Synthesis of New trans-2-Benzyl-3-(furan-2-yl)-4-substituted-1,2,3,4-tetrahydroisoquinolinones
Abstract
:Introduction
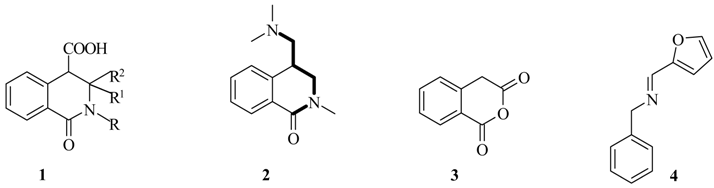
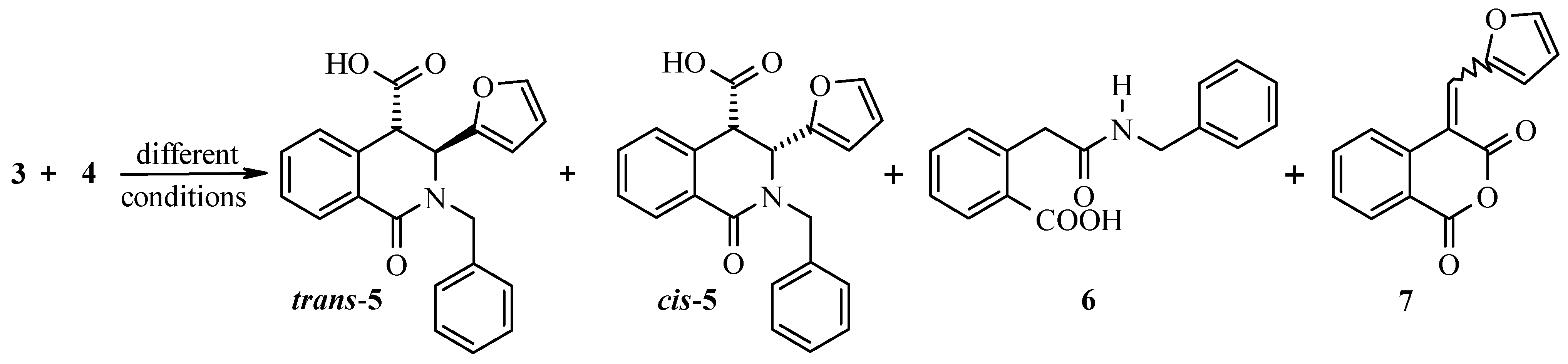
Results and Discussion

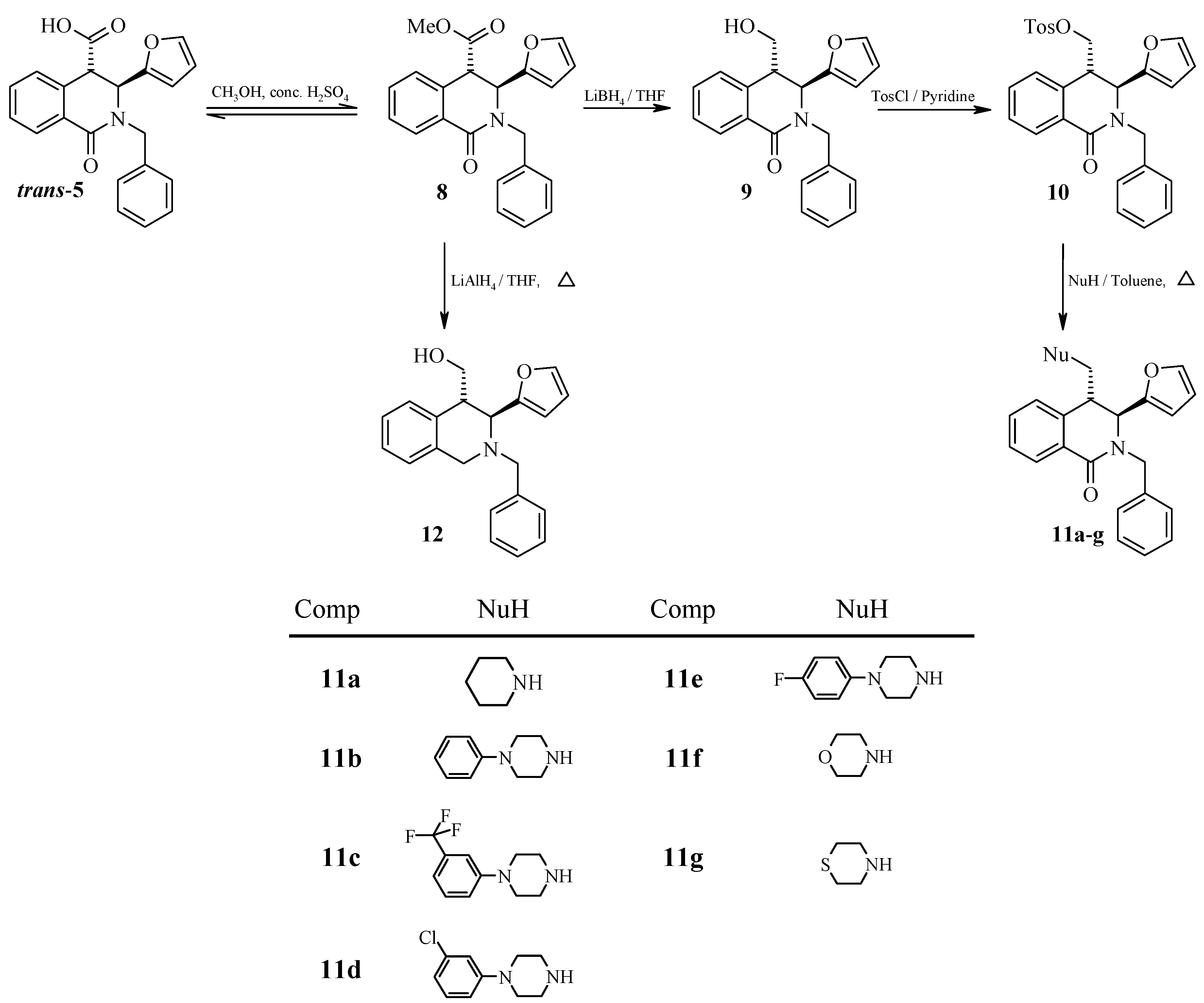
Conclusions
Experimental
General
(±)-trans- and (±)-cis-2-Benzyl-3-(furan-2-yl)-1-oxo-1,2,3,4-tetrahydro-4-isoquinoline Carboxylic Acid (trans-5 and cis-5) and By-products 6 and 7: General Procedure for Determination of the Ratios of the Products Obtained from 3 and 4 on a Small Scale Experiments
Reaction between 3 and 4 Performed on a Large Scale in Dichloroethane.
(±)-cis-2-Benzyl-3-(furan-2-yl)-1-oxo-1,2,3,4-tetrahydro-4-isoquinoline Carboxylic Acid (cis-5).
(±)-trans-2-Benzyl-3-(furan-2-yl)-1-oxo-1,2,3,4-tetrahydro-4-isoquinoline Carboxylic Acid (trans-5).
2-[2-(Benzylamino)-2-oxoethyl]benzenecarboxylic Acid (6).
4-(Furan-2-yl)methylidene-1H-isochromene-1,3-dione (7).
Reaction between 3 and 4 Performed on a Large Scale in Pyridine.
(±)-trans-Methyl-2-Benzyl-3-(furan-2-yl)-1-oxo-1,2,3,4-tetrahydro-4-isoquinoline Carboxylate (8).
(±)-trans-2-Benzyl-3-(furan-2-yl)-4-hydroxymethyl-1,2,3,4-tetrahydroisoquinolin-1-one (9).
(±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-tosyloxymethyl-1,2,3,4-tetrahydroisoquinolin-1-one (10).
General Procedure for the Preparation of (±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-(N,N-disubstitutedaminomethyl)-1,2,3,4-tetrahydroisoquinolin-1-ones (11a-g).
(±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-[(piperidin-1-yl)methyl)]-1,2,3,4-tetrahydroisoquinolin-1-one (11a).
(±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-[(4-phenyl-piperazin-1-yl)methyl)]-1,2,3,4-tetrahydroisoquinolin-1-one (11b).
(±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-[4-(3-trifluoromethyl-phenyl)-piperazin-1-yl)methyl)]-1,2,3,4-tetrahydroisoquinolin-1-one (11c).
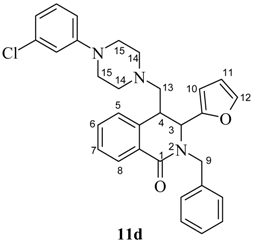
(±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-[4-(3-chlorophenyl)-piperazin-1-yl)methyl)]-1,2,3,4-tetrahydroisoquinolin-1-one (11d).
(±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-[4-fluorophenylpiperazin-1-yl)methyl)]-1,2,3,4-tetrahydroisoquinolin-1-one (11e).
(±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-[4-(morpholin-4-yl)methyl]-1,2,3,4-tetrahydroisoquinolin-1-one (11f).
(±)-trans-2-Benzyl-1-oxo-3-(furan-2-yl)-4-[4-(thiomorpholin-4-yl)methyl]-1,2,3,4-tetrahydroisoquinolin-1-one (11g).
(±)-trans-2-Benzyl-3-(furan-2-yl)-4-hydroxymethyl-1,2,3,4-tetrahydroisoquinoline (12).
Acknowledgements
References
- Kametani, T. The chemistry of the isoquinoline alkaloids; Elsevier: New York, 1969. [Google Scholar]
- Shamma, M.; Moniot, J. Isoquinoline alkaloid research: 1972-1977; Plenum Press: New York, 1978. [Google Scholar]
- Ito, N. 4-alkoxycarbonyl-1,2,3,4-tetrahydo-1-isoquinolones. Japanese Patent 6825,971, (1968). [Chem. Abstr. 1969, 70, 57685b]. [Google Scholar]
- Muller, G.; Poittevin, A. 2,3,10-Trimethoxy-8-oxoberbine. French Patent M. 5,415, (1967). [Chem. Abstr. 1969, 71, 91735y]. [Google Scholar]
- Bonnaud, B.; Carlessi, A.; Bigg, D. C. H. Synthesis of novel Isoquinoline derivatives as potential CNS-agents. J. Heterocycl. Chem. 1993, 30, 257–265. [Google Scholar] [CrossRef]
- Gitto, R.; Barreca, M.; Francica, E.; Caruso, R.; De Luca, L.; Russo, E.; De Sarro, G.; Chimirri, A. Synthesis and anticonvulsant properties of 1,2,3,4-tetrahydroisoquinolin-1-ones. Arkivoc 2004, 170–180. [Google Scholar] [CrossRef]
- Venkov, A. P.; Temnyalova, T. A.; Ivanov, I. I. Synthesis of N, N′-bis(1,2,3,4-tetrahydroisoquinolinyl)dicarboxamides by an intramolecular α-diamidoalkylation reaction. Synth. Comm. 1995, 25, 1419–1425. [Google Scholar] [CrossRef]
- Kim, H. J.; Yoon, U. C.; Yung, Young-Shik; Park, N. S.; Cederstrom, E. M.; Mariano, P. S. Oxidative Pictet-Spengler cyclization. J. Org. Chem. 1998, 63, 860–863. [Google Scholar]
- Kane, T. R.; Ly, C. Q.; Kelly, D. E.; Denner, J. M. Solid-phase synthesis of 1,2,3,4-tetrahydroisoquinoline derivatives emploing suppord-bound tyrosine esters in the Pictet-Spengler reaction. J. Comb. Chem. 2004, 6, 564–572. [Google Scholar] [CrossRef] [PubMed]
- Jones, R. C. F.; Smallridge, M. J.; Chapleo, C. B. Tetrahydrofolate Coenzyme models: Synthesis of tetrahydroimidazoisoquinolines and tetrahydroimidazoquinolines. J. Chem. Soc. Perkin Trans 1 1990, 385–391. [Google Scholar] [CrossRef]
- Zalán, Z.; Martinek, T. A.; Lázár, L.; Fülöp, F. Synthesis and conformational analysis of 1,3,2-diazaphosphorino[6,1-a]isoquinolines, a new ring system. Tetrahedron 2003, 59, 9117–9125. [Google Scholar]
- Naramsimhan, N. S.; Patil, P. A. An efficient Synthesis of 4-aryl-1,2,3,4-tetrahydroisoquinolines. J. Chem. Soc., Chem. Commun. 1987, 191–192. [Google Scholar]
- Boyd, G. V.; Monteli, R. L.; Lindley, P. F.; Mahmoud, M. M. The chemistry of N-substituted 3-amino-1H-2-benzopyran-1-ones and 5-amino-2,3-dihydrofuran-2-ones. Ene-type reactions involving transfer of acyl groups. X-Ray crystal structure of cis-3,4-dihydro-4-morpholinocarbonyl-3-p-nitrophenyl-1H-2-benzopyran-1-one. J. Chem. Soc. PerkinTrans I 1978, 1351–1360. [Google Scholar]
- Semple, J. E.; Rydzewski, R. M.; Gardner, G. An efficient synthetic route to ethyl 2-aryl-4-hydroxy-1,3(2H,4H)-dioxoisoquinoline-4-carboxylates. J. Org. Chem. 1996, 61, 7967–7972. [Google Scholar] [CrossRef] [PubMed]
- Haimova, M. A.; Mollov, N. M.; Ivanova, S. C.; Dimitrova, A. I. A highly stereoselective synthesis of 3,4-dihydro-1(2H)-isoquinolinones and 8-oxoberbines from homophthalic anhydride and azomethines. Tetrahedron 1977, 33, 331–336. [Google Scholar] [CrossRef]
- Atanassova, I.; Haimova, M.; Stanoeva, E. Curtius rearrangement of 1(2H)-isoquinolinone and 1H-2-benzopyran-1-one carboxylic acids. Comm. Dept. Chem. Bulg. Acad. Sci. 1984, 17, 172–179. [Google Scholar]
- Cushman, M.; Gentry, J.; Dekow, F. W. Condensation of imines with homophthalic anhydrides. A convergent synthesis of cis- and trans-13-methyltetrahydroprotoberberines. J. Org. Chem. 1977, 42, 1111–1116. [Google Scholar]
- Cushman, M.; Madaj, E. J. A study and mechanistic interpretation of the electronic and steric effects that determine the stereochemical outcome of the reaction of Schiff bases with homophthalic anhydride and 3-phenylsuccinic anhydride. J. Org. Chem. 1987, 52, 907–915. [Google Scholar] [CrossRef]
- Cushman, M.; Chen, Jer-kang. Utilization of the ferrocenyl-2-methylpropyl substituent as a chiral auxiliary in the asymmetric synthesis of the benzophenanthridine alkaloids (+)- and (-)-corynoline. J. Org. Chem. 1987, 52, 1517–1521. [Google Scholar]
- Georgieva, A.; Stanoeva, E.; Spassov, S.; Macicek, J.; Angelova, O.; Haimova, M.; De Kimpe, N. Reactions of α-chloroimines with homophthalic anhydrides. Synthesis and molecular structure of 3,3a-dihydrofuro[3,4-c]isoquinoline-1,5(4H,9bH)-diones, furo[3′,4′:9,9a}-8,9,16,16a-tetrahydro-1H,3H,11H-dibenzo[a,g]quinolizine-1,11-diones and related compounds. Tetrahedron 1991, 47, 3375–3388. [Google Scholar]
- Kozekov, I. D.; Koleva, R. I.; Palamareva, M. D. New trans/cis tetrahydroisoquinolines. 1. trans-2-Benzyl-3-(1-methyl-1H-pyrrol-2-yl)-4-substituted-1,2,3,4-tetrahydroisoquinolin-1-ones and corresponding tetrahydroisoquinolines. J. Heterocyclic Chem. 2002, 39, 229–236. [Google Scholar]
- Stoyanova, M. P.; Kozekov, I. D.; Palamareva, M. D. New trans/cis tetrahydroisoquinolines. 2. trans- and cis-3-(1-methyl-1H-pyrrol-2-yl)-1-(2H)-oxo2-phenethyl-1,2,3,4-tetrahydroisoquinolin-4-carboxylic acids and subsequent transformation. J. Heterocycl. Chem. 2003, 40, 795–803. [Google Scholar]
- Georgieva, A.; Stanoeva, E.; Karamfilova, K.; Spassov, S.; Angelova, O.; Haimova, M.; De Kimpe, N.; Boelens, M. Synthesis of 11H-4b, 10b-dihydro[1]benzopyrano[4,3-c]isoquinoline-6,11(5H)-diones and 13H-6c,12b-dihydronaphthol[1′,2′:5,6]-pyrano[4,3-c]isoquinoline-8,13-(7H)- dione from homophthalic anhydride and N-(2-hydroxyarylidene)alkylamines. Tetrahedron 1994, 50, 9399–9410. [Google Scholar] [CrossRef]
- Georgieva, A.; Spassov, S.; Stanoeva, E.; Topalova, I.; Tchanev, Ch. Effect of the structure of 1-aza-1,3-dienes on 1,2-versus 3,4-selectivity in cycloaddition reactions with homophthalic anhydride. J. Chem. Res. (S) 1997, 148–149. [Google Scholar] [CrossRef]
- Yadav, J. S.; Reddy, B. V. S.; Saritha Raj, K.; Prasad, A. R. Room temperature ionic liquid promoted three component coupling reactions: a facile synthesis of cis- isoquinolonic acids. Tetrahedron 2003, 59, 1805–1809. [Google Scholar] [CrossRef]
- Azizian, J.; Mohammadi, A. A.; Karimi, A. R.; Mohammadizadeh, M. R. A stereoselective three-component reaction: KAl(SO4)2 12H2O, an efficient and reusable catalyst for the one-pot synthesis of cis-isoquinolonic acids. J. Org. Chem. 2005, 70, 350–352. [Google Scholar] [CrossRef] [PubMed]
- El-Rayyes, N. R.; Ali, A. H. A. The Stobbe condensation with dimethyl homophthalate. II. J. Heterocycl. Chem. 1976, 13, 83–88. [Google Scholar]
- Sample Availability: Compounds cis-5, trans-5,6-9, trans-11a-e, and trans-11g are available in 20 mg amounts from the authors.
© 2006 by MDPI (http:www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Kandinska, M.I.; Kozekov, I.D.; Palamareva, M.D. Synthesis of New trans-2-Benzyl-3-(furan-2-yl)-4-substituted-1,2,3,4-tetrahydroisoquinolinones. Molecules 2006, 11, 403-414. https://doi.org/10.3390/11060403
Kandinska MI, Kozekov ID, Palamareva MD. Synthesis of New trans-2-Benzyl-3-(furan-2-yl)-4-substituted-1,2,3,4-tetrahydroisoquinolinones. Molecules. 2006; 11(6):403-414. https://doi.org/10.3390/11060403
Chicago/Turabian StyleKandinska, Meglena I., Ivan D. Kozekov, and Mariana D. Palamareva. 2006. "Synthesis of New trans-2-Benzyl-3-(furan-2-yl)-4-substituted-1,2,3,4-tetrahydroisoquinolinones" Molecules 11, no. 6: 403-414. https://doi.org/10.3390/11060403
APA StyleKandinska, M. I., Kozekov, I. D., & Palamareva, M. D. (2006). Synthesis of New trans-2-Benzyl-3-(furan-2-yl)-4-substituted-1,2,3,4-tetrahydroisoquinolinones. Molecules, 11(6), 403-414. https://doi.org/10.3390/11060403




