Synthesis, Antibacterial, Antifungal and Antiviral Activity Evaluation of Some New bis-Schiff Bases of Isatin and Their Derivatives
Abstract
:Introduction
Results and Discussion
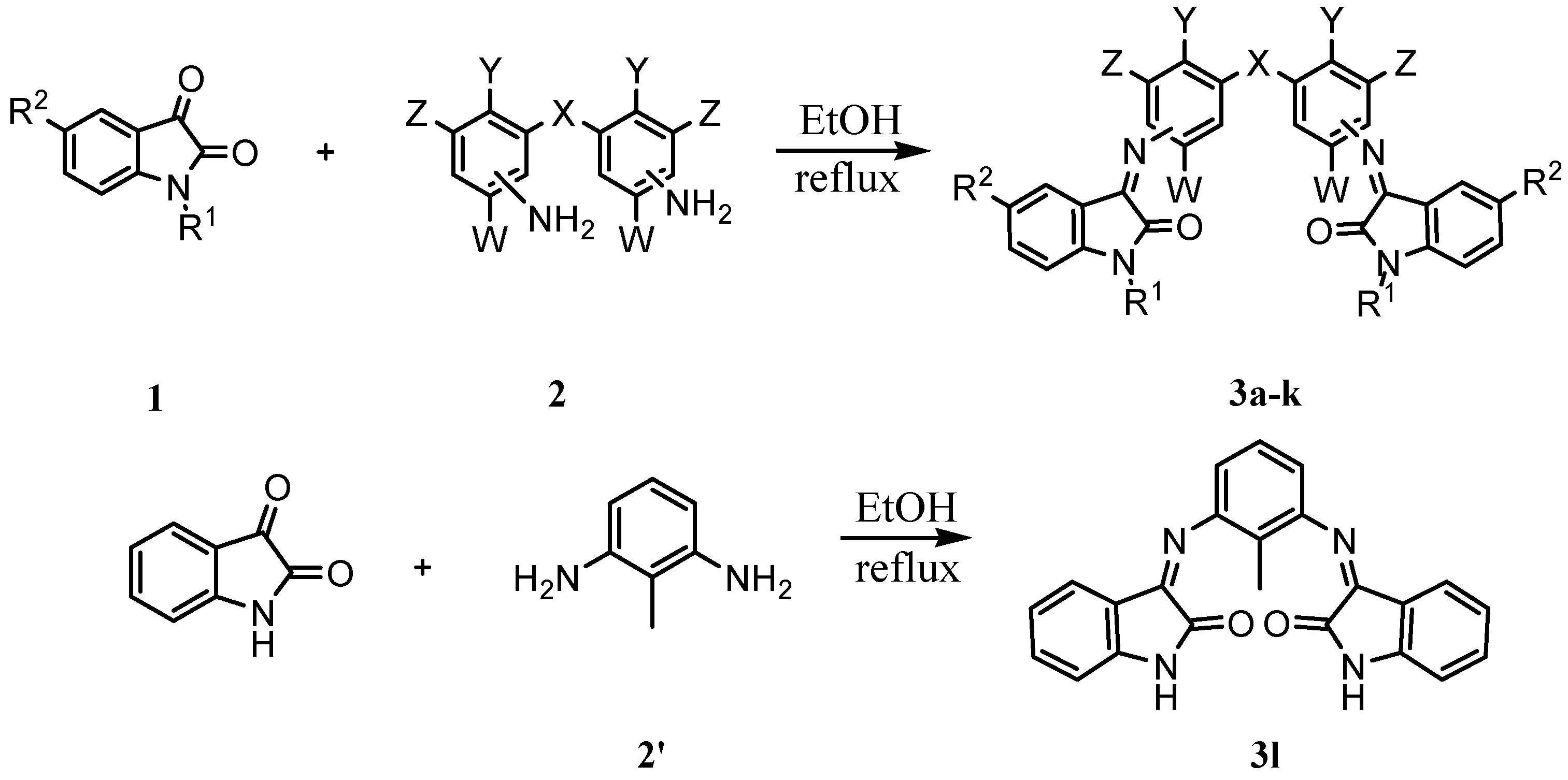
| Compd. | X | Y | Z | W | R1 | R2 | Position of C=N relative to X | m.p. (˚C) | color | time(h) | Yield (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 3a | CH2 | H | H | H | H | H | 3,3΄ | >260 | ochre | 17 | 77 |
| 3b | CH2 | H | H | H | H | H | 3,4΄ | 248-250 | light ochre | 20 | 70 |
| 3c | CH2 | H | H | H | H | F | 4,4΄ | >260 | ochre | 0.5 | 99 |
| 3d | CH2 | H | H | H | H | F | 3,3΄ | >260 | light brown | 5 | 68 |
| 3e | CH2 | Cl | Et | Et | H | H | 4,4΄ | >260 | orange-yellow | 15 | 74 |
| 3f | CH2 | H | H | H | Bn | H | 4,4΄ | 180-182 | orange | 8 | 98 |
| 3g | CH2 | H | H | H | Bn | H | 3,3΄ | 204-206 | oval-yellow | 3 | 82 |
| 3h | O | H | H | H | H | H | 3,4΄ | >260 | canary-yellow | 28 | 91 |
| 3i | O | H | H | H | H | F | 4,4΄ | >260 | brown-yellow | 0.5 | 87 |
| 3j | O | H | H | H | Bn | H | 4,4΄ | 204-206 | dark orange | 1 | 88 |
| 3k | CO | H | H | H | H | H | 4,4΄ | 242-244 | yellow | 0.5 | 70 |
| 3l | - | - | - | - | - | - | - | >260 | brick-red | 19 | 80 |
| Sample CIP | Antimicrobial activity (MIC) (μg/mL) | |||||||
|---|---|---|---|---|---|---|---|---|
| S. cerevisiae (28383)a | S. aureus (4.83) | C. albicans (1180-79) | E. coli (54127) | |||||
| 3a | <50 | <50 | <50 | <50 | ||||
| 3b | <50 | <50 | <50 | <50 | ||||
| 3c | <50 | <50 | <50 | <50 | ||||
| 3d | <50 | <50 | <50 | <50 | ||||
| 3e | <50 | <50 | <50 | <50 | ||||
| 3f | <50 | <50 | <50 | <50 | ||||
| 3g | <50 | <50 | <50 | <50 | ||||
| 3h | <50 | <50 | <50 | <50 | ||||
| 3i | <50 | <50 | <50 | <50 | ||||
| 3j | <50 | <50 | <50 | <50 | ||||
| 3k | <50 | <50 | <50 | <50 | ||||
| 3l | <100 | <100 | <100 | <100 | ||||
| compound | Minimum cytotoxic concentration(µg/mL)* | ||
|---|---|---|---|
| HEL | Vero | HeLa | |
| 3a | 80 | 80 | ≥16 |
| 3b | 80 | 80 | ≥16 |
| 3c | 16 | ≥16 | 80 |
| 3d | 80 | 80 | 80 |
| 3e | 80 | 16 | 16 |
| 3f | 40 | 40 | 200 |
| 3g | 200 | 200 | 200 |
| 3h | 400 | 400 | ≥16 |
| 3i | 8 | 40 | ≥16 |
| 3j | 400 | ≥80 | 80 |
| 3k | 80 | ≥16 | 80 |
| 3l | 400 | 400 | ≥400 |
| Brivudin | >500 | 500 | >500 |
| Ribavirin | >500 | >500 | >500 |
| Acyclovir | >500 | - | - |
| Ganciclovir | >100 | - | - |
| (S)-DHPA | - | 500 | >500 |
| Compound | Minimum virus-inhibitory concentration* (µg/mL) | ||||||
| HEL | |||||||
| Herpes simplex virus-1(KOS) | Herpes simplex virus-2(G) | Vaccinia virus | Vesicular stomatitis virus | Herpes simplex virus-1 KOS ACVr (TK-) | |||
| 3a | >16 | >16 | >16 | >16 | >16 | ||
| 3b | >16 | >16 | >16 | >16 | >16 | ||
| 3c | >3.2 | >3.2 | >3.2 | >3.2 | >3.2 | ||
| 3d | >16 | >16 | >16 | >16 | >16 | ||
| 3e | >16 | >16 | 9.6 | >16 | >16 | ||
| 3f | >8 | >8 | >8 | >8 | >8 | ||
| 3g | >40 | >40 | >40 | >40 | >40 | ||
| 3h | >80 | >80 | >80 | >80 | >80 | ||
| 3i | >1.6 | >1.6 | >1.6 | >1.6 | >1.6 | ||
| 3j | >80 | >80 | >80 | >80 | >80 | ||
| 3k | >16 | >16 | >16 | >16 | >16 | ||
| 3l | >80 | >80 | >80 | >80 | 16 | ||
| Brivudin | 0.16 | >500 | 60 | >500 | 500 | ||
| Ribavirin | 500 | >500 | 300 | >500 | >500 | ||
| Acyclovir | 2.4 | 20 | >500 | >500 | 300 | ||
| Ganciclovir | 0.48 | 4 | >100 | >100 | 12 | ||
| (S)-DHPA | - | - | - | - | - | ||
| Compound | Vero | ||||||
| Para-influenza-3 virus | Reovirus-1 | Sindbis virus | Coxsackie virus B4 | PuntaToro virus | |||
| 3a | >16 | >16 | >16 | >16 | >16 | ||
| 3b | >16 | >16 | >16 | >16 | >16 | ||
| 3c | ≥16 | >16 | >16 | >16 | >16 | ||
| 3d | >16 | >16 | >16 | >16 | >16 | ||
| 3e | >3.2 | >3.2 | >3.2 | >3.2 | >3.2 | ||
| 3f | >8 | >8 | >8 | >8 | >8 | ||
| 3g | >40 | >40 | >40 | >40 | >40 | ||
| 3h | >80 | >80 | >80 | >80 | >80 | ||
| 3i | >8 | >8 | >8 | >8 | >8 | ||
| 3j | >80 | >80 | >80 | >80 | >80 | ||
| 3k | >16 | >16 | >16 | >16 | >16(80) | ||
| 3l | >80 | >80 | >80 | >80 | >80 | ||
| Brivudin | >100 | >100 | >100 | >100 | >100 | ||
| Ribavirin | 300 | 300 | 300 | >500 | 60 | ||
| Acyclovir | - | - | - | - | - | ||
| Ganciclovir | - | - | - | - | - | ||
| (S)-DHPA | >100 | 300 | >100 | >100 | |||
| Compound | Minimum virus-inhibitory concentration* (µg/mL) | ||||||
| HeLa | |||||||
| Vesicular stomatitis virus | Coxsackie virus B4 | Respiratory syncytial virus | |||||
| 3a | 9.6 | >16 | >16 | ||||
| 3b | 3.2 | >16 | >16 | ||||
| 3c | >16 | >16 | >16 | ||||
| 3d | 9.6 | >16 | >16 | ||||
| 3e | >3.2 | >3.2 | >3.2 | ||||
| 3f | >40 | >40 | >40 | ||||
| 3g | >40 | >40 | >40 | ||||
| 3h | >16 | >16 | >16 | ||||
| 3i | >16 | >16 | >16 | ||||
| 3j | >16 | >16 | >16 | ||||
| 3k | 48 | >16 | >16 | ||||
| 3l | >400 | >400 | >400 | ||||
| Brivudin | >500 | >500 | >500 | ||||
| Ribavirin | 60 | >500 | 60 | ||||
| Acyclovir | - | - | - | ||||
| Ganciclovir | - | - | - | ||||
| (S)-DHPA | 500 | >500 | >500 | ||||
Experimental
General
General procedure for preparation of bis-Schiff bases of isatin and its derivatives
Acknowledgements
References
- Somogyi, L. Transformation of Isatin 3-acylhydrazones under acetylating conditions: Synthesis and structure elucidation of 1,5′-disubstituted 3′-acetylspiro[oxindole-3,2′-[1,3,4]oxadiazolines]. Bull. Chem. Soc. Jpn. 2001, 74, 873–881. [Google Scholar]
- Da Silva, J. F. M.; Garden, S. J.; Pinto, A. C. The Chemistry of Isatins: a Review from 1975 to 1999. J. Braz. Chem. Soc. 2001, 12(3), 273–324. [Google Scholar]
- Elliott, J.; Gardner, D. L. Proline determination with isatin, in the presence of amino acids. Anal. Biochem. 1976, 70, 268–273. [Google Scholar] [CrossRef]
- Palfi, G.; Palfi, Z. Determination of viality of pollen on the basis of its amino acids content. Mydica 1982, 27, 107. [Google Scholar]
- Eriksen, A. B. Quantitative determination of the amino acid proline by glass fiber paper chromatography. Medd. Nor. Inst. Skogforsk. 1976, 32, 389–404. [Google Scholar]
- Shah, A.; Rahman, S. S.; de Biasi, V.; Camilleri, P. Development of Colorimetric Method for the Detection of Amines Bound to Solid Support. Anal. Commun. 1997, 34(11), 325–328. [Google Scholar]
- Chiyanzu, I.; Clarkson, C.; Smith, P. J.; Lehman, J.; Gut, J.; Rosenthal, P. J.; Chibale, K. Design, synthesis and anti-plasmodial evaluation in vitro of new 4-aminoquinoline isatin derivatives. Bioorg. Med. Chem. 2005, 13, 3249–3261. [Google Scholar]
- Pandeya, S. N.; Sriram, D. Synthesis and screening for antibacterial activity of Schiff’s and Mannich bases of Isatin and its derivatives. Acta. Pharm. Turc. 1998, 40, 33–38. [Google Scholar]
- Sarangapani, M.; Reddy, V. M. Synthesis and antimicrobial activity of 1-[(N,N-disubstituted amino)methyl]-3-[(2-phenyl-3,4-dihydro-4-oxoquinazoline-3-yl]indole-2-one. Indian J. Hetero-cycl. Chem. 1994, 3, 257–260. [Google Scholar]
- Varma, R. S.; Nobles, W. L. Antiviral, antibacterial, and antifungal activities of isatin N-Mannich bases. J. Pharm. Sci. 1975, 64, 881–882. [Google Scholar]
- Sridhar, S. K.; Pandeya, S. N.; Stables, J. P.; Ramesh, A. The Wide Pharmacological Versatility of Semicarbazones, Thiosemicarbazones and Their Metal Complexes. Eur. J. Med. Chem. 2002, 16, 129–132. [Google Scholar]
- Varma, M.; Pandeya, S, N.; Singh, K. N.; Stables, J. P. Anticonvulsant activity of Schiff bases of isatin derivatives. Acta Pharm. 2004, 54, 49–56. [Google Scholar]
- Pandeya, S. N.; Yogeeswari, P.; Sriram, D.; De Clercq, E.; Pannecouque, C.; Witvrouw, M. Synthesis and Screening for Anti-HIV Activity of Some N-Mannich Bases of Isatin Derivatives. Chemotherapy 1999, 45, 192–196. [Google Scholar]
- Pandeya, S. N.; Sriram, D.; Nath, G.; De Clercq, E. Synthesis, antibacterial, antifungal and anti-HIV activities of Norfloxacin Mannich bases. Eur. J. Med. Chem. 2000, 35, 249–255. [Google Scholar]
- Pandeya, S. N.; Sriram, D.; Nath, G.; De Clercq, E. Synthesis, antibacterial, antifungal and anti-HIV evaluation of Schiff and Mannich bases of isatin and its derivatives with triazole. Arzneim Forsch./Drug Res. 2000, 50, 55–59. [Google Scholar]
- Pandeya, S. N.; Yogeeswari, P.; Sriram, D.; Nath, G. Synthesis and antimicrobial activity of N-Mannich bases of 3-[N′-sulphadooximino]isatin and its methyl derivatives. Bull. Chim. Farm. 1998, 137, 321–324. [Google Scholar]
- Pandeya, S. N.; Sriram, D.; Nath, G.; De Clercq, E. Synthesis and antimicrobial activity of Schiff and Mannich bases of isatin and its derivatives with pyrimidine. Farmaco 1999, 54, 624–628. [Google Scholar]
- Pandeya, S. N.; Sriram, D.; Nath, G.; De Clercq, E. Synthesis, antibacterial, antifungal and anti-HIV activity of Schiff and Mannich bases of isatin with N-[6-chlorobenzthiazole-2-yl] thiosemicarbazid. Indian J. Pharm. Sci. 1999, 61, 358–361. [Google Scholar]
- Pandeya, S. N.; Sriram, D.; Nath, G.; De Clercq, E. Synthesis, antibacterial, antifungal and anti-HIV evaluation of Schiff and Mannich bases of isatin derivatives with 3-amino-2-methylmercapto quinazolin-4(3H)-one. Pharm. Acta Helv. 1999, 74, 11–17. [Google Scholar] [CrossRef]
- Pandeya, S. N.; Sriram, D.; Nath, G.; De Clercq, E. Synthesis, antibacterial, antifungal and anti-HIV activities of Schiff and Mannich bases derived from isatin derivatives and N-[4-(4′-chlorophenyl)thiazol-2-yl] thiosemicarbazide. Eur. J. Pharm. Sci. 1999, 9, 25–31. [Google Scholar]
- Singh, S. P.; Shukla, S. K.; Awasthi, L. P. Synthesis of some 3-(4′-nitrobenzoylhydrazono)-2-indolinones as a potential antiviral agents. Curr. Sci. 1983, 52, 766–769. [Google Scholar]
- Marcu, G. Chimica complecsilor coordinative; Ed. Academiei Bucuresti: Bucarest, 1984; pp. 44–73. [Google Scholar]
- Cerchiaro, G.; Micke, G. A.; Tavares, M. F. M.; Ferriera, A. M. D. C. Kinetic studies of carbohydrate oxidation catalyzed by novel isatin–Schiff base copper(II) complexes. J. Mol. Catal. A: Chem 2004, 221, 29–39. [Google Scholar] [CrossRef]
- Takeuchi, T.; Bottcher, A.; Quezada, C. M.; Simon, M. I.; Meade, T. J.; Gray, H. B. Selective Inhibition of Human -Thrombin by Cobalt(III) Schiff Base Complexes. J. Am. Chem. Soc. 1998, 120, 8555–8556. [Google Scholar]
- Bacchi, A.; Carcelli, M.; Pelagatti, P.; Pelizzi, G.; Rodriguez-Arguelles, M. C.; Rogolino, D.; Solinas, C.; Zani, F. Antimicrobial and mutagenic properties of organotin(IV) complexes with isatin and N-alkylisatin bisthiocarbonohydrazones. J. Inorg. Biochem. 2005, 99, 397–408. [Google Scholar] [CrossRef]
- Cerchiaro, G.; Aquilano, K.; Filomeni, G.; Rotilio, G.; Ciriolo, M. R.; Ferriera, A. M. D. C. Isatin-Schiff base copper(II) complexes and their influence on cellular viability. J. Inorg. Biochem. 2005, 99, 1433–1440. [Google Scholar] [CrossRef]
- Garden, S. J.; Torres, J. C.; da Silva, L. E.; Pinto, A. C. A convenient methodlogy for the N-alkylation of isatin compounds. Synth. Commun. 1998, 28, 1679–1689. [Google Scholar]
- Dei Cas, E.; Dujardin, L.; Ribeiro Pinto, M. E.; Ajana, F.; Fruit, J.; Poulain, D.; Camus, D.; Vernes, A.; Francois, N. Growth was measured in vitro using a liquid-phase method according to NCCLS guidelines from the American Society of Microbiology for 24 hours using various concentrations of drugs. Mycoses 1991, 34, 167–172. [Google Scholar]
- De Clercq, E.; Descamps, J.; Verhelst, G.; Walker, R.T.; Jones, A.S.; Torrence, P. F.; Shugar, D. Relative potencies of different anti-herpes agents in the topical treatment of cutaneous herpes simples virus infection of athymic nude mice. J. Infect. Dis. 1980, 141, 563–574. [Google Scholar]
- De Clercq, E. Antiviral and antimetabolic activities of neplanocins. Antimicrob. Agents Chemother. 1985, 28, 84–89. [Google Scholar]
- De Clercq, E.; Holý, A.; Rosenberg, I.; Sakuma, T.; Balzarini, J.; Maudgal, P.C. A novel selective broad-spectrum anti-DNA virus agent. Nature 1986, 323, 464–467. [Google Scholar]
- Sample availability: Contact the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Jarrahpour, A.; Khalili, D.; De Clercq, E.; Salmi, C.; Brunel, J.M. Synthesis, Antibacterial, Antifungal and Antiviral Activity Evaluation of Some New bis-Schiff Bases of Isatin and Their Derivatives. Molecules 2007, 12, 1720-1730. https://doi.org/10.3390/12081720
Jarrahpour A, Khalili D, De Clercq E, Salmi C, Brunel JM. Synthesis, Antibacterial, Antifungal and Antiviral Activity Evaluation of Some New bis-Schiff Bases of Isatin and Their Derivatives. Molecules. 2007; 12(8):1720-1730. https://doi.org/10.3390/12081720
Chicago/Turabian StyleJarrahpour, Aliasghar, Dariush Khalili, Erik De Clercq, Chanaz Salmi, and Jean Michel Brunel. 2007. "Synthesis, Antibacterial, Antifungal and Antiviral Activity Evaluation of Some New bis-Schiff Bases of Isatin and Their Derivatives" Molecules 12, no. 8: 1720-1730. https://doi.org/10.3390/12081720
APA StyleJarrahpour, A., Khalili, D., De Clercq, E., Salmi, C., & Brunel, J. M. (2007). Synthesis, Antibacterial, Antifungal and Antiviral Activity Evaluation of Some New bis-Schiff Bases of Isatin and Their Derivatives. Molecules, 12(8), 1720-1730. https://doi.org/10.3390/12081720




