The Entropy of Laughter: Discriminative Power of Laughter’s Entropy in the Diagnosis of Depression
Abstract
:1. Introduction
1.1. The Social Meaning of Laughter
1.2. Biomedical Applications
1.3. The Present Study
2. Material and Methods
2.1. Subjects
2.2. Psychological Test
2.3. Compilation of Laughter
2.4. Laughter Processing
2.5. Statistical Analyses
3. Results
3.1. Decision Tree
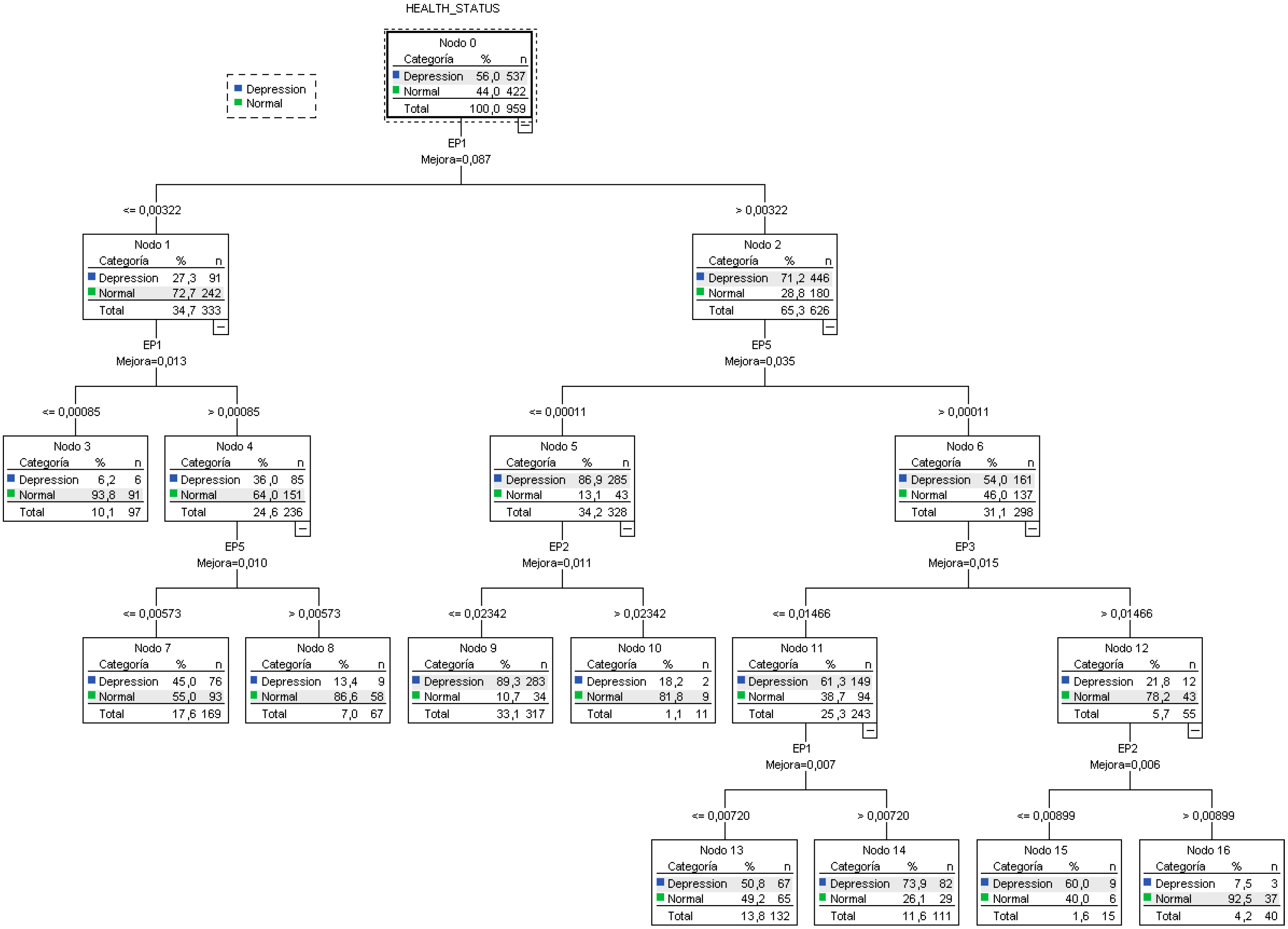
| Observed | Predicted | ||
|---|---|---|---|
| Patients | Controls | Percent Correct | |
| Patients | 441 | 96 | 82.1% |
| Controls | 134 | 288 | 68.2% |
| Overall Percentage | 60.0% | 40.0% | 76.0% |
3.2. Cluster Analysis
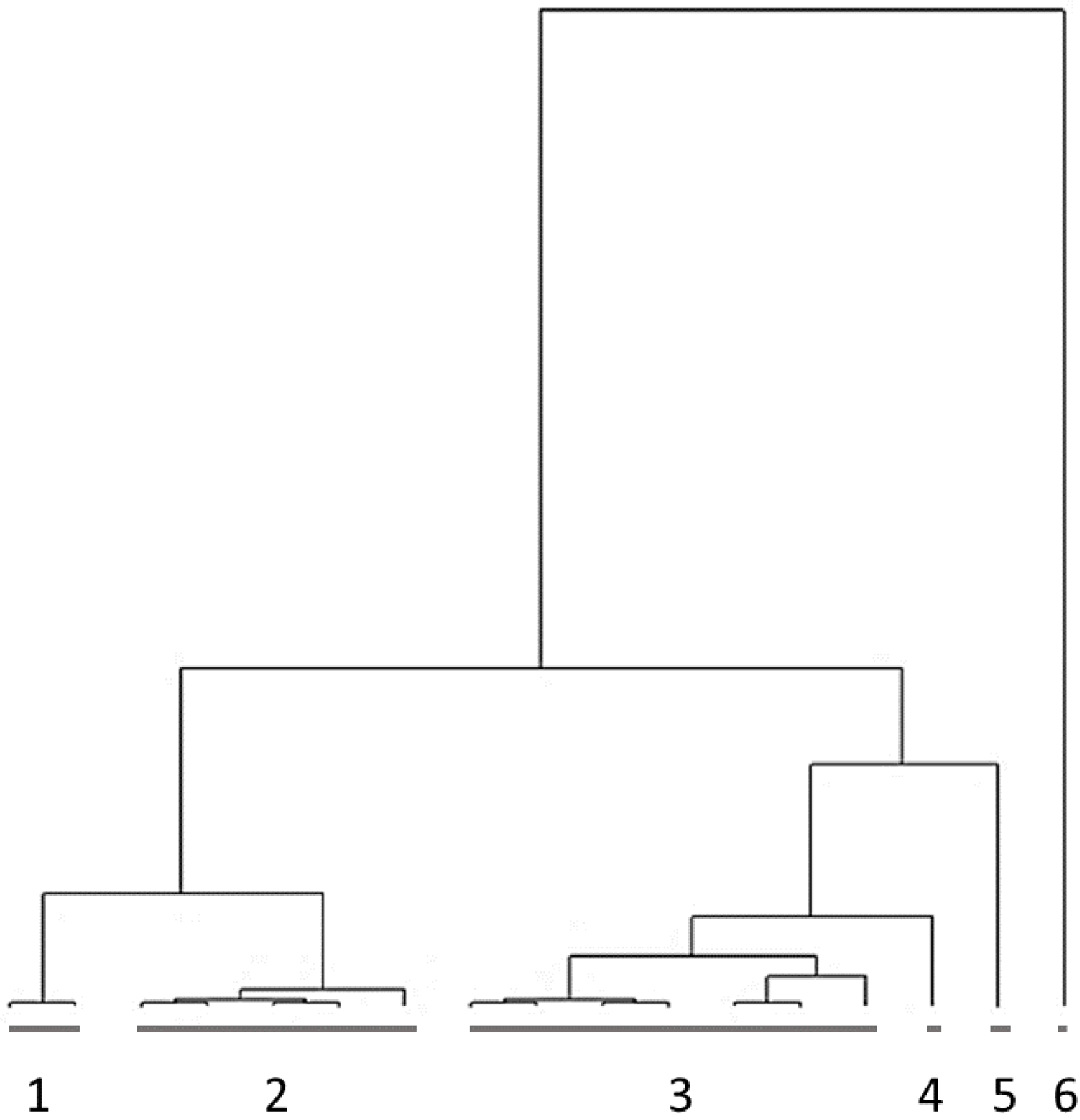
| EP1 | EP2 | EP3 | EP4 | EP5 | |
|---|---|---|---|---|---|
| Cluster 1 | − | − | − | − | − |
| Cluster 2 | ++ | − | − | − | − |
| Cluster 3 | + | + | − | − | |
| Cluster 4 | + | + | + | ||
| Cluster 5 | ++ | + | + | ||
| Cluster 6 | + | + | ++ | ++ | ++ |
| Entropy | Health Statuts | N | Mean | Standard Deviation | Median | Minimum | Maximum | p-Value * |
|---|---|---|---|---|---|---|---|---|
| EP1 | Depression | 537 | 0.0076 | 0.0054 | 0.0066 | 0.0005 | 0.0409 | 0.0000 |
| Normal | 422 | 0.0045 | 0.0059 | 0.0026 | 0.0002 | 0.0472 | 0.0000 | |
| Total | 959 | 0.0062 | 0.0059 | 0.0050 | 0.0002 | 0.0472 | – | |
| EP2 | Depression | 537 | 0.0078 | 0.0055 | 0.0070 | 0.0000 | 0.0483 | 0.0000 |
| Normal | 422 | 0.0098 | 0.0118 | 0.0062 | 0.0000 | 0.0818 | 0.0000 | |
| Total | 959 | 0.0087 | 0.0089 | 0.0067 | 0.0000 | 0.0818 | – | |
| EP3 | Depression | 537 | 0.0058 | 0.0058 | 0.0052 | 0.0000 | 0.0560 | 0.0000 |
| Normal | 422 | 0.0097 | 0.0128 | 0.0064 | 0.0000 | 0.0954 | 0.0000 | |
| Total | 959 | 0.0075 | 0.0097 | 0.0056 | 0.0000 | 0.0954 | – | |
| EP4 | Depression | 537 | 0.0040 | 0.0048 | 0.0025 | 0.0000 | 0.0317 | 0.0000 |
| Normal | 422 | 0.0077 | 0.0115 | 0.0050 | 0.0000 | 0.1068 | 0.0000 | |
| Total | 959 | 0.0056 | 0.0086 | 0.0036 | 0.0000 | 0.1068 | – | |
| EP5 | Depression | 537 | 0.0026 | 0.0040 | 0.0000 | 0.0000 | 0.0231 | 0.0000 |
| Normal | 422 | 0.0064 | 0.0107 | 0.0030 | 0.0000 | 0.0916 | 0.0000 | |
| Total | 959 | 0.0042 | 0.0079 | 0.0012 | 0.0000 | 0.0916 | – |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Provine, R.R. Laughter; Viking: New York, NY, USA, 2000. [Google Scholar]
- Ross, M.D.; Owren, M.J.; Zimmermann, E. Reconstructing the Evolution of Laughter in Great Apes and Humans. Curr. Biol. 2009, 19, 1106–1111. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, R. The Human Story: A New History of Mankind’s Evolution; Faber & Faber: London, UK, 2004. [Google Scholar]
- Ruch, W.; Ekman, P. The Expressive Pattern of Laughter. In Emotion, Qualia, and Consciousness; World Scientific: Singapore, Singapore, 2001; pp. 426–443. [Google Scholar]
- Bachorowski, J.-A.; Owren, M.J. Vocal Acoustics in Emotional Intelligence. In The Wisdom in Feeling: Psychological Processes in Emotional Intelligence; Barrett, L.F., Salovey, P., Eds.; The Guilford Press: New York, NY, USA, 2002; pp. 11–36. [Google Scholar]
- Szameitat, D.P.; Alter, K.; Szameitat, A.J.; Wildgruber, D.; Sterr, A.; Darwin, C.J. Acoustic profiles of distinct emotional expressions in laughter. J. Acoust. Soc. Am. 2009, 126, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Weems, S. Ha! The Science of When We Laugh and Why; Basic Books: New York, NY, USA, 2014. [Google Scholar]
- Kozintsev, A. The Mirror of Laughter; Transaction Publishers: Piscataway, NJ, USA, 2010. [Google Scholar]
- Oldenburg, R. The Great Good Place, 3rd ed.; Marlowe: New York, NY, USA, 1999. [Google Scholar]
- Allman, J. Evolving Brains; Scientifc American Library: New York, NY, USA, 1999. [Google Scholar]
- Baron-Cohen, S.; Ring, H.; Wheelwright, S.; Bullmore, E.; Brammer, M.; Simmons, A.; Williams, S. Social intelligence in the normal and autistic brain: An fMRI Study. Eur. J. Neurosci. 1999, 11, 1891–1898. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, R. The Social Brain: Mind, Language, and Society in Evolutionary Perspective. Annu. Rev. Anthropol. 2003, 32, 163–181. [Google Scholar] [CrossRef]
- Marijuán, P.C.; Navarro, J. The Bonds of Laughter: A Multidisciplinary Inquiry into the Information Processes of Human Laughter. 2011. arXiv:1010.5602v1. Available online: http://arxiv.org/abs/1010.5602 (accessed on 19 January 2016).
- Kashdan, T.; Yarbro, J.; McKnight, P.; Nezlek, J. Laughter with someone else leads to future social rewards: Temporal change using experience sampling methodology. Personal. Individ. Differ. 2014, 58, 15–19. [Google Scholar] [CrossRef]
- Urbain, J.; Cakmak, H.; Dutoit, T. Automatic Phonetic Transcription of Laughter and Its Application to Laughter Synthesis. In Proceedings of the 2013 Humaine Association Conference on Affective Computing and Intelligent Interaction (ACII), Geneva, Switzerland, 2–5 September 2013.
- Bea, J.; Marijuán, P. The Informational Patterns of Laughter. Entropy 2003, 5, 205–213. [Google Scholar] [CrossRef]
- Takahashi, D.; Fenley, A.; Teramoto, Y.; Narayanan, D.; Borjon, J.; Holmes, P.; Ghazanfar, A. The developmental dynamics of marmoset monkey vocal production. Science 2015, 349, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Friston, K. The free-energy principle: A unified brain theory? Nat. Rev. Neurosci. 2010, 11, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Carhart-Harris, R.; Leech, R.; Hellyer, P.; Shanahan, M.; Feilding, A.; Tagliazucchi, E.; Chialvo, D.; Nutt, D. The entropic brain: A theory of conscious states informed by neuroimaging research with psychedelic drugs. Front. Hum. Neurosci. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Bryant, G.; Aktipis, C. The animal nature of spontaneous human laughter. Evol. Hum. Behav. 2014, 35, 327–335. [Google Scholar] [CrossRef]
- Hasan, H.; Hasan, T. Laugh Yourself into a Healthier Person: A Cross Cultural Analysis of the Effects of Varying Levels of Laughter on Health. Inter. J. Med. Sci. 2009, 6, 200–211. [Google Scholar] [CrossRef]
- Takeda, M.; Hashimoto, R.; Kudo, T.; Okochi, M.; Tagami, S.; Morihara, T.; Sadick, G.; Tanaka, T. Laughter and humor as complementary and alternative medicines for dementia patients. BMC Complement. Altern. Med. 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Gelkopf, M. The Use of Humor in Serious Mental Illness: A Review. Evid. Based Complement. Altern. Med. 2011, 2011, 342837. [Google Scholar] [CrossRef] [PubMed]
- Ganz, F.; Jacobs, J. The effect of humor on elder mental and physical health. Geriatr. Nurs. 2014, 35, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Falkenberg, I.; Kiángel, K.; Bartels, M.; Wild, B. Sense of humor in patients with schizophrenia. Schizophr. Res. 2007, 95, 259–261. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.; Haonni, B.; Haug, M.; Amrhein, I.; Krebs-Roubicek, E.; Mäller-Spahn, F.; Savaskan, E. Humour therapy in patients with late-life depression or Alzheimer's disease: A Pilot Study. Int. J. Geriatr. Psychiatry 2006, 22, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Uekermann, J.; Channon, S.; Lehmkámper, C.; Abdel-Hamid, M.; Vollmoeller, W.; Daum, I. Executive function, mentalizing and humor in major depression. J. Inter. Neuropsychol. Soc. 2008, 14, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.; Youn, C. Effects of laughter therapy on depression, cognition and sleep among the community-dwelling elderly. Geriatr. Gerontol. Inter. 2011, 11, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Ruch, W.F.; Heintz, S. Humour styles, personality and psychological well-being: What’s humour got to do with it? Eur. J. Humour Res. 2014, 1, 1–24. [Google Scholar]
- Navarro, J.; del Moral, R.; Alonso, M.; Loste, P.; Garcia-Campayo, J.; Lahoz-Beltra, R.; Marijuán, P. Validation of laughter for diagnosis and evaluation of depression. J. Affect. Disord. 2014, 160, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Brieva, J.; Cordero-Villafafila, A. A new validation of the Hamilton Rating Scale for depression. J. Psychiatr. Res. 1988, 22, 21–28. [Google Scholar] [CrossRef]
- Breiman, L.; Friedman, J.H.; Stone, C.J.; Olshen, R.A. Classification and Regression Trees; Chapman&Hall: London, UK, 1984. [Google Scholar]
- Lebart, L.; Morineau, A.; Piron, M. Statistique Exploratoire Multidimensionnelle; Dunod: Paris, France, 1995. (In French) [Google Scholar]
- Lahoz-Beltrá, R. Bioinformática: Simulación, Vida Artificial e Inteligencia Artificial; Ediciones Díaz de Santos: Madrid, Spain, 2004. (In Spanish) [Google Scholar]
- Van Orden, G.C.; Kloos, H.; Wallot, S. Living in the Pink: Intentionality, Wellbeing, and Complexity. In Philosophy of Complex Systems; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Blanco, S.; Garay, A.; Coulombie, D. Comparison of Frequency Bands Using Spectral Entropy for Epileptic Seizure Prediction. ISRN Neurol. 2013, 2013, 287327. [Google Scholar] [CrossRef] [PubMed]
- Staniczenko, P.; Lee, C.; Jones, N. Rapidly detecting disorder in rhythmic biological signals: A spectral entropy measure to identify cardiac arrhythmias. Phys. Rev. E 2009, 79, 011915. [Google Scholar] [CrossRef] [PubMed]
- Danev, S.; St. Stoyanov, D. Early Noninvasive Diagnosis of Neurodegenerative Diseases. Folia Med. 2010, 52, 5–13. [Google Scholar] [CrossRef]
- Stoessl, A.J. Neuroimaging in the early diagnosis of neurodegenerative disease. Transl. Neurodegener. 2012, 1. [Google Scholar] [CrossRef] [PubMed]
- Grasso, M.; Piscopo, P.; Confaloni, A.; Denti, M. Circulating miRNAs as Biomarkers for Neurodegenerative Disorders. Molecules 2014, 19, 6891–6910. [Google Scholar] [CrossRef] [PubMed]
- Pievani, M.; Filippini, N.; van den Heuvel, M.; Cappa, S.; Frisoni, G. Brain connectivity in neurodegenerative diseases—from phenotype to proteinopathy. Nat. Rev. Neurol. 2014, 10, 620–633. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Campbell, N. Classification of social laughter in natural conversational speech. Comput. Speech Lang. 2014, 28, 314–325. [Google Scholar] [CrossRef]
- Bonin, F.; Campbell, N.; Vogel, C. Time for laughter. Knowl. Based Syst. 2014, 71, 15–24. [Google Scholar] [CrossRef]
- Bachorowski, J.; Owren, M. The acoustic features of human laughter. J. Acoust. Soc. Am. 2002, 111, 2480. [Google Scholar] [CrossRef]
- Panksepp, J. Psychology. Beyond a joke: From animal laughter to human joy? Science 2005, 308, 62–63. [Google Scholar] [CrossRef] [PubMed]
- Doyle, L.; McCowan, B.; Hanser, S.; Chyba, C.; Bucci, T.; Blue, J. Applicability of Information Theory to the Quantification of Responses to Anthropogenic Noise by Southeast Alaskan Humpback Whales. Entropy 2008, 10, 33–46. [Google Scholar] [CrossRef]
- Bering, J. The Rat that Laughed. Sci. Am. 2012, 307, 74–77. [Google Scholar] [CrossRef] [PubMed]
- Scherer, K. Vocal communication of emotion: A Review of Research paradigms. Speech Commun. 2003, 40, 227–256. [Google Scholar] [CrossRef]
- Bachorowski, J.; Owren, M. Vocal Expressions of Emotion. In Handbook of Emotions; Lewis, M., Haviland-Jones, J., Barrett, L., Eds.; The Guilford Press: New York, NY, USA, 2008. [Google Scholar]
- Amador, A.; Perl, Y.; Mindlin, G.; Margoliash, D. Elemental gesture dynamics are encoded by song premotor cortical neurons. Nature 2013, 495, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Coen, P.; Clemens, J.; Weinstein, A.; Pacheco, D.; Deng, Y.; Murthy, M. Dynamic sensory cues shape song structure in Drosophila. Nature 2014, 507, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Collins, K.P.; Marijuán, P.C. El Cerebro Dual; Hacer: Barcelona, Spain, 1997. (In Spanish) [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro, J.; Del Moral, R.; Cuesta-Alvaro, P.; Lahoz-Beltra, R.; Marijuán, P.C. The Entropy of Laughter: Discriminative Power of Laughter’s Entropy in the Diagnosis of Depression. Entropy 2016, 18, 36. https://doi.org/10.3390/e18010036
Navarro J, Del Moral R, Cuesta-Alvaro P, Lahoz-Beltra R, Marijuán PC. The Entropy of Laughter: Discriminative Power of Laughter’s Entropy in the Diagnosis of Depression. Entropy. 2016; 18(1):36. https://doi.org/10.3390/e18010036
Chicago/Turabian StyleNavarro, Jorge, Raquel Del Moral, Pedro Cuesta-Alvaro, Rafael Lahoz-Beltra, and Pedro C. Marijuán. 2016. "The Entropy of Laughter: Discriminative Power of Laughter’s Entropy in the Diagnosis of Depression" Entropy 18, no. 1: 36. https://doi.org/10.3390/e18010036
APA StyleNavarro, J., Del Moral, R., Cuesta-Alvaro, P., Lahoz-Beltra, R., & Marijuán, P. C. (2016). The Entropy of Laughter: Discriminative Power of Laughter’s Entropy in the Diagnosis of Depression. Entropy, 18(1), 36. https://doi.org/10.3390/e18010036







