Interactive Effects of Biosynthesized Nanocomposites and Their Antimicrobial and Cytotoxic Potentials
Abstract
1. Introduction
2. Materials and Methods
2.1. Biosynthesis of Silver (AgNPs), Zinc Oxide (ZnONPs) and Titanium Dioxide (TiO2NPs) Nanoparticles
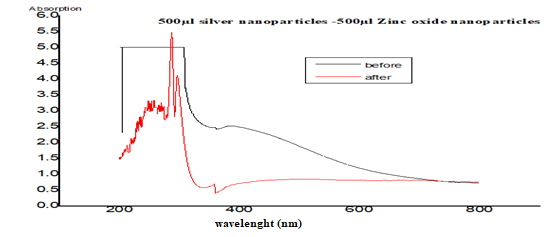
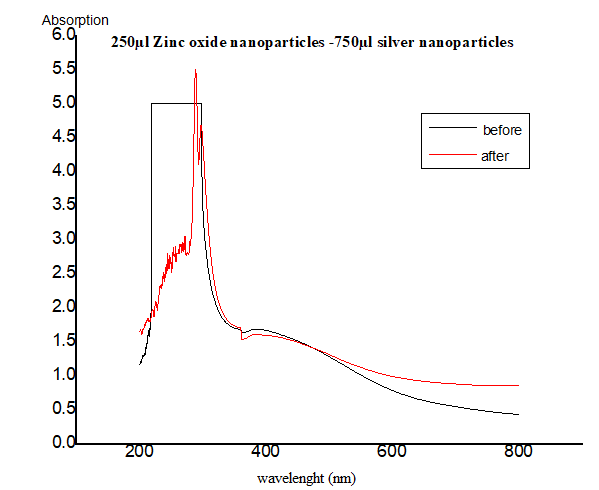
2.2. Preparation of Nanocomposites
2.3. Characterization of Biosynthesized AgNPs, ZnONPs, TiO2NPs and Nanocomposites
2.4. Evaluation of Antibacterial Activity
2.5. Determination of the Antitumor Activity
2.6. The statistical Analysis
3. Results
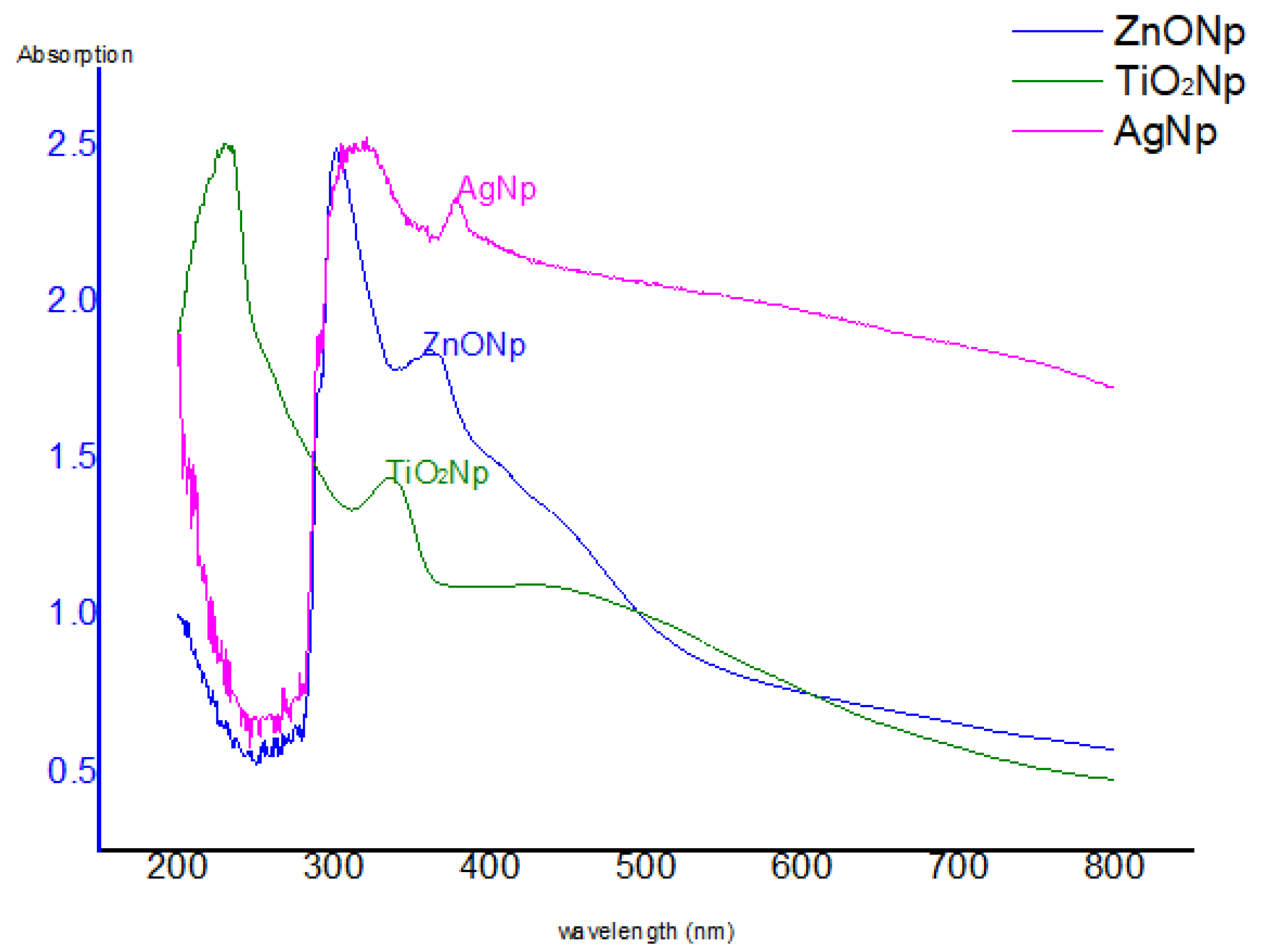
3.1. The UV–Vis Spectra
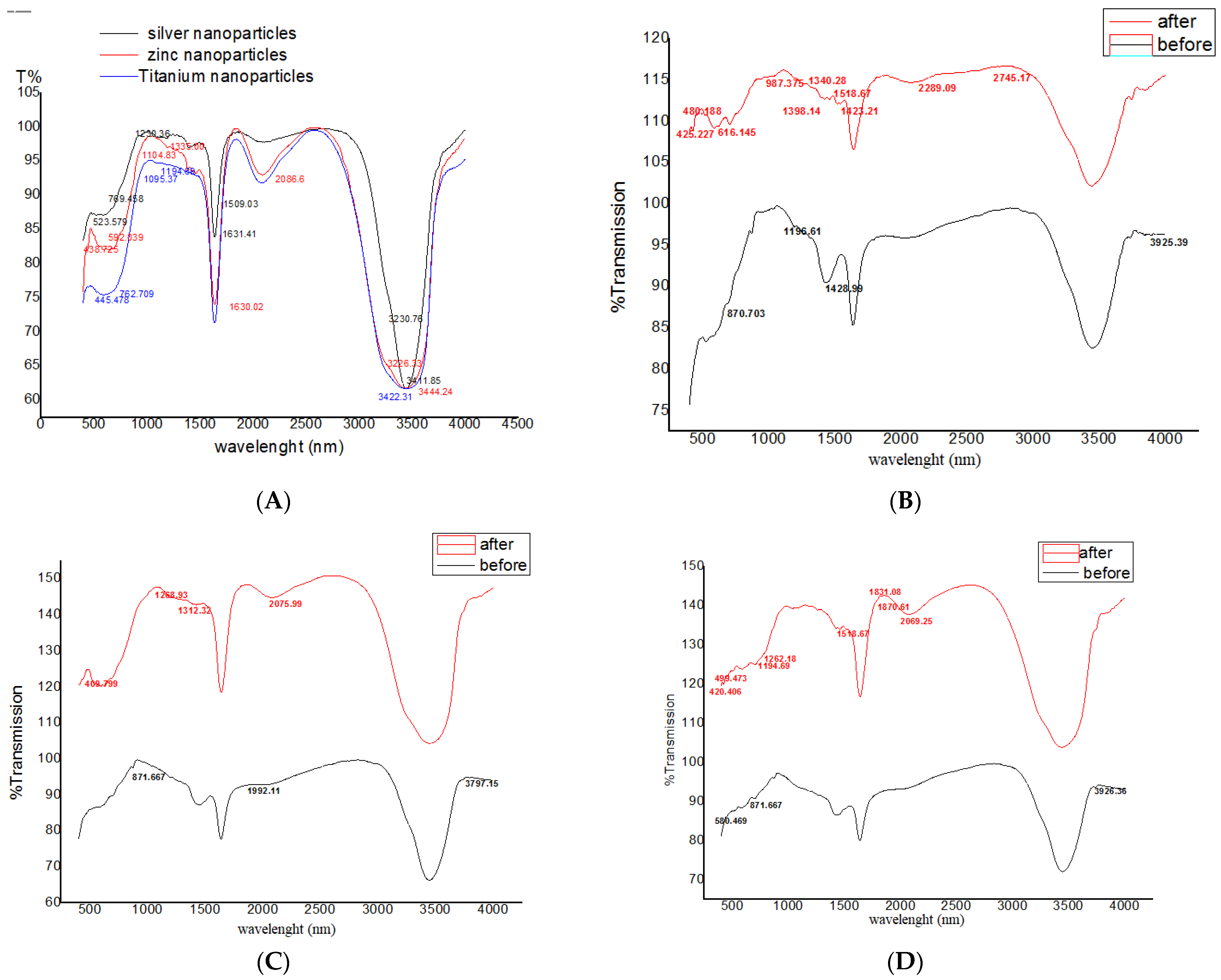
3.2. FTIR Spectrum of Nanoparticles
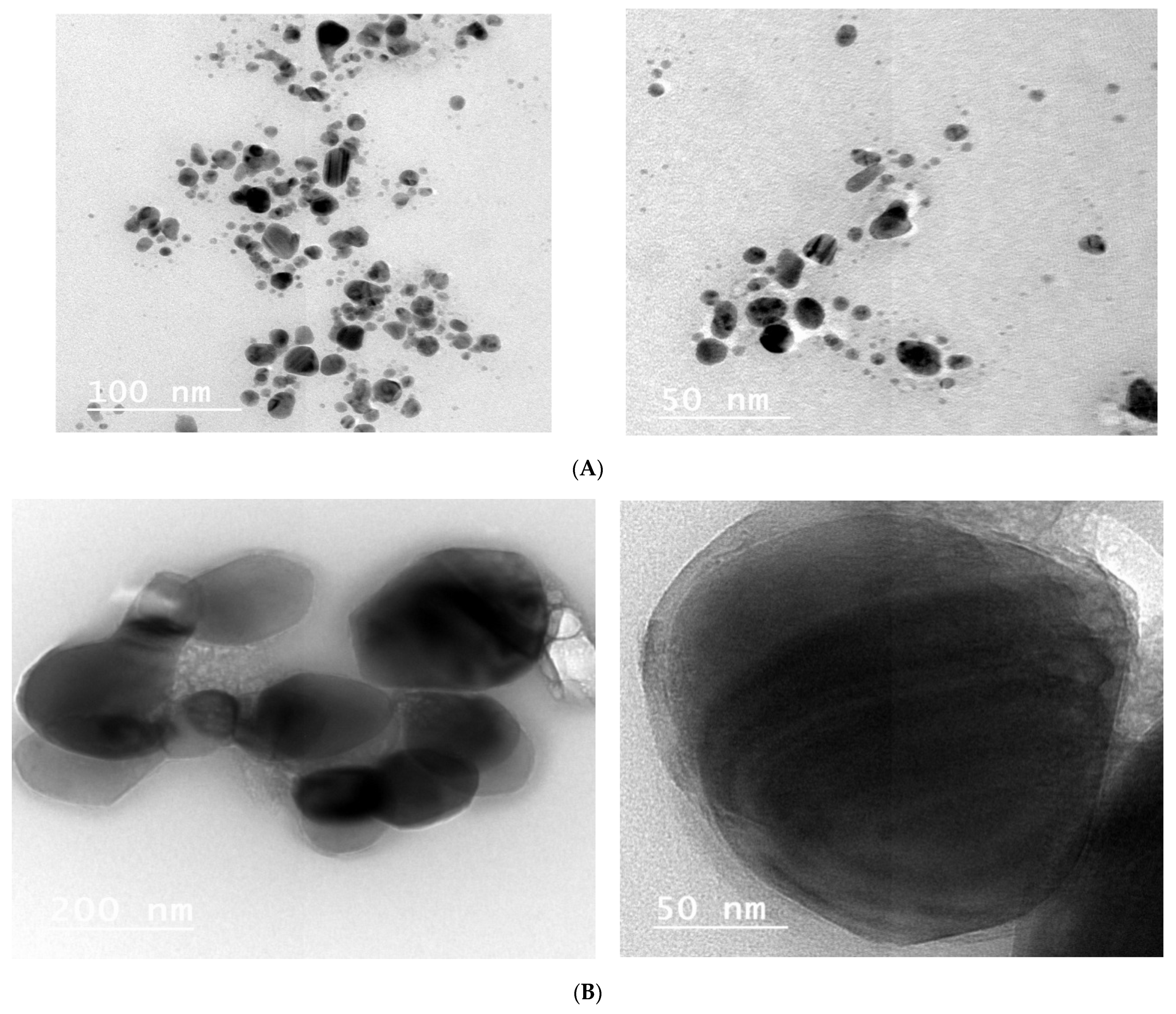
3.3. Analysis of Transmission Electron Microscope Images
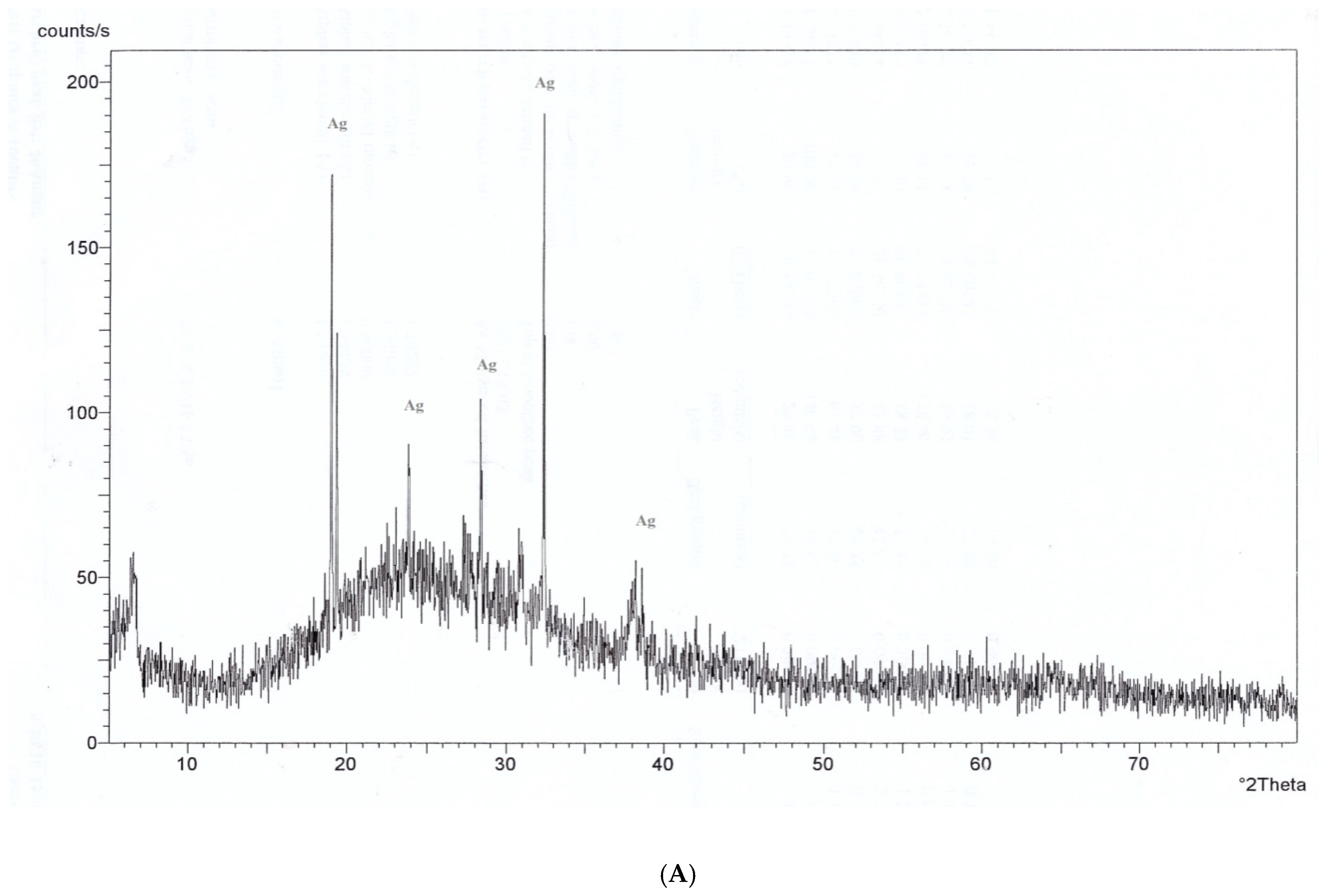
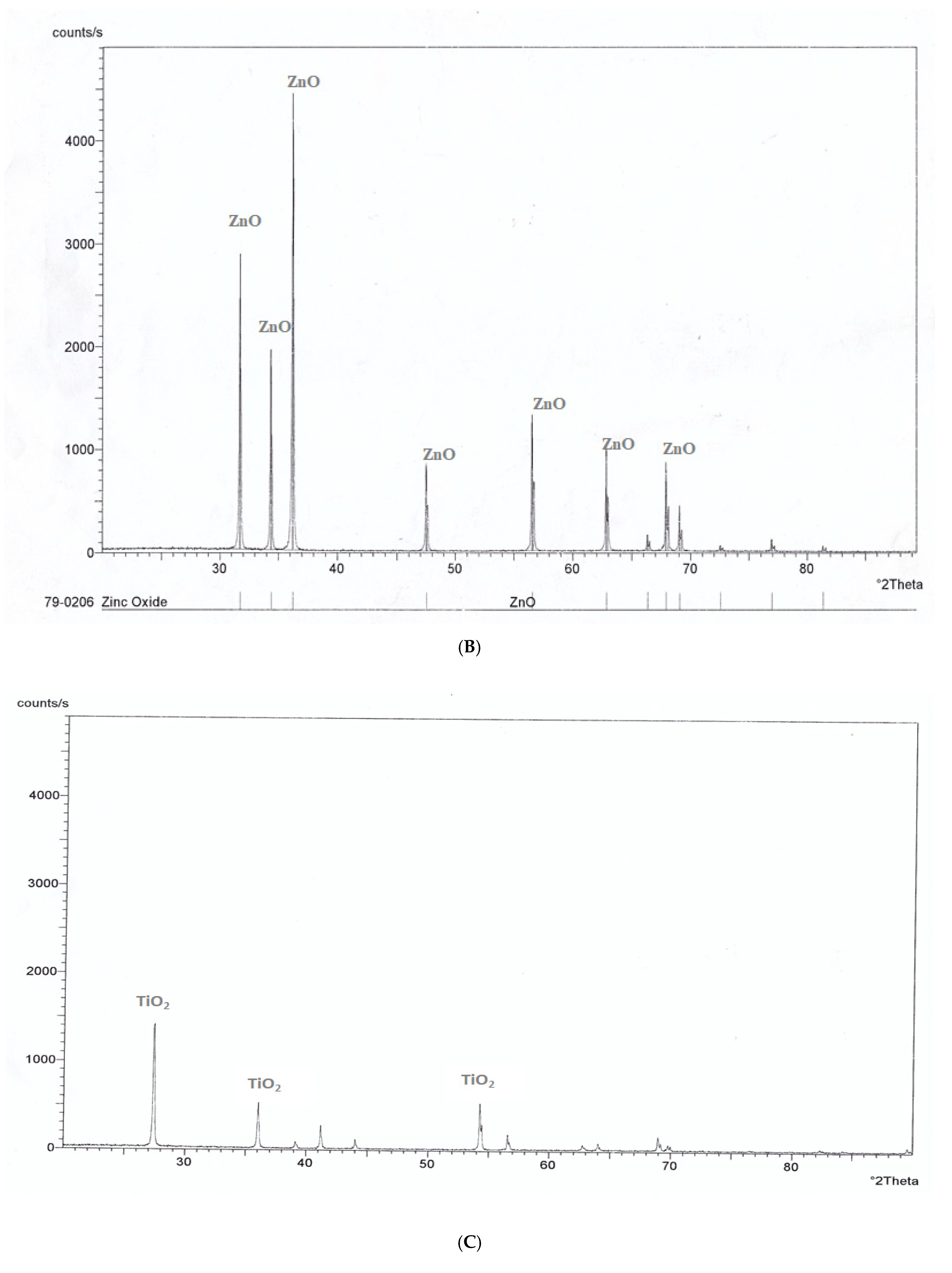
3.4. X-Ray Diffraction (XRD) Pattern Analysis
3.5. Application Studies of the Produced Nanoparticles
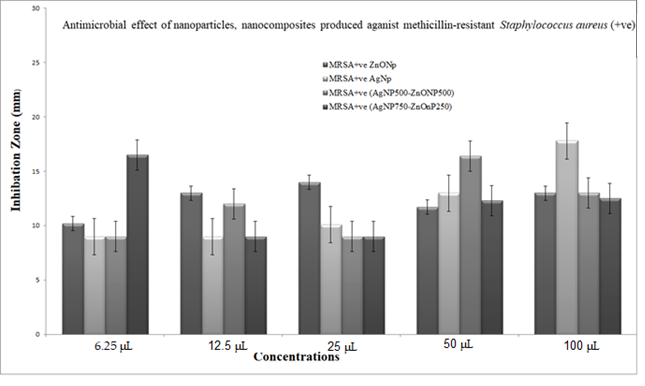
3.5.1. Comparative Toxicity Study of the Biosynthesized Nanoparticles and Nanocomposite on the Human Bacterial Diseases
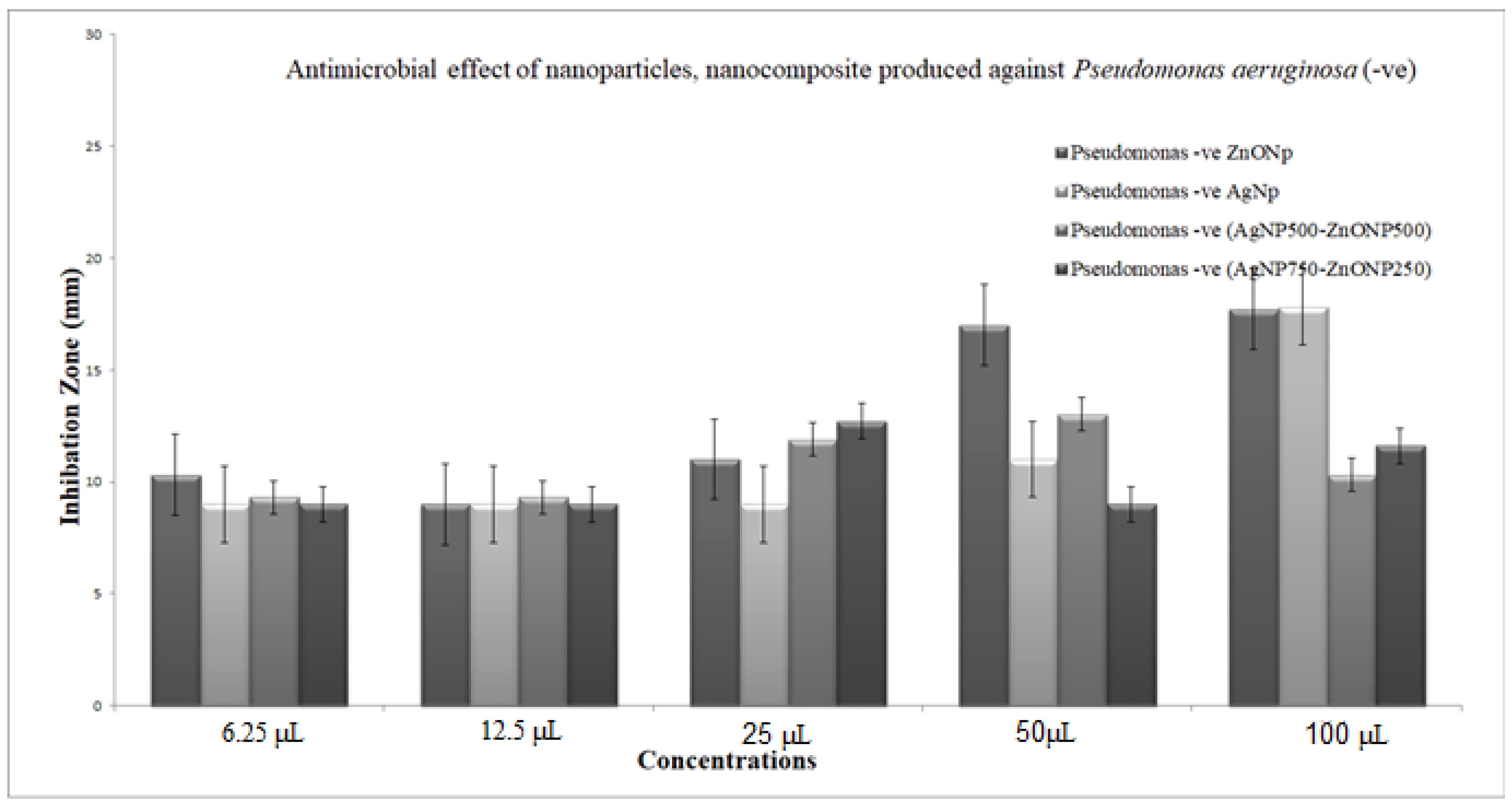
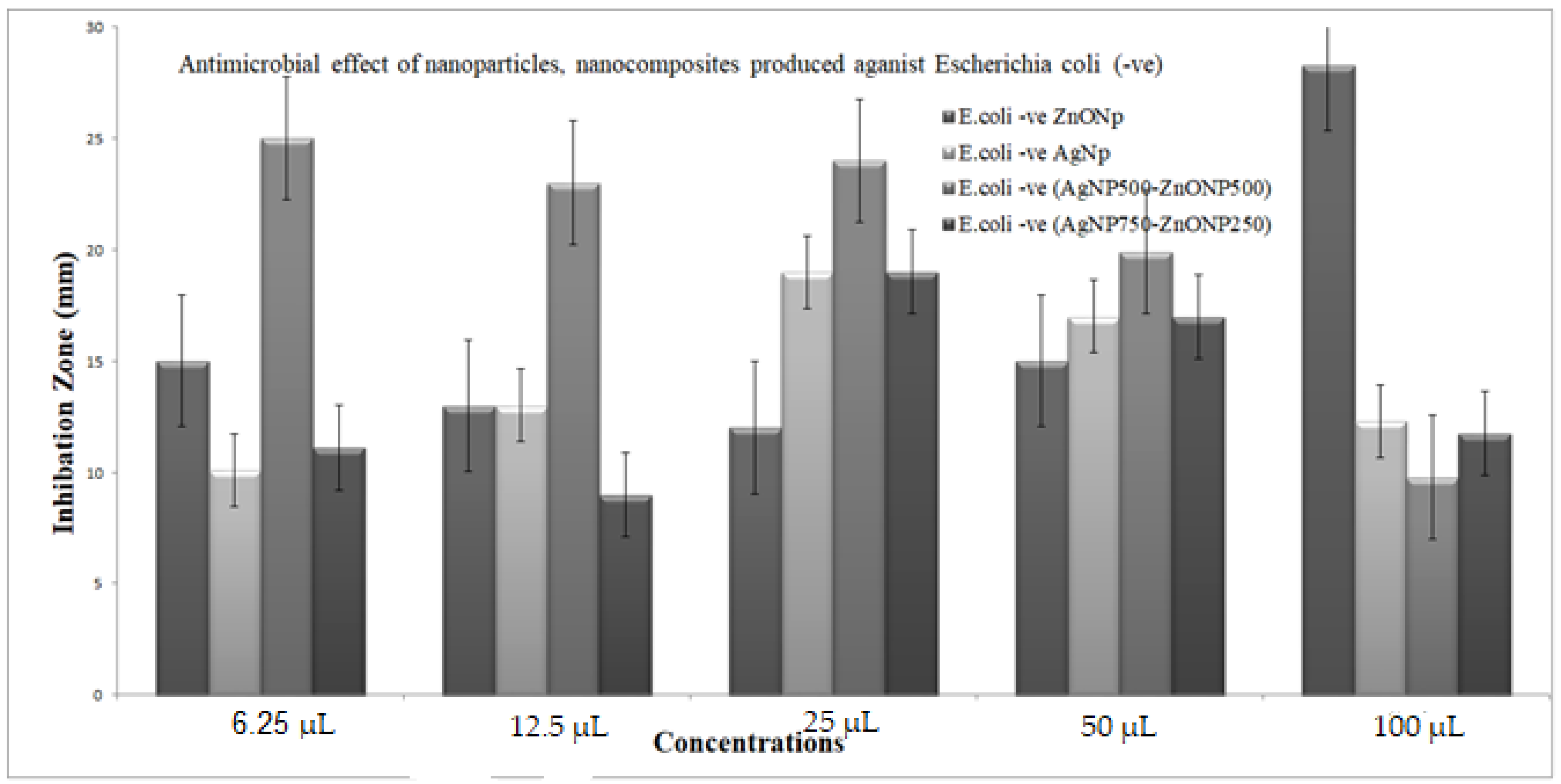
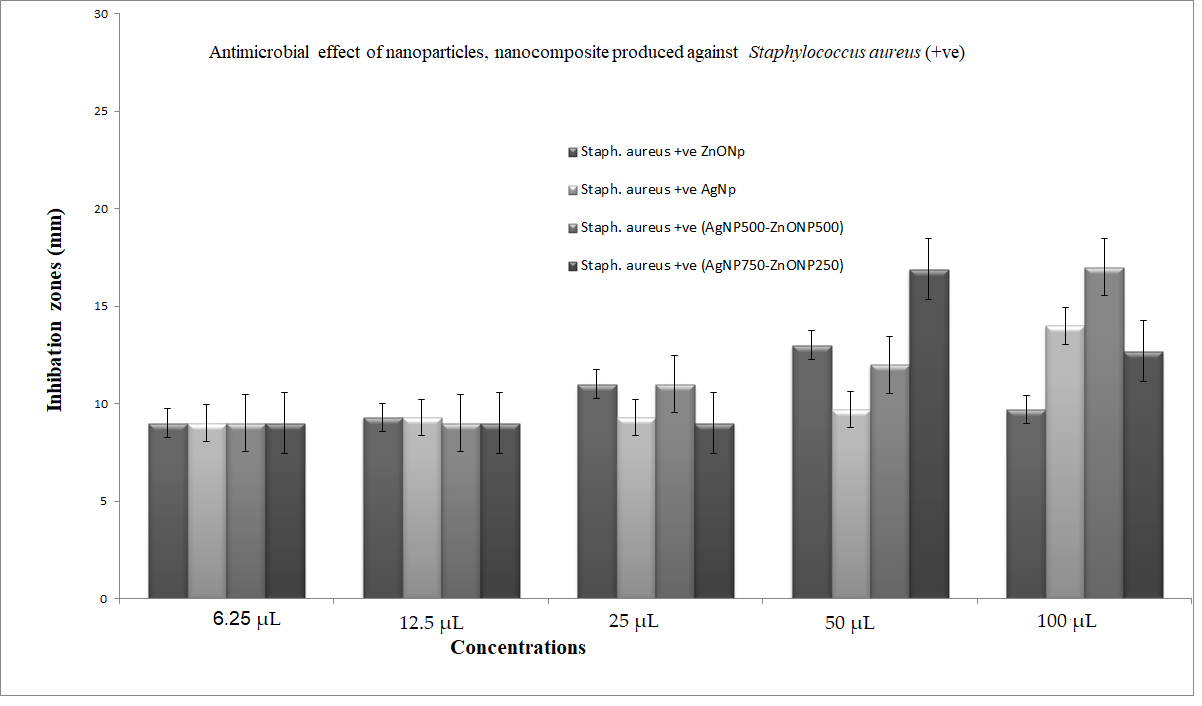
3.5.2. Antimicrobial Effect of Nanoparticles and Nanocomposite
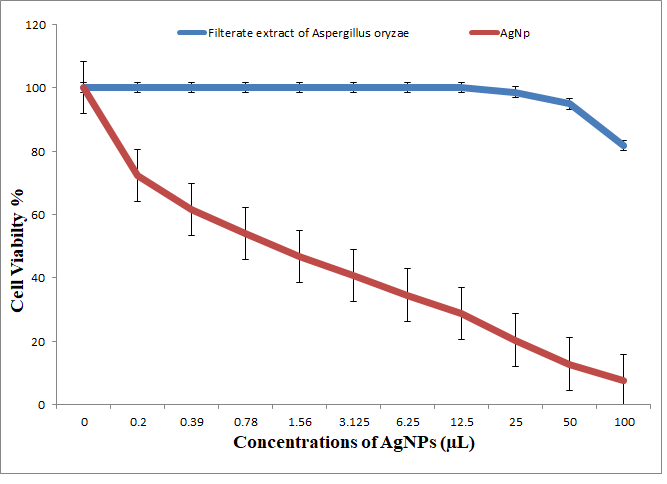
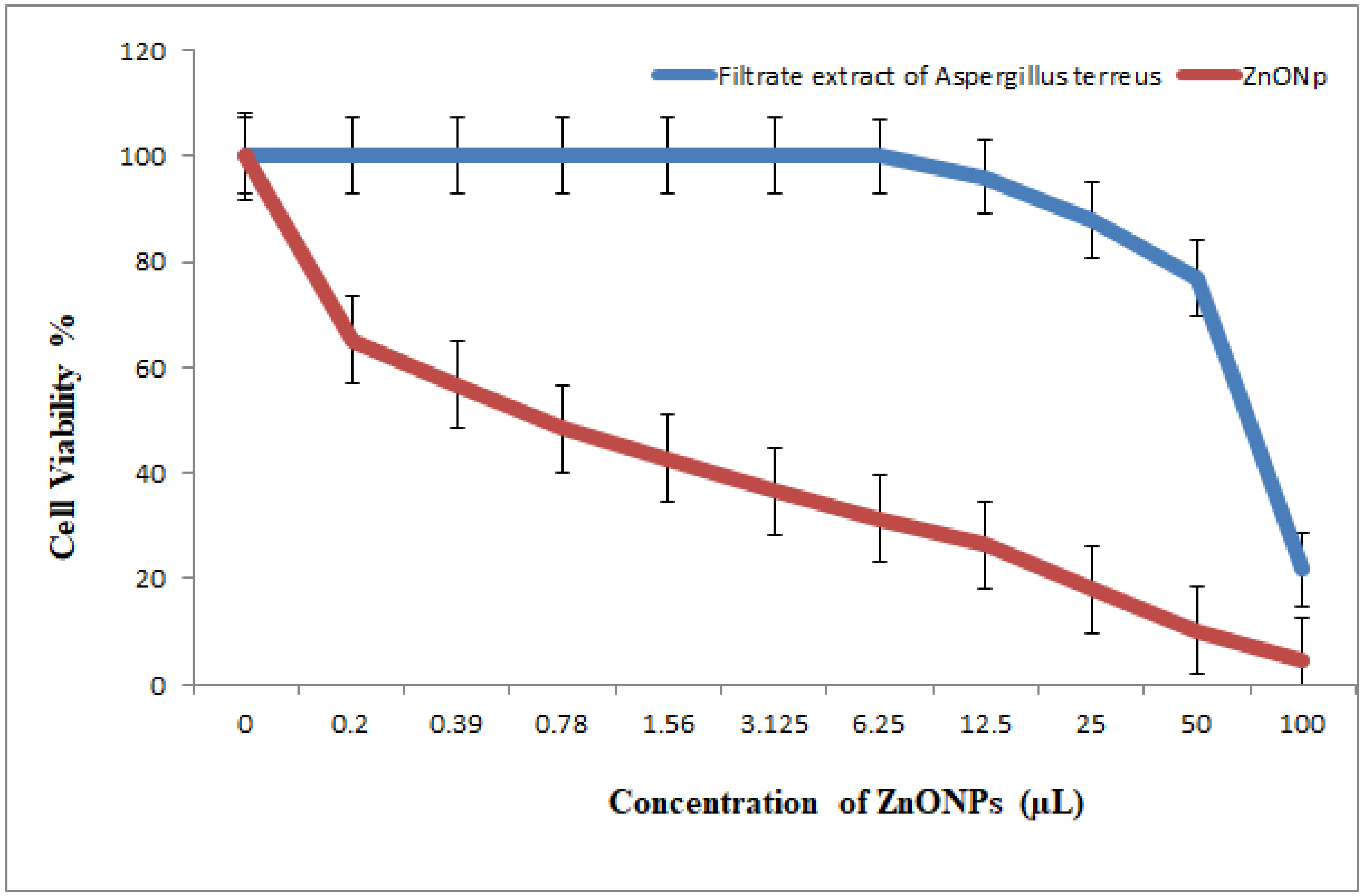
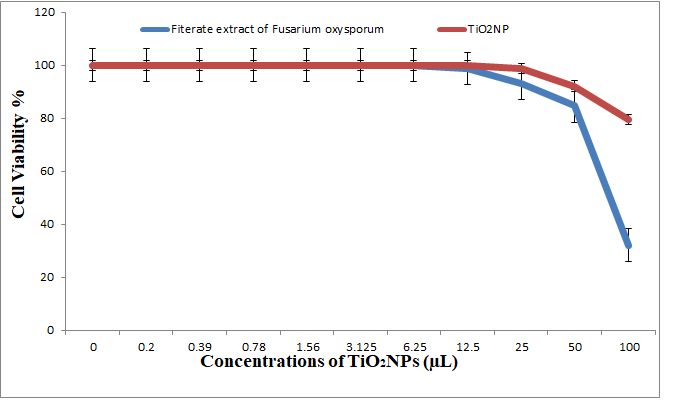
3.5.3. Antitumor Activity of Nanoparticles and Nanocomposites
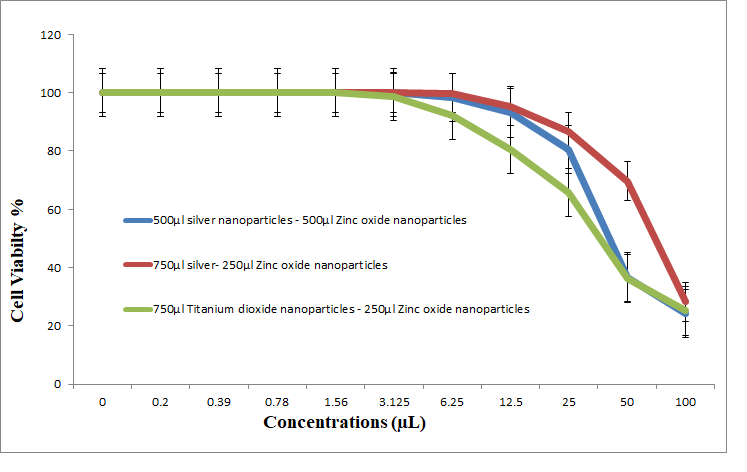
3.5.4. Antitumor Activity of Nanocomposites
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kalpana, V.N.; Devi Rajeswari, V. A Review on Green Synthesis, Biomedical Applications, and Toxicity Studies of ZnO NPs. Bioinorg. Chem. Appl. 2018, 2018, 3569758. [Google Scholar] [CrossRef]
- Fang, X.; Wang, Y.; Wang, Z.; Jiang, Z.; Dong, M. Microorganism Assisted Synthesized Nanoparticles for Catalytic Applications. Energies 2019, 12, 190. [Google Scholar] [CrossRef]
- Verma, P.; Maheshwari, S.K. Applications of Silver Nanoparticles in Diverse Sectors. Int. J. Nano Dimens. 2019, 10, 18–36. [Google Scholar]
- Ghareib, M.M.; Attia, M.; Awad, N.M.; Matter, M.Z.; El-Kahky, D. Screening of Fusariumspp Resistance to Silver Ions for Ability to Synthesize Silver Nanoparticles from Egyptian Soils. Glob. Adv. Res. J. 2015, 4, 366–382. [Google Scholar]
- Roy, A.; Bulut, O.; Some, S.; Mandal, A.; Yilmaz, M. Green Synthesis of Silver Nanoparticles: Biomole-cule-Nanoparticle Organizations Targeting Antimicrobial Activity. RSC Adv. 2018, 9, 2673–2702. [Google Scholar] [CrossRef]
- Kvítek, L.; Panáček, A.; Soukupová, J.; Kolář, M.; Večeřová, R.; Prucek, R.; Holecová, M.; Zbořil, R. Effect of Surfactants and Polymers on Stability and Antibacterial Activity of Silver Nanoparticles (NPs). J. Phys. Chem. C 2008, 112, 5825–5834. [Google Scholar] [CrossRef]
- Gaidau, C.; Petica, A.; Ignat, M.; Iordache, O.; Ditu, L.-M.; Ionescu, M. Enhanced Photocatalysts based on Ag-TiO2 and Ag-N-TiO2 Nanoparticles for Multifunctional Leather Surface Coating. Open Chem. 2016, 14, 383–392. [Google Scholar] [CrossRef]
- Khalandi, B.; Asadi, N.; Milani, M.; Davaran, S.; Abadi, A.J.; Abasi, E.; Akbarzadeh, A. A Review on Potential Role of Silver Nanoparticles and Possible Mechanisms of their Actions on Bacteria. Drug Res. 2017, 67, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Qing, Y.A.; Cheng, L.; Ruiyan, L.; Liu, G.; Zhang, Y.; Tang, X.; Wang, J.; Liu, H.; Qin, Y. Potential Antibacterial Mechanism of Silver Nanoparticles and the Optimization of Orthopedic Implants by Advanced Modification Technologies. Int. J. Nanomed. 2018, 13, 3311–3327. [Google Scholar] [CrossRef]
- Fernando, S.; Gunasekara, T.; Holton, J. Antimicrobial Nanoparticles: Applications and Mechanisms of Action. Sri Lankan J. Infect. Dis. 2018, 8, 2–11. [Google Scholar] [CrossRef]
- Wang, Y.-W.; Cao, A.; Jiang, Y.; Zhang, X.; Liu, J.-H.; Liu, Y.; Wang, H. Superior Antibacterial Activity of Zinc Oxide/Graphene Oxide Composites Originating from High Zinc Concentration Localized around Bacteria. ACS Appl. Mater. Interfaces 2014, 6, 2791–2798. [Google Scholar] [CrossRef]
- Ancona, A.; Dumontel, B.; Garino, N.; Demarco, B.; Chatzitheodoridou, D.; Fazzini, W.; Engelke, H.; Cauda, V. Lipid-Coated Zinc Oxide Nanoparticles as Innovative ROS-Generators for Photodynamic Therapy in Cancer Cells. Nanomaterials 2018, 8, 143. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Vaseeharan, B.; Sudhakaran, R.; Jeyakandan, J.; Ramasamy, P.; Sonawane, A.; Padhi, A.; Velusamy, P.; Anbu, P.; Faggio, C. Bioinspired Zinc Oxide Nanoparticles using Lycopersiconesculentum for Antimicrobial and Anticancer Applications. J. Clust. Sci. 2019, 30, 1465–1479. [Google Scholar] [CrossRef]
- El-kahky, D.; Attia, M.; Easa, S.; Awad, N.; Helmy, E. Biosynthesized of Zinc Oxide Nanoparticles Using Aspergillusterreus and their Application as Antitumor and Antimicrobial Activity. Glob. Adv. Res. J. Agric. Sci. 2019, 8, 90–100. [Google Scholar]
- Dayakar, T.; VenkateswaraRao, K.; Bikshalu, K.; Rajendar, V.; Park, S.-H. Novel Synthesis and Structural Analysis of Zinc Oxide Nanoparticles for the Non Enzymatic Glucose Biosensor. Mater. Sci. Eng. C 2017, 75, 1472–1479. [Google Scholar] [CrossRef] [PubMed]
- Punitha, V.N.; Vijayakumar, S.; Sakthive, B.; Praseetha, P.K. Protection of Neuronal Cell Lines, Antimicrobial and Photocatalytic Behaviours of Eco-Friendly TiO2 Nanoparticles. J. Environ. Chem. Eng. 2020, 8, 104343. [Google Scholar]
- Shih, Y.; Lin, C. Effect of particle size of titanium dioxide nanoparticleaggregates on the degradation of one azo dye. Environ. Sci. Pollut. Res. 2012, 19, 1652–1658. [Google Scholar] [CrossRef]
- Akhtar, M.S.; Swamy, M. Biosynthesis and Characterization of Silver Nanoparticles from Methanol Leaf Extract of Cassia didymobotyra and Assessment of Their Antioxidant and Antibacterial Activities. J. Nanosci. Nanotechnol. 2015, 15, 9818–9823. [Google Scholar] [CrossRef]
- Kim, J.S.; Kuk, E.; Yu, K.N.; Kim, J.H.; Park, S.J.; Lee, H.J. Antimicrobial effects of silver nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2007, 3, 95–101. [Google Scholar] [CrossRef]
- Qiong, L.J.; Li, X.Z.; Dai, Z.; Lu, Z.; Yang, H.; Chen, R. Enhanced antibacterial activity and mechanism studies of Ag/Bi2O3 nanocomposites. Adv. Powder Technol. 2018, 29, 2082–2090. [Google Scholar]
- Gunalan, S.; Rajeswari, S.; Venckatesh, R. Green synthesizedzinc oxide nanoparticles against bacterial and fungal pathogens. Prog. Nat. Sci. Mater. Int. 2012, 22, 693–700. [Google Scholar] [CrossRef]
- Gupta, J.; Mohapatra, J.; Bahadur, D. Visible light drivenmesaporous Ag-embedded ZnO nanocomposites: Reactive oxygen species enhanced photocatalysis, bacterial inhibition and photodynamic therapy. Dalton Trans. 2017, 46, 685–696. [Google Scholar] [CrossRef] [PubMed]
- Youssef, A.M.; El-Nahrawy, A.M.; Hammad, A.B.A. Sol-gel synthesisand characterizations of hybrid chitosan-PEG/calcium silicate nanocomposite modified with ZnO-NPs and (E102) for optical and antibacterial applications. Int. J. Biol. Macromol. 2017, 97, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Amin, S.A.; Pazouki, M.; Hosseinnia, A. Synthesis of TiO2–Ag Nanocomposite with Sol–Gel Method and Investigation of Its Antibacterial Activity against E. coli. Powder Technol. 2009, 196, 241–245. [Google Scholar] [CrossRef]
- Brook, L.A.; Evans, P.; Foster, H.A.; Pemble, M.E.; Steele, A.; Sheel, D.W.; Yates, H.M. Highly Bioactive Silver and Silver/Titania Composite Films Grown by Chemical Vapour Deposition. J. Photochem. Photobiol. A 2007, 187, 53–63. [Google Scholar] [CrossRef]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.; Kim, T.; Kim, J. A Mechanistic Study of the Antibacterial Effect of Silver Ions on Escherichia coli and Staphylococcus aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Endo Kimura, M.; Janczarek, M.; Bielan, Z.; Zhang, D.; Wang, K.; Markowska-Szczupak, A.; Kowalska, E. Photocatalytic and Antimicrobial Properties of Ag2O/TiO2 Heterojunction. ChemEngineering 2019, 3, 3. [Google Scholar] [CrossRef]
- Zhang, H.; Chen, B.; Jiang, H.; Wang, C.; Wang, H.; Wang, X. A strategy for ZnO Nanorod Mediated Multi-Mode Cancer Treatment. Biomaterials 2011, 32, 1906–1914. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Liu, Z.; Liu, Y.; Fang, P.; Dai, Y. Enhanced Adsorption and Photocatalytic Degradation of High-Concentration Methylene Blue on Ag2O-Modified TiO2-Based Nanosheet. Chem. Eng. J. 2013, 221, 283–291. [Google Scholar] [CrossRef]
- Akhavan, O. Lasting Antibacterial Activities of Ag–TiO2/Ag/a-TiO2 Nanocomposite thin Film Photocatalysts under Solar Light Irradiation. J. Colloid Interface Sci. 2009, 336, 117–124. [Google Scholar] [CrossRef]
- Dawson, A.; Kamat, P.V. Semiconductor−Metal Nanocomposites. Photoinduced Fusion and Photocatalysis of Gold-Capped TiO2 (TiO2/Gold) Nanoparticles. J. Phys. Chem. B 2001, 105, 960–966. [Google Scholar] [CrossRef]
- El-Kahky, D. Improvement of the Antimicrobial Properties of Nanoparticles and Application in Water Purification. Ph.D. Thesis, Ain Shams University, Cairo, Egypt, 2019. [Google Scholar]
- Christine, O.D. Modified Czapek Dox Agar (Dehydrated). Saboutaudia 1962, 1, 214–219. [Google Scholar]
- Bottoni, P.; Scatena, R. The Role of CA 125 as Tumor Marker: Biochemical and Clinical Aspects. In Advances in Cancer Biomarkers: From Biochemistry to Clinic for a Critical Revision; Scatena, R., Ed.; Springer: Dordrecht, The Netherlands, 2015; pp. 229–244. [Google Scholar]
- Ahmad, A.; Mukherjee, P.; Senapati, S.; Mandal, D.; Khan, M.I.; Kumar, R.; Sastry, M. Extracellular Biosynthesis of Silver Nanoparticles Using the Fungus Fusariumoxysporum. Colloids Surf. B 2003, 28, 313–318. [Google Scholar] [CrossRef]
- Ahmad, A.; Senapati, S.; Khan, M.I.; Kumar, R.; Sastry, M. Extracellular Biosynthesis of Monodisperse Gold Nanoparticles by a Novel ExtremophilicActinomycete, Thermomonospora sp. Langmuir 2003, 19, 3550–3553. [Google Scholar] [CrossRef]
- Ahmad, A.; Senapati, S.; Khan, M.I.; Kumar, R.; Ramani, R.; Srinivas, V.; Sastry, M. Intracellular Synthesis of Gold Nanoparticles by a Novel AlkalotolerantActinomycete, Rhodococcus Species. Nanotechnology 2003, 14, 824–828. [Google Scholar] [CrossRef]
- Krumm, S. WINFIT 1.0—A Computer Program for X-Ray Diffraction Line Profile Analysis. Acta Univ. Carol. Geol. 1994, 38, 253–261. [Google Scholar]
- Williams, D.; Carter, C.B. Transmission Electron Microscopy: A Textbook for Materials Science; Plenum Press: New York, NY, USA, 1996. [Google Scholar]
- Jeevan, R.; Cromwell, D.A.; Trivella, M.; Lawrence, G.; Kearins, O.; Pereira, J.; Sheppard, C.; Caddy, C.; van der Meulen, J. Reoperation Rates after Breast Conserving Surgery for Breast Cancer among Women in England: Retrospective Study of Hospital Episode Statistics. BMJ 2012, 345, e4505. [Google Scholar] [CrossRef]
- Saotome, K.; Morita, H.; Umeda, M. Cytotoxicity Test with Simplified Crystal Violet Staining Method using Microtitre Plates and Its Application to Injection Drugs. Toxicol. In Vitro 1989, 3, 317–321. [Google Scholar] [CrossRef]
- Shivananda, C.S.; Asha, S.; Madhukumar, R.; Satish, S.; Narayana, B.; Byrappa, K.; Wang, Y.; Sangappa, Y. Biosynthesis of Colloidal Silver Nanoparticles: Their Characterization and Potential Antibacterial Activity. Macromol. Res. 2016, 24, 684–690. [Google Scholar] [CrossRef]
- Rajan, A.; Cherian, E.; Gurunathan, D.B. Biosynthesis of Zinc Oxide Nanoparticles Using Aspergillusfumigatus JCF and Its Antibacterial Activity. Int. J. Mod. Sci. Technol. 2016, 1, 52–57. [Google Scholar]
- Kirthi, A.V.; Rahuman, A.A.; Rajakumar, G.; Marimuthu, S.; Santhoshkumar, T.; Jayaseelan, C.; Elango, G.; Zahir, A.A.; Kamaraj, C.; Bagavan, A. Biosynthesis of Titanium Dioxide Nanoparticles Using Bacterium Bacillus subtilis. Mater. Lett. 2011, 65, 2745–2747. [Google Scholar] [CrossRef]
- Sun, B.; Reale, T.; Schroeder, S.; Pettey, M.; Smith, R. On the Accuracy of Vaisala RS41 versus RS92 Upper-Air Temperature Observations. J. Atmos. Ocean. Technol. 2019, 36, 635–653. [Google Scholar] [CrossRef]
- Yanari, S.; Bovey, F.A. Interpretation of the Ultraviolet Spectral Changes of Proteins. J. Biol. Chem. 1960, 235, 2818–2826. [Google Scholar] [CrossRef]
- Nir, S. Polarizability Calculations on Water, Hydrogen, Oxygen, and Carbon Dioxide. J. Chem. Phys. 1973, 59, 3341–3355. [Google Scholar] [CrossRef]
- Bregadze, V.; Melikishvili, Z.; Giorgadze, T.; Monaselidze, J.; Jaliashvili, Z.; Khuskivadze, T. Point Defects in Double Helix Induced by Interaction of Silver Nanoparticles with DNA. Russ. Chem. Bull. 2012, 62, 1914–1918. [Google Scholar]
- Jyoti, K.; Baunthiyal, M.; Singh, A. Characterization of Silver Nanoparticles Synthesized Using Urticadioica Linn. Leaves and their Synergistic Effects with Antibiotics. J. Radiat. Res. Appl. Sci. 2016, 9, 217–227. [Google Scholar] [CrossRef]
- Ahlam, A.A.; Shaniba, V.S.; Jayasree, P.R.; Manish Kumar, P.R. Spondiaspinnata (L.f.) Kurz Leaf Extract Derived Zinc Oxide Nanoparticles Induce Dual Modes of Apoptotic-Necrotic Death in HCT 116 and K562 Cells. Biol. Trace Elem. Res. 2020. [Google Scholar] [CrossRef]
- Abdel-Raouf, N.; Al-Enazi, N.; Ibraheem, I.; Alharbi, R.; Alkhulaifi, M. Bactericidal Efficacy of Ag and Au Nanoparticles Synthesized by the Marine Alga Laurenciacatarinensis. Int. J. Pharm. Res. Allied Sci. 2017, 2017, 213–226. [Google Scholar]
- Harish Kumar, K.; Savalgi, V. Microbial Synthesis of Zinc Nanoparticles Using Fungus Isolated from Rhizosphere Soil. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 2359–2364. [Google Scholar]
- Tang, J.; Fu, X.; Ou, Q.; Gao, K.; Man, S.-Q.; Guo, J.; Liu, Y. Hydroxide Assisted Synthesis of Monodisperse and Biocompatible Gold Nanoparticles with Dextran. Mater. Sci. Eng. C 2018, 93, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Nugrahani, I.; Ibrahim, S.; Mauludin, R.; Almira, M. Hydrate Transformation Study of Fluoroquinolone Antibiotics Using Fourier Transform Infrared Spectroscopy (FTIR). Int. J. Pharm. Pharm. Sci. 2015, 7, 246–252. [Google Scholar]
- Tiernan, H.; Byrne, B.; Kazarian, S.G. ATR-FTIR Spectroscopy and Spectroscopic Imaging for the Analysis of Biopharmaceuticals. Spectrochim. Acta Part A 2020, 241, 118636. [Google Scholar] [CrossRef] [PubMed]
- Ravulapalli, S.; Mariserla, B.M.K.; Soma, V.R.; Saritha, R.; Rao, D. Biosynthesis of Silver Nanoparticles Using CoriandrumSativum Leaf Extract and Their Application in Nonlinear Optics. Adv. Sci. Lett. 2010, 3, 138–144. [Google Scholar]
- Smith, R.C. Infrared Spectra of Substituted1, 10-Phenanthrolines. Ph.D. Thesis, Iowa State University, Ames, IA, USA, 1961. [Google Scholar]
- Rahman, M.T.; Vargas, M.; Ramana, C.V. Structural Characteristics, Electrical Conduction and Dielectric Properties of Gadolinium Substituted Cobalt Ferrite. J. Alloy. Compd. 2014, 617, 547–562. [Google Scholar] [CrossRef]
- Suresh, S.; Karthikeyan, S.; Jayamoorthy, K. FTIR and Multivariate Analysis to Study the Effect of Bulk and Nano Copper Oxide on Peanut Plant Leaves. J. Sci. Adv. Mater. Devices 2016, 1, 343–350. [Google Scholar] [CrossRef]
- Rahim, A.; Amiri, S. Anti-Corrosion Hybrid Nanocomposite Coatings with Encapsulated Organic Corrosion Inhibitors. J. Polym. Res. 2015, 12, 587–593. [Google Scholar]
- Ding, D.; Liu, K.; He, S.; Gao, C.; Yin, Y. Ligand-Exchange Assisted Formation of Au/TiO2Schottky Contact for Visible-Light Photocatalysis. Nano Lett. 2014, 14, 6731–6736. [Google Scholar] [CrossRef]
- Ansari, S.; Khan, M.M.; Ansari, M.; Cho, M.H. Silver Nanoparticles and Defect-Induced Visible Light Photocatalytic and Photoelectrochemical Performance of Ag@m-TiO2 Nanocomposite. Sol. Energy Mater. Sol. Cells 2015, 141, 162–170. [Google Scholar] [CrossRef]
- Ottoni, C.A.; Simões, M.F.; Fernandes, S.; dos Santos, J.G.; da Silva, E.S.; de Souza, R.F.B.; Maiorano, A.E. Screening of Filamentous Fungi for Antimicrobial Silver Nanoparticles Synthesis. AMB Express 2017, 7, 31. [Google Scholar] [CrossRef]
- Chaudhuri, S.K.; Malodia, L. Biosynthesis of Zinc Oxide Nanoparticles Using Leaf Extract of Calotropisgigantea: Characterization and Its Evaluation on Tree Seedling Growth in Nursery Stage. Appl. Nanosci. 2017, 7, 501–512. [Google Scholar] [CrossRef]
- Boningari, T.; Inturi, S.N.R.; Suidan, M.; Smirniotis, P.G. Novel One-Step Synthesis of Sulfur Doped-TiO2 by Flame Spray Pyrolysis for Visible Light Photocatalytic Degradation of Acetaldehyde. Chem. Eng. J. 2018, 339, 249–258. [Google Scholar] [CrossRef]
- Wannes, B.; Zaghouania, B.R.; Dimassia, W. Effect of Experimental Condition on Properties of Zinc Oxide Films Prepared by Sol-Gel Deposition with Ammonium Hydroxide as an Additive. J. Text. Sci. Eng. 2018, 8, 2165–3064. [Google Scholar]
- Roy, A.; Parveen, A.; Koppalkar, A.; Prasad, M. Effect of Nano-Titanium Dioxide with Different Antibiotics against Methicillin- Resistant Staphylococcus Aureus. J. Biomater. Nanobiotechnol. 2010, 1, 37. [Google Scholar] [CrossRef]
- Hajimohammad, A.; Fozouni, L. Antibacterial Effect of Zinc Oxide Nanoparticles on Mupirocin-Resistant Staphylococcus aureus Isolated from Nasal Carriers. Int. J. Basic Sci. Med. 2018, 3, 78–82. [Google Scholar] [CrossRef]
- Jesline, A.; John, N.P.; Narayanan, P.M.; Vani, C.; Murugan, S. Antimicrobial Activity of Zinc and Titanium Dioxide Nanoparticles against Biofilm-Producing Methicillin-Resistant Staphylococcus aureus. Appl. Nanosci. 2015, 5, 157–162. [Google Scholar] [CrossRef]
- Jiang, H.S.; Qiu, X.-N.; Li, G.-B.; Li, W.; Yin, L. Silver Nanoparticles Induced Accumulation of Reactive Oxygen Species and Alteration of Antioxidant Systems in the Aquatic Plant Spirodelapolyrhiza. Environ. Toxicol. Chem. 2014, 33, 1398–1405. [Google Scholar] [CrossRef]
- Tewari, S. Green Synthesis, Characterization and Antibacterial Activity of ZnO Nanoparticles. Am. J. Plant Sci. 2018, 9, 2158–2742. [Google Scholar]
- Chang, T.C.; Chen, C.-C.; Cheng, K.M.; Chin, C.Y.; Chen, Y.H.; Chen, X.A.; Sun, J.-R.; Young, J.J.; Chiueh, T.-S. Trimethyl Chitosan-Capped Silver Nanoparticles with Positive Surface Charge: Their Catalytic Activity and Antibacterial Spectrum Including Multidrug-Resistant Strains of Acinetobacterbaumannii. Colloids Surf. B 2017, 155, 61–70. [Google Scholar] [CrossRef]
- Ajitha, B.; Ashok Kumar Reddy, Y.; Reddy, P.S. Biogenic Nano-Scale Silver Particles by Tephrosiapurpurea Leaf Extract and their Inborn Antimicrobial Activity. Spectrochim. Acta Part A 2014, 121, 164–172. [Google Scholar] [CrossRef]
- Palanikumar, L.; Ramasamy, S.; Balachandran, C. Size-Dependent Antimicrobial Response of Zinc Oxide Nanoparticles. Nanobiotechnol. IET 2014, 8, 111–117. [Google Scholar] [CrossRef]
- Jones, N.; Ray, B.; Ranjit, K.; Manna, A. Antibacterial Activity of ZnO Nanoparticle Suspensions on a Broad Spectrum of Microorganisms. FEMS Microbiol. Lett. 2008, 279, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yang, J.; Xie, J.; Luo, Z.; Jiang, J.; Yang, Y.; Liu, S. The Potent Antimicrobial Properties of Cell Penetrating Peptide-Conjugated Silver Nanoparticles with Excellent Selectivity for Gram-Positive Bacteria over Erythrocytes. Nanoscale 2013, 5, 3834–3840. [Google Scholar] [CrossRef]
- Hsiao, I.L.; Huang, Y.-J. Effects of Various Physicochemical Characteristics on the Toxicities of ZnO and TiO2 Nanoparticles toward Human Lung Epithelial Cells. Sci. Total Environ. 2011, 409, 1219–1228. [Google Scholar] [CrossRef]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef] [PubMed]
- Gupta, G.; Senapati, V.; Dhawan, A.; Shanker, R. Heteroagglomeration of Zinc Oxide Nanoparticles with Clay Mineral Modulates the Bioavailability and Toxicity of Nanoparticle in Tetrahymena pyriformis. J. Colloid Interface Sci. 2017, 495, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Gopal, J.; Muraleedharan, P.; Tyagi, A.; Raj, B. Physics and Chemistry of Photocatalytic Titanium Dioxide: Visualization of Bactericidal Activity Using Atomic Force Microscopy. Curr. Sci. 2006, 90, 1378–1383. [Google Scholar]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial Activity of the Metals and Metal Oxide Nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef]
- Barua, S.; Konwarh, D.R.; Bhattacharya, S.S.; Das, P.; Devi, K.S.; Maiti, T.; Mandal, M.; Karak, N. Non-Hazardous Anticancerous and Antibacterial Colloidal ‘Green’ Silver Nanoparticles. Colloids Surf. B 2012, 105, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Gomaa, E.Z. Antimicrobial, Antioxidant and Antitumor Activities of Silver Nanoparticles Synthesized by Allium cepa Extract: A Green Approach. J. Genet. Eng. Biotechnol. 2017, 15, 49–57. [Google Scholar] [CrossRef]
- Ahamed, M.; Khan, M.A.M.; Akhtar, M.J.; Alhadlaq, H.A.; Alshamsan, A. Ag-Doping Regulates the Cytotoxicity of TiO2 Nanoparticles via Oxidative Stress in Human Cancer Cells. Sci. Rep. 2017, 7, 17662. [Google Scholar] [CrossRef]
- Behzadi, S.; Serpooshan, V.; Tao, W.; Hamaly, M.A.; Alkawareek, M.Y.; Dreaden, E.C.; Brown, D.; Alkilany, A.M.; Farokhzad, O.C.; Mahmoudi, M. Cellular Uptake of Nanoparticles: Journey inside the Cell. Chem. Soc. Rev. 2017, 46, 4218–4244. [Google Scholar] [CrossRef]
- Lu, P.J.; Fang, S.W.; Cheng, W.L.; Huang, S.C.; Huang, M.C.; Cheng, H.F. Characterization of Titanium Dioxide and Zinc Oxide Nanoparticles in Sunscreen Powder by Comparing Different Measurement Methods. J. Food Drug Anal. 2018, 26, 1192–1200. [Google Scholar] [CrossRef]
- Lucky, S.S.; Soo, K.C.; Zhang, Y. Nanoparticles in Photodynamic Therapy. Chem. Rev. 2015, 115, 1990–2042. [Google Scholar] [CrossRef]
- Sakhrani, N.; Padh, H. Organelle Targeting: Third Level of Drug Targeting. Drug Des. Dev. Ther. 2013, 7, 585–599. [Google Scholar]
- Hou, X.-S.; Wang, H.-S.; Mugaka, B.P.; Yang, G.-J.; Ding, Y. Mitochondria: Promising Organelle Targets for Cancer Diagnosis and Treatment. Biomater. Sci. 2018, 6, 2786–2797. [Google Scholar] [CrossRef]
- Taş, A.; Cakmak, N.; Ağbektaş, T.; Silig, Y. Cytotoxic Activity of Zinc Oxide/Titanium Dioxide Nanoparticles on Prostate Cancer Cells. Int. J. Chem. Technol. 2019, 3, 113–120. [Google Scholar] [CrossRef]
- Yoon, J.Y. Basic Principles of Electrochemical Bio sensing Using a Smartphone; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Dong, X.-X.; Yang, J.-Y.; Luo, L.; Zhang, Y.-F.; Mao, C.; Sun, Y.-M.; Lei, H.-T.; Shen, Y.-D.; Beier, R.C.; Xu, Z.-L. Portable AmperometricImmunosensor for Histamine Detection Using Prussian Blue-Chitosan-Gold Nanoparticle Nanocomposite Films. Biosens. Bioelectron. 2017, 98, 305–309. [Google Scholar] [CrossRef]
- Ma, H.; Gao, P.; Qian, P.; Su, Y. Size-Dependent Electrochemical Properties of Pure Metallic Nanoparticles. J. Phys. Chem. C 2020, 124, 3403–3409. [Google Scholar] [CrossRef]
- Braun, T.; Ghatkesar, M.K.; Backmann, N.; Grange, W.; Boulanger, P.; Letellier, L.; Lang, H.-P.; Bietsch, A.; Gerber, C.; Hegner, M. Quantitative Time-Resolved Measurement of Membrane Protein–Ligand Interactions Using Microcantilever Array Sensors. Nat. Nanotechnol. 2009, 4, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Murugesan, B.; Sonamuthu, J.; Samayanan, S.; Arumugam, S.; Mahalingam, S. Highly Biological Active Antibiofilm, Anticancer and Osteoblast Adhesion Efficacy from MWCNT/PPy/Pd Nanocomposite. Appl. Surf. Sci. 2018, 434, 400–411. [Google Scholar] [CrossRef]



| No. | Nanoparticles μL | No. | Nanoparticles μL | ||
|---|---|---|---|---|---|
| 1 | 250 μL AgNPs | 750 μL ZnONPs | 8 | 250 μL ZnONPs | 750 μL TiO2NPs |
| 2 | 250 μL AgNPs | 750 μL TiO2NPs | 9 | 333 μL ZnONPs | 667 μL AgNPs |
| 3 | 333 μL AgNPs | 667 μL ZnONPs | 10 | 333 μL ZnONPs | 667 μL TiO2NPs |
| 4 | 333 μL AgNPs | 667 μL TiO2NPss | 11 | 250 μL TiO2NPs | 750 μL AgNPs |
| 5 | 500 μL AgNPs | 500 μL ZnONPs | 12 | 250 μL TiO2NPs | 750 μL ZnONPs |
| 6 | 500 μL AgNPs | 500 μL TiO2NPs | 13 | 333 μL TiO2NPs | 667 μL AgNPs |
| 7 | 250 μL ZnONPs | 750 μL AgNPs | 14 | 333 μL TiO2NPs | 667 μL ZnONPs |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Kahky, D.; Attia, M.; Easa, S.M.; Awad, N.M.; Helmy, E.A. Interactive Effects of Biosynthesized Nanocomposites and Their Antimicrobial and Cytotoxic Potentials. Nanomaterials 2021, 11, 903. https://doi.org/10.3390/nano11040903
El-Kahky D, Attia M, Easa SM, Awad NM, Helmy EA. Interactive Effects of Biosynthesized Nanocomposites and Their Antimicrobial and Cytotoxic Potentials. Nanomaterials. 2021; 11(4):903. https://doi.org/10.3390/nano11040903
Chicago/Turabian StyleEl-Kahky, Dina, Magdy Attia, Saadia M. Easa, Nemat M. Awad, and Eman A. Helmy. 2021. "Interactive Effects of Biosynthesized Nanocomposites and Their Antimicrobial and Cytotoxic Potentials" Nanomaterials 11, no. 4: 903. https://doi.org/10.3390/nano11040903
APA StyleEl-Kahky, D., Attia, M., Easa, S. M., Awad, N. M., & Helmy, E. A. (2021). Interactive Effects of Biosynthesized Nanocomposites and Their Antimicrobial and Cytotoxic Potentials. Nanomaterials, 11(4), 903. https://doi.org/10.3390/nano11040903






