Synthesis of 1-Aryl-3-phenethylamino-1-propanone Hydrochlorides as Possible Potent Cytotoxic Agents
Abstract
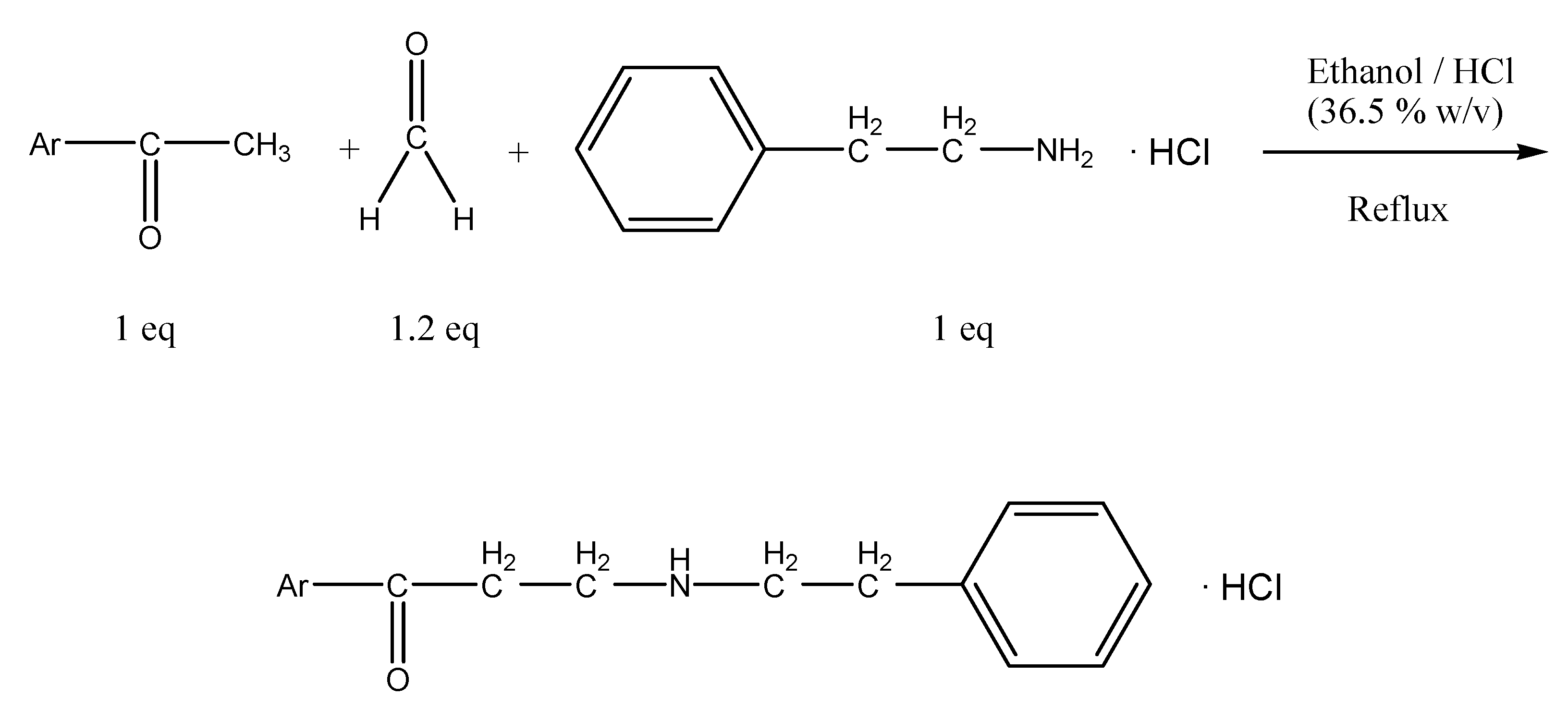
:Introduction
Results and Discussion

| Compound | 1H-NMR (DMSO-d6) | 13 C-NMR (DMSO-d6) |
|---|---|---|
| 1 | δ 2.96-3.17 (2H, m), 3.18-3.21 (2H, m), 3.28 (2H, t, J = 6.7 Hz), 3.55 (2H, t, J = 6.9 Hz), 7.22-7.34 (5H, m), 7.53-7.69 (3H, m), 7.96-7.98 (2H, m), 9.23 (1H, br s). | δ 32.2, 35.1, 42.4, 48.4, 127.4, 128.6, 129.31, 129.34, 129.5, 134.4, 136.5, 137.9, 197.6. |
| 2 | δ 2.37 (1H, s), 2.48-2.99 (2H, m), 3.18-3.37 (4H, m), 3.49 (2H, t, J = 6.9 Hz), 7.24-7.36 (7H, m), 7.87 (2H, d, J = 8.0 Hz ), 9.10 (1H, br s). | δ 21.8, 32.2, 34.9, 42.5, 48.4, 127.4, 128.7, 129.3, 129.4, 130.0, 134.1, 137.9, 144.8, 197.1. |
| 3 | δ 2.96-3.00 (2H, m), 3.16-3.20 (2H, m), 3.26 (2H, t, J = 6.9 Hz), 3.47 (2H, t, J = 6.7 Hz), 3.83 (3H, s), 7.94 (2H, d, J = 8.7 Hz ), 7.06 (2H, d, J = 8.7 Hz ), 7.22-7.34 (5H, m), 9.16 (1H, br s). | δ 32.2, 34.7, 42.6, 48.4, 56.3, 114.7, 127.4, 129.31, 129.33, 129.5, 131.0, 137.9, 164.2, 195.9. |
| 4 | δ 2.94-2.99 (2H, m), 3.17-3.21 (2H, m), 3.28 (2H, t, J = 6.5 Hz), 3.52 (2H, t, J = 6.7 Hz), 7.22-7.34 (5H, m), 7.63 (2H, d, J = 8.4 Hz ), 7.98 (2H, d, J = 8.7 Hz ), 9.10 (1H, br s). | δ 32.2, 35.1, 42.3, 48.4, 127.4, 129.3, 129.4, 129.6, 130.6, 135.3, 137.9, 139.2, 196.7. |
| 5 | δ 2.95-2.99 (2H, m), 3.16-3.20 (2H, m), 3.27 (2H, t, J = 6.7 Hz), 3.53 (2H, t, J = 6.9 Hz), 7.21-7.41 (7H, m), 8.05 (2H, dd, J = 8.7, 5.4 Hz ), 9.18 (1H, br s). | δ 32.2, 35.0, 42.4, 48.4, 116.5, 127.4, 129.3, 131.7, 133.3, 137.9, 164.7, 167.2, 196.2. |
| 6 | δ 2.95-3.00 (2H, m), 3.16-3.20 (2H, m), 3.27 (2H, t, J = 6.6 Hz), 3.52 (2H, t, J = 6.8 Hz), 7.22-7.34 (5H, m), 7.76 (2H, d, J = 8.4 Hz ), 7.90 (2H, d, J = 8.4 Hz ), 9.20 (1H, br s). | δ 32.2, 35.1, 42.3, 48.4, 127.4, 128.4, 129.31, 129.34, 130.6, 132.6, 135.6, 137.9, 196.9. |
| 7 | δ 2.96-3.00 (2H, m), 3.14-3.18 (2H, m), 3.26 (2H, t, J = 6.7 Hz), 3.50 (2H, t, J = 6.9 Hz), 7.20-7.33 (5H, m), 7.58 (1H, dd, J = 8.4, 2.4 Hz ), 7.73 (1H, d, J = 2.2 Hz), 7.82 (1H, d, J = 8.4 Hz ), 9.20 (1H, br s). | δ 32.2, 38.9, 42.1, 48.4, 127.4, 128.4, 129.30, 129.31, 130.9, 132.01, 132.05, 136.5, 137.4, 137.9, 198.4. |
| 8 | δ 3.12-3.16 (2H, m), 3.37-3.45 (2H, m), 3.57-3.61 (2H, m), 3.80 (2H, t, J = 6.7 Hz), 7.22-7.35 (5H, m), 8.24 (2H, d, J = 8.7 Hz), 8.37 (2H, d, J = 9.1 Hz), 11.09 (1H, br s). | δ 30.0, 33.8, 47.8, 54.3, 124.5, 127.4, 129.2, 129.5, 130.1, 137.7, 141.3, 150.8, 196.6. |
| 9 | δ 3.08-3.58 (8H, m), 6.88 (2H, d, J = 8.7 Hz), 7.21-7.34 (5H, m), 7.89 (2H, d, J = 8.7 Hz), 10.57(1H, s), 10.60 (1H, br s). | δ 30.0, 32.5, 48.3, 54.2, 115.9, 127.4, 128.3, 129.2, 129.5, 131.3, 137.7, 163.2, 195.4. |
| 10 | δ 2.95-2.99 (2H, m), 3.14-3.18 (2H, m), 3.26 (2H, t, J = 6.9 Hz), 3.48 (2H, t, J = 6.9 Hz), 7.22-7.34 (5H, m), 7.98 (2H, dd, J = 4.0, 1.1 Hz), 8.05 (2H, dd, J = 4.7, 1.1 Hz), 9.20 (1H, br s). | δ 32.2, 35.4, 42.3, 48.4, 127.4, 129.31, 129.33, 129.6, 134.5, 136.1, 137.9, 143.4, 190.5. |
| Compound | MW | UV* | MS m/z M+ | IR (KBr, cm-1) | ||
|---|---|---|---|---|---|---|
| λmax | logε | C(M) | C=O stretching | |||
| 1 | 289.80 | 244 | 4.12 | 5.1x10-5 | 253.3 | 1678 |
| 2 | 303.83 | 255 | 4.2 | 6.58x10-5 | 267.7 | 1677 |
| 3 | 319.83 | 286 | 3.81 | 12.5x10-5 | 283.4 | 1655 |
| 4 | 324.24 | 253 | 4.21 | 3.8x10-5 | 287.7 | 1675 |
| 5 | 307.79 | 245 | 4.03 | 6.5x10-5 | 271.6 | 1671 |
| 6 | 368.70 | 258 | 4.28 | 5.4x10-5 | 331.0 | 1672 |
| 7 | 427.58 | 248 | 3.85 | 13.9x10-5 | 321.3 | 1701 |
| 8 | 334.80 | 262 | 4.33 | 4.48x10-5 | 298.5 | 1695 |
| 9 | 305.80 | 283 | 4.48 | 3.2x10-5 | 269.3 | 1666 |
| 10 | 295.83 | 262, 287 | 3.95, 3.86 | 13.5x10-5 | 259.4 | 1651 |
Product characterization
Synthetic condition optimization
| Entry | Target compound | Reagent ratioa | Solvent | Acid | Time | Yield (%) |
|---|---|---|---|---|---|---|
| A | 1 | 1:1.2:1 | Ethanol | Yes | 7 hrs | 95 |
| B | 10 | 1:1.2:1 | Ethanol | Yes | 7 hrs | 93 |
| C | 1 | 1:1.2:1 | Ethanol | No | 13 hrs | 95 |
| D | 10 | 1:1.2:1 | Ethanol | No | 19 hrs | 96 |
| E | 1 | 2:2:1 | Ethanol | Yes | 8 hrs | 74 |
| F | 10 | 2:2:1 | Ethanol | Yes | 14 hrs | 69 |
| G | 1 | 2:2:1 | Ethanol | No | 7 hrs | 0 |
| H | 10 | 2:2:1 | Ethanol | No | 16 hrs | 74 |
| I | 1 | 2:2:1 | None | No | – b | 18c |
| J | 10 | 2:2:1 | None | No | – b | 16d |
| K | 1 | 1:1.2:1 | None | No | – b | 37 |
| L | 10 | 1:1.2:1 | None | No | – b | 47 |
- a Ketone:paraformaldehyde:phenethylamine hydrochloride
- b Reaction mixture heated to 83-86°C (ca. 10 minutes); then an exothermic reaction ocurred (see text for details)
- c 15 % of 1-phenethyl-3-benzoyl-4-phenyl-4-piperidinol was also formed
- d 21 % of 1-phenethyl-3-(2-thienylcarbonyl)-4-(2-thienyl)-4-piperidinol was also formed
Conclusions
Experimental
General
Synthesis of 1-aryl-3-phenethylamino-1-propanone hydrochlorides 1-10
| Compound | Ketone | Paraformaldehyde | Phenethylamine hydrochloride | Time (hours) | Crystallisation solvent | Yield % | Melting Point (°C) |
|---|---|---|---|---|---|---|---|
| 1 | 8.3 | 9.9 | 8.3 | 7 | Ethyl acetate | 95 | 169-171 |
| 2 | 7.4 | 8.9 | 7.4 | 8 | Methanol | 97 | 163-165 |
| 3 | 6.6 | 7.9 | 6.6 | 13 | Ethanol | 94 | 164-165a |
| 4 | 6.4 | 7.7 | 6.4 | 16 | Ethanol | 96 | 195-197b |
| 5 | 7.2 | 8.6 | 7.2 | 12 | Ethanol | 87 | 192-194c |
| 6 | 5.0 | 6.0 | 5.0 | 14 | Methanol. | 97 | 205-207 |
| 7 | 5.2 | 6.3 | 5.2 | 26 | Methanol | 91 | 167-169 |
| 8 | 6.0 | 7.2 | 6.0 | 8 | Methanol | 98 | 194-195 |
| 9 | 7.3 | 8.8 | 7.3 | 19 | Methanol | 93 | 167-169 |
| 10 | 7.9 | 9.5 | 7.9 | 7 | Ethanol | 93 | 156-157 |
Acknowledgments
References
- Dimmock, J. R.; Kumar, P. Anticancer and cytotoxic properties of Mannich bases. Curr. Med. Chem. 1997, 4, 1–22. [Google Scholar]
- Erciyas, E.; Erkaleli, H. I.; Cosar, G. Antimicrobial evaluation of some styryl ketone derivatives and related thiol adducts. J. Pharm. Sci. 1994, 83, 545–548. [Google Scholar] [CrossRef]
- Manavathu, E. K.; Vashishtha, S. C.; Alangaden, G. J.; Dimmock, J. R. In vitro antifungal activity of some Mannich bases of conjugated styryl ketones. Can. J. Microbiol. 1998, 44, 74–79. [Google Scholar] [CrossRef]
- Gul, H. I.; Denizci, A. A.; Erciyas, E. Antimicrobial evaluation of some Mannich bases of acetophenones and their representative quaternary derivatives. Arzneimittel-Forsch. 2002, 52, 773–777. [Google Scholar]
- Gul, H. I.; Ojanen, T.; Hanninen, O. Antifungal evaluation of some bis Mannich bases derived from acetophenone and their corresponding piperidinols and stability studies. Biol. Pharm. Bull. 2002, 25, 1307–1310. [Google Scholar]
- Gul, H. I.; Ojanen, T.; Vepsalainen, J.; Gul, M.; Erciyas, E.; Hanninen, O. Antifungal activity of some mono, bis and quaternary Mannich bases derived from acetophenone. Arzneimittel-Forsch. 2001, 51, 72–75. [Google Scholar]
- Gul, H. I.; Sahin, F.; Gul, M.; Ozturk, S.; Yerdelen, K. O. Evaluation of antimicrobial activities of several Mannich bases and their derivatives. Arch. Pharm. (Weinheim) 2005, 338, 335–338. [Google Scholar] [CrossRef]
- Gul, H. I.; Vepsalainen, J.; Gul, M.; Erciyas, E.; Hanninen, O. Cytotoxic activities of mono and bis Mannich bases derived from acetophenone against Renca and Jurkat cells. Pharm. Acta Helv. 2000, 74, 393–398. [Google Scholar] [CrossRef]
- Gul, M.; Gul, H. I.; Vepsalainen, J.; Erciyas, E.; Hanninen, O. Effect of acetophenone derived Mannich bases on cellular glutathione level in Jurkat cells: a possible mechanism of action. Arzneimittel-Forsch. 2001, 51, 679–682. [Google Scholar]
- Gul, H. I.; Gul, M.; Erciyas, E. Syntheses and stability studies of some Mannich bases of acetophenones and evaluation of their cytotoxicity against Jurkat cells. Arzneimittel-Forsch. 2002, 52, 628–635. [Google Scholar]
- Gul, H. I.; Gul, M.; Hänninen, O. Cytotoxic activities of some mono and bis Mannich bases derived from acetophenone in brine shrimp bioassay. Arzneimittel-Forsch. 2002, 52, 840–843. [Google Scholar]
- Gul, H. I.; Gul, M.; Vepsalainen, J.; Erciyas, E.; Hanninen, O. Cytotoxicity of some azines of acetophenone derived mono-Mannich bases against Jurkat cells. Biol. Pharm. Bull. 2003, 26, 631–637. [Google Scholar]
- Gul, M.; Gul, H. I.; Das, U.; Hanninen, O. Biological evaluation and structure-activity relationships of bis-(3-aryl-3-oxo-propyl)-methylamine hydrochlorides and 4-aryl-3-arylcarbonyl-1-methyl-4-piperidinol hydrochlorides as potential cytotoxic agents and their alkylating ability towards cellular glutathione in human leukemic T cells. Arzneimittel-Forsch. 2005, 55, 332–337. [Google Scholar]
- Gul, M.; Atalay, M.; Gul, H. I.; Nakao, C.; Lappalainen, J.; Hanninen, O. The effects of some Mannich bases on heat shock proteins HSC70 and GRP75, and thioredoxin and glutaredoxin levels in Jurkat cells. Toxicol. In Vitro 2005, 19, 573–580. [Google Scholar] [CrossRef]
- Suleyman, H.; Gul, H. I.; Gul, M.; Alkan, M.; Gocer, F. Anti-inflammatory activity of bis(3-aryl-3-oxo-propyl)methylamine hydrochloride in rat. Biol. Pharm. Bull. 2007, 30, 63–67. [Google Scholar]
- Atwal, M. S.; Bauer, l.; Dixit, S. N.; Gearien, J. E.; Megahy, M.; Morris, R.; Pokorny, C. Analgetics. II. Relationship between structure and activity of some beta-aminoketones. J. Med. Chem. 1969, 12, 994–997. [Google Scholar] [CrossRef]
- Chen, H.; Ji, Z.; Wong, L. K.; Siuda, J. F.; Narayanan, V. L. Synthesis, antiinflammatory, and cytotoxic activities of 2-alkyl and 2-benzyl-2-dimethylaminomethyl-5-(E)-arylidene cyclo-pentanone hydrochlorides. Pharm. Res. 1996, 13, 1482–1487. [Google Scholar] [CrossRef]
- Suleyman, H.; Gul, H. I.; Asoglu, M. Antiinflammatory activity of 3-benzoyl-1-methyl-4-phenyl-4-piperidinol hydrochloride. Pharmacol. Res. 2003, 47, 471–475. [Google Scholar] [CrossRef]
- Koechel, D. A.; Rankin, G. O. Diuretic activity of Mannich base derivatives of ethacrynic acid and certain ethacrynic acid analogues. J. Med. Chem. 1978, 21, 764–769. [Google Scholar] [CrossRef]
- Lee, C. M.; Plattner, J. J.; Ours, C. W.; Horrom, B. W.; Smital, J. R.; Pernet, A. G.; Bunnell, P. R.; El-Masry, S. E.; Dodge, P. W. [[(Aminomethyl)aryl]oxy]acetic acid esters. A new class of high-ceiling diuretics. 1. Effects of nitrogen and aromatic nuclear substitution. J. Med. Chem. 1984, 27, 1579–1587. [Google Scholar] [CrossRef]
- Borenstein, M. R.; Doukas, P. H. Anticonvulsant activity of indanylspirosuccinimide Mannich bases. J. Pharm. Sci. 1987, 76, 300–302. [Google Scholar] [CrossRef]
- Dimmock, J. R.; Jonnalagadda, S. S.; Phillips, O. A.; Erciyas, E.; Shyam, K.; Semple, H. A. Anticonvulsant properties of some Mannich bases of conjugated arylidene ketones. J. Pharm. Sci. 1992, 81, 436–440. [Google Scholar] [CrossRef]
- Gursoy, A.; Karali, N.; Buyuktimkin, S.; Demirayak, S.; Ekinci, A. C.; Ozer, H. Some 3-hydrazono-2-indolinones and N-Mannich bases as potential anticonvulsants. Farmaco 1996, 51, 437–442. [Google Scholar]
- Gul, H. I.; Calis, U.; Vepsalainen, J. Synthesis and evaluation of anticonvulsant activities of some bis-Mannich bases and corresponding piperidinols. Arzneimittel-Forsch. 2002, 52, 863–869. [Google Scholar]
- Gul, H. I.; Calis, U.; Vepsalainen, J. Synthesis of some mono-Mannich bases and corresponding azine derivatives and evaluation of their anticonvulsant activity. Arzneimittel-Forsch. 2004, 54, 359–364. [Google Scholar]
- Gul, M.; Gul, H. I.; Hanninen, O. Effects of Mannich bases on cellular glutathione and related enzymes of Jurkat cells in culture conditions. Toxicol. In Vitro 2002, 16, 107–112. [Google Scholar] [CrossRef]
- Roth, H. J.; Schwarz, D.; Hundeshagen, G. Photoreactions of phthalimide Mannich bases in methanol. Arch. Pharm. (Weinheim) 1976, 309, 48–52. [Google Scholar] [CrossRef]
- Collino, F.; de-Nardo, M. Mannich ketobases with narcotic antagonist activity. Boll. Chim. Farm. 1983, 122, 393–404. [Google Scholar]
- Prost, M.; Urbain, M.; Charlier, R. 3-Amino-4'-Hydroxybutyrophenones. Belg. Chim. Therapeut. 1970, 5, 303–311. [Google Scholar]
- Sample Availability: Samples of compounds 1-10 are available from the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Mete, E.; Gul, H.I.; Kazaz, C. Synthesis of 1-Aryl-3-phenethylamino-1-propanone Hydrochlorides as Possible Potent Cytotoxic Agents. Molecules 2007, 12, 2579-2588. https://doi.org/10.3390/12122579
Mete E, Gul HI, Kazaz C. Synthesis of 1-Aryl-3-phenethylamino-1-propanone Hydrochlorides as Possible Potent Cytotoxic Agents. Molecules. 2007; 12(12):2579-2588. https://doi.org/10.3390/12122579
Chicago/Turabian StyleMete, Ebru, Halise Inci Gul, and Cavit Kazaz. 2007. "Synthesis of 1-Aryl-3-phenethylamino-1-propanone Hydrochlorides as Possible Potent Cytotoxic Agents" Molecules 12, no. 12: 2579-2588. https://doi.org/10.3390/12122579




