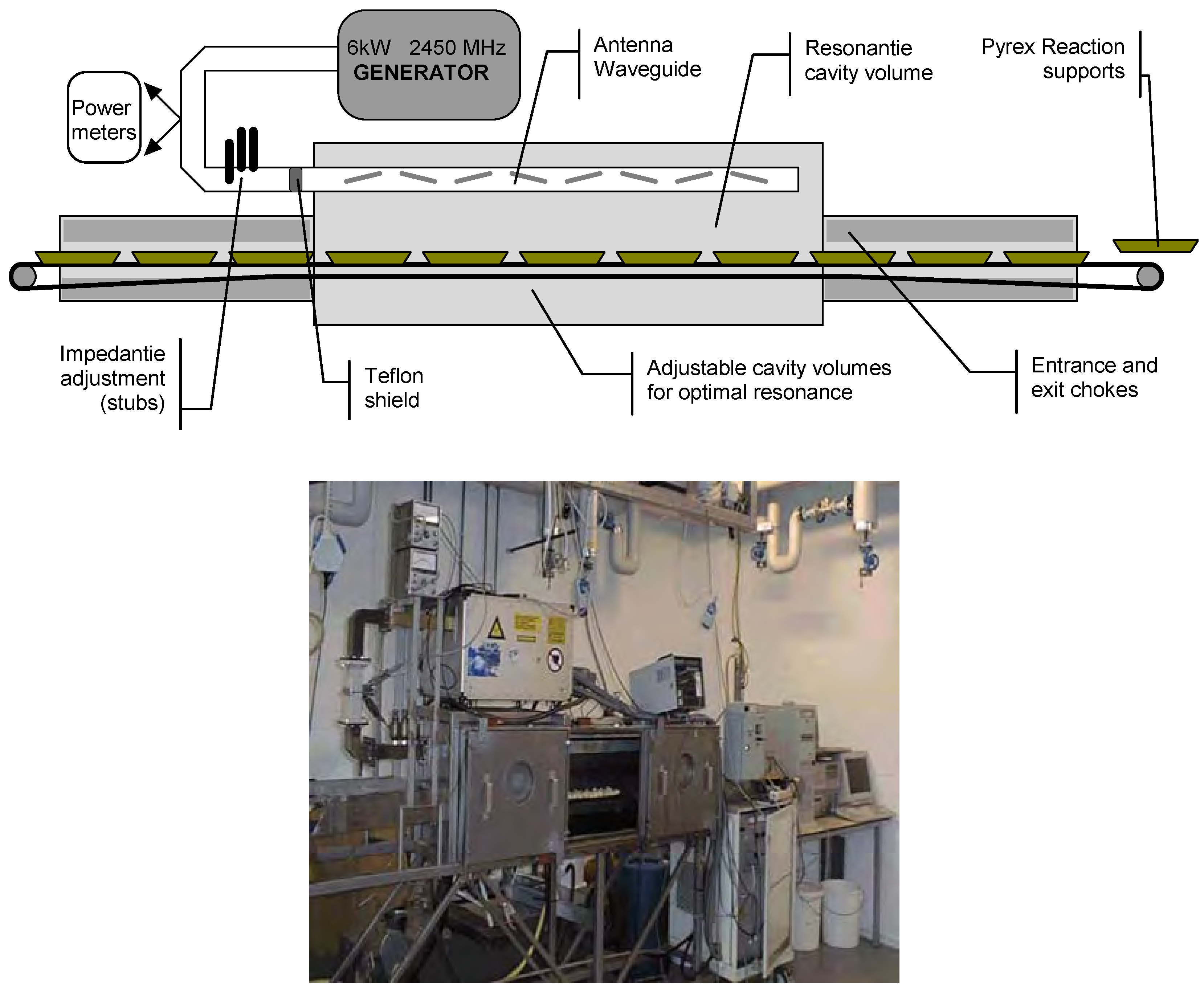
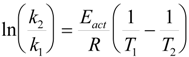Original thermal 2.45 GHz microwave effect
Frequencies ranging from 3 MHz to 30 GHz i.e. from radio-frequencies to the infrared are being used to process food. Depending on the chosen frequency and the particular design of the applicator, treatment by electromagnetic energy at different wavelengths has distinct features. For example, in microwave ovens electromagnetic waves with centimeter wavelengths freely propagate and are absorbed by solid or liquid phase food products. The principle of microwave heating is that the changing electrical field interacts with the molecular dipoles and charged ions. The heat generated by the molecular rotation is due to friction of this motion.
The influence of microwave energy on chemical or biochemical reactions is strictly thermal. The microwave energy quantum is given by the usual equation W = h ν. Within the frequency domain of microwaves and hyper-frequencies (300 MHz - 300 GHz), the corresponding energies are 1.24 10
-6 - 1.24 10
-3 eV, respectively. These energies are much lower than the usual ionisation energies of biological compounds (13.6 eV), of covalent bond energies like OH (5 eV), hydrogen bonds (2 eV), Van der Waals intermolecular interactions (lower than 2 eV) and even lower than the energy associated to Brownian motion at 37°C (2.7 10
-3eV). From this scientific point of view, direct molecular activation of microwaves should be excluded. Some kind of stepwise accumulation of the energy, giving rise to a high-activated state should be totally excluded due to fast relaxation [
2]. Like Peterson [
3] wrote in many of his articles: “The question and the debate of the non thermal effect of microwave give a lot of damage for the reputation of this technology and its application in industry”. Microwaves are only absorbed by dipoles, transforming their energy into heat.
Heat transfer advantages of applying microwave power, a non contact energy source, into the bulk of a material include: faster energy absorption, reduced thermal gradients, selective heating and virtually unlimited final temperatures. For chemical production, the resultant value could include: more effective heating, fast heating of catalysts, reduced equipment size, faster response to process heating control, faster start-up, increased production, and elimination of process steps.
Microwave assisted heterogeneous catalytic reactions
Most industrial chemical reactions are carried out in a solvent in the presence of solid catalysts. When the reaction is conducted in a heterogeneous medium using a dissipative and/or catalytic solid phase and under microwave heating, the reaction rate increases compared to classical heating under the same conditions (pressure, temperature and reagent concentrations). In this paper, several reactions of industrial interest have been studied: isomerisation of m-xylene to p-xylene in presence of aluminum intercalated montmorillonite, Wacker oxidation of cyclohexene with PdCl2 in the presence of heptane as solvent, hydrolysis of hexanenitrile to hexanoic acid and esterification of fatty acids. Aluminum intercalated montmorillonite or granulated ceramics (diameter 5 mm) are used as a catalyst and dissipative solid phase.
When the catalyst is introduced in solid granular form, the yield and the rate of the heterogeneous isomerisation, oxidation, esterification and hydrolysis reactions increases with microwave heating as compared to conventional heating under the same reaction.
Table 1 shows an increase yield of 200% for the oxidation and 150% for hydrolysis when the reaction is conducted in the microwave batch reactor.
Table 1.
Heterogeneous reactions under microwave and classical heating.
Table 1.
Heterogeneous reactions under microwave and classical heating.
| Chemical reaction | T(°C) | time (min) | MW Yield (%) | Classical Yield (%) |
|---|
| Isomerization of m-xylene | 400 | 30 | 25 | 16 |
| Hydrolysis of hexanenitrile | 100 | 60 | 40 | 26 |
| Oxidation of cyclohexene | 80 | 60 | 26 | 12 |
| Esterification of stearic acid | 140 | 120 | 97 | 83 |
The increase in reaction rate corresponds to a virtual difference in reaction temperature. Since the bulk temperature is equal for both the conventional and microwave heated systems, there must be an elevated temperature at the local reaction site; i.e. the catalytic surface. This is possible since the heat transfer by microwaves depends on the specific loss factor of the different materials - the catalyst and the solvent. Many metallic catalysts are semi-conductors and will readily absorb microwave energy, especially at higher temperatures.
The apparent temperature of the catalytic site under microwave irradiation can be estimated from the initial reaction rates. The reaction rate is connected to the temperature by the Arrhenius equation k = A exp (-Eact/RT). For two temperatures, the ratio of the respective reaction rates is related to:
From a kinetic analysis of the esterification reaction, the activation energy (Eact) for the hydrolysis of hexanenitrile has been determined to be 70 ± 3 kJ/mole. From equation (1), it follows that the apparent elevated temperature of the catalyst for the batch reactions was calculated to be 9 ± 1 K higher than the measured bulk temperature.
In the steady state, the microwave heat transfer to the catalyst is equal to the heat loss of the catalyst to its surroundings. The resulting ΔT will be linearly dependent on the difference in loss factor and the radius of the catalyst.
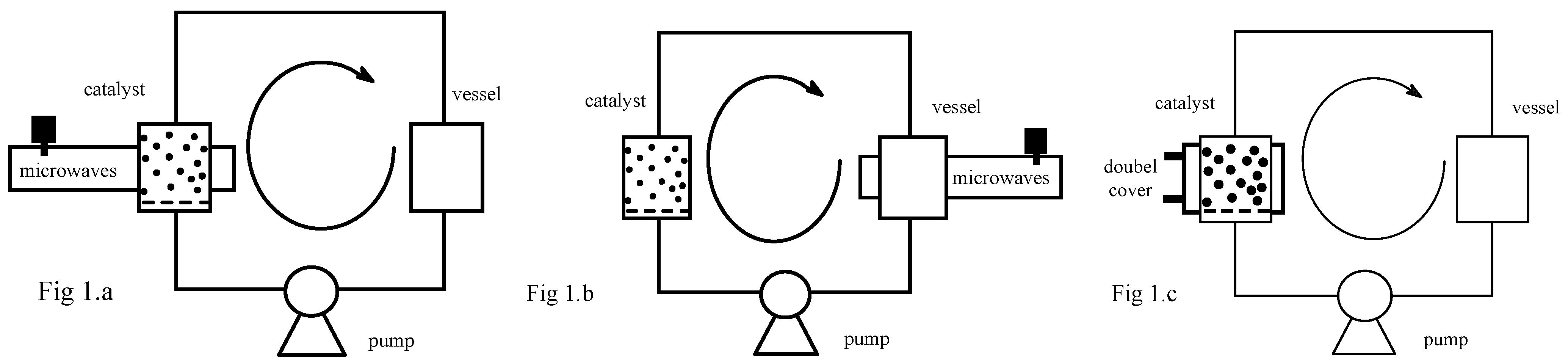
Figure 1.
Microwave and classical continuous reactors [
4].
Figure 1.
Microwave and classical continuous reactors [
4].
To prove the essential combination of microwave and catalyst, the continuous flow microwave reactor (
Figure 1) has been used in two different set-ups. In the first experimental set-up (
Figure 1a) the solid catalyst is submitted to microwave irradiation and the liquid is heated when it passes through the catalyst. In the second experiment (
Figure 1b), only the liquid is heated by microwaves when it circulates in the cavity vessel and the catalyst is placed outside the microwave cavity. The temperature was maintained and checked to be constant throughout the circuit, since the heat loss to the surrounding occurs mainly in the cavity where it is compensated by microwave energy. In the third experiment (
Figure 1c), the reaction was conducted in a conventional continuous reactor operating under the same conditions (temperature, concentration and pressure) as the microwave continuous reactor.
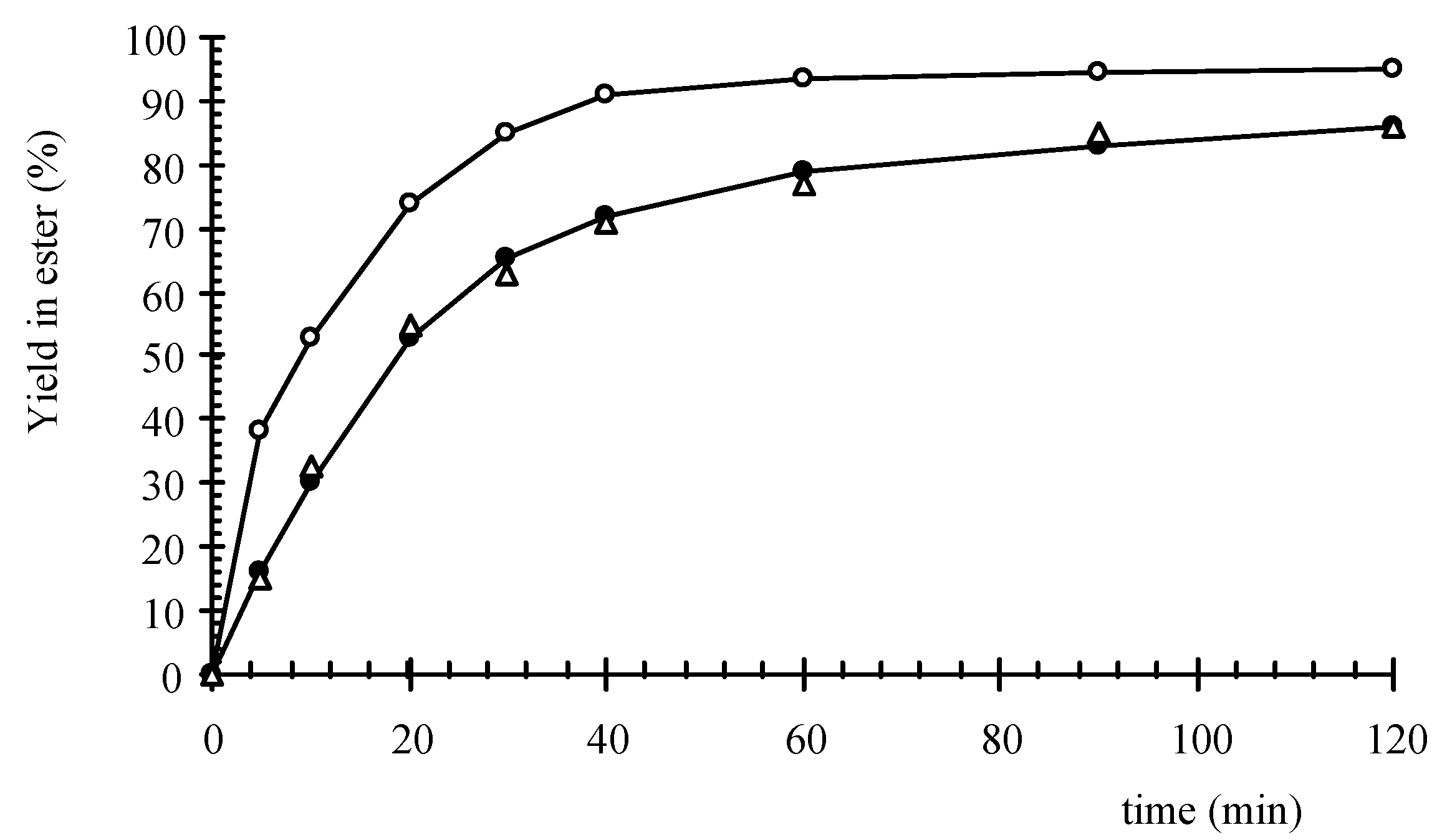
Figure 2.
Influence of the location of the catalyst in continuous microwave reactor (○)
Fig 1.a (∆)
Fig 1.b (●)
Fig 1.c.
Figure 2.
Influence of the location of the catalyst in continuous microwave reactor (○)
Fig 1.a (∆)
Fig 1.b (●)
Fig 1.c.
In
Figure 2, the yield is plotted as a function of time for the three experiments at 80 °C and for a flow rate of 1.33 mL/s. Only a direct heating of the catalyst by microwaves (
Figure 1a) gave an increase of initial esterification reaction rate of 150%. Conventional heating of the catalyst vessel (
Figure 1c) and microwave heating with the catalyst outside the cavity (
Figure 1b) gave identical results because the temperature at the catalytic surface was equal to the bulk temperature. Although the bulk temperature in all experiments was the same, there will be a higher temperature at the surface of the catalyst when it is directly heated by microwaves.
Prediction model: Arrhenius equation
The principal question which microwave chemists ask is why a category of reactions is accelerated by microwaves, like esterification of fatty acids, acylation of aromatic ethers or Diels-Alder reaction, and a second one is not accelerated by microwaves, like arylation of alkenes, hydrolysis of sugars or cyclisation of citronellal [
1].
In the field of microwave assisted organic synthesis, scientist report their results in terms of the yield obtained after a certain time. In order to estimate the influence of the microwaves on the different reaction times or yields obtained when either microwave or conventional heating is applied, it is useful to have a numerical approximation for the processes involved [
5,
6].
We present a tool which will help microwave chemists estimate microwave reaction kinetics and yield parameters related to the process setting for commercial microwave reactors. This model predicting reaction kinetics and yields under microwave heating is based on the Arrhenius equation, in agreement with experimental data and procedures. Chemists could answer to such questions as “my reaction yields 90% after 7 days at reflux; is it possible to obtain the same yield after a few minutes under microwaves?” or “my reaction is not activated by microwaves, what is the problem?” The prediction tool is freely available from the authors and is hopefully helpful for the comprehension of microwave chemistry, to rapidly explore the ‘chemistry space to increase the diversity of the microwave accelerated reactions, or to compare results with scientists in the microwave chemistry field.
For a homogeneous chemical reaction system, it is mainly the fast heating rate that accounts for the differences observed. In comparison with classical heated systems, microwaves give us the opportunity to reach process condition in time and/or temperatures that are not normally accessible. A high temperature results in a fast reaction rate; thus a fast heating results in a quick completion of the reaction without essentially changing the reaction kinetics. That is, if the reaction time is in the order of magnitude of the heating time. Although classical heat transfer is often not considered to be a limiting factor in laboratory scale experiments, the successful application of closed plastic reaction vessels, proves that even at a small scale, the application of microwave heating makes a reduction in reaction time practical. Heating rates can also influence the selectivity between two competing processes. The reaction rate of one path might increase more with the temperature then the other. Through analysis of the classical Arrhenius behaviour, it will be shown that the region in which we can discriminate between two similar reactions is in practice quite limited. Rapid volumetric heating was also found to improve the reaction in processes with a phase transfer.
The volumetric heating aspect is particularly beneficial in solvent free reaction systems, where the concentrations are high and the conductive heat transfer limited. A heat transfer rate similar to that of microwaves is often not feasible by conventional means at a larger scale. This is inherent to the severe temperature gradient, which causes degradation at the surface of the reactor. The application of volumetric heating and the fact that microwave can be directly regulated, make it feasible to work just below the degradation limit temperature. In solvent reaction systems, the volumetric heating might result in super heated boiling. The absence of nucleation sides in the bulk of the homogeneous liquid results in a reverse thermal gradient. The liquid can only loose heat by boiling at the reactor surface or top liquid surface. The super heated temperature of the bulk is very depended on the thermodynamic and dielectric properties of the solvent. The phenomena can be applied to conduct homogeneous reactions at 30 degrees higher temperature then classical possible under atmospheric pressure, resulting in a corresponding reaction rate increase.
Volumetric heating does not imply a homogeneous temperature distribution. Especially in batch processing, the result of a high standing wave ratio is local hot spots, which might be several 10 to 100 °C hotter than the average temperature. As the reaction rate increases non-linear with the temperature, will the observed average reaction rate be somewhat higher than the reaction rate corresponding with the average temperature.
The material selective nature of microwave heating results in more defined and localised hot spots in a heterogeneous system because of the difference in loss between the two media. In heterogeneous catalysis, the catalyst can have a steady state temperature well above the continuous medium. Although the catalytic side might actually occupy a small fraction of the total systems volume, the reaction only proceeds there. Consequently will the observed reaction rate be faster then what would be expected on basis of the measured bulk temperature. From a simple two-compartment model it can be concluded that in this case, the mass exchange and reactor cooling are crucial factors. The effect on the overall kinetic as a function of the thermal energy flow will be discussed.
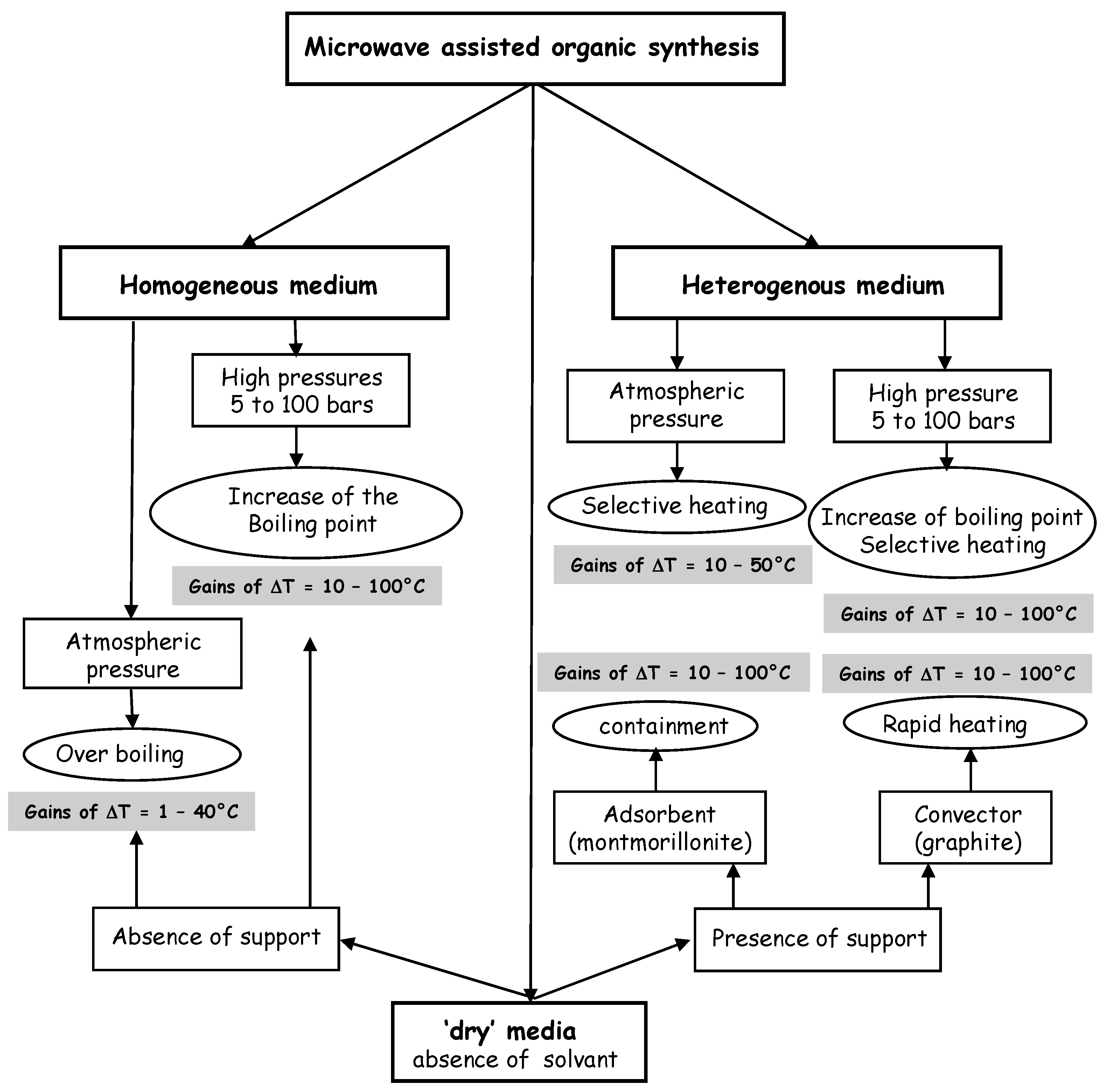
Figure 3.
Experimental procedures generally used in microwave assisted organic synthesis.
Figure 3.
Experimental procedures generally used in microwave assisted organic synthesis.
According to the selected experimental procedure, a reaction conducted under microwaves gains 10 to 100°C in comparison with the maximum temperature reached by conventional heating (
Figure 3). Advantages of applying microwave power, a no contact energy source, into the bulk of a material include: faster energy absorption, reduced thermal gradients, selective heating and virtually unlimited final temperature. For chemical synthesis, the resultant value could include: more effective heating, fast heating of catalysts, reduced equipment size, faster response to process heating control, faster start-up, increased production, and elimination of process steps.
The ideal model to which every researcher seeks is obviously the theoretical and explanatory model which is built from fundamental laws. The simplest example of this type of model, in the field of chemistry, is the Arrhenius equation:
where E
act : Activation energy ; k : rate constant ; T : reaction temperature ; A : frequency factor ; R : constant of ideal gas. Another relation exists between activation energy and the temperature factor which mesure the relative enhancement of reaction rate with the increase of reaction temperature ΔT:
We deducted: Ln (kT+ΔT / kT) = ΔT * Eact / (R * T * (T+ΔT))
In general chemistry textbooks, it is usually assumed that the temperature factor kT+ΔT/kT doubles when the reaction temperature is increased by a factor of ΔT = 10°C. This assumption could give incorrect conclusions, because it is valid only for specific values of temperature and activation energy.
In this work, we have considered the temperature induced by microwaves as the major factor influencing acceleration of organic synthesis. We have made a similarity between the temperature factor and the microwave ’acceleration effect’ noted: M = kMW / kConv. = kT+ΔT / kT
This microwave factor M represents the ratio between the reaction rate under microwaves (at a temperature of T + ΔT) and the reaction rate under conventional heating (at a temperature T). For first order organic reactions, the microwave factor also represents the ratio between reaction times under conventional heating and microwaves: M = treact. (Conv.)/ t react. (MW).
Table 1 is generated from the equation of the microwave factor in function of reaction temperature, activation energy and elevated temperature induced by microwaves in function of the experimental procedure used. The reactions could be divided into two categories according to the activation energy and to the microwave ‘acceleration effect’ factor: a) Reactions accelerated by microwaves: high activation energy > 100 kJ/mole. In this category, an increase of temperature by a few degrees could enhance dramatically the organic reaction by dividing the reaction temperature by 100 to 1,000,000. b) In the second category, we have reactions not accelerated by microwaves that have low activation energy. They need a maximum of microwave energy to have a great increase in reaction temperature of more than 100°C to see a minor effect in reaction yields and kinetics. To have the maximum microwave ‘acceleration effect’ factor, the reaction has to possess high activation energy, conducted at low temperature in the conventional procedure, and realized with a maximum temperature gradient under appropriate microwave procedure.
The table could be used also for yield and selectivity calculations by entering the appropriate kinetic equations regarding the order of the reaction and the number of reagents used. For example, we could calculate time and yield for a first order reaction by these equations.
| | Reagent | → | Product |
| t=0 | a | | 0 |
| t=t | a-x | | x |
The reaction rate for a first order reaction: v = k * (a-x). By integration, we could find the kinetic equation: kT * t = Ln (a / (a-x)) for conventional heating; and kT+ΔT * t = Ln (a / (a-x)) for microwaves.
Table 2.
Prediction of microwave assisted organic synthesis [
7].
Table 2.
Prediction of microwave assisted organic synthesis [7].
| | reaction temperature T = 50 °C | reaction temperature T = 100 °C | reaction temperature T = 200 °C | reaction temperature T = 400 °C |
|---|
| ΔT = TMO - T Conv. | ΔT = TMO - T Conv. | ΔT = TMO - T Conv. | ΔT = TMO - T Conv. |
|---|
| Eact. (J/mole) | ΔT = 10 | ΔT = 50 | ΔT = 100 | ΔT = 10 | ΔT = 50 | ΔT = 100 | ΔT = 10 | ΔT = 50 | ΔT = 100 | ΔT = 10 | ΔT = 50 | ΔT = 100 |
|---|
| 20 000 | 1 | 3 | 6 | 1 | 2 | 4 | 1 | 2 | 2 | 1 | 1 | 2 |
| 40 000 | 2 | 7 | 34 | 1 | 5 | 15 | 1 | 3 | 6 | 1 | 2 | 3 |
| 60 000 | 2 | 20 | 197 | 2 | 10 | 60 | 1 | 4 | 14 | 1 | 2 | 4 |
| 80 000 | 2 | 54 | 1144 | 2 | 21 | 234 | 2 | 7 | 35 | 1 | 3 | 6 |
| 100 000 | 3 | 147 | 6656 | 2 | 45 | 914 | 2 | 11 | 85 | 1 | 3 | 10 |
| 120 000 | 4 | 399 | 38716 | 3 | 97 | 3572 | 2 | 18 | 205 | 1 | 4 | 6 |
| 140 000 | 5 | 1084 | 225182 | 3 | 208 | 13966 | 2 | 30 | 499 | 1 | 6 | 25 |
| 160 000 | 6 | 2942 | 1309730 | 4 | 445 | 54604 | 2 | 49 | 1213 | 2 | 7 | 40 |
| Microwave 'acceleration' factor M = kMO / kConv. = kT+ΔT / kT = treact (Conv.) / treact (MW) |
Time to yield 90% (x/a = 0.9) is equal to:
The yield could be calculated by:
If an organic reaction (activation energy of 100 kJ/mole) is conducted in a conventional procedure at 100°C for 7 days, it will be finished after 3 days in an homogeneous media with an over-boiling phenomena (TMO=110°C; ΔT=10°C), or in 4 hours in microwave ‘dry media’ (TMO=150°C; ΔT=50°C), or in 11 minutes in a microwave reactor under pressure or using a convector like graphite (TMO=200°C ; ΔT=100°C).
The prediction model can be applied to organic reactions with high or low activation energies. Hydrolysis of nitriles to carboxylic acids has an activation energy of 120 kJ/mole. This reaction yields 76% after heating conventionally at 140°C for 7 days. The prediction model shows that this reaction could be finished with a yield of 99% after only 30 minutes when applying microwaves under high pressure (in a Teflon bomb) at a reaction temperature of 240°C. This prediction is in accordance with published results [
8].