Discussion
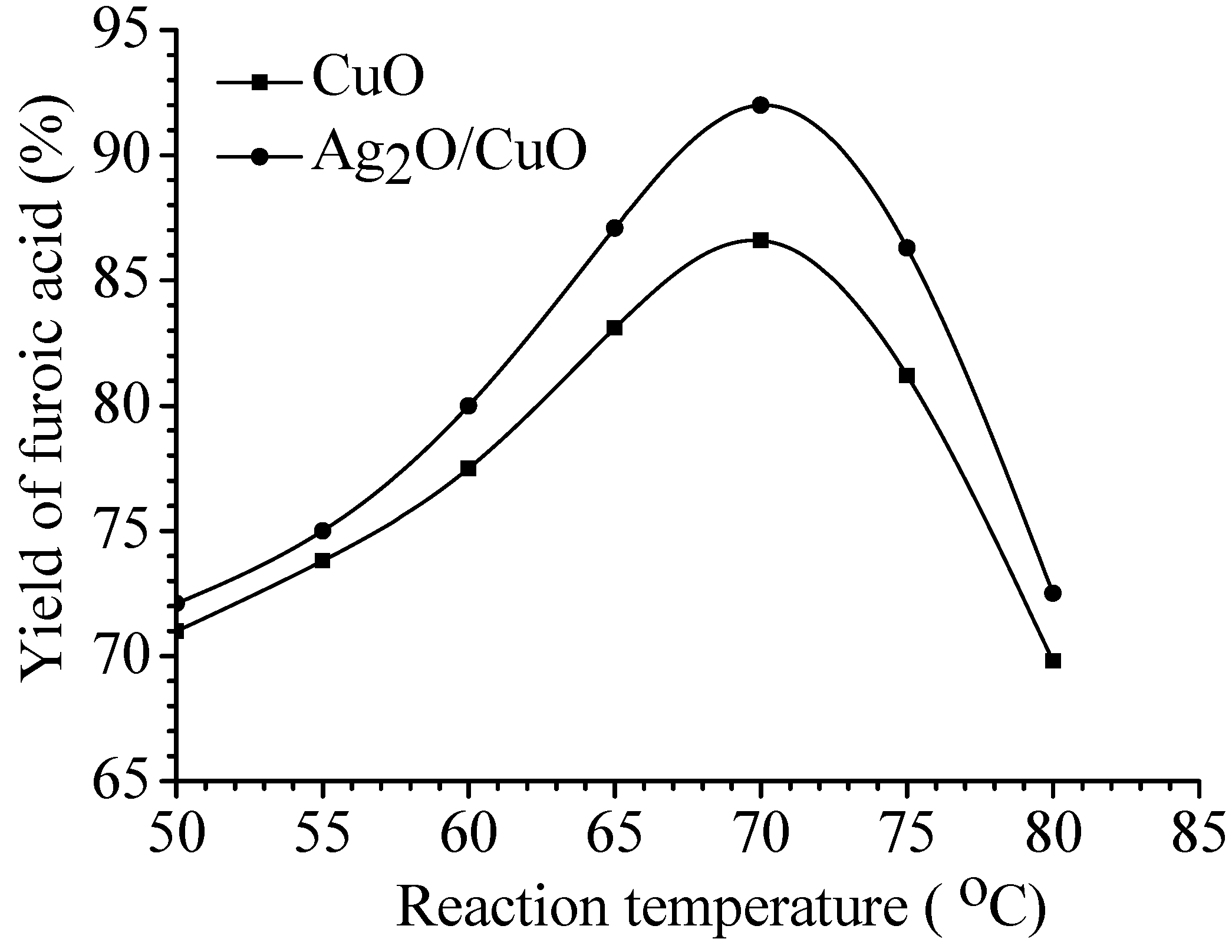
Reaction conditions: Reaction temperature
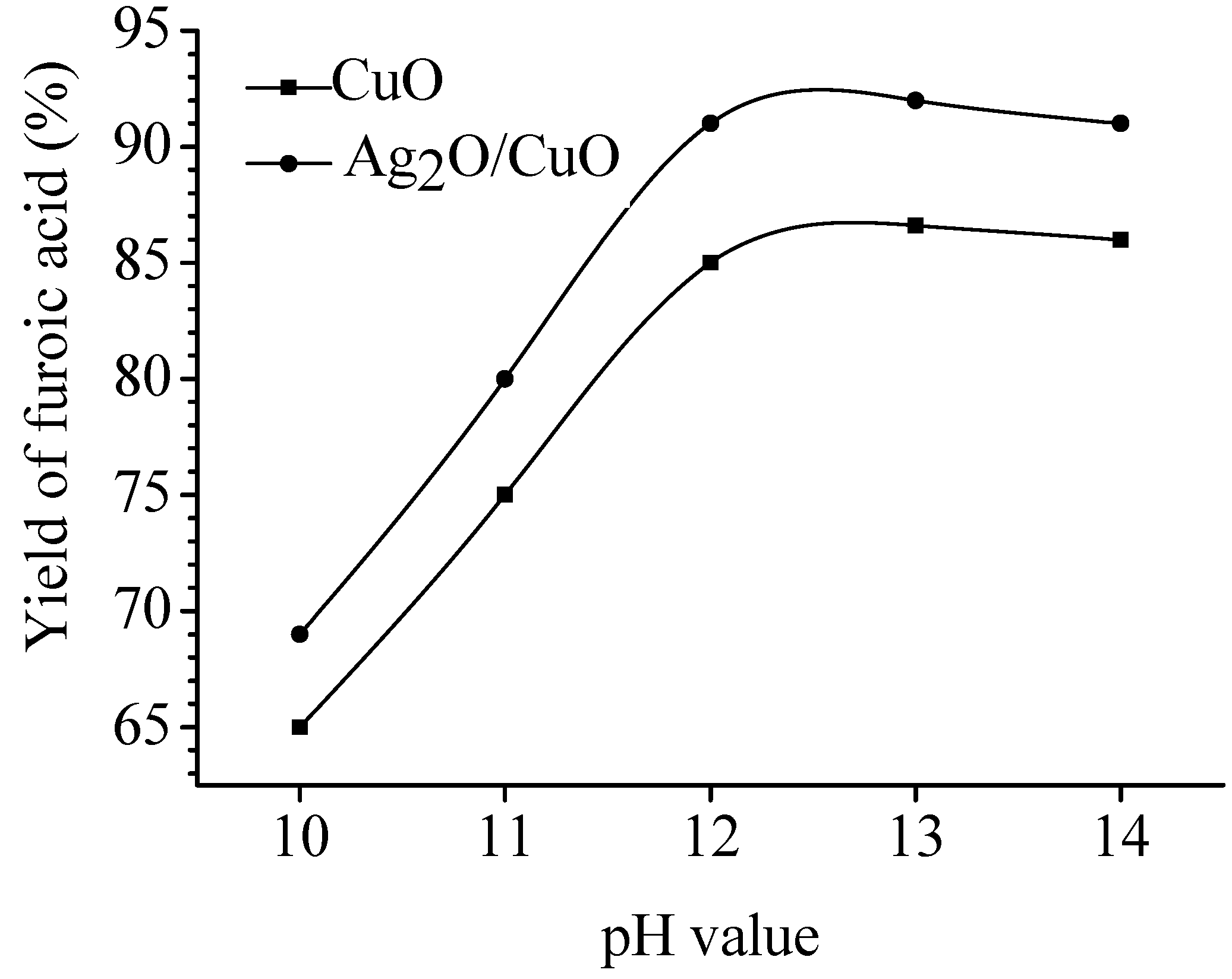
pH value
Amount of catalyst

Size of catalyst

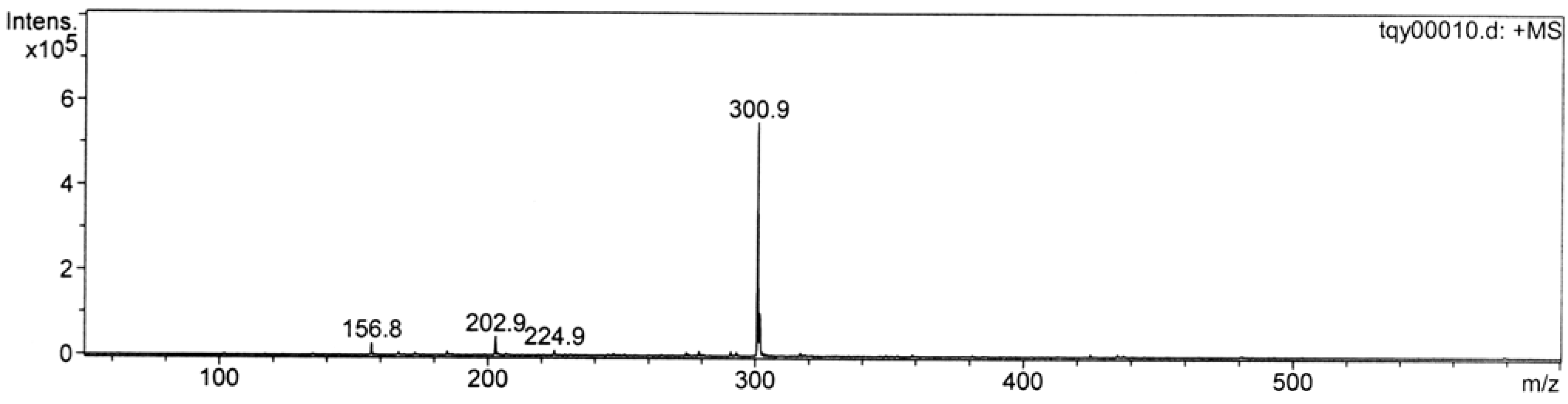
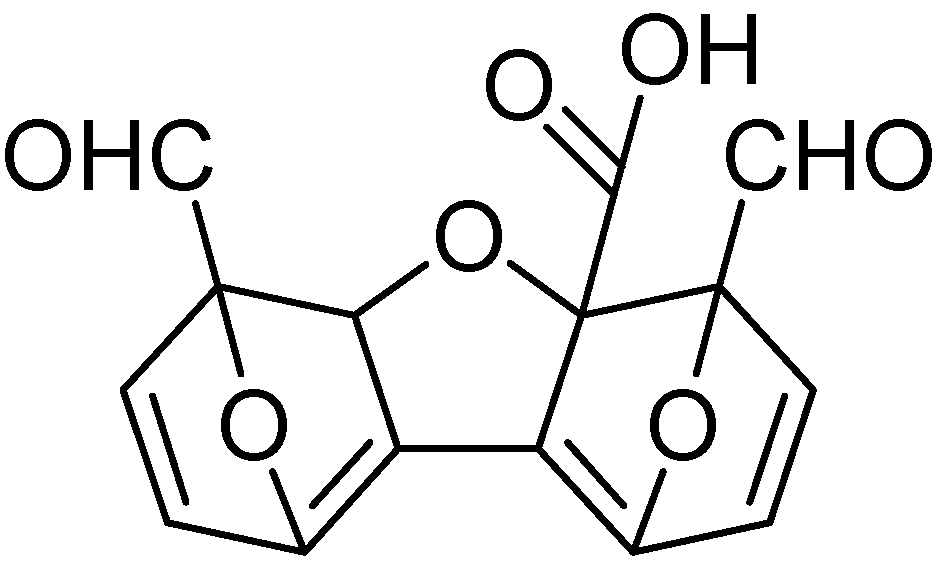
Cation poisoning
Catalyst regeneration and aging
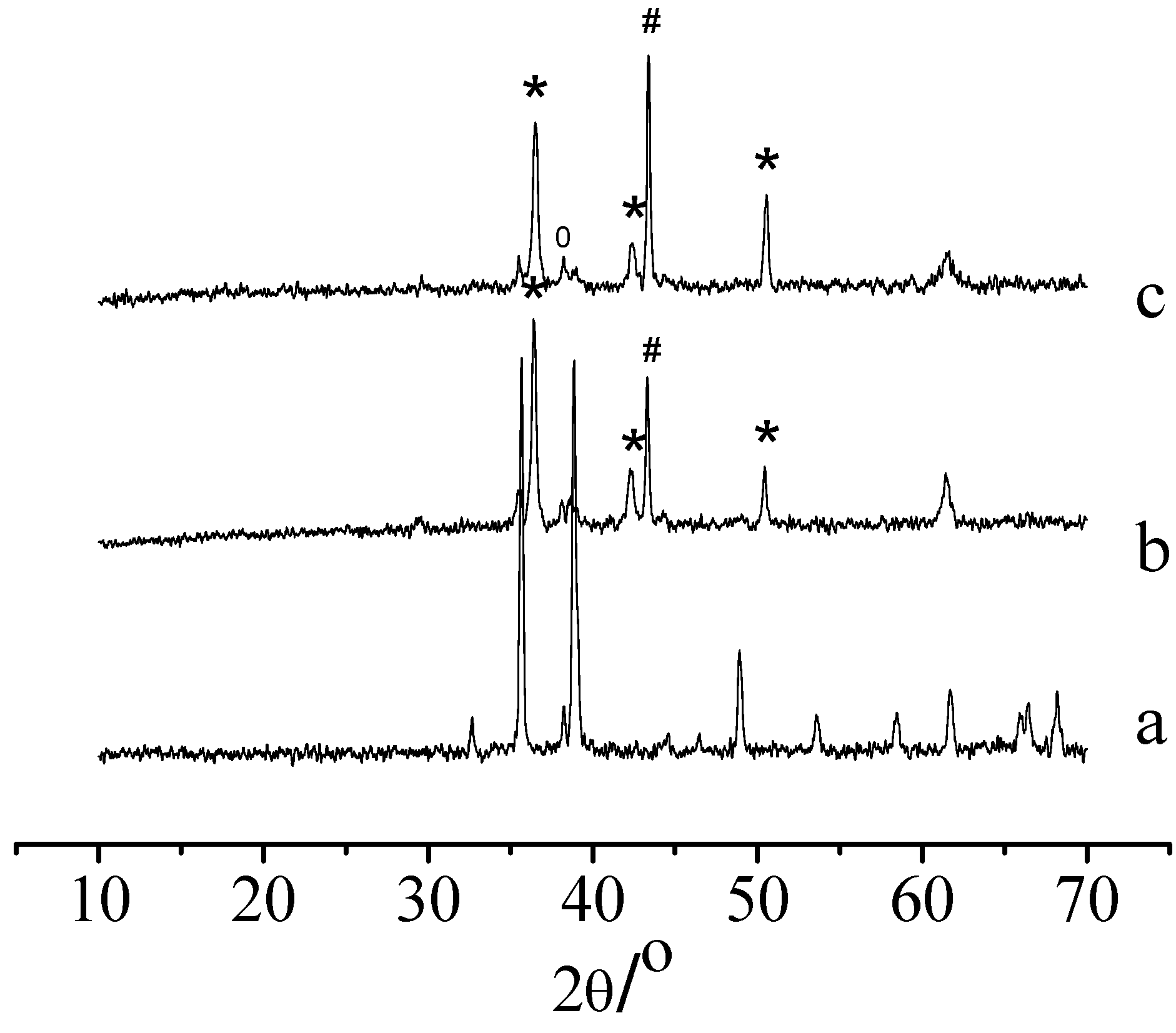



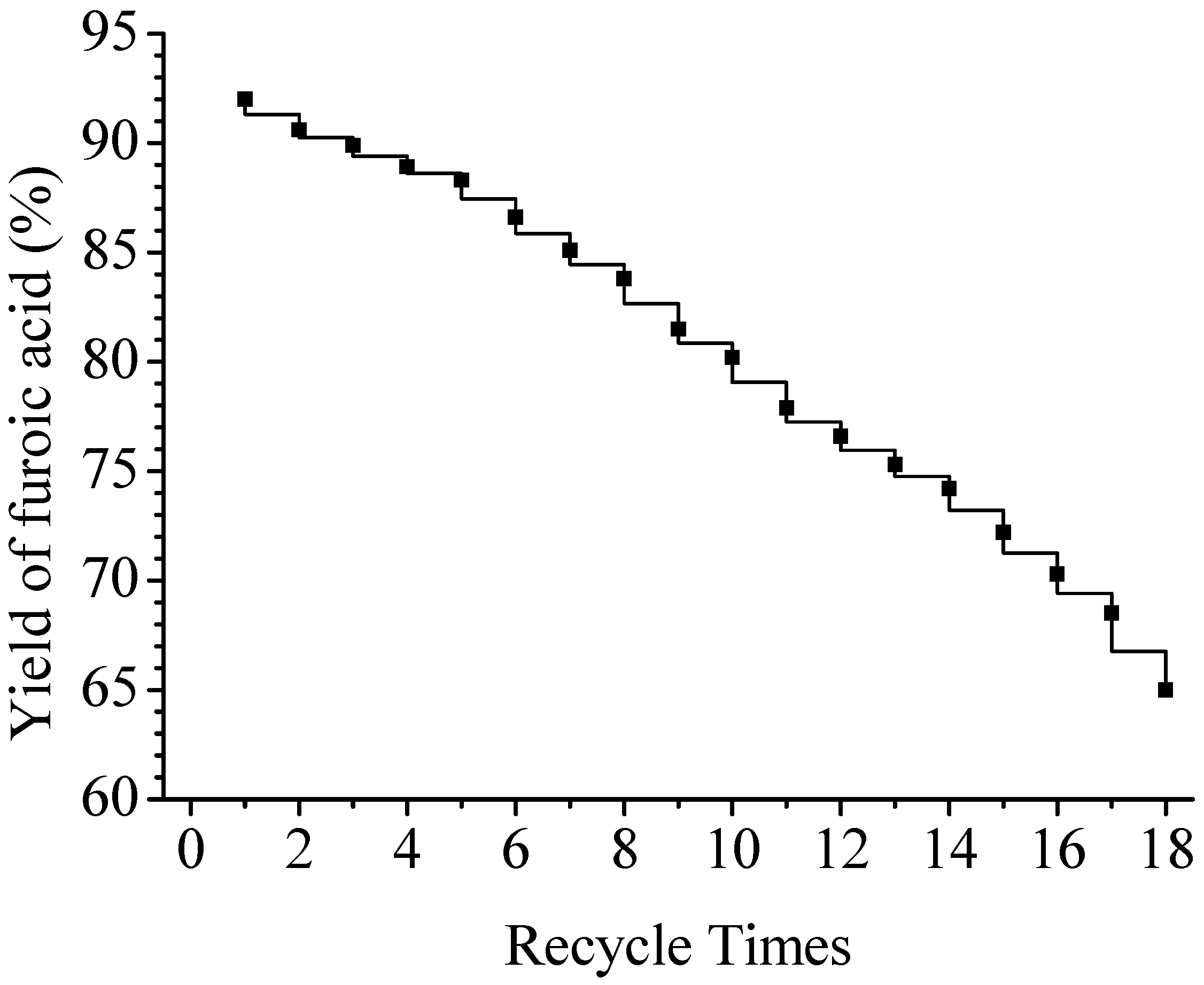
Application of the catalysts
| Aldehyde | Catalyst | Yield of carboxylic acid/% |
|---|---|---|
| Furfural | Ag2O/CuO | 92.0 |
| Furfural | CuO | 86.6 |
| Benzaldehyde | Ag2O/CuO | 74.5 |
| Benzaldehyde | CuO | 66.0 |
| 4-OH-3-MeO-benzaldehyde | Ag2O/CuO | 81.3* |
| 4-OH-3-MeO-benzaldehyde | CuO | 76.0* |
| 4-Nitro-benzaldehyde | Ag2O/CuO | 78.6** |
| 4-Nitro-benzaldehyde | CuO | 71.5** |
| 4-MeO-benzaldehyde | Ag2O/CuO | 73.4 |
| 4-MeO-benzaldehyde | CuO | 64.2 |
Conclusions
Experimental
General
Preparation of Ag2O/CuO
Oxidation of aldehydes
References and Notes
- Zanka, A. A simple and highly practical oxidation of primary alcohols to acids mediated by 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO). Chem. Pharm. Bull. 2003, 51, 888–889. [Google Scholar] [CrossRef]
- Babu, B.R.; Balasubramaniam, K.K. Simple and facile oxidation of aldehydes to carboxylic acids. Org. Prep. Proced. Int. 1994, 26, 123–125. [Google Scholar] [CrossRef]
- Dalcanale, E.; Montanari, F. Selective oxidation of aldehydes to carboxylic acids with sodium chlorite-hydrogen peroxide. J. Org. Chem. 1986, 51, 567–569. [Google Scholar] [CrossRef]
- Bal, B.S.; Childers, W.E.; Pinnick, H.W. Oxidation of α,β-unsaturated aldehydes. Tetrahedron 1981, 37, 2091–2096. [Google Scholar] [CrossRef]
- Bayle, J.P.; Perez, F.; Courtieu, J. Oxidation of aldehydes by sodium chlorite. Bull. Soc. Chim. Fr. 1990, 4, 565–567. [Google Scholar]
- Kumaraswamy, G.; Jena, N.; Sastry, M.N.V.; Kumar, B.A. An expeditious, practical large scale synthesis of 4-amino-2-chloro-6,7-dimethoxyquinazoline. Org. Prep. Proced. Int. 2004, 36, 341–346. [Google Scholar] [CrossRef]
- Kamal, A.; Reddy, B.S.N.; Reddy, G.S.K.; Ramesh, G. Design and synthesis of C-8 linked pyrrolobenzodiazepine-naphthalimide hybrids as anti-tumor agents. Bioorg. Med. Chem. Lett. 2002, 12, 1933–1936. [Google Scholar] [CrossRef]
- Gupta, K.K.S.; Dey, S.; Gupta, S.S.; Adhikari, M.; Banerjee, A. Evidence of esterification during the oxidation of some aromatic aldehydes by chromium(VI) in acid medium and the mechanism of the oxidation process. Tetrahedron 1990, 46, 2431–2444. [Google Scholar] [CrossRef]
- Hurd, C.D.; Garrett, J.W.; Osborne, E.N. Furan reactions. IV. Furoic acid from furfural. J. Am. Chem. Soc. 1933, 55, 1082–1084. [Google Scholar] [CrossRef]
- Salehi, P.; Firouzabadi, H.; Farrokhi, A.; Gholizadeh, M. Solvent-free oxidations of alcohols, oximes, aldehydes and cyclic acetals by pyridinium chlorochromate. Synthesis 2001, 15, 2273–2276. [Google Scholar]
- Chaubey, G.S.; Das, S.; Mahanti, M.K. Kinetics of the oxidation of heterocyclic aldehydes by quinolinium dichromate. Bull. Chem. Soc. Jpn. 2002, 75, 2215–2220. [Google Scholar] [CrossRef]
- Balasubramanian, K.; Prathiba, V. Quinolinium dichromate - a new reagent for oxidation of alcohols. Indian J. Chem. Sect. B 1986, 25, 326–327. [Google Scholar]
- Chaubey, G.S.; Kharsyntiew, B.; Mahanti, M.K. Oxidation of substituted 2-furaldehydes by quinolinium dichromate: A kinetic study. J. Phys. Org. Chem. 2004, 17, 83–87. [Google Scholar] [CrossRef]
- Takemoto, T.; Yasuda, K.; Ley, S.V. Solid-supported reagents for the oxidation of aldehydes to carboxylic acids. Syn. Lett. 2001, 10, 1555–1556. [Google Scholar]
- Zhivich, A.B.; Myznikov, Y.E.; Koldobskii, G.I.; Ostrovskii, V.A. XXV. Tetrazolium salts - new phase-transfer catalysts. J. Gen. Chem. USSR (Engl.Transl.) 1988, 58, 1701–1708. [Google Scholar]
- Jursic, B. Surfactant-assisted permanganate oxidation of aromatic compounds. Can. J. Chem. 1989, 67, 1381–1383. [Google Scholar] [CrossRef]
- Heaney, H. Newbold; Newbold, A.J. The oxidation of aromatic aldehydes by magnesium monoperoxyphthalate and urea-hydrogen peroxide. Tetrahedron Lett. 2001, 42, 6607–6610. [Google Scholar] [CrossRef]
- Sato, K.; Hyodo, M.; Takagi, J.; Aoki, M.; Noyori, R. Hydrogen peroxide oxidation of aldehydes to carboxylic acids: an organic solvent-, halide- and metal-free procedure. Tetrahedron Lett. 2000, 41, 1439–1442. [Google Scholar] [CrossRef]
- Vocanson, F.; Guo, Y.P.; Namy, J.L.; Kagan, H.B. Dioxygen oxidation of alcohols and aldehydes over a cerium dioxide-ruthenium system. Synth. Commun. 1998, 28, 2577–2582. [Google Scholar] [CrossRef]
- Harrison, R.J.; Moyle, M. 2-Furoic acid. Org. Syn. 1956, 36, 36. [Google Scholar] [CrossRef]
- Chakraborty, T.K.; Chandrasekaran, S. Facile oxidation of aldehydes to carboxylic acids with chromium(V) reagents. Synth. Commun. 1980, 10, 951–956. [Google Scholar] [CrossRef]
- Kharnaior, G.G.; Chaubey, G.S.; Mahanti, M.K. Kinetics of oxidation of 2-furfural by quinolinium dichromate. Oxid. Commun. 2001, 24, 377–381. [Google Scholar]
- Moore, J.A.; Partain, E.M., III. Oxidation of furfuraldehydes with sodium chlorite. Org. Prep. Proced. Int. 1985, 17, 203–205. [Google Scholar] [CrossRef]
- Pan, Z.Z.; Sha, Y.W. Effect and regeneration of heterogeneous palladium/charcoal catalysts poisoned by tetramethylthiourea-quinoline in the stereoselective hydrogenation of methacycline to α-doxycycline. Appl. Catal. A-Gen. 2003, 252, 347–352. [Google Scholar] [CrossRef]
- Sample availability: Contact the authors.
© 2008 by MDPI (http://www.mdpi.org) Reproduction is permitted for noncommercial purposes.
Share and Cite
Tian, Q.; Shi, D.; Sha, Y. CuO and Ag2O/CuO Catalyzed Oxidation of Aldehydes to the Corresponding Carboxylic Acids by Molecular Oxygen. Molecules 2008, 13, 948-957. https://doi.org/10.3390/molecules13040948
Tian Q, Shi D, Sha Y. CuO and Ag2O/CuO Catalyzed Oxidation of Aldehydes to the Corresponding Carboxylic Acids by Molecular Oxygen. Molecules. 2008; 13(4):948-957. https://doi.org/10.3390/molecules13040948
Chicago/Turabian StyleTian, Qingyong, Daxin Shi, and Yaowu Sha. 2008. "CuO and Ag2O/CuO Catalyzed Oxidation of Aldehydes to the Corresponding Carboxylic Acids by Molecular Oxygen" Molecules 13, no. 4: 948-957. https://doi.org/10.3390/molecules13040948
APA StyleTian, Q., Shi, D., & Sha, Y. (2008). CuO and Ag2O/CuO Catalyzed Oxidation of Aldehydes to the Corresponding Carboxylic Acids by Molecular Oxygen. Molecules, 13(4), 948-957. https://doi.org/10.3390/molecules13040948




