Machine Learning Models and Statistical Complexity to Analyze the Effects of Posture on Cerebral Hemodynamics
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Measurement
2.2. Methods
2.2.1. Machine Learning Models of dCA
2.2.2. Statistic Complexity of Hemodynamics
2.3. Statistical Analysis
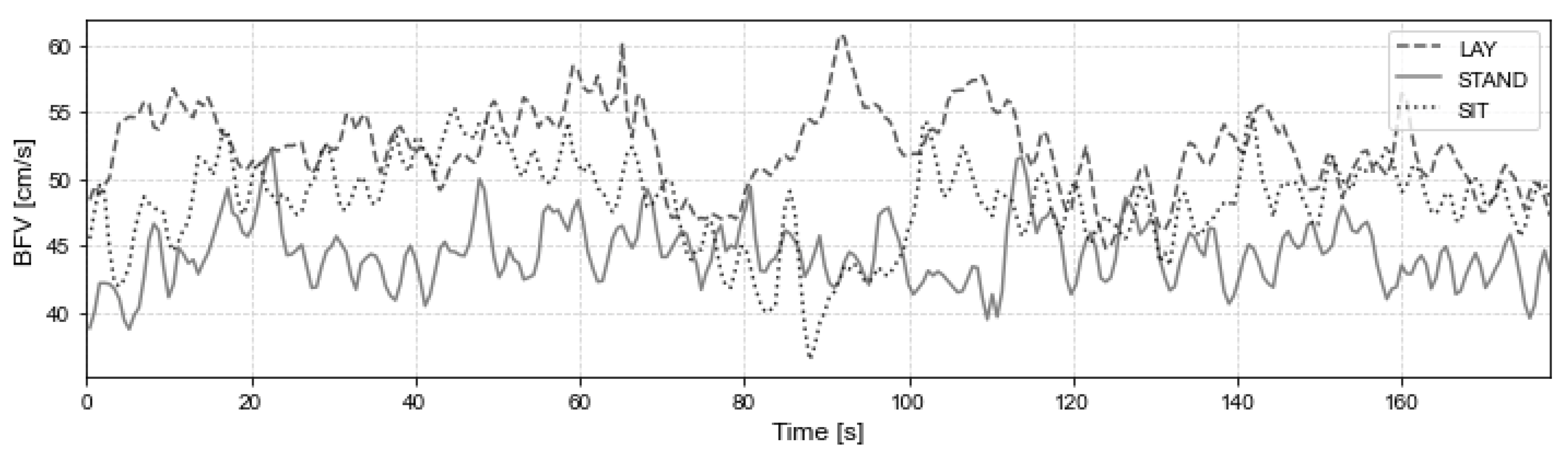
3. Results
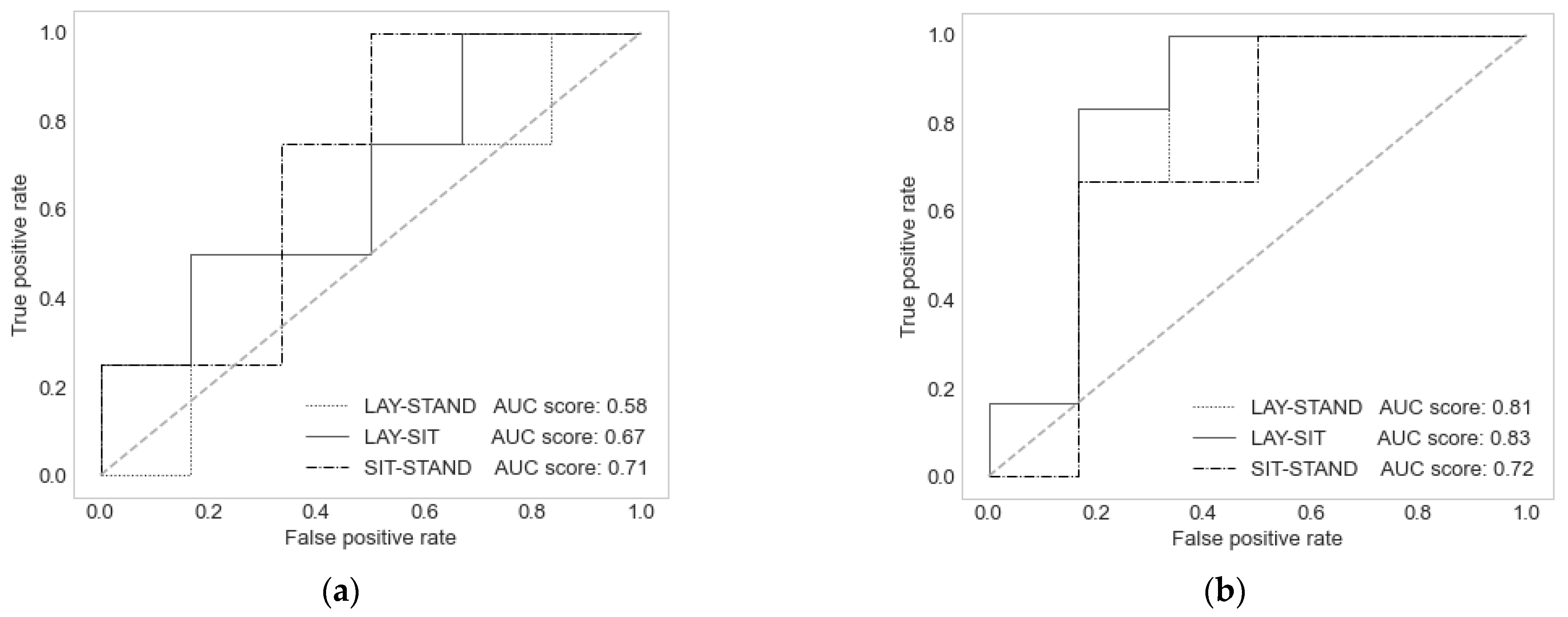
3.1. SVM Models
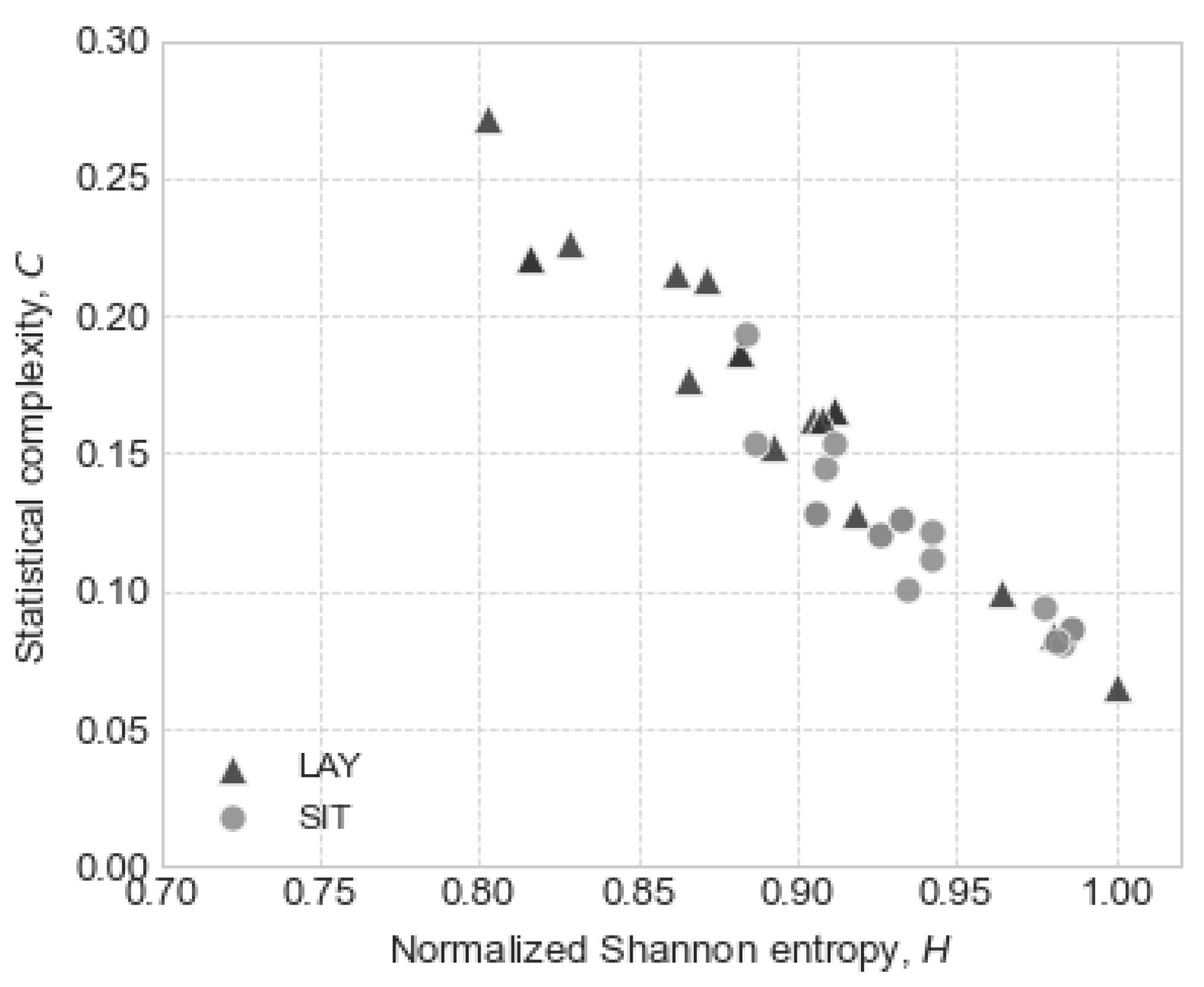
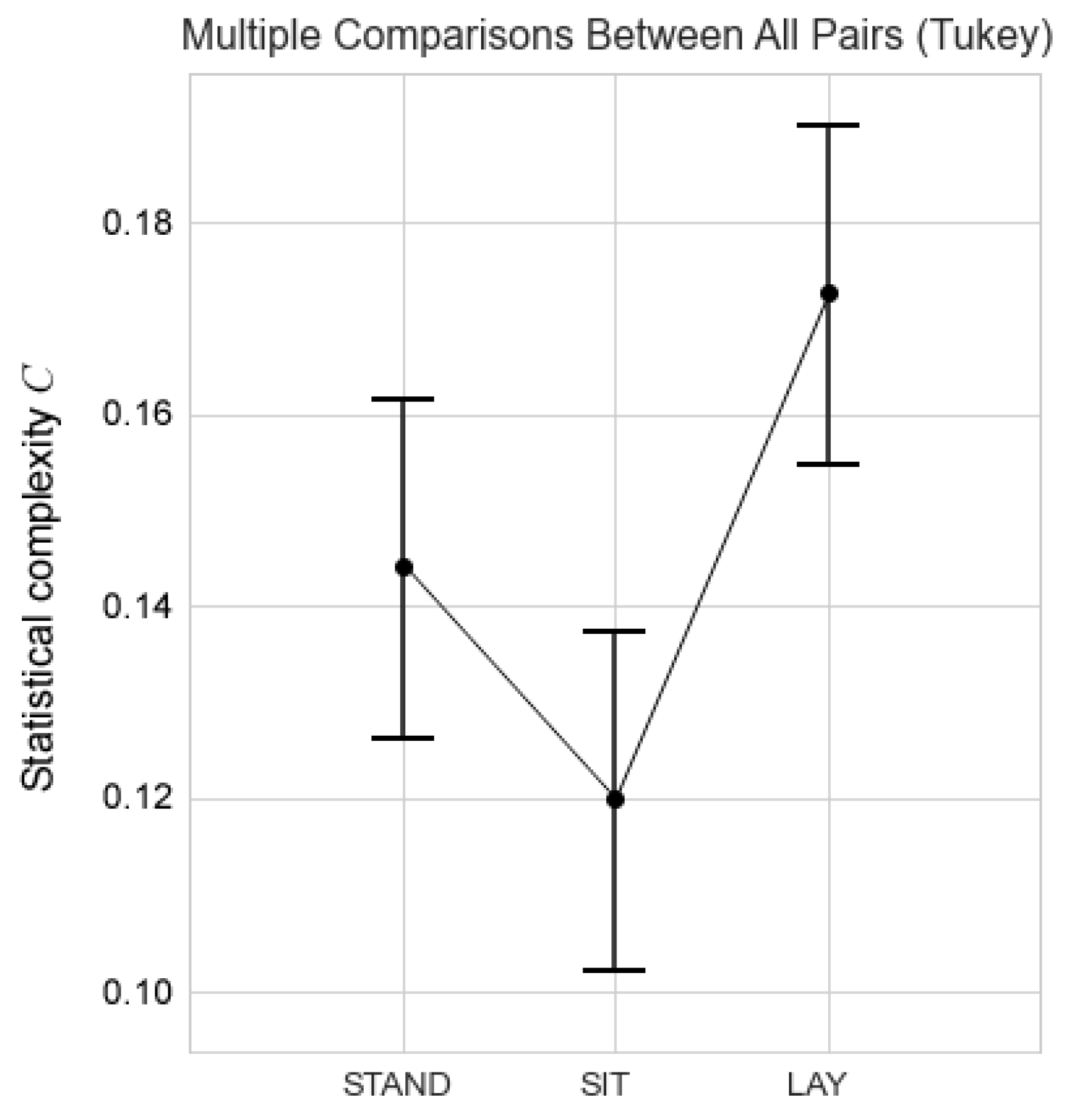
3.2. Statistic Entropy and Complexity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Classic Dynamic Autoregulation Index
| T | D | K | ARI |
|---|---|---|---|
| 2.00 | 1.70 | 0.00 | 0 |
| 2.00 | 1.60 | 0.20 | 1 |
| 2.00 | 1.50 | 0.40 | 2 |
| 2.00 | 1.15 | 0.60 | 3 |
| 2.00 | 0.90 | 0.80 | 4 |
| 1.90 | 0.75 | 0.90 | 5 |
| 1.60 | 0.65 | 0.94 | 6 |
| 1.20 | 0.55 | 0.96 | 7 |
| 0.87 | 0.52 | 0.97 | 8 |
| 0.65 | 0.50 | 0.98 | 9 |
References
- Aaslid, R.; Lindegaard, K.F.; Sorteberg, W.; Nornes, H. Cerebral autoregulation dynamics in humans. Stroke 1989, 20, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Paulson, O.B.; Strandgaard, S.; Edvinsson, L. Cerebral autoregulation. Cerebrovasc. Brain Metab. Rev. 1990, 2, 161–192. [Google Scholar] [PubMed]
- Tiecks, F.P.; Lam, A.M.; Aaslid, R.; Newell, D.W. Comparison of static and dynamic cerebral autoregulation measurements. Stroke 1995, 26, 1014–1019. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, R.C.; Bor-Seng-Shu, E.; Santos, M.R.; Negrão, C.E.; Teixeira, M.J.; Panerai, R.B. Dynamic cerebral autoregulation changes during sub-maximal handgrip maneuver. PLoS ONE 2013, 8, e70821. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Fisher, J.P.; Seifert, T.; Overgaard, M.; Secher, N.H.; Ogoh, S. Blood flow in internal carotid and vertebral arteries during orthostatic stress. Exp. Physiol. 2012, 97, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Romero, S.A.; Moralez, G.; Rickards, C.A.; Ryan, K.L.; Convertino, V.A.; Fogt, D.L.; Cooke, W.H. Control of cerebral blood velocity with furosemide-induced hypovolemia and upright tilt. J. Appl. Physiol. 2011, 110, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Sorond, F.A.; Serrador, J.M.; Jones, R.N.; Shaffer, M.L.; Lipsitz, L.A. The sit-to-stand technique for the measurement of dynamic cerebral autoregulation. Ultrasound Med. Biol. 2009, 35, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Claassen, J.A.H.R.; Levine, B.D.; Zhang, R. Dynamic cerebral autoregulation during repeated squat-stand maneuvers. J. Appl. Physiol. 2009, 106, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Barnes, S.C.; Ball, N.; Panerai, R.B.; Robinson, T.G.; Haunton, V.J. Random squat/stand maneuvers: A novel approach for assessment of dynamic cerebral autoregulation? J. Appl. Physiol. 2017, 123, 558–566. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barnes, S.C.; Ball, N.; Haunton, V.J.; Robinson, T.G.; Panerai, R.B. How many squat–stand manoeuvres to assess dynamic cerebral autoregulation? Eur. J. Appl. Physiol. 2018, 118, 2377–2384. [Google Scholar] [CrossRef] [PubMed]
- Batterham, A.P.; Panerai, R.B.; Robinson, T.G.; Haunton, V.J. Does depth of squat-stand maneuver affect estimates of dynamic cerebral autoregulation? Physiol. Rep. 2020, 8, e14549. [Google Scholar] [CrossRef] [PubMed]
- Corsini, C.; Cervi, E.; Migliavacca, F.; Schievano, S.; Hsia, T.Y.; Pennati, G. Mathematical modelling of the maternal cardiovascular system in the three stages of pregnancy. Med. Eng. Phys. 2017, 47, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Favre, M.E.; Serrador, J.M. Sex differences in cerebral autoregulation are unaffected by menstrual cycle phase in young, healthy women. Am. J. Physiol. Heart Circ. Physiol. 2019, 316, H920–H933. [Google Scholar] [CrossRef] [PubMed]
- Deegan, B.M.; Sorond, F.A.; Galica, A.; Lipsitz, L.A.; O’Laighin, G.; Serrador, J.M. Elderly women regulate brain blood flow better than men do. Stroke 2011, 42, 1988–1993. [Google Scholar] [CrossRef] [PubMed]
- Van Beek, A.H.; Rikkert, M.G.O.; Pasman, J.W.; Hopman, M.T.; Claassen, J.A. Dynamic cerebral autoregulation in the old using a repeated sit-stand maneuver. Ultrasound Med. Biol. 2010, 36, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Van Beek, A.H.; Claassen, J.A.; Rikkert, M.G.O.; Jansen, R.W. Cerebral autoregulation: An overview of current concepts and methodology with special focus on the elderly. J. Cereb. Blood Flow Metab. 2008, 28, 1071–1085. [Google Scholar] [CrossRef] [PubMed]
- Deegan, B.M.; Devine, E.R.; Geraghty, M.C.; Jones, E.; ÓLaighin, G.; Serrador, J.M. The relationship between cardiac output and dynamic cerebral autoregulation in humans. J. Appl. Physiol. 2010, 109, 1424–1431. [Google Scholar] [CrossRef]
- Garrett, Z.K.; Pearson, J.; Subudhi, A.W. Postural effects on cerebral blood flow and autoregulation. Physiol. Rep. 2017, 5, e13150. [Google Scholar] [CrossRef] [PubMed]
- Gelinas, J.C.; Marsden, K.R.; Tzeng, Y.C.; Smirl, J.D.; Smith, K.J.; Willie, C.K.; Ainslie, P.N. Influence of posture on the regulation of cerebral perfusion. Aviat. Space Environ. Med. 2012, 83, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Panerai, R.B.; Barnes, S.C.; Nath, M.; Ball, N.; Robinson, T.G.; Haunton, V.J. Directional sensitivity of dynamic cerebral autoregulation in squat-stand maneuvers. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 315, R730–R740. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, A.; Nikolic, D.; Birch, A.A.; Olufsen, M.S.; Panerai, R.B.; Simpson, D.M.; Payne, S.J. Increased blood pressure variability upon standing up improves reproducibility of cerebral autoregulation indices. Med. Eng. Phys. 2017, 47, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Giller, C.A.; Mueller, M. Linearity and non-linearity in cerebral hemodynamics. Med. Eng. Phys. 2003, 25, 633–646. [Google Scholar] [CrossRef]
- Panerai, R.B.; Deverson, S.T.; Mahony, P.; Hayes, P.; Evans, D.H. Effect of CO2 on dynamic cerebral autoregulation measurement. Physiol. Meas. 1999, 20, 265. [Google Scholar] [CrossRef]
- Panerai, R.B. The critical closing pressure of the cerebral circulation. Med. Eng. Phys. 2003, 25, 621–632. [Google Scholar] [CrossRef]
- Mitsis, G.D.; Zhang, R.; Levine, B.D.; Marmarelis, V.Z. Modeling of nonlinear physiological systems with fast and slow dynamics. II. Application to cerebral autoregulation. Ann. Biomed. Eng. 2002, 30, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Mitsis, G.D.; Poulin, M.J.; Robbins, P.A.; Marmarelis, V.Z. Nonlinear modeling of the dynamic effects of arterial pressure and CO2 variations on cerebral blood flow in healthy humans. IEEE Trans. Biomed. Eng. 2004, 51, 1932–1943. [Google Scholar] [CrossRef]
- Marmarelis, V.Z.; Shin, D.C.; Zhang, R. Linear and nonlinear modeling of cerebral flow autoregulation using principal dynamic modes. Open Biomed. Eng. J. 2012, 6, 42. [Google Scholar] [CrossRef]
- Hu, K.; Lo, M.T.; Peng, C.K.; Liu, Y.; Novak, V. A nonlinear dynamic approach reveals a long-term stroke effect on cerebral blood flow regulation at multiple time scales. PLoS Comput. Biol. 2012, 8, e1002601. [Google Scholar] [CrossRef]
- Hu, K.; Peng, C.K.; Czosnyka, M.; Zhao, P.; Novak, V. Nonlinear assessment of cerebral autoregulation from spontaneous blood pressure and cerebral blood flow fluctuations. Cardiovasc. Eng. 2008, 8, 60–71. [Google Scholar] [CrossRef]
- Panerai, R.B.; Chacon, M.; Pereira, R.; Evans, D.H. Neural network modelling of dynamic cerebral autoregulation: Assessment and comparison with established methods. Med. Eng. Phys. 2004, 26, 43–52. [Google Scholar] [CrossRef]
- Peng, T.; Rowley, A.B.; Ainslie, P.N.; Poulin, M.J.; Payne, S.J. Wavelet phase synchronization analysis of cerebral blood flow autoregulation. IEEE Trans. Biomed. Eng. 2010, 57, 960–968. [Google Scholar] [CrossRef] [PubMed]
- Chacon, M.; Araya, C.; Panerai, R.B. Non-linear multivariate modeling of cerebral hemodynamics with autoregressive Support Vector Machines. Med. Eng. Phys. 2011, 33, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Schölkopf, B.; Smola, A.J.; Williamson, R.C.; Bartlett, P.L. New support vector algorithms. Neural Comput. 2000, 12, 1207–1245. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, P.J. The use of balanced half-sample replication in cross-validation studies. J. Am. Stat. Assoc. 1976, 71, 596–604. [Google Scholar] [CrossRef]
- Ramos, E.G.; Simpson, D.M.; Panerai, R.B.; Nadal, J.; Lopes, J.M.D.A.; Evans, D.H. Objective selection of signals for assessment of cerebral blood flow autoregulation in neonates. Physiol. Meas. 2005, 27, 35. [Google Scholar] [CrossRef] [PubMed][Green Version]
- R Development Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Available online: http://www.R-project.org/ (accessed on 20 December 2021).
- Chang, C.C.; Lin, C.J. LIBSVM: A library for support vector machines. ACM Trans. Intell. Syst. Technol. 2011, 2, 1–27. [Google Scholar] [CrossRef]
- Meyer, D.; Dimitriadou, E.; Hornik, K.; Weingessel, A.; Leisch, F.; Chang, C.C.; Lin, C. Misc Functions of the Department of Statistics (e1071); Version, 1–6; TU Wien: Vienna, Austria, 2012. [Google Scholar]
- Olkkonen, H. Discrete Wavelet Transforms: Biomedical Applications; BoD–Books on Demand: Norderstedt, Germany, 2011. [Google Scholar]
- Rosso, O.A.; Blanco, S.; Yordanova, J.; Kolev, V.; Figliola, A.; Schürmann, M.; Başar, E. Wavelet entropy: A new tool for analysis of short duration brain electrical signals. J. Neurosci. Methods 2001, 105, 65–75. [Google Scholar] [CrossRef]
- Shannon, C.E. A mathematical theory of communication. Bell System Tech. J. 1948, 27, 623–656. [Google Scholar] [CrossRef]
- Martin, M.T.; Plastino, A.; Rosso, O.A. Statistical complexity and disequilibrium. Phys. Lett. A 2003, 311, 126–132. [Google Scholar] [CrossRef]
- Lopez-Ruiz, R.; Mancini, H.L.; Calbet, X. A statistical measure of complexity. Phys. Lett. A 1995, 209, 321–326. [Google Scholar] [CrossRef]
- Anteneodo, C.; Plastino, A.R. Some features of the López-Ruiz-Mancini-Calbet (LMC) statistical measure of complexity. Phys. Lett. A 1996, 223, 348–354. [Google Scholar] [CrossRef]
- Calbet, X.; López-Ruiz, R. Tendency towards maximum complexity in a nonequilibrium isolated system. Phys. Rev. E 2001, 63, 066116. [Google Scholar] [CrossRef] [PubMed]
- Wootters, W.K. Statistical distance and Hilbert space. Phys. Rev. D 1981, 23, 357. [Google Scholar] [CrossRef]
- Zanin, M.; Zunino, L.; Rosso, O.A.; Papo, D. Permutation entropy and its main biomedical and econophysics applications: A review. Entropy 2012, 14, 1553–1577. [Google Scholar] [CrossRef]
- Goldberger, A.L.; Penga, C.-K.; Lipsitzb, L.A. What is physiologic complexity and how does it change with aging and disease? Neurobiol. Aging 2002, 23, 23–26. [Google Scholar] [CrossRef]
- Crowder, M.J.; Hand, D.J. Analysis of Repeated Measures; Routledge: Abingdon, UK, 2017. [Google Scholar]
- Martínez-Camblor, P.; Carleos, C.; Corral, N. General nonparametric ROC curve comparison. J. Korean Stat. Soc. 2013, 42, 71–81. [Google Scholar] [CrossRef]
- Rosso, O.A.; Carpi, L.C.; Saco, P.M.; Gómez Ravetti, M.; Plastino, A.; Larrondo, H.A. Causality and the entropy–complexity plane: Robustness and missing ordinal patterns. Phys. A 2012, 391, 42–55. [Google Scholar] [CrossRef]
- Zarjam, P.; Epps, J.; Chen, F.; Lovell, N.H. Estimating cognitive workload using wavelet entropy-based features during an arithmetic task. Comput. Biol. Med. 2013, 43, 2186–2195. [Google Scholar] [CrossRef] [PubMed]
- Cavieres, R.; Landerretche, J.; Jara, J.L.; Chacon, M. Analysis of cerebral blood flow entropy while listening to music with emotional content. Physiol. Meas. 2021, 42, 055002. [Google Scholar] [CrossRef] [PubMed]
- Shen, K.; McFadden, A.; McIntosh, A.R. Signal Complexity Indicators of Health Status in Clinical-EEG; BioRxiv: Cold Spring Harbor, NY, USA, 2021. [Google Scholar]

| Posture | Lay | Stand | Sit | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Signal | BFV | BP | BFV | BP | BFV | BP | ||||||
| Side | Right | Left | Mean | - | Right | Left | Mean | - | Right | Left | Mean | - |
| Mean | 58.52 | 60.02 | 59.27 | 74.96 | 54.36 | 52.65 | 53.50 | 84.81 | 53.69 | 52.07 | 52.88 | 81.45 |
| Std | 13.48 | 14.23 | 13.68 | 9.03 | 11.47 | 10.31 | 10.79 | 10.93 | 13.50 | 11.22 | 12.26 | 11.87 |
| CoV | 0.23 | 0.24 | 0.23 | 0.12 | 0.21 | 0.20 | 0.20 | 0.13 | 0.25 | 0.22 | 0.23 | 0.15 |
| Model | np | nv | C | v | σ | CC Lay | CC Stand | CC Sit |
|---|---|---|---|---|---|---|---|---|
| FIR linear | [1–10] | - | [−2, 14 einf] | [0, 1–0, 9] | [−1, 5] | 0.611 | 0.721 | 0.626 |
| FIR non-linear | [1–10] | - | [−2, 14 einf] | [0, 1–0, 9] | [−1, 5] | 0.655 | 0.742 | 0.682 |
| AR linear | [1–8] | [1–6] | [−2, 14 einf] | [0, 1–0, 9] | [−1, 5] | 0.553 * | 0.706 | 0.599 |
| AR non-linear | [1–8] | [1–6] | [−2, 14 einf] | [0, 1–0, 9] | [−1, 5] | 0.749 | 0.809 * | 0.761 |
| Model | Lay | Sit | Stand | p-Values ANOVA |
|---|---|---|---|---|
| FIR ARI | 4.69 ± 2.51 | 3.6 ± 2.15 | 4.76 ± 2.23 | 0.2522 |
| NFIR ARI | 4.41 ± 1.94 | 4.12 ± 2.58 | 4.97 ± 2.03 | 0.3201 |
| ARX ARI | 4.41 ± 2.57 | 4.25 ± 1.72 | 4.42 ± 1.92 | 0.9683 |
| NARX ARI | 4.92 ± 2.31 | 3.84 ± 2.28 | 4.51 ± 2.77 | 0.6991 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chacón, M.; Rojas-Pescio, H.; Peñaloza, S.; Landerretche, J. Machine Learning Models and Statistical Complexity to Analyze the Effects of Posture on Cerebral Hemodynamics. Entropy 2022, 24, 428. https://doi.org/10.3390/e24030428
Chacón M, Rojas-Pescio H, Peñaloza S, Landerretche J. Machine Learning Models and Statistical Complexity to Analyze the Effects of Posture on Cerebral Hemodynamics. Entropy. 2022; 24(3):428. https://doi.org/10.3390/e24030428
Chicago/Turabian StyleChacón, Max, Hector Rojas-Pescio, Sergio Peñaloza, and Jean Landerretche. 2022. "Machine Learning Models and Statistical Complexity to Analyze the Effects of Posture on Cerebral Hemodynamics" Entropy 24, no. 3: 428. https://doi.org/10.3390/e24030428
APA StyleChacón, M., Rojas-Pescio, H., Peñaloza, S., & Landerretche, J. (2022). Machine Learning Models and Statistical Complexity to Analyze the Effects of Posture on Cerebral Hemodynamics. Entropy, 24(3), 428. https://doi.org/10.3390/e24030428






