Characterization of Flavonoid Subgroups and Hydroxy Substitution by HPLC-MS/MS
Abstract
:Introduction
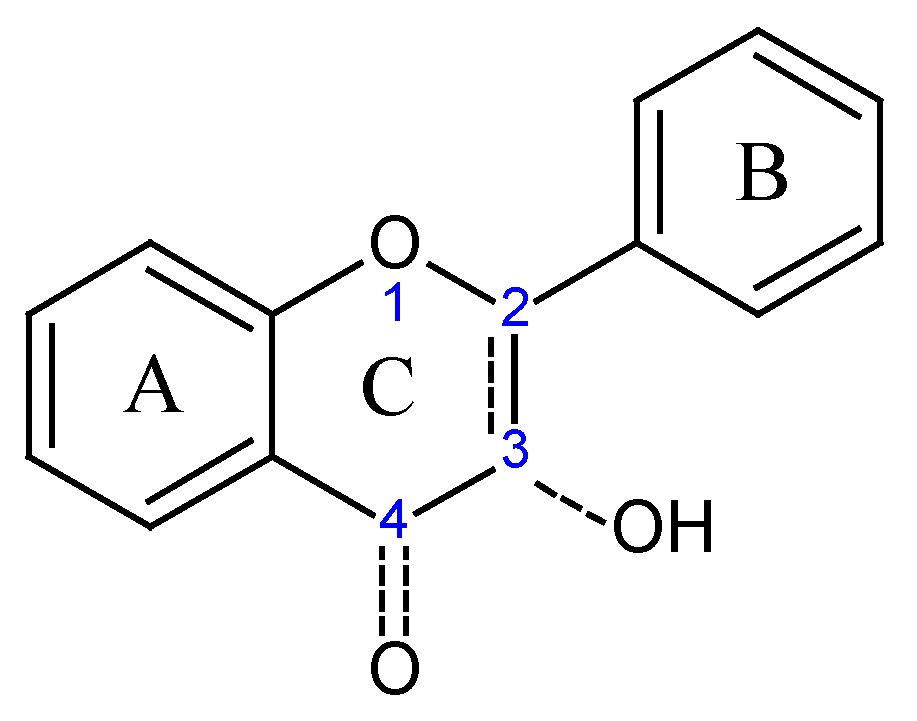
Results and Discussion

| Standard compound | R.t. (min) | Absorbance maxima (nm) | oregano flavonoid | R.t. (min) | Absorbance maxima (nm) | |||
|---|---|---|---|---|---|---|---|---|
| Band A | Band B | Band A | Band B | |||||
| quercetin (1) | 43.0 | 372 | 256 | A | 27.3 | 340 (sh) | 288 | |
| fisetin (2) | 40.1 | 362 | 248 | B | 35.1 | 334 (sh) | 292 | |
| kaempferol (3) | 44.7 | 366 | 267 | C | 40.0 | 336 (sh) | 288 | |
| flavonol (4) | 51.8 | 344 | 240 | D | 43.5 | 332 (sh) | 290 | |
| luteolin (5) | 42.4 | 350 | 254 | |||||
| apigenin (6) | 47.0 | 336 | 269 | |||||
| chrysin (7) | 49.1 | 314 | 268 | |||||
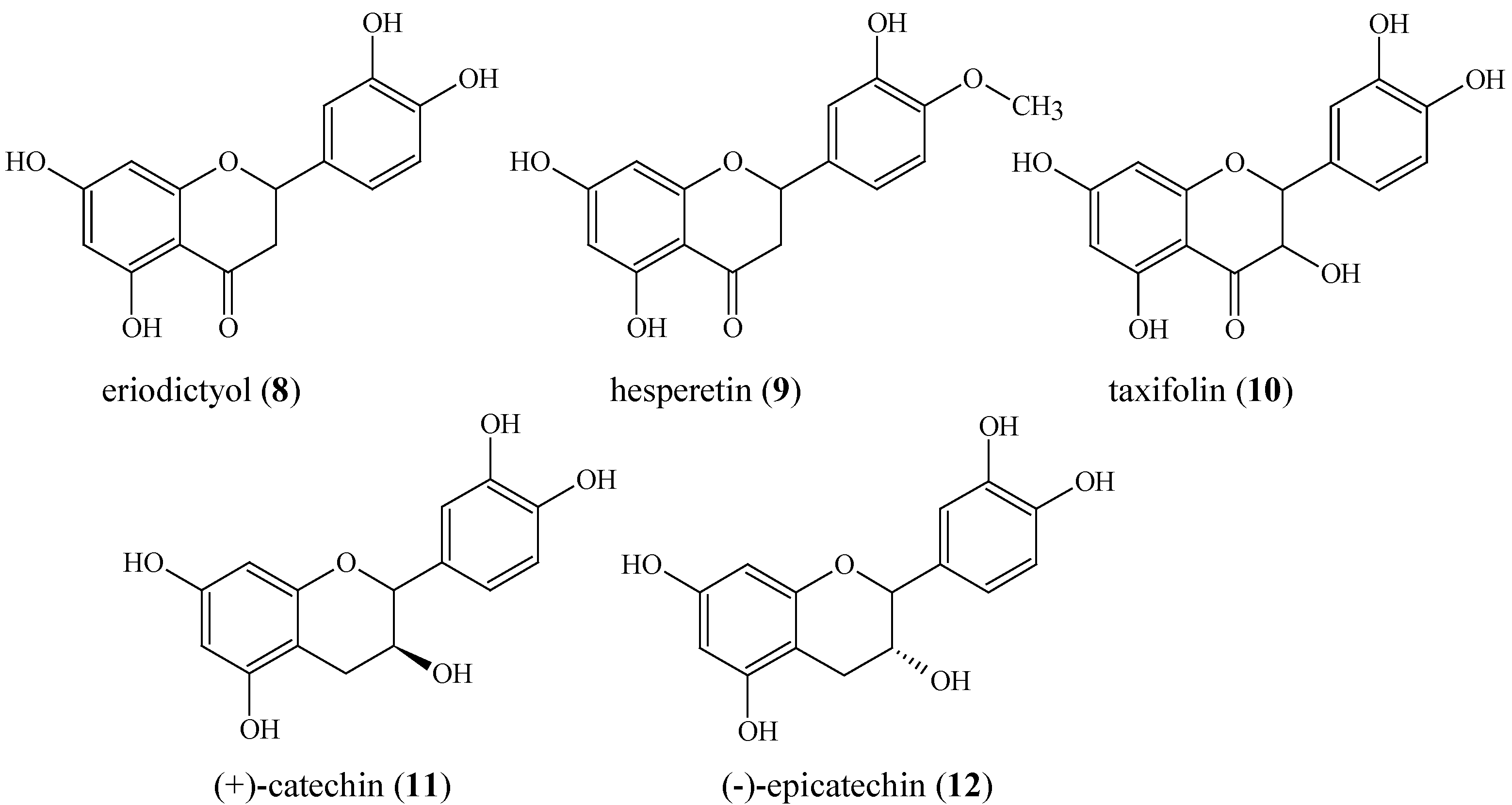
| eriodictyol (8) | 39.5 | 336 (sh) | 288 | |||||
| hesperetin (9) | 45.4 | 336 (sh) | 288 | |||||
| taxifolin (10) | 27.5 | 340 (sh) | 288 | |||||
| (+)-catechin (11) | 13.5 | - | 280 | |||||
| (-)-epicatechin (12) | 19.8 | - | 278 | |||||
| Standard flavonoid aglycones | |||||||
|---|---|---|---|---|---|---|---|
| Assessed fragments | Flavonols | Flavones | |||||
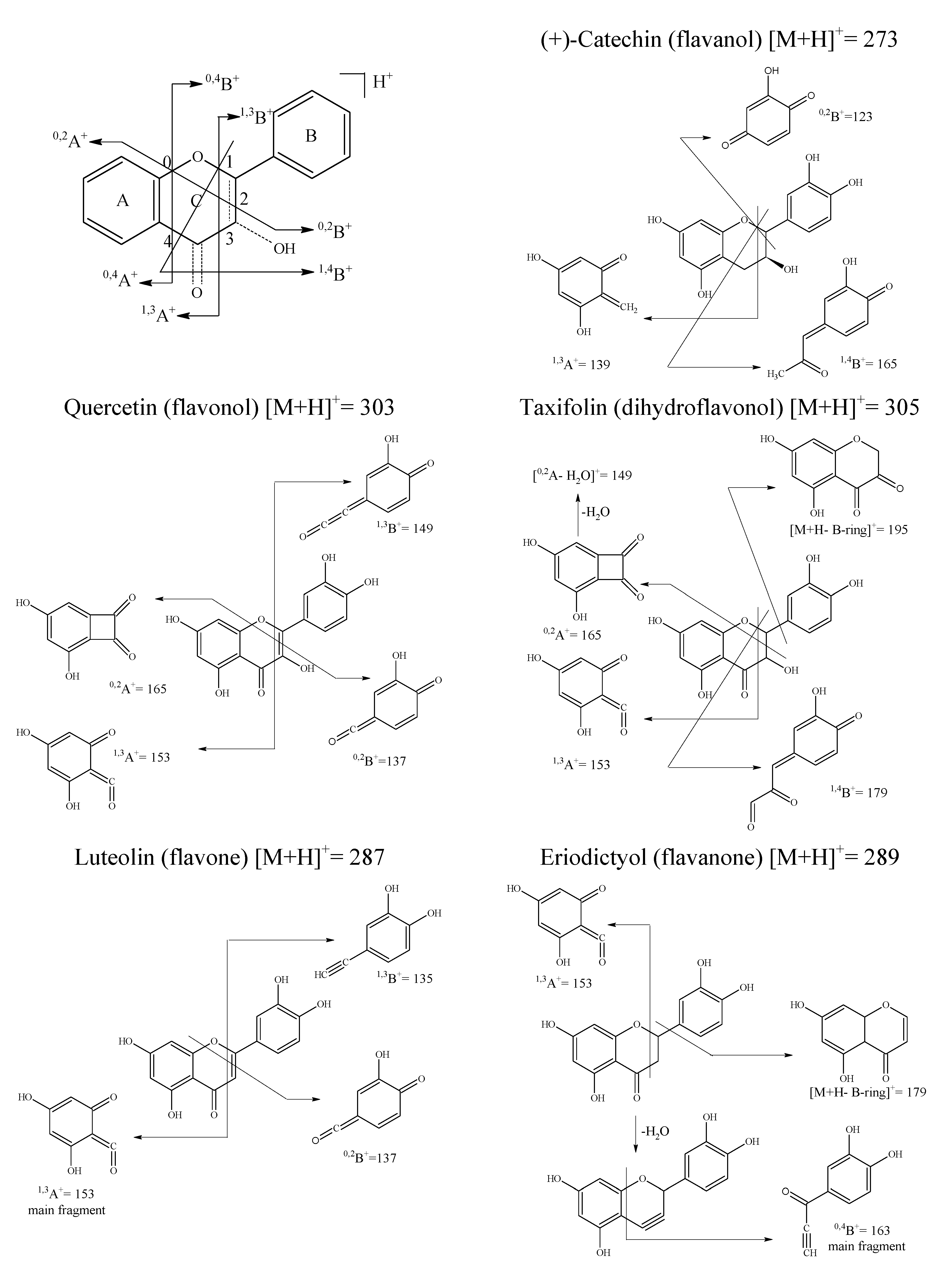
| Quercetin (1) | Fisetin (2) | Kaempferol (3) | Flavonol (4) | Luteolin (5) | Apigenin (6) | Chrysin (7) | |
| [M+H]+ | 303 | 287 | 287 | 239 | 287 | 271 | 255 |
| Defining subgroup fragments | |||||||
| [M+H-H2O]+ | 285 (69) a | 269 (59) | 269 (39.5) | 221 (28) | 269 (36) | 253 (20) | 237 (36.5) |
| [M+H-H2O-CO]+ | 257 (100) | 241 (100) | 241 (100) | 193 (31.5) | 241 (36.5) | 225 (18) | 209 (23) |
| [M+H-H2O-2CO]+ | 229 (42) | 213 (62) | 213 (59) | 165 (64) | – | – | – |
| [M+H-CO]+ | 275 (14) | 259 (25) | 259 (32) | 211 (7) | – | – | – |
| [M+H-2CO]+ | 247 (30) | 231 (70) | 231 (29.5) | 183 (77) | – | – | – |
| [M+H-CH2CO]+ | – | – | – | – | – | – | – |
| Defining A- & B- ring fragments | |||||||
| 0,2A+ | 165 (35.5) | 149 | 165 (52) | 133 (78) | – | – | – |
| 0,2B+ | 137 (11) | 137 | 121 (21) | 105 (48) | 137 (19) | 121 (7) | – |
| 0,4B+ | – | – | – | – | – | – | – |
| 1,2B+ | – | – | – | – | – | – | – |
| 1,3A+ | 153 (7) | 137 (22) | 153 (27) | 121 (100) | 153 (100) | 153 (100) | 153 (100) |
| 1,3B+ | 149 (5.5) | 149 (50) | 133 (13) | – | 135 (17) | 119 (18) | 102 (16) |
| 1,4B+ | – | – | – | – | – | – | – |
| [0,2A-H2O]+ | – | – | – | – | – | – | – |
| [M+H-B-ring]+ | – | – | – | – | – | – | – |
| Assessed fragments | Flavanones | Dihydroflavonol | Flavanols | ||||
| Eriodictyol (8) | Hesperetin (9) | Taxifolin (10) | (+)-Catechin (11) | (–)-Epicatechin (12) | |||
| [M+H]+ | 289 | 303 | 305 | 291 | 291 | ||
| Defining subgroup fragments | |||||||
| [M+H-H2O]+ | 271 (15.5) | 285 (11) | 287 (98) | 273 (15) | 273 (16) | ||
| [M+H-H2O-CO]+ | – | – | 259 (100) | – | – | ||
| [M+H-H2O-2CO]+ | – | – | 231 (2) | – | – | ||
| [M+H-CO]+ | – | – | – | – | – | ||
| [M+H-2CO]+ | – | – | – | – | – | ||
| [M+H-CH2CO]+ | 247 (5) | 261 (4) | – | 249 (2) | 249 (3) | ||
| Defining A- & B- ring fragments | |||||||
| 0,2A+ | – | – | – | – | – | ||
| 0,2B+ | – | – | – | – | – | ||
| 0,4B+ | 163 (100) | 177 (100) | – | – | – | ||
| 1,2B+ | – | – | – | 123 (91) | 123 (93) | ||
| 1,3A+ | 153 (21) | 153 (13) | 153 (35) | 139 (100) | 139 (100) | ||
| 1,3B+ | – | – | – | – | – | ||
| 1,4B+ | – | – | 179 (2) | 165 (77.5) | 165 (79) | ||
| [0,2A-H2O]+ | – | – | 149 (3) | – | – | ||
| [M+H-B-ring]+ | 179 (20) | 179 (26) | 195 (16) | – | – | ||
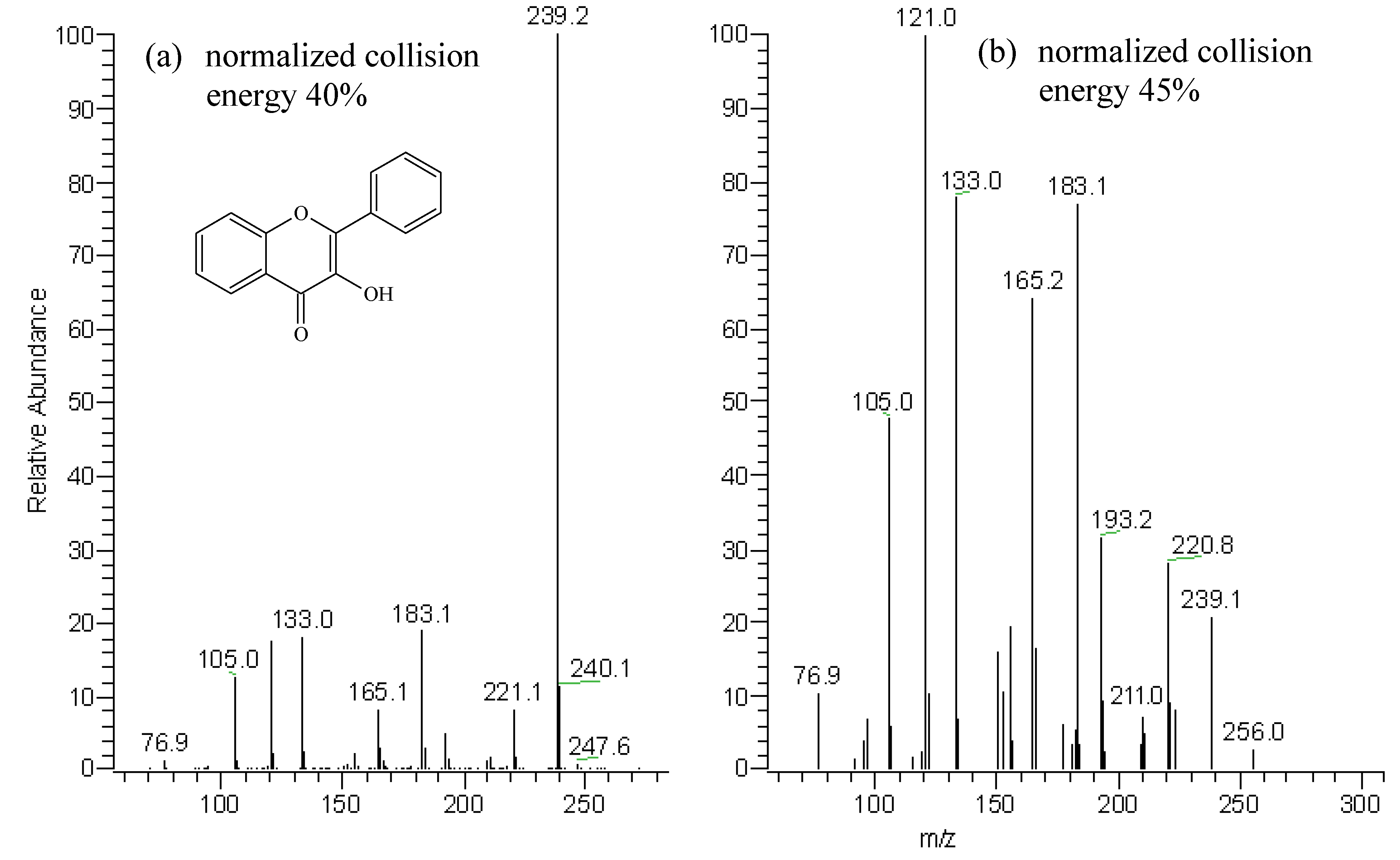
Identification of oregano flavonoid aglycones
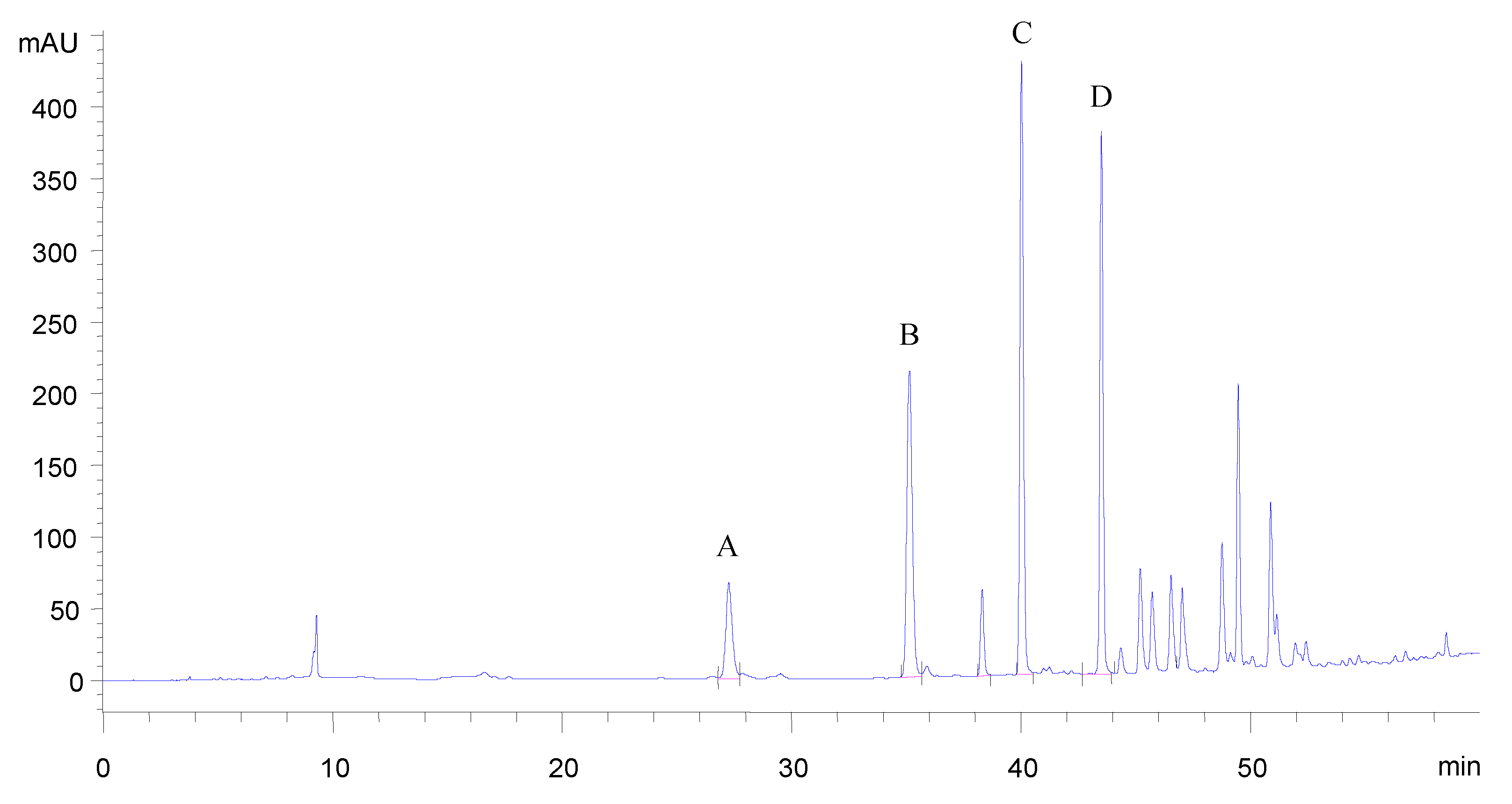
| Assessed fragments | Flavonoid aglycone peaks | |||
| A | B | C | D | |
| [M+H]+ | 305 | 289 | 289 | 273 |
| Defining subgroup fragments | ||||
| [M+H-H2O]+ | 287 (98) a | 271 (99) | 271 (15.5) | 255 (4) |
| [M+H-H2O-CO]+ | 259 (100) | 243 (100) | - | - |
| [M+H-H2O-2CO]+ | 231 (2) | 215 (3) | - | - |
| [M+H-CO]+ | - | - | - | - |
| [M+H-2CO]+ | - | - | - | - |
| [M+H-CH2CO]+ | - | - | 247 (5) | 231 (6) |
 dihydroflavonol- type fragments |  flavanone-type fragments | |||
| Defining A- & B-ring fragments | ||||
| 0,2A+ | - | - | - | - |
| 0,2B+ | - | - | - | - |
| 0,4B+ | - | - | 163 (100) | 147 (100) |
| 1,3A+ | 153 (35) | 153 (33) | 153 (21) | 153 (81) |
| 1,3B+ | - | - | - | - |
| 1,4B+ | 179 (2) | 163 (3) | - | - |
| [0,2A-H2O]+ | 149 (3) | 149 (4.5) | - | - |
| [M+H-B-ring]+ | 195 (16) | 195 (15) | 179 (20) | 179 (5) |
Conclusions
Experimental
Materials
Extraction of oregano flavonoid aglycones
HPLC-DAD Analysis
MS/MS Analysis
References
- Shahidi, F. Flavonoids as Antioxidants. In Natural Antioxidants. Chemistry, Health Effects and Practical Applications; AOCS Press: Champaign Illinois, 1997; pp. 174–194. [Google Scholar]
- Tsimogiannis, D.I.; Oreopoulou, V. Free-radical scavenging and antioxidant activity of 5,7,3´,4´-hydroxy-substituted flavonoids. Innov. Food Sci. Emerg. Technol. 2004, 5, 523–528. [Google Scholar] [CrossRef]
- Dangles, O.; Fargeix, G.; Dufour, C. One-electron oxidation of quercetin and quercetin derivatives in protic and non protic media. J. Chem. Soc, Perkin Trans. 2 1999, 1387–1395.
- Tsimogiannis, D.; Oreopoulou, V. Defining the role of flavonoid structure on cottonseed oil stabilization: Study of A- and C-ring substitution. JAOCS 2007, 84, 129–136. [Google Scholar]
- Tsimogiannis, D.I.; Oreopoulou, V. The contribution of flavonoid C-ring on the DPPH free radical scavenging efficiency. A kinetic approach for the 3´,4´-hydroxy substituted members. Innov. Food Sci. Emerg. Technol. 2006, 7, 140–146. [Google Scholar]
- Boué, M.S.; Carter-Wientjes, H.C.; Shih, Y.B.; Cleveland, E.T. Identification of flavone aglycones and glycosides in soybean pods by liquid chromatography–tandem mass spectrometry. J. Chromatogr. A. 2003, 991, 61–68. [Google Scholar] [CrossRef]
- Bohm, B. Extraction, Purification and Identification of Flavonoids. In Introduction to Flavonoids; Harwood Academic Publishers: Amsterdam, The Netherlands, 1998; pp. 200–204. [Google Scholar]
- Cuyckens, F.; Claeys, M. Mass spectrometry in the structural analysis of flavonoids. J. Mass Spectrom. 2004, 39, 1–15. [Google Scholar]
- Fabre, N.; Rustan, I.; de Hoffmann, E.; Quetin-Leclercq, J. Determination of flavone, flavonol, and flavanone aglycones by negative ion liquid chromatography electrospray ion trap mass spectrometry. J. Am. Soc. Mass Spectrom. 2001, 12, 707–715. [Google Scholar]
- March, E.R.; Miao, Xiu-Sheng. A fragmentation study of kaempferol using electrospray quadrupole time-of-flight mass spectrometry at high mass resolution. Int. J. Mass Spectrom. 2004, 231, 157–167. [Google Scholar]
- Hughes, J.R.; Croley, R.T.; Metcalfe, D.C.; March, E.R. A tandem mass spectrometric study of selected characteristic flavonoids. Int. J. Mass Spectrom. 2001, 210-211, 371–385. [Google Scholar]
- Wu, W.; Yan, C.; Li, L.; Liu, Z.; Liu, S. Studies on the flavones using liquid chromatography–electrospray ionization tandem mass spectrometry. J. Chromatogr. A 2004, 1047, 213–220. [Google Scholar]
- Wu, W.; Liu, Z.; Song, F.; Liu, S. Structural Analysis of Selected Characteristic Flavones by Electrospray Tandem Mass Spectrometry. Anal. Sci. 2004, 20, 1103–1105. [Google Scholar]
- Ma, Y.L.; Li, Q.M.; Van den Heuvel, H.; Claeys, M. Characterization of flavone and flavonol aglycones by collision-induced dissociation tandem mass spectrometry. Rapid Commun. Mass Spectrom. 1997, 11, 1357–1364. [Google Scholar] [CrossRef]
- Huck, C.W.; Buchmeiser, M.R.; Bonn, G.K. Fast analysis of flavonoids in plant extracts by liquid chromatography–ultraviolet absorbance detection on poly(carboxylic acid)-coated silica and electrospray ionization tandem mass spectrometric detection. J. Chromatogr. A 2001, 943, 33–38. [Google Scholar]
- Chen, L. J.; Games, D. E.; Jones, J.; Kidwell, H. Separation and Identification of Flavonoids in an Extract from the Seeds of Oroxylum indicum by CCC. J. Liq. Chromatogr. Relat. Technol. 2003, 26, 1623–1636. [Google Scholar]
- Huck, W.C.; Huber, G.C.; Ongania, K.H.; Bonn, K.G. Isolation and characterization of methoxylated flavones in the flowers of Primula veris by liquid chromatography and mass spectrometry. J. Chromatogr. A 2000, 870, 453–462. [Google Scholar] [CrossRef]
- Hasrat, J. A.; Pieters, L.; Claeys, M.; Vlietinck, A. Adenosine-1 Active Ligands: Cirsimarin, a Flavone Glycoside from Microtea debilis. J. Nat. Prod. 1997, 60, 638–641. [Google Scholar] [CrossRef]
- Stevens, J. F.; Ivancic, M.; Deinzer, M. L.; Wollenweber, E. A Novel 2-Hydroxyflavanone from Collinsonia Canadensis. J. Nat. Prod. 1999, 62, 392–394. [Google Scholar]
- Lai, Jia-Ping; Lim, Yew Heng; Su, Jin; Shen, Han-Ming; Ong, Choon Nam. Identification and characterization of major flavonoids and caffeoylquinic acids in three Compositae plants by LC/DAD-APCI/MS. J. Chromatogr. B 2007, in press (corrected proof). [Google Scholar]
- Grayer, R. J.; Kite, G. C.; Abou-Zaid, M.; Archer, L. J. The Application of Atmospheric Pressure Chemical Ionisation Liquid Chromatography– Mass Spectrometry in the Chemotaxonomic Study of Flavonoids: Characterisation of Flavonoids from Ocimum gratissimum var. gratissimum. Phytochem. Anal. 2000, 11, 257–267. [Google Scholar]
- Stevens, F.J.; Taylor, W.A.; Deinzer, L.M. Quantitative analysis of xanthohumol and related prenylflavonoids in hops and beer by liquid chromatography–tandem mass spectrometry. J. Chromatogr. A 1999, 832, 97–107. [Google Scholar]
- Le Gall, G.; DuPont, M.S.; Mellon, A.F.; Davis, L.A.; Collins, J.G.; Verhoeyen, E.M.; Colquhoun, J.I. Characterization and Content of Flavonoid Glycosides in Genetically Modified Tomato (Lycopersicon esculentum) Fruits. J. Agric. Food Chem. 2003, 51, 2438–2446. [Google Scholar] [CrossRef]
- Cren-Olivé, C.; Déprez, S.; Leburn, S.; Codeville, B.; Rolando, C. Characterization of methylation site of monomethylflavan-3-ols by liquid chromatography/electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2000, 14, 2312–2319. [Google Scholar]
- Tsimogiannis, D.; Stavrakaki, M.; Oreopoulou, V. Isolation and characterisation of antioxidant components from oregano (Origanum heracleoticum). Int. J. Food Sci. Technol. 2006, 41, (Supplement 1). 39–48. [Google Scholar] [CrossRef]
- Vekiari, S.A.; Oreopoulou, V.; Tzia, C.; Thomopoulos, C.D. Oregano flavonoids as lipid antioxidants. JAOCS 1993, 70, 483–487. [Google Scholar]
- Bocchini, P.; Russo, M.; Galletti, G.C. Pyrolysis – gas chromatography/mass spectrometry used as a microanalytical technique for the characterization of Origanum heracleoticum from Calabria, Southern Italy. Rapid Commun. Mass Spectrom. 1998, 12, 1555–1563. [Google Scholar] [CrossRef]
- Cavero, S.; Garcia-Risco, M.R.; Marin, F.R.; Jaime, L.; Santoyo, S.; Senorans, F. J.; Reglero, G.; Ibanez, E. Supercritical fluid extraction of antioxidant compounds from oregano. J. Supercrit. Fluids 2006, 38, 62–69. [Google Scholar] [CrossRef]
- Exarchou, V.; Godejohann, M.; Van Beek, T.A.; Gerothanassis, I.P.; Vervoort, J. LC-UV-Solid-Phase Extraction-NMR-MS Combined with a Cryogenic Flow Probe and its Application to the Identification of Compounds Present in Greek Oregano. Anal. Chem. 2003, 75, 6288–6294. [Google Scholar]
- Kosar, M.; Dorman, H.J.D.; Hiltunen, R. Effect of an acid treatment on the phytochemical and antioxidant characteristics of extracts from selected Lamiaceae species. Food Chem. 2005, 91, 525–533. [Google Scholar]
- Mercken, H.M.; Beecher, G.M. Liquid chromatographic method for the separation and quantification of prominent flavonoid aglycones. J. Chromatogr. A 2000, 897, 177–184. [Google Scholar]
- Sample availability: Samples are available from the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Tsimogiannis, D.; Samiotaki, M.; Panayotou, G.; Oreopoulou, V. Characterization of Flavonoid Subgroups and Hydroxy Substitution by HPLC-MS/MS. Molecules 2007, 12, 593-606. https://doi.org/10.3390/12030593
Tsimogiannis D, Samiotaki M, Panayotou G, Oreopoulou V. Characterization of Flavonoid Subgroups and Hydroxy Substitution by HPLC-MS/MS. Molecules. 2007; 12(3):593-606. https://doi.org/10.3390/12030593
Chicago/Turabian StyleTsimogiannis, Dimitrios, Martina Samiotaki, George Panayotou, and Vassiliki Oreopoulou. 2007. "Characterization of Flavonoid Subgroups and Hydroxy Substitution by HPLC-MS/MS" Molecules 12, no. 3: 593-606. https://doi.org/10.3390/12030593
APA StyleTsimogiannis, D., Samiotaki, M., Panayotou, G., & Oreopoulou, V. (2007). Characterization of Flavonoid Subgroups and Hydroxy Substitution by HPLC-MS/MS. Molecules, 12(3), 593-606. https://doi.org/10.3390/12030593





