An Electron Transfer Approach to the Preparation of Highly Functionalized Anthraquinones
Abstract
:Introduction

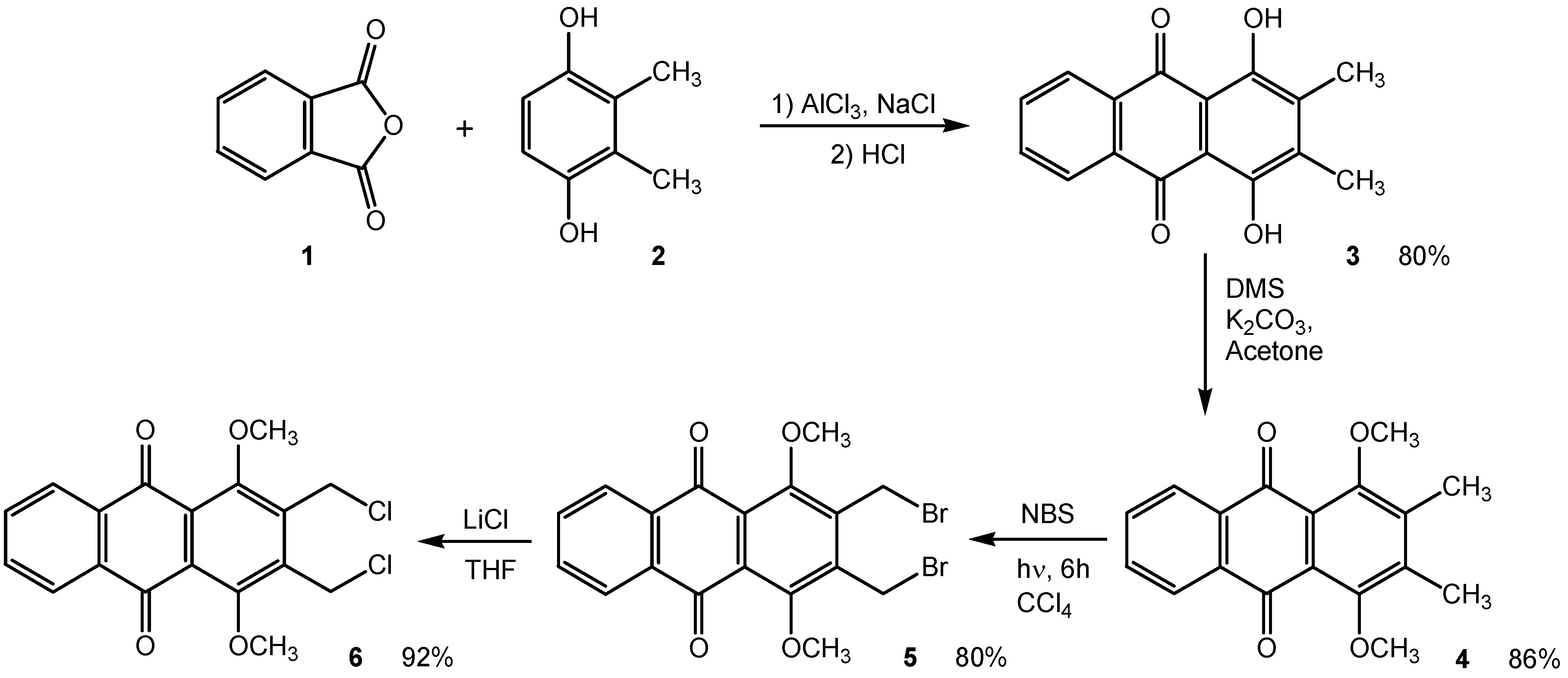
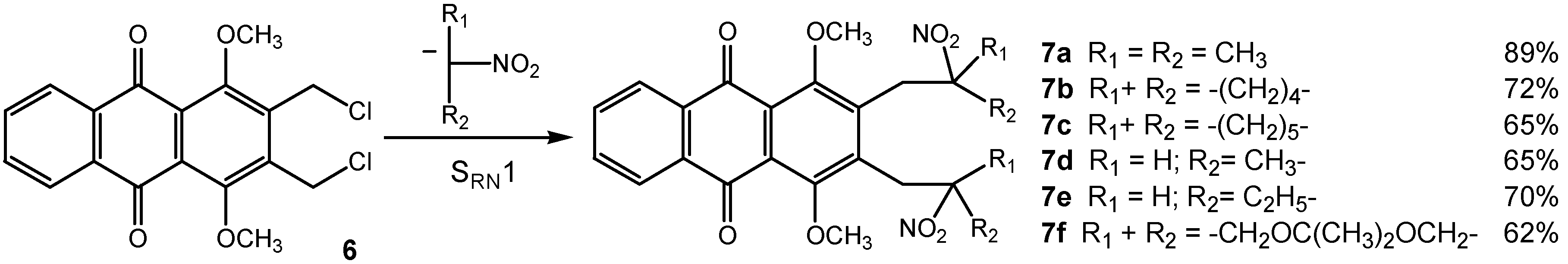
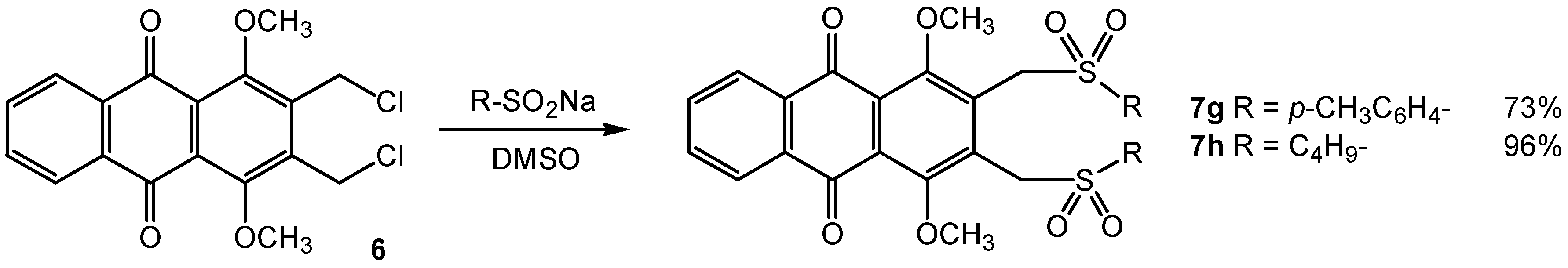
Results and Discussion
Conclusions
Experimental
General
2,3-Bis(chloromethyl)-1,4-dimethoxyanthraquinone (6).
General procedure for bis-SRN1 reaction with aliphatic and cyclic nitronate anions.
General procedure for reactions with substituted sulfinate sodium salt
References and Notes
- Lin, A. J.; Cosby, L. A.; Shansky, C. W.; Sartorelli, A. C. Potential bioreductive alkylating agents. 1. Benzoquinone derivatives. J. Med. Chem. 1972, 15, 1247. [Google Scholar]
- Moore, H. W. Bioactivation as a model for drug design bioreductive alkylation. Science 1977, 197, 527. [Google Scholar] [CrossRef]
- Arcamone, F. Properties of antitumor anthracyclines and new developments in their application: Cain Memorial Award Lecture. Cancer Res. 1985, 45, 5995. [Google Scholar]
- Vanelle, P.; Terme, T.; Giraud, L.; Crozet, M. P. Progress in electron transfer reactions of new quinone bioreductive alkylating agents. Recent Adv. Devel. Org. Chem. 2000, 4, 1. [Google Scholar]
- Terme, T.; Crozet, M. P.; Maldonado, J.; Vanelle, P. Electron Transfer Reactions in Organic Synthesis; Vanelle, P., Ed.; Research Signpost: Trivandrum, 2002; p. 1. [Google Scholar]
- Kerdesky, F. A. J.; Ardecky, R. J.; Lakshmikantham, M. V.; Cava, M. P. Simple o-quino-dimethane route to (±)-4-demethoxydaunomycinone. J. Am. Chem. Soc. 1981, 103, 1992. [Google Scholar] [CrossRef]
- Terme, T.; Crozet, M. P.; Giraud, L.; Vanelle, P. Original annulation in anthraquinone series: synthesis of new 2,3-dialkylnaphthacene-5,12-diones. Tetrahedron 2000, 56, 1097. [Google Scholar] [CrossRef]
- Liu, L. K.; Chi, Y.; Jen, K.-Y. Copper-catalyzed additions of sulfonyl iodides to simple and cyclic alkenes. J. Org. Chem. 1980, 45, 406. [Google Scholar] [CrossRef]
- Gilbert, K. E.; Borden, W. T. Peracid oxidation of aliphatic amines: general synthesis of nitroalkanes. J. Org. Chem. 1979, 44, 659. [Google Scholar] [CrossRef]
- Linden, G. B.; Gold, M. H. Preparation of 2- and 5-substituted 1,3-dioxanes. J. Org. Chem. 1956, 21, 1175. [Google Scholar]
- Samples Availability: Available from the authors.
© 2005 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Beziane, A.; Terme, T.; Vanelle, P. An Electron Transfer Approach to the Preparation of Highly Functionalized Anthraquinones. Molecules 2005, 10, 289-294. https://doi.org/10.3390/10010289
Beziane A, Terme T, Vanelle P. An Electron Transfer Approach to the Preparation of Highly Functionalized Anthraquinones. Molecules. 2005; 10(1):289-294. https://doi.org/10.3390/10010289
Chicago/Turabian StyleBeziane, Abdelouahab, Thierry Terme, and Patrice Vanelle. 2005. "An Electron Transfer Approach to the Preparation of Highly Functionalized Anthraquinones" Molecules 10, no. 1: 289-294. https://doi.org/10.3390/10010289
APA StyleBeziane, A., Terme, T., & Vanelle, P. (2005). An Electron Transfer Approach to the Preparation of Highly Functionalized Anthraquinones. Molecules, 10(1), 289-294. https://doi.org/10.3390/10010289



