Coatings were deposited on glass substrates by dip coating and slot-die. A silicon oxide layer was pre-deposited on each substrate to prevent the migration of Na+ cations. Three types of photocatalytic layers were prepared using different surfactants. Specifically, the selected surfactants were ammonium lauryl sulfate, cetyltrimethylammonium bromide and polyoxyethylene sorbitan monooleate, which gave rise to porous photocatalytic coatings labeled PPC-ALS, PPC-CTAB and PPC-TWEEN. Besides, a fourth type of photocatalytic coating (PPC-0) was prepared without using any surfactant in the sol formulation. Further details on the preparation of the coatings are presented in the methods section.
2.1. Characterization of the Photocatalyst
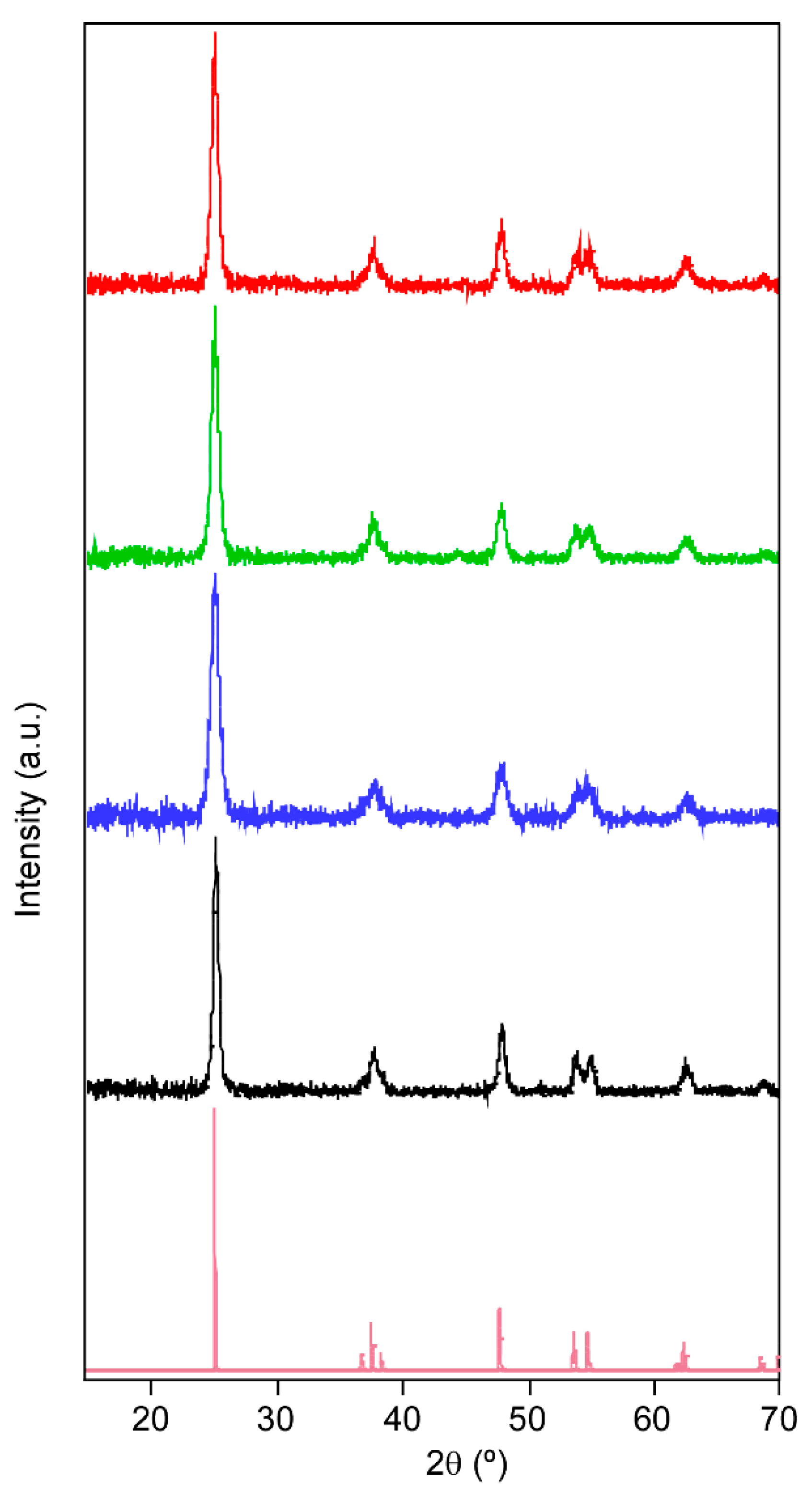
The PXRD analysis of xerogels showed anatase as the main crystalline phase and no traces of rutile were observed on the diffractograms (
Figure 1). Therefore, the heat treatment conditions were suitable, and the presence of surfactant did not affect the obtained crystalline phase.
A broadening of the peaks is observed (
Figure 2) depending on whether or not a surfactant is used and the type used, which can be related to the crystal size formed. This size has a direct influence on the kinetics of the reaction because by reducing the crystal size the photoactive area will be increased. The crystallite size (
Dhkl) was calculated from the Scherrer equation:
where
K is the particle shape factor (0.94),
λ is the X-ray wavelength,
βhkl is the half-width of reflection, and
θ is the Bragg angle corresponding to the reflection [
13,
14].
Table 1 shows the full width at half maximum (FWHM) for the reflection (101) and estimated crystallite size (
Dhkl). All coatings present nanoscopic crystallites. Without the presence of tensioactive, the size of crystallites is ca. 17 nm. However, the addition of a surfactant into the sol formulation promotes a meaningful size reduction, achieving the smallest crystallite with the formulation containing ALS surfactant.
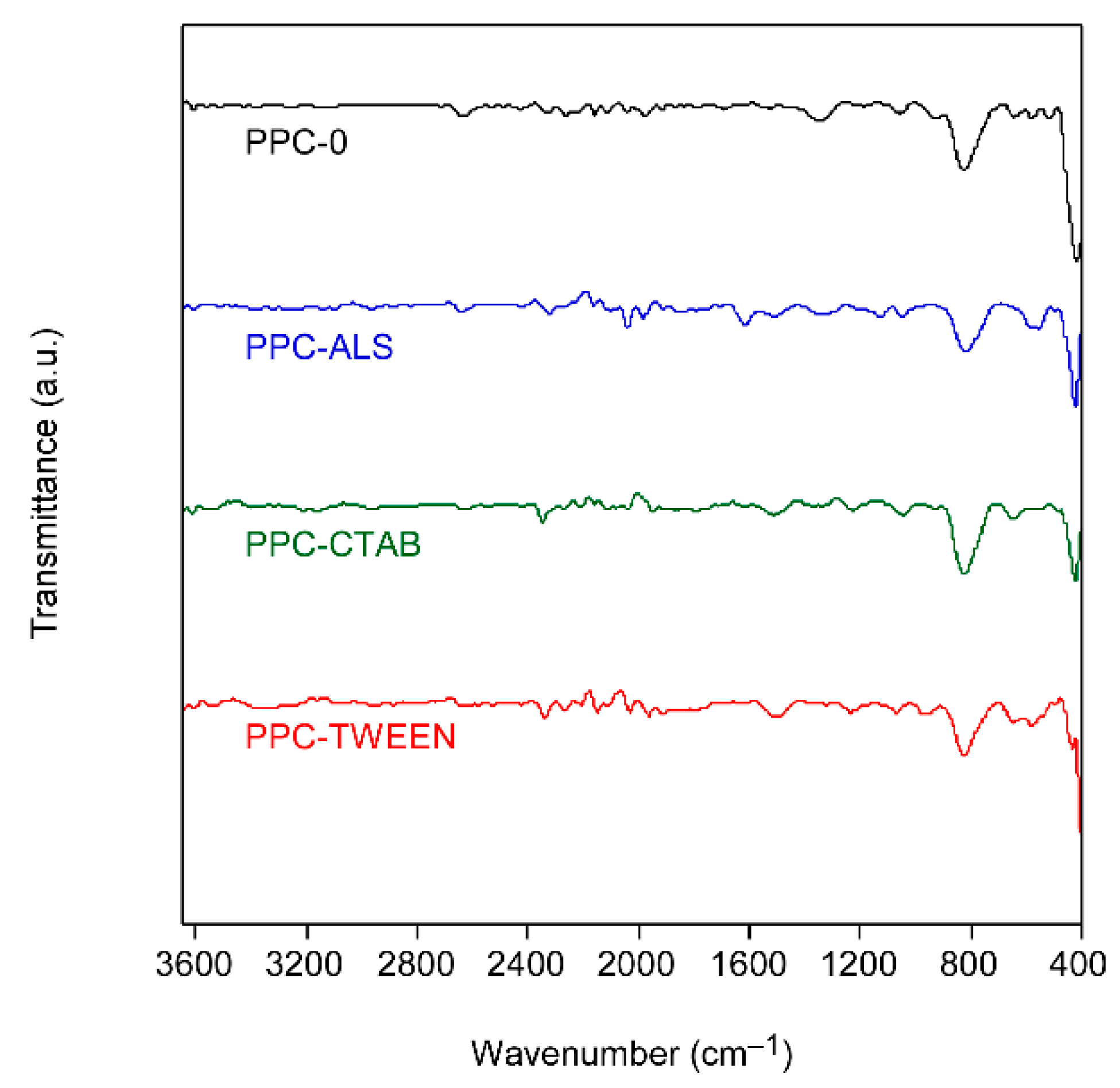
Figure 3 shows the infrared spectra of all xerogels. It can be verified that no traces of the surfactants remain in the powder after the thermal process, because the characteristic bands of these organic compounds are not detected.
The bands at 800 and 400 cm
−1 correspond to Ti-O and Ti-O-Ti bonds [
15], respectively, confirming the pure nature of TiO
2 samples.
It is well known that the presence of surfactants in liquids decreases their viscosity, an obvious effect on the thickness of these coatings. As can be seen in
Table 2, the use of different surfactants provides films thinner than PPC-0. In addition, compared with PPC-0, the presence of the template creates more irregular surfaces, with increasing roughness, due to the emergence of a porous surface. In any case, all rugosity values are relatively low (i.e., nanometric scale), which is desirable to minimize the scattering and to retain a high light transmittance.
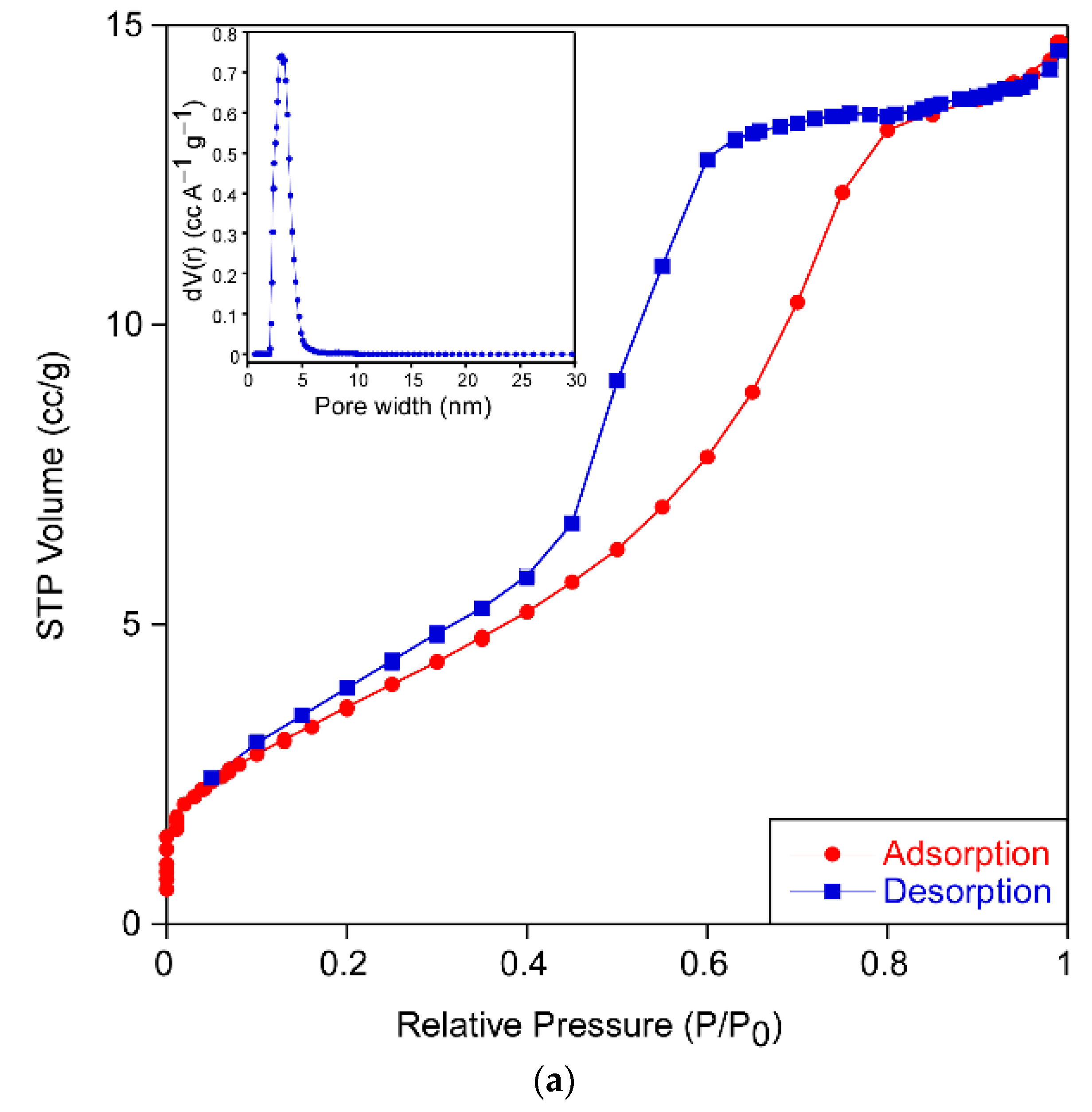
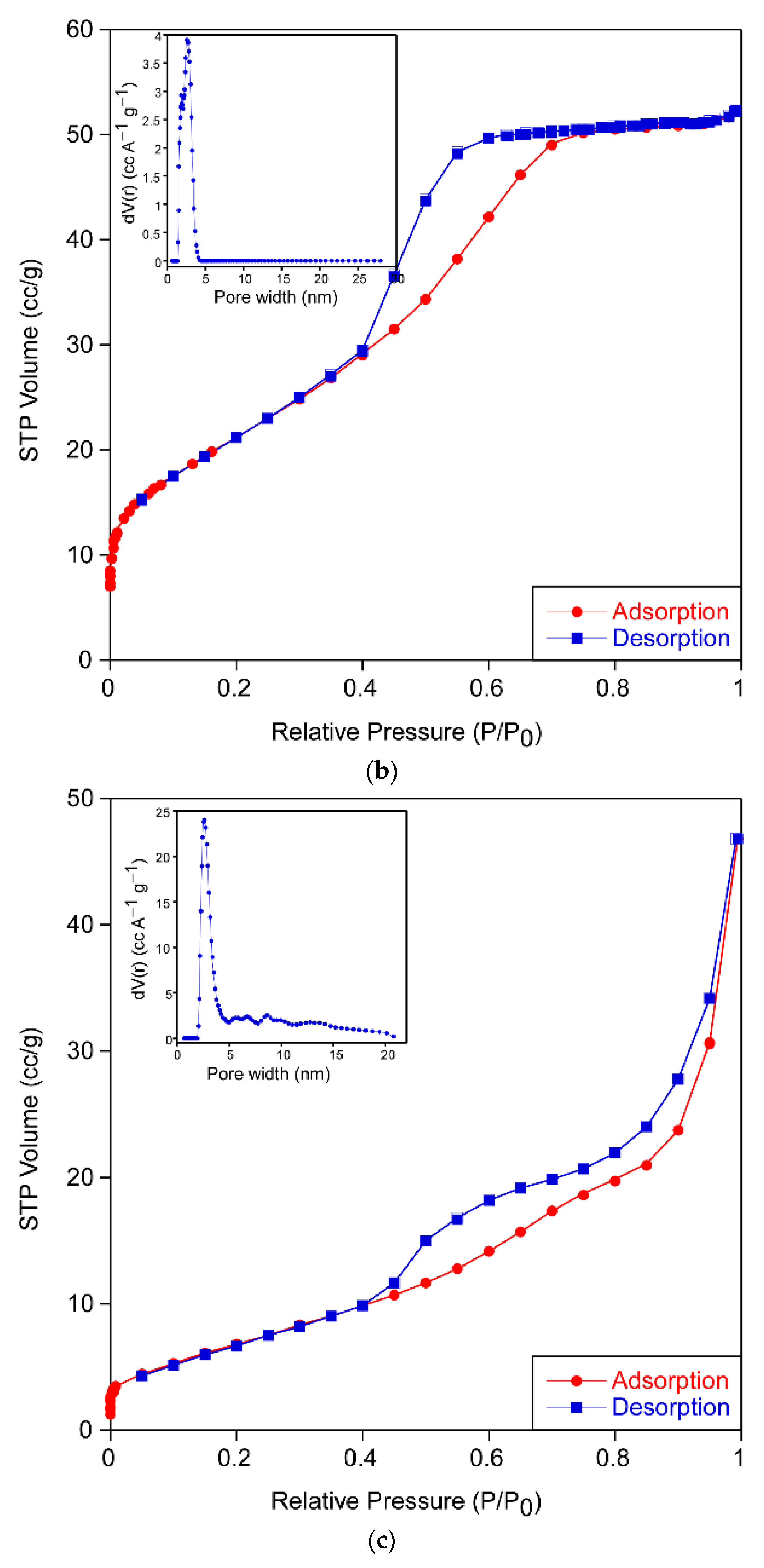
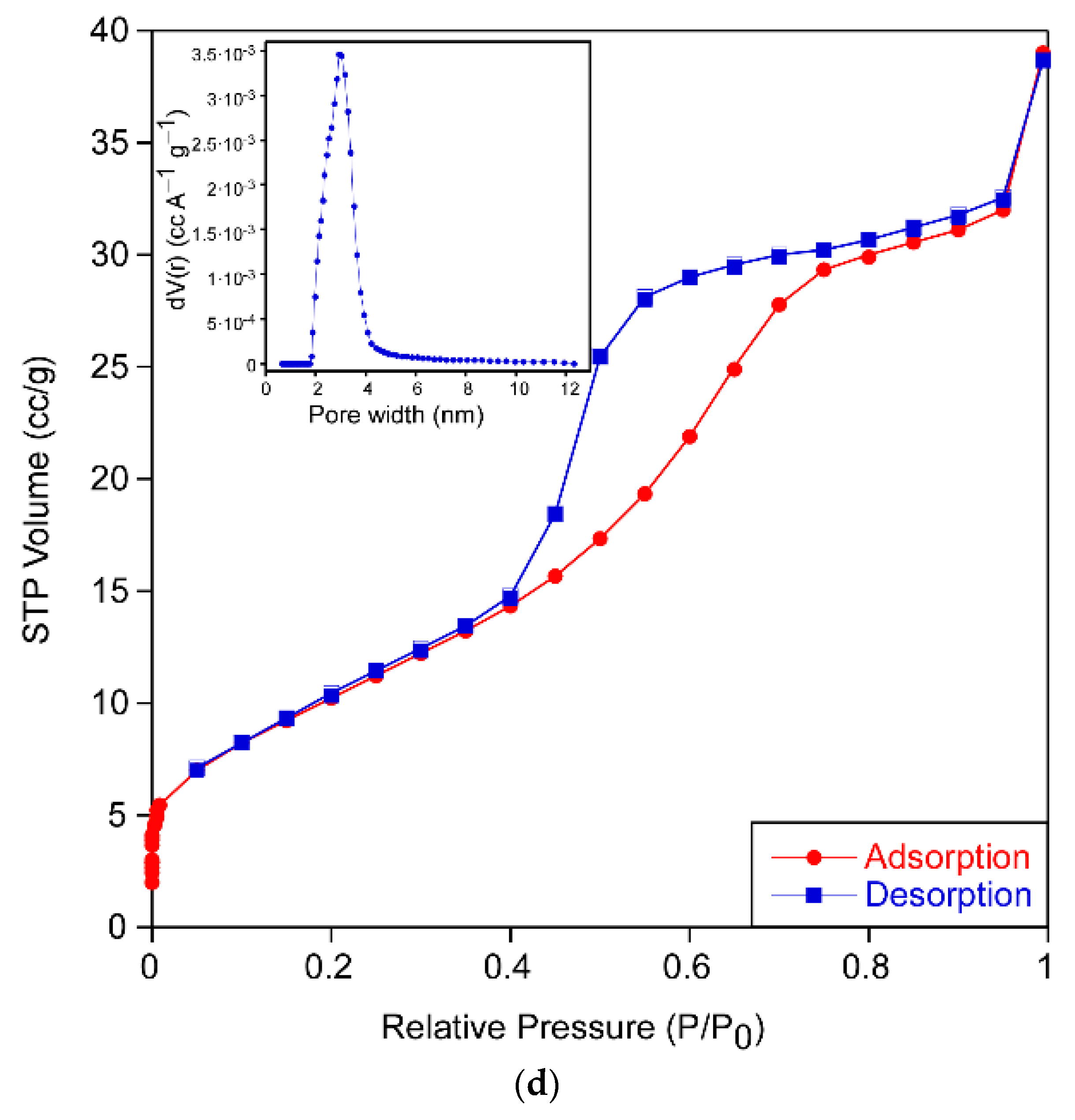
The results of the analysis from N
2 adsorption isotherms (
Figure 4) for each sample are gathered in
Table 3. All of them exhibited type IV adsorption–desorption isotherms with an elongated S-type hysteresis loop that indicates the mesoporous nature of the xerogels. The form of the hysteresis cycle allows one to identify the shape of the pores of the sample according to the IUPAC classification [
16]. The samples PPC-0, PPC-ALS and PPC-TWEEN show a type H2 hysteresis cycle, which describes materials that are often disordered where the distribution of pore size and shape is not well defined and indicative of a narrow distribution of pore bodies with a wide neck size distribution. However, the sample PPC-CTAB shows type H3 hysteresis often associated with slit-shaped pores. Samples exhibit pore sizes between 3 and 12 nm except for the PPC-CTAB sample. This pore size should ensure easy diffusion of the compounds through the porous network during the photocatalytic reaction. No meaningful differences were appreciated in the pore size distribution of the sample PPC-CTAB, because most of its pores were also in the 3–12 nm range, despite the fact that a minor contribution of pores up to 41 nm can be found.
The obtained surface area and porosity was highly influenced by the kind of surfactant used in the sol formulation, increasing in all cases in which surfactant was added. Certainly, the increase in specific surface area is related to the crystalline size (
Table 1). Based on these data, the PPC-ALS formulation appears to have the most promising properties for photocatalysis.
Table 4 compares the transmittance and band gap of the photocatalytic films. The substrates have a SiO
2 precoating to protect the titania form sodium migration, but it also increases the overall transmittance, allowing more photons to reach the TiO
2 layer. In fact, without this SiO
2 precoating, the transmittance decreases by 2–3%. Due to the refractive index of titania, the reflectance in the photocatalytic layers increases, causing the transmittance of these layers to be lower in the visible region.
Tauc’s formula was used to estimate the band gap of the photocatalytic layers [
17,
18]:
where
α is the absorption coefficient,
hν is photon energy and
Eg is the band gap. Depending on whether the band gap electron transition is direct or indirect, the factor
n has a value of 0.5 or 2. In this case, the value of
n was set to 2 since anatase exhibits an indirect band gap [
19]. The factor
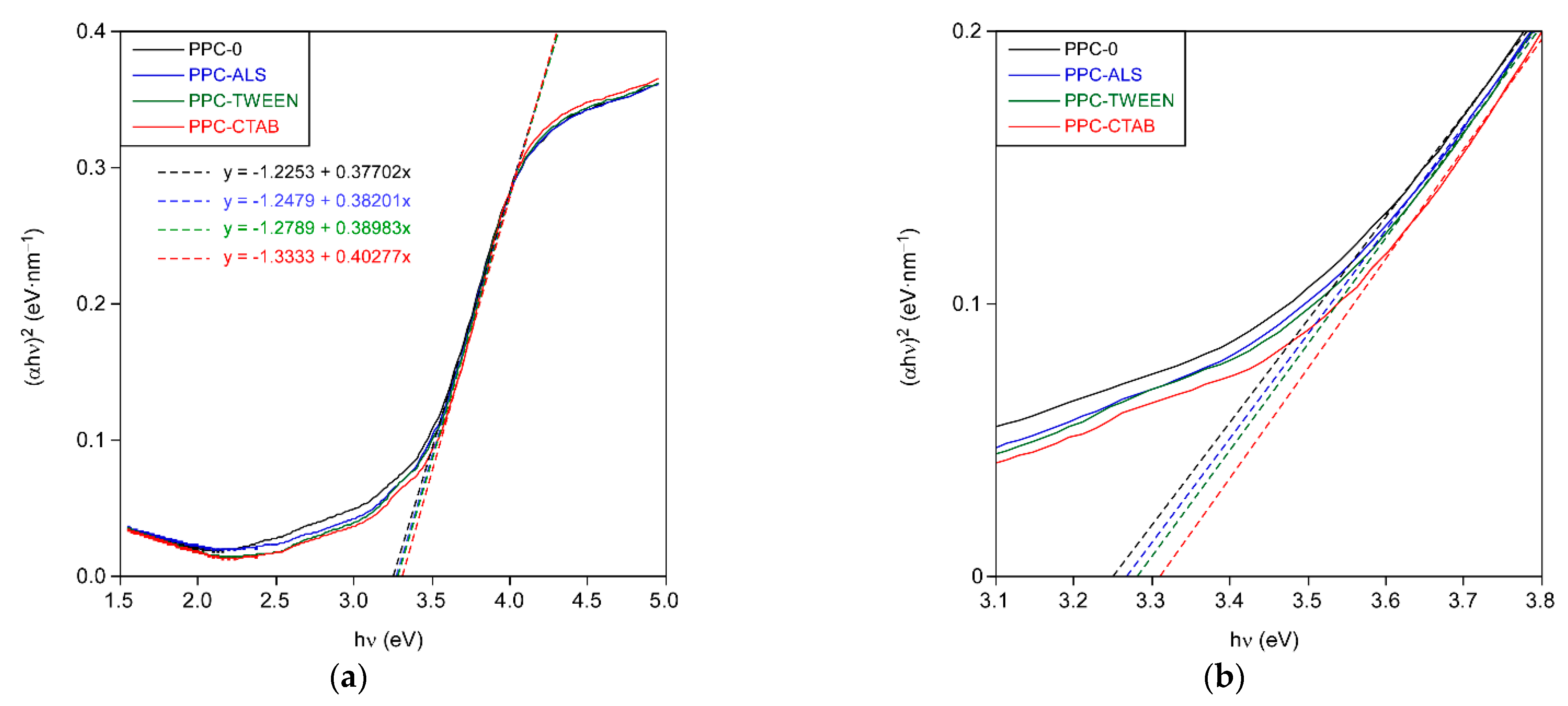
A is a constant. The absorbance spectrum of the TiO
2 layers is plotted as a function of photon energy in
Figure 5 according to Equation (2). The intersection of the linear fit with the abscissa axis gives an estimate of the bandgap energy.
These values are comparable to that expected for anatase bulk phase (3.2 eV) [
20,
21]. The differences in the band gap between the formulations are small, always increasing with the presence of surfactant. The PPC-CTAB formulation is the one whose band gap is most affected. This increase in the band gap energy is related to the decrease in layer thickness. Lower layer thicknesses are obtained with the presence of surfactant, which, as mentioned previously, is related to the decrease in crystallite size. This is due to the quantum size effects of the semiconductors, whereby the band gap energy decreases as the crystallite size increases [
22,
23,
24].
Figure 6 shows scanning electron microscopy (SEM) images for two representative photocatalytic coatings. These images reveal a porous microstructure consisting of nanoscopic TiO
2 particles (ca. 5–10 nm) that enclose pores with sizes below ca. 20 nm (
Figure 6). The estimated particle size exceeds crystallite size, which indicates that each particle is formed by the sinterization of several crystallites. It is worth mentioning that the resolution limitation of the SEM images did not allow one to perceive any clear differences among the different types of coatings.
The adhesion of the coatings was assessed by a cross-cut test. As can be seen in the images (
Figure 7), all samples exhibited good adhesion to the substrate, as none of them showed any delamination.
2.2. Study of Photocatalytic Activity
Different dyes were used to evaluate the photocatalytic activity of the coatings.
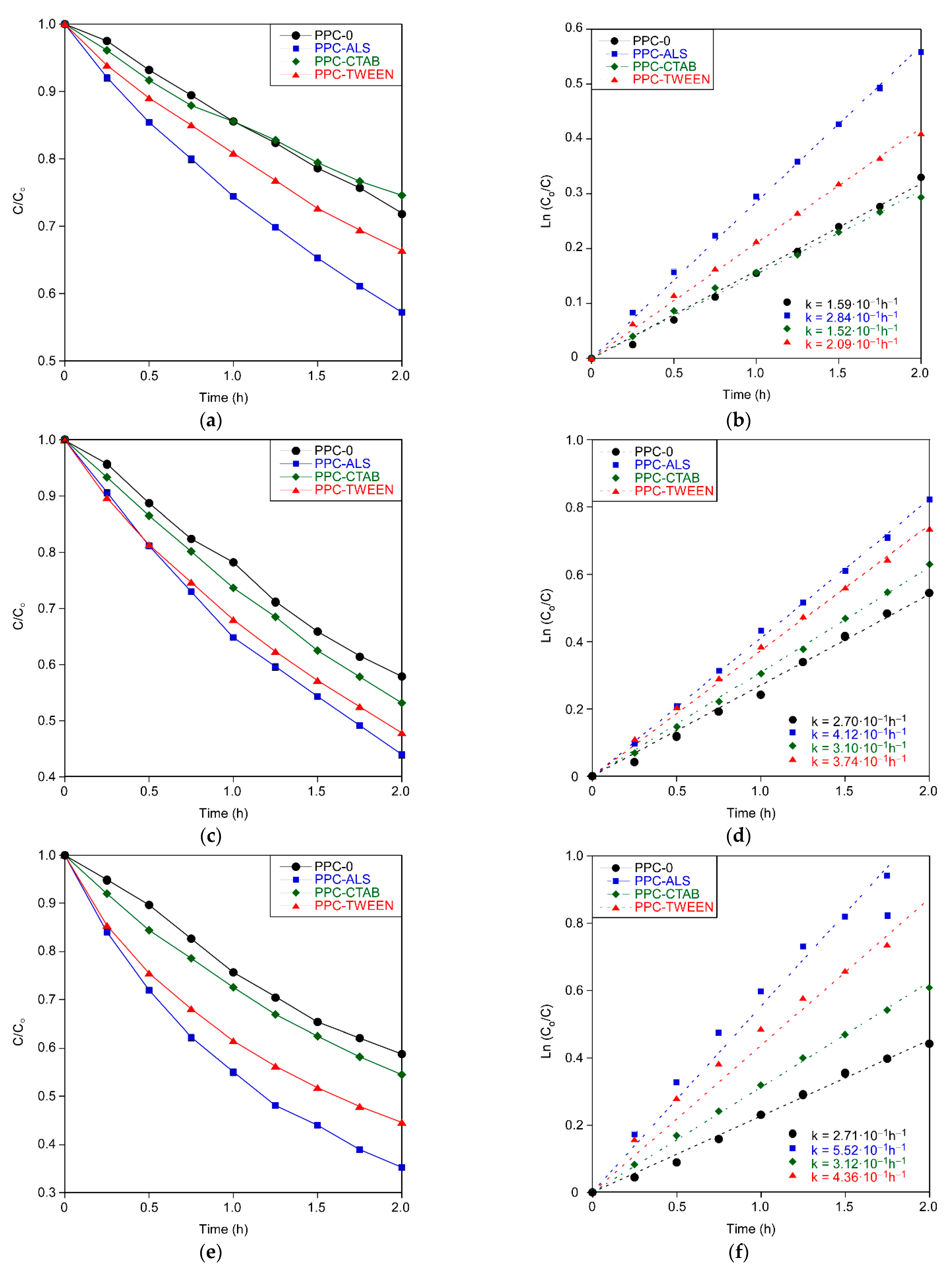
Figure 8a,c,e show the relative concentration of each dye (C/C
o, where C and C
o represent the dye concentration at a given time and the initial concentration, respectively) with respect to time. Previously, the substrates were kept immersed in the problem solution without applying UV radiation for 2 h. It was observed that, in all the photocatalytic layers, the removal of the dye was less than 1% after 2 h, so it was concluded that, regardless of the formulation studied, the adsorption phenomenon on the material hardly occurs.
The photocatalytic degradation kinetic of all dyes fits the first-order reaction [
25]. This fit is shown in
Figure 8b,d,f, representing the natural logarithm of the relative concentration (Ln(C
o/C) with respect to time.
Table 5 shows the percentages of dye removed and the reaction constants calculated through the fit for each coating and dye.
Considering the microstructural characteristics of the coatings (
Table 1) in the photoactivity, PPC-CTAB shows a performance comparable to PPC-0, despite the fact that PPC-CTAB has a somewhat larger surface area and porosity with respect to PPC-0.
However, those coatings exhibiting higher surface area values (PPC-TWEEN and PPC-ALS) also promote a meaningful improvement in photodegradation performance.
Regarding the type of dye, the size of the molecule to be degraded seems to be the major influencing factor on the rate at which the compound is removed, although other factors such as functional groups or steric hindrance might have a minor influence. In all cases, with different surfactants and different pore distributions the order of degradation is the same, from highest to smallest activity: PPC-ALS, PPC-TWEEN, PPC-CTAB and PPC-0. It is evident that a higher porosity is beneficial for the photocatalytic activity, but if a lower or similar band gap energy is maintained, the crystallite size is minimized.
Keeping in mind the obtained results, the formulation of ALS has been selected for its application by the slot-die method.
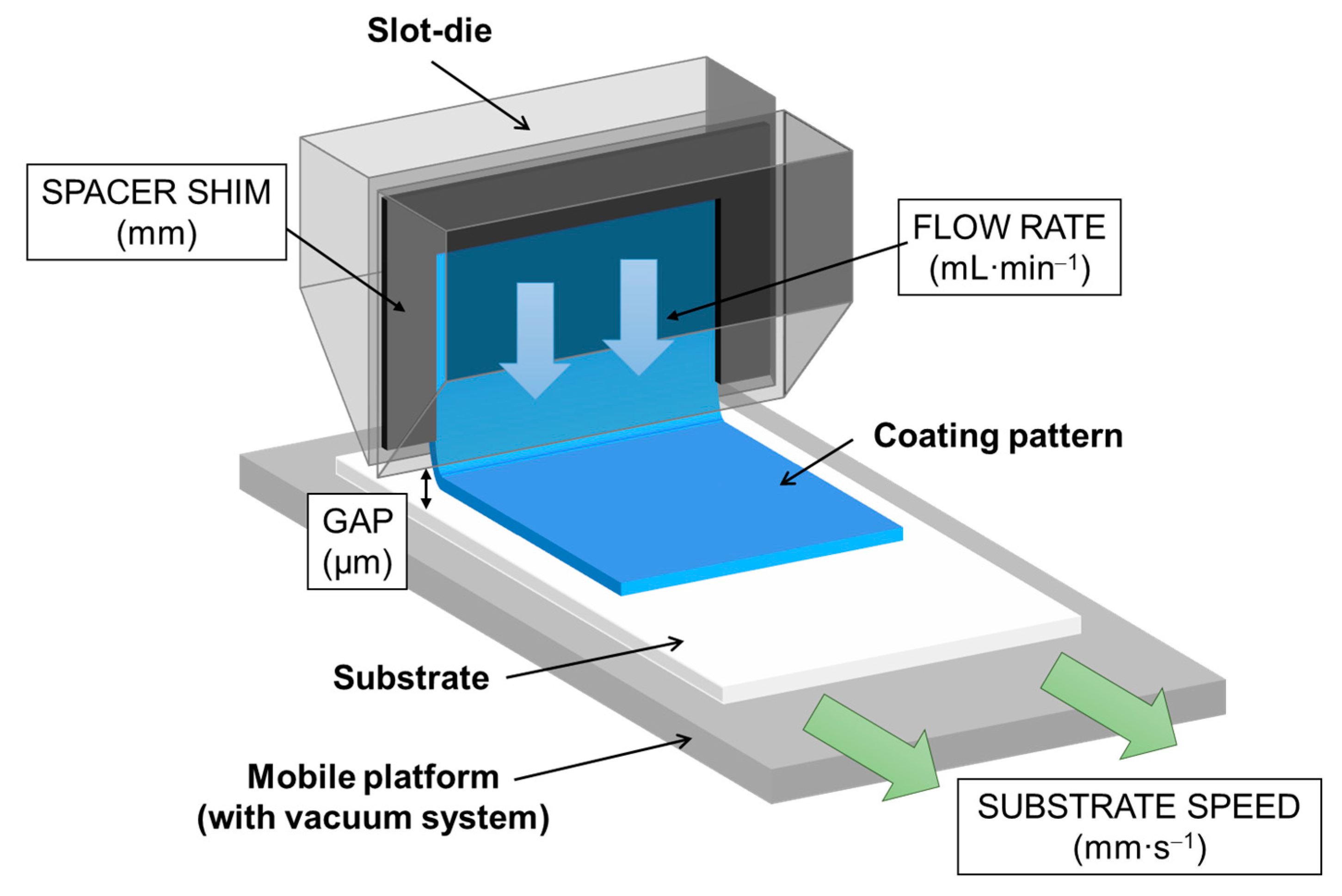
2.3. Slot-Die Analysis of Variance
The slot-die process needs a previous study due to all the variables involved. For this reason, a design of experiments (DoE) was proposed.
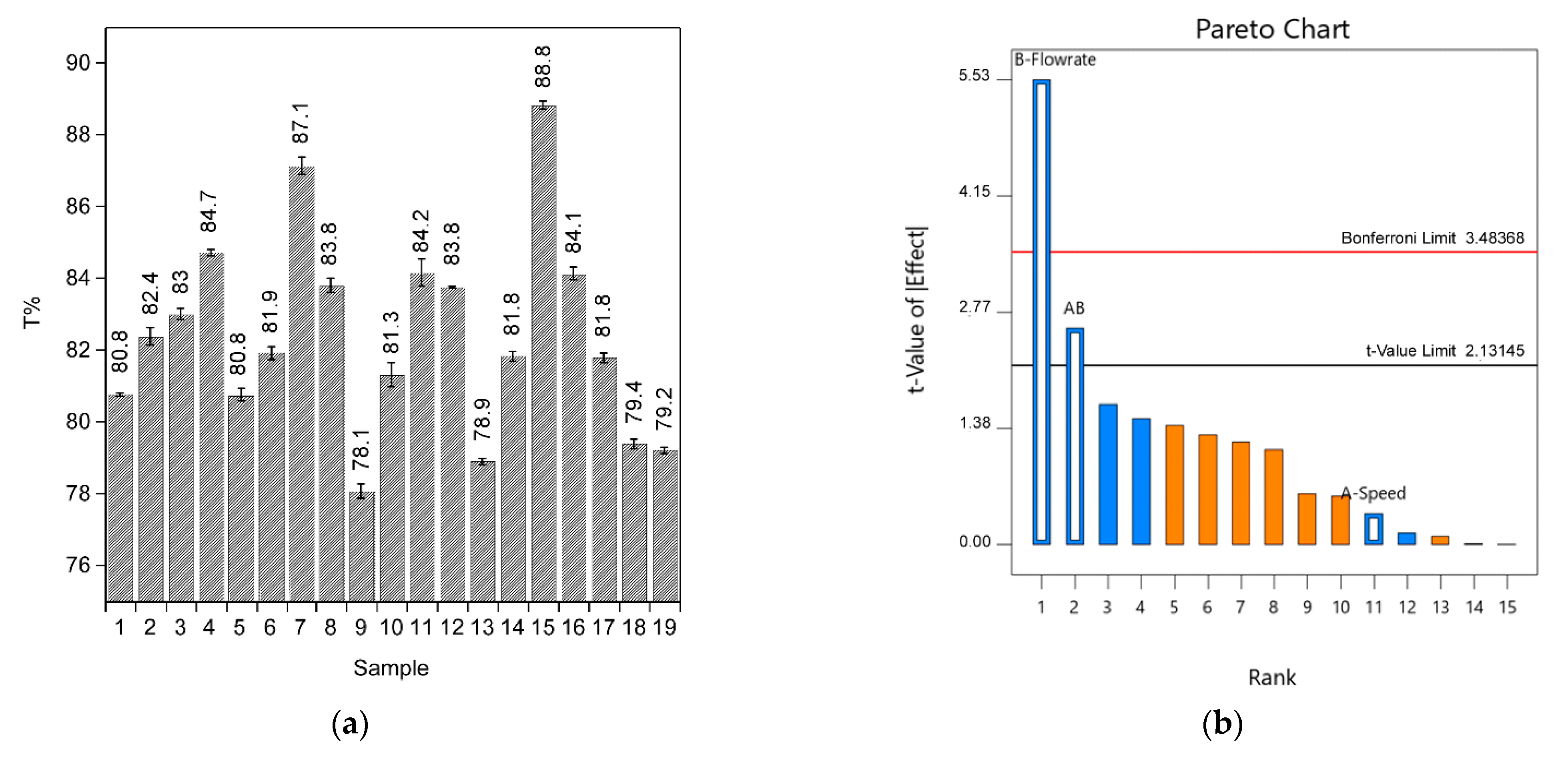
Figure 9a shows the results obtained for the DoE. TiO
2 coating was measured by three points per sample. These responses were analyzed by the Unscrambler X software. The pareto chart (
Figure 9b) was used to evaluate which variables and effects are the most important. Factor B (Flow rate) and interaction AB (Velocity-Flow rate) were found to be the most significant factors. The analysis model forces variable A (speed) to be considered as a main factor, but the pareto chart and ANOVA resolve it as a non-significant factor.
Table 6 shows the ANOVA results. The Model F-value of 12.46 implies the model is significant.
p-values less than 0.0500 indicate that the model terms are significant. In this case, B and AB are significant model terms.
In view of these results, it was considered that an optimization of the parameters was not necessary. The DoE provided parameters to obtain coatings by the slot-die process with similar characteristics to those obtained by immersion. Thus, coatings obtained by both deposition techniques were compared.
2.4. Comparison between Dip Coatings and Slot-Die Coatings
One of the objectives of this work was to assess if coatings of similar characteristics can be obtained by two different application methods (dip coating and slot-die) while retaining the same sol formulation. Therefore, with the help of the DoE, the parameters were established to obtain a coating by the slot-die process with the same optical characteristics to the one obtained by dip coating. These parameters were 50 mm·s−1 speed, 1 mL·min−1 flow rate, 200 μm gap and 0.127 mm shim. To evaluate the slot-die results, substrates were coated by immersion on one side (shielding the other side). Photocatalytic performance was tested by immersing the coated substrates in a borosilicate glass reactor containing 45 mL of an aqueous solution (0.11 mmol dye per m2 coating).
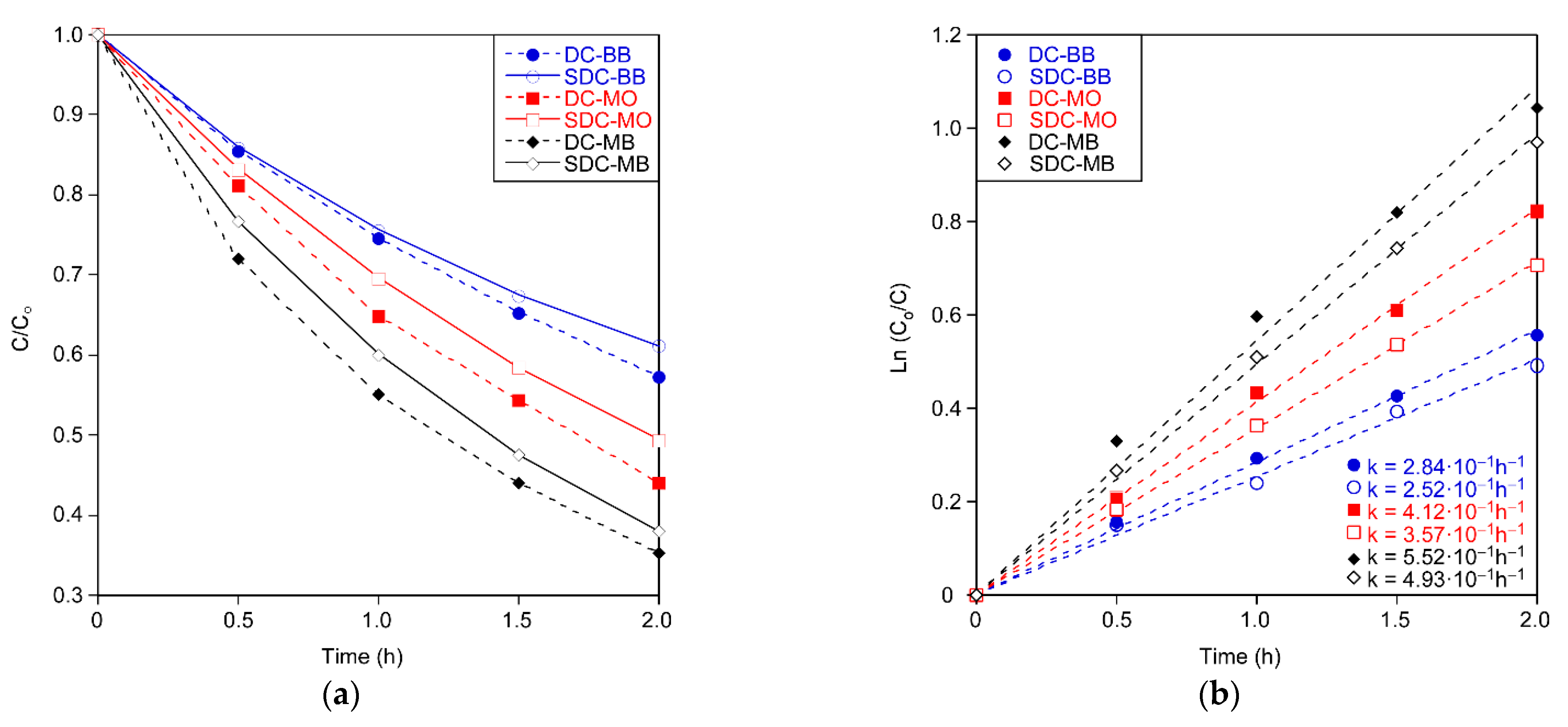
Both types of coatings showed similar photocatalytic activity, but in all cases it is slightly higher in the dip coated substrate (
Figure 10), as the degradation constants are higher for the dip coating technology (
Table 7). One of the hypotheses of this behavior is associated to the edge effect generated by immersion (area with a thicker coating). This effect is due to the dip coating method. During extraction from the substrate, the liquid slowly evaporates while being subjected to drag forces, including gravity, and more liquid accumulates at the edge of the substrate. Another common defect of this technique is rippling [
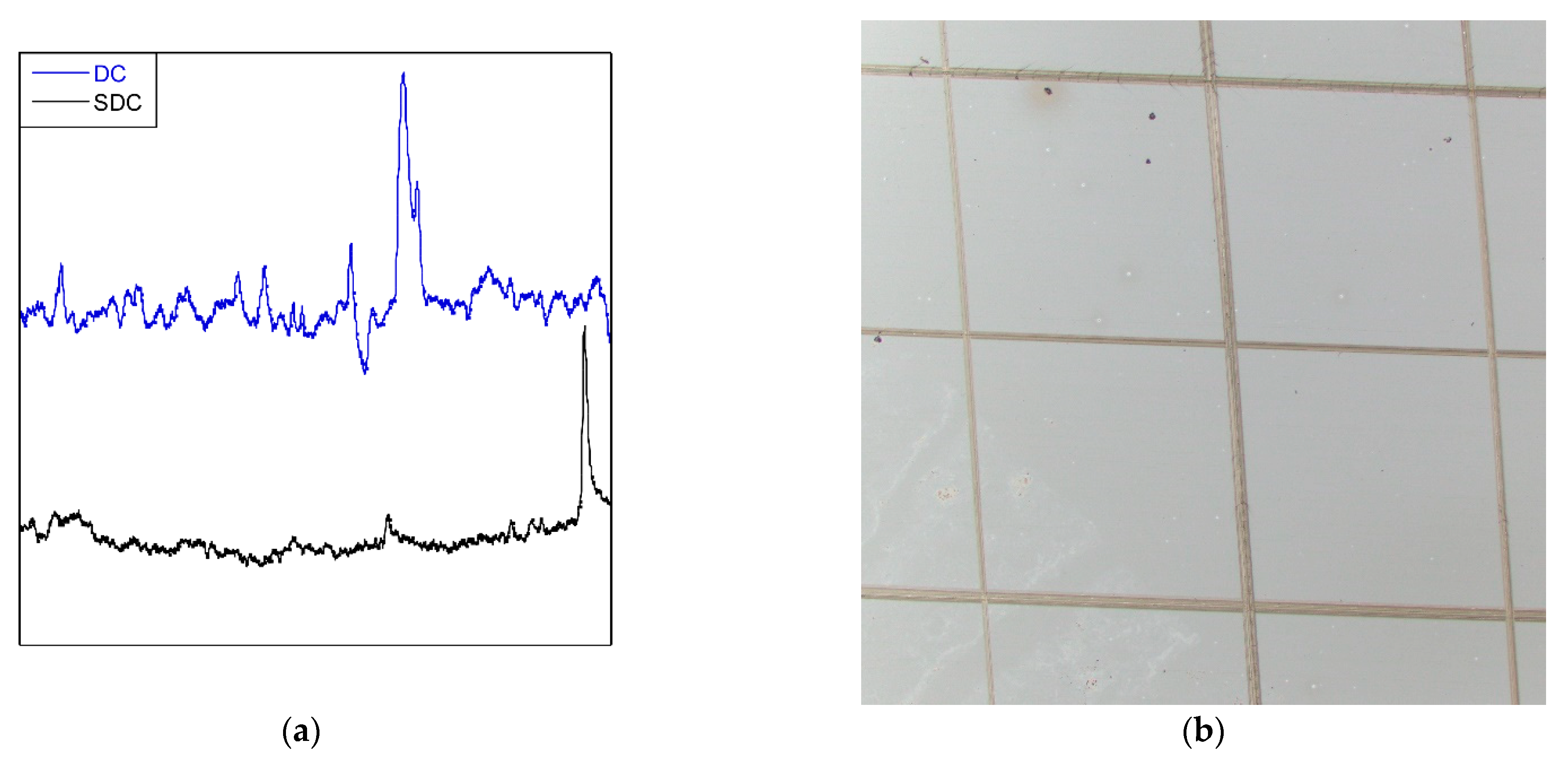
26]. Therefore, slot-die coatings have less roughness due to the process, with Ra values of 0.0074 μm for SDC versus 0.0248 μm for DC. It is an obvious difference in roughness when looking at
Figure 11a, which shows the surface profiles of coatings obtained by profilometry. Due to all the aforementioned reasons, dip coating is more active, though the difference between the two methods is small. These are promising results for large-scale sol–gel application, due to its advantages over the immersion method.
As a dip coating sample (
Figure 7), the adhesion of the slot-die coating was tested by the cross-cut test, exhibiting good adhesion to the substrate (
Figure 11b).