fNIRS Complexity Analysis for the Assessment of Motor Imagery and Mental Arithmetic Tasks
Abstract
1. Introduction
2. Materials and Methods
2.1. Block Design
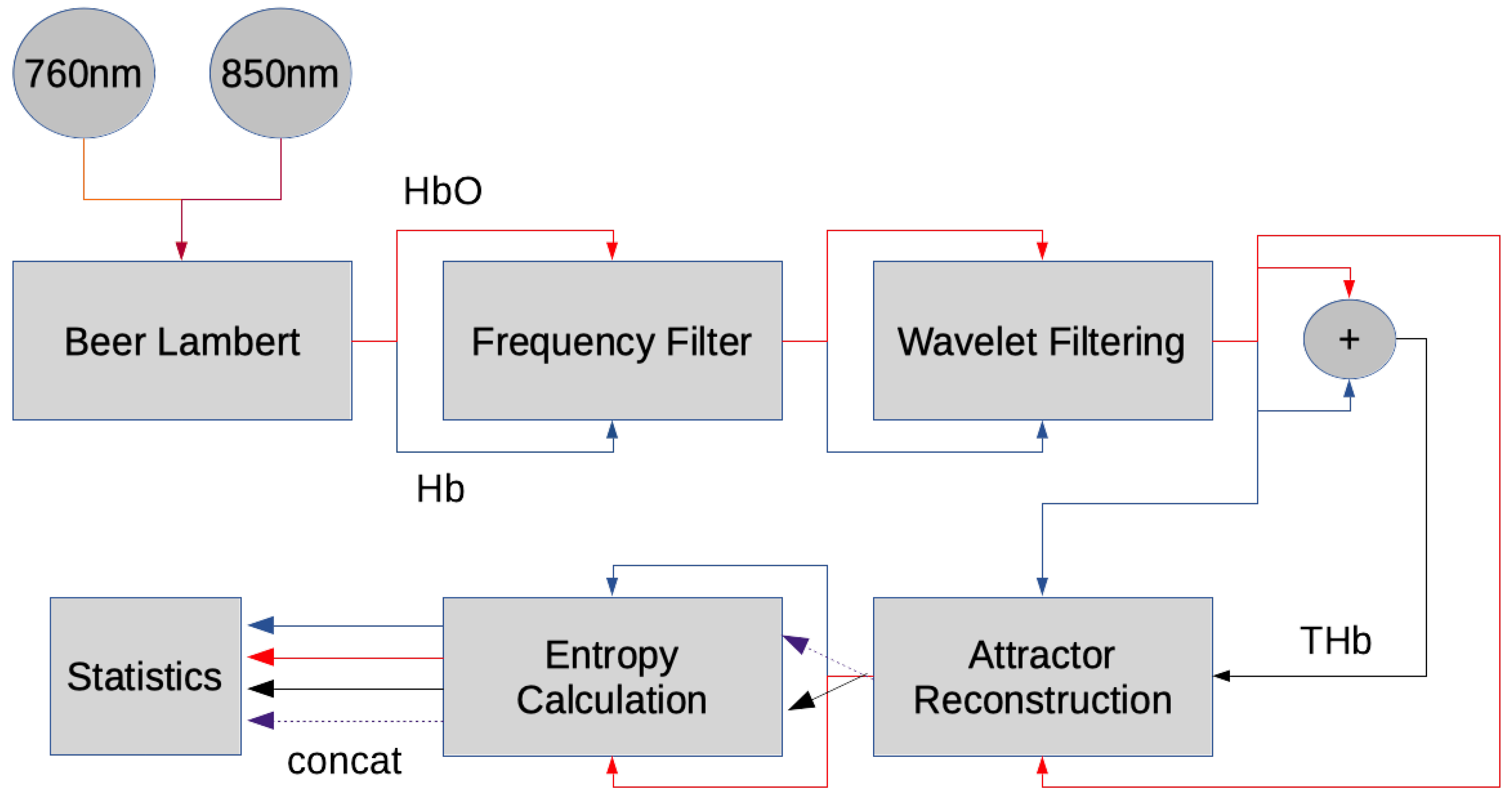
2.2. Hemoglobin Extraction from fNIRS Signals
2.3. fNIRS Data Preprocessing
2.4. Entropy Analysis
2.5. Statistical Analysis
3. Results
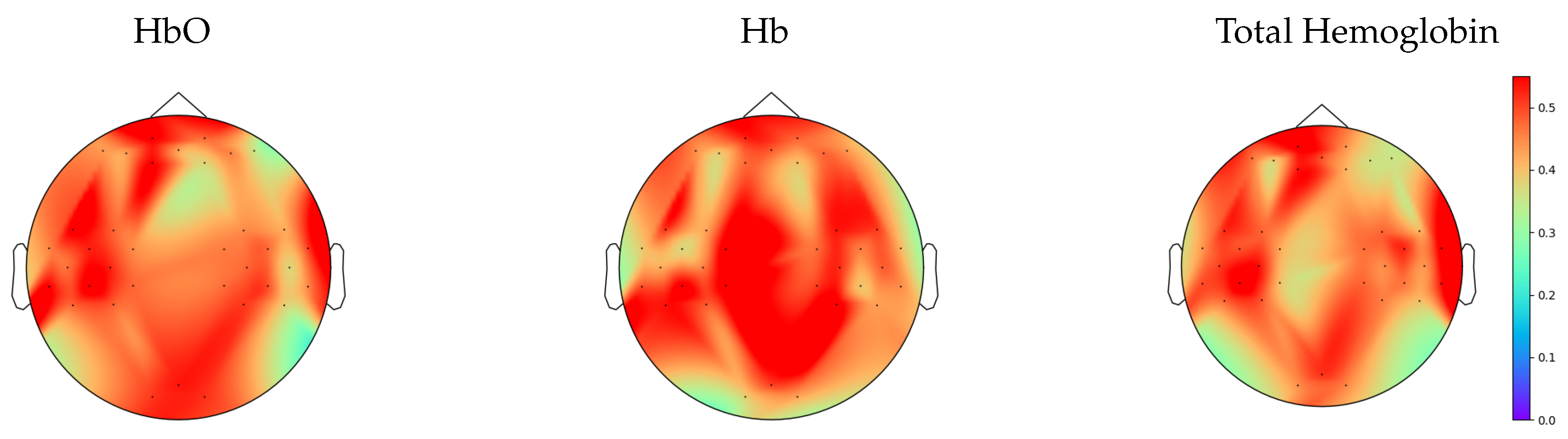
3.1. Nonlinearity Test
3.2. Analysis of Repetitions within Tasks
3.3. Between-Task Statistical Analysis
3.4. Multiple Comparison Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Strangman, G.; Culver, J.P.; Thompson, J.H.; Boas, D.A. A Quantitative Comparison of Simultaneous BOLD fMRI and NIRS Recordings during Functional Brain Activation. NeuroImage 2002, 17, 719–731. [Google Scholar] [CrossRef] [PubMed]
- Jobsis, F. Noninvasive, infrared monitoring of cerebral and myocardial oxygen sufficiency and circulatory parameters. Science 1977, 198, 1264–1267. [Google Scholar] [CrossRef] [PubMed]
- Naseer, N.; Hong, K.S. fNIRS-based brain-computer interfaces: A review. Front. Hum. Neurosci. 2015, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Goldberger, A.L.; Peng, C.K.; Lipsitz, L.A. What is physiologic complexity and how does it change with aging and disease? Neurobiol. Aging 2002, 23, 23–26. [Google Scholar] [CrossRef]
- Marmarelis, V.Z. Nonlinear Dynamic Modeling of Physiological Systems; John Wiley & Sons: Hoboken, NJ, USA, 2004; Volume 10. [Google Scholar]
- Sunagawa, K.; Kawada, T.; Nakahara, T. Dynamic nonlinear vago-sympathetic interaction in regulating heart rate. Heart Vessel. 1998, 13, 157–174. [Google Scholar] [CrossRef]
- Toyoda, H.; Kashikura, K.; Okada, T.; Nakashita, S.; Honda, M.; Yonekura, Y.; Kawaguchi, H.; Maki, A.; Sadato, N. Source of nonlinearity of the BOLD response revealed by simultaneous fMRI and NIRS. NeuroImage 2008, 39, 997–1013. [Google Scholar] [CrossRef]
- Banaji, M.; Tachtsidis, I.; Delpy, D.; Baigent, S. A physiological model of cerebral blood flow control. Math. Biosci. 2005, 194, 125–173. [Google Scholar] [CrossRef]
- Jeong, J. EEG dynamics in patients with Alzheimer’s disease. Clin. Neurophysiol. 2004, 115, 1490–1505. [Google Scholar] [CrossRef]
- Friston, K.J. Book Review: Brain Function, Nonlinear Coupling, and Neuronal Transients. Neuroscientist 2001, 7, 406–418. [Google Scholar] [CrossRef]
- Elbert, T.; Ray, W.J.; Kowalik, Z.J.; Skinner, J.E.; Graf, K.E.; Birbaumer, N. Chaos and physiology: Deterministic chaos in excitable cell assemblies. Physiol. Rev. 1994, 74, 1–47. [Google Scholar] [CrossRef]
- Sinaĭ, Y.G. On the Notion of Entropy of a Dynamical System. Dokl. Russ. Acad. Sci. 1959, 124, 768–771. [Google Scholar]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, H2039–H2049. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Wang, Z.; Xie, H.; Yu, W. Characterization of Surface EMG Signal Based on Fuzzy Entropy. IEEE Trans. Neural Syst. Rehabil. Eng. 2007, 15, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of biological signals. Phys. Rev. E 2005, 71, 021906. [Google Scholar] [CrossRef]
- Li, P.; Liu, C.; Li, K.; Zheng, D.; Liu, C.; Hou, Y. Assessing the complexity of short-term heartbeat interval series by distribution entropy. Med. Biol. Eng. Comput. 2015, 53, 77–87. [Google Scholar] [CrossRef]
- Bandt, C.; Keller, G.; Pompe, B. Entropy of interval maps via permutations. Nonlinearity 2002, 15, 1595–1602. [Google Scholar] [CrossRef]
- Bandt, C.; Shiha, F. Order Patterns in Time Series. J. Time Ser. Anal. 2007, 28, 646–665. [Google Scholar] [CrossRef]
- Gu, Y.; Miao, S.; Han, J.; Zeng, K.; Ouyang, G.; Yang, J.; Li, X. Complexity analysis of fNIRS signals in ADHD children during working memory task. Sci. Rep. 2017, 7, 829. [Google Scholar] [CrossRef]
- Jin, L.; Jia, H.; Li, H.; Yu, D. Differences in brain signal complexity between experts and novices when solving conceptual science problem: A functional near-infrared spectroscopy study. Neurosci. Lett. 2019, 699, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Perpetuini, D.; Chiarelli, A.M.; Cardone, D.; Filippini, C.; Bucco, R.; Zito, M.; Merla, A. Complexity of Frontal Cortex fNIRS Can Support Alzheimer Disease Diagnosis in Memory and Visuo-Spatial Tests. Entropy 2019, 21, 26. [Google Scholar] [CrossRef]
- Perpetuini, D.; Bucco, R.; Zito, M.; Merla, A. Study of memory deficit in Alzheimer’s disease by means of complexity analysis of fNIRS signal. Neurophotonics 2018, 5, 011010. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhu, Z.; Zhao, W.; Sun, Y.; Wen, D.; Xie, Y.; Liu, X.; Niu, H.; Han, Y. Decreased resting-state brain signal complexity in patients with mild cognitive impairment and Alzheimer’s disease: A multi-scale entropy analysis. Biomed. Opt. Express 2018, 9, 1916–1929. [Google Scholar] [CrossRef]
- Angsuwatanakul, T.; Iramina, K.; Kaewkamnerdpong, B. Brain complexity analysis of functional near infrared spectroscopy for working memory study. In Proceedings of the 2015 8th Biomedical Engineering International Conference (BMEiCON), Pattaya, Thailand, 25–27 November 2015; pp. 1–5. [Google Scholar] [CrossRef]
- Keshmiri, S.; Sumioka, H.; Yamazaki, R.; Ishiguro, H. Differential Entropy Preserves Variational Information of Near-Infrared Spectroscopy Time Series Associated With Working Memory. Front. Neuroinform. 2018, 12, 33. [Google Scholar] [CrossRef] [PubMed]
- Keshmiri, S.; Sumioka, H.; Okubo, M.; Ishiguro, H. An Information-Theoretic Approach to Quantitative Analysis of the Correspondence Between Skin Blood Flow and Functional Near-Infrared Spectroscopy Measurement in Prefrontal Cortex Activity. Front. Neurosci. 2019, 13, 79. [Google Scholar] [CrossRef]
- Shin, J.; von Lühmann, A.; Blankertz, B.; Kim, D.; Jeong, J.; Hwang, H.; Müller, K. Open Access Dataset for EEG+NIRS Single-Trial Classification. IEEE Trans. Neural Syst. Rehabil. Eng. 2017, 25, 1735–1745. [Google Scholar] [CrossRef] [PubMed]
- Villringer, A.; Chance, B. Non-invasive optical spectroscopy and imaging of human brain function. Trends Neurosci. 1997, 20, 435–442. [Google Scholar] [CrossRef]
- Pinti, P.; Scholkmann, F.; Hamilton, A.; Burgess, P.; Tachtsidis, I. Current Status and Issues Regarding Pre-processing of fNIRS Neuroimaging Data: An Investigation of Diverse Signal Filtering Methods Within a General Linear Model Framework. Front. Hum. Neurosci. 2019, 12, 505. [Google Scholar] [CrossRef]
- Strangman, G.; Boas, D.A.; Sutton, J.P. Non-invasive neuroimaging using near-infrared light. Biol. Psychiatry 2002, 52, 679–693. [Google Scholar] [CrossRef]
- Molavi, B.; Dumont, G.A. Wavelet-based motion artifact removal for functional near-infrared spectroscopy. Physiol. Meas. 2012, 33, 259–270. [Google Scholar] [CrossRef]
- Abarbanel, H.D.I.; Brown, R.; Sidorowich, J.J.; Tsimring, L.S. The analysis of observed chaotic data in physical systems. Rev. Mod. Phys. 1993, 65, 1331–1392. [Google Scholar] [CrossRef]
- Delgado-Bonal, A.; Marshak, A. Approximate Entropy and Sample Entropy: A Comprehensive Tutorial. Entropy 2019, 21, 541. [Google Scholar] [CrossRef]
- Lake, D.E.; Richman, J.S.; Griffin, M.P.; Moorman, J.R. Sample entropy analysis of neonatal heart rate variability. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002, 283, R789–R797. [Google Scholar] [CrossRef] [PubMed]
- Azami, H.; Escudero, J. Refined composite multivariate generalized multiscale fuzzy entropy: A tool for complexity analysis of multichannel signals. Phys. A Stat. Mech. Its Appl. 2017, 465, 261–276. [Google Scholar] [CrossRef]
- Scott, D.W. On optimal and data-based histograms. Biometrika 1979, 66, 605–610. [Google Scholar] [CrossRef]
- Barnett, A.G.; Wolff, R.C. A time-domain test for some types of nonlinearity. IEEE Trans. Signal Process. 2005, 53, 26–33. [Google Scholar] [CrossRef]
- Khoa, T.Q.D.; Thang, H.M.; Nakagawa, M. Testing for nonlinearity in functional near-infrared spectroscopy of brain activities by surrogate data methods. J. Physiol. Sci. JPS 2008, 58, 47–52. [Google Scholar] [CrossRef]
- Amalric, M.; Dehaene, S. Origins of the brain networks for advanced mathematics in expert mathematicians. Proc. Natl. Acad. Sci. USA 2016, 113, 4909–4917. [Google Scholar] [CrossRef]
- Soltanlou, M.; Sitnikova, M.A.; Nuerk, H.C.; Dresler, T. Applications of Functional Near-Infrared Spectroscopy (fNIRS) in Studying Cognitive Development: The Case of Mathematics and Language. Front. Psychol. 2018, 9, 277. [Google Scholar] [CrossRef]
- Fazli, S.; Mehnert, J.; Steinbrink, J.; Curio, G.; Villringer, A.; Müller, K.R.; Blankertz, B. Enhanced performance by a hybrid NIRS-EEG brain computer interface. NeuroImage 2012, 59, 519–529. [Google Scholar] [CrossRef]
- Agrell, B.; Dehlin, O. The clock-drawing test. Age Ageing 1998, 27, 399–403. [Google Scholar] [CrossRef]
- Nardelli, M.; Greco, A.; Danzi, O.P.; Perlini, C.; Tedeschi, F.; Scilingo, E.P.; Del Piccolo, L.; Valenza, G. Cardiovascular assessment of supportive doctor-patient communication using multi-scale and multi-lag analysis of heartbeat dynamics. Med. Biol. Eng. Comput. 2019, 57, 123–134. [Google Scholar] [CrossRef]
- Li, X.; Song, D.; Zhang, P.; Zhang, Y.; Hou, Y.; Hu, B. Exploring EEG Features in Cross-Subject Emotion Recognition. Front. Neurosci. 2018, 12, 162. [Google Scholar] [CrossRef]
- Hu, X.; Yu, J.; Song, M.; Yu, C.; Wang, F.; Sun, P.; Wang, D.; Zhang, D. EEG Correlates of Ten Positive Emotions. Front. Hum. Neurosci. 2017, 11, 26. [Google Scholar] [CrossRef] [PubMed]
- Buckner, R.L. Event-related fMRI and the hemodynamic response. Hum. Brain Mapp. 1998, 6, 373–377. [Google Scholar] [CrossRef]
- Zhang, Y.; Brooks, D.H.; Franceschini, M.A.; Boas, D.A. Eigenvector-based spatial filtering for reduction of physiological interference in diffuse optical imaging. J. Biomed. Opt. 2005, 10, 011014. [Google Scholar] [CrossRef] [PubMed]

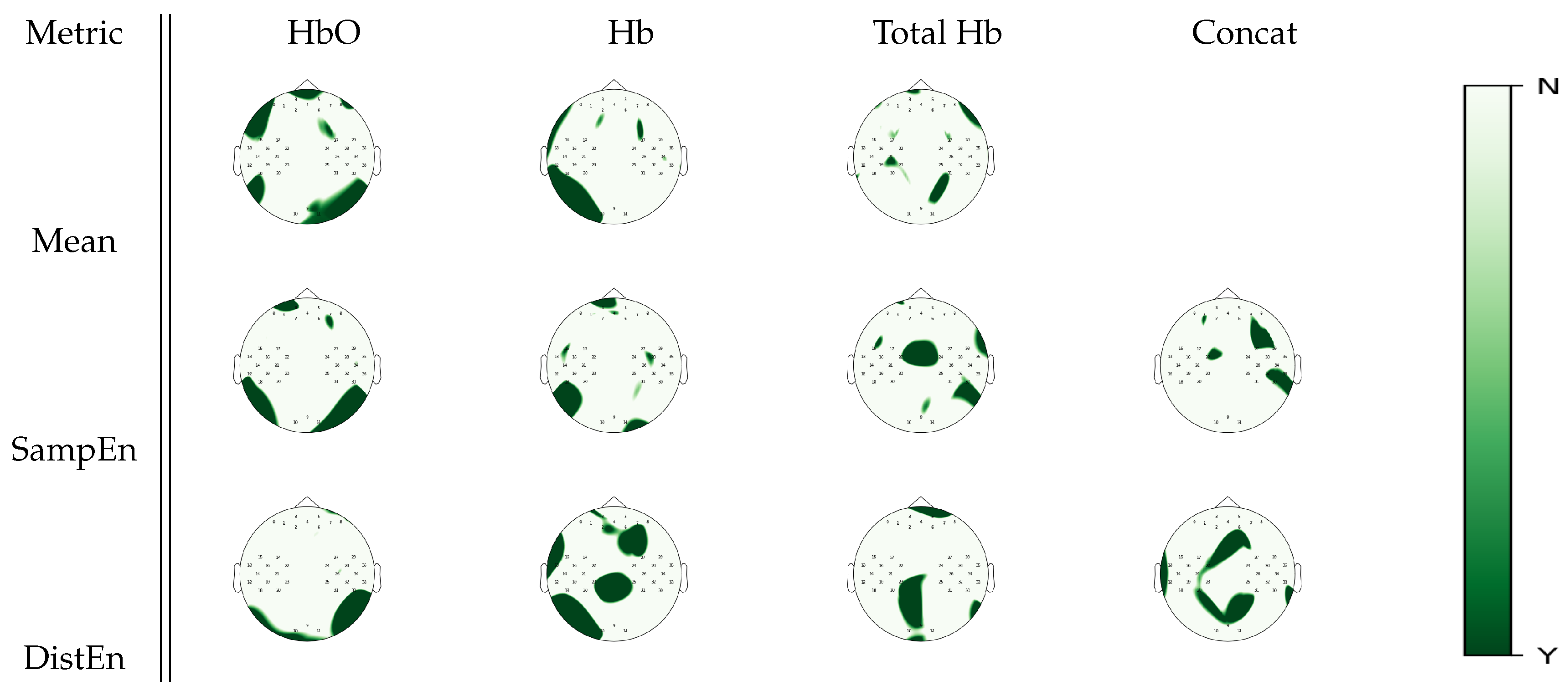
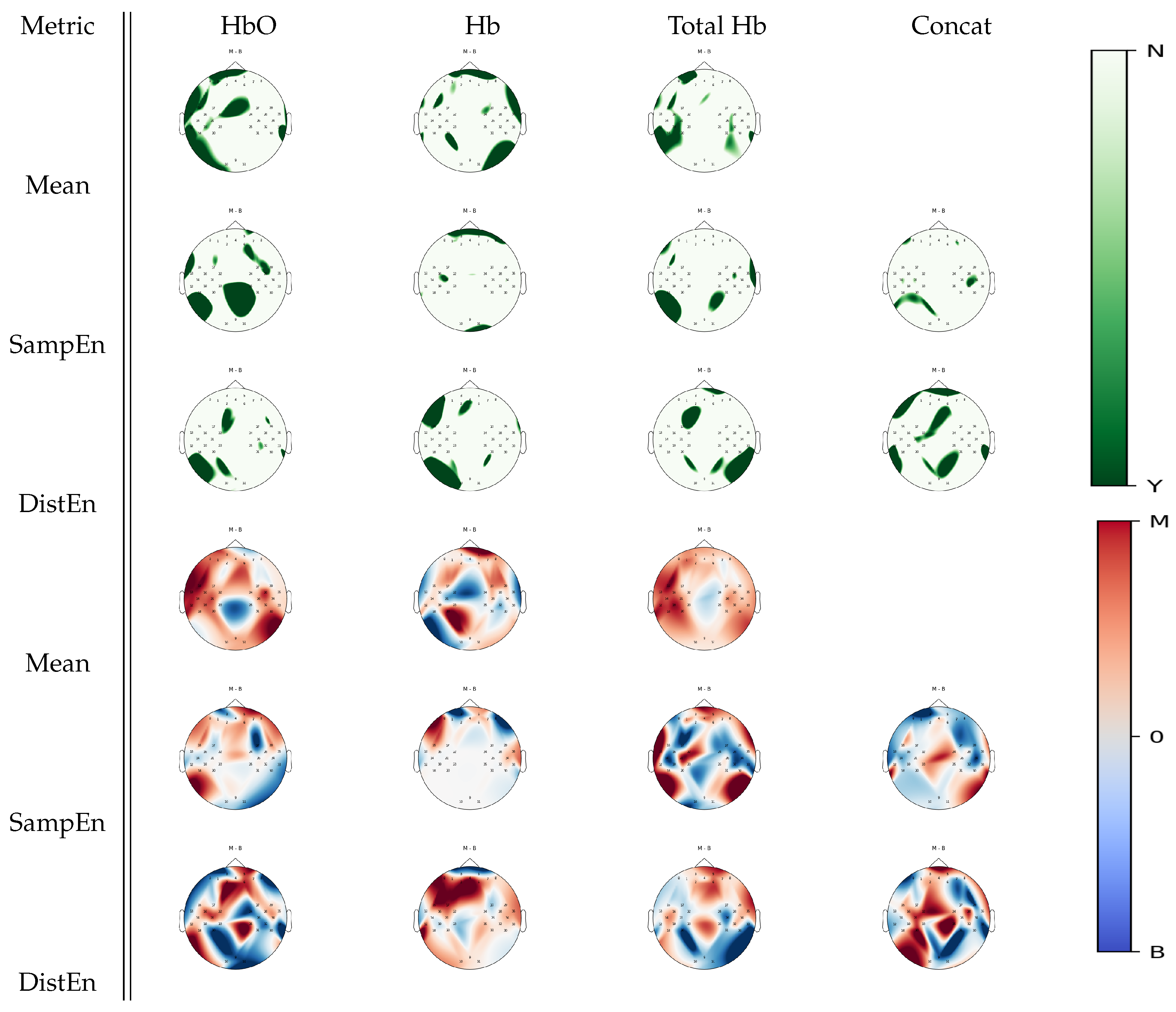

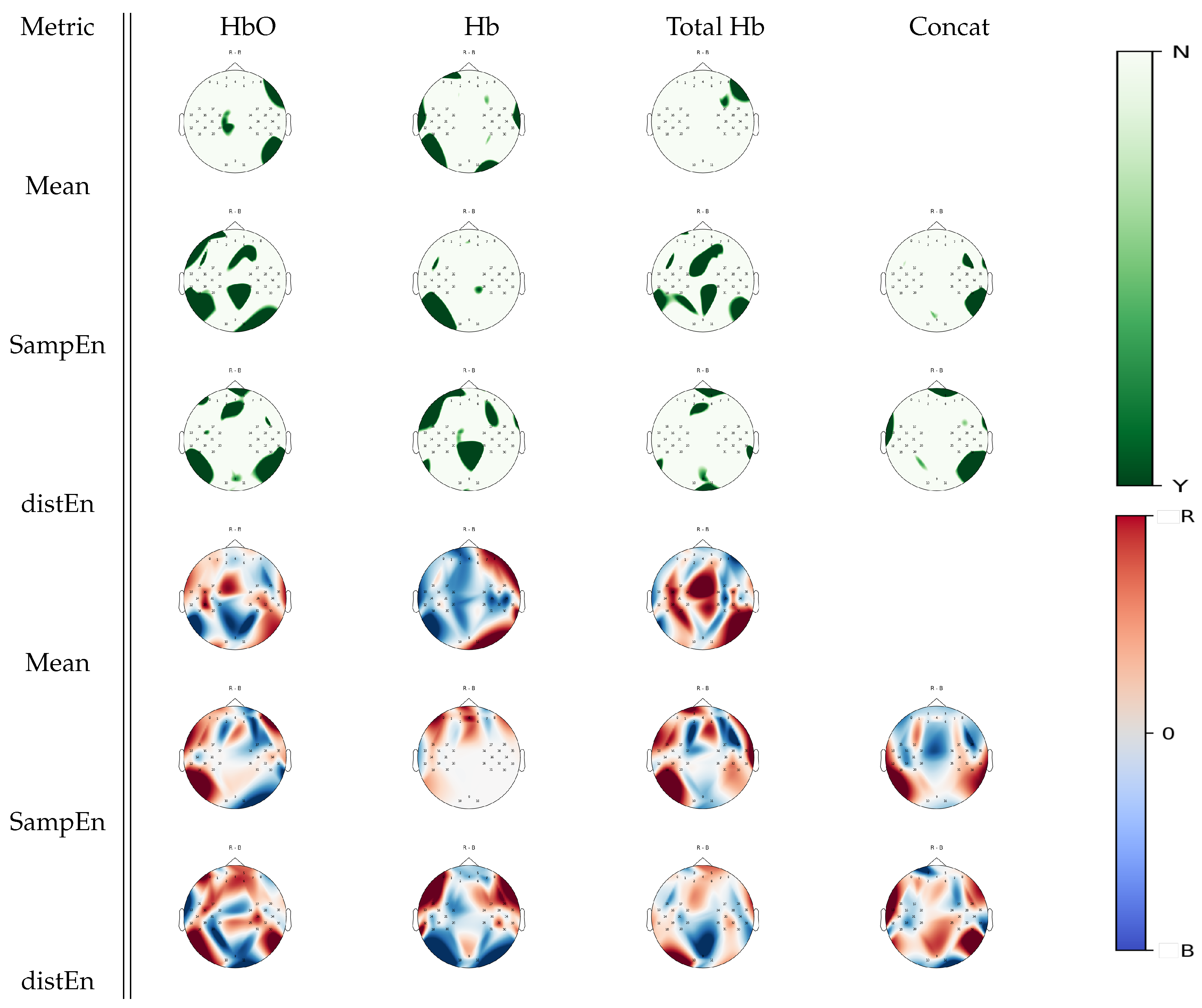
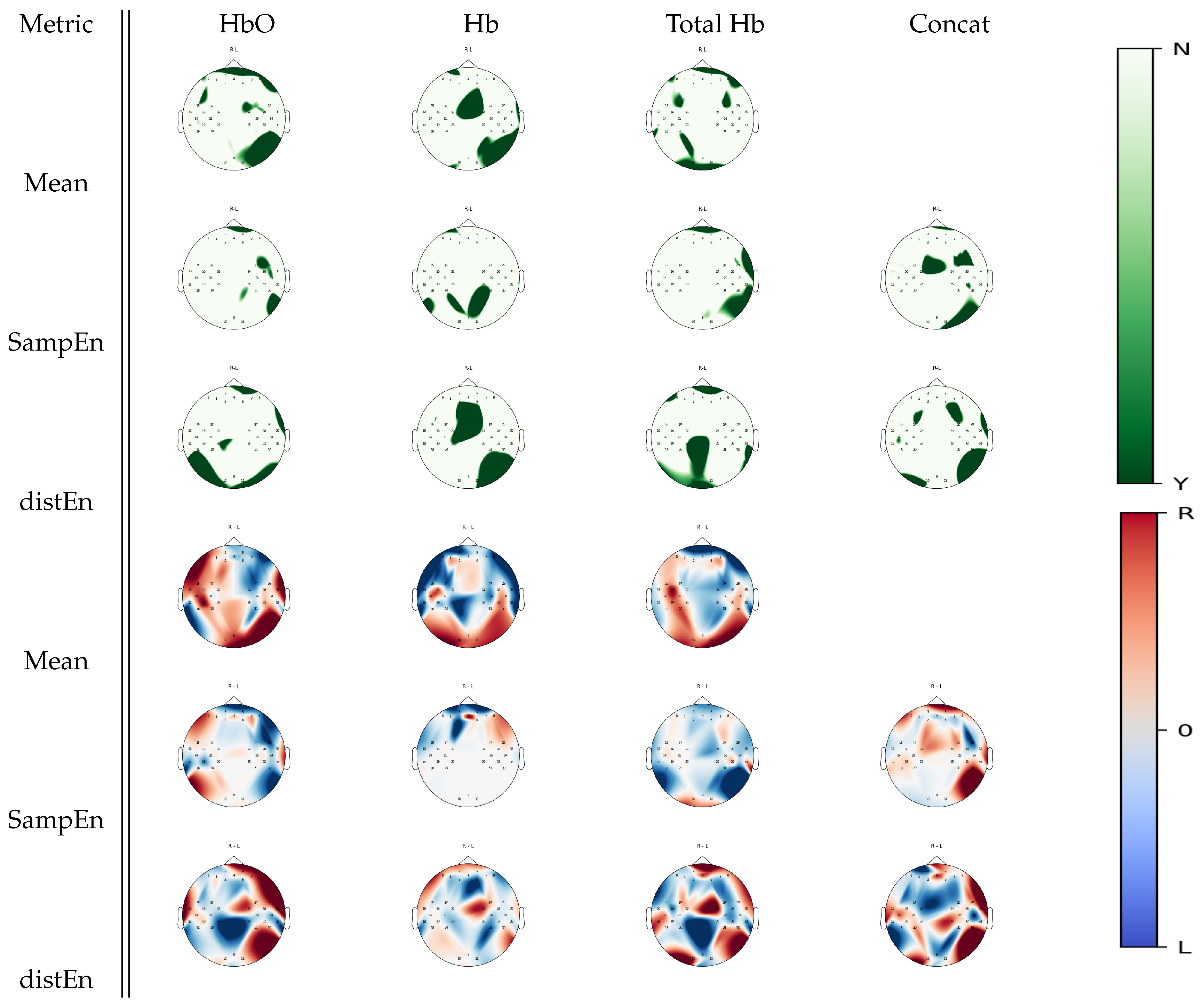
| Metric | Mental Arithmetic | Left Hand Imagery | Right Hand Imagery | Baseline |
|---|---|---|---|---|
| HbO | 0.1735 | 0.1147 | 0.0331 | 0.7383 |
| Hb | 0.0870 | 0.0841 | 0.1735 | 0.0039 |
| Total Hb | 0.0331 | 0.2449 | 0.0965 | 0.0501 |
| 0.0610 | 0.1414 | 0.0976 | 0.1375 | |
| 0.0891 | 0.2844 | 0.0101 | 0.0262 | |
| 0.2013 | 0.2528 | 0.0501 | 0.0554 | |
| 0.0408 | 0.1147 | 0.0106 | 0.1735 | |
| 0.0934 | 0.0023 | 0.0501 | 0.0219 | |
| 0.0145 | 0.0106 | 0.0556 | 0.0408 | |
| 0.0708 | 0.0051 | 0.0243 | 0.0243 | |
| 0.0219 | 0.0078 | 0.1735 | 0.0078 | |
| 0.6658 | 0.1735 | 0.1147 | 0.1619 | |
| 0.1272 | 0.0115 | 0.0709 | 0.2209 | |
| 0.0871 | 0.0501 | 0.1411 | 0.0874 | |
| 0.0408 | 0.1619 | 0.0118 | 0.0408 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghouse, A.; Nardelli, M.; Valenza, G. fNIRS Complexity Analysis for the Assessment of Motor Imagery and Mental Arithmetic Tasks. Entropy 2020, 22, 761. https://doi.org/10.3390/e22070761
Ghouse A, Nardelli M, Valenza G. fNIRS Complexity Analysis for the Assessment of Motor Imagery and Mental Arithmetic Tasks. Entropy. 2020; 22(7):761. https://doi.org/10.3390/e22070761
Chicago/Turabian StyleGhouse, Ameer, Mimma Nardelli, and Gaetano Valenza. 2020. "fNIRS Complexity Analysis for the Assessment of Motor Imagery and Mental Arithmetic Tasks" Entropy 22, no. 7: 761. https://doi.org/10.3390/e22070761
APA StyleGhouse, A., Nardelli, M., & Valenza, G. (2020). fNIRS Complexity Analysis for the Assessment of Motor Imagery and Mental Arithmetic Tasks. Entropy, 22(7), 761. https://doi.org/10.3390/e22070761






