Phenolic Molecules in Virgin Olive Oils: a Survey of Their Sensory Properties, Health Effects, Antioxidant Activity and Analytical Methods. An Overview of the Last Decade Alessandra
Abstract
:Phenolic molecules in virgin olive oil
| Compound | Substituent (MW) | Structure |
|---|---|---|
| Benzoic and derivatives acids | 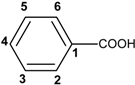 | |
| 3-Hydroxybenzoic acid | 3-OH (138) | |
| p- Hydroxybenzoic acid | 4-OH (138) | |
| 3,4-Dihydroxybenzoic acid | 3,4-OH (154) | |
| Gentisic acid | 2,5-OH (154) | |
| Vanillic acid | 3-OCH3, 4-OH (168) | |
| Gallic acid | 3,4,5-OH (170) | |
| Syringic acid | 3,5-OCH3, 4-OH (198) | |
| Cinnamic acids and derivatives | 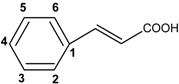 | |
| o-Coumaric acid | 2-OH (164) | |
| p-Coumaric acid | 4-OH (164) | |
| Caffeic Acid | 3,4-OH (180) | |
| Ferulic Acid | 3-OCH3, 4-OH (194) | |
| Sinapinic Acid | 3,5-OCH3, 4-OH (224) | |
| Phenyl ethyl alcohols | 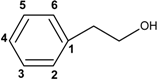 | |
| Tyrosol [(p-hydroxyphenyl)ethanol] or p-HPEA | 4-OH (138) | |
| Hydroxytyrosol [(3,4-dihydroxyphenyl)ethanol] or 3,4-DHPEA | 3,4-OH (154) | |
| Other phenolic acids and derivatives | 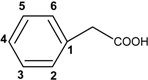 | |
| p-Hydroxyphenylacetic acid | 4-OH (152) | |
| 3,4-Dihydroxyphenylacetic acid | 3,4-OH (168) | |
| 4-Hydroxy-3-methoxyphenylacetic acid | 3-OCH3, 4-OH (182) | |
| 3-(3,4-Dihydroxyphenyl)propanoic acid | (182) | 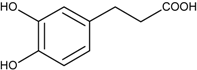 |
| Dialdehydic forms of secoiridoids | 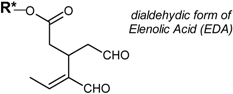 | |
| Decarboxymethyloleuropein aglycon (3,4-DHPEA-EDA) | R1-OH (304) | |
| Decarboxymethyl ligstroside aglycon (p-HPEA-EDA) | R1-H (320) | |
| Compound | Substituent (MW) | |
| Secoiridoid Aglycons | ||
| Oleuropein aglycon or 3,4-DHPEA-EA | R1-OH (378) | |
| Ligstroside aglycon or p-HPEA-EA | R1-H (362) | |
| Aldehydic form of oleuropein aglycon | R1-OH (378) | |
| Aldehydic form ligstroside aglycon | R1-H (362) | |
| Structure | ||
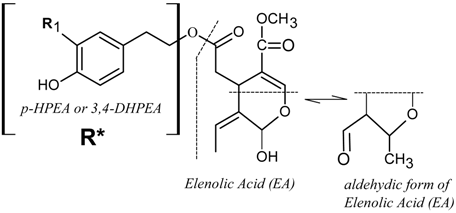 | ||
| Compound | Substituent (MW) | Structure |
| Flavonols | 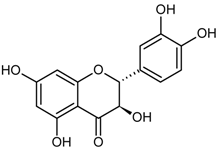 | |
| (+)-Taxifolin | (304) | |
| Flavones | 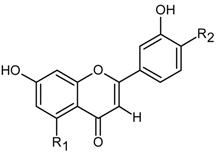 | |
| Apigenin | R1-OH, R2-H (270) | |
| Luteolin | R1-OH, R2-OH (286) | |
| Lignans | 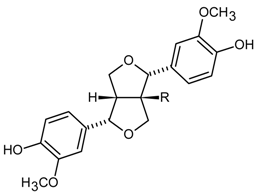 | |
| (+)-Pinoresinol | R-H (358) | |
| (+)-1-Acetoxypinoresinol | R-OCOCH3 (416) | |
| (+)-1-Hydroxypinoresinol | R-OH (374) | |
| Hydroxyisochromans | 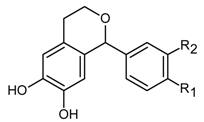 | |
| 1-Phenyl-6,7-dihydroxyisochroman | R1,R2-H (242) | |
| 1-(3’-Methoxy-4’-hydroxy)phenyl-6,7-dihydroxyisochroman | R1-OH, R2-OCH3 (288) | |
Why are the phenolic compounds present in virgin olive oil so important? Why is their determination so interesting and difficult?
- –
- The levels and chemical structure of antioxidant phenols in different plant foods, aromatic plants and various plant materials.
- –
- The probable role of plant phenols in the prevention of various diseases associated with oxidative stress such as cardiovascular and neurodegenerative diseases and cancer.
- –
- The ability of plant phenols to modulate the activity of enzymes, a biological action not yet understood.
- –
- The ability of certain classes of plant phenols such as flavonoids (also called polyphenols) to bind to proteins. Flavonol–protein binding, such as binding to cellular receptors and transporters, involves mechanisms which are not related to their direct activity as antioxidants.
- –
- The stabilization of edible oils, protection from formation of off-flavors and stabilization of flavours.
- –
- The preparation of food supplements.
Health aspects linked to phenols in VOO
- 1)
- 2)
- 3)
- inhibit platelet-induced aggregation [81];
- 4)
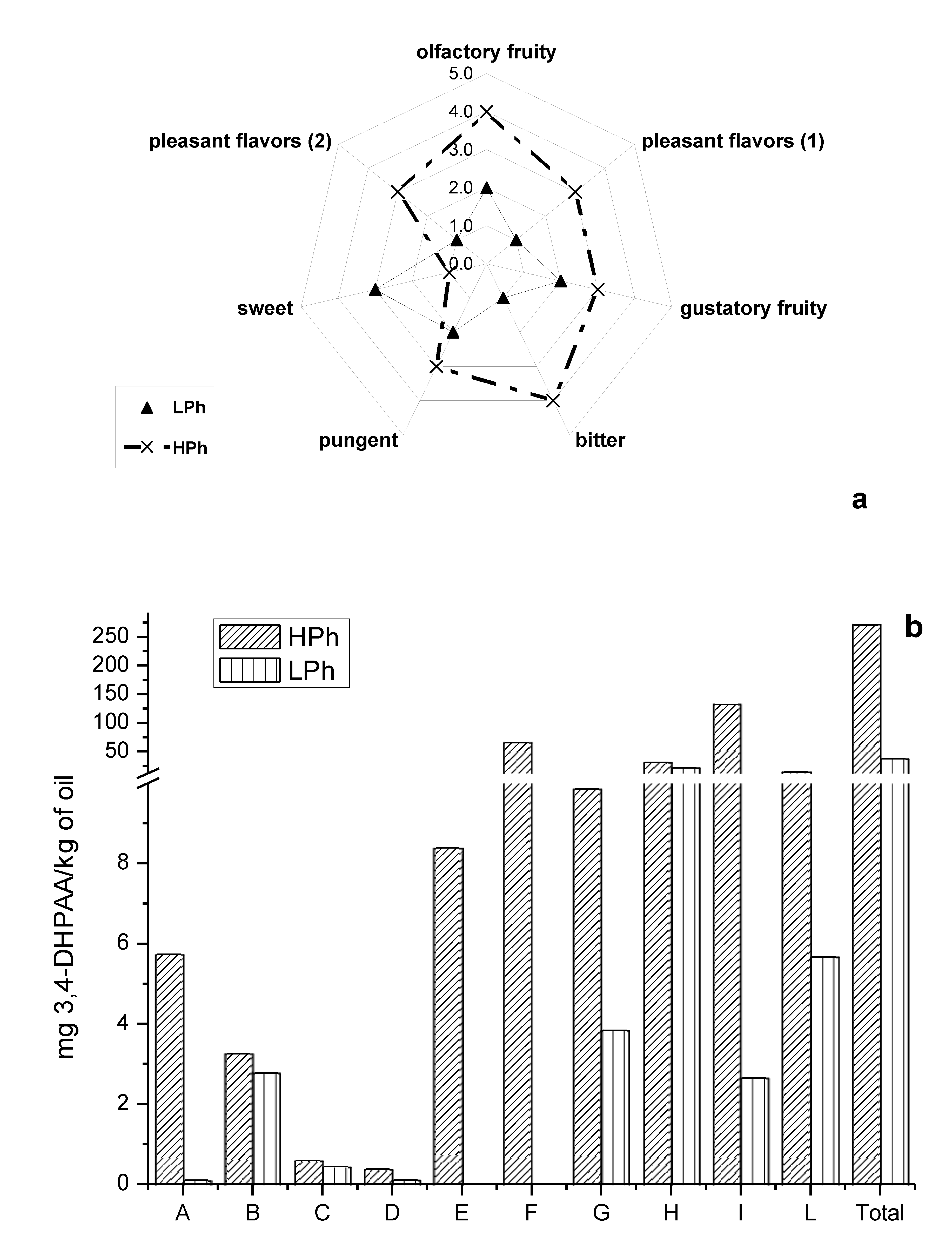
Sensory properties elicited by phenols in VOO
New analytical approaches to characterization of the phenolic profile and applied studies during the last decade
| Time of analysis | Mobile phase | Stationary phase | Type of elution | Extraction System | Detection System | Observations | References | |
|---|---|---|---|---|---|---|---|---|
| 120 min | A: H2O B: MeOH | Spherisorb ODS 25 cm x 4.6 mm i.d.; 10µm | Gradient | Combination between LLE [42] and SPE | UV in HPLC. MS off-line | Separation of the polar fraction of VOO in two parts. Antioxidant activity assessment | [175] | |
| 93 min | A: H2O + CH3COOH 0.5% B: MeCN | Spherisorb ODS 2, 25 cm x 4.6 mm i.d. | Gradient | LLE with methanol/water (80:20 v/v) | UV; MS (ESI) in positive ion mode | Flavonoids such as luteolin and apigenin were detected as phenolic components of VOO | [41] | |
| 25 min | H2O:CH3CN (82:18 v/v) + CH3COOH 0.02% | Nucleosil ODS, 25 cm x 2.1 mm or 25 cm x 1.1 mm i.d. 5 µm | Isocratic | LLE with buffer; SPE with phenyl cartridges (acidification) | UV, fluorescence, MS, MS/MS HPLC-APCI (negative ion mode) | Phenolic acids | [30] | |
| 60 min approx. | A: H2O + HCOOH 0.045% B: MeOH + HCOOH 0.045% | Nucleosil ODS, 25 cm x 2.1 mm i.d. 5 µm | Gradient | -Phenolic acids as Cartoni [30] -HYTY and TY: 3 g oil across cartridge phenylic | MS; MS/MS | Olives and VOOs. MS/MS using Multiple Reaction Monitoring (MRM) (high specificity and sensitivity in MS spectra) | [176] | |
| HPLC method and conditions of Cortesi et al. [177] | C18 column (RP) Alltech 25 cm x 4.6 mm i.d. | Gradient | LLE: Montedoro et al. [1] using butylated hydroxytoluene (BHT) | MS; MS/MS | Analysis of oleuropein aglycon by APCI-MS. Phenolic compound profile | [178] | ||
| HPLC method of Romani et al. [179] | Lichrosorb RP18, 25 cm x 4.6 mm i.d. 5 µm | Gradient | LLE with EtOH/water (70:30 v/v), the water was acidified with formic acid (pH 2.5) | DAD; MSD | HPLC analysis of phenolic acids, secoiridoids and flavonoids | [180] | ||
| 60 min | A: H2O + CH3COOH 2mM B: MeOH + CH3COOH 2mM | Nucleosil ODS, 25 cm x 2.1 mm i.d. 5 µm | Gradient | LLE with methanol/water (80:20 v/v), acidification and passed through a C18 cartridge | MS and MS/MS (API/MS in negative ion mode) | Identification of a new class of phenolic compounds in olive oils: hydroxy-isochromans | [35] | |
| HPLC method and conditions of Brenes et al. [44] | UV; electrochemical, fluorescence, MS. | Use of a lignan (1-acetoxypinoresinol) to authenticate Picual VOOs. Use of GC too. | [35] | |||||
| 50 min or 70 min | A: H2O + H3PO4 0.5% B: MeOH/ MeCN (50:50 v/v) | Lichrospher 100 RP18, 25 cm x 4.0 mm i.d. 5 µm | Gradient | SPE (diol-bound phase) | UV, HPLC-MS in ESI(positive ion mode) | Dialdehydic and aldehydic forms of oleuropein aglycon and ligstroside aglycon | [181] | |
| 65 min | A: H2O + CH3COOH 2% B: MeOH/ MeCN (50:50 v/v) | C18 Luna column, 25 cm x 3.0 mm i.d. 5 µm | Gradient | Comparative study of 5 extraction methods (LLE and SPE) | UV, DAD; MS | HPLC and CE methods. (HYTY, TY, oleuropein, ligstroside aglycon and decarboxymethyl oleuropein aglycon) | [182] | |
| 65 min | A: H2O + CH3COOH 2% B: EtOH | Phenomenex Luna (phenyl-hexyl)phase; 25 cm x 4.6 mm i.d. 5 µm | Isocratic | LLE with methanol/water (80:20 v/v) Montedoro et al. [1] | UV; MS (ESI in negative ion mode) | Isolation of individual polyphenols to study sensory properties | [137] | |
| 60 min approx. | A: H2O + HCOOH 0.09% B: MeOH + HCOOH 0.09% | Nucleosil ODS, 25 cm x 2.1 mm i.d. 5 µm | Gradient | Separation of phenolic compounds in two fractions after C18 cartridge. Group A: 12 g oil Group B. 3 g oil | UV; fluorescence; MS; MS/MS | Improve of extraction system of [176]. Determination of isomer of dihydroxy- and dimethoxybenzoic acids. Comparison among LOD in HPLC-UV, HPLC-FL and HPLC-MS/MS | [183] | |
| 75 min | A: H2O + CH3COOH 0.5% B: MeCN | C18 Luna column, 25 cm x 3.0 mm i.d. 5 µm | Gradient | LLE with methanol/water (60:40 v/v) | DAD; MS (ESI in negative ion mode) | Effect of olive ripening degree on the oxidative stability and organoleptic properties of olive oil | [184] | |
| HPLC method of Rotondi et al. [184]. | Gradient | LLE with methanol/water from olive oil. SLE from olive fruits | DAD; MS (ESI in positive and negative ion mode) | HPLC and CE analysis. 3 simple phenols, a secoiridoid derivative and 2 lignans | [185] | |||
| 60 min | A: H2O + 0.2% acetic acid B: MeCN | Lichrospher 100, 12.5 cm x 4.0 mm i.d. 5 µm | Gradient | LLE with methanol (500 mg of oil) | Refractive index detector; MS | TY, Vanillic acid, Lut and Apig.Squalene (with Refractive Index detector). Quantitation in 7 samples. | [45] | |
| 45 min | A: H2O + 0.2% acetic acid B: MeCN | Inertsil ODS-3, 15 cm x 4.6 mm i.d. 5 µm | Gradient | LLE with methanol/water (80:20, v:v) (45 g of oil) | UV; ESI-MS | Antioxidant activity of olive pulp and olive of Arbeq. cv | [46] | |
| 40 min | A: H2O + 0.2% acetic acid B: MeCN | C18 Luna column, 15 cm x 2.0 mm i.d. 5 µm | Gradient | Diol cartridge (3 g of oil) | UV (DAD); MS; MS/MS (QqQ) | Quantification of 23 compounds in 3 olive oils. Possible models of derived secoiridoids (nine basic models of Lig and Ol aglycons found in bibliography) | [186] | |
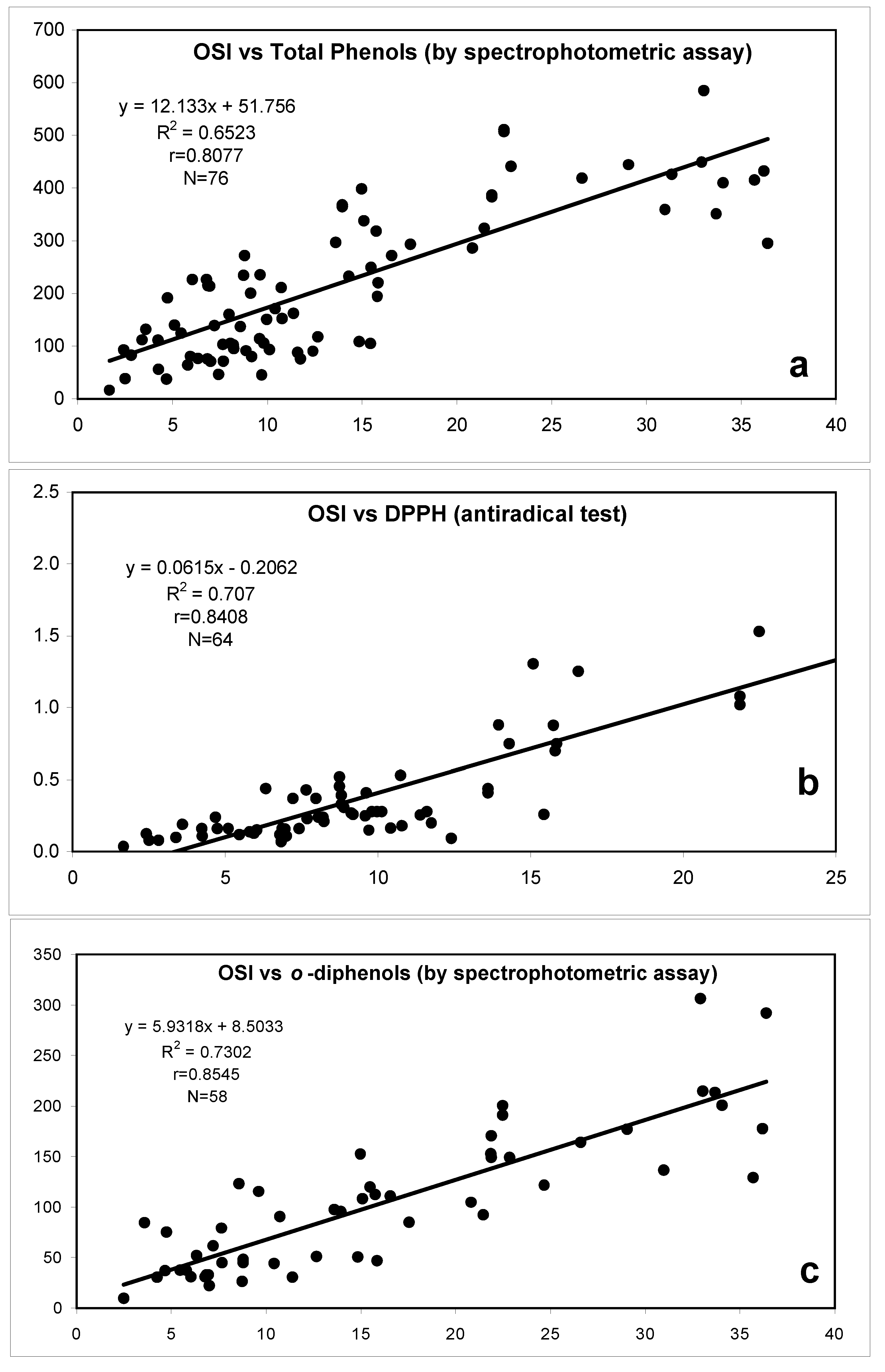
| 70 min | A: H2O + 0.5% acetic acid B: MeCN | C18 Luna column, 25 cm x 4.6 mm i.d. 5 µm | Gradient | LLE with methanol/water (60:40, v:v) (60 g of oil) | UV (DAD); MS | Isolation of several phenolic compounds and study of their antioxidants properties (DPPH, OSI and electrochemical method) | [117] | |
| 50 min | A: H2O + HCOOH (pH 3.2) B: MeCN | C18 Luna column, 25 cm x 3.0 mm i.d. 5 µm | Gradient | LLE with ethanol/water (7:3, v:v) (25 ml of oil) | UV (DAD); ESI-MS | Evaluation of lignans free and linked HYTY and TY in VOO. TLC to determine the presence of lignans. | [187] | |
| 30 min | A: H2O + 0.1% acetic acid B: MeCN | RP C18 2.1 x 100 mm, 3.5 mm particle size; XTerra MS | Gradient | SPE-Diol (60 g of oil) diluted 1:10 | ESI-TOF (TOF) | Determination of all the well-known phenolic compounds of oil and more than 25 “new” compounds | [188] | |
| Time of analysis | Mobile phase | Stationary phase | Type of elution | Extraction System | Detection System | Observations | References |
|---|---|---|---|---|---|---|---|
| 45 min | A: H2O + CH3COOH 2% (pH 3.1) B: MeOH | Erbasil C18, 15 cm x 4.6 mm i.d. | Gradient | LLE with methanol/water | UV; NMR and IR | Spectroscopic characterization of secoiridoid derivatives | [3] |
| 60 min | A: H2O + CH3COOH 0.2% B: MeOH | Spherisorb ODS 2, 25 cm x 4.6 mm i.d. | Gradient | Same as Montedoro et al. [1] | Photodiode array; MS; NMR | Simple phenols, flavonoids, secoiridoids | [44] |
| HPLC method of Montedoro et al. [1] | Column RP18 Latex; 25 cm x 4.0 mm i.d. 5 μm | Gradient | LLE with methanol (500 g of olive oil) | UV; MS (ESI) in negative and positive ion mode; NMR | Identification of lignans as major components in polar fraction of olive oil. Preparative thin-layer chromatography (PLC). | [34] | |
| HPLC method of Montedoro et al. [1,2,3] | Column RP18 Latex; 25 cm x 4.0 mm i.d. 5 μm | Gradient | LLE with absolute methanol and methanol/water (80:20 v/v) | UV; MS (ESI) in negative and positive ion mode; NMR | Use of TLC, GC, GC-MS Study of antioxidant/anticancer capacity | [191] | |
| 50 min | A: H2O + CH3COOH 3% B: MeCN:MeOH (50:50 v/v) | Lichrosphere 100 RP18, 25 cm x 4.0 mm i.d. 5 μm | Gradient | Comparative studies of LLE and SPE using diol-phase cartridges; unwanted substances washed out with hexane and hexane/ethyl acetate (90:10, v/v) | UV; DAD NMR (for ligstroside aglycon) | Phenols, flavones and lignans. Colorimetric determination of o-diphenols. GC-MS | [192] |
| 60 min | A: H2O + CH3COOH 0.5% B: MeOH/ MeCN (50:50 v/v) | Lichrospher 100 RP18, 25 cm x 4.0 mm i.d. 5 μm | Gradient | Comparative study of LLE and SPE (diol and C18-phase) | Photodiode array detector; MS, NMR. | Simple phenols, secoiridoids and lignans | [193] |
| 30 min | A: H2O + 0.1% trifluoroacetic (TFA-d) B: MeCN+ 0.1% (TFA-d) | Phenomenex RP-C18, 25 cm x 4.6 mm i.d. 5 μm | Gradient | LLE with methanol/water (80:20 v/v) (50 g of oil) | LC-SPE-NMR system | Complete characterization of 27 phenolic compounds in olive oil. 7 compounds not detected in the past | [174] |
| Instrumental Variables | Experimental Variables | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| References | λd [nm] | V [kV] | T [ºC] | i. d. [μm] | Lef [cm] | tinj [s] | Type of Buffer | [Buffer] [mM] | pH | Organic modifiers and other variables | |
| [182] | 200 | 27 | 30 | 50 | 40 | 3 s (0.5 p.s.i) | Sodium Tetraborate | 45 | 9.6 | - | |
| [215] | CZE method of Bendini et al.[182] | ||||||||||
| [185] | CZE method of Bendini et al.[182] | ||||||||||
| [31] | 210 | 25 | 25 | 75 | 50 | 8 s (0.5 p.s.i) | Sodium Tetraborate | 25 | 9.6 | - | |
| [29] | 200 | 18 | 25 | 50 | 36 | 2 s (1.5 p.s.i) | Sodium Tetraborate | 40 | 9.2 | - | |
| [32] | 210 | -25 | 25 | 75 | 50 | 8 s (0.5 p.s.i) | Sodium Tetraborate | 50 | 9.6 | 20% 2-propanol | |
| [216] | 214/250 | 25 | 25 | 75 | 100 | 8 s (0.5 p.s.i) | Sodium Tetraborate | 30 | 9.3 | ||
| [217] | 214/MS (ESI-IT) | 25 | 25 | 50 | 100 | 10 s (0.5 p.s.i) | NH4OAc | 60 | 9.5 | 5% 2-propanol Sheath liquid (60:40 v/v 2-propanol/ water and 0.1% v/v of TEA at a flow rate of 0.28 mL/h) | |
| [218] | 200/240/280/330 | 28 | 22 | 50 | 40 | 8 s (0.5 p.s.i) | Sodium Tetraborate | 45 | 9.3 | ||
| [219] | CZE method of Carrasco-Pancorbo [218] | ||||||||||
| [188] | MS (ESI-TOF) | 30 | 25 | 50 | 85 | 10 s (50 mBar) | Ammonium hydrogen carbonate | 25 | 9.0 | Sheath liquid (2-propanol/ water 50:50 v/v at a flow rate of 4 μL/min) | |
| References | Initial quantity of oil Final quantity of solvent (MeOH/H2O (50:50 v/v) ) in the extraction process (kind of extraction) | Detected compounds in olive oil | Other relevant aspects | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| [182] | 2 g → 1 mL (LLE [220]) | HYTY, TY, unidentified secoiridoids compounds | 1st paper where CE is used for the analysis of phenolic compounds from oils | ||||||||
| [215] | 2 g → 1 mL (LLE [220]) | HYTY, TY, DHPE, unidentified oleuropein aglycone derivatives | |||||||||
| [185] | 2 g → 0.5 mL (LLE [220], as modified in [184]) | HYTY, TY, VA, DOA, Ac Pin | |||||||||
| [31] | 60 g → 0.5 mL (LLE [31]) | 13 phenolic acids + taxifolin (flavanonol | Potent extraction system which permits detection of small quantities of phenolic acids | ||||||||
| [29] | 10 g → non specified (Combination of LLE-SPE [29]) | 5 phenolic acids | |||||||||
| [32] | 60 g → 0.5 mL (LLE [31]) | 13 phenolic acids + taxifolin (flavanonol | Co-electroosmotic CE | ||||||||
| [216] | 60 g → 2 mL (SPE-Diol [216]) | TY, Pin, Ac Pin, DOA, Lig Agl, HYTY, Ol Agl, EA | Use of standards obtained by semipreparative-HPLC | ||||||||
| [217] | 60 g → 2 mL (SPE-Diol [216]) | 11 phenols (simple phenols, lignans, complex phenols and EA) | 1st paper in which CZE-ESI-IT MS is used for the analysis of phenolic compounds from oils | ||||||||
| [218] | 60 g → 2 mL (SPE-Diol [216]) | 26 compounds belonging to all the different families of phenolic compounds present in olive oil | 26 compounds in less than 10 min. 1st paper in which flavonoids are detected by CE, and 1st “multicomponent” method for the determination of olive oil phenols | ||||||||
| [219] | 60 g → 2 mL (SPE-Diol [216]) | Applicative work using a previously method [218] | Interesting from a quantitative and applicative point of view | ||||||||
| [188] | 60 g → 2 mL (SPE-Diol [216]) and diluted 1:10 | All the “well-known” phenolic compounds and 28 other analytes | 1st paper in which CZE-ESI-TOF MS is used for the analysis of phenolic compounds from oils. TOF permits the “identification” of new compounds in the oil’s profile | ||||||||
Concluding remarks and future outlook
Abbreviations
| (3,4-DHPEA) | 3,4-dihydroxyphenyl-ethanol or hydroxytyrosol |
| (3,4-DHPEA-AC) | 3,4-dihydroxyphenyl-ethanol acetate or hydroxytyrosol acetate |
| (3,4-DHPEA-EA) | 3,4-dihydroxyphenyl-ethanol linked to elenolic acid |
| (3,4-DHPEA-EDA) | 3,4-dihydroxyphenyl-ethanol linked to dialdehydic form of elenolic acid |
| (Ac Pin) | acetoxypinoresinol |
| (BHT) | buthylated hydroxytoluene |
| (CE) | Capillary Electrophoresis |
| (COX) | cyclooxygenase |
| (DPPH) | diphenylpicrylhydrazyl |
| (EA) | elenolic acid |
| (ESI-MS) | electrospray ionization mass spectrometry |
| (FID) | Flame Ionization Detector |
| (GC) | gas chromatography |
| (HPLC) | High Performance Liquid Chromatography |
| (HPLC-APCI-MS) | HPLC-atmospheric pressure chemical ionization mass spectrometry |
| (IT-MS) | ion-trap mass spectrometry |
| (LDL) | low-density lipoprotein |
| (LLE) | Liquid-Liquid- extraction |
| (LOD) | limit of detection |
| (MS) | mass spectrometry |
| (MS/MS) | tandem mass spectrometry |
| (MSn) | multiple-stage mass spectrometry |
| (NMR) | nuclear magnetic resonance |
| (OSI) | Oxidative Stability Instrument |
| (p-HPEA) | p-hydroxyphenyl-ethanol or tyrosol |
| (p-HPEA-EA) | p-hydroxyphenyl-ethanol linked to elenolic acid |
| (p-HPEA-EDA) | p-hydroxyphenyl-ethanol linked to dialdehydic form of elenolic acid |
| (Pin) | pinoresinol |
| (QqQ) | triple quadrupole |
| (SPE) | Solid-Phase Extraction |
| (VOO) | virgin olive oil |
| (SID) | secoiridoid |
| (TOF-MS) | time of flight mass spectrometry |
| (TPAR) | Total Peak Area Ratio |
| (UV) | ultraviolet |
References and Notes
- Montedoro, G.F.; Servili, M.; Baldioli, M.; Miniati, E. Simple and Hydrolyzable Phenolic Compounds in Virgin Olive Oil. 1. Their Extraction, Separation, and Quantitative and Semiquantitative Evaluation by HPLC. J. Agric. Food. Chem. 1992, 40, 1571–1576. [Google Scholar] [CrossRef]
- Montedoro, G.F.; Servili, M.; Baldioli, M.; Miniati, E. Simple and Hydrolyzable Phenolic Compounds in Virgin Olive Oil. 2. Initial Characterization of the Hydrolyzable Fraction. J. Agric. Food. Chem. 1992, 40, 1577–1580. [Google Scholar] [CrossRef]
- Montedoro, G.F.; Servili, M.; Baldioli, M.; Selvaggini, R.; Miniati, E.; Macchioni, A. Simple and Hydrolyzable Compounds in Virgin Olive Oil. 3. Spectroscopic Characterizations of the Secoiridoid Derivatives. J. Agric. Food Chem. 1993, 41, 2228–2234. [Google Scholar] [CrossRef]
- Amiot, M.J.; Fleuriet, A.; Macheix, J.J. Importance and evolution of phenolic compounds in olive during growth and maturation. J. Agric. Food Chem. 1986, 34, 823–825. [Google Scholar] [CrossRef]
- Amiot, M.J.; Fleuriet, A.; Mecheix, J.J. Accumulation of oleuropein derivatives during olive maturation. Phytochemistry 1989, 28, 67–69. [Google Scholar] [CrossRef]
- Gutierrez Gonzales-Quijano, R.; Janer del Valle, C.; Janier del Valle, M.L.; Gutierrez Rosales, F.; Vazquez Roncero, A. Relacion entre los polifenoles y la calidad y estabilidad del aeite de oliva virgen. Grasas Aceites 1977, 28, 101–106. [Google Scholar]
- Gutfinger, T. Polyphenols in olive oils. J. Am. Oil Chem. Soc. 1981, 58, 966–968. [Google Scholar] [CrossRef]
- Tsimidou, M. Polyphenols and quality of virgin olive oil in retrospective. Ital. J. Food Sci. 1998, 10, 99–115. [Google Scholar]
- Montedoro, G.F.; Cantarelli, C. Indagini sulle sostanze fenoliche presenti negli oli d’oliva. Riv. Ital. Sost. Grasse 1969, 46, 115–124. [Google Scholar]
- Kuwajima, H.; Uemura, T.; Takaishi, K.; Inoue, K.; Inouye, H. A secoiridoid glucoside from Olea europea. Phytochemistry 1988, 27, 1757–1759. [Google Scholar] [CrossRef]
- Cortesi, N.; Azzolini, M.; Rovellini, P.; Fedeli, E. I componenti minori polari degli oli vergini di oliva: ipotesi di struttura mediante LC-MS. Riv. Ital. Sost. Grasse 1995, 72, 241–251. [Google Scholar]
- Cortesi, N.; Azzolini, M.; Rovellini, P.; Fedeli, E. Dosaggio dei componenti minori polari (CMP) in oli vergini di oliva. Riv. Ital. Sostanze Grasse 1995, 72, 333–337. [Google Scholar]
- Angerosa, F.; D’Alessandro, N.; Konstantinou, P.; Di Giacinto, L. Characterization of phenolic and secoiridoid aglycons present in virgin olive oil by gas chromatography-chemical ionization mass spectrometry. J. Chromatogr. A 1996, 736, 195–203. [Google Scholar] [CrossRef]
- Brenes, M.; Garcıa, A.; Garcıa, P.; Rios, J. J.; Garrido, A. Phenolic Compounds in Spanish Olive Oils. J. Agric. Food Chem. 1999, 47, 3535–3540. [Google Scholar] [CrossRef]
- Prim, N.; Pastor, F.I.J.; Diaz, P. Biochemical studies on cloned Bacillus sp. BP-7 phenolic acid decarboxylase PadA. Appl. Microbiol. Biot. 2003, 63, 51–56. [Google Scholar] [CrossRef]
- Exarchou, V.; Godejohann, M.; van Beek, T. A.; Gerothanassis, I. P.; Vervoort, J. LC-UV-Solid-Phase Extraction-NMR-MS Combined with a Cryogenic Flow Probe and Its Application to the Identification of Compounds Present in Greek Oregano. Anal. Chem. 2003, 75, 6288–6294. [Google Scholar] [CrossRef]
- Andreasen, M.F.; Christensen, L.P.; Meyer, A.S.; Hansen, A. Content of phenolic acids and ferulic acid dehydrodimers in 17 rye (Secale cereale L.) varieties. J. Agric. Food Chem. 2000, 48, 2837–2842. [Google Scholar] [CrossRef]
- Barthelmebs, L.; Divies, C.; Cavin, J.F. Knockout of the p-coumarate decarboxylase gene from lactobacillus plantarum reveals the existence of two other inducible enzymatic activities involved in phenolic acid metabolism. Appl. Environ. Micro. 2000, 66, 3368–3375. [Google Scholar] [CrossRef]
- Hakkinen, S.; Heinonen, M.; Karenlampi, S.; Mykkanen, H.; Ruuskanen, J.; Torronen, R. Screening of selected flavonoids and phenolic acids in 19 berries. Food Res. Int. 1999, 32, 345–353. [Google Scholar] [CrossRef]
- Xu, L.; Diosady, L.L. Rapid method for total phenolic acid determination in rapeseed/canola meals. Food Res. Int. 1997, 30, 571–574. [Google Scholar] [CrossRef]
- Maga, J. A. Simple phenol and phenolic compounds in food flavor. Crit. Rev. Food Sci. Nutr. 1978, 10, 323–372. [Google Scholar] [CrossRef]
- Nergiz, C.; Unal, K. Determination of phenolic acids in virgin olive oil. Food Chem. 1991, 39, 237–240. [Google Scholar] [CrossRef]
- Harborne, J. B. The Flavonoids: Advances in Research since 1986; Chapman and Hall: London, U.K, 1994. [Google Scholar]
- Peleg, H.; Naim, M.; Rouseff, R. L.; Zehavi, U. Distribution of bound and free phenolic acids in oranges (Citrus sinensis) and Grapefruits (Citrus paradisi). J. Sci. Food Agric. 1991, 57, 417–426. [Google Scholar] [CrossRef]
- Shahidi, F.; Nacsk, M. Food phenolics: Sources, Chemistry, Effects, and Applications; Technomic Publishing Company, Inc.: Lancaster, PA, 1995. [Google Scholar]
- Robbins, R. J. Phenolic Acids in Foods: An Overview of Analytical Methodology. J. Agric. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef]
- Gomes, C. A.; Girao da Cruz, T.; Andrade, J. L.; Milhazes, N.; Borges, F.; Marques, M. P. M. Anticancer Activity of Phenolic Acids of Natural or Synthetic Origin: A Structure-Activity Study. J. Med. Chem. 2003, 46, 5395–5401. [Google Scholar] [CrossRef]
- Masaki, H.; Okamoto, N.; Sakaki, S.; Sakurai, H. Protective effects of hydroxybenzoic acids and their esters on cell damage induced by hydroxyl radicals and hydrogen peroxides. Biol. Pharm. Bull. 1997, 20, 304–308. [Google Scholar] [CrossRef]
- Buiarelli, F.; Di Berardino, S.; Coccioli, F.; Jasionowska, R.; Russo, M. V. Determination of phenolic acids in olive oil by capillary electrophoresis. Anal. Chim. 2004, 94, 699–705. [Google Scholar] [CrossRef]
- Cartoni, G. P.; Coccioli, F.; Janionowska, R.; Ramirez, D. HPLC analysis of the benzoic and cinnamic acids in edible vegetable oils. Ital. J. Food Sci. 2000, 12, 163–173. [Google Scholar]
- Carrasco Pancorbo, A.; Cruces-Blanco, C.; Segura Carretero, A.; Fernández Gutiérrez, A. Sensitive determination of phenolic acids in extra-virgin olive oil by capillary zone electrophoresis. J. Agric. Food. Chem. 2004, 52, 6687–6693. [Google Scholar] [CrossRef]
- Carrasco-Pancorbo, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Co-electroosmotic capillary electrophoresis determination of phenolic acids in comercial olive oil. J. Sep. Sci. 2005, 28, 925–934. [Google Scholar] [CrossRef]
- Davin, B.D.; Bedgar, D.L.; Katayama, T.; Lewis, N.G. On the stereoselective synthesis of (1)-pinoresinol in Forsythia suspensa from its achiral precursor, coniferyl alcohol. Phytochemistry 1992, 31, 3869–3874. [Google Scholar] [CrossRef]
- Owen, R. W.; Mier, W.; Giacosa, A.; Hull, W. E.; Spiegelhalder, B.; Bartsch, H. Identification of Lignans as Major Components in the Phenolic Fraction of Olive Oil. Clinical Chem. 2000, 46, 976–988. [Google Scholar]
- Brenes, M.; Garcıa, A.; Rios, J. J.; Garcıa, P.; Garrido, A. Use of 1-acetoxypinoresinol to authenticate Picual olive oils. Int. J. Food Sci. Technol. 2002, 37, 615–625. [Google Scholar] [CrossRef]
- Bianco, A.; Coccioli, F.; Guiso, M.; Marra, C. The occurrence in olive oil of a new class of phenolic compounds: hydroxy-isochromans. Food Chem. 2001, 77, 405–411. [Google Scholar]
- Le March, L. Cancer preventive effects of flavonoids—a review. Biomed. Pharmacother. 2002, 56, 296–301. [Google Scholar] [CrossRef]
- Kanadaswami, C.; Lee, L. T.; Lee, P. P. H.; Hwang, J. J.; Ke, F. C.; Huanh, Y. T.; Lee, M.T. The antitumour activities of flavonoids. In vivo 2005, 19, 895–909. [Google Scholar]
- Steinberg, F. M.; Bearden, M. M.; Keen, C. L. Cocoa and chocolate flavonoids: implications for cardiovascular health. J. Am. Diet Assoc. 2003, 103, 215–223. [Google Scholar] [CrossRef]
- Nestel, P. Isoflavones: their effects on cardiovascular risk and functions. Curr. Opin. Lipidol. 2003, 14, 3–8. [Google Scholar] [CrossRef]
- Rovellini, P.; Cortesi, N.; Fedeli, E. Analysis of flavonoids from Olea europaea by HPLC-UV and HPLC-electrospray-MS. Riv. Ital. Sost. Grasse 1997, 74, 273–279. [Google Scholar]
- Vázquez-Roncero, A.; Janer Del Valle, L.; Janer Del Valle, C. Componentes fenòlicos de la aceituna. III. Polifenoles del aceite. Grasas Aceites 1976, 27, 185–191. [Google Scholar]
- Carrasco-Pancorbo, A.; Gómez-Caravaca, A. M.; Cerretani, L.; Bendini, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Rapid quantification of the phenolic fraction of spanish virgin olive oils by capillary electrophoresis with uv detection. J. Agric. Food Chem. 2006, 54, 7984–7991. [Google Scholar] [CrossRef]
- Brenes, M.; García, A.; García, P.; Ríos, J. J.; Garrido, A. Phenolic compounds in Spanish olive oils. J. Agric. Food Chem. 1999, 47, 3535–3540. [Google Scholar] [CrossRef]
- Murkovic, M.; Lechner, S.; Pietzka, A.; Bratacos, M.; Katzogiannos, E. Analysis of minor components in olive oil. J. Biochem. Methods 2004, 61, 155–160. [Google Scholar] [CrossRef]
- Morelló, J. R.; Vuorela, S.; Romero, M. P.; Motilva, M. J.; Heinonen, M. Antioxidant activity of olive pulp and olive oil phenolic compounds of the arbequina cultivar. J. Agric. Food Chem. 2005, 53, 2002–2008. [Google Scholar] [CrossRef]
- Bouaziz, M; Grayer, R. J; Simmonds, M. S. J; Damak, M; Sayadi, S. Identification and antioxidant potential of flavonoids and low molecular weight phenols in olive cultivar Chemlali growing in Tunisia. J. Agric. Food Chem. 2005, 53, 236–241. [Google Scholar] [CrossRef]
- Vlahov, G. Flavonoids in three olives (Olea europaea) fruit varieties during maturation. J. Sci. Food Agric. 1992, 58, 157–159. [Google Scholar] [CrossRef]
- Ryan, D.; Prenzler, P. D.; Lavee, S.; Antolovich, M.; Robards, K. Quantitative changes in phenolic content during physiological development of olive (Olea europaea) cultivar Hardy´s Mammoth. J. Agric. Food Chem. 2003, 51, 2532–2538. [Google Scholar] [CrossRef]
- Romani, A.; Pinelli, P.; Mulinacci, N.; Vincieri, F. F.; Gravano, E.; Tattini, M. HPLC analysis of flavonoids and secoiridoids in leaves of Ligustrum vulgare L. (Oleaceae). J. Agr. Food Chem. 2000, 48, 4091–4096. [Google Scholar] [CrossRef]
- Boskou, D. Sources of natural antioxidants. Trends Food Sci. Technol. 2006, 17, 505–512. [Google Scholar] [CrossRef]
- Stark, A.H.; Madar, Z. Olive oil as a functional food: Epidemiology and nutritional approaches. Nutr. Rev. 2002, 60, 170–176. [Google Scholar] [CrossRef]
- Perez-Jimenez, F. (coordinator) International conference on the healthy effect of virgin olive oil. Eur. J. Clinic. Invest. 2005, 35, 421–424. [Google Scholar] [CrossRef]
- Bisignano, G.; Tomaino, A.; Lo Cascio, R.; Crisafi, G.; Uccella, N.; Saija, A. On the in-vitro antiomicrobial activity of oleuropein and hydroxytyrosol. J. Pharm. Pharmacol. 1999, 31, 971–974. [Google Scholar]
- Glatzle, J.; Beckert, S.; Kasparek, M. S.; Mueller, M. H.; Mayer, P.; Meile, T.; Konigsrainer, A.; Steurer, W. Olive oil is more potent than fish oil to reduce septic pulmonary dysfunctions in rats. Langenbecks Arch Surg 2007, 392, 323–329. [Google Scholar] [CrossRef]
- Tuck, K.L.; Hayball, P.J. Major phenolic compounds in olive oil: metabolism and health effects. J. Nutr. Biochem. 2002, 13, 636–644. [Google Scholar] [CrossRef]
- Tuck, K.L.; Hayball, P.J.; Stupans, I. Structural characterisation of the metabolites of hydroxytyrosol, the principal phenolic component in olive oil, in rats. J. Agric. Food Chem. 2002, 50, 2404–2409. [Google Scholar] [CrossRef]
- Visioli, F.; Bellomo, G.; Galli, C. Free radical-scavenging of olive oil phenols. Biochem. Biophys. Res. Commun. 1998, 247, 60–64. [Google Scholar] [CrossRef]
- Visioli, F.; Galli, C.; Bornet, F.; Mattei, A.; Patelli, R.; Galli, G.; Caruso, D. Olive oil phenolics are dose-dependently absorbed in humans. FEBS Lett. 2000, 468, 159–160. [Google Scholar] [CrossRef]
- Tuck, K.L.; Freeman, M.P.; Hayball, P.J.; Stretch, G.L.; Stupans, I. The in vivo fate of hydroxytyrosol and tyrosol, antioxidant phenolic constituents of olive oil, following intravenous and oral dosing of labelled compounds to rats. J. Nutr. 2001, 131, 1993–1996. [Google Scholar]
- Visioli, F.; Galli, C. Olive oil phenols and their potential effects on human health. J. Agric. Food Chem. 1998, 46, 4292–4296. [Google Scholar] [CrossRef]
- Coni, E.; Di Benedetto, R.; Di Pasquale, M.; Masella, R.; Modesti, D.; Attei, R.; Carlini, E.A. Protective effect of Oleuropein, and olive oil biophenol, on low density lipoprotein oxidizability in rabbits. Lipids 2000, 35, 45–54. [Google Scholar] [CrossRef]
- Menéndez, J. A.; Vázquez-Martín, A.; Colomer, R.; Carrasco-Pancorbo, A.; García-Villalba, R.; Fernández-Gutiérrez, A.; Segura-Carretero, A. Oleuropein aglycone, the bitter principle of olives and Olive oil, is a potent anti-HER2 oncogene agent capable to reverse breast cancer acquired autoresistance to trastuzumab (Herceptin). BMC Cancer 2007, 7, 80. [Google Scholar] [CrossRef]
- Beauchamp, G.K.; Keast, R. S. J.; Morel, D.; Lin, J.; Pika, J.; Han, Q.; Lee, C-H.; Smith, A. B.; Breslin, P. A. S. Ibuprofen-like activity in extra-virgin olive oil. Nature 2005, 437, 45–46. [Google Scholar] [CrossRef]
- Smith, A. B.; Han, Q.; Breslin, P. A. S.; Beauchamp, G. K. Synthesis and assignment of absolute configuration of (-)-oleocanthal: A potent, naturally occurring non-steroidal anti-inflammatory and anti-oxidant agent derived from extra virgin olive oils. Org. Lett. 2005, 7, 5075–5078. [Google Scholar] [CrossRef]
- Fogliano, V.; Sacchi, R. Oleocanthal in olive oil: Between myth and reality. Mol. Nutr. Food Res. 2006, 50, 5–6. [Google Scholar] [CrossRef]
- Visioli, F.; Bellomo, G.; Montedoro, G.; Galli, C. Low-density lipoprotein oxidation is inhibited in vitro by olive oil constituents. Atherosclerosis 1995, 117, 25–32. [Google Scholar] [CrossRef]
- Fito, M.; Covas, M. I.; Lamuela-Raventos, R. M.; Vila, J.; Torrents, J.; de la Torre, C.; Marrugat, J. Protective effect of olive oil and its phenolic compounds against low density lipoprotein oxidation. Lipids 2000, 35, 633–638. [Google Scholar] [CrossRef]
- Leenen, R.; Roodenburg, A. J. C.; Vissers, M. N.; Schuubiers, J. A. E.; van Putte, K. P. A. M.; Wiseman, S. A.; van de Put, F.H. M. M. Supplementation of plasma with olive oil phenols and extracts: Influence on LDL oxidation. J. Agric. Food Chem. 2002, 50, 1290–1297. [Google Scholar] [CrossRef]
- Bogani, P.; Galli, C.; Villa, M.; Visioli, F. Postprandial anti-inflammatory and antioxidant effects of extra virgin olive oil. Atherosclerosis 2007, 190, 181–186. [Google Scholar] [CrossRef]
- Serraino, M.; Thompson, L.U. The effect of flaxseed consumption on the initiation and promotional stages of mammary carcinogenesis. Nutr. Cancer. 1992, 17, 153–159. [Google Scholar] [CrossRef]
- Togna, G.I.; Togna, A.R.; Franconi, M.; Marra, C.; Guiso, M. Olive oil isochromans inhibit human platelet reactivity. J. Nutr. 2003, 133, 2532–2536. [Google Scholar]
- Covas, M. I.; Ruiz-Gutiérrez, V.; de la Torre, R.; Kafatos, A.; Lamuela-Raventós, R. M.:; Osada, J; Owen, R. W.; Visioli, F. Minor components of olive oil: Evidence to date of health benefits in humans. Nutr. Rev. 2006, 64, S20–S30. [Google Scholar] [CrossRef]
- Fitó, M.; Covas, M.I.; Lamuela-Raventós, R.M.; Vila, J.; Torrents, J.; De La Torre, C.; Marrugat, J. Protective effect of olive oil and its phenolic compounds against low density lipoprotein oxidation. Lipids 2000, 35, 633–638. [Google Scholar] [CrossRef]
- Masella, R.; Varì, R.; D'Archivio, M.; Di Benedetto, R.; Matarrese, P.; Malorni, W.; Scazzocchio, B.; Giovannini, C. Extra virgin olive oil biophenols inhibit cell-mediated oxidation of LDL by increasing the mRNA transcription of glutathione-related enzymes. J. Nutr. 2004, 134, 785–791. [Google Scholar]
- De la Puerta, R.; Ruiz-Gutiérez, V.; Hoult, J. R. Inhibition of leukocyte 5-lipooxigenase by phenolics from virgin olive oil. Biochem. Pharmacol. 1999, 157, 445–449. [Google Scholar] [CrossRef]
- Owen, R. W.; Giacosa, A.; Hull, W. E.; Haubner, R.; Spigelhalder, B.; Bartsch, H. The antioxidant/anticancer potencial of phenolic compounds from olive oil. Eur. J. Cancer 2000, 36, 1235–1247. [Google Scholar] [CrossRef]
- Carluccio, M.A.; Siculella, L.; Ancora, M.A.; Massaro, M.; Scoditti, E.; Storelli, C.; Visioli, F.; Distante, A.; De Caterina, R. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: antiatherogenic properties of the Mediterranean diet phytochemicals. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 622–629. [Google Scholar] [CrossRef]
- Moreno, J. J. Effect of olive oil minor components on oxidative stress and arachidonic acid mobilization and metabolism by macrophages RAW 264.7. Free Radic. Biol. Med. 2003, 35, 1073–1081. [Google Scholar] [CrossRef]
- Massaro, M.; Basta, G.; Lazzerini, G.; Carluccio, M.A.; Bosetti, F.; Solaini, G.; Visioli, F.; Paolicchi, A.; De Caterina, R. Quenching of intracellular ROS generation as a mechanism for oleate-induced reduction of endothelial activation in early atherogenesis. Thromb. Haemost. 2002, 88, 335–344. [Google Scholar]
- Petroni, A.; Blasevich, M.; Salami, M.; Papini, N.; Montedoro, G. F.; Galli, C. Inhibition of platelet aggregation and eicosanoid production by phenolic components of olive oil. Thromb. Res. 1995, 78, 151–160. [Google Scholar] [CrossRef]
- Quiles, J. L.; Farquharson, A. J.; Simpson, D. K.; Grant, I.; Wahle, K. W. Olive oil phenolics: effects on DNA oxidation and redoenzyme RNA in prostate cells. Br. J. Nutr. 2002, 88, 225–234. [Google Scholar] [CrossRef]
- Hashim, Y. Z. H. Y.; Eng, M. E.; Gill, C. I. R.; McGlynn, H.; Rowland, I. R. Components of olive oil and chemoprevention of colorectal cancer. Nutr. Rev. 2005, 63, 374–386. [Google Scholar] [CrossRef]
- Stavric, B. Role of chemopreventers in human diet. Clin. Biochem. 1994, 27, 319–332. [Google Scholar] [CrossRef]
- Yang, C.S.; Landau, J.M.; Huang, M.-T.; Newmark, H.L. Inhibition of carcinogenesis by dietary polyphenolic compounds. Ann. Rev. Nutr. 2001, 21, 381–406. [Google Scholar] [CrossRef]
- Frankel, E. N. Volatile lipid oxidation products. Prog. Lipid Res. 1982, 22, 1–33. [Google Scholar] [CrossRef]
- Montedoro, G. Costituenti fenolici presenti negli oli vergini di oliva Nota I:Identificazione di alcuni acidi fenolici e loro potere antiossidante. Sci. Tecnol. Aliment. 1972, 3, 177–186. [Google Scholar]
- Frankel, E. N. Chemistry of autoxidation: mechanism, products and flavor significance. In Flavor Chemistry of Fats and Oils; Min, D. B., Smouse, T. H., Eds.; AOCS Press: Champaign, IL (USA), 1985; pp. 1–37. [Google Scholar]
- Porter, W.L.; Black, E.D.; Drolet, A.M. Use of polyamide oxidative fluorescence test on lipid emulsions: contrast in relative effectiveness of antioxidants in bulk versus dispersed systems. J. Agric. Food Chem. 1989, 37, 615–624. [Google Scholar] [CrossRef]
- Frankel, E. N. Antioxidants in lipid foods and their impact on food quality. Food Chem. 1996, 57, 51–55. [Google Scholar] [CrossRef]
- Paiva-Martins, F.; Santos, V.; Mangericao, H.; Gordon, M. H. Effects of Copper on the Antioxidant Activity of Olive Polyphenols in Bulk Oil and Oil-in-Water Emulsions. J. Agric. Food Chem. 2006, 54, 3738–3743. [Google Scholar] [CrossRef]
- Fregapane, G.; Lavelli, V.; Leon, S.; Kapuralin, J.; Salvador, M.D. Effect of filtration on virgin olive oil stability during storage. Eur. J. Lipid Sci. Technol. 2006, 108, 134–142. [Google Scholar] [CrossRef]
- Mendez, A.I.; Falque, E. Effect of storage time and container type on the quality of extra-virgin olive oil. Food Control 2007, 18, 521–529. [Google Scholar] [CrossRef]
- Lercker, G.; Bendini, A.; Cerretani, L. Quality composition and production process of virgin olive oils. Progr. Nutr. 2007, 9, 9–23. [Google Scholar]
- Gómez-Caravaca, A.M.; Cerretani, L.; Bendini, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Lercker, G. Effect of filtration systems on the phenolic content in virgin olive oil by HPLC-DAD-MSD. Am. J. Food Technol. in press.
- Tsimidou, M. Z.; Georgiou, A.; Koidis, A.; Boskou, D. Loss of stability of ‘‘veiled’’ (cloudy) virgin olive oils in storage. Food Chem. 2005, 93, 377–383. [Google Scholar] [CrossRef]
- Shahidi, F.; Wanasundra, P. D. Phenolic antioxidants. Crit. Rev. Food Sci. Nutr. 1992, 32, 67–103. [Google Scholar] [CrossRef]
- Roginsky, V.; Lissi, E. A. Review of methods to determine chainbreaking antioxidant activity in food. Food Chem. 2005, 92, 235–254. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B; Prior, R. L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Prior, R.; Wu, X.; Schich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Visioli, F.; Bellomo, G.; Galli, C. Free radical-scavenging properties of olive oil polyphenols. Biochem Biophys Res. Commun. 1998, 247, 60–64. [Google Scholar] [CrossRef]
- Paiva-Martins, F.; Gordon, M. H. Effects of pH and ferric ions on the antioxidant activity of olive polyphenols in oil-in-water emulsions. J. Am. Oil Chem. Soc. 2002, 79, 571–576. [Google Scholar] [CrossRef]
- Paiva-Martins, F.; Gordon, M.H. Interactions of Ferric Ions with Olive Oil Phenolic Compounds. J. Agric. Food Chem. 2005, 53, 2704–2709. [Google Scholar] [CrossRef]
- Bendini, A.; Cerretani, L.; Vecchi, S.; Carrasco-Pancorbo, A.; Lercker, G. Protective Effects of Extra Virgin Olive Oil Phenolics on Oxidative Stability in the Presence or Absence of Copper Ions. J. Agric. Food Chem. 2006, 54, 4880–4887. [Google Scholar] [CrossRef]
- Pedrielli, P.; Skibsted, L. H. Antioxidant synergy and regeneation effect of quercetin, (-)-epicatechin, and (+)-catechin on R-tocopherol in homogeneous solutions of peroxidating methyl linoleate. J. Agric. Food Chem. 2002, 50, 7138–7144. [Google Scholar] [CrossRef]
- Cai, Y. J.; Ma, L. P.; Hou, L. F.; Zhou, B.; Yang, L.; Liu, Z. L. Antioxidant effects of green tea polyphenols on free radical initiated peroxidation of rat liver microsomes. Chem. Phys. Lipids 2002, 120, 109–117. [Google Scholar] [CrossRef]
- Pazos, M.; Andersen, M.L.; Medina, I.; Skibsted, L.H. Efficiency of Natural Phenolic Compounds Regenerating a-Tocopherol from a-Tocopheroxyl Radical. J. Agric. Food Chem. 2007, 55, 3661–3666. [Google Scholar] [CrossRef]
- Baldioli, M.; Servili, M.; Perretti, G.; Montedoro, G. Antioxidant Activity of Tocopherols and Phenolic Compounds in Virgin Olive Oil. J. Am. Oil Chem. Soc. 1996, 73, 1589–1593. [Google Scholar] [CrossRef]
- Trujillo, M.; Mateos, R.; Collantes De Teran, L.; Espartero, J. L.; Cert, R.; Jover, J.; Alcudia, F.; Bautista, J.; Cert, A.; Parrado, J. Lipophilic Hydroxytyrosyl Esters. Antioxidant Activity in Lipid Matrices and Biological Systems. J. Agric. Food Chem. 2006, 54, 3779–3785. [Google Scholar] [CrossRef]
- Fki, I.; Allouche, N.; Sayadi, S. The use of polyphenolic extract, purified hydroxytyrosol and 3,4-dihydroxyphenyl acetic acid from olive mill wastewater for the stabilization of refined oils: a potential alternative to synthetic antioxidants. Food Chem. 2005, 93, 197–204. [Google Scholar] [CrossRef]
- Perez-Bonilla, M.; Salido, S.; van Beek, T. A.; Linares-Palomino, P. J.; Altarejos, J.; Nogueras, M.; Sanchez, A. Isolation and identification of radical scavengers in olive tree (Olea europaea) wood. J. Chromatogr. A. 2006, 1112, 311–318. [Google Scholar]
- Tsimidou, M.; Papadopoulos, G.; Boskou, D. Phenolic Compounds and Stability of Virgin Olive Oil - Part I. Food Chem. 1992, 45, 141–144. [Google Scholar] [CrossRef]
- Visioli, F.; Bellomo, G.; Galli, C. Free Radical-Scavenging Properties of Olive Oil Polyphenols. Biochem. Biophys. Res. Commun. 1998, 247, 60–64. [Google Scholar] [CrossRef]
- Chimi, H.; Cillard, J.; Cillard, P.; Rahmani, M. Peroxyl and Hydroxyl Radical Scavenging Activity of Some Natural Phenolic Antioxidants. J. Am. Oil Chem. Soc. 1991, 68, 307–312. [Google Scholar] [CrossRef]
- Gordon, M.H.; Paiva-Martins, F.; Almeida, M. Antioxidant Activity of Hydroxytyrosol Acetate Compared with That of Other Olive Oil Polyphenols. J. Agric. Food Chem. 2001, 49, 2480–2485. [Google Scholar] [CrossRef]
- Aparicio, R.; Roda, L.; Albi, M. A.; Gutierrez, F. Effect of Various Compounds on Virgin Olive Oil Stability Measured by Rancimat. J. Agric. Food Chem. 1999, 47, 4150–4155. [Google Scholar] [CrossRef]
- Carrasco-Pancorbo, A.; Cerretani, L.; Bendini, A.; Segura-Carretero, A.; Del Carlo, M.; Gallina-Toschi, T.; Lercker, G.; Compagnone, D.; Fernandez-Gutierrez, A. Evaluation of the Antioxidant Capacity of Individual Phenolic Compounds in Virgin Olive Oil. J. Agric. Food Chem. 2005, 53, 8918–8925. [Google Scholar] [CrossRef]
- Briante, R.; Febbraio, F.; Nucci, R. Antioxidant properties of low molecular weight phenols present in the Mediterranean diet. J. Agric. Food Chem. 2003, 51, 6975–6981. [Google Scholar] [CrossRef]
- Nenadis, N.; Wang, L. F.; Tsimidou, M. Z.; Zhang, H.Y. Radical Scavenging Potential of Phenolic Compounds Encountered in O. europaea Products as Indicated by Calculation of Bond Dissociation Enthalpy and Ionization Potential Values. J. Agric. Food Chem. 2005, 53, 295–299. [Google Scholar] [CrossRef]
- Lorenz, P.; Zeh, M.; Martens-Lobenhoffer, J.; Schmidt, H.; Wolf, G.; Horn, T.F.W. Natural and newly synthesized hydroxy-1-aryl-isochromans: A class of potential antioxidants and radical scavengers. Free Rad. Res. 2005, 39, 535–545. [Google Scholar] [CrossRef]
- Franconi, F.; Coinu, R.; Carta, S.; Urgeghe, P. P.; Ieri, F.; Mulinacci, N.; Romani, A. Antioxidant Effect of Two Virgin Olive Oils Depends on the Concentration and Composition of Minor Polar Compounds. J. Agric. Food Chem. 2006, 54, 3121–3125. [Google Scholar] [CrossRef]
- Rìos, J.J.; Gil, M.J.; Gutiérrez-Rosales, F. Solid-phase extraction gas chromatography-ion trap-mass spectrometry qualitative method for evaluation of phenolic compounds in virgin olive oil and structural confirmation of oleuropein and ligstroside aglycons and their oxidation products. J. Chromatogr. A 2005, 1093, 167–176. [Google Scholar]
- Rovellini, P.; Cortesi, N. Liquid chromatography-mass spectrometry in the study of oleuropein and ligstroside aglycons in virgin olive oils: aldehydic, dialdehydic forms and their oxidized products. Riv. Ital. Sost. Grasse 2002, 79, 1–14. [Google Scholar]
- Carrasco-Pancorbo, A.; Cerretani, L.; Bendini, A.; Segura-Carretero, A.; Lercker, G.; Fernández-Gutiérrez, A. Evaluation of the Influence of Thermal Oxidation in the Phenolic Composition and in Antioxidant Activity of Extra-Virgin Olive Oils. J. Agric. Food Chem. 2007, 55, 4771–4780. [Google Scholar] [CrossRef]
- Armaforte, E.; Mancebo-Campos, V.; Bendini, A.; Salvador, M. D.; Fregapane, G.; Cerretani, L. Retention effects of oxidized polyphenols during analytical extraction of phenolic compounds of virgin olive oil. J. Sep. Sci. 2007, 15. [Google Scholar] [CrossRef]
- Kilcast, D. Sensory science. Chem. Brit. 2003, 39, 62–62. [Google Scholar]
- Kiritsakis, A. K. Flavor components of olive oil – a review. J. Am. Oil Chem. Soc. 1998, 75, 673–681. [Google Scholar] [CrossRef]
- Angerosa, F.; Mostallino, R.; Basti, C.; Vito, R. Virgin olive oil odour notes: their relationships with volatile compounds from the lipoxygenase pathway and secoiridoid compounds. Food Chem. 2000, 68, 283–287. [Google Scholar] [CrossRef]
- Caponio, F.; Gomes, T.; Pasqualone, A. Phenolic compounds in virgin olive oils: influence of the degree of olive ripeness on organoleptic characteristics and shelf-life. Eur Food Res. Technol. 2001, 212, 329–333. [Google Scholar] [CrossRef]
- Rotondi, A.; Bendini, A.; Cerretani, L.; Mari, M.; Lercker, G.; Gallina Toschi, T. Effect of Olive Ripening Degree on the Oxidative Stability and Organoleptic Properties of Cv. Nostrana di Brisighella Extra Virgin Olive Oil. J. Agric. Food Chem. 2004, 52, 3649–3654. [Google Scholar] [CrossRef]
- Perez, A. G.; Luaces, P.; Rios, J. J.; Garcia, J. M.; Sanz, C. Modification of Volatile Compound Profile of Virgin Olive Oil Due to Hot-Water Treatment of Olive Fruit. J. Agric. Food Chem. 2003, 51, 6544–6549. [Google Scholar] [CrossRef]
- García, J. M.; Yousfi, K.; Mateos, R.; Olmo, M.; Cert, A. Reduction of oil bitterness by heating of olive (Olea europaea) fruits. J. Agric. Food Chem. 2001, 49, 4231–4235. [Google Scholar] [CrossRef]
- European Commission Regulation EEC/2568/91 of 11 July on the Characteristics of Olive and Olive Pomace Oils and on Their Analytical Methods. Off. J. Eur. Commun. 1991, L 248, 1–82.
- Gutiérrez, F.; Perdiguero, S.; Gutiérrez, R.; Olías, J.M. Evaluation of the Bitter Taste in Virgin Olive Oil. J. Am. Oil Chem. Soc. 1992, 69, 394–395. [Google Scholar] [CrossRef]
- Mateos, R.; Cert, A.; Pérez-Camino, C. M.; García, J. M. Evaluation of Virgin Olive Oil Bitterness by Quantification of Secoiridoid Derivatives. J. Am. Oil Chem. Soc. 2004, 81, 71–75. [Google Scholar] [CrossRef]
- Frank, O.; Ottinger, H.; Hofmann, T. Characterization of an intense bitter-tasting 1H,4H-quinolizinium-7-olate by application of the taste dilution analysis, a novel bioassay for the screening and identification of taste-active compounds in foods. J. Agric. Food Chem. 2001, 49, 231–238. [Google Scholar] [CrossRef]
- Andrewes, P.; Busch, J. L. H. C.; De Joode, T.; Groenewegen, A.; Alexandre, H. Sensory Properties of Virgin Olive Oil Polyphenols: Identification of Deacetoxy-ligstroside Aglycon as a Key Contributor to Pungency. J. Agric. Food Chem. 2003, 51, 1415–1420. [Google Scholar] [CrossRef]
- Gutierrez-Rosales, F.; Rios, J. J.; Gomez-Rey, MA. L. Main Polyphenols in the Bitter Taste of Virgin Olive Oil. Structural Confirmation by On-Line High-Performance Liquid Chromatography Electrospray Ionization Mass Spectrometry. J. Agric. Food Chem. 2003, 51, 6021–6025. [Google Scholar] [CrossRef]
- Sinesio, F.; Moneta, E.; Esti, M. The dynamic sensory evaluation of bitterness and pungency in virgin olive oil. Food Qual. Pref. 2005, 16, 557–564. [Google Scholar] [CrossRef]
- Gutierrez, F.; Albi, M.A.; Palma, R.; Rios, J.J.; Olias, J.M. Bitter taste of virgin olive oil: Correlation of sensory evaluation and instrumental HPLC analysis. J. Food Sci. 1989, 54, 68–70. [Google Scholar] [CrossRef]
- Busch, J. L. H. C.; Hrncirik, K.; Bulukin, E.; Boucon, C.; Mascini, M. Biosensor measurements of polar phenolics for the assessment of the bitterness and pungency of virgin olive oil. J Agric Food Chem. 2006, 54, 4371–4377. [Google Scholar] [CrossRef]
- Beltrán, G.; Ruano, M. T.; Jiménez, A.; Uceda, M.; Aguilera, M. P. Evaluation of virgin olive oil bitterness by total phenol content analysis. Eur. J. Lipid Sci. Technol. 2007, 108, 193–197. [Google Scholar]
- Vazquez-Roncero, A.; Janer del Valle, C.; Janer del Valle, M.L. Determinación de polifenoles totales en el aceite de oliva. Grasas Aceites. 1973, 24, 350–357. [Google Scholar]
- Graciani Costante, E.; Vazquez Roncero, A. Estudio de los components polares del aceite de oliva por cromatografia liquida de alta eficacia (HPLC) II Cromatografia en fase inverse. Grasas y Aceites. 1980, 31, 237–243. [Google Scholar]
- Solinas, M.; Cichelli, A. Sulla determinazione delle sostanze fenoliche dell'olio di oliva. Riv. Ital. Sost. Grasse. 1981, 58, 159–164. [Google Scholar]
- Solinas, M.; Cichelli, A. GLC and HPLC quantitation of phenolic compounds in olive oil: the possible role of tyrosol in assessing virgin olive oil amount in mixture with refined oils. Riv. Ital. Soc. Aliment. 1982, 11, 4–12. [Google Scholar]
- Tsimidou, M.; Papadopoulos, G.; Boskou, D. Determination of phenolic compounds in virgin olive oil by reverse-phase HPLC with emphasis on UV detection. Food Chem. 1992, 45, 141–144. [Google Scholar] [CrossRef]
- Akasbi, M.; Shoeman, D.W.; Saari Csallany, A. High Performance Liquid Chromatography of selected phenolic compounds in olive oils. J. Am. Oil Chem. Soc. 1993, 70, 367–370. [Google Scholar] [CrossRef]
- Mannino, S.; Cosio, M.S.; Bertuccioli, M. High performance liquid chromatography of phenolic compounds in virgin olive oils using amperometric detection. Ital. J. Food Sci. 1993, 4, 363–370. [Google Scholar]
- Tsimidou, M.; Lytridou, M.; Boskou, D.; Pappa-Louis, A.; Kotsifaki, F.; Petrakis, C. On determination of minor phenolic acids of virgin olive oil by RP-HPLC. Grasas y Aceites. 1996, 47, 151–157. [Google Scholar] [CrossRef]
- Janer Del Valle, C.; Vazquez Roncero, A. A study of the polar compounds in olive oil by gas chromatography. Grasas y Aceites. 1980, 31, 309–316. [Google Scholar]
- Solinas, M. HRGC analysis of phenolic components in virgin olive oils in relation to the ripening and the variety of olives. Riv. Ital. Sost. Grasse. 1987, 64, 255–262. [Google Scholar]
- Angerosa, F.; D’Alessandro, N.; Konstantinou, P.; Di Giacinto, L. GC-MS evaluation of phenolic compounds in virgin olive oil. J. Agric. Food Chem. 1995, 43, 1802–1807. [Google Scholar] [CrossRef]
- Angerosa, F.; D’Alessandro, N.; Corana, F.; Mellerio, G. Characterisation of phenolic and secoiridoid aglycons present in virgin olive oil by gas chromatography-chemical ionisation mass spectrometry. J. Chromatogr. A. 1996, 736, 195–203. [Google Scholar] [CrossRef]
- Ohnesorge, J.; Sänger-van de Griend, C.; Wätzig, H. Quantitation in capillary electrophoresis-mass spectrometry. Electrophoresis 2005, 26, 2360–2375. [Google Scholar] [CrossRef]
- Ryan, D.; Robards, K.; Prenzler, P.; Jardine, D.; Herlt, T. Antolovich. Liquid chromatography with electrospray ionisation mass spectrometric detection of phenolic compounds from Olea elropaea. J. Chromatogr. A. 1999, 855, 529–537. [Google Scholar] [CrossRef]
- Soler, C.; Hamilton, B.; Furey, A.; James, K. J.; Mañes, J.; Picó, Y. Comparison of tour mass analyzers for determining carbosulfan and its metabolites in citrus by liquid chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2006, 20, 2151–2164. [Google Scholar] [CrossRef]
- Bristow, A. W. T.; Webb, K. S. Intercomparison study on accurate mass measurement of small molecules in mass spectrometry. J. Am. Soc. Mass Spectrom. 2003, 14, 1086–1098. [Google Scholar] [CrossRef]
- Pelzing, M.; Decker, J.; Neusüß, C.; Räther, O. talk A042670. In presented on 52nd ASMS Conference on Mass Spectrometry and Allied Topics, Nashville, TN, May 23–27, 2004.
- Bringmann, G.; Kajahn, I.; Neusüß, C.; Pelzing, M.; Laug, S.; Unger, M.; Holzgrabe, U. Analysis of the glucosinolate pattern of Arabidopsis thaliana seeds by capillary zone electrophoresis coupled to electrospray ionization-mass spectrometry. Electrophoresis 2005, 26, 1513–1522. [Google Scholar] [CrossRef]
- Owen, R.W.; Haubner, R.; Mier, W.; Giacosa, A.; Hull, W.E.; Spiegelhalder, B.; Bartsch, H. Isolation, structure elucidation and antioxidant potential of the major phenolic and flavonoid compounds in brined olive drupes. Food Chem. Toxicol. 2003, 41, 703–717. [Google Scholar] [CrossRef]
- Cardoso, S.M.; Guyot, S.; Marnet, N.; Lopes-da-Silva, J.A.; Renard, C.M.G.C.; Coimbra, M.A. Characterisation of phenolic extracts from olive pulp and olive pomace by electrospray mass spectrometry. J. Sci. Food Agric. 2005, 85, 21–32. [Google Scholar] [CrossRef]
- Di Donna, L.; Mazzotti, F.; Napoli, A.; Salerno, R.; Sajjad, A.; Sindona, G. Secondary metabolism of olive secoiridoids. New microcomponents detected in drupes by electrospray ionization and high-resolution tandem mass spectrometry. Rapid Commun. Mass. Spectrom. 2007, 21, 273–278. [Google Scholar] [CrossRef]
- Silva, S.; Gomes, L.; Leitao, F.; Coelho, A.V.; Boas, L.V. Phenolic compounds and antioxidant activity of Olea europaea L. fruits and leaves. Food Sci. Technol. Intern. 2006, 12, 385–395. [Google Scholar] [CrossRef]
- Vinha, A. F.; Ferreres, F.; Silva, B. M.; Valentao, P.; Gonçalves, A.; Pereira, J. A.; Oliveira, M. B.; Seabra, R. M.; Andrade, P. B. Phenolic profiles of Portuguese olive fruits (Olea europaea L.): Influences of cultivar and geographical origin. Food Chem. 2005, 89, 561–568. [Google Scholar] [CrossRef]
- Mulinacci, N.; Innocenti, M.; La Marca, G.; Mercalli, E.; Giaccherini, C.; Romani, A.; Erica, S.; Vincieri, F.F. Solid olive residues: Insight into their phenolic composition. J. Agric. Food Chem. 2005, 53, 8963–8969. [Google Scholar] [CrossRef]
- Innocenti, M.; La Marca, G.; Malvagia, S.; Giaccherini, C.; Vincieri, F.F.; Mulinacci, N. Electrospray ionisation tandem mass spectrometric investigation of phenylpropanoids and secoiridoids from solid olive residue. Rapid Commun. Mass. Spectrom. 2006, 20, 2013–2022. [Google Scholar] [CrossRef]
- Mulinacci, N.; Romani, A.; Galardi, C.; Pinelli, P.; Giaccherini, C.; Vincieri, F. F. Polyphenolic contentt in olive oil waste waters and related olive samples. J. Agric. Food Chem. 2001, 49, 3509–3514. [Google Scholar] [CrossRef]
- De la Torre-Carbot, K.; Jauregui, O.; Castellote, A.I.; Lamuela-Raventos, R.M.; Covas, M.I.; Casals, I.; Lopez-Sabater, M.C. Rapid high-performance liquid chromatography-electro spray ionization tandem mass spectrometry method for qualitative and quantitative analysis of virgin olive oil phenolic metabolites in human low-density lipoproteins. J. Chromatogr. A 2006, 1116, 69–75. [Google Scholar]
- De la Torre-Carbot, K.; Chavez-Servin, J.L.; Jauregui, O.; Castellote, A.I.; Lamuela-Raventos, R.M.; Fito, M.; Covas, M.I.; Munoz-Aguayo, D.; Lopez-Sabater, M.C. Presence of virgin olive oil phenolic metabolites in human low density lipoprotein fraction: Determination by high-performance liquid chromatography-electrospray ionization tandem mass spectrometry. Anal. Chim. Acta 2007, 583, 402–410. [Google Scholar] [CrossRef]
- Sacchi, R.; Addeo, F.; Paolillo, L. 1H and 13C NMR of virgin olive oil. An overview. Magn. Reson. Chem. 1997, 35, S5133–S5145. [Google Scholar]
- Vlahov, G. Application of NMR to the study of olive oils. Prog. Nucl. Magn. Reson. Spectrosc. 1999, 35, 341–357. [Google Scholar] [CrossRef]
- Gerothanassis, I. P.; Exarchou, V.; Lagouri, V.; Troganis, A.; Tsimidou, M.; Boskou, D. Methodology fro identification of phenolic acids in complex mixtures by high-resolution two-dimensional nuclear magnetic resonance. Application to methanolic extracts of two oregano species. J. Agric. Food Chem. 1998, 46, 4185–4192. [Google Scholar] [CrossRef]
- Christophoridou, S.; Dais, P.; Tseng, L. H.; Spraul, M. Separation and identification of phenolic compounds in olive oil by coupling High-performance liquid chromatography with postclumn solid-phase extraction to nuclear magnetic resonance spectroscopy (LC-SPE-NMR). J. Agric. Food Chem 2005, 53, 4667–4679. [Google Scholar] [CrossRef]
- Litridou, M.; Linssen, J.; Schols, H.; Bergmans, M.; Posthumus, M.; Tsimidou, M.; Boskou, D. Phenolic compounds in virgin olive oils: Fractionation by solid phase extraction and antioxidant activity assessment. J. Sci. Food Agric. 1997, 74, 169, 174. [Google Scholar]
- Bianco, A.; Buiarelli, F.; Cartoni, G.; Coccioli, F.; Muzzalupo, I.; Polidori, A.; Uccella, N. Analysis by HPLC-MS/MS of biophenolic components of olive oil. Anal. Letters 2001, 34, 1033, 1051. [Google Scholar]
- Cortesi, N.; Azzolini, M.; Rovellini, P.; Fedeli, E. Minor polar components of virgin olive oils: a hypothetical structure by LC-MS. Riv. Ital. Sost. Grasse 1995, 72, 241–251. [Google Scholar]
- Caruso, D.; Colombo, R.; Patelli, R.; Giavarini, F.; Galli, G. Rapid evaluation of phenolic component profile and analysis of oleuropein aglycon in olive oil by atmospheric pressure chemical ionization-mass spectrometry (APCI-MS). J. Agric. Food Chem. 2000, 48, 1182–185. [Google Scholar] [CrossRef]
- Romani, A.; Mulinacci, N.; Pinelli, P.; Vincieri, F. F.; Cimato, A. Polyphenolic content in five Tuscany cultivars of Olea europaea L. J. Agric. Food Chem. 1999, 47, 964–967. [Google Scholar] [CrossRef]
- Romani, A.; Pinelli, P.; Mulinacci, N.; Galardi, C.; Vincieri, F. F.; Liberatore, L.; Cichelli, A. HPLC and HRGC analyses of polyphenols and secoiridoid in olive oil. Chromatographia 2001, 53, 279–284. [Google Scholar] [CrossRef]
- Gutiérrez-Rosales, F.; Ríos, J. J.; Gómez-Rey, M. L. Main polyphenols in the bitter taste of virgin olive oil. Structural confirmation by on-line high-performance liquid chromatography electrospray ionization mass spectrometry. J. Agric. Food Chem. 2003, 51, 6021–6025. [Google Scholar] [CrossRef]
- Bendini, A.; Bonoli, M.; Cerretani, L.; Biguzzi, B.; Gallina-Toschi, T.; Lercker, G. Liquid-liquid and solid-phase extraction from phenols from virgin olive oil and their separation by chromatographic and electrophoretic methods. J. Chromatogr. A 2003, 985, 425–433. [Google Scholar] [CrossRef]
- Bianco, A.; Buiarelli, F.; Cartoni, G.; Coccioli, F.; Jasionowska, R.; Margherita, P. Analysis by liquid chromatography-tandem mass spectrometry of biophenolic compounds in virgin olive oil. Part II. J. Sep. Sci. 2003, 26, 417–424. [Google Scholar] [CrossRef]
- Rotondi, A.; Bendini, A.; Cerretani, L.; Mari, M.; Lercker, G.; Gallina Toschi, T. Effect of olive ripening degree on the oxidative stability and organoleptic properties of cv. Nostrana di Brisighella extra virgin olive oil. J. Agric. Food Chem. 2004, 52, 3649–3654. [Google Scholar] [CrossRef]
- Bonoli, M.; Bendini, A.; Cerretani, L.; Lercker, G.; Gallina-Toschi, T. Qualitative and semiquantitative analysis of phenolic compounds in extra virgin olive oils as a function of the ripening degree of olive fruits by different analytical techniques. J. Agric. Food Chem. 2004, 52, 7026–7032. [Google Scholar] [CrossRef]
- De la Torre-Carbot, K.; Jauregui, O.; Gimeno, E.; Castellote, A. I.; Lamuela-Raventós, R. M.; López-Sabater, M. C. Characterization and quantification of phenolic compounds in olive oils by solid-phase extraction, HPLC-DAD and HPLC-MS/MS. J. Agric. Food Chem. 2005, 53, 4331–4340. [Google Scholar] [CrossRef]
- Mulinaci, N.; Giaccherini, C.; Ieri, F.; Innoccenti, M.; Romani, A.; Vincieri, F. F. Evaluation of lignans and free and linked hydroxy-tyrosol and tyrosol in extra virgin olive oil after hydrolysis processes. J. Sci. Food Agric. 2006, 86, 757, 764. [Google Scholar]
- Carrasco-Pancorbo, A.; Neusüß, C.; Pelzing, M.; Segura-Carretero, A.; Fernández-Gutiérrez, A. CE- and HPLC-TOF-MS for the characterization of phenolic compounds in olive oil. Electrophoresis 2007, 28, 806–821. [Google Scholar] [CrossRef]
- Limiroli, R.; Consonni, R.; Ottolina, G.; Marsilio, V.; Bianchi, G.; Zetta, L. H-1 and C-13 NMR characterization of new oleuropein aglycones. J. Chem. Soc-Perkin. 1995, 1, 1519–1523. [Google Scholar]
- Gariboldi, P.; Jommi, G.; Verotta, L. Secoiridoids from olea-europaea. Phytochemistry 1986, 25, 865–869. [Google Scholar] [CrossRef]
- Owen, R. W.; Mier, W.; Giacosa, A.; Hull, W. E.; Spiegelhalder, B.; Bartsch, H. Phenolic compounds and squalene in olive oils: the concentration and antioxidant potential of total phenols, simple phenols, secoiridoids, lignans and squalene. Food Chem. Toxicol. 2000, 38, 647–659. [Google Scholar] [CrossRef]
- Mateos, R.; Espartero, J. L.; Trujillo, M.; Ríos, J. J.; León-Camacho, M.; Alcudia, F.; Cert, A. Determination of phenols, flavones, and lignans in virgin olive oils by solid-phase extraction and high-performance liquid chromatography with diode array ultraviolet detection. J. Agric. Food Chem. 2001, 49, 2185–2192. [Google Scholar] [CrossRef]
- Hrncirik, K.; Fritsche, S. Comparability and reliability of different techniques for the determination of phenolic compounds in virgin olive. Eur. J. Lipid Sci. Technol. 2004, 106, 540–549. [Google Scholar] [CrossRef]
- Fernández-Bolanos, J.; Rodríguez, G.; Rodríguez, R.; Heredia, A.; Guillén, R.; Jiménez, A. Production in large quantities of highly purified hydroxytyrosol from liquid-solid waste of two-phase olive oil processing or “alperujo”. J. Agric. Food Chem. 2002, 50, 6804–6811. [Google Scholar] [CrossRef]
- Bastoni, L.; Bianco, A.; Piccioni, F.; Uccella, N. Biophenolic profile in olives by nuclear magnetic resonance. Food Chem. 2001, 73, 145–151. [Google Scholar] [CrossRef]
- Savournin, C.; Baghdikian, B.; Elías, R.; Dargouth-Kesraoui, F.; Boukef, K.; Balansard, G. Rapid high-performance liquid chromatography analysis for the quantitative determination of oleuropein in Olea europaea leaves. J. Agric. Food Chem. 2001, 49, 618–621. [Google Scholar] [CrossRef]
- Limiroli, R.; Consonni, R.; Ranalli, A.; Bianchi, G.; Zetta, L. H-1 NMR study of phenolics in the vegetation water of three cultivars of Olea europaea: Similarities and differences. J. Agric. Food Chem. 1996, 44, 2040–2048. [Google Scholar] [CrossRef]
- Bianco, A.; Uccella, N. Biophenolic components of olives. Food Res. Int. 2000, 33, 475–485. [Google Scholar] [CrossRef]
- Obied, H. K.; Karuso, P.; Prenzler, P. D.; Robards, K. Novel secoiridoids with antioxidant activity from Australian olive mill waste. J. Agric. Food Chem. 2007, 55, 2848–2853. [Google Scholar] [CrossRef]
- Servili, M.; Baldioli, M.; Selvagginni, R.; Miniati, E.; Macchioti, A.; Montedoro, G. High-performance liquid chromatography evaluation of phenols in olive fruit, virgin olive oil, vegetation waters, and pomace and 1D- and 2D-nuclear magnetic resonance. J. Am. Oil Chem. Soc. 1999, 76, 873–882. [Google Scholar] [CrossRef]
- Capasso, R. A review on the electron ionization and fast atom bombardment mass spectrometry of polyphenols naturally occurring in olive wastes and some of their synthetic derivatives. Phytochem. Anal. 1999, 10, 299–306. [Google Scholar] [CrossRef]
- De Nino, A.; Mazzotti, F.; Perri, E.; Procopio, A.; Raffaelli, A.; Sindona, G. Virtual freezing of hemiacetal-aldehyde equilibrium of the aglycones of oleuropein and ligstroside present in olive oils from Carolea and Coratina cultivars by ionspray ionization tandem mass spectrometry. J. Mass Spectrom. 2000, 35, 461–467. [Google Scholar] [CrossRef]
- Cardoso, S. M.; Guyot, S.; Marnet, N.; Lopes-da-Silva, J. A.; Silva, A. M. S.; Renard, C. M. G. C.; Coimbra, M. A. Identification of oleuropein oligomers in olive pulp and pomace. J. Sci. Food Agric. 2006, 86, 1495–1502. [Google Scholar] [CrossRef]
- Christophoridou, S.; Dais, P. Novel approach to the detection and quantification of phenolic compounds in olive oil based on P-31 nuclear magnetic resonance spectroscopy. J. Agric. Food Chem. 2006, 54, 656–664. [Google Scholar] [CrossRef]
- Frazier, R. A. Recent advances in capillary electrophoresis methods for food analysis. Electrophoresis 2001, 22, 4197–4206. [Google Scholar] [CrossRef]
- Ibáñez, E.; Cifuentes, A. New analytical techniques in food science. Crit. Rev. Food Sci. Nutr. 2001, 41, 413–450. [Google Scholar] [CrossRef]
- Senorans, F. J.; Ibañez, E.; Cifuentes, A. New trends in food processing. Crit. Rev. Food Sci. Nutr. 2003, 43, 507–526. [Google Scholar] [CrossRef]
- Escarpa, A.; González, M. C. An overview of analytical chemistry of phenolic compounds in foods. Crit. Rev. Food Sci. Nutr. 2001, 31, 57–139. [Google Scholar]
- Frazier, R. A.; Ames, J. M.; Nursten, H. E. The development and application of capillary electrophoresis methods for food analysis. Electrophoresis 1999, 20, 3156–3180. [Google Scholar] [CrossRef]
- Boyce, M. C. Determination of additives in food by capillary electrophoresis. Electrophoresis 2001, 22, 1447–1459. [Google Scholar] [CrossRef]
- Andrade, P.; Ferreres, F.; Gil, M. I.; Tomás Barberán, F. A. Determination of phenolic compounds in honeys with different floral origin by capillary zone electrophoresis. Food Chem. 1997, 60, 79–84. [Google Scholar] [CrossRef]
- Issaq, H. J. Capillary electrophoresis of natural products-II. Electrophoresis 1999, 20, 3190–3202. [Google Scholar] [CrossRef]
- Pazourek, J.; González, G.; Revilla, A. L.; Havel, J. Separation of polyphenols in Canary Islands wine by capillary zone electrophoresis without preconcentration. J. Chromatogr. A. 2000, 874, 111–119. [Google Scholar] [CrossRef]
- Schmitt-Kopplin, P.; Englmann. Capillary electrophoresis-mass spectrometry: Survey on developments and applications 2003-2004. Electrophoresis 2005, 26, 1209–1220. [Google Scholar] [CrossRef]
- Bonoli, M.; Montanucci, M.; Gallina-Toschi, T.; Lercker, G. Fast separation and determination of tyrosol, hydroxytyrosol and other phenolic compounds in extra-virgin olive oil by capillary zone electrophoresis with ultraviolet-diode array detection. J. Chromatogr. A. 2003, 1011, 163–172. [Google Scholar]
- Gómez-Caravaca, A. M.; Carrasco-Pancorbo, A.; Cañabate-Díaz, B.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Electrophoretic identification and quantitation of compounds in the polyphenolic fraction of extra-virgin olive oil. Electrophoresis 2005, 26, 3538–3551. [Google Scholar] [CrossRef]
- Carrasco-Pancorbo, A.; Arráez-Román, D.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Capillary electrophoresis-electrospray ionization-mass spectrometry method to determine the phenolic fraction of extra-virgin olive oil. Electrophoresis 2006, 27, 2182–2196. [Google Scholar] [CrossRef]
- Carrasco-Pancorbo, A.; Gómez-Caravaca, A. M.; Cerretani, L.; Bendini, A.; Segura-Carretero, Fernández-Gutiérrez. A simple and rapid electrophoretic method to characterize simple phenols, lignans, complex phenols, phenolic acids, and flavonoids in extravirgin olive oil. J. Sep. Sci. 2006, 29, 2221–2233. [Google Scholar] [CrossRef]
- Carrasco-Pancorbo, A.; Gómez-Caravaca, A. M.; Cerretani, L.; Bendini, A.; Segura-Carretero, Fernández-Gutiérrez. Rapid quantification of the phenolic fraction of Spanish virgin olive oils by capillary electrophoresis with UV detection. J. Agric. Food Chem. 2006, 54, 7984–7991. [Google Scholar] [CrossRef]
- Pirisi, F. M.; Cabras, P.; Falqui Cao, C.; Migliorini, M.; Muggelli, M. Phenolic compounds in virgin olive oil. 2. Reappraisal of the extraction, HPLC separation, and quantification procedures. J. Agric. Food Chem. 2000, 48, 1191–1196. [Google Scholar] [CrossRef]
© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Bendini, A.; Cerretani, L.; Carrasco-Pancorbo, A.; Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Lercker, G. Phenolic Molecules in Virgin Olive Oils: a Survey of Their Sensory Properties, Health Effects, Antioxidant Activity and Analytical Methods. An Overview of the Last Decade Alessandra. Molecules 2007, 12, 1679-1719. https://doi.org/10.3390/12081679
Bendini A, Cerretani L, Carrasco-Pancorbo A, Gómez-Caravaca AM, Segura-Carretero A, Fernández-Gutiérrez A, Lercker G. Phenolic Molecules in Virgin Olive Oils: a Survey of Their Sensory Properties, Health Effects, Antioxidant Activity and Analytical Methods. An Overview of the Last Decade Alessandra. Molecules. 2007; 12(8):1679-1719. https://doi.org/10.3390/12081679
Chicago/Turabian StyleBendini, Alessandra, Lorenzo Cerretani, Alegria Carrasco-Pancorbo, Ana Maria Gómez-Caravaca, Antonio Segura-Carretero, Alberto Fernández-Gutiérrez, and Giovanni Lercker. 2007. "Phenolic Molecules in Virgin Olive Oils: a Survey of Their Sensory Properties, Health Effects, Antioxidant Activity and Analytical Methods. An Overview of the Last Decade Alessandra" Molecules 12, no. 8: 1679-1719. https://doi.org/10.3390/12081679
APA StyleBendini, A., Cerretani, L., Carrasco-Pancorbo, A., Gómez-Caravaca, A. M., Segura-Carretero, A., Fernández-Gutiérrez, A., & Lercker, G. (2007). Phenolic Molecules in Virgin Olive Oils: a Survey of Their Sensory Properties, Health Effects, Antioxidant Activity and Analytical Methods. An Overview of the Last Decade Alessandra. Molecules, 12(8), 1679-1719. https://doi.org/10.3390/12081679