Influencing Multi-Walled Carbon Nanotubes for the Removal of Ismate Violet 2R Dye from Wastewater: Isotherm, Kinetics, and Thermodynamic Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Purification and Functionalization of MWCNTs
2.3. Dye Solution Preparation
2.4. Adsorption Experiments
2.5. Characterization of MWCNTs
2.6. Adsorption Isotherm Study
2.6.1. Isotherm Experiment
2.6.2. Theoretical Background of Isotherm Models
The Freundlich Model
The Langmuir Model
The Henderson and Halsey Isotherm Models
The Harkins–Jura Model
The Smith Model
The Tempkin Model
2.6.3. Error Functions Test
Average Percentage Error (ABE)
Nonlinear Chi-Square Test (χ2)
Sum of Absolute Errors (EABS)
2.7. Adsorption Kinetics
2.7.1. Pseudo-First-Order Kinetics Model
2.7.2. Pseudo-Second-Order Kinetics Model
2.7.3. The Intraparticle Diffusion Model
2.8. Adsorption Thermodynamics
2.9. Application on Real Wastewater
3. Results and Discussion
3.1. Characterization of the MWCNTs
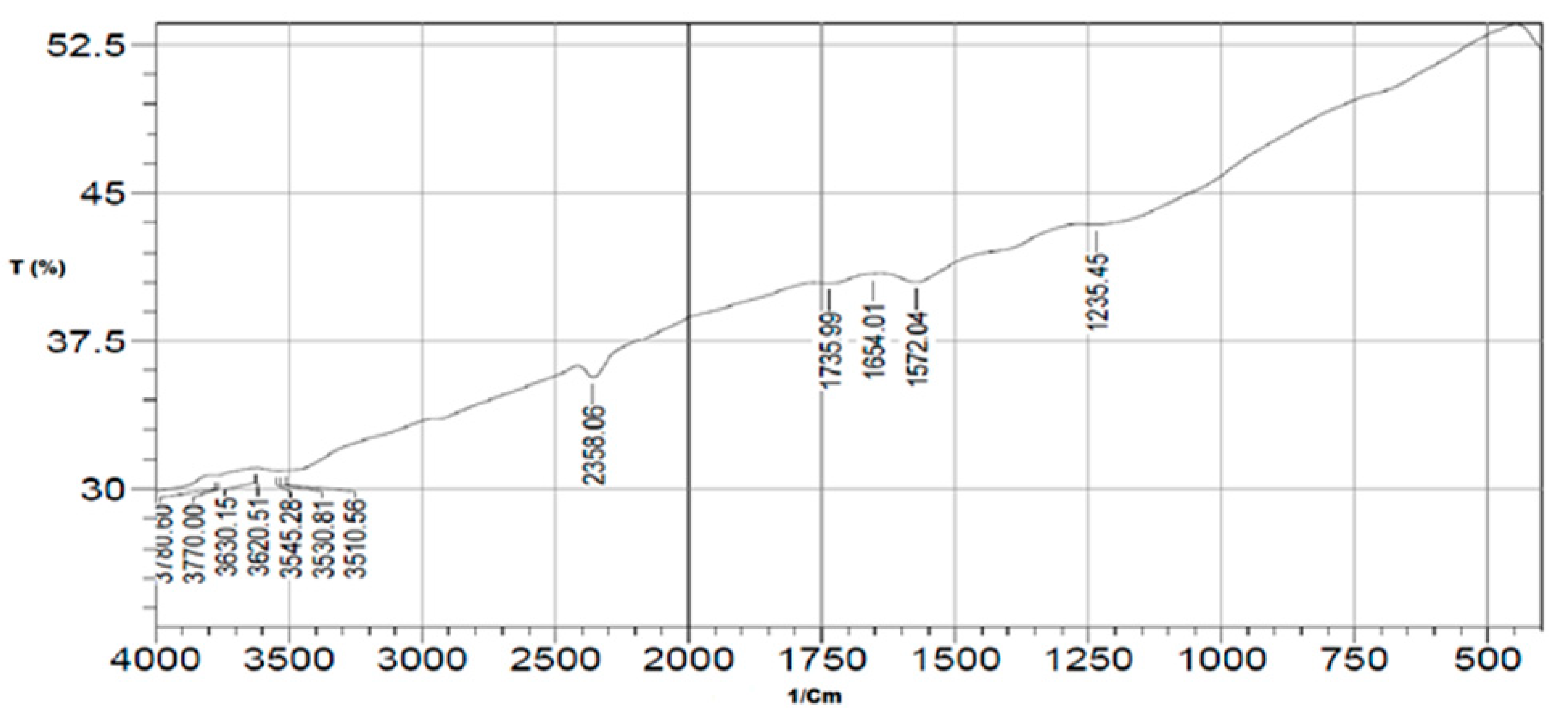
3.1.1. FT-IR Analysis
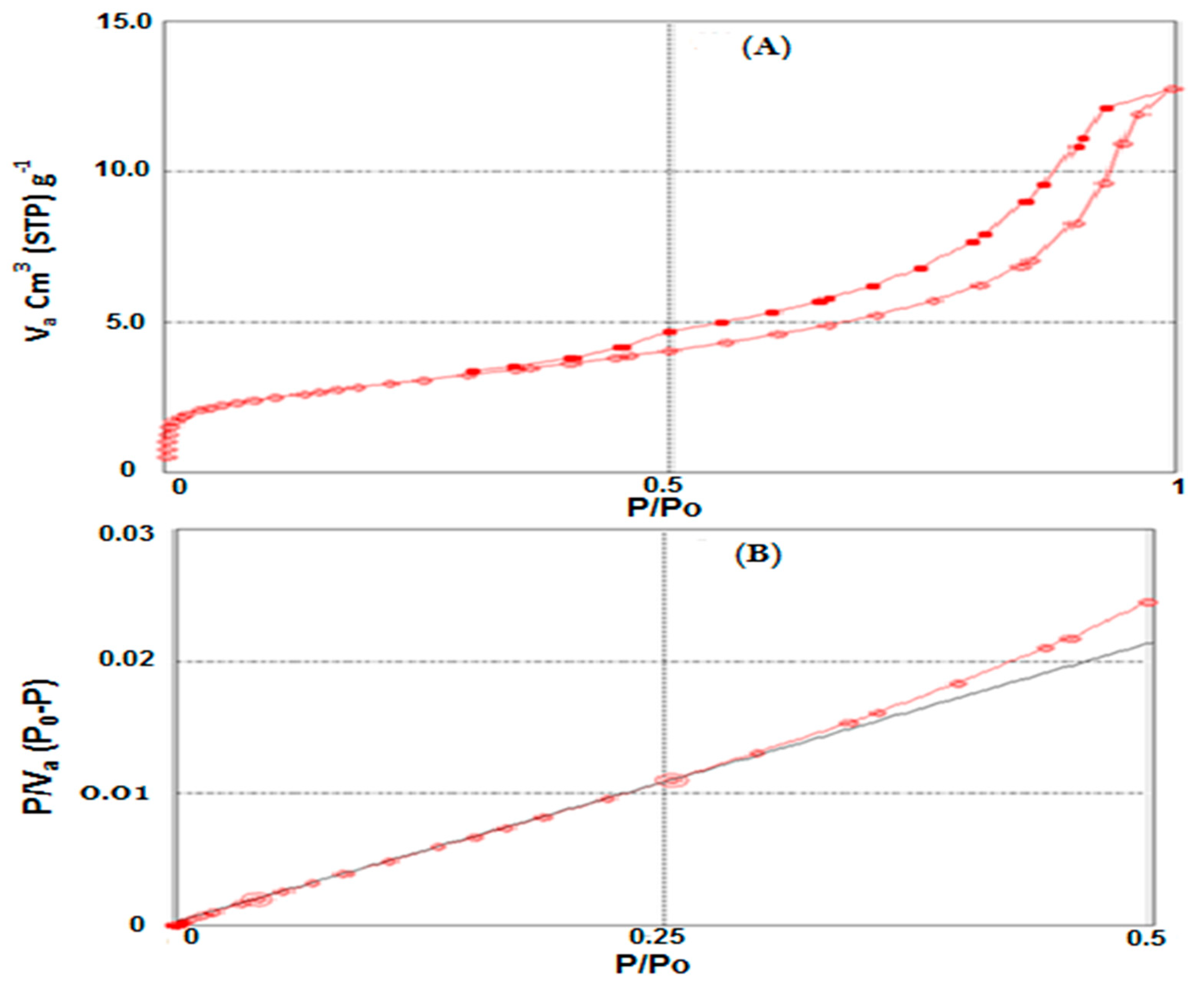
3.1.2. BET Surface Analysis
3.1.3. SEM Examination
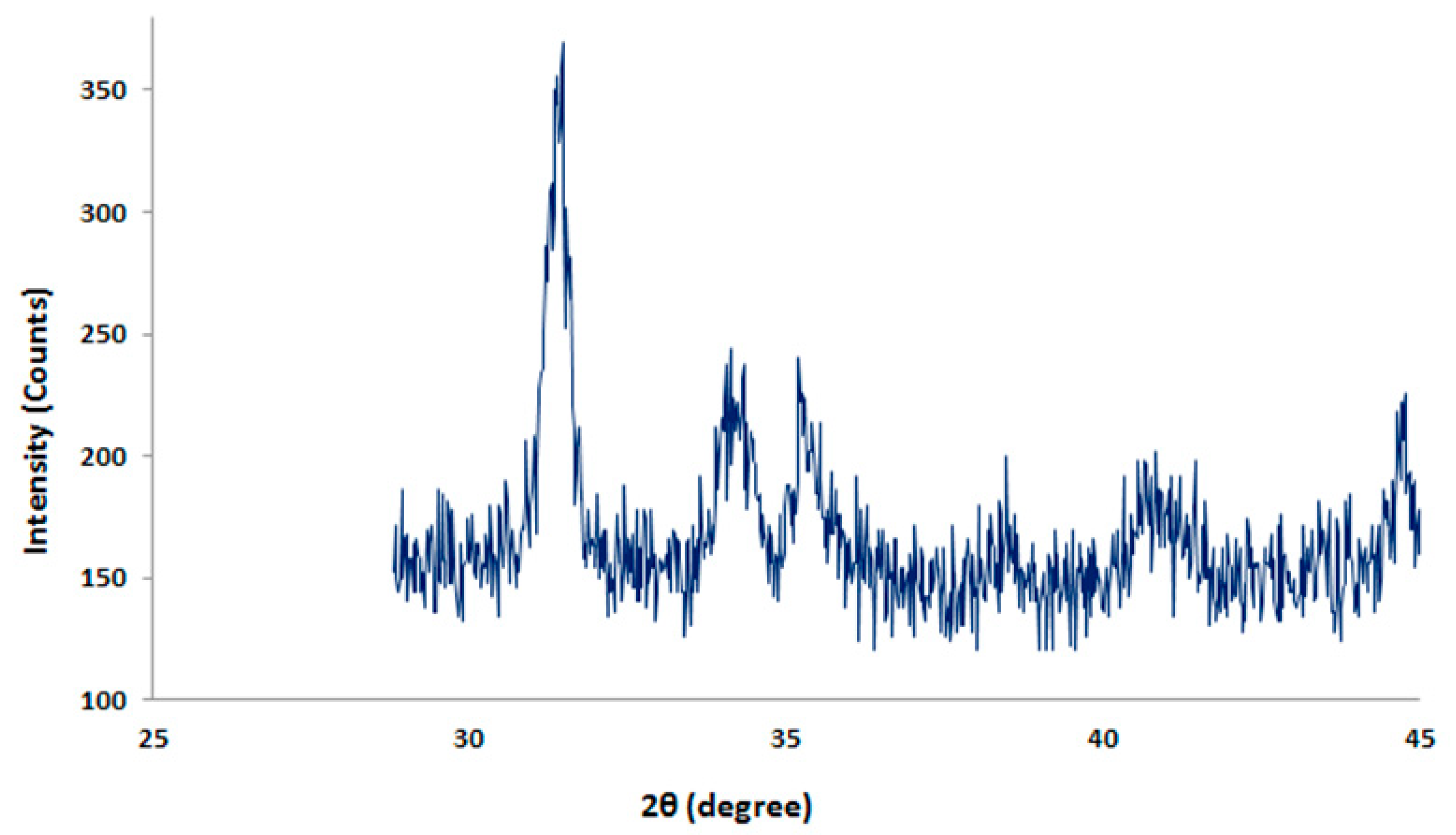
3.1.4. XRD Measurement Analysis
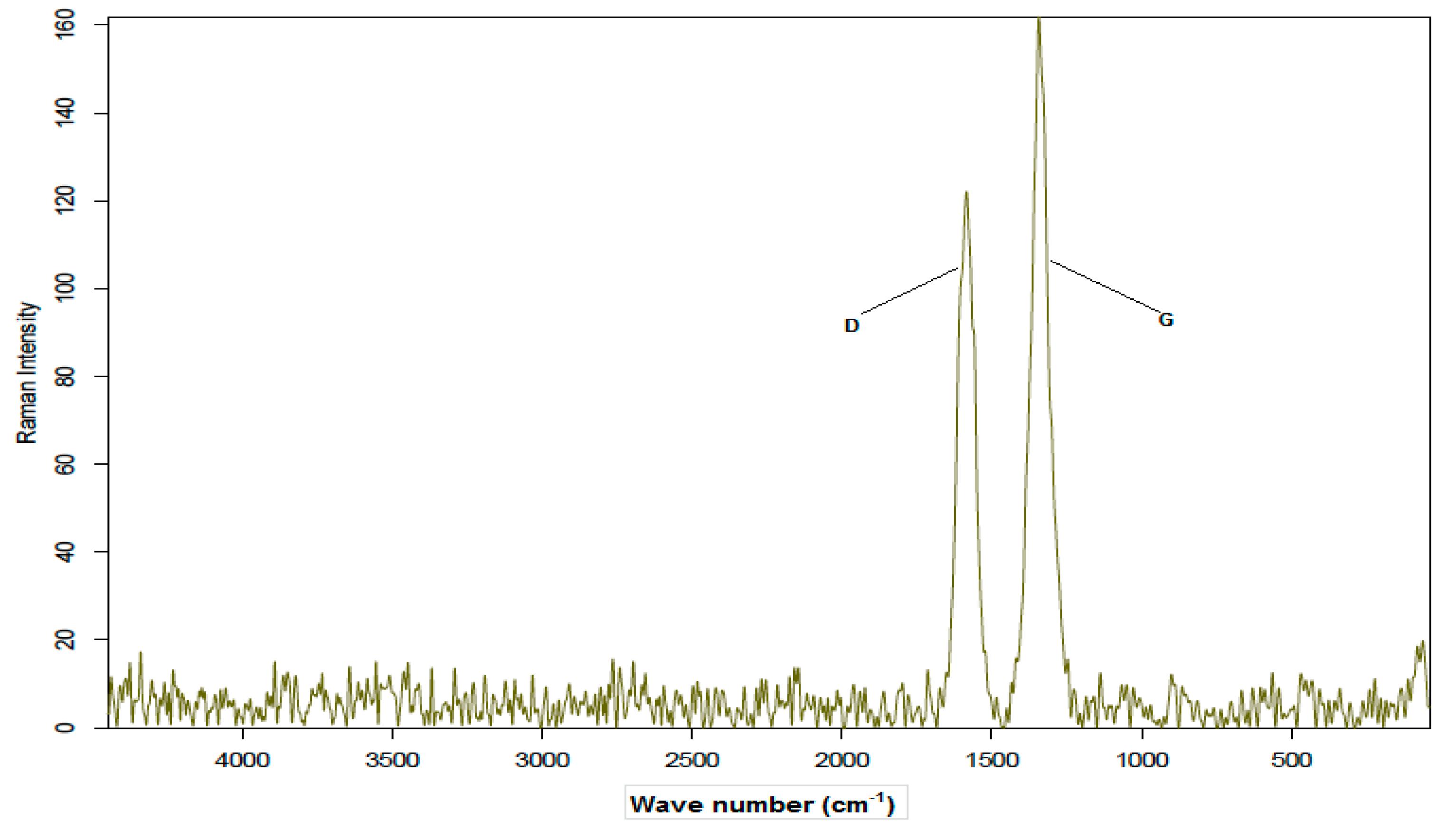
3.1.5. Raman Characterization
3.2. Adsorption Experiments
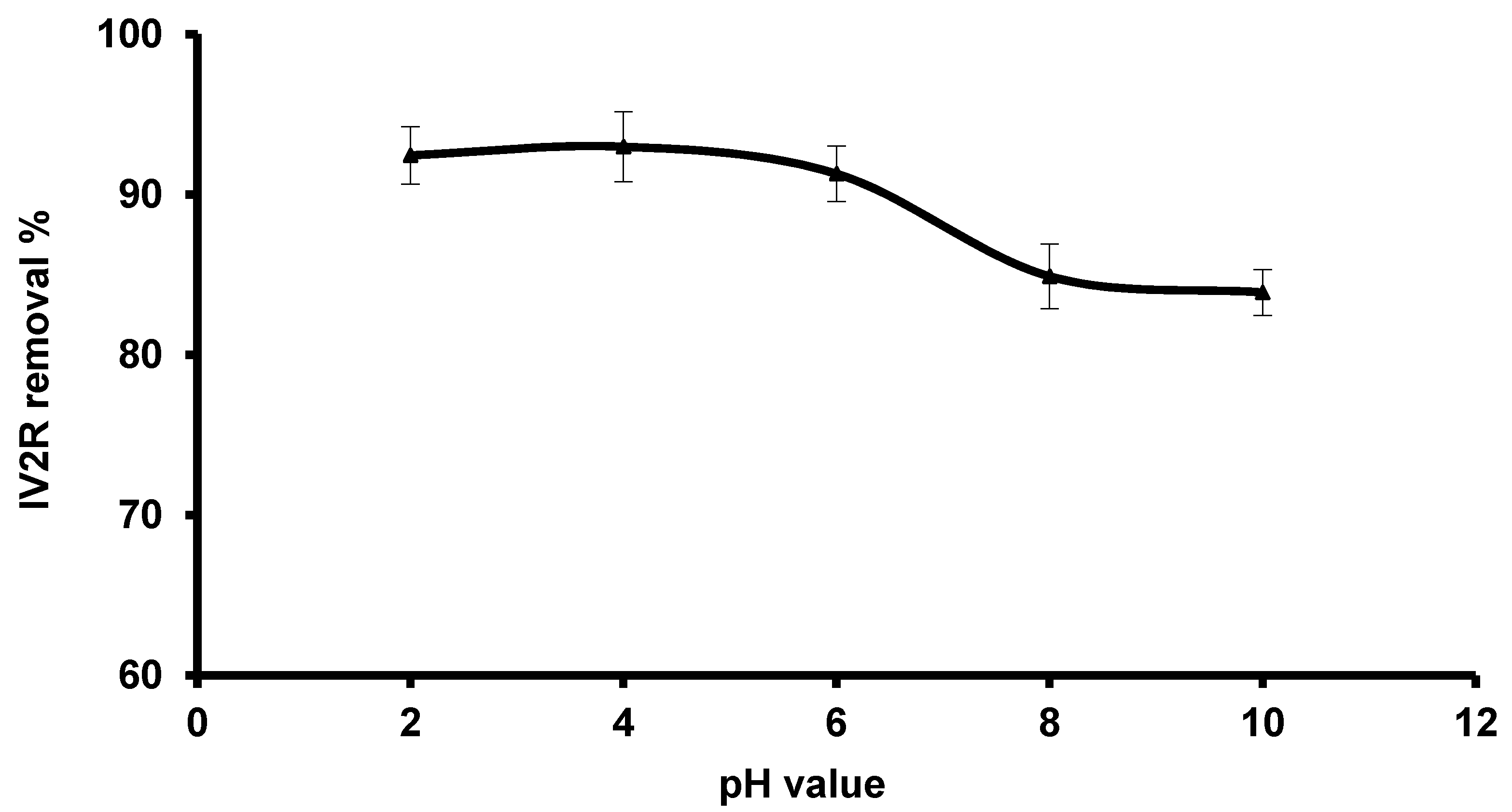
3.2.1. Influence of pH
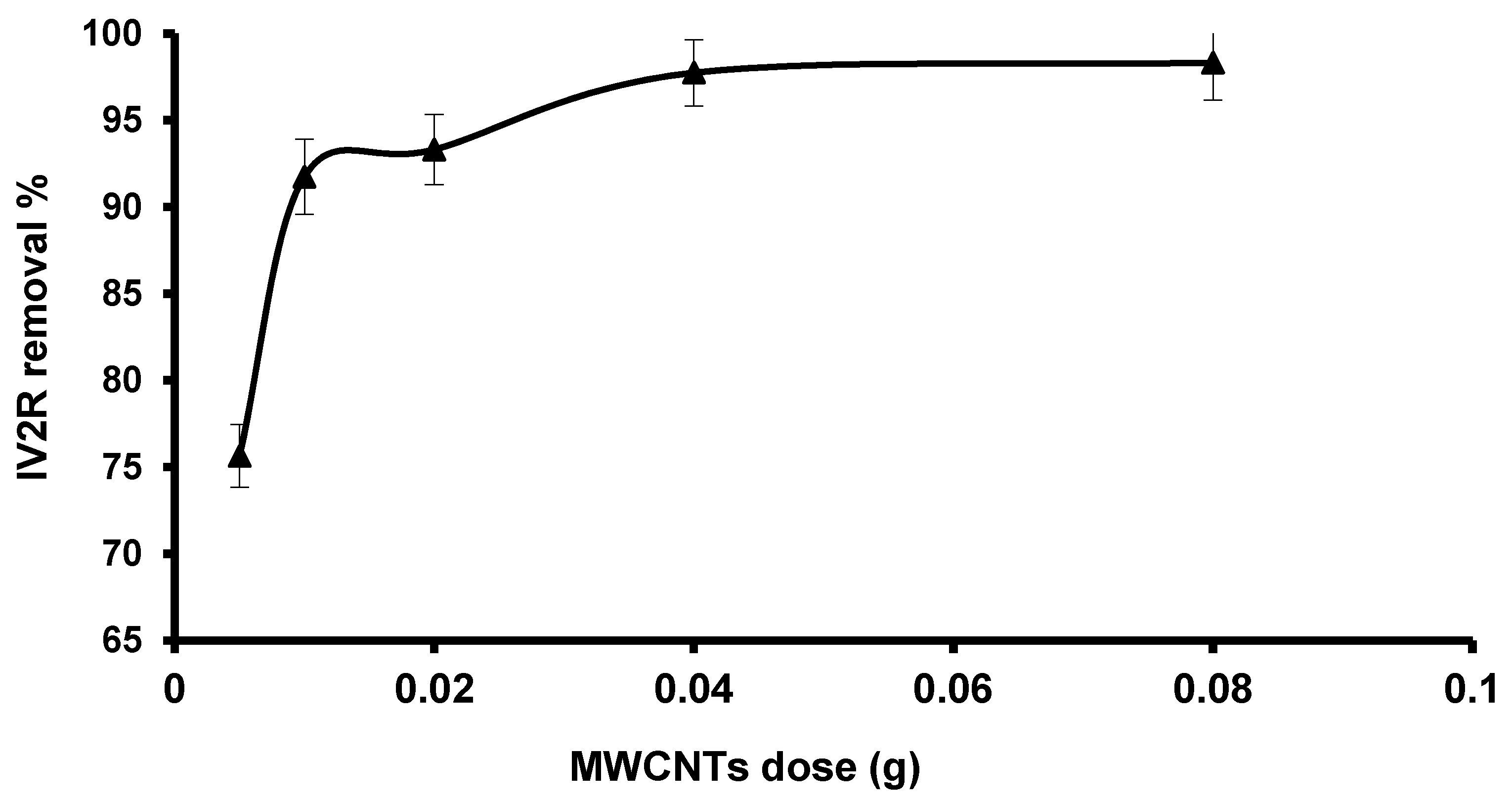
3.2.2. Influence of Adsorbent Dose
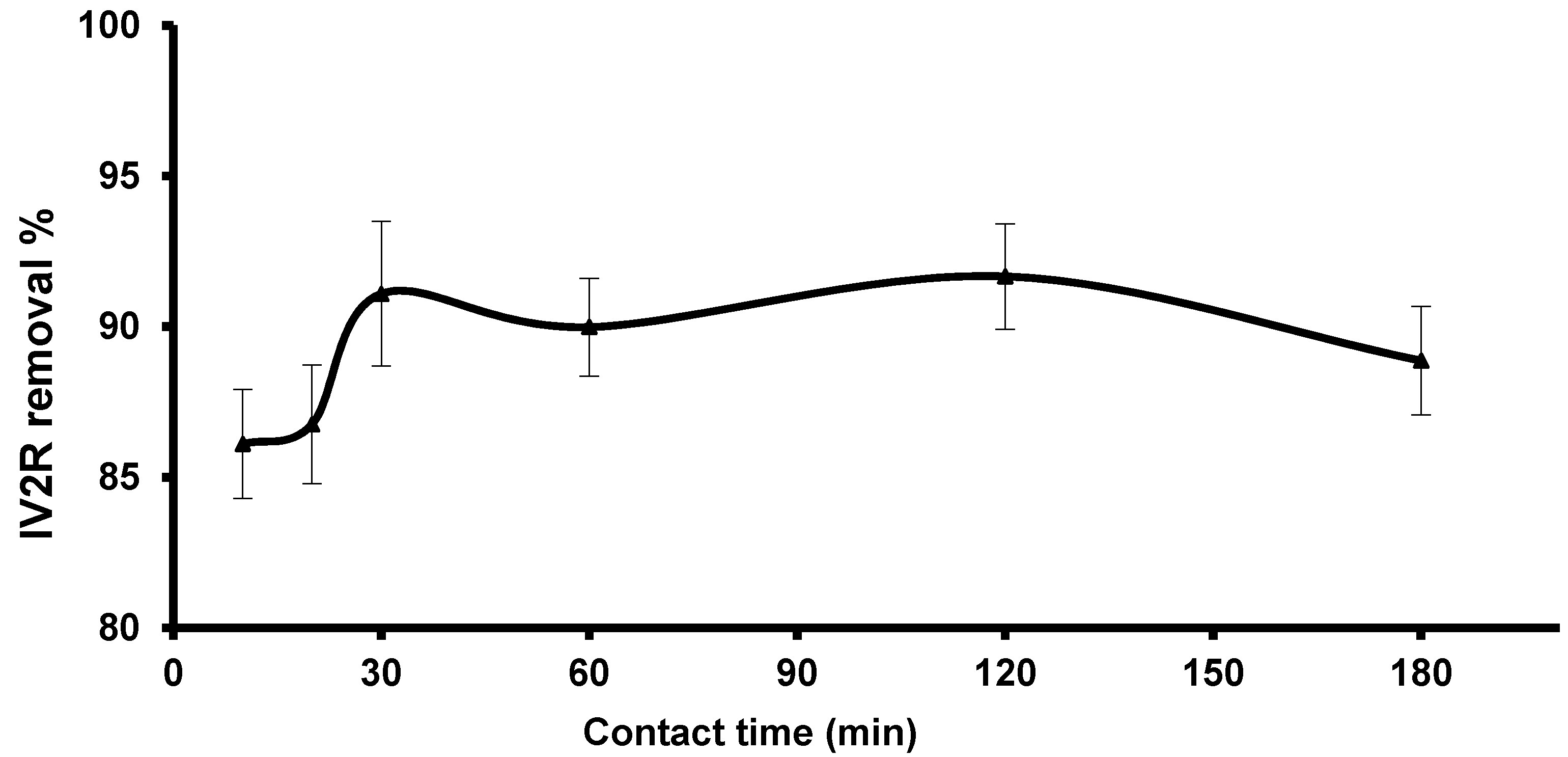
3.2.3. Influence of Contact Time
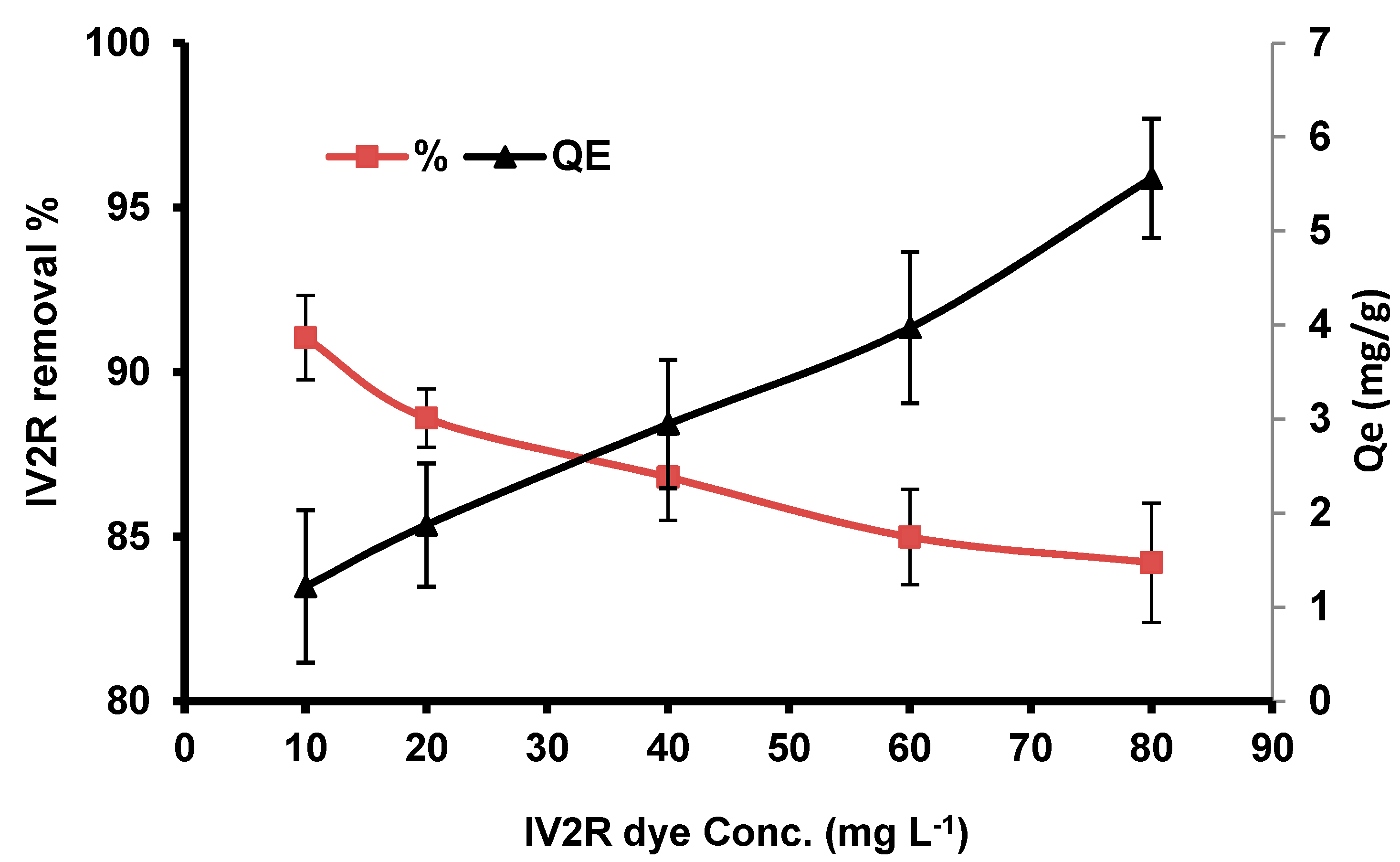
3.2.4. Influence of the Initial Dye Concentration
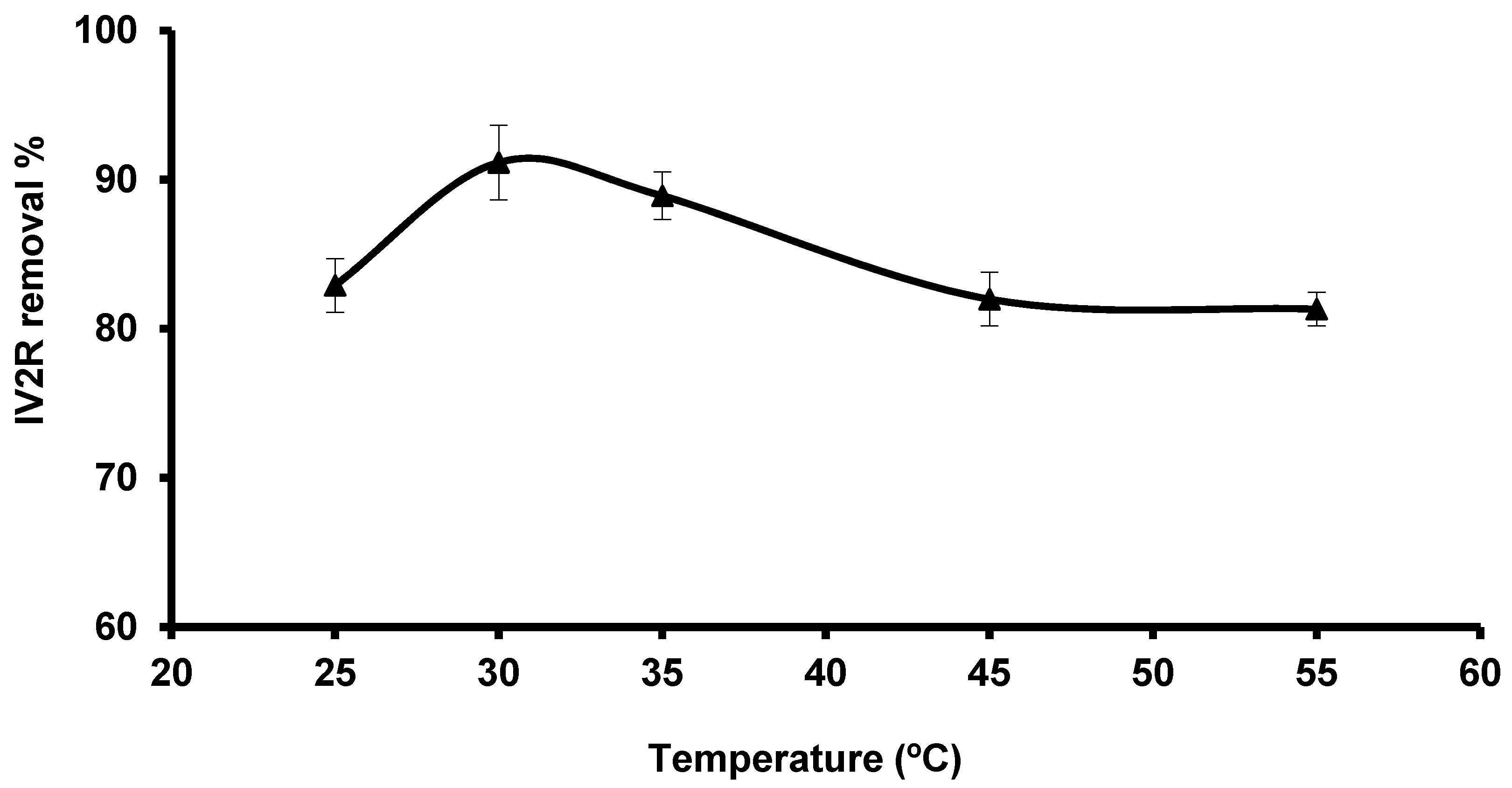
3.2.5. Influence of Temperature
3.3. Isothermal Analysis
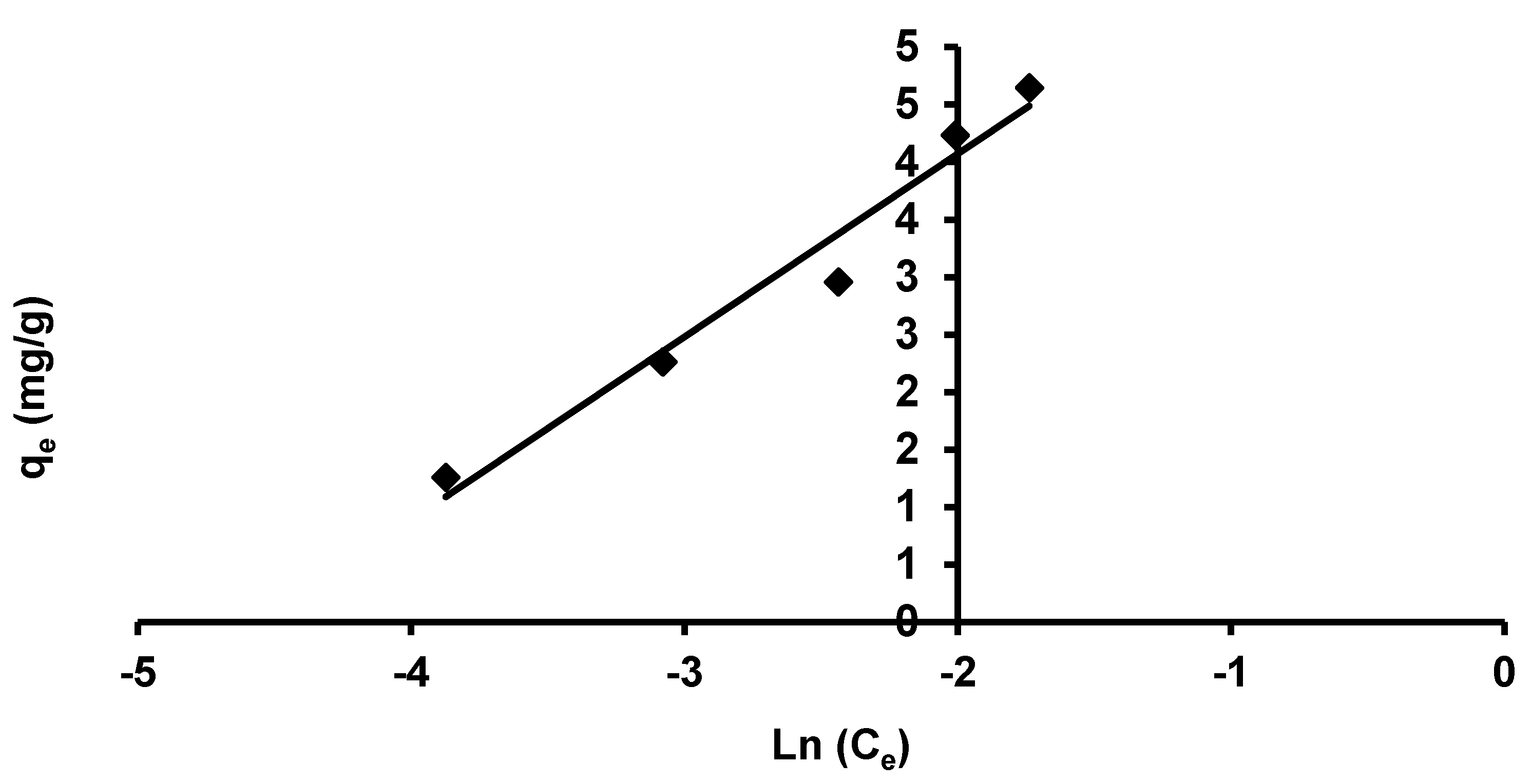
3.3.1. Freundlich Isotherm
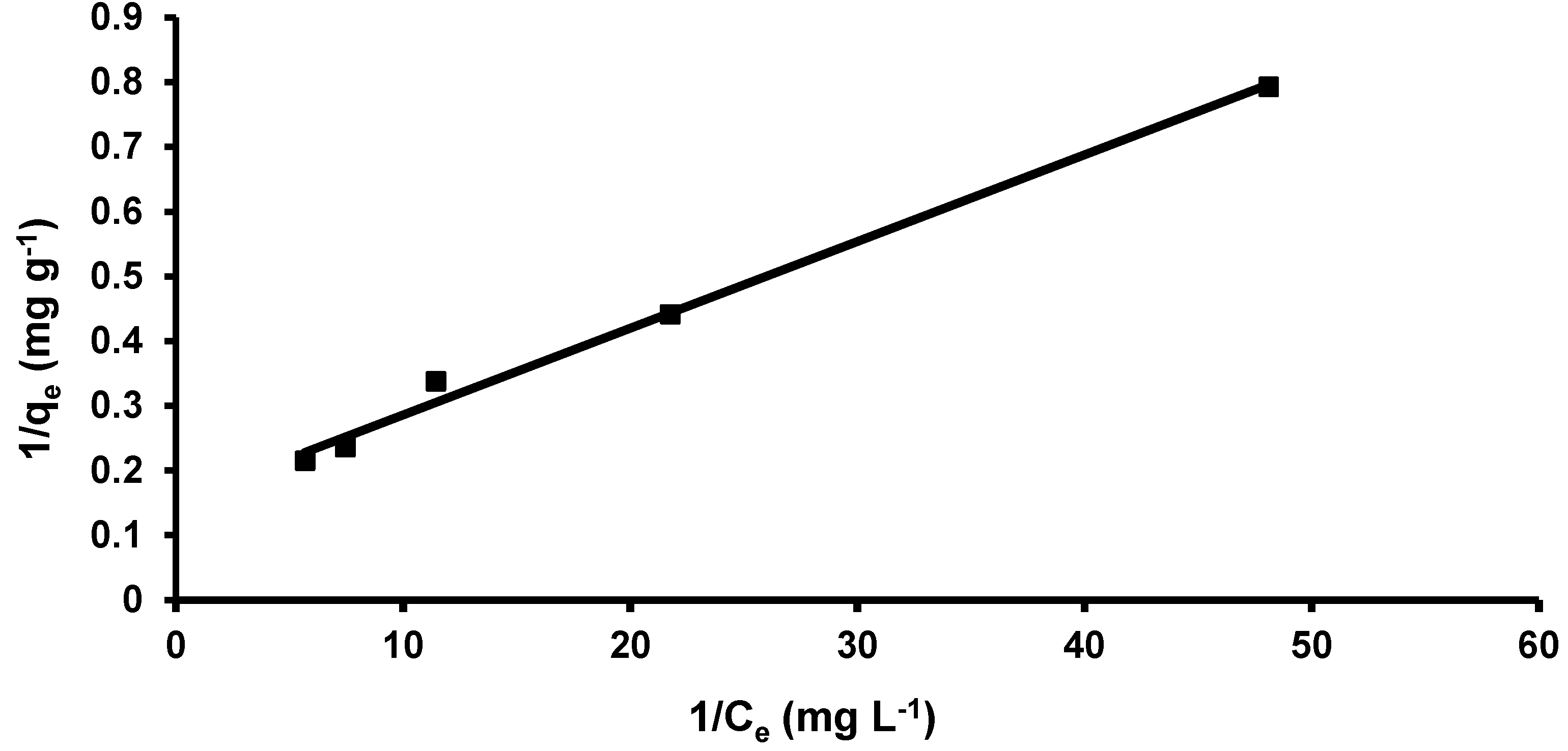
3.3.2. Langmuir Isotherm
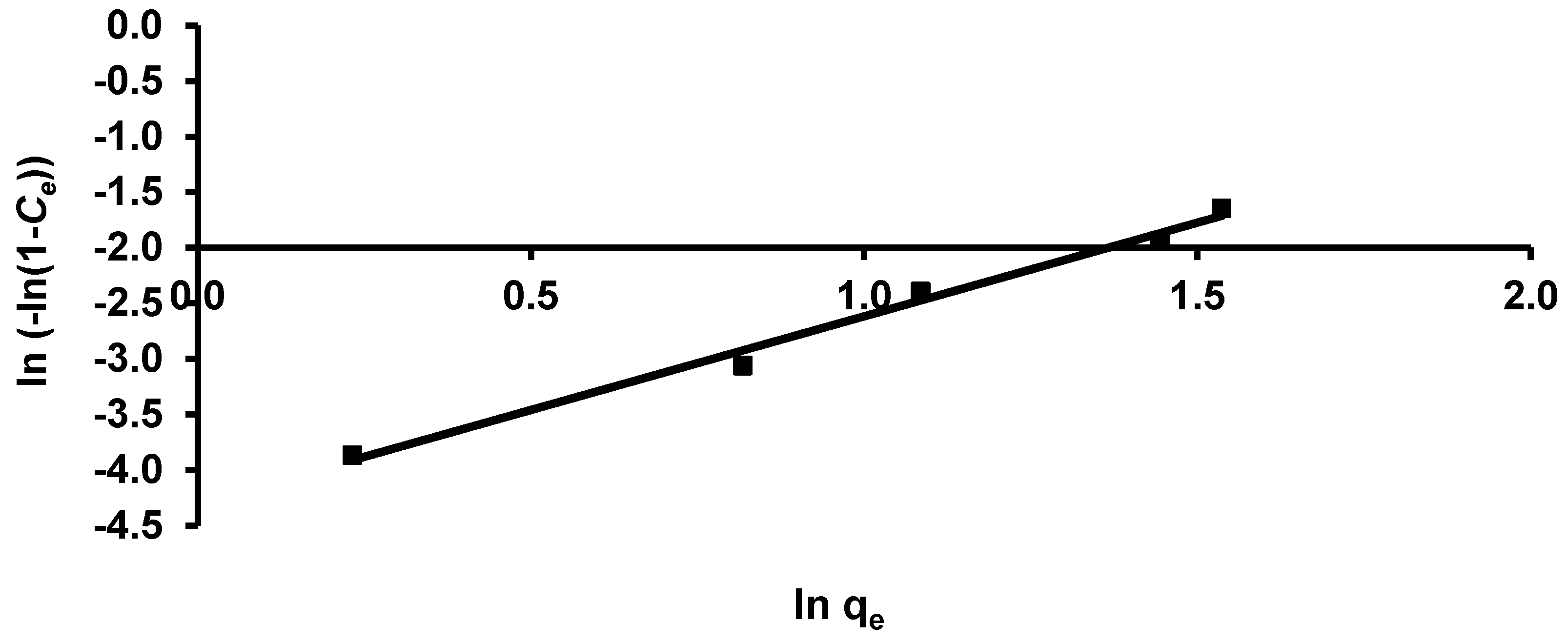
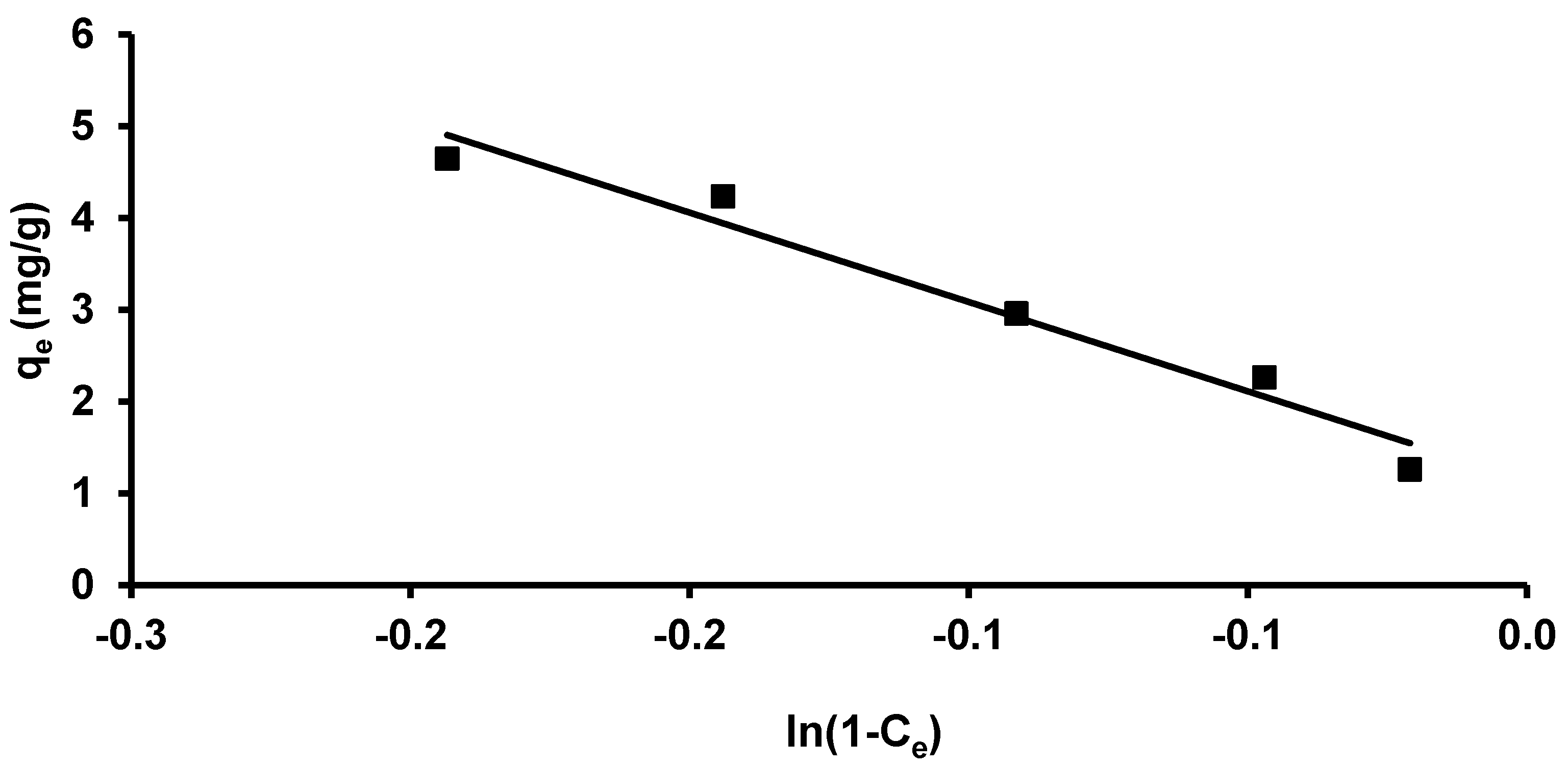
3.3.3. Halsey and Henderson Isotherm
3.3.4. Harkins–Jura Isotherm
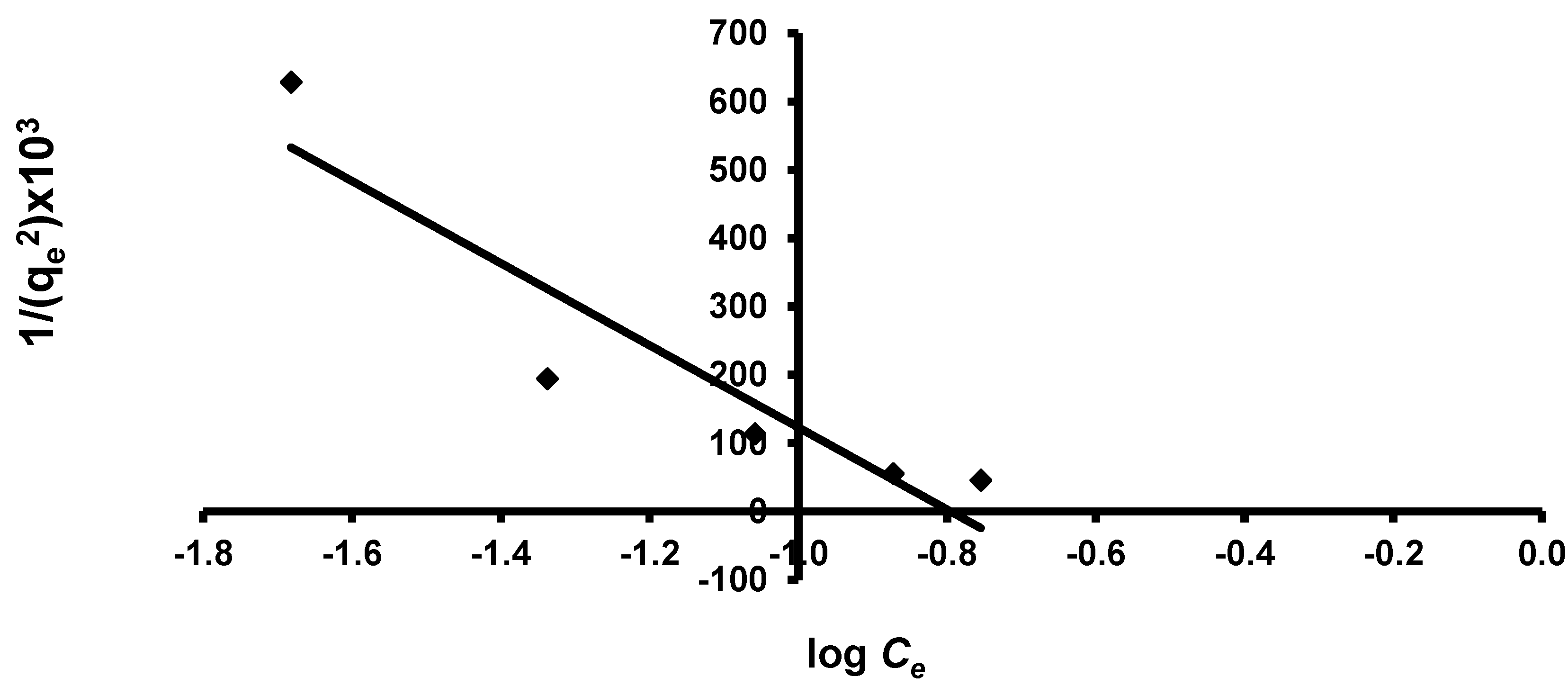
3.3.5. Smith Isotherm
3.3.6. Tempkin Isotherm
3.3.7. Error Function Examination for the Best and Most Appropriate Isotherm Model
3.4. Adsorption Kinetics
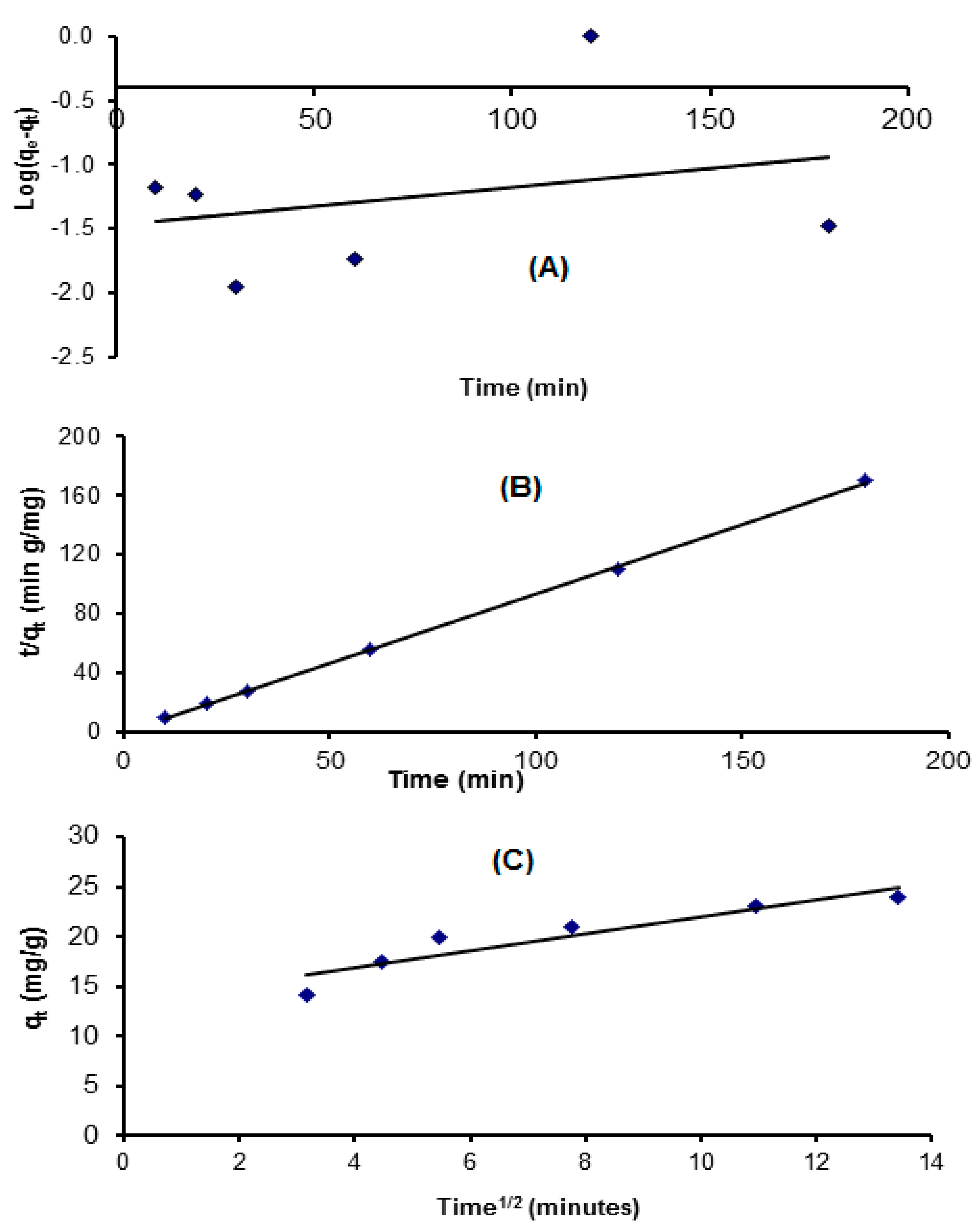
3.4.1. Pseudo-First-Order Kinetics Model
3.4.2. Pseudo-Second-Order Kinetics Model
3.4.3. The Intraparticle Diffusion Equation
3.5. Adsorption Thermodynamics
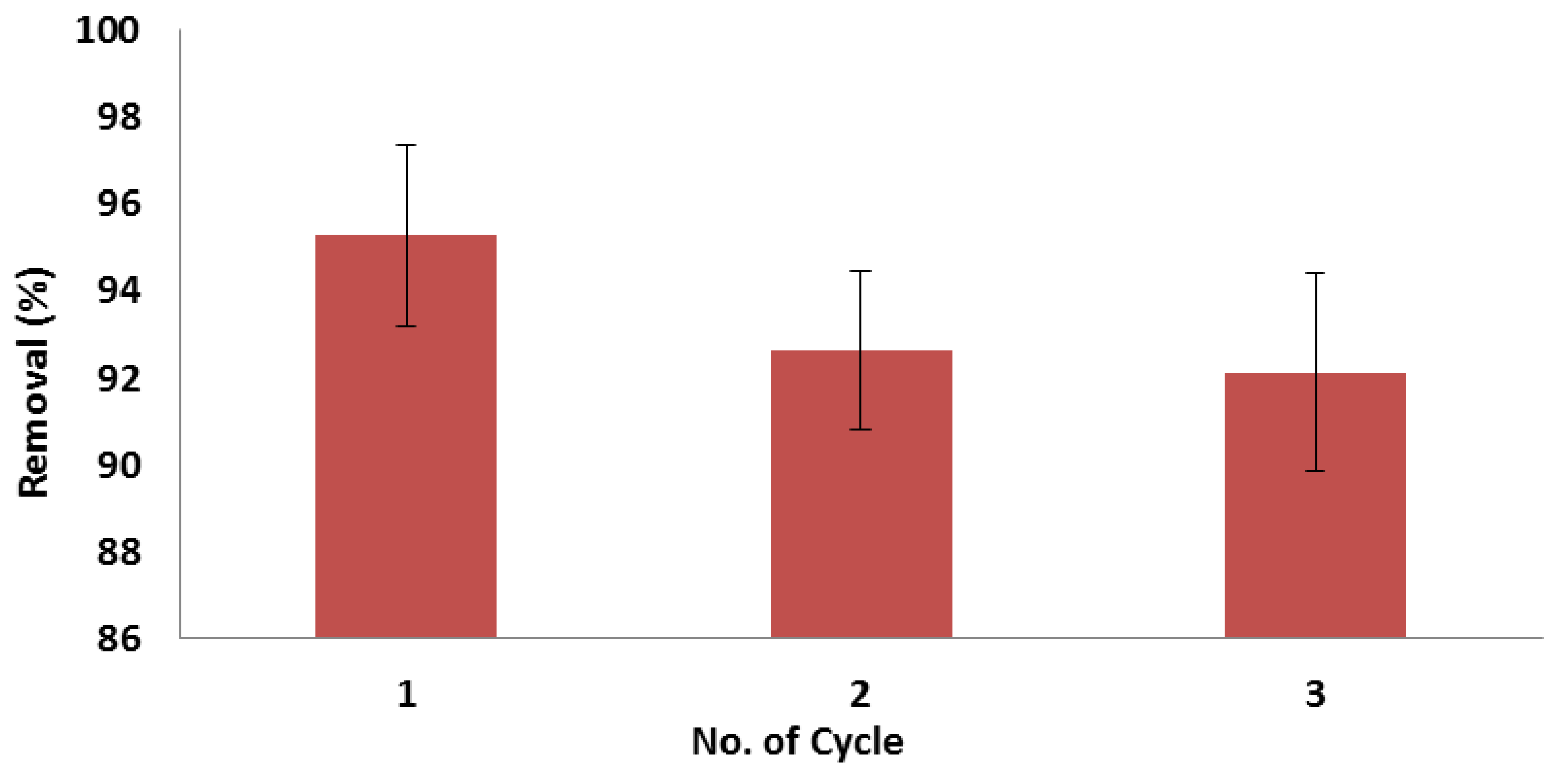
3.6. Regeneration and Reusability Study
3.7. Applicability on Actual Wastewater
3.8. Comparative Studies of the Sorption Capacity of MWCNTs
3.9. Future Research Perspectives and Hallenges
- (a)
- Improving CNT filters, films, and sheets for real industrial wastewater purification on an experimental scale.
- (b)
- Creating purified and functionalized compounds of CNT in commercial amounts with little environmental impact at a reasonable price.
- (c)
- Producing CNTs with comparable adsorption capabilities through various techniques such as laser ablation, chemical vapor deposition, and arc discharge processes.
- (d)
- Predicting the adsorption mechanism and dye elimination capability from real industrial wastewater under a range of operating conditions in batch and column processes, as well as on a larger scale.
- (e)
- Examining the toxicity of CNTs to the environment.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rajabi, M.; Mahanpoor, K.; Moradi, O. Removal of dye molecules from aqueous solution by carbon nanotubes and carbon nanotube functional groups: Critical review. RSC Adv. 2017, 7, 47083–47090. [Google Scholar] [CrossRef]
- El Moselhy, K.M.; Azzem, M.A.; Amer, A.; AlProl, A.E. Al Adsorption of Cu (II) and Cd (II) from Aqueous Solution by Using Rice Husk Adsorbent. Phys. Chem. Indian J. 2017, 12, 1–13. [Google Scholar]
- Al Prol, A.E.; El-Azzem, M.A.; Amer, A.; El-Metwally, M.E.A.; El-Hamid, H.T.A.; El-Moselhy, K.M. Adsorption of Cadmium (II) Ions (II) from Aqueous Solution onto Mango Leaves. Asian J. Phys. Chem. Sci. 2017, 2, 1–11. [Google Scholar] [CrossRef]
- Saratale, G.D.; Saratale, R.G.; Chang, J.S.; Govindwar, S.P. Fixed-bed decolorization of Reactive Blue 172 by Proteus vulgaris NCIM-2027 immobilized on Luffa cylindrica sponge. Int. Biodeterior. Biodegrad. 2011, 65, 494–503. [Google Scholar] [CrossRef]
- Kishor, R.; Purchase, D.; Saratale, G.D.; Saratale, R.G.; Ferreira, L.F.R.; Bilal, M.; Chandra, R.; Bharagava, R.N. Ecotoxicological and health concerns of persistent coloring pollutants of textile industry wastewater and treatment approaches for environmental safety. J. Environ. Chem. Eng. 2021, 9, 105012. [Google Scholar] [CrossRef]
- Al Prol, A.E. Study of Environmental Concerns of Dyes and Recent Textile Effluents Treatment Technology: A Review. Asian J. Fish. Aquat. Res. 2019, 3, 1–18. [Google Scholar] [CrossRef]
- El-hamid, H.T.A.; Al-Prol, A.E.; Hafiz, M.A. Plackett-Burman and Response Surface Methodology for Optimization of Oily Wastewater Bioremediation by Aspergillus sp. South Asian J. Res. Microbiol. 2018, 2, 1–9. [Google Scholar] [CrossRef]
- Miricioiu, M.G.; Niculescu, V.-C. Fly Ash, from Recycling to Potential Raw Material for Mesoporous Silica Synthesis. Nanomaterials 2020, 10, 474. [Google Scholar] [CrossRef]
- Niculescu, V. Mesoporous silica—Efficient media for phenols removal from wastewater. Sect. Hydrol. Water Resour. 2017. [Google Scholar] [CrossRef]
- Niculescu, V. Experimental set-up for simultaneously wastewater treatment and carbon dioxide separation through porous materials. In Proceedings of the 17th International Multidisciplinary Scientific GeoConference SGEM2017, Energy and Clean Technologies, Vienna, Austria, 27–29 November 2017. [Google Scholar] [CrossRef]
- Nourmoradi, H.; Ghiasvand, A.; Noorimotlagh, Z. Removal of methylene blue and acid orange 7 from aqueous solutions by activated carbon coated with zinc oxide (ZnO) nanoparticles: Equilibrium, kinetic, and thermodynamic study. Desalanation Water Treat. 2015, 55, 252–262. [Google Scholar] [CrossRef]
- Samad, S.A.; Huq, D.; Masum, M.M.A.S.M. Textile Dye Removal from Wastewater Effluents Using Chitosan-ZnO Nanocomposite. J. Text. Sci. Eng. 2015, 5, 5–8. [Google Scholar] [CrossRef]
- Nakkeeran, E.; Varjani, S.J.; Dixit, V.; Kalaiselvi, A. Synthesis, characterization and application of zinc oxide nanocomposite for dye removal from textile industrial wastewater. Indian J. Exp. Biol. 2018, 56, 498–503. [Google Scholar]
- Sampranpiboon, P.; Charnkeitkong, P.; Feng, X. Equilibrium isotherm models for adsorption of zinc (II) ion from aqueous solution on pulp waste. WSEAS Trans. Environ. Dev. 2014, 10, 35–47. [Google Scholar]
- Al Prol, A.E.; El-Metwally, M.E.A.; Amer, A. Sargassum latifolium as eco-friendly materials for treatment of toxic nickel (II) and lead (II) ions from aqueous solution. Egypt. J. Aquat. Biol. Fish. 2019, 23, 285–299. [Google Scholar] [CrossRef]
- Eid, A.; Prol, A. Adsorption and Bioremediation as Technologies of Wastewater Treatment. Ph.D. Thesis, University of Almonifia, Tanta, Egypt, 2017. [Google Scholar]
- Zhao, D.; Zhang, W.; Chen, C.; Wang, X. Adsorption of Methyl Orange Dye onto Multiwalled Carbon Nanotubes. Procedia Environ. Sci. 2013, 18, 890–895. [Google Scholar] [CrossRef]
- Saber-Samandari, S.; Saber-Samandari, S.; Joneidi-Yekta, H.; Mohseni, M. Adsorption of anionic and cationic dyes from aqueous solution using gelatin-based magnetic nanocomposite beads comprising carboxylic acid functionalized carbon nanotube. Chem. Eng. J. 2017, 308, 1133–1144. [Google Scholar] [CrossRef]
- Machado, F.M.; Bergmann, C.P.; Lima, E.C.; Royer, B.; de Souza, F.E.; Jauris, I.M.; Calvete, T.; Fagan, S.B. Adsorption of Reactive Blue 4 dye from water solutions by carbon nanotubes: Experiment and theory. Phys. Chem. Chem. Phys. 2012, 14, 11139–11153. [Google Scholar] [CrossRef]
- Mashkoor, F.; Nasar, A. Inamuddin Carbon nanotube-based adsorbents for the removal of dyes from waters: A review. Environ. Chem. Lett. 2020, 18, 605–629. [Google Scholar] [CrossRef]
- Yao, Y.; Bing, H.; Feifei, X.; Xiaofeng, C. Equilibrium and kinetic studies of methyl orange adsorption on multiwalled carbon nanotubes. Chem. Eng. J. 2011, 170, 82–89. [Google Scholar] [CrossRef]
- Shirmardi, M.; Mahvi, A.H.; Hashemzadeh, B.; Naimabadi, A.; Hassani, G.; Niri, M.V. The adsorption of malachite green (MG) as a cationic dye onto functionalized multi walled carbon nanotubes. Korean J. Chem. Eng. 2013, 30, 1603–1608. [Google Scholar] [CrossRef]
- Kareem, A.; Alrazak, N.A.; Aljebori, K.H.; Aljeboree, A.M.; Algboory, H.L.; Alkaim, A.F. Removal of methylene blue dye from aqueous solutions by using activated carbon/ureaformaldehyde composite resin as an adsorbent. Int. J. Chem. Sci. 2016, 14, 635–648. [Google Scholar]
- Machado, F.; Bergmann, C.P.; Lima, E.C.; Adebayo, M.; Fagan, S.B. Adsorption of a textile dye from aqueous solutions by carbon nanotubes. Mater. Res. 2014, 17, 153–160. [Google Scholar] [CrossRef]
- Yu, J.; Zhao, X.; Yang, H.; Chen, X.; Yang, Q.; Yu, L. Aqueous adsorption and removal of organic contaminants by carbon nanotubes. Sci. Total Environ. 2014, 483, 241–251. [Google Scholar] [CrossRef]
- Mohammed, M.I.; Razak, A.A.A.; Al-Timimi, D.A.H. Modified Multiwalled Carbon Nanotubes for Treatment of Some Organic Dyes in Wastewater. Adv. Mater. Sci. Eng. 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Bahgat, M.; Farghali, A.; El-Rouby, W.; Khedr, M. Synthesis and modification of multi-walled carbon nano-tubes (MWCNTs) for water treatment applications. J. Anal. Appl. Pyrolysis 2011, 92, 307–313. [Google Scholar] [CrossRef]
- El-Mohdy, H.L.A.; Mostafa, T.B. Synthesis of Polyvinyl Alcohol/Maleic Acid Hydrogels by Electron Beam Irradiation for Dye Uptake. J. Macromol. Sci. Part A Pure Appl. Chem. 2013, 50, 6–17. [Google Scholar] [CrossRef]
- Ghoneim, M.M.; El-Desoky, H.S.; El-Moselhy, K.M.; Amer, A.; El-Naga, E.H.A.; Mohamedein, L.I.; Al-Prol, A.E. Removal of cadmium from aqueous solution using marine green algae, Ulva lactuca. Egypt. J. Aquat. Res. 2014, 40, 235–242. [Google Scholar] [CrossRef]
- Langmuir, A.O.D.; Temkin, F.; Radushkevich, D. Isotherms Studies of Equilibrium Sorption of Zn2+ Unto Phosphoric Acid Modified Rice Husk. IOSR J. Appl. Chem. 2012, 3, 38–45. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Über die adsorption in solution. J. Phys. Chem 1906, 57, 1100–1107. [Google Scholar]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. Part II—Liquids. J. Frankl. Inst. 1917, 184, 721. [Google Scholar] [CrossRef]
- Halsey, G. Physical Adsorption on Non-Uniform Surfaces. J. Chem. Phys. 1948, 16, 931–937. [Google Scholar] [CrossRef]
- Harkins, D.; Jura, G. An adsorption method for the determination of the area of a solid without the assumption of a molecular area, and the area occupied by nitrogen molecules on the surfaces of solids. J. Chem. Phys. 1944, 66, 1366–1373. [Google Scholar] [CrossRef]
- Mall, I.D.; Srivastava, V.C.; Agarwal, N.K. Removal of Orange-G and Methyl Violet dyes by adsorption onto bagasse fly ash—Kinetic study and equilibrium isotherm analyses. Dye Pigment. 2006, 69, 210–223. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Potgieter, J.H.; Pearson, S.; Pardesi, C. Kinetic and Thermodynamic Parameters for the Adsorption of Methylene Blue Using Fly Ash under Batch, Column, and Heap Leaching Configurations. Coal Combust. Gasif. Prod. 2018, 11, 22–33. [Google Scholar] [CrossRef]
- Kumar, Y.P.; King, P.; Prasad, V.S.R.K. Removal of copper from aqueous solution using Ulva fasciata sp.—A marine green algae. J. Hazard. Mater. 2006, 137, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Al-Aoh, H.A.; Yahya, R.; Maah, M.J.; Bin-Abas, M.R. Adsorption of methylene blue on activated carbon fiber prepared from coconut husk: Isotherm, kinetics and thermodynamics studies. Desalination Water Treat. 2013, 52, 6720–6732. [Google Scholar] [CrossRef]
- Özacar, M.; Şengil, I. Application of kinetic models to the sorption of disperse dyes onto alunite. Colloids Surf. A Physicochem. Eng. Asp. 2004, 242, 105–113. [Google Scholar] [CrossRef]
- Babarinde, N.A.A.; Oyesiku, O.O.; Dairo, O.F. Isotherm and thermodynamic studies of the biosorption of copper (II) ions by Erythrodontium barteri. Int. J. Phys. Sci. 2007, 2, 300–304. [Google Scholar]
- El-Feky, A.M.; Alprol, A.E.; Heneash, A.M.M.; Abo-Taleb, H.A.; Yousif, M. Evaluation of Water Quality and Plankton for Mahmoudia Canal in Northern West of Egypt. Egypt. J. Aquat. Biol. Fish. 2019, 22, 461–474. [Google Scholar] [CrossRef][Green Version]
- Thamri, A.; Baccar, H.; Struzzi, C.; Bittencourt, C.; Abdel-Ghani, A.; Llobet, E. MHDA-Functionalized Multiwall Carbon Nanotubes for detecting non-aromatic VOCs. Nat. Publ. Gr. 2016, 6, 35130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zou, H.; Qing, Q.; Yang, Y.; Li, Q.; Liu, Z. Effect of Chemical Oxidation on the Structure of Single-Walled Carbon Nanotubes. J. Chem. Phys. B 2003, 107, 3712–3718. [Google Scholar] [CrossRef]
- Dutta, D.P.; Venugopalan, R.; Chopade, S. Manipulating Carbon Nanotubes for Efficient Removal of Both Cationic and Anionic Dyes from Wastewater. Mater. Sci. Inc. Nanomater. Polym. 2017, 2, 3878–3888. [Google Scholar] [CrossRef]
- Salam, M.A.; Burk, R. Synthesis and characterization of multi-walled carbon nanotubes modified with octadecylamine and polyethylene glycol. Arab. J. Chem. 2017, 10, S921–S927. [Google Scholar] [CrossRef]
- Das, R.; Abd Hamid, S.B.; Ali, M.E.; Ramakrishna, S.; Yongzhi, W. Carbon Nanotubes Characterization by X-ray Powder Diffraction—A Review. Curr. Nanosci. 2014, 11, 23–35. [Google Scholar] [CrossRef]
- Maryam, M.; Suriani, A.; Shamsudin, M.S.; Rusop, M.; Mahmood, M.R. BET Analysis on Carbon Nanotubes: Comparison between Single and Double Stage Thermal CVD Method. Adv. Mater. Res. 2012, 626, 289–293. [Google Scholar] [CrossRef]
- Romantsova, I.; Burakov, A.; Kucherova, A.; Neskoromnaya, E.; Babkin, A.; Tkachev, A. Liquid-Phase Adsorption of an Organic Dye on Non-Modified and Nanomodified Activated Carbons: Equilibrium and Kinetic Analysis. Adv. Mater. Technol. 2016, 42–48. [Google Scholar] [CrossRef]
- Ngoh, Y.Y.; Leong, Y.-H.; Gan, C.Y. Optimization study for synthetic dye removal using an agricultural waste of Parkia speciosa pod: A sustainable approach for waste water treatment. Int. Food Res. J. 2015, 22, 2351–2357. [Google Scholar]
- Sadik, R.; Lahkale, R.; Hssaine, N. Removal of textile dye by mixed oxide-LDH: Kinetics, isotherms of the adsorption and retention mechanisms studies. IOSR J. Environ. Sci. Toxicol. Food Technol. 2014, 8, 28–36. [Google Scholar] [CrossRef]
- Ananta, S.; Saumen, B.; Vijay, V. Adsorption Isotherm, Thermodynamic and Kinetic Study of Arsenic (III) on Iron Oxide Coated Granular Activated Charcoal. Int. Res. J. Environ. Sci. 2015, 4, 64–77. [Google Scholar]
- Joseph, D.A. The effect of ph and biomass concentration on lead (pb) adsorption by aspergillus niger from simulated waste water. Sci. China Life Sci. 2008, 49, 69–73. [Google Scholar]
- Senthil-Kumar, P.; Gayathri, R. Adsorption of Pb2+ ions from aqueous solutions onto bael tree leaf powder: Isotherms, kinetics and thermodynamics study. J. Eng. Sci. Technol. 2009, 4, 381–399. [Google Scholar]
- Hashem, F.S. Removal of Methylene Blue by Magnetite-Covered Bentonite Nano-Composite. Chem. Bull 2013, 2013, 524–529. [Google Scholar] [CrossRef]
- Elzain, A.A.; El-Aassar, M.R.; Hashem, F.S.; Mohamed, F.M.; Ali, A.S.M. Removal of methylene dye using composites of poly (styrene-co-acrylonitrile) nanofibers impregnated with adsorbent materials. J. Mol. Liq. 2019, 291, 111335. [Google Scholar] [CrossRef]
- Putra, W.P.; Kamari, A.; Najiah, S.; Yusoff, M.; Ishak, C.F. Biosorption of Cu (II), Pb (II) and Zn (II) Ions from Aqueous Solutions Using Selected Waste Materials: Adsorption and Characterisation Studies. J. Encapsulation Adsorpt. Sci. 2014, 4, 25–35. [Google Scholar] [CrossRef]
- Kumar, P.S.; Ramakrishnan, K.; Kirupha, S.D.; Sivanesan, S. Thermodynamic and kinetic studies of cadmium adsorption from aqueous solution onto rice husk. Braz. J. Chem. Eng. 2010, 27, 347–355. [Google Scholar] [CrossRef]
- Malakootian, M.; Toolabi, A.; Moussavi, S.G.; Ahmadian, M. Equilibrium and Kinetic Modeling of Heavy Metals Biosorption from Three Different Real Industrial Wastewaters onto Ulothrix Zonata Algae. Aust. J. Basic Appl. Sci. 2011, 5, 1030–1037. [Google Scholar]
- Volesky, B. Biosorption: Application Aspects—Process Simulation Tools. Hydrometallurgy 2001, 11, 69–80. [Google Scholar]
- Kooh, M.R.R.; Dahri, M.K.; Lim, L.B. The removal of rhodamine B dye from aqueous solution using Casuarina equisetifolia needles as adsorbent. Cogent Environ. Sci. 2016, 2. [Google Scholar] [CrossRef]
- Abdelkarim, S.; Mohammed, H.; Nouredine, B. Sorption of Methylene Blue Dye from Aqueous Solution Using an Agricultural Waste. Trends Green Chem. 2017, 3, 1–7. [Google Scholar] [CrossRef]
- Abdelwahab, O.; Amin, N. Adsorption of phenol from aqueous solutions by Luffa cylindrica fibers: Kinetics, isotherm and thermodynamic studies. Egypt. J. Aquat. Res. 2013, 39, 215–223. [Google Scholar] [CrossRef]
- Nessim, R.B.; Bassiouny, A.R.; Zaki, H.R.; Moawad, M.N.; Kandeel, K.M. Biosorption of lead and cadmium using marine algae. Chem. Ecol. 2011, 27, 579–594. [Google Scholar] [CrossRef]
- Ghaedi, M.; Shojaeipour, E.; Ghaedi, A.M.; Sahraei, R. Isotherm and kinetics study of malachite green adsorption onto copper nanowires loaded on activated carbon: Artificial neural network modeling and genetic algorithm optimization. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2015, 142, 1–57. [Google Scholar] [CrossRef]
- Mondal, P.; Baksi, S.; Bose, D. Study of Environmental Issues in Textile Industries and Recent Wastewater Treatment Technology. World Sci. News 2017, 61, 98–109. [Google Scholar]
- Yao, Y.; Xu, F.; Chen, M.; Xu, Z.; Zhu, Z. Bioresource Technology Adsorption behavior of methylene blue on carbon nanotubes. Bioresour. Technol. 2010, 101, 3040–3046. [Google Scholar] [CrossRef]
- Rodriguez, A.; Ovejero, G.; Sotelo, J.L.; Mestanza, M.; Garcia, J. Adsorption of dyes on carbon nanomaterials from aqueous solutions. J. Environ. Sci. Health Part A 2010, 45, 1642–1653. [Google Scholar] [CrossRef] [PubMed]
- Moradi, O. Adsorption Behavior of Basic Red 46 by Single-Walled Carbon Nanotubes Surfaces. Full Nanotub. Carbon Nanostruct. 2013, 21, 37–41. [Google Scholar] [CrossRef]
- Jamshidi, H.; Ghaedi, M.; Sabzehmeidani, M.M.; Bagheri, A.R. Comparative study of acid yellow 119 adsorption onto activated carbon prepared from lemon wood and ZnO nanoparticles loaded on activated carbon. Appl. Organomet. Chem. 2018, 32, e4080. [Google Scholar] [CrossRef]
- Das, D.; Charumathi, D.; Das, N. Bioaccumulation of the synthetic dye Basic Violet 3 and heavy metals in single and binary systems by Candida tropicalis grown in a sugarcane bagasse extract medium: Modelling optimal conditions using response surface methodology (RSM) and inhibition kinetics. J. Hazard. Mater. 2011, 186, 1541–1552. [Google Scholar] [CrossRef]
- Konicki, W.; Pełech, I.; Mijowska, E.; Jasińska, I. Adsorption of anionic dye Direct Red 23 onto magnetic multi-walled carbon nanotubes-Fe3C nanocomposite: Kinetics, equilibrium and thermodynamics. Chem. Eng. J. 2012, 210, 87–95. [Google Scholar] [CrossRef]
- Inyinbor, A.A.; Adekola, F.A.; Olatunji, G.A. Kinetics, isotherms and thermodynamic modeling of liquid phase adsorption of Rhodamine B dye onto Raphia hookerie fruit epicarp. Water Resour. Ind. 2016, 15, 14–27. [Google Scholar] [CrossRef]



| Characteristics | Value |
|---|---|
| Dye name | ISMATE violate 2R |
| Wavelength (λ max) | 550 nm |
| Mol. wt. | 700 |
| Molecular formula | C22H14N4O11S3CuCl |
| C.I. name | IV2R |
| Molecular structure | 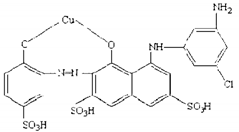 |
| Isotherm Model | Isotherm Parameter | Value |
|---|---|---|
| Freundlich | 1/n | 13.77 |
| KF (mg1−1/nL1/ng−1) | 0.608 | |
| R2 | 0.989 | |
| Langmuir | Qmax (mg g−1) | 76.92 |
| b | 1.42 | |
| R2 | 0.99 | |
| Harkins–Jura | AHJ | 1.31 |
| BHJ | 1.73 | |
| R2 | 0.858 | |
| Halsey | 1/nH | 0.6089 |
| KH | 2.714 | |
| R2 | 0.989 | |
| Henderson | 1/nh | 1.688 |
| Kh | 0.014 | |
| R2 | 0.988 | |
| Smith | Wbs | 1.138 |
| Ws | 19.46 | |
| R2 | 0.964 | |
| Tempkin | AT | 95.58 |
| BT | 1.59 | |
| bT | 1558.2 | |
| R2 | 0.965 |
| Isotherm Model | APE% | χ2 | EABS |
|---|---|---|---|
| Freundlich | 0.031 | 0 | 0.034 |
| Langmuir | 2.079 | 0.326 | 2.236 |
| Harkins–Jura | 85.602 | 551.664 | 92.065 |
| Halsey | 12.38 | 11.539 | 13.315 |
| Henderson | 0.022 | 0 | 0.024 |
| Smith | 0.004 | 0 | 0.004 |
| Tempkin | 0.001 | 0 | 0.001 |
| Kinetic Models | Parameters | Value |
|---|---|---|
| First-order | qe (calc.) (mg g−1) | 29.3 |
| k1 × 103 (min−1) | 6.6787 | |
| R2 | 0.0797 | |
| Second-order | qe (calc.) (mg g−1) | 1.066 |
| k2 × 103 (mg g−1 min−1) | 9300.677 | |
| R2 | 0.9993 | |
| Intraparticle diffusion | Kdif (mg g−1 min−0.5) | 13.434 |
| C cal (mg g−1) | 0.862 | |
| R2 | 0.867 |
| Temperature (°C) | ∆G° (kJ mol−1) | ∆H° (kJ mol−1) | ΔS° (J mol−1) |
|---|---|---|---|
| 25 | −7.87669 | 21.877 | −98.76 |
| 35 | −7.94698 | ||
| 45 | −9.763 | ||
| 55 | −10.5637 |
| Parameters | Before Treatment | After Treatment | Standards for Cotton Textile Industries [66] |
|---|---|---|---|
| pH | 9.5 | 2–6 | 5.5–9.0 |
| TSS (mg L−1) | 2050 | 110 | 100 |
| TDS (mg L−1) | 3240 | 652 | 500 |
| CNTs | Dye Adsorbed | qe (mg g−1) | Ref. |
|---|---|---|---|
| MWCNTs | Sufranine O | 43.48 | [65] |
| MWCNTs | Methylene blue | 35.4 | [67] |
| Oxidize MWCNTs | Bromothymol blue | 55 | [25] |
| SWCNT–COOH | Basic red 46 | 45.33 | [1] |
| MWCNTs | methylene blue | 64.7 | [67] |
| MWNTs | Orange II | 66.12 | [68] |
| SWCNT | Basic red 46 | 38.35 | [69] |
| MWNTs | Reactive blue | 335.7 | [19] |
| MWNTs | Alizarin red S | 161.290 | [70] |
| MWCNTs | Reactive blue 4 | 502.5 | [19] |
| MWNTs | Methyl orange | 52.86 | [17] |
| CNTs/activated carbon fiber | Basic violet 10 | 220 | [71] |
| MWCNTs/Fe3C | Direct Red 23 | 172 | [72] |
| Chitosan/Fe2O3/MWCNTs | Methyl orange | 66.90 | [21] |
| MWCNTs/Fe2O3 | Methylene blue | 42.3 | [73] |
| MWCNT | 76.9 | Present study | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abualnaja, K.M.; Alprol, A.E.; Ashour, M.; Mansour, A.T. Influencing Multi-Walled Carbon Nanotubes for the Removal of Ismate Violet 2R Dye from Wastewater: Isotherm, Kinetics, and Thermodynamic Studies. Appl. Sci. 2021, 11, 4786. https://doi.org/10.3390/app11114786
Abualnaja KM, Alprol AE, Ashour M, Mansour AT. Influencing Multi-Walled Carbon Nanotubes for the Removal of Ismate Violet 2R Dye from Wastewater: Isotherm, Kinetics, and Thermodynamic Studies. Applied Sciences. 2021; 11(11):4786. https://doi.org/10.3390/app11114786
Chicago/Turabian StyleAbualnaja, Khamael M., Ahmed E. Alprol, Mohamed Ashour, and Abdallah Tageldein Mansour. 2021. "Influencing Multi-Walled Carbon Nanotubes for the Removal of Ismate Violet 2R Dye from Wastewater: Isotherm, Kinetics, and Thermodynamic Studies" Applied Sciences 11, no. 11: 4786. https://doi.org/10.3390/app11114786
APA StyleAbualnaja, K. M., Alprol, A. E., Ashour, M., & Mansour, A. T. (2021). Influencing Multi-Walled Carbon Nanotubes for the Removal of Ismate Violet 2R Dye from Wastewater: Isotherm, Kinetics, and Thermodynamic Studies. Applied Sciences, 11(11), 4786. https://doi.org/10.3390/app11114786








