Phase I and Phase II Ocular Metabolic Activities and the Role of Metabolism in Ophthalmic Prodrug and Codrug Design and Delivery
Abstract
:Introduction
1. Anatomy of the Eye
1a. Blood Flow and Membrane Properties of the Eye
1b. Transport and Distribution Mechanisms in the Eye
2. Metabolic Activity in the Eye
2a. Phase 1 metabolic activity in the eye
| Enzyme | Substrate | Ocular Tissue | Ref. |
|---|---|---|---|
| 7-Ethoxyresorufin-O-dealkylase (EROD) | 7-Ethoxyresorufin | Porcine ciliary body, and bovine ciliary body | 18, 10 |
| 7-Pentoxyresorufin-O-dealkylase (PROD) | 7-Pentoxyresorufin | Porcine ciliary body, and bovine ciliary body | 18, 10 |
| 7-Ethoxycoumarin O-deethylase (ECOD) | 7-Ethoxycoumarin | Bovine corneal epithelium, retinal pigment epithelium, retina, ciliary body and corneal epithelium | 12, 14 |
| Aryl hydrocarbon hydroxylase (AHH) | Benzo(a)pyrene | Bovine retina, cornea, iris, choroid, ciliary body; Bovine retinal pigment epithelium, corneal endothelium; Mouse eye; Chick embryo retinal pigmented epithelium | 12, 14, 15 |
| Benzphetamine demethylase (BPDM) | Benzphetamine | Bovine retinal pigment epithelium, and ciliary body | 12, 14 |
| Prostaglandin hydroxylase | Prostaglandin | Porcine iris, ciliary body | 13 |
| Cyclooxygenase | Arachidonic acid | Bovine and rabbit corneal epithelium; Bovine corneal epithelium, ciliary body and corneal endothelium | 12, 16 |
2b. Phase II metabolic activity in the eye
3. Metabolism-focused Ophthalmic-specific Drug Design and Delivery Systems
3a. The Prodrug Approach
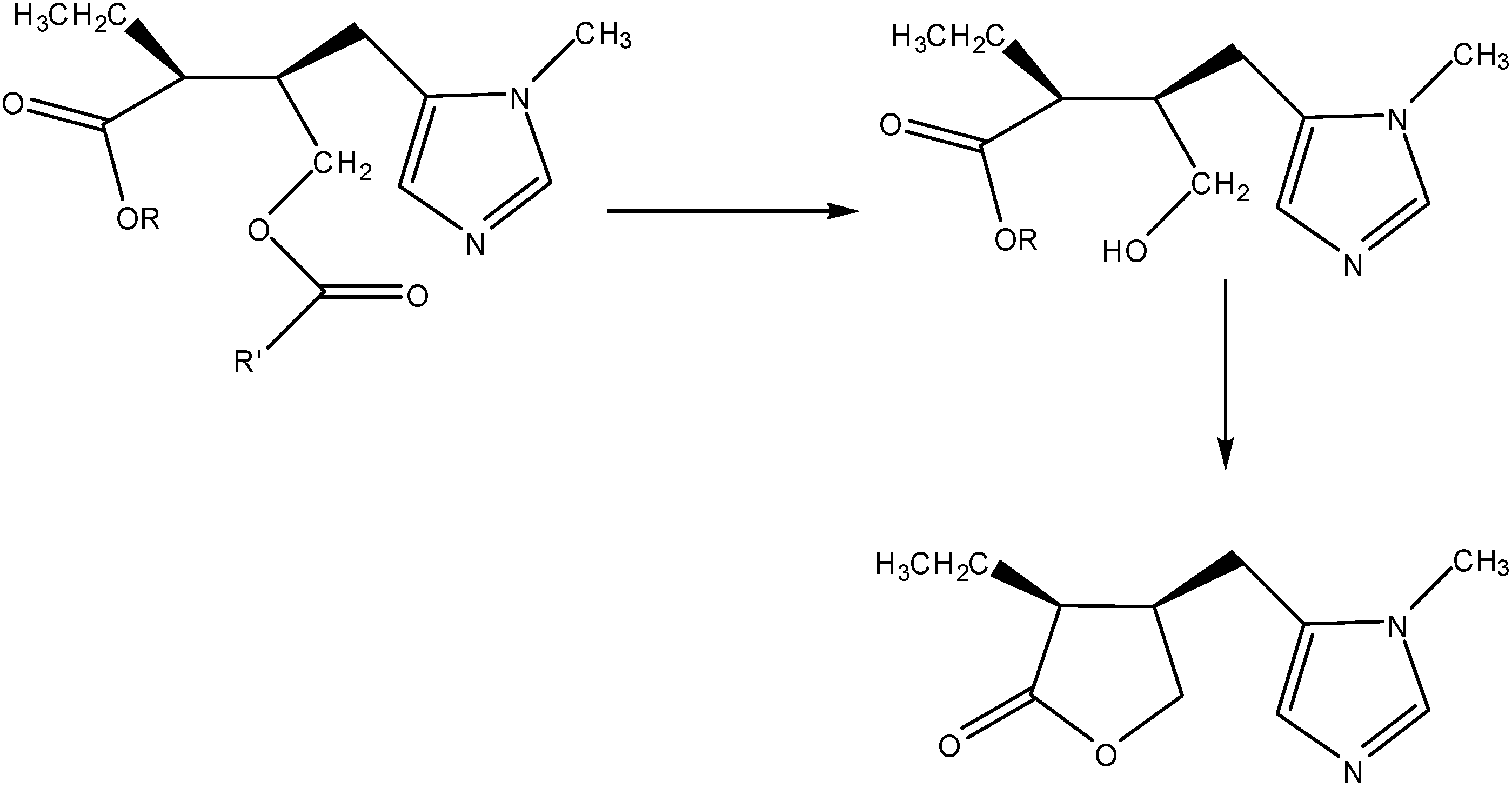
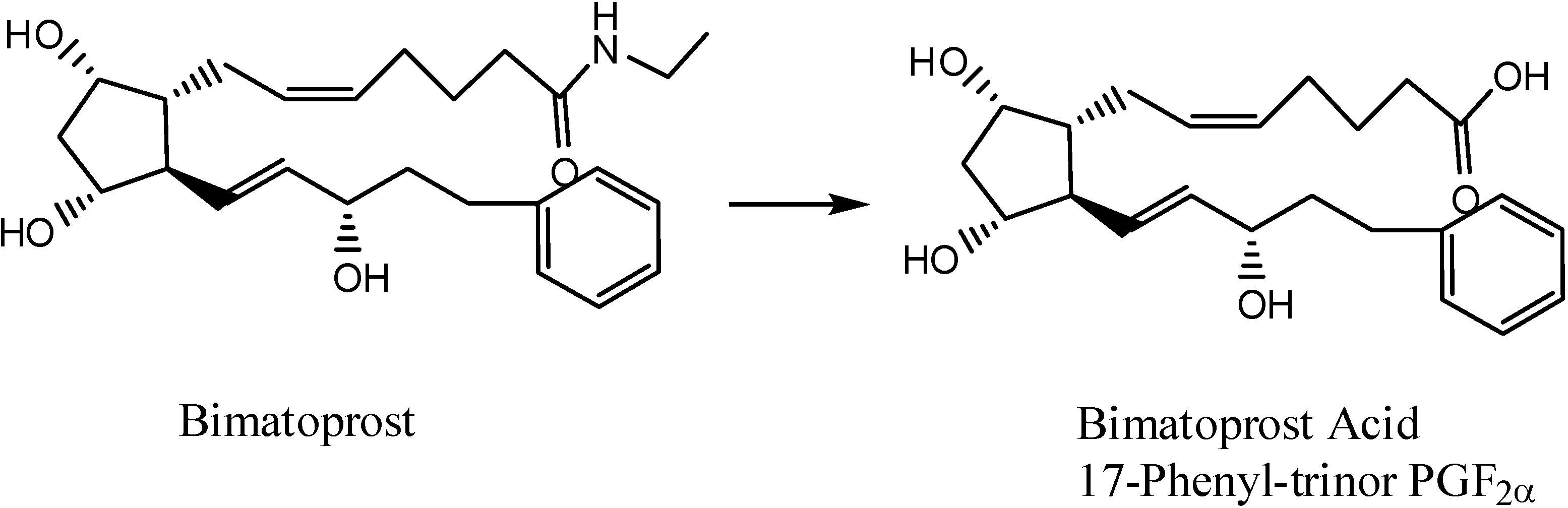
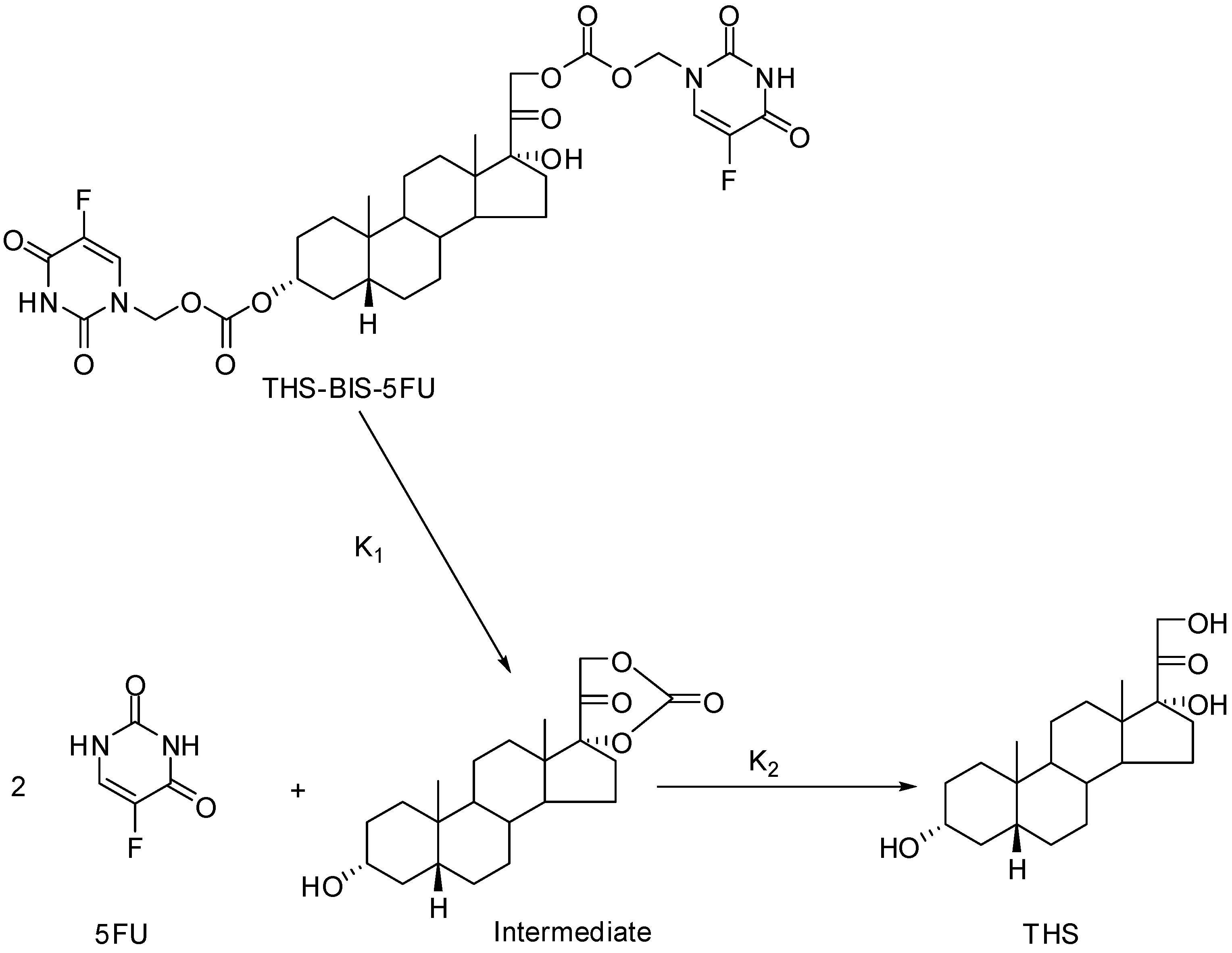
3b. The Codrug Approach
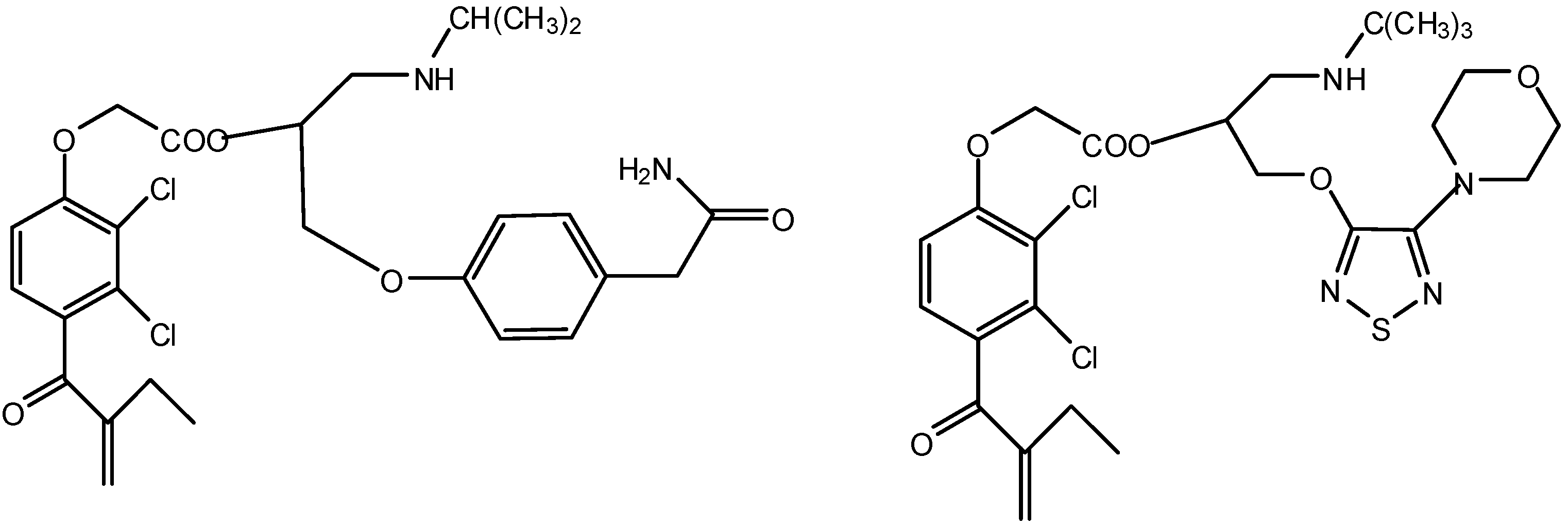
4. Enzyme Inhibitors to Modulate Prodrugs transport and metabolism
5. Eye Irritation and Corneal Damage Assessment
Conclusions
References and Notes
- Maurice, D.M.; Mishima, S. Ocular pharmacokinetics. Handbook of Experimental Pharmacology; Springer Verlag: Berlin-Heidelberg, 1984; Volume 69, (Pharmacol. Eye); pp. 19–116. [Google Scholar]
- Visor, G.C. Drug design strategies for ocular therapeutics. Adv. Drug Deliv. Rev. 1994, 14, 269–279. [Google Scholar] [CrossRef]
- Geroski, D.H.; Edelhauser, H.F. Transscleral drug delivery for posterior segment disease. Adv. Drug Deliv. Rev. 2001, 52, 37–48. [Google Scholar] [CrossRef]
- Duvvuri, S.; Majumdar, S.; Mitra, A.K. Drug delivery to the retina: challenges and opportunities. Expert Opin. Biol. Ther. 2003, 3, 45–56. [Google Scholar] [CrossRef]
- Duvvuri, S.; Majumdar, S.; Mitra, A.K. Role of metabolism in ocular drug delivery. Curr. Drug Metab. 2004, 5, 507–515. [Google Scholar] [CrossRef]
- Bodor, N.; Buchwald, P. Ophthalmic drug design based on the metabolic activity of the eye: soft drugs and chemical delivery systems. AAPS J. 2005, 7, 4. [Google Scholar]
- Guyto, A.C. Textbook of Medical Physiology, 8th Ed. ed; W.B. Saunders Co.: Philadelphia, PA, 1991. [Google Scholar]
- Leinweber, F.J. Drug Disposition in the mammalian eye and brain: a comparison of mechanisms. Drug Metab. Rev. 1991, 23, 133–246. [Google Scholar] [CrossRef]
- Moorthy, R.; Valluri, S. Ocular toxicity associated with systemic drug therapy. Curr. Opin Ophthalmol. 1999, 10, 438–46. [Google Scholar] [CrossRef]
- Jaanus, S.D. Ocular side effects of selected systemic drugs. Optom Clin. 1992, 2, 73–96. [Google Scholar]
- Bourlais, C.L.; Acar, L.; Zia, H.; Sado, P.A.; Needham, T.; Leverge, R. Ophthalmic drug delivery systems recent advances. Prog. Retin. Eye Res. 1998, 17, 33–58. [Google Scholar] [CrossRef]
- Kishida, K.; Matsumoto, K.; Manabe, R.; Sugiyama, T. Cytochrome P450 and related components of the microsomal electron transport system in the bovine ciliary body. Curr. Eye Res. 1986, 5, 529–533. [Google Scholar] [CrossRef]
- Schwartzman, M.L.; Abraham, N.G.; Masferrer, J.; Dunn, W.; Mcgiff, J.C. Cytochrome P450 dependent metabolism of arachidonic acid in bovine corneal epithelium. Biochem. Biophys. Res. Comm. 1985, 132, 343–351. [Google Scholar] [CrossRef]
- Schwartzman, M.L.; Masferrer, J.; Dunn, M.W.; Mcgiff, J.C.; Abraham, N.G. Cytochrome P450, drug metabolizing enzymes and arachidonic acid metabolism in bovine ocular tissues. Curr. Eye Res. 1987, 6, 623–630. [Google Scholar] [CrossRef]
- Asakura, T.; Shishi, H. Cytochrome P450-mediated prostaglandin omega/omega-1 hydroxylase activities in porcine ciliary body epithelial cells. Exp. Eye Res. 1992, 55, 377–384. [Google Scholar] [CrossRef]
- Shichi, H.; Atlas, S.A.; Nebert, D.W. Genetically regulated arylhydrocarbon hydroxylase induction in the eye: possible significance of the drug-metabolising enzyme system for the retinal pigmented epithelium-choriod. Exp. Eye Res. 1975, 21, 557–567. [Google Scholar] [CrossRef]
- Shichi, H.; Tsunematsu, Y.; Nebert, D.W. Aryl hydrocarbon hydroxylase induction in retinal pigmented epithelium: possible associated of genetic differences in a drug-metabolizing enzyme system with retinal degeneration. Exp. Eye Res. 1976, 23, 165–176. [Google Scholar] [CrossRef]
- Schwartzman, M.L.; Pageno, P.J.; Mcgriff, J.C.; Abraham, N.G. Immunochemical studies on the contribution of NADPH cytochrome P450 reductase to the cytochrome P450 dependent metabolism of arachidonic acid. Arch. Biochem. Biophys. 1987, 252, 635–645. [Google Scholar] [CrossRef]
- Shichi, H.; Nebert, D.W. Generic differences in drug metabolism associated with ocular toxicity. Envir. Health Perspec. 1982, 44, 107–117. [Google Scholar] [CrossRef]
- Sakamoto, S; Shichi, H. Induction of alkoxyresorufin O-dealkylases and UDP-glucuronosyl transferase by Phenobarbital and 3-methylcholanthrene in primary cultures of porcine ciliary epithelial cells. Biochem. Pharmacol. 1991, 41, 611–616. [Google Scholar]
- Shanthaveerappa, T.R.; Bourne, G.H. Monoamine oxidase distribution in the rabbit eye. J. Histochem. Cytochem. 1964, 12, 281–287. [Google Scholar] [CrossRef]
- Sparks, D.L.; Thomas, T.N.; Buckholtz, N.S. Monoamine oxidase activity in bovine retina: sub-cellular distribution and drug sensitivities. Neurosci. Lett. 1981, 21, 201–6. [Google Scholar] [CrossRef]
- Crabbe, M.J.C. Ocular diamine oxidase activity. Exp. Eye Res. 1985, 41, 777–778. [Google Scholar] [CrossRef]
- Kremzner, L.T.; Roy, D.; Spector, A. Polyamines in normal and cataractous human lenses. Evidence for post-translational modifications. Exp. Eye Res. 1983, 37, 649–659. [Google Scholar] [CrossRef]
- Lee, V.H.L. Esterase activities in adult rabbit eyes. J. Pharm. Sci. 1983, 72, 239–244. [Google Scholar] [CrossRef]
- Sanchez-Chavez, G.; Vidal, C. J.; Salceda, R. Acetyl- and butyrylcholinesterase activities in the rat retina and retinal pigment epithelium. J. Neurosci. Res. 1995, 41, 655–62. [Google Scholar] [CrossRef]
- Dutton, G.J. Glucuronidation of Drugs and Other Compounds; CRC Press: Boca Raton, FL, 1966. [Google Scholar]
- Coupland, S.E.; Penfold, P.L.; Billson, F.A. Hydrolases of anterior segment tissues in the normal human, pig and rat eye: a comparative study. Arch. Clin. Exp. Ophthalmol. 1994, 232, 182–92. [Google Scholar] [CrossRef]
- Coupland, S.E.; Hoffman, H.H.; Penfoldand, P.L.; Billson, F.A. Increased hydroxylase activities in the human trabecular meshwork of glaucomatous eyes. Ger. J. Ophthalmol. 1993, 2, 107–112. [Google Scholar]
- Physlactos, A.C. The lysosomal enzymes of the iris-ciliary body are retained in their organelles and exhibit increased activities during acute uveal inflamation. Acta Ophthamol. 1991, 69, 33–38. [Google Scholar] [CrossRef]
- Rao, P.V.; Zigler, J.S., Jr. Zeta-crystallin from guinea pig lens is capable of functioning catalytically as an oxidoreductase. Arch. Biochem. Biophys. 1991, 284, 181–185. [Google Scholar] [CrossRef]
- Gondhowiardojo, T.D.; van Haeringen, N.J.; Hoekzema, R.; Pels, L.; Kijlstra, A. Detection of aldehyde dehydrogenase activity in human corneal extracts. Curr. Eye Res. 1991, 10, 1001–1007. [Google Scholar]
- Godbout, R. High levels of aldehyde dehydrogenase transcripts in the undifferentiated chick retina. Exp. Eye Res. 1992, 54, 297–305. [Google Scholar] [CrossRef]
- Holmes, R.S.; Vandeberg, J.L. Ocular NAD-dependent alcohol dehydrogenase and aldyhyde dehydrogenase in the baboon. Exp. Eye Res. 1986, 43, 383–398. [Google Scholar]
- Watkins, J.; Wirthwein, D.P.; Sanders, R.A. Comparative study of phase II biotransformation in rabbit ocular tissues. Drug Metab. Dispos. 1991, 19, 708–13. [Google Scholar]
- Saneto, R.P.; Awasthi, Y.C.; Srivastava, S.K. Mercapturic acid pathway enzymes in bovine ocular lens, cornea, retina and retinal pigmented epithelium. Exp. Eye Res. 1982, 34, 107–111. [Google Scholar]
- Saneto, R.P.; Awasthi, Y.C.; Srivastava, S.K. Glutathione S-transferases of the bovine retina: evidence that glutathione peroxidase activity is the result of glutathione S-transferase. Biochem. J. 1982, 205, 213–217. [Google Scholar]
- Ahmed, H.; Singh, S.V.; Medh, R.D.; Ansari, G.A.S.; Kurosoku, A.; Awasthi, Y.C. Differential expression of Omega, Beta, and pi classes isozymes of glutathione S-transferase in bovine lens, cornea and retina. Arch. Biochem. Biophys. 1988, 266, 416–426. [Google Scholar] [CrossRef]
- Miller, L.; Stier, M.; Lovenburg, W. Evidence for the presence of N-acetyl transferase in the rat retina. Comp. Biochem. Physiol. 1980, 66, 213–216. [Google Scholar]
- Sastry, B.V.; Vidaver, P.S.; Janson, V.E.; Franks, J.J. S-adenosyl-L-methionine-mediated enzymatic methylations in the rat retinal membranes. J. Ocular Pharmacol. 1994, 10, 253–263. [Google Scholar] [CrossRef]
- Maurice, D.M.; Mishima, S. Pharmacology of the eye. In Handbook of experimental pharmacology; Sears, M.L., Ed.; Springer-Verlag: Berlin, Heidelberg, 1984; pp. 16–119. [Google Scholar]
- Mosher, G.L.; Mikkelson, T.J. Permeability of the N-alkyl p-aminobenzoate esters across the isolated corneal membrane of the rabbit. Int. J. Pharm. 1979, 2, 239–243. [Google Scholar] [CrossRef]
- Bundgaard, H.; Falch, E.; Larsen, C.; Mosher, G. L.; Mikkelson, T. J. Pilocarpic acid esters as novel sequentially labile pilocarpine prodrugs for improved ocular delivery. J. Med. Chem. 1985, 28, 79–81. [Google Scholar] [CrossRef]
- Bundgaard, H.; Falch, E.; Larsen, C.; Mosher, G. L.; Mikkelson, T. J. Pilocarpine prodrugs. II. Synthesis, stability, bioconversion, and physicochemical properties of sequentially labile pilocarpine acid diesters. J. Pharm. Sci. 1986, 75, 775–83. [Google Scholar]
- Druzgala, P.l; Winwood, D.; Drewniak-Deyrup, M.; Smith, S.; Bodor, N.; Kaminski, J.J. New water-soluble pilocarpine derivatives with enhanced and sustained muscarinic activity. Pharm. Res. 1992, 9, 372–7. [Google Scholar] [CrossRef]
- Druzgala, P.; Bodor, N. Water soluble pilocarpine prodrugs with sustained intraocular activity in normotensive rabbits and in glaucomatous beagles. J. Control. Release 1994, 28, 282–3. [Google Scholar] [CrossRef]
- Thombre, A.G.; Himmelstein, K.J. Quantitative evaluation of topically applied pilocarpine in the precorneal area. J. Pharm. Sci. 1984, 73, 219–22. [Google Scholar] [CrossRef]
- Meseguer, G.; Gurny, R.; Buri, P.; Rozier, A.; Plazonnet, B. Gamma scintigraphic study of precorneal drainage and assessment of miotic response in rabbits of various ophthalmic formulations containing pilocarpine. Int. J. Pharm. 1993, 95, 229–34. [Google Scholar] [CrossRef]
- Patton, T.F. Pharmacokinetic evidence for improved ophthalmic drug delivery by reduction of instilled volume. J. Pharm. Sci. 1977, 66, 1058–9. [Google Scholar] [CrossRef]
- Bundgaard, H.; Falch, E.; Larsen, C.; Mikkelson, T.J. Pilocarpine prodrugs I. Synthesis, physico-chemical properties and kinetics of lactonization of pilocarpic acid esters. J. Pharm. Sci. 1986, 75, 36–43. [Google Scholar] [CrossRef]
- Suhonen, P.; Jarvinen, T.; Peura, P.; Urtti, A. Permeability of pilocarpic acid diesters across albino rabbit cornea in vitro. Int. J. Pharm. 1991, 74, 221–8. [Google Scholar] [CrossRef]
- Saarinen-Savolainen, P.; Jaervinen, T.; Suhonen, P.; Urtti, A. Amphiphilic properties of pilocarpine prodrugs. Int. J. Pharm. 1996, 133, 171–178. [Google Scholar] [CrossRef]
- Suhonen, P.; Jaervinen, T.; Lehmussaari, K.; Reunamaeki, T.; Urtti, A. Ocular absorption and irritation of pilocarpine prodrug is modified with buffer, polymer, and cyclodextrin in the eyedrop. Pharm. Res. 1995, 12, 529–33. [Google Scholar]
- Jarho, P.; Jaervinen, K.; Urtti, A.; Stella, V.J.; Jaervlinen, T. Modified β-cyclodextrin (SBE7-β-CyD) with viscous vehicle improves the ocular delivery and tolerability of pilocarpine prodrug in rabbits. J. Pharm. Pharmacol. 1996, 48, 263–269. [Google Scholar] [CrossRef]
- Sznitowska, M.; Zurowska-Pryczkowska, K.; Janicki, S.; Jarvinen, T. Miotic effect and irritation potential of pilocarpine prodrug incorporated into a submicron emulsion vehicle. Int. J. Pharm. 1999, 184, 115–120. [Google Scholar] [CrossRef]
- Chrai, S.S.; Patton, T.F.; Mehta, A.; Robinson, J.R. Lacrimal and instilled fluid dynamics in rabbit eyes. J. Pharm. Sci. 1973, 62, 1112–1121. [Google Scholar] [CrossRef]
- Himmelstein, K.J.; Guvenir, I.; Patton, T.F. Preliminary pharmacokinetic model of pilocarpine uptake and distribution in the eye. J. Pharm. Sci. 1978, 67, 603–606. [Google Scholar] [CrossRef]
- Van Buskirk, E.M. Adverse reactions from timolol administration. Ophthalmology 1980, 87, 447–450. [Google Scholar]
- Nelson, W.L.; Fraunfelder, F.T.; Sills, J.M.; Arrowsmith, J.B.; Kuritsky, J.N. Adverse respiratory and cardiovascular events attributed to timolol ophthalmic solution, 1978-1985. Am. J. Ophthalmol. 1986, 102, 606–611. [Google Scholar]
- Sasaki, H.; Igarashi, Y.; Nishida, K.; Nakamura, J. Ocular delivery of the β-blocker, tilisolol, through the prodrug approach. Int. J. Pharm. 1993, 93, 49–60. [Google Scholar] [CrossRef]
- Hellberg, M.R.; Ke, T-L.; Haggard, K.; Klimko, P.G.; Dean, T.R.; Graff, G. The Hydrolysis of the Prostaglandin Analog Prodrug Bimatoprost to 17-Phenyltrinor PGF2α by Human and Rabbit Ocular Tissue. J. Ocular Pharm. Therapeut. 2003, 19, 97–103. [Google Scholar] [CrossRef]
- Hussain, A.; Truelove, J. E. Prodrug approaches to enhancement of physicochemical properties of drugs. IV. Novel epinephrine prodrug. J. Pharm. Sci. 1976, 65, 1510–12. [Google Scholar] [CrossRef]
- Howard-Sparks, M.; Al-Ghananeem, A.M.; Pearson, A.P.; Crooks, P.A. Evaluation of O3α-, O21-Di-(N1-methyloxycarbonyl-2,4-dioxo-5-fluoropyrimidinyl)17α-hydroxy-5β-pregnan-20-one as a novel potential antiangiogenic codrug. J. Enzyme Inhib. Med. Chem. 2005, 20, 417–428. [Google Scholar]
- Hamad, M.O.; Kiptoo, P.K.; Stinchcomb, A.L.; Crooks, P.A. Synthesis and hydrolytic behavior of two novel tripartate codrugs of naltrexone and 6β-naltrexol with hydroxybupropion as potential alcohol abuse and smoking cessation agents. Bioorg. Med. Chem. 2006, 14, 7051–7061. [Google Scholar] [CrossRef]
- Howard-Sparks, M.; Al-Ghananeem, A.; Pearson, A.; Crooks, P. A Novel Chemical Delivery System Comprising an Ocular Sustained Release Formulation of a 3α,17α,21-trihydroxy-5β-pregnan-20-one-BIS-5-Flourouracil Codrug. Drug Dev. Ind. Pharm. In Press.
- Cardillo, J.A.; Farah, M.E.; Mitre, J.; Morales, P.H.; Costa, R.A.; Melo, L.A.S.; Kuppermann, B.; Jorge, R.; Ashton, P. An intravitreal biodegradable sustained release naproxen and 5-fluorouracil system for the treatment of experimental post-traumatic proliferative vitreoretinopathy. Br. J. Ophthalmol. 2004, 88, 1201–5. [Google Scholar] [CrossRef]
- Cynkowska, G.; Cynkowski, T.; Al-Ghananeem, A.M.; Al-Ghananeem, A.A.; Guo, H.; Ashton, P.; Crooks, P.A. Novel antiglaucoma prodrugs and codrugs of ethacrynic acid. Bioorg. Med. Chem Lett. 2005, 15, 3524–7. [Google Scholar] [CrossRef]
- Cynkowska, G.; Cynkowski, T.; Guo, H.; Ashton, P.; Crooks, P. A. Synthesis of novel codrugs of prostaglandin F2α with β-adrenergic receptor blockers for the treatment of ocular diseases. In Book of Abstracts, 211th ACS National Meeting, New Orleans, LA, March 24-28, 1996.
- Katragadda, S.; Talluri, R.S.; Mitra, A.K. Simultaneous modulation of transport and metabolism of acyclovir prodrugs across rabbit cornea: An approach involving enzyme inhibitors. Int. J. Pharm. 2006, 320, 104–113. [Google Scholar] [CrossRef]
- Anand, B.S.; Mitra, A.K. Mechanism of Corneal Permeation of L-Valyl Ester of Acyclovir: Targeting the Oligopeptide Transporter on the Rabbit Cornea. Pharm. Res. 2002, 19, 1194–1202. [Google Scholar] [CrossRef]
- Balakrishnan, A.; Jain-Vakkalagadda, B.; Yang, C.; Pal, D.; Mitra, A.K. Carrier mediated uptake of L-tyrosine and its competitive inhibition by model tyrosine linked compounds in a rabbit corneal cell line (SIRC)-strategy for the design of transporter/receptor targeted prodrugs. Int. J. Pharm. 2002, 247, 115–125. [Google Scholar] [CrossRef]
- Katragadda, S.; Talluri, R.S.; Mitra, A.K. Modulation of P-glycoprotein-mediated efflux by prodrug derivatization: an approach involving peptide transporter-mediated influx across rabbit cornea. J. Ocul. Pharmacol. Ther. 2006, 22, 110–120. [Google Scholar] [CrossRef]
- Majumdar, S.; Nashed, Y. E.; Patel, K.; Jain, R.; Itahashi, M.; Neumann, D. M.; Hill, J. M.; Mitra, A. K. Dipeptide Monoester Ganciclovir Prodrugs for Treating HSV-1-Induced Corneal Epithelial and Stromal Keratitis: In Vitro and In Vivo Evaluations. J. Ocul. Pharmacol. Ther. 2005, 21, 463–474. [Google Scholar] [CrossRef]
- Ashton, P.; Smith, T.J.; Glavinos, P.G.; Conklin, J.D., Jr.; Crooks, P.A.; Riggs, R.M.; Cynkowski, T.; Cynkowska, G. Permeable, non-irritating prodrugs of nonsteroidal and steroidal agents. U.S. Pat. 5,681,964, 1997. [Google Scholar]
- Lallemand, F.; Furrer, P.; Felt-Baeyens, O.; Gex-Fabry, M.; Dumont, J.-M.; Besseghir, K.; Gurny, R. A novel water-soluble cyclosporine A prodrug: ocular tolerance and in vivo kinetics. Int. J. Pharm. 2005, 295, 7–14. [Google Scholar] [CrossRef]
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Al-Ghananeem, A.M.; Crooks, P.A. Phase I and Phase II Ocular Metabolic Activities and the Role of Metabolism in Ophthalmic Prodrug and Codrug Design and Delivery. Molecules 2007, 12, 373-388. https://doi.org/10.3390/12030373
Al-Ghananeem AM, Crooks PA. Phase I and Phase II Ocular Metabolic Activities and the Role of Metabolism in Ophthalmic Prodrug and Codrug Design and Delivery. Molecules. 2007; 12(3):373-388. https://doi.org/10.3390/12030373
Chicago/Turabian StyleAl-Ghananeem, Abeer M, and Peter A Crooks. 2007. "Phase I and Phase II Ocular Metabolic Activities and the Role of Metabolism in Ophthalmic Prodrug and Codrug Design and Delivery" Molecules 12, no. 3: 373-388. https://doi.org/10.3390/12030373
APA StyleAl-Ghananeem, A. M., & Crooks, P. A. (2007). Phase I and Phase II Ocular Metabolic Activities and the Role of Metabolism in Ophthalmic Prodrug and Codrug Design and Delivery. Molecules, 12(3), 373-388. https://doi.org/10.3390/12030373




