EEG Characterization of the Alzheimer’s Disease Continuum by Means of Multiscale Entropies
Abstract
1. Introduction
2. Materials
2.1. Subjects
2.2. EEG Recording
3. Methods
3.1. Multiscale Sample Entropy
- Firstly, given a one-dimensional discrete time series, [], successive coarse-grained time series are built according to the scale factor . This process is performed by calculating the average value of the data points from non-overlapping windows of length [20,24]:It is worth noting that the number of samples for each subsequent coarse-grained series is reduced by a factor of , hence resulting in a sequence equal to the original time series.
- Afterwards, SampEn is calculated for each . SampEn algorithm is detailed in the contribution of Richman and Moorman [12]. SampEn is a non-linear measure that allows assessing the degree of irregularity of a signal [12], developed as an improvement of ApEn [11,12]. SampEn accounts as an embedding entropy, the calculation of which is based on the similarity with a delayed version of the time series itself [32]. Given m, a positive integer, and r, a positive real number, SampEn is defined as the negative logarithm of the conditional probability that two sequences of length m from the time series are similar within a r threshold at the next point, excluding self matches [12]. Setting tolerance as r times the standard deviation of the original time series grants robustness to variations, such as magnification, reduction or shift by a constant [20,24]. Setting the values of r and m is crucial in the performance of SampEn. However, there are no absolute guidelines to optimize these variables [24]. It has been found that excessively low r values may cause the calculation to fail, while excessively high values may introduce some bias [24,33]. Previous SampEn studies obtained good statistical reproducibility setting m = 1 and r = 0.25 for sequences larger than 100 points [24,33]. For this reason, the maximum scale factor was established to = 25, implying a minimum length of 100 samples for any coarse-grained time series.
3.2. Refined Multiscale Spectral Entropy
- Given a one-dimensional discrete time series, [], a coarse-grained process is applied using the method described in Equation (1).
- The PSD of the coarse-grained time series is calculated. The PSD consists of the distribution of power into frequency components. In this work, the PSD was obtained by calculating the fast Fourier transform (FFT) of the autocorrelation function of the EEG data. For each scale factor , the normalized PSD (nPSD) of the coarse-grained time series is computed as , where , being the length of the power spectrum of . The value of for the rMSSE study was established to 25 to maintain consistency with the MSE analysis. The nPSD is evaluated as follows:
- Then, the normalized rMSSE is calculated as the Shannon entropy of the PSD:The resulting rMSSE values are within the range between 0 and 1, assuming the lowest value according to a signal with a unique frequency component (i.e., a sinusoidal signal), and the highest value corresponding to a signal with an equally distributed power in all frequencies (i.e., white noise).
3.3. Statistical and Classification Analyses
4. Results
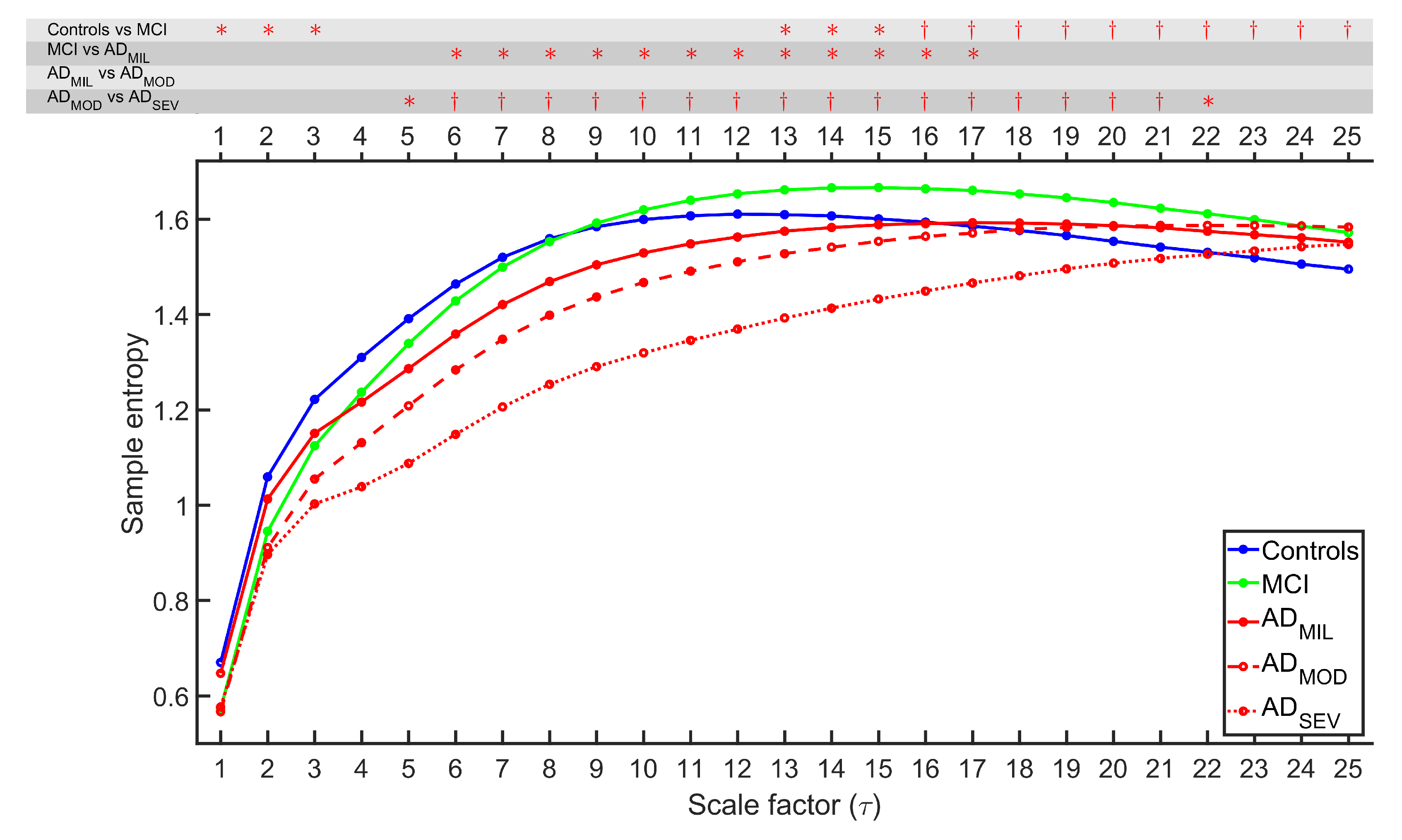
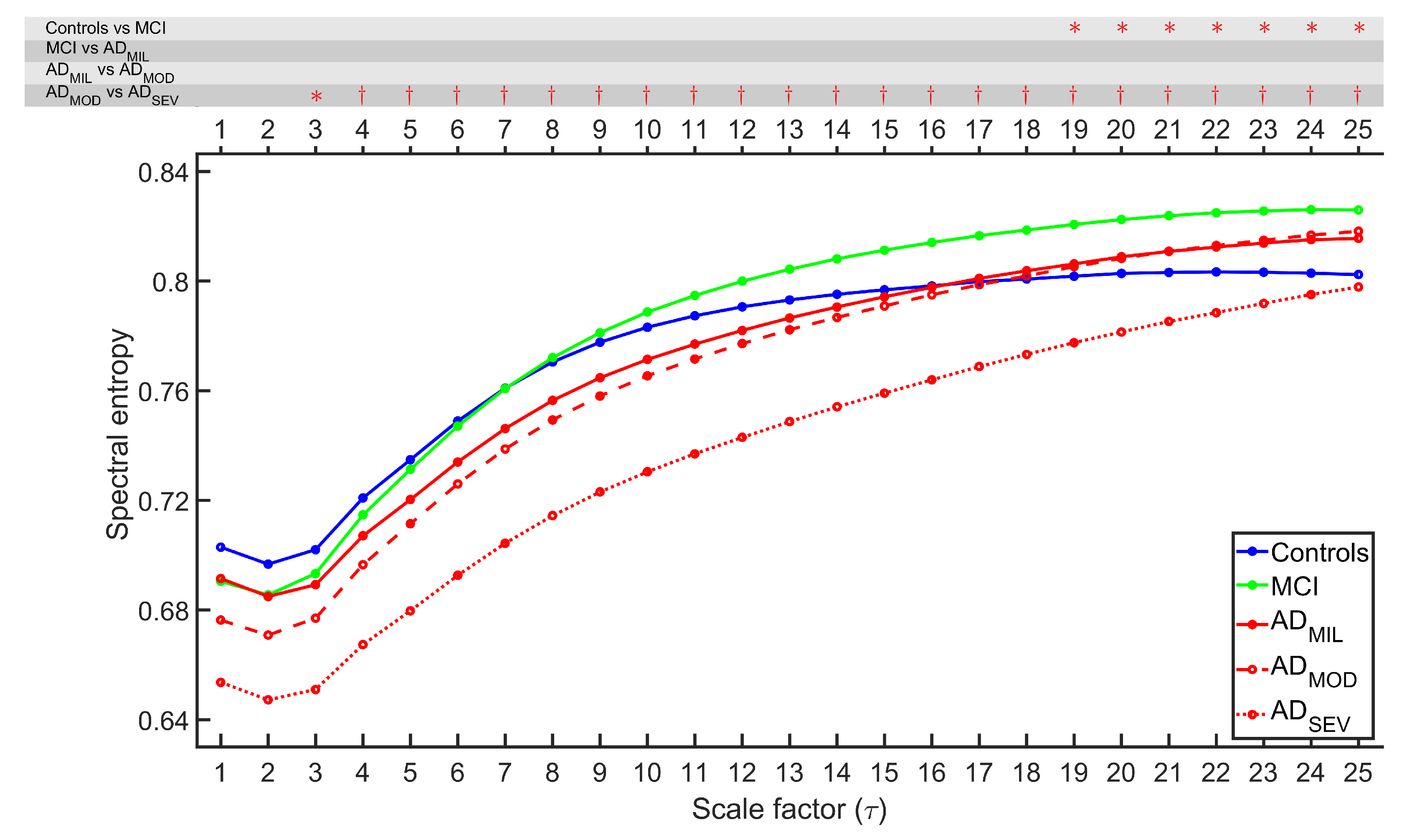
4.1. Irregularity Analyses
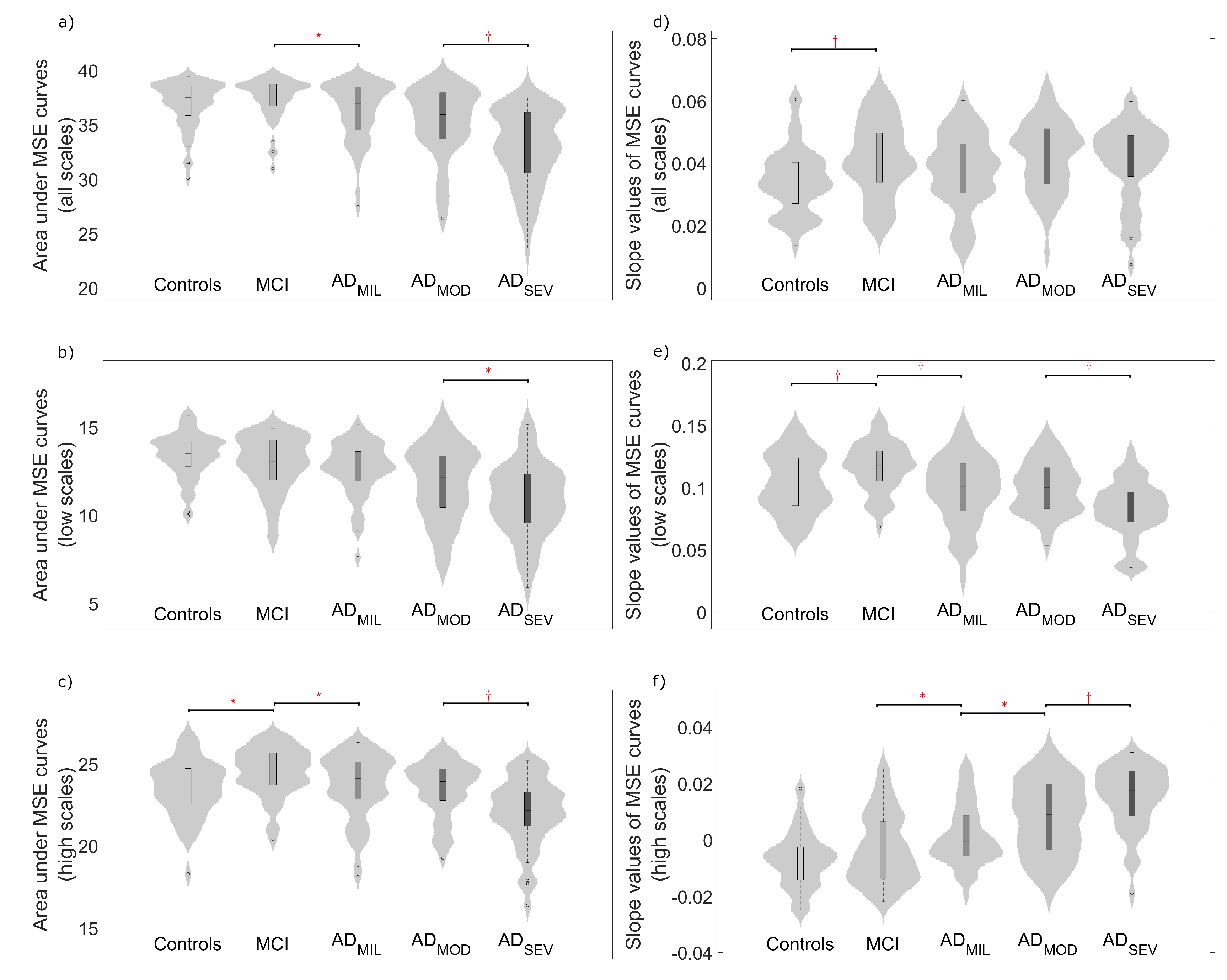
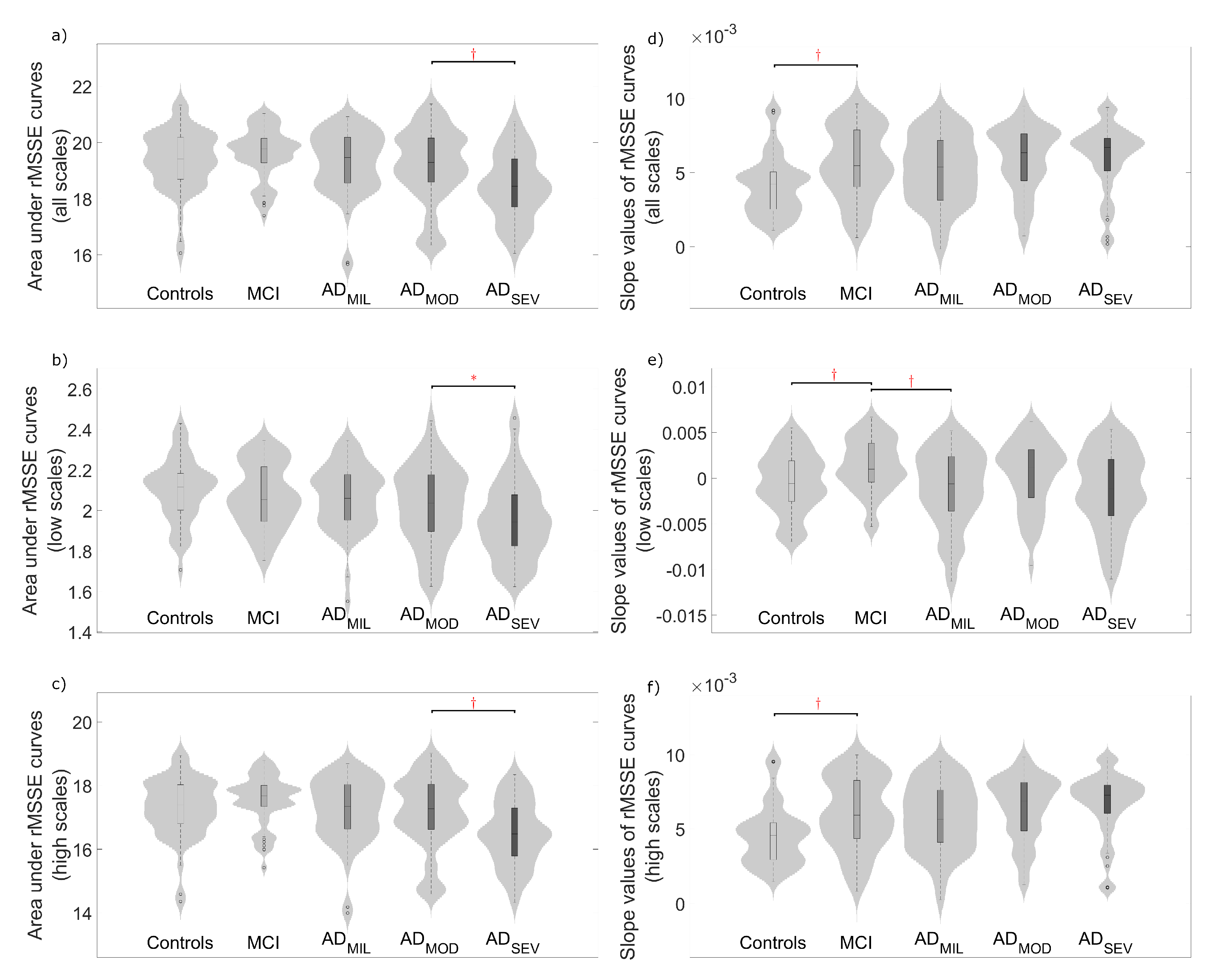
4.2. Complexity Analyses
4.3. Classification Analysis
5. Discussion
5.1. Multiscale Entropies for Irregularity Estimation
5.2. Multiscale Entropies for Complexity Estimation
5.3. Multiscale Entropies for Classification Purposes
5.4. Limitations
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AD | Alzheimer’s Disease |
| ADMIL | Mild Alzheimer’s Disease |
| ADMOD | Moderate Alzheimer’s Disease |
| ADSEV | Severe Alzheimer’s Disease |
| ApEn | Approximate Entropy |
| CV | Cross Validation |
| EEG | Electroencephalogram |
| FDR | False Discovery Rate |
| FFT | Fast Fourier Transform |
| FuzEn | Fuzzy Entropy |
| ICA | Independent Component Analysis |
| LDA | Linear Discriminant Analysis |
| LOO | Leave-One-Out |
| MCI | Mild Cognitive Impairment |
| MMSE | Minimental State Examination |
| MSE | Multiscale Sample Entropy |
| MSSE | Multiscale Spectral Entropy |
| NIA-AA | National Institute on Aging and Alzheimer’s Association |
| nPSD | Normalized Power Spectral Density |
| PermEn | Permutation Entropy |
| PSD | Power Spectral Density |
| QDA | Quadratic Discriminant Analysis |
| rMSSE | Refined Multiscale Spectral Entropy |
| SampEn | Sample Entropy |
| SD | Standard Deviation |
| SMR | Stepwise Multilinear Regression |
| SpecEn | Spectral Entropy |
References
- Alzheimer’s Association. 2018 Alzheimer’s disease facts and figures. Alzheimers Dement. 2018, 14, 367–429. [Google Scholar] [CrossRef]
- Alzheimer’s Disease International: World Alzheimer Report. 2018. Available online: https://www.alz.co.uk/research/world-report-2018 (accessed on 4 April 2019).
- Klimova, B.; Kuca, K.; Maresova, P. Global View on Alzheimer’s Disease and Diabetes Mellitus: Threats, Risks and Treatment Alzheimer’s Disease and Diabetes Mellitus. Curr. Alzheimer Res. 2018, 15, 1277–1282. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.C. Mild cognitive impairment as a diagnostic entity. J. Intern. Med. 2004, 256, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Reisberg, B. Dementia: A systematic approach to identifying reversible causes. Geriatrics 1986, 41, 30–46. [Google Scholar] [PubMed]
- Pasquier, F. Early diagnosis of dementia: Neuropsychology. J. Neurol. 1999, 246, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Jack, C.R., Jr.; Benett, D.A.; Blennow, K.; Carrillo, M.C.; Dunn, B.; Budd, S.; Holtzman, D.M.; Jagust, W.; Jessen, F.; Karlawish, J.; et al. NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement. 2018, 14, 535–562. [Google Scholar] [CrossRef]
- Daulatzai, M.A. Early stages of pathogenesis in memory impairment during normal senescence and Alzheimer’s disease. J. Alzheimers Dis. 2010, 20, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Miskovic, V.; MacDonald, K.J.; Rhodes, L.J.; Cote, K.A. Changes in EEG multiscale entropy and power-law frequency scaling during the human sleep cycle. Hum. Brain Mapp. 2018, 40, 538–551. [Google Scholar] [CrossRef]
- Powell, E.; Percival, I.C. A spectral entropy method for distinguishing regular and irregular motion of Hamiltonian systems. J. Phys. A: Math. Gen. 1979, 12, 2053. [Google Scholar] [CrossRef]
- Pincus, S. Approximate entropy as a measure of system complexity. Proc. Natl. Acad. Sci. USA 1991, 88, 2297–2301. [Google Scholar] [CrossRef]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am. J. Physiol. Heart. Circ. Physiol. 2000, 278, H2039–H2049. [Google Scholar] [CrossRef]
- Kosko, B. Fuzzy Entropy and Conditioning. Inf. Sci. 1986, 40, 165–174. [Google Scholar] [CrossRef]
- Bandt, C.; Pompe, B. Permutation entropy: A natural complexity measure for time series. Phys. Rev. Lett. 2002, 88, 174102. [Google Scholar] [CrossRef]
- Gómez, C.; Poza, J.; Gomez-Pilar, J.; Bachiller, A.; Juan-Cruz, C.; Tola-Arribas, M.A.; Carreres, A.; Cano, M.; Hornero, R. Analysis of Spontaneous EEG Activity in Alzheimer’s Disease Using Cross-Sample Entropy and Graph Theory. In Proceedings of the The 38th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Lake Buena Vista (Orlando), FL, USA, 17–20 August 2016; pp. 2830–2833. [Google Scholar] [CrossRef]
- Abásolo, D.; Hornero, R.; Espino, P. Approximate entropy of EEG background activity in Alzheimer’s disease patients. Intell. Autom. Soft. Co. 2009, 15, 591–603. [Google Scholar] [CrossRef]
- Abásolo, D.; Hornero, R.; Espino, P.; Alvarez, D.; Poza, J. Entropy analysis of the EEG background activity in Alzheimer’s disease patients. Physiol. Meas. 2006, 27, 241–253. [Google Scholar] [CrossRef] [PubMed]
- Hornero, R.; Abásolo, D.; Escudero, J.; Gómez, C. Nonlinear analysis of electroencephalogram and magnetoencephalogram recordings in patients with Alzheimer’s disease. Phil. Trans. Math. Phys. Eng. Sci. 2008, 367, 317–336. [Google Scholar] [CrossRef]
- Jeong, J. EEG dynamics in patients with Alzheimer’s disease. Clin. Neurophysiol. 2004, 115, 1490–1505. [Google Scholar] [CrossRef]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of biological signals. Phys. Rev. E 2005, 71, 021906. [Google Scholar] [CrossRef]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale Entropy Analysis of Complex Physiologic Time Series. Phys. Rev. Lett. 2002, 89, 068102. [Google Scholar] [CrossRef]
- Goldberger, A.L. Non-linear dynamics for clinicians: Chaos theory, fractals, and complexity at the bedside. Lancet 1996, 347, 1312–1314. [Google Scholar] [CrossRef]
- Fan, M.; Yang, A.C.; Fuh, J.L.; Chou, C.A. Topological Pattern Recognition of Severe Alzheimer’s Disease Via Regularized Supervised Learning of EEG Complexity. Front. Neurosci. 2018, 12, 685. [Google Scholar] [CrossRef]
- Escudero, J.; Abásolo, D.; Hornero, R.; Espino, P.; López, M. Analysis of electroencephalograms in Alzheimer’s disease patients with multiscale entropy. Physiol. Meas. 2006, 27, 1091–1106. [Google Scholar] [CrossRef] [PubMed]
- Stam, C.J.; Van Der Made, Y.; Pijnenburg, Y.A.L.; Scheltens, P.H. EEG synchronization in mild cognitive impairment and Alzheimer’s disease. Acta Neurol. Scand. 2003, 108, 90–96. [Google Scholar] [CrossRef]
- Stam, C.J.; Montez, T.; Jones, B.F.; Rombouts, S.A.R.B.; Van Der Made, Y.; Pijnenburg, Y.A.L.; Scheltens, P. Disturbed fluctuations of resting state EEG synchronization in Alzheimer’s disease. Clin. Neurophysiol. 2005, 116, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Poza, J.; Hornero, R.; Escudero, J.; Fernández, A.; Sánchez, C.I. Regional Analysis of Spontaneous MEG Rhythms in Patients with Alzheimer’s Disease Using Spectral Entropies. Ann. Biomed. Eng. 2007, 36, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Humeau-Heurtier, A.; Wu, C.W.; Wu, S.D.; Mahé, G.; Abraham, P. Refined Multiscale Hilbert-Huang Spectral Entropy and its Application to Central and Peripheral Cardiovascular Data. IEEE Trans. Biomed. Eng. 2016, 63, 2405–2415. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Núñez, P.; Poza, J.; Bachiller, A.; Gomez-Pilar, J.; Lubeiro, A.; Molina, V.; Hornero, R. Exploring non-stationarity patterns in schizophrenia: Neural reorganization abnormalities in the alpha band. J. Neural Eng. 2017, 14, 046001. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Gómez, S.J.; Gómez, C.; Poza, J.; Martínez-Zarzuela, M.; Tola-Arribas, M.Á.; Cano, M.; Hornero, R. Measuring Alterations of Spontaneous EEG Neural Coupling in Alzheimer’s Disease and Mild Cognitive Impairment by Means of Cross-Entropy Metrics. Front. Neuroinform. 2018, 12, 76. [Google Scholar] [CrossRef]
- Sleigh, J.W.; Steyn-Ross, D.A.; Steyn-Ross, M.L.; Grant, C.; Ludbrook, G. Cortical entropy changes with general anaesthesia: Theory and experiment. Physiol. Meas. 2004, 25, 921–934. [Google Scholar] [CrossRef]
- Lake, D.E.; Richman, J.S.; Griffin, M.P.; Moorman, J.R. Sample entropy analysis of neonatal heart rate variability. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002, 283, R789–R797. [Google Scholar] [CrossRef]
- McIntosh, A.R.; Kovacevic, N.; Lippe, S.; Garrett, D.; Grady, C.; Jirsa, V. The development of a noisy brain. Arch. Ital. Biol. 2010, 148, 323–337. [Google Scholar] [CrossRef] [PubMed]
- Weng, W.C.; Jiang, G.J.; Chang, C.F.; Lu, W.Y.; Lin, C.Y.; Lee, W.T.; Shieh, J.S. Complexity of multi-channel electroencephalogram signal analysis in childhood absence epilepsy. PLoS ONE 2015, 10, e0134083. [Google Scholar] [CrossRef] [PubMed]
- Valencia, J.F.; Porta, A.; Vallverdu, M.; Claria, F.; Baranowski, R.; Orlowska-Baranowska, E.; Caminal, P. Refined multiscale entropy: Application to 24-h Holter recordings of heart period variability in healthy and aortic stenosis subjects. IEEE Trans. Biomed. Eng. 2009, 56, 2202–2213. [Google Scholar] [CrossRef] [PubMed]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Series B Stat. Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Dominguez, L.G. On the risk of extracting relevant information from random data. J. Neural. Eng. 2009, 6, 058001. [Google Scholar] [CrossRef] [PubMed]
- Bishop, C.M. Pattern recognition and machine learning. J. Electron. Imaging 2007, 16, 049901. [Google Scholar] [CrossRef]
- Simon, R.; Radmacher, M.D.; Dobbin, K.; McShane, L.M. Pitfalls in the use of DNA microarray data for diagnostic and prognostic classification. J. Natl. Cancer Inst. 2003, 95, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Simons, S.; Abásolo, D.; Escudero, J. Classification of Alzheimer’s disease from quadratic sample entropy of electroencephalogram. Healthc. Technol. Lett. 2015, 2, 70–73. [Google Scholar] [CrossRef]
- Jelles, B.; Van Birgelen, J.H.; Slaets, J.P.J.; Hekster, R.E.M.; Jonkman, E.J.; Stam, C.J. Decrease of non-linear structure in the EEG of Alzheimer patients compared to healthy controls. Clin. Neurophysiol. 1999, 110, 1159–1167. [Google Scholar] [CrossRef]
- Tononi, G.; Edelman, G.M.; Sporns, O. Complexity and coherency: Integrating information in the brain. Trends Cogn. Sci. 1998, 2, 474–484. [Google Scholar] [CrossRef]
- Mizuno, T. Assessment of EEG dynamical complexity in Alzheimer’s disease using multiscale entropy. Clin. Neurophysiol. 2010, 121, 1438–1446. [Google Scholar] [CrossRef] [PubMed]
- Azami, H.; Escudero, J. Coarse-graining approaches in univariate multiscale sample and dispersion entropy. Entropy 2018, 20, 138. [Google Scholar] [CrossRef]
- Chai, X.; Weng, X.; Zhang, Z.; Lu, Y.; Liu, G.; Niu, H. Quantitative EEG in Mild Cognitive Impairment and Alzheimer’s Disease by AR-Spectral and Multi-scale Entropy Analysis. In World Congress on Medical Physics and Biomedical Engineering; Springer: Singapore, 2018; pp. 159–163. [Google Scholar]
- Labate, D.; La Foresta, F.; Morabito, G.; Palamara, I.; Morabito, F.C. Entropic measures of EEG complexity in Alzheimer’s disease through a multivariate multiscale approach. IEEE Sens. J. 2013, 13, 3284–3292. [Google Scholar] [CrossRef]
- Becker, J.T.; Mintun, M.A.; Aleva, K.; Wiseman, M.B.; Nichols, T.; DeKosky, S.T. Compensatory reallocation of brain resources supporting verbal episodic memory in Alzheimer’s disease. Neurology 1996, 46, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Vina, J.; Lloret, A. Why women have more Alzheimer’s disease than men: Gender and mitochondrial toxicity of amyloid-β peptide. J. Alzheimers Dis. 2010, 20, S527–S533. [Google Scholar] [CrossRef]
| Group | N | Age (Mean ± SD) (Years) | Gender (Female:Male) | MMSE Score (Mean ± SD) |
|---|---|---|---|---|
| Controls | 51 | 80.14 ± 7.09 | 25:26 | 28.82 ± 1.13 |
| MCI subjects | 51 | 85.53 ± 7.25 | 36:15 | 23.33 ± 2.84 |
| ADMIL patients | 51 | 80.69 ± 7.05 | 30:21 | 22.49 ± 2.27 |
| ADMOD patients | 50 | 81.30 ± 8.04 | 43:7 | 13.60 ± 2.76 |
| ADSEV patients | 50 | 79.98 ± 7.82 | 43:7 | 2.42 ± 3.70 |
| Est. Class | Controls | MCI Subjects | ADMIL Patients | ADMOD Patients | ADSEV Patients | |
|---|---|---|---|---|---|---|
| True Class | ||||||
| Controls | 22 | 16 | 6 | 4 | 3 | |
| MCI subjects | 14 | 27 | 2 | 7 | 1 | |
| ADMIL patients | 12 | 14 | 4 | 10 | 11 | |
| ADMOD patients | 4 | 13 | 3 | 15 | 15 | |
| ADSEV patients | 4 | 1 | 1 | 10 | 34 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maturana-Candelas, A.; Gómez, C.; Poza, J.; Pinto, N.; Hornero, R. EEG Characterization of the Alzheimer’s Disease Continuum by Means of Multiscale Entropies. Entropy 2019, 21, 544. https://doi.org/10.3390/e21060544
Maturana-Candelas A, Gómez C, Poza J, Pinto N, Hornero R. EEG Characterization of the Alzheimer’s Disease Continuum by Means of Multiscale Entropies. Entropy. 2019; 21(6):544. https://doi.org/10.3390/e21060544
Chicago/Turabian StyleMaturana-Candelas, Aarón, Carlos Gómez, Jesús Poza, Nadia Pinto, and Roberto Hornero. 2019. "EEG Characterization of the Alzheimer’s Disease Continuum by Means of Multiscale Entropies" Entropy 21, no. 6: 544. https://doi.org/10.3390/e21060544
APA StyleMaturana-Candelas, A., Gómez, C., Poza, J., Pinto, N., & Hornero, R. (2019). EEG Characterization of the Alzheimer’s Disease Continuum by Means of Multiscale Entropies. Entropy, 21(6), 544. https://doi.org/10.3390/e21060544








