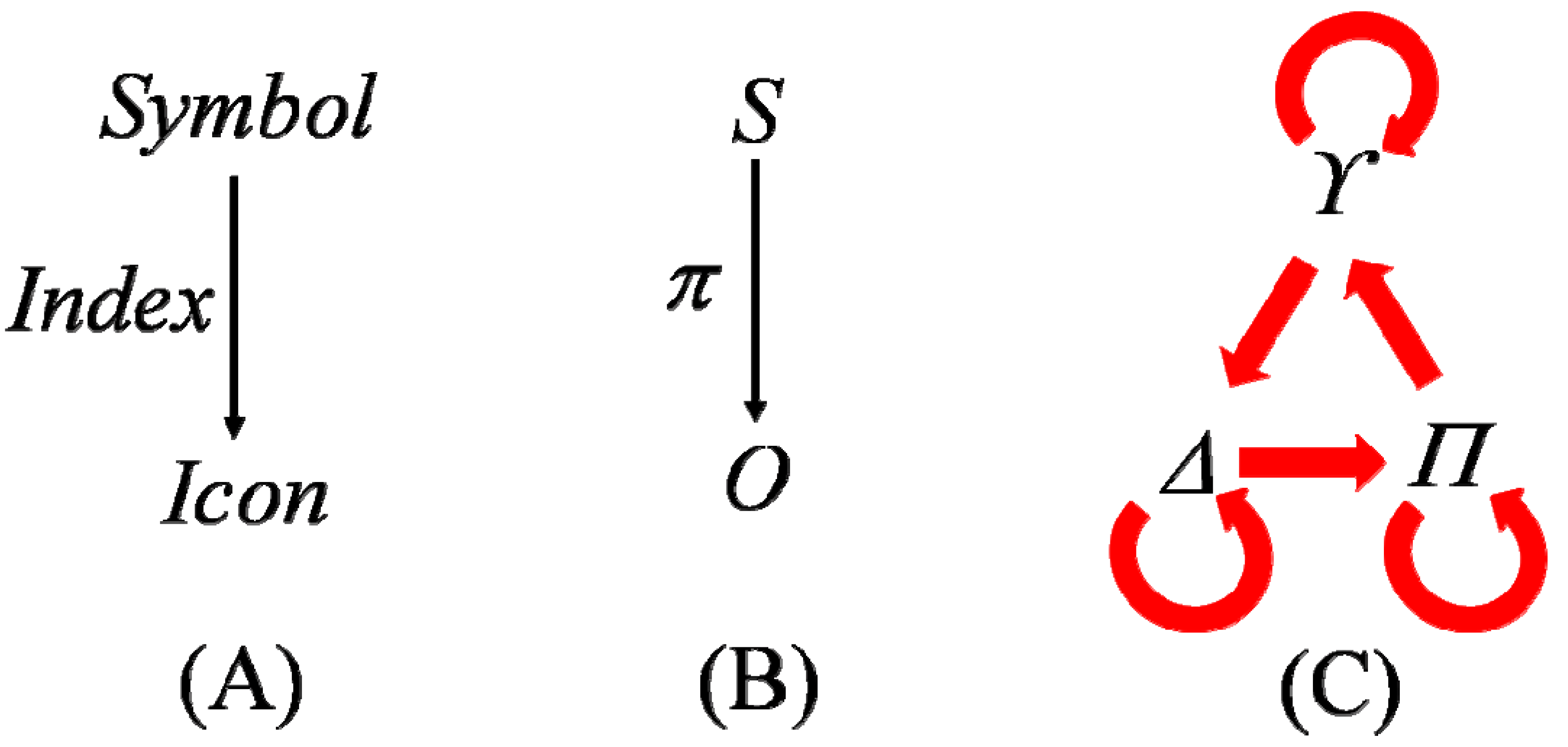
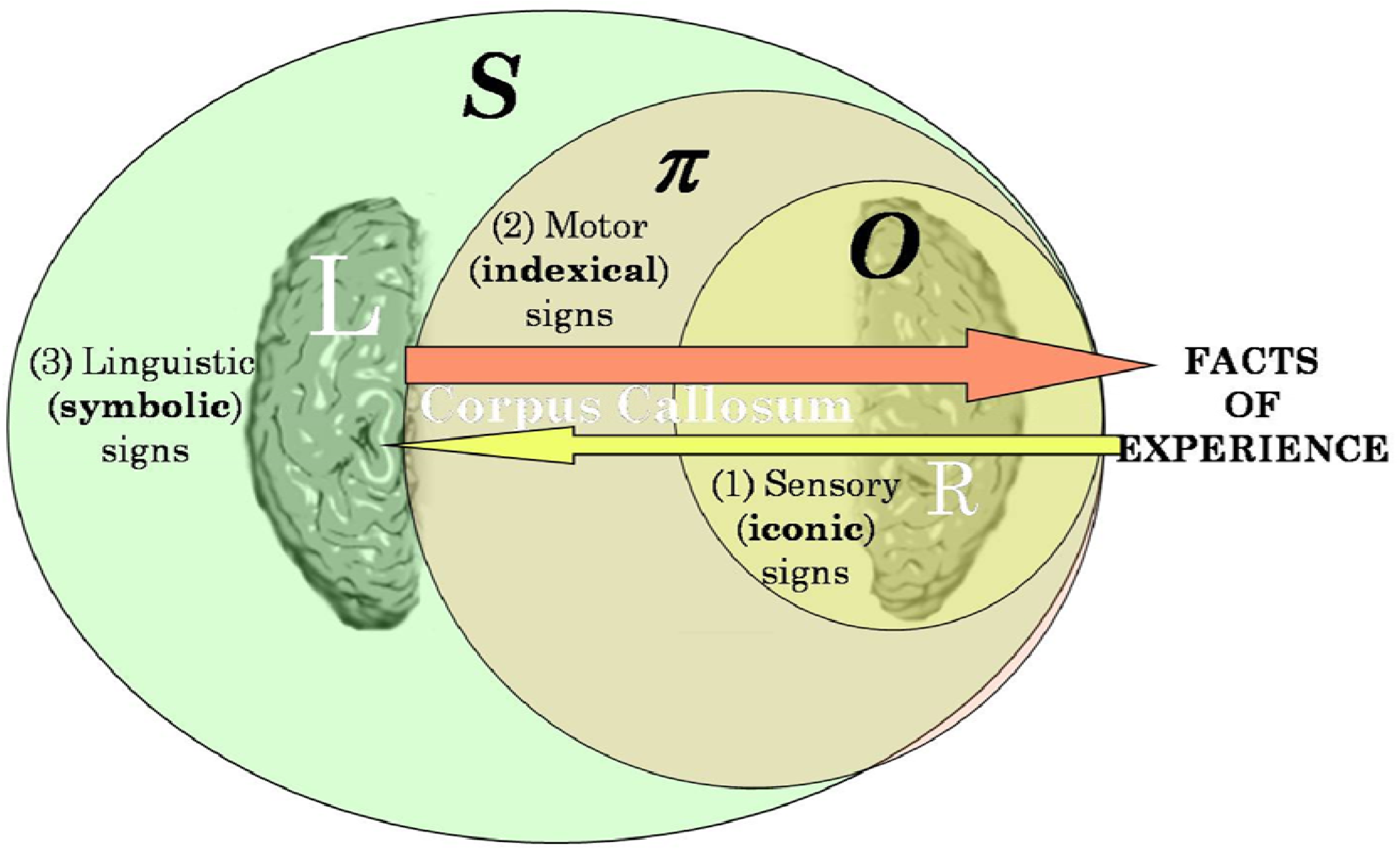
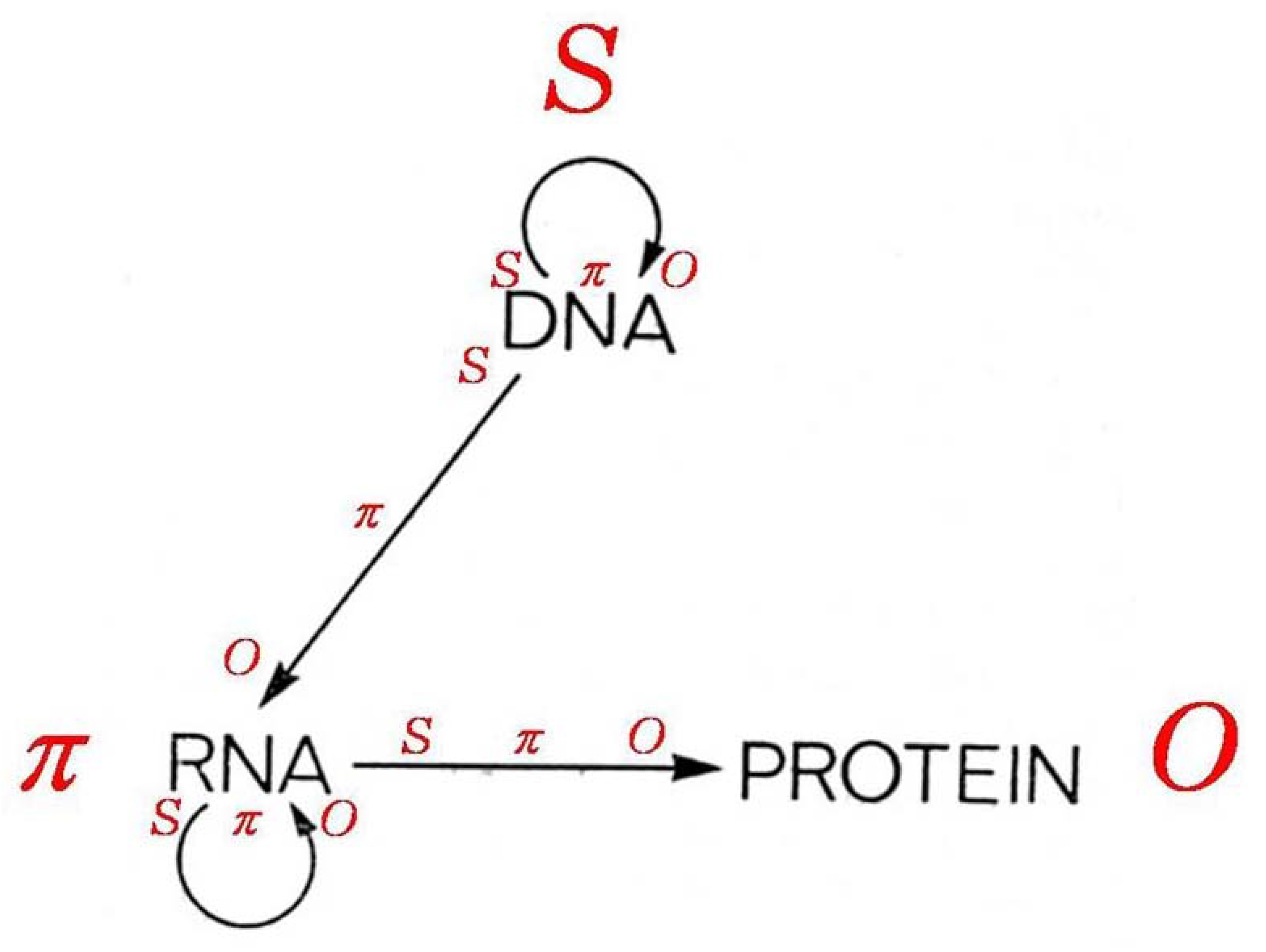
4.2. Symbol to Object, Genotype to Phenotype
The direction of the design information flows from the
S through
π to the
O by logical necessity—a necessity hypothesized to fit the relevant empirical facts according to Crick’s controversial “central dogma” of molecular biology [
74,
75]—see the diagram given here as
Figure 8. The controversy about the central dogma has centered on questions about the extent to which the sequence and stereoscopic arrangement of amino acids in particular proteins—the acknowledged building blocks of cells, organs, and whole organisms—could be sufficiently specified in DNA [
95,
96].
Among the particular objections to the central dogma addressed by Crick in 1970 [
75] were early speculations by Howard Temin about how cancer producing viruses could modify a cell’s nuclear DNA so as to cause its subsequent out-of-control (cancerous) metastatic reproduction in competition with normal cells, tissues, and organs as guided by the body’s original DNA [
97]. Other problems that would come to light were the existence of disease agents consisting, seemingly, of nothing but badly formed proteins that seemed almost to replicate themselves—leading Prusiner in 1982 to propose the term prion as a portmanteau constructed from “proteinaceous infectious” matter [
98]. Best known examples today of multiplying prions include the ultimately lethal beta amyloid plaques of mad cow disease which in humans is known as Creutzfeldt–Jakob disease, and, perhaps also the similar plaques in the brains of persons with Alzheimer’s disease. In addition, there were complications in the transfer of information from RNAs to proteins and in the necessary stereoscopic folding of the latter to form fully functional variants.
The hypothetical action of viral RNA in rewriting some portion of cellular DNA was demonstrated with the independent discovery of reverse transcriptase by Temin and by David L. Baltimore in 1970 [
99,
100]—a finding that would make Nobel laureates of both of them in 1975. It would also provide—according to some, e.g., see Morange [
101,
101], Rosenberg [
104], and Stotz [
103]—a basis for complicating the “central dogma” of molecular biology and for making it, superficially at least, more compatible with another underlying doctrine—an as yet to be determined variant of Darwinian and/or Lamarckian evolution—that would absolutely require some means of getting the information about sequences and arrangements of aminos to flow from nonliving matter backward, as it were, in order to inform DNA of its teleological functions with respect to organisms and the biosphere. Rephrasing the problem faced by the apparent incompatibility of Crick’s “central dogma” with the requirements of the reverse flow of biological information from any theory of emergence or evolution in the terms of a general theory of signs as suggested by
Figure 8, the dominant
S in the foundational sign relation,
SπO, would have to be temporarily subordinated in some manner, or made a co-equal with the
O of that relation.
To get that to happen, the critical directionality of the
π mapping in the formula would have to be reversed. Logically speaking, this is difficult on account of the fact that the relation between any determining
S and its
O is both abstract and arbitrary. Whereas two abstract
Ss can implicate and presuppose one another simultaneously in a completely general manner, as suggested in the recursive association of DNA with DNA as seen at the top of the diagram of the “central dogma” in
Figure 8—that is, DNA (also RNA) can serve as its own object of representation—conceiving of a means by which raw aminos could form up into the many proteins required for even a bacterium or fungus, never ming how they could generate RNAs which might then be transformed into DNAs, remain as unsolved mysteries. Also, the central dogma as originally formulated would seem to preclude any means by which randomly formed amino sequences of the appropriate chirality for the proteins of a life form could operate in the “backward” direction to produce RNA and/or DNA representations of themselves.
Because the prior teaching of evolution—as noted by Carl Woese in 2001 [
105]—seemed to require modifications of Crick’s “central dogma” of molecular biology in order to allow for backward flow of information from proteins to RNA and from RNA to DNA—retroviruses held out hope for the reconciliation of the conflicting claims—e.g., along that line see Hoyle and Wickramasinghe in 1991 [
106], also Forterre in 2006 [
107]. However, the hope for making viruses the engines of DNA production seems to reach an impasse at just the point where the retroviruses are most critically needed as pointed out in 2009 by Forterre and Prangishvili [
108]. They wrote:
. . . structural analyses of capsid proteins have revealed that at least two types of virions originated independently before the LUCA (the Last Universal Cellular Ancestor). Although several hypotheses have been recently proposed to explain the origin of viruses, the emergence of virions, as a specific mechanism for gene dissemination, remains unexplained (p. 466).
A dual common problem, it would seem, for each and every one of the objections raised against Crick’s central dogma is that the suspected means of reversing the flow of information from proteins to nuclear DNA is that all of the modifications seem only to impact DNA outside of the germ line and are limited in their scope and/or are harmful or lethal to the organism in which they occur. Lethality, of course, is a detriment to the possibility of reproduction, but even if reproduction occurs, unless the modified DNA is in the germ line, even if the modification itself is not lethal, neither can it be passed on. The problem was well put by August Weismann [1834–1914] in what is commonly referred to as the “Weismann barrier”—the principle that genetic information flows from the germ cells to the rest of the body but not, at least not normally, in the other direction.
The Weismann principle [
109]—that the “essential nature of the germ-cell dominates over the organism which will grow from it” (p. 103)—came near anticipating Crick’s central dogma. With respect to the latter, Morange [
101] concluded that
the only way to justify its existence [that of the central dogma] is through the description of the evolutionary history that shaped the relations between DNA, RNAs and proteins (p. 247).
4.4. The Developing Individual
In the case of the initial DNA mapping of any given individual, the symbol and its object are almost indistinguishable except for the fact that in every pairing there is evidently a dominant and subordinate member. By convention we suppose that the dominant member is the symbolic element and the subordinate is the object. The mapping, at the level of the initial DNA, consists of the bond between the haploids with the dominant member of the pair on the strand that determines the “sense” rather than the “antisense”—and during the first mitotic division the prior strand in each case will be the dominant (S) and the derived strand the subordinate (O). When the validation implicit in the successful mating of the complementary halves of the double helical DNA is completed, the unfolding of the life story of the newly formed individual begins to be expressed with mitotic division.
In the process of the union of sperm and egg, the sperm’s outer wall and organelles, including the mitochondria of the sperm [
111] are dismantled possibly for reuse or disposal. For this reason, the father’s mitochondrial DNA, except possibly in very rare instances [
112], plays no role in the mitochondria of the offspring which, in effect, receives all of its mitochondrial DNA (mtDNA) from its mother. Although some researchers have claimed mixing of mtDNA from both parents, Bandelt and colleagues have argued that all such reported observations can be attributed to errors in data analysis [
114,
115]. Setting the possibility of mtDNA inheritance from the father aside, as soon as the first complete DNA is formed, it replicates to provide for the formation of the first two stem cells of the new organism. These cells are termed totipotent because, assuming they are well formed, they have the capacity to authorize the development of every kind of cell in the new individual [
116].
In doing so, they must communicate with the host cells identifying themselves, presumably, as friend rather than foe. It seems likely that they do so, in part at least, through the common mitochondrial DNA shared between mother and child (the father’s mitochondrial DNA having been eliminated as noted). Fallarino,
et al. [
113], in any case, showed that mother’s cytotoxic immune cells that might otherwise attack the cells of the developing embryo are largely neutralized by chemical factors (cytochrome C and caspase-8). It may well turn out that indeed the mtDNA is a key factor in helping to resolve what has been called a paradox of immunology [
117]—namely, how is it that the maternal immune system does not attack and destroy the developing cells of the embryo? To avoid such destruction, the developing individual must in some way engage in effective biochemical cross-talk with its mother.
Over the first four days after conception, the initial pair of stem cells will divide to 4, 8, 16, and then 32 cells to form a sphere known as a morula. The sphere will then differentiate itself by the fifth day into a blastocyst. A particular part of the surface of the blastocyst known as the trophoblast will first migrate down the fallopian tube in which it was first formed to the uterus where it will attach itself to the inner wall. At the point of attachment it will begin to form that part of the placenta that will develop into the umbilical cord and the thickened multi-layered sponge-like portion of the placenta enabling it to bring nutrients to its three distinct components: the outer layer of cells at its surface that will become the placental sack in which the embryo itself will develop, the inner area known as the blastocele containing a fluid consisting mainly of globin and nutrients that will eventually be incorporated in a yolk sac, and the pluripotent cells within the container and developing outward from its inner surface within the inner chamber of the blastocyst known as the embryoblast.
As the embryo continues to develop, after the blastocyst has implanted itself to connect with the uterine wall, the embryo will undergo the transformation known as gastrulation—a term that refers specifically to the development of the stomach and insides of the individual. While gastrulation is occurring in the developing embryo itself the blastocyst is transformed into a gastrula with multiple chambers. At the junction with the uterine wall a complex multilayered connection, communication, and circulation system is developed, including the umbilical cord and differentiated tissues of the placenta, that will provide the developing individual with access to its critical supply of oxygen and nutrients coming from its mother while also providing separation from its mother’s blood and from potential attack by its mother’s own mobile immune cells circulating in her bloodstream.
As it is being transformed into the larger and more complex gastrula, inside the blastocyst beneath its upper chamber that is developing into the placental circulation system and umbilical connection, the inner cell mass, now shaped roughly like a saucer thicker at the middle than at the edges, is differentiating itself into three distinct pluripotent germ layers. The disk will rotate, elongate, and divide itself along a center line while at the same time folding inward as it forms the tube for the spinal cord and its surfaces connect as its inward parts begin to form along with the head and limbs of the individual [
118].
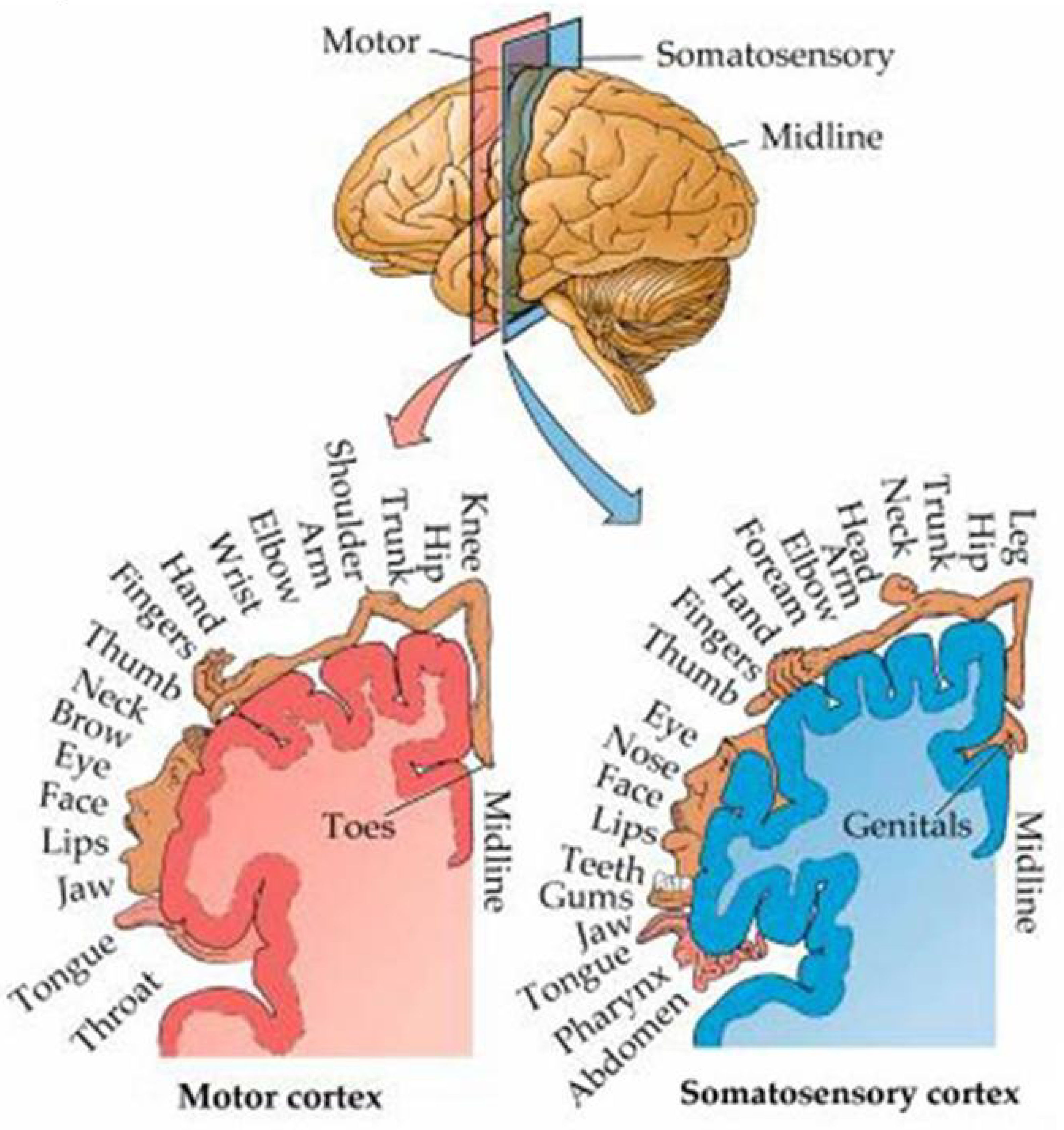
4.4.1. The Ectoderm
The first of the three main pluripotent stem layers in the disk shape that will be transformed in gastrulation is referred to as (1) the ectoderm, or outer layer, that will develop into the skin, the lenses of the eyes, the connective tissues of the head, the face, lips, mouth and tongue, as well as all of the nervous system. The ectoderm provides the cells needed to form the head and brain as well as the openings for the eyes, ears, nose, and mouth inward as far down as the pharynx and larynx, and from the outer skin inward to connect the openings to the urethra in both males and females and the labia of the vagina in females, and of course the anus.
The ectoderm also contains the germ cells that will form gametes (the reproductive cells critical to the survivability of the species) as well as those that produce the “neural crest” which is sometimes regarded as a separate pluripotent germ cell line that develops into the brain and nervous system. In terms of the pragmatic mapping formula, it can be argued that the ectoderm as a defining element of the entire surface and the future of the organism as a progenitor can be construed as the S portion of the SπO formula.
The ectoderm is critically involved producing the body’s information and control systems, including the brain and articulatory organs of the mouth, tongue, lips, vocal folds, pharynx, and probably the larynx as well [
119,
120], and is the source from which the germ cells will migrate to the gonads to form gametes. Therefore, it is the repository of crucial information for control and development of the present individual as well as future generations and it is logically the dominant element in the three distinct germ layers of the developing embryo.
4.4.2. The Mesoderm
The mesoderm, or middle layer of the embryonic disk, will develop into the skeleton, the bones and its marrow, as well as striated and smooth muscles. It provides the pluripotent stem cells that will form the heart and circulatory systems, the mobile parts of the digestive/excretory systems as well as the urinary systems and the kidneys, the lymphatic systems, and the sweat glands [
118,
119]. These elements are the main transport and mobility systems of the body corresponding to the
π portion of the
SπO formula.
They include both the mobility systems that come under the control of conscious intentions and volitions as well as the autonomous movements of the body. The latter are especially involved in the acquisition of critical supplies such as oxygen, water, and nutrients, as well as their transport to all of the organ systems throughout the body, and the exportation of waste products, toxins, and harmful disease agents through exhalation of carbon dioxide and the excretion of liquid wastes in sweat, urine, and feces. The heart is of particular interest as its movements are certainly affected by voluntary movements of other striated muscles and yet this highly organized system of striated muscle tissues, unlike the striated muscle tissues that normally fall under voluntary control, is ordinarily under the control of the autonomous nervous system. The rate of the heart’s pumping, of course, is normally indirectly controlled by demands for oxygen that can be increased or decreased owing to intentional (volitional) movements of the body.
The mesoderm is also, as well we might expect seeing that it is the source of the essential autonomous mobility systems of the body, the primary source of the body’s immune systems. These systems are deeply integrated into the transport involved in the importation of needed provisions, their distribution to all the organs of the body, and the exportation of gaseous, liquid, and solid waste products. The mesoderm is not only involved in producing those organ systems involved in nourishing and maintaining the body with respect to needed provisions that must be imported and wastes that need to be exported, but also with respect to the production of the elaborate defense systems that are commonly, and somewhat incorrectly referred to with a singular umbrella phrase as “the immune system.” The body’s internal defenses, however, are actually constituted by a deeply layered and richly integrated system of systems.
4.4.3. The Endoderm
The endoderm, or inner layer of the embryonic disk, will develop into the inner layers of bodily surfaces in the lungs, the digestive tube except for the mouth, pharynx, and probably the larynx. The endoderm will provide the cells for the inner part of the rectum and the inner linings of glands which connect to the gut, including the stomach, colon, liver, pancreas, urinary bladder, urethra, epithelial linings of the trachea, lungs, pharynx, thyroid, parathyroid, and the intestines. The endoderm provides for the generation of the inner linings of the inner cavities of the ears, the urinary bladder, the inner parts of urethra as well as the inner linings of the thyroid gland and thymus [
119]. Nearly all of the endocrine glands originate in this layer with the exception of part of the pituitary which arises in the ectoderm. When the work of the endoderm is completed, however, the envelope of the bodily icon with its many “surfaces within surfaces”—to use a phrase from Hoffmeyer [
24] is well defined and fitted together as a whole. It seems strange to realize that the internal surfaces of the body are much larger and, with respect to the internal functions of the whole body, just as critical to its viability as its outer surface. For instance, the inner surface of the intestinal lining of an adult, if flattened out would, according to plausible estimates, cover an area about the size of a doubles tennis court [
121].
The outer surfaces of the body, as already noted, are produced by the pluripotent cells of the ectoderm while all the internal organs not already provided for by the ectoderm, it seems, are generated by the mesoderm, but the innermost organ surfaces, those of the gut and of the organs that empty into it, or into the urinary tract, together with nearly all of the endocrine control and maintenance systems come from the endoderm. Thus, the endoderm effectively completes the boundaries of the bodily icon, the O, and the pragmatic mapping formula, SπO, is completed as the developing embryo comes to term and is readied for birth.
4.5. Fetal Immune Defenses, Nutrition, and Clean-Up
During the development of the viable fetus, the newly formed individual is provided oxygen and nutrients from mother’s blood through a remarkable temporary circulation system that ingeniously prevents the mingling of the mother’s blood with the baby’s. The system not only protects the developing child from attack by infections, even ones that might affect the mother, but it also buffers the fetus from exposure to potentially harmful interactions that may take place in the mother’s bloodstream and elsewhere while the baby is undergoing delicate and rapid development—the kind where a seemingly small problem could easily cascade into a series of harmful or even lethal effects downstream.
The red blood cells of the developing fetus are produced from the mesoderm of the blastocyst. However, the defense against infectious disease must be primarily provided by the mother’s filtration and immune systems which clean her own bloodstream and secondarily by the only known type of the mother’s immunoglobulin (IgG) that can pass across what is loosely known as the “placental barrier”—that barrier consisting of essential membranes that separate the mother’s blood from that of the developing fetus: for one, there is the basal wall of the trophoblast bordering the intervillous space of the placenta that is richly penetrated by fetal capillaries; and, second, there is the basal wall of the fetal capillaries themselves. These membranes, and any space between them, must both be crossed in order for chemical materials—oxygen, carbon-dioxide, nutrients, and wastes—to pass between fetus and mother.
The whole placenta itself develops in the upper chamber of the blastocyst at the place where it is joined to the wall of the mother’s uterus. It is a distinct organ generated by the germ cells of the blastocyst that is crucial to the development of the fetus and yet will be completely abandoned at the time of the newborn infant’s separation from the mother with the severing of the umbilical cord. The placenta itself is a multilayered compartment contained within the mother’s uterus. The outer layer of the placenta consists of a spongy material penetrated by maternal blood vessels, both veins and arteries, that supply maternal blood to the inside of an oddly shaped sac at the center of the placenta referred to as the intervillous space. The membrane forming the oddly shaped sac containing the intervillous space, sometimes merged with the outer basal membrane of the fetal capillaries, constitutes the essential “placental barrier.” It is effectively wrapped around the branching tree like structures of blood vessels emanating from the developing fetus though the umbilical cord. The barrier membranes allow oxygen and nutrients to pass from mother’s blood to the blood of the infant and at the same time to permit carbon dioxide and waste products from the fetal blood to pass to the mother’s bloodstream.
The blood flow on the mother’s side of the barrier is from the higher pressure arterial inputs toward the lower pressure venal tubes assuring an oxygen/nutrient rich supply of blood on the mother’s side of the placental barrier that enables the fetal blood supply to expel its wastes while loading up with oxygen and nutrients for the return trip to the developing baby. Thus throughout the process the placenta does the work of the lungs, digestive, excretory, and immune systems of the developing baby.
The fetal circulatory system, though powered by the fetal heartbeat beginning at about the fifth to seventh week of gestation, is very different during fetal development than after birth. For one, the fetal red blood cells originating in the mesoderm have a higher affinity for oxygen than the ones that will be constructed after birth in the infant’s bone marrow [
122]. For another the flow of deoxygenated blood from the fetus back to the placenta occurs through the two higher pressure arteries flowing away from the fetal heart through the umbilical cord back to the placenta. This is unusual because, of course, arteries normally carry oxygenated blood.
Meanwhile oxygenated blood flows from the placenta to the fetus through a single large vein. Again, this flow contrasts with the later arrangement because in the fetus the large umbilical vein carries oxygenated blood toward the heart. This is almost exactly the opposite of the flow of oxygenated versus deoxygenated blood after birth when the umbilical cord will be cut and the neonate must breathe on its own. Another interesting peculiarity of the fetal circulation is that the oxygenated blood returning from the placenta to the fetus is divided at the liver with about half of it being filtered through the fetal liver while the other half goes on to the heart to be pumped through the developing fetal body, most of it bypassing the fetal lungs [
123], and then returning after being deoxygenated in the fetal capillaries to the placenta through the dual umbilical arteries. The two umbilical arteries return deoxygenated blood and liquid wastes from the fetal bladder, to the placenta.
Although much of the blood flowing to the fetal liver bypasses it, the routing of roughly half the placental blood through the fetal liver suggests that some monitoring and filtering of materials in the oxygenated and nutrient laden blood is already taking place in the fetal liver. The developing liver also contains some blood stem cells that migrate to the thymus, where mobile lymphocytes, called T-cells (see
section 4.9.4 below), mature and are tested and screened for autoimmunity. After birth, the formation of mature red blood cells and of the cells involved in the infant’s immune systems will take place in the bone marrow.
4.6. Neonatal Immune Defenses, Nutrition, Repair, and Clean-Up
Presumably, during the rapid development of the blastocyst, so long as sufficiently capable totipotent or pluripotent stem cells survive an insult, damaged cells may be replaced or repaired on the fly. However, defense, repair, and clean-up capabilities internal to the individual become more critical after birth. For the first few months, especially if the baby is nursed with its mother’s milk, it continues to benefit first from the residual IgG it received from its mother during gestation, and, subsequently, after birth it will continue to receive additional immune benefits through its mother’s milk [
124]. Of course, there is also the risk that even mother’s milk may also be a source of toxins. For instance, contamination from heavy metals, e.g., from dental mercury, appears to be a risk during breast feeding, especially if the mother’s exposure is substantial. For instance, the research shows plainly that placing or otherwise disturbing dental mercury (in dental amalgam) increases the exposure of the infant to this highly reactive toxin [
125]. Also, it is known that combining risk factors, e.g., putting metal toxins together, may have a multiplicative effect rather than a summative one owing to the potential interactions. Likewise, combining toxins with disease agents provides for unpredictable opportunistic interactions [
67].
High-risk combinations of toxins and disease agents may result from such diverse environmental sources as contaminated food or water, industrial wastes, pesticides, food preservatives, and even common medical procedures involving prescription drugs, dentistry, and vaccination. Even with prescribed drugs, antibiotics, vaccines (containing attenuated disease agents, adjuvants, animal proteins, and other adventitious components), and implantations of any kind placed in the body, clinical trials examining all possible interactions are prohibitively expensive even when the number of variables is less than or equal to three. The cost of clinical trials to examine possible interactions for numbers of variables greater than three grows at such an astronomical rate as to render studies of them essentially nonexistent. The lethal cases that involve multiple injuries from such interactions are apt to be recorded as some form of unexplained death—such as sudden infant death syndrome [
67,
126], anaphylactic shock, or simply as some unexplained variant of multiple systems damage attributed to unknown causes.
In nonlethal instances, interactive injuries may result in little more than a temporary malaise while in other cases the effects may result in a cascade of chronic, life-long and life-shortening illnesses, e.g., the disease condition known as emphysema associated with smoking and related hazards, or mesothelioma associated with asbestos exposure, and skin cancers related to excessive exposure to ultraviolet radiation come to mind. Because of the fact that damaged tissues are more susceptible to disease than healthy tissues, it follows that disease agents also have a greater chance of infecting persons who have already been damaged by toxins, radiation, or other injurious agents than persons who are in the peak of health.
For all of the foregoing reasons, it is unsurprising to discover that the body’s developing systems of waste disposal and detoxification, are closely related to its systems of defense against disease agents. Similarly, we should expect, presumably, to find also that the body’s systems of tissue repair will be intimately coordinated with both waste/toxin disposal and defense against invasive diseases. Likewise, when viewed from the side of potential disease agents that can invade the body—viruses, fungi, bacteria, and parasites—again it is unsurprising to find that undesirable microbes and organisms such as the common gut parasites known as pinworms and hookworms (along with many others that are less common) tend to bring a lot of undesirable company with them, both on their insides and on their outsides. Disease agents and parasites also tend to be loaded with toxic components that may sometimes be more harmful than the carrier. For all of these reasons, it is to be expected that the provisional requirements of breathing in and out, as well as nutritional requirements for water and food, must be expected to be intrinsically and deeply related to the detox, defense, and repair systems.
Therefore, it makes little or no sense to consider the immune systems as if they were isolated from or independent from the ordinary supply and maintenance systems that require interactions and communication between the biochemistry of the body and that of the external ecoment—to borrow an apt term from Brier [
15]—with all of its potential hazards for injuries and diseases. However, in this paper, I have committed to deal primarily with the communication systems, in particular with the role of TNR type representations, crucial to the functioning of the immune defenses.
4.7. Communication: Who Am I?
Initially, the identity of self cells is defined in terms of the major histocompatibility complex (MHC)—derived from the nuclear DNA. This set of identity papers—consisting at maturity of hundreds of millions of copies of this TNR identifying the self, in effect, like a complex medical record and passport—provides the proof of identity of essentially every cell in the body of the neonate. At the beginning, in ways not yet fully understood, the body’s totipotent cells were authorized to form any and every part of the human body. Progress from the totipotent cells to the differentiation that has occurred by six months after birth has presumably proceeded in large measure by communications from cell to cell during and after many mitotic divisions that have systematically constrained the expression of the DNA. The fertilized egg divides progressively into the cells of the blastocyst, the gastrula with its various layers, and so forth, right on up to the time of birth, after which, in typical cases the individual progresses to maturity.
About six months after the baby is born, just as the so-called “passive” immunity inherited from mother, fades in its effectiveness, the baby’s own immune defenses, which have been ramping up from before its birth, are already, as Beck and Habicht [
127] have said, “fabulously complex” (p. 60). In all its aspects and at almost every conceivable step, these incredible defense systems critically depend on successful communications in order to attain the homeostatic balance that we loosely refer to as “health and well-being.” In medical jargon the contrast is between thriving and that vague status that often precedes a descent into disease loosely (and negatively) described as “failure to thrive” [
119].
Questions concerning the identity of cells—whether they are legitimately part of the developing body, the self, or whether they are potentially harmful antigens, such as a toxin, an invading virus, fungus, microbe, or part of some other foreign organism (e.g., a gut worm or other parasite)—must be asked and answered correctly in order for the immune cells to do their work well. Also, it is useful for immune cells to be able to determine whether the cells of the self—the body to be defended, maintained, and possibly repaired—are healthy or possibly infected. If the cell is damaged and in need of help the immune cells may inquire concerning the nature and seriousness of the damage, infection, or deficiency in order to either correct it or send for help. In cases where the cell cannot properly identify itself, and/or report its status, the immune cell may be authorized either to kill and destroy it, or to call for help to accomplish the same purposes.
The cells that constitute the personnel for “immune” defense systems undergo processes that resemble production, nurturing and growth, program recruitment, training, hiring/firing, testing (sometimes of a do or die type), commissioning/decommissioning, arming/licensing, dispatching/recalling, reassignment in some cases, long-term inactive status subject to call-up in case of war, and eventual retirement and/or disassembly in what would appear to be analogous to radical organ donation of recyclable chemical parts. Comparisons of the body’s immune systems at a functional level can be drawn with ordinary municipal transport, supply, maintenance, waste disposal and sanitation departments, as well as many of the normal functions associated with fire and police departments. On a grander scale, the immune systems can be compared to defense systems on a national level by comparisons with the various branches of the military including information gathering resources, immigration control, Coast Guard/Border Patrol, common foot soldiers, elite commando units, and various other highly mobile forces performing such functions as reconnaisance, arrest, transport, detention, interrogation, execution, and disposal.
4.9. A Multilayered System of Interacting Systems
To truly appreciate the crucial importance of the balance that is ordinarily maintained by the normal interactions between the multiple layers of immune systems in a healthy human being, it is instructive to consider some of the ways that things can go radically wrong whenever one or more components of the multilayered system are thrown out of balance.
There are documented instances where this can be done unintentionally by a deliberate medical intervention. For instance, when penicillin has been administered in an effort to minimize the impact of an
E. coli O157 infection, for instance, the fatality rate increases from approximately 15% to 50% of the persons infected [
130]. The tripling of the potential lethality of the disease threat is attributable to the collateral damage to the ecoment of the gut by the administration of the antibiotic. To understand how such harmful effects—potentially deadly ones—can and do occur, it is necessary to take account of the interactions of the multilayered immune systems.
According to widely held and taught views of immunology three distinct levels of immune systems have been recognized. These include: (1) what is somewhat misleadingly called the “innate immune system,” (2) the “complement system,” and (3) the so-called “adaptive immune system” [
131]. The inter-relatedness and cross-talk between these systems is increasingly evident in the current research and it is now widely agreed that a fourth layer of the vast immune network must be recognized in the gut microbial ecosystem. The ecoment of the gut, formerly a “forgotten organ” [
132]—as O’Hara and Shanahan described it—is now increasingly thought of as an integral part of a normal and healthy body, perhaps even the “seat of the soul” as Andrew Wakefield has argued [
133].
As can easily be seen from the current research, all four of the well-recognized (if not well-defined) layers of the system of immune systems are in fact each multilayered in its own. They are also deeply interrelated and exquisitely complex from top to bottom. The known functions of the immune system defy any piecemeal explanation. In a healthy body, the many layers of the systems work together with such efficiency that it seems necessary to suppose that almost any single component presupposes the whole. Here I want to suggest the hypothesis that the immune system of systems as a whole must be not only provided for within the initial DNA of the organism, but that its epigenetic and probabilistic expression must be implicit in systems provided for there as well. This is certainly not to deny that countless slips can occur between the DNA and its expression in bodily systems, but I do intend to suggest that there are compelling theoretical reasons to suppose that Crick’s central dogma about the dominant role of DNA over RNA and both of them over proteins is probably more valid than many contemporary theoreticians might now suppose, e.g., Gottlieb, Stotz, and others [
96,
103].
There are also empirical evidences suggesting that the role of retroviruses and other potential means by which DNA can be modified on the fly tend to apply almost exclusively to the DNA of somatic cells rather than to the better protected and less accessible DNA of gamete cells. Generally epigenetic factors that can be shown to impact germ cell DNA do so in ways that either have unknown consequences, or, where the effects can be seen in the short term, they are disorder-producing, sometimes teratogenic, or lethal.
With all of the foregoing in mind, we will consider each of the foregoing systems one by one with special attention to their dependence on TNR type representations. Because each of the systems seems to presuppose the others, the task of introducing them is already difficult, but it is made even moreso by the fact that much of what has been claimed, and still is regarded by some as received wisdom, has already been shown to be false on the basis of empirical findings. Interestingly, the entire complex of inter-related immune systems begins from the premise that the self is to be protected from potential invasion or contamination by anything that is not either the self or identifiable as belonging in the neighborhood. This foundational premise of immune systems in general presupposes that every living organism with immune capacities, especially mammals and human beings, has a certain self-consciousness built into its cells right down to the level of its DNA.
It is intriguing to consider the possibility that self-consciousness, and perhaps consciousness its very self, consists of a certain resonance between the self cells of an entire body with all of its own organ systems. They seem to be tuned to a particular biochemical frequency shared by all those cells and organs that constitute the self. Theoreticians have variously tried to associate the “soul” and its consciousness—that ineffable whatever-it-is-ness that we call the “self” in modern talk—alternately with the whole body when it is awake, or especially with the brain or heart, or even with the gut, or possibly with the “personality” and its history, and so forth. However, what I want to suggest at this point is the possibility that the unifying character of consciousness, what is termed the “self”—as contrasted with “non-self”—resides within the very heart of our cells right down at the level of our initial DNA where the biologically viable and unique individual story of each of our respective selves begins.
For every mature human being capable of abstract thought, at some point there is a strange realization of self-awareness, self-consciousness, where the mundane ordinary sense of looking out toward an external world from within our body morphs into a representation of the fact that although we are in this body we cannot imagine how we got inside it, and yet, strangely, we can imagine what it would be like to be outside it as an observer. In fact, we find ourselves in such a relation to essentially all other human beings like ourselves. With such realizations come the great quandaries of existence. How did we get here? Where in the universe is the earth, anyway? And, how did the cosmos itself come to be? More significantly, how comes it that we are able to put these questions? In such a context, the question of what consciousness itself is seems to be intrinsically a problem of representation and of our capacity to produce at least some mundane true representations of the universe—apropos of the miracle referred to by Einstein [
73] and of the resonance referred to by Peirce [
72] and the profound quandary defined by Brier [
14].
From the cellular point of view, the major histocompatibility complex (MHC) which, because of its crucial role in defining self to the body’s immune systems, is also referred to as the human leukocyte antigen (HLA), actually involves several distinct levels of MHC/HLA. Only the red blood cells and the body’s noncellular components do not rely on MHC to identify and define their roles to themselves and to other cells. The MHC inside each cell is the gene-richest part of the DNA. In higher vertebrates including humans, the MHC enables the production of certain proteins that are displayed on the surface of cells as identifiers of self and as a kind of written record of the history and current status of the cell.
In addition to its self identifier which marks it as a legitimate part of the body, the cell may also display non-self proteins or foreign proteins from invading microbiota (viruses and bacteria) that show it to be diseased or injured. Because these proteins are examined by specialized immune cells which may then trigger the production of antibodies that can identify, capture, and eventually destroy foreign microbiota that are the source of foreign proteins, the proteins displayed on the surface of cells are referred to as antigens (antibody generators). If the cell is damaged beyond repair, the immune inquirer may also call for it to be dismantled and removed from the body.
Abstracting and generalizing across the many complex layers of the immune systems, the key to success in protecting and maintaining the health of the self, is for the cells of the immune systems to correctly identify the body’s own MHC proteins as self and to correctly recognize foreign, damaged, or non-self proteins as indicators of problems that need to be dealt with. If an MHC self-protein is validly reported by the cell to an immune cell inquirer, and if it is correctly taken as such by the immune cell, the result is a mundane exchange of valid TNRs between the cells. The construction of representations, however, is never completely without risk. As Peirce noted, chance is always a factor in present tense actions—and as Brier insists, we must acknowledge “chance as real” [
14,
15,
134]. There is genuine risk in mistaking a mere fiction for an established TNR. This gives rise to the possibility of error and of deliberate deception as succinctly pointed out by Einstein in 1941 [
135]. If misrepresentations accumulate sufficiently, as it seems they must by chance, they will eventually lead to dissolution and the death of the organism.
In individual cells the dissolution can come about gradually and naturally over the course of time resulting in what is known as apoptosis or it can come about in the context of mortal combat in conflicts of various sorts. In all instances, the success or failure of any given representation, all of which are more or less at risk in the present tense, depends ultimately on its truth or falsehood in the most mundane sense. In any individual body, truth is defined as agreement with the representations provided for by the organism’s defining DNA, with special focus on the MHC.
4.9.1. The Zero-Order Layer of Defenses: The Ecoment of the Gut
According to standard doctrine—e.g., Alberts,
et al., 1994 [
136]—the most basic level of the immune defense systems consists of a whole complex of interrelated systems that are called “innate” to distinguish them from those that appear to be “acquired” or “learned” and that are accordingly referred to as the “adaptive immune system.” However, it is somewhat misleading for a couple of reasons, to suppose that the so-called “innate” and “adaptive” systems are distinct in the manner the terms suggest.
First, it is evidently incorrect to suppose that all the various layers of the human immune systems are not provided for innately. The DNA—including its unfolding epigenetic expression and downstream consequences, however probabilistic and at risk they may be—nonetheless provides for that part of immunity which is referred to as “adaptive” just as it provides for the part that is referred to as “innate.” The whole of the multilayered system of immune systems is undoubtedly provided for at least in an abstract way, though not yet in concrete detail, with the successful initial union of sperm and egg.
Second, the presumption that what is commonly referred to as “the innate immune system” is the most basic level is apparently also incorrect. There is a still more basic level of defense that confers a large part of the normal human’s immunity to infection that precedes the entry of anything into the inside of the body itself—that is, into the blood, lymph, or bodily tissues beneath the epithelial layers that mark both the outside and the inside of the bodily icon.
Obviously, the skin and mucous membranes of the bodily orifices for the eyes, ears, nose, mouth, anus, urethra, and in females the vagina, provide an active line of defense against potentially harmful foreign substances and potential disease agents. They also provide necessary entry points for sensations of sight, hearing, smell, touch, taste, and sexual intercourse, as well as needed air, water, and food, and exit points for liquid and solid wastes. What is not so obvious is that material outside the inner linings of the body, including the linings of the digestive tube—the entire gut—is generally regarded as if it were still “outside” the body. The contents of the gut, for instance, according to gastroenterologists enter the body, by their reckoning, only by crossing through the lining of the gut into the blood, lymph, or other bodily tissues. Similarly, it can be argued that the gases in the lungs are not fully inside the body until they pass through the mucosal membranes into the blood and internal tissues.
Setting to one side the logical impreciseness of any such claims—an impreciseness that is partly unavoidable because the edges must be joined smoothly and the surfaces are not completely impermeable—it is nonetheless true that the borders of the gut in particular, especially the small and large intestines where much of the biochemical work of digestion is performed, are closely guarded. Also, it is in the human gut that we find what might well be termed the zero-order immune system of a healthy body. Vighi, Marcucci, Sensi, Di Cara, and Frati [
137] estimate that “almost 70% of the entire immune system” (p. 3) is contained within the gut—also see H. Miller,
et al. [
138]. No doubt this is one of the reasons why the United States National Institutes of Health has committed $115 million to study to microbes of the body and especially of the gut [
139]. Within the gut, there are, it is estimated, about 100 trillion biological microbes [
140].
The vast majority of these gut inhabitants—commonly referred to as “microflora” but better described by the more general term “microbiota” (both of which are in common use)—are not only not harmful but, on the contrary, help us to digest materials essential to the body and stand as a zero-order defense against unwelcome intruders. O’Hara and Shanahan [
132] have argued that the gut microbial system “has a collective metabolic activity equal to a virtual organ within an organ” (p. 688). It is evidently the case that there is considerable “cross-talk” between the microbiota of the gut and the host organism [
141]. Serino,
et al. point out that the communications between the microbiota of the gut and the host are evidently of vital importance to the host:
Over the past few years, it has been established that, in obesity, type I diabetes and Crohn's disease—to cite but a few—the intestinal microflora play a pathophysiological role and can induce, transfer or prevent the outcome of such conditions. A few of the molecular vectors responsible for this regulatory role have been determined. Some are related to control of the immune, vascular, endocrine and nervous systems located in the intestines. However, more important is the fact that the intestinal microflora-to-host relationship is bidirectional [
142], with evidence of an impact of the host genome on the intestinal microbiome (especially see p. 162).
That is, it appears that the genetic specifications of the host determine in large measure the shaping and composition of the microbiota of the gut. Taking into consideration the extent to which the integumentary system of the host’s body (the
O of the
SπO formula) is united through the tubes in the body’s orifices with the inner mucosal linings of the gut and with all the organ systems that have contact with the external world, perhaps the interactions of the host genome with the ecoment of the gut logically suggests a great deal more about the unplumbed depths of functions of the genome. It seems likely that the genome represents much more about the extended neighborhood, the whole ecoment, of the organism than has been previously realized—a possibility more in keeping with the logical requirements of ethology as suggested by Brier in 2008 [
14].
Elsewhere, Brier [
16] has referred to the “transmitters in the brain and the immune system” which he suggests involve “interactions” that may be “very important for the establishment of the autopoeitic system of the second order” (p. 64). With reference to the gut microbiota, Round and Mazmanian [
143] tantalizingly go so far as to suggest that the “disturbances in the bacterial microbiota” that evidently “result in dysregulation of adaptive immune cells raises the possibility that the mammalian immune system, which seems to be designed to control micro-organisms, is in fact controlled by micro-organisms” (p. 313). Surely, they go too far in supposing that a collection of many disparate, often conflicting micro-organisms, could do more than the coordinated system of self-cells, organs, and living tissues in the body of any individual mammal, much less any self-conscious ordinary human being, but there is no doubt that there is considerable cross-talk between the microbiota in the gut of a human being and the immune systems of that same person [
141,
144].
Among the recently discovered peculiarities of the interactions between the immune systems and the microbiota of the gut is evidence of changing roles of high level immune cells known as T regulators (Treg cells). These cells are part of what is thought of as the highest layer of the four global layers of the whole immune system—namely, the “adaptive immune system” (see
section 4.9.4 below)—and they are involved in communications that inform and shape the development of the immune systems of infants as they mature toward adulthood. The research of Grindebacke,
et al. suggests that the infant’s Treg cells first focus attention on antigens that are appearing in the gut but increasingly attend to ones that originate outside the gut as the infant matures beyond its first 18 months of life [
110]. What is interesting about the current research into such regulatory communications is that they show the various layers within the immune systems to be integrated in ways that are only just beginning to come to light. This will be even clearer as we proceed through the other global layers of the immune systems.
4.9.2. The “Innate Immune System”
Turning next to the somewhat misleadingly so-called “innate immune system” (ISS), we find again, a richly layered system of systems closely integrated with the other systems that theoretically, at least, reside above and below this major layer. Theoretically, the ISS ranks above the layer of immune functions of the gut microbiota and beneath the so-called “complement system” (CS)—the latter being in its turn is beneath the layer known as the “adaptive immune system” (AIS). However, because of the communications between the various hypothetically distinct layers, the boundaries between them are fuzzy to say the least.
The distinction between layers is more a matter of how far beneath the outer surfaces of the body the immune cells of the system in question penetrate. The microbiota of the gut, for instance, do not normally penetrate the lining of the gut to enter more deeply into the bloodstream or lymphatic ducts. However, the immune cells of the higher systems generally have access to the bloodstream and/or the lymphatic ducts—often referred to as the “humoral” immune system (with fast moving components that have access to all the major layers of the whole system of systems).
Another traditional basis for distinguishing the ISS and the AIS layers was the fact that only the latter layer can produce immunoglobulins (also known as antibodies) enabling defenses targeted against particular classes of disease agents. That distinction still stands. However, along that line it was commonly supposed that only AIS cells were able to acquire the information necessary to describe the identity of particular pathogen—as in a labeled wanted poster—in order to display it and thus communicate the nature of a particular attacker, an antigen, to other cells especially the attack cells of the immune systems. Immune cells that can display the identity of particular pathogens in this way are called antigen presenting cells (APCs). Until recently, it was supposed that only AIS cells could perform this function, but it is now known that all immune cells can probably serve as APCs.
All of this, however, goes to show that the foundational premise of the ISS—and, indeed the immune systems in general—is that there is a real difference between self cells and non-self cells. As already noted this distinction depends initially on the self defining major histocompatibility complex (MHC) which is also known as human leukocyte antigen (HLA). The most general type of this self-defining ID and health report is displayed by every cell in the body excepting those that lack a nucleus, e.g., red blood cells and blood platelets which are so generally necessary to the survival of every other self cell that to require identification from them would be like requiring yourself to show yourself your passport to verify your identity before every breath, every swallow of liquid, and every bite of food. It would be superfluous to the point of defeating the self nurturing functions of the red blood cells and platelets for them to display MHC to themselves and other self cells. It seems a little odd to call the self-defining MHC an “antigen” because it is—provided that it is a well-formed TNR faithfully representing the identity of the self—the foundational basis for defining non-self entities that can reasonably be referred to as “anti-gens”—or valid generators of antibodies. The self-defining MHC certainly is not normally intended to generate antibodies against itself or the cells and organs that it defines as self. That said, the self-defining MHC is commonly expressed on the outside of a cell. This common, self-MHC, then, mutually affirms itself to neighboring cells and to inquiring leukocytes somewhat in the manner of a license or passport photograph. All healthy self-cells, excepting red blood cells and thrombocytes for the reasons already noted, can identify themselves with MHC and ones that can also present information concerning the identity of pathogens are considered APCs. It has turned out that the APC function is much more general than formerly supposed.
Among the authorities checking for the identity of cells or standing guard against foreign biological matter inside the body are the immune cells of the ISS layer. With the exception of cells formed during fetal development and before the individual has developed bones, all the immune cells originate in the bone marrow from what are termed hematopoeitic stem cells. In addition to the basic workhorses of the bloodstream which consist first and foremost of the exceedingly numerous red blood cells—with about 200 billion being produced each day [
145]—and secondarily of megakaryocytes that produce platelets and the various thrombocytes making up key components of blood plasma enabling blood clotting and the healing of injured tissues, the hematopoeitic stem cells of the bone marrow also produce various white blood cells—leukocytes of various types which are the main agents of the ISS, and, also, the AIS (discussed below in
145section 4.9.4). The hematopoietic stem cells that produce leukocytes, according to current understanding, are differentiated in the bone marrow into myeloid and lymphoid progenitors.
The leukocytes of the ISS can be differentiated into three main groups. First, there are five major kinds highly mobile ISS leukocytes, ones that move rapidly in the bloodstream and that can move out into the lymph and other tissues. Among the ISS mobile leukocytes are four that are believed to have a myeloid progenitor —neutrophils, eosinophils, basophils, and monocytes—and there is just one type, the natural killer (NK), that has a lymphoid progenitor. All of them are involved in dealing with perceived pathogens although the monocytes are also specialized for clean up and waste disposal. The NK cells are large lymphocytes critical for defense against cancerous cells. Normally they constitute about 10% to 15% of blood lymphocytes and were called “natural killers” because they were originally thought to be licensed to kill, like a kind of James Bond of the ISS, any cell not displaying self-identifying MHC. However, it has more recently been learned that NK cells are regulated by special “killer Ig[immunoglobulin]-like receptors” determining activation thresholds for these cells [
146]. Again, contrary to prior thinking, NK cells though not yet shown to be fully functional APCs as the other ISS leukocytes evidently are (see discussion below), nevertheless, have turned out to be at least weak APCs [
147].
Next, turning to the myeloid leukocytes, the most plentiful and most common type consists of the neutrophils whose main function is to consume and kill, by a process much like eating and digesting, invading bacteria or fungi. They do this by capturing one or several of the perceived foreign microbiota, engulfing them and then decomposing them through an internal digestive process that also results in the death of the leukocyte after only a few of the invaders have been dispatched. An accumulation of neutrophils is what appears as pus at the site of an infection. Neutrophils collectively account for about 50% to 70% of the total number of leukocytes circulating in a normal adult’s blood at any given time. They are like police on patrol exercising lethal force against certain kinds of invaders that cannot identify themselves as “self” by displaying self-defining MHC.
In spite of the fact that traditional teaching in immunology suggests that neutrophils are generic attack units, it is evident that they sometimes attack certain kinds of invaders while ignoring others. In
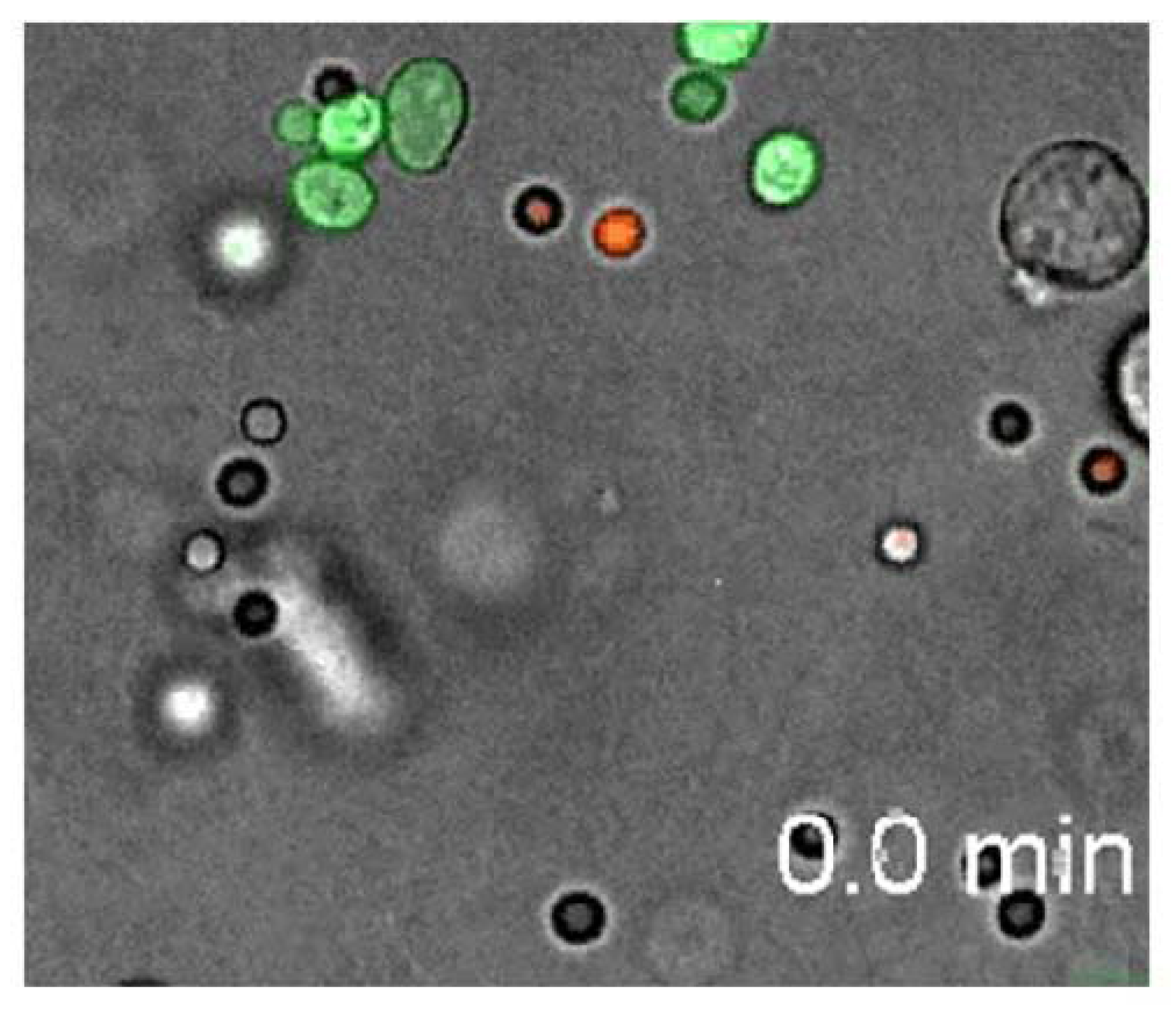
Figure 9, from Behnsen
et al. [
148], a neutrophil can be seen attacking and disposing of several Candida spores (the green spheres) while it ignores various Conidia (the white spheres). Does the neutrophil know the difference or is it constrained in a purely physical way as suggested by Behnsen
et al.? It seems that neutrophils may be informed concerning what sort of bad guy to hunt down at the moment. The sort of representation required, in any case, for a deliberate search (hunt) is a hypostatic abstraction of a Candida spore that enables one to be recognized when found—perhaps as distinct from a Conidium. At that point the hypostatic icon of a Candida is matched with a perceived icon and the
SπO requirements are fulfilled—a TNR is achieved. It can easily be shown that more is required for a specific search, but certainly no less will work. Whether neutrophils are capable of such a search and destroy mission may be in doubt, but there is no doubt that adaptive immune cells can perform such missions.
In fact, the notion that neutrophils are naive generic immune cells was shown to be false by Cassatella,
et al. in 1995 when they discovered that neutrophils themselves play a regulatory role in “adaptive” immunity [
149]. This result has generally been confirmed and accepted [
150,
151]. Also, it has been shown that the premature apoptosis of large numbers of neutrophils is indicative of toxic stress [
152] showing, again, the close coordination of detoxification and immune functions.
Figure 9.
A neutrophil (the fast moving phagocytic leukocyte) can be seen attacking and disposing of several Canadian spores (the green spheres) while it ignores various Conidia (white spheres)−from Behrens, et al. (2007, Video S15).
Figure 9.
A neutrophil (the fast moving phagocytic leukocyte) can be seen attacking and disposing of several Canadian spores (the green spheres) while it ignores various Conidia (white spheres)−from Behrens, et al. (2007, Video S15).
A less numerous type of myeloid mobile leukocytes, eosinophils, are particularly useful in attacking and warding off various parasites (e.g., helminths such as the common pinworms or hookworms often found in the intestines of mammals and human beings). Eosinophils account for about 1% to 6% of the total supply of leukocytes in a healthy adult bloodstream. They release toxic cytokines that are harmful to invading parasites but also to the host tissues. While they were formerly thought to be exclusively part of the traditionally described ISS it is now known that eosinophils also play an immunoregulatory role linking them both to the adaptive and innate layers of the immune systems [
153,
154,
155].
A third type of myeloid mobile leukocyte, one traditionally associated with the ISS, is the basophil. It is a type accounting for only about .01 to .3 percent of leukocytes and as might be expected basophils also have a more specialized function. As granulocytes circulating in the blood they seem to be involved as signaling cells to notify other immune cells of an infection site or injury. They also are involved in healing and especially in producing cytokines involved in producing new blood vessels in damaged tissues [
156]. Again, the implication arises that basophils are not only involved in the ISS but also in the AIS.
The three main mobile myeloid types of leukocytes (at the ISS level) seem to function as police with different specialties, and ranks, but they appear to be equipped to handle routine common problems rather than the sort of major attack from outside the body that would require a more highly trained professional military response (at the AIS level). The common neutrophils are like cops on patrol who can use lethal force in dealing with common domestic problems—bacteria and fungi. For bigger domestic problems, the eosinophils are like the SWAT team specialists on the police force that are called out to deal with difficult large problems—parasites such as the helminths that contain huge loads of bacteria, viruses, and toxins that need to be managed carefully and preferably encouraged to stay in their mobile homes and exit the body along with other wastes. The basophils are evidently a less common sort of detective specialized in gathering information, sounding the alarm when a problem occurs to call for assistance at the site of an injury or infection, and, in the meantime, to establish a perimeter and initiate damage control measures.
While all three types of mobile leukocytes are associated with the ISS—a layer just above the zero-order rank of the gut microbiota—all of the mobile police in the ISS seem to be able to communicate with and to present problems about specific kinds of bad guys (antigens) to specialists in the better equipped and more highly trained professional military—the folks in the AIS. At any rate, neutrophils [
157], eosinophils [
153,
154,
155], and basophils as well [
158], can function as antigen presenting cells (APCs)—that is, immune cells that report the existence of a particular perceived threat along with something like a labeled description of the suspects to be rounded up. Even when these mobile police on patrol are dying or already dead, any one of the three is capable of sending out an alarm to call for help. Bird points out that eosinophils and basophils can do so [
159], and Yang,
et al. [
160] describe how it works with neutrophils:
alarmins released by neutrophil degranulation and/or death can link neutrophils to dendritic cells by promoting their recruitment and activation, resulting in the augmentation of innate and adaptive immune responses (p. 531).
The fourth class of myeloid mobile leukocytes consists of the highly versatile monocytes. Their specialty seems to be in cleaning up the scene of a crime—disposing of the bodies and debris left behind and putting things back in order. However, they too are capable of functioning as APCs [
161] and can exercise lethal force. They are also able to move from the bloodstream into bodily tissues where they muscle up and specialize into either of two types of tissue resident outpost guard cells, macrophages or dendritic cells. Both are larger than the monocytes in the bloodstream. The macrophages still specialize in debris disposal and the clean up of tissues by phagocytosis. The other type of tissue resident cell that a monocyte may become is known as a dendritic cell because of its dendrite-like appendages that it uses to grab hold of foreign particles or antigens—e.g., see
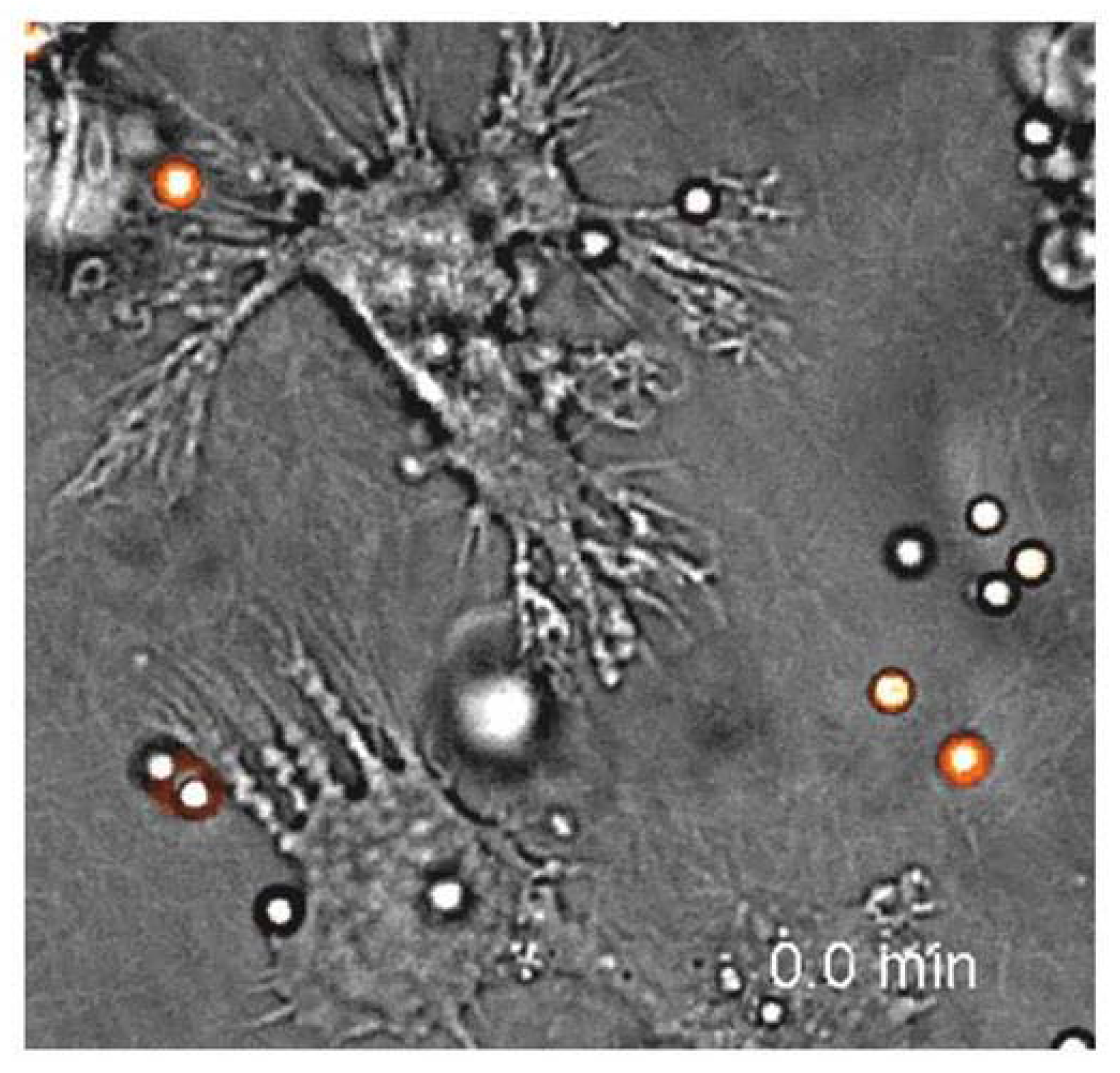
Figure 10 in which a dendritic cell can be observed to drag a conidium about 9 μm. All of these cells are capable of killing functions and of serving as APCs [
157,
162].
Interestingly, in 2008 Hume argued that the distinction between macrophages and dendritic cells with respect to their APC capabilities has been exaggerated. He insisted that the traditionally distinguished types should be collapsed into a single category and that macrophages are just as able to serve APC functions as dendritic cells [
162]. With that idea in mind it seems reasonable to wonder if perhaps other current distinctions in levels and types of leukocytes and other immune cells may also need eventually to be reconsidered. For instance, are the basophil granulocytes in the bloodstream really distinct in origin and type from the category of leukocytes known as mastocytes (which are also called as “mast cells”)? Whatever the case may be, the mast cells are very much like basophils in their form but like the so-called “fixed” location monocytes the mast cells also migrate into tissues where they specialize either as resident guards in mucosal membranes or as distinct types of specialized outposts in various other tissues [
163,
164]. Mast cells, it seems, have a lineage from the bone marrow distinct from that of the basophils which they greatly resemble. From the bloodstream, the mast cells migrate out to mucosal linings and bodily tissues where they take up residence as on location guards. Like basophils, they are able to regulate levels of inflammation and to mediate tissue repair [
156].
Figure 10.
A dendritic cell with extened appendages drags in a conidium, but does not ingest it−from Behrens, et al. (2007, Video S8).
Figure 10.
A dendritic cell with extened appendages drags in a conidium, but does not ingest it−from Behrens, et al. (2007, Video S8).
Summing up the functions of the ISS, they can be described as (1) the discovery of non-self agents and debris—a symbolic (S) function; (2) the neutralization of non-self elements—an active indexical (π) function; and (3) disposal of non-self agents and debris—an iconic (O) function.
4.9.3. The “Complement System”
What is known as the “complement system” (CS) is a complex of several similar biochemical cascades enabling the mass destruction and disposal of non-self enemy combatants (pathogens) and/or the debris of non-self proteins. The basic components of the system are said to be produced by the ISS. The basic complement components consist of a substantial supply of proteins produced in the liver and circulating in the bloodstream that may be compared to a kind of complex weapon system consisting of multiple parts waiting for a signal to go active. The yet to be activated proteins circulating in the blood constitute something like a ready weapons cache awaiting a sequence of interlinked activating signals ultimately enabling the launching of ever so many tiny attack complexes that can be guided to pathogenic targets of opportunity where they form up on the surface of the marked target cell, perforate its outer surface and kill it. Before the complement protein system is activated, the targets, hopefully consisting of pathogenic non-self cells—rather than healthy self cells—are marked for destruction and removal in a variety of ways by the ISS or the AIS (see
section 4.9.4 below).
The targeting of pathogens by the AIS involves their being marked by immunoglobulins (antibodies) IgG, IgM, or IgA bound to their surface. The marking by the ISS, by contrast, is less specific and involves some as yet to be determined means of chemical marking merely described as “pathogen-associated molecular patterns” (PAMPS) which supposedly enables CS targeting of common pathogens. Recent research by Paul-Clark,
et al., however, suggest that oxidative damage may be sufficient in some cases at least to initiate CS targeting of cells as pathogens [
165].
When the CS works well, the elements (antigens and wastes) to be disposed of are correctly identified and marked for destruction and removal after which the CS layer is triggered by some threshold of cytokine activity released by inflamed tissues, ISS, and AIS immune cells. When that happens, the biochemical cascade of the CS layer kicks into action and rapidly cleans house. It does so by bursting the cell membranes of identified pathogens (lysis), by enhancing the phagocytosis of perforated pathogens, and by speeding up the clearance of debris through the spleen and liver. In view of the fact that about 5% of the total blood serum may be recruited into the activity of the CS, the amplification of the capacity of the body to rid itself of disease producing pathogens and to clean up the debris of the collateral damage is enormous. The positive effects of the CS, when it works well—that is when the vast majority of targeted elements are pathogens and collateral damage to self cells is minimal—the impact may appear as something akin to overnight healing. The fever and symptoms may suddenly abate because of the impact of the CS response. On the other hand, because of the global impact of the CS on much of the body and on multiple organ systems, there is a concomitant risk of catastrophic results if things go wrong.
In worst case scenarios, the result can be anaphylactic shock followed by the sudden death of the organism. In less severe but still acute instances, violent seizures may result. From these examples it is easy to infer that combinations of difficulties and gradations of severity can fall anywhere on a scale from acute life-threatening multiple system failures to milder cases that may develop gradually to the point of being hardly detectible. By contrast, if the CS is working correctly, non-self elements are correctly identified, marked for destruction and removal, and subsequently are swept up in an extremely efficient way. The analogy that comes to mind is that of a killing hail storm followed by a tornado that sweeps through a war zone—the ballistic hail selectively targets and kills enemy combatants while leaving the defending immune cells and the civilian population virtually untouched. Right behind the hail of guided bullets comes a tornado-like vacuum sweeping up the debris of the war and the dead combatants.
4.9.4. The “Adaptive Immune System”
By now it is already clear that there are “adaptive” components throughout all of the foregoing immune system layers. Even the zero-order level of the ecoment of the gut microbiota involves cross-talk with higher levels suggesting that the gut microbiota also constitute, to some extent, an “adaptive” system. Also, as already pointed out, all of the cells of the ISS are capable of performing as APCs—a function formerly thought to be limited to the AIS.
Nevertheless, it remains true that the AIS is distinguished by its unique capacity to produce specific antibodies tuned to particular disease agents. In this respect, it stands above the microbiota of the gut, the ISS, and the CS layer. Together in communication with the other layers of the immune systems the lymphocyte cells of the AIS can target particular pathogens, track them down, immobilize, capture, and interrogate them to the point of dismantling them piece by piece, remember the results of prior engagements, and, thus, prepare in advance for possible repeat attacks.
From the lymphoid progenitors produced by the hematopoetic stem cells of the bone marrow, in addition to the large granular lymphocytes known as NK cells of the ISS which were discussed above, there are lymphoid dendritic cells [
166]. These lymphoid cells, like myeloid dendritic cells, have the capacity to capture pathogens and deliver them to the lymph nodes. They both also have the capacity to phagocytose pathogens and cut their proteins into small pieces. Later, these fragments can be presented as antigen defining components on the surface of the dendritic cell. These cells function something like intelligence specialists teaching other cells about potentially harmful antigens—thus placing themselves among the so-called “professional” antigen-presenting cells—showing other cells in the AIS the differences between self-MHC (commonly referred to as Class I) and particular pathogen MHCs (Class II). There are evidently differences in the functions of lymphoid dendritic cells in contrast with their myeloid counterparts, but they are not yet well understood [
144,
167].
In addition to the lymphoid dendritic cells, the AIS also has small lymphocytes that originate in the bone marrow. These small lymphocytes are subsequently differentiated into B cells destined to develop antibodies against pathogenic non-self invaders and T cells that will go to the thymus for something like an advanced series of specialized military degrees in intelligence gathering and communication, command and control, and, some of the T cells will be specially equipped for hand-to-hand fighting against particular pathogens of which the T cells will be informed by APCs of the “professional” kind—those that enable the distinction between MHC-I, somewhat misleadingly called “self antigen,” and MHC-II antigens that define perceived pathogens. It almost goes without saying that every step along the way, the representations that are provided to and by immune cells must conform to the requirements of TNRs, or disease, dissolution, and death of the organism will be the inevitable outcome.
The B cells, emanating from the bone marrow, must pass a series of do or die tests in before they can graduate to the level of B cells judged to be competent to produce antibodies. If the B cell happens to fail any of the tests along the way, it will either never mature, or it will dismantle itself by apoptosis. Upon its graduation as a B cell competent to produce antibodies, if it turns out that the antibody which it specifies happens to match up with the MHC-I that defines self-cells, that B cell will either become inactive or succumb to apoptosis. According to the current understanding, millions of different B cells with distinct antibodies are produced to circulate in the blood seeking a pathogen matching the description of the one that the particular B cell is able to produce and already has on hand. When a match is found, the B cell is authorized to engulf the enemy combatant, interrogate it to the point of digesting it, and then display the enemy’s identity on its own (the B cell’s) surface.
At this point the B cell is something like a well equipped military industrialist in a fast moving vehicle (a B cell in the bloodstream or lymphatic system) who has captured a prisoner and has extracted potentially vital information about an invasion possibly underway. The industrialist is able and willing to immediately set up one or more factories to produce the weapons (antibodies) that will enable the capture and destruction of the pathogen that has been identified, but needs confirmation that the information obtained is valid and the authorization to begin mass production. If and when the validation comes, the B cell will first clone itself multiple times, and then, its clones will convert themselves into plasma cells each of which engages in the manufacture of the needed antibody weapons. The confirmation and authorization to initiate the cloning of B cells and the production of plasma cell factories will come from T cells. A small fraction of the B cell clones will be transformed into long-lived B memory cells that will remain in the bloodstream conducting surveillance in case of a re-emergence of the same antigen at any time in the future. Some of these memory cells are known to last for the entire life of the host organism.
Consider next the T cells that are sent to the thymus for their training and education. It seems that the major classes of these specialists, all of which are in the military intelligence business, are critically dependent on gathering and confirming information from APCs. In the do or die training that these cells must undergo in the thymus, only about 2% of those that start the course will complete it. The rest will generally undergo inactivation and/or apoptosis. Initially, some of the graduating T cells will be immediately equipped with lethal cytotoxic capability involving a particular glycoprotein cluster of differentiation (CD8) that appears on its surface and binds to the MHC-I molecule [
168]. The same glycoprotein is also found in NK cells within the ISS. Here, in the AIS, as soon as an APC informs the cytotoxic T-CD8+ cell about the precise antigen—one defined in an MHC-II molecule—it is ready to be authorized to go weapons hot and start hunting for the determined pathogen.
Meantime, another class of T cell, known in the immunology literature as a T helper (Th) cell—which is a specialist in intelligence gathering and communication with other immune system cells—will also be informed by a professional APC concerning an antigen that has been identified by that APC as an immediate threat. The informed Th cell—at this stage at Th0 cell, then—though it will not engage in direct combat with pathogenic cells, will proceed first to authorize the rapid deployment of informed killer T cells looking for the particular antigen of which the Th0 cell has been informed by the professional APC. The Th0 cell may next follow one of three branching routes: it may be biochemically transformed into a Th1 cell that will inform and encourage macrophages to attack the particular antigen the Th1 cell learned of from the professional APC; or the Th1 cell may be biochemically transformed into a memory T cell that will guard against possible future attacks by the same antigen.
Alternatively, the Th0 cell may be biochemically transformed into a Th2 cell that proceeds to authorize B cells to clone themselves and thus to initiate mass production of antibodies against the particular antigen the Th2 cell is able to confirm as an actual threat. After B cells clone themselves and produce plasma cells that manufacture antibodies against a particular antigen, the progress of the ongoing conflict will be monitored and regulated by interactions between B cells and Th1 cells. The result of the early Th0 activity in informing and authorizing killer T cells and macrophages to attack a particular pathogen will be to enhance the probability that the informed and active killer T cells and macrophages—provided that the MHC-II report is a TNR and that the killer T can correctly distinguish the MHC-I that defines the self cells via TNRs reported to the killer T from self-cells—may be able to stop the pathogen before the infective agent has the chance to proliferate.
Finally, a few observations must be made about the remarkably simple and yet effective and vast differentiation of types of distinct immunoglobulins (particular antibodies). It is also important to include the caveat that in doing so, we have barely penetrated the inner workings of the four global layers of the immense system of immune systems. It should be noted as well that it is estimated that the number of distinct antibodies that can be produced by the human AIS exceeds 10 billion [
169,
170]—an observation by Susumu Tonegawa leading to a Nobel Prize awarded to him in 1987 [
171]. At that time, the press release concerning Tonegawa’s findings effusively claimed:
We are accordingly well prepared for an encounter with any possible antigen. It is likely that normally only a minor part of the antibody variance will ever be put into usage [
172].
In fact, in 2009, Glanville,
et al. showed that the combinatory possibilities for just one of the immunoglobulin types—namely, IgM, which is the third most plentiful of accounting for about 10% of the total antibodies—exceed 3.5 X 10
10 [
169]. There are, in addition to this particular type of antibody, four other types: IgG, the general and most common type and the only one believed to cross the placental barrier; IgD, which seems to be involved in decommissioning immune cells; IgA, found mainly in mucosal linings; and the relatively rare, IgE, which is associated with immune responses that can occur far from the evident site where the antigen impinges.
Although it is commonly supposed that the immune systems evolved gradually and in independent bits and pieces—on the theory that their known components seem to operate independently to a high degree—there are in fact, many indications that the systems are in communication with each other. The processes of manufacturing distinct types of cells in the bone marrow, their subsequent differentiation through various filters and developmental tests, along with the intricate systems of communication suggest that communications are occurring from the ecoment in the gut all the way to the highest levels of the AIS. Some of the means by which this communication occurs are understood, but it is plausible to suppose that a great deal more remains to be discovered.