Ameliorative Potential of L-Alanyl L-Glutamine Dipeptide in Colon Cancer Patients Receiving Modified FOLFOX-6 Regarding the Incidence of Diarrhea, the Treatment Response, and Patients’ Survival: A Randomized Controlled Trial
Abstract
1. Introduction
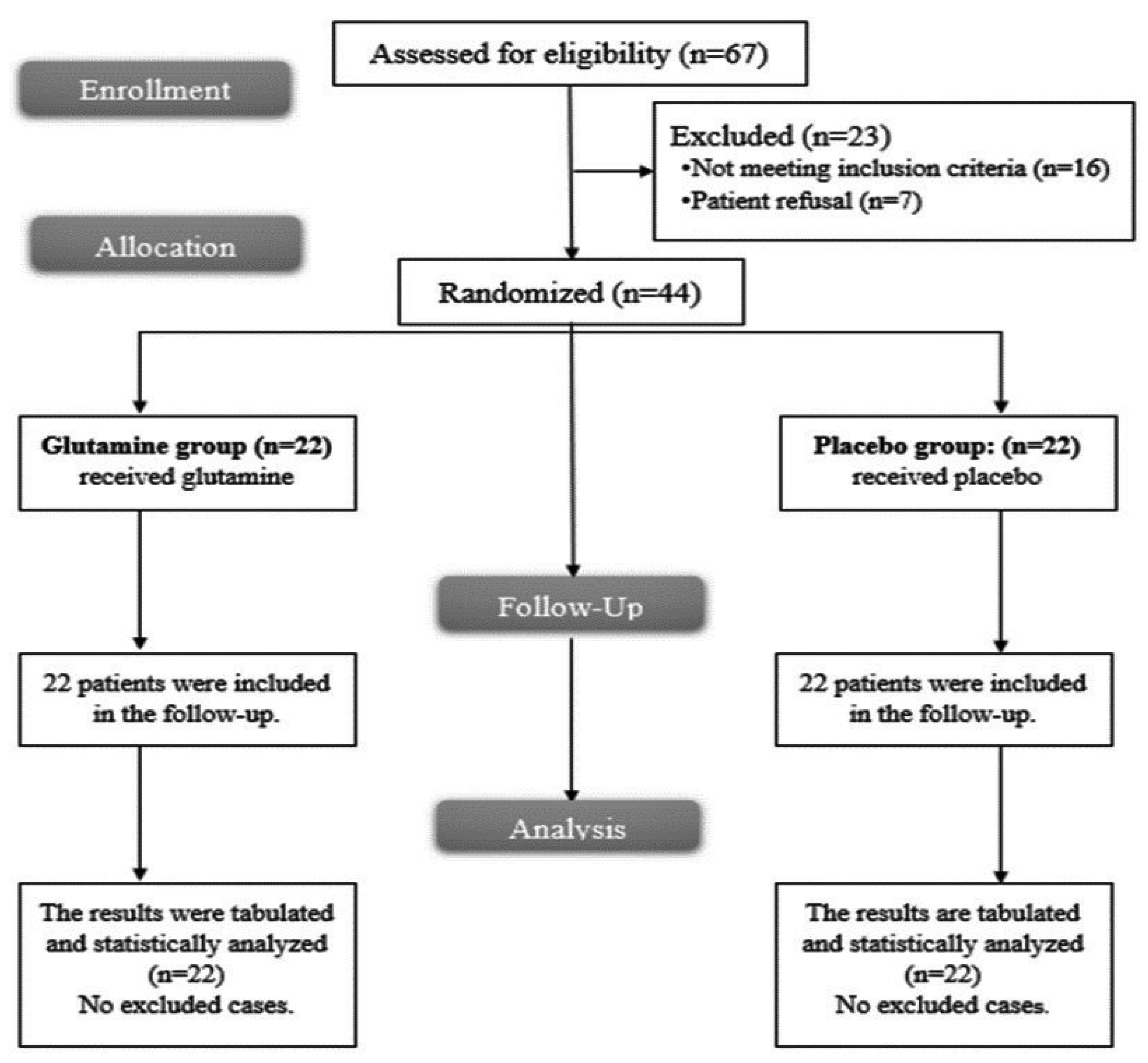
2. Materials and Methods
2.1. Eligible Patients
2.2. Inclusion Criteria
2.3. Exclusion Criteria
2.4. Treatment Plan (Chemotherapy)
2.5. Follow Up
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Freitas, A.; Silva, M.; Silva, C.; Prata, M.; Rodrigues, F.; Siqueira, R.; Lima, A.; Santos, A.A.; Havt, A. Alanyl-glutamine protects the intestinal barrier function in trained rats against the impact of acute exhaustive exercise. Braz. J. Med. Biol. Res. Rev. Bras. Pesqui. Med. E Biol. 2020, 53, e9211. [Google Scholar] [CrossRef]
- Alaa el-Din, A.K.; Labib, H.A.; Salah, D. Studying the Effect of Parenterally Administered l-Alanyl l-Glutamine Dipeptide in Diabetes and New Onset Diabetes in Liver Transplantation. Egypt. J. Anaesth. 2016, 32, 415–420. [Google Scholar] [CrossRef][Green Version]
- Krishna Rao, R. Role of Glutamine in Protection of Intestinal Epithelial Tight Junctions. J. Evid. Based Psychother. 2012, 5, 47–54. [Google Scholar] [CrossRef]
- Jiang, H.; Zhang, N.; Tang, T.; Feng, F.; Sun, H.; Qu, W. Target the human Alanine/Serine/Cysteine Transporter 2(ASCT2): Achievement and Future for Novel Cancer Therapy. Pharmacol. Res. 2020, 158, 104844. [Google Scholar] [CrossRef]
- Kim, M.H.; Kim, H. Oncogenes and Tumor Suppressors Regulate Glutamine Metabolism in Cancer Cells. J. Cancer Prev. 2013, 18, 221–226. [Google Scholar] [CrossRef]
- Choi, Y.-K.; Park, K.-G. Targeting Glutamine Metabolism for Cancer Treatment. Biomol. Ther. 2018, 26, 19–28. [Google Scholar] [CrossRef]
- Jiang, L.; Shestov, A.A.; Swain, P.; Yang, C.; Parker, S.J.; Wang, Q.A.; Terada, L.S.; Adams, N.D.; McCabe, M.T.; Pietrak, B.; et al. Reductive Carboxylation Supports Redox Homeostasis during Anchorage-Independent Growth. Nature 2016, 532, 255–258. [Google Scholar] [CrossRef]
- Anderson, P.M.; Lalla, R.V. Glutamine for Amelioration of Radiation and Chemotherapy Associated Mucositis during Cancer Therapy. Nutrients 2020, 12, 1675. [Google Scholar] [CrossRef]
- Hassan, A.; Khalaf, A.; Elias, A. Colorectal Cancer in Egypt: Clinical, Life-Style, and Socio-Demographic Risk Factors. Al-Azhar Int. Med. J. 2021, 2, 6–15. [Google Scholar] [CrossRef]
- Makhlouf, N.A.; Abdel-Gawad, M.; Mahros, A.M.; Lashen, S.A.; Zaghloul, M.; Eliwa, A.; Elshemy, E.E.; Ali-Eldin, Z.; Abdeltawab, D.; El-Raey, F.; et al. Colorectal cancer in Arab world: A systematic review. World J. Gastrointest. Oncol. 2021, 13, 1791–1798. [Google Scholar] [CrossRef]
- Okunaka, M.; Kano, D.; Matsui, R.; Kawasaki, T.; Uesawa, Y. Evaluation of the Expression Profile of Irinotecan-Induced Diarrhea in Patients with Colorectal Cancer. Pharmaceuticals 2021, 14, 377. [Google Scholar] [CrossRef]
- Abraham, J.P.; Magee, D.; Cremolini, C.; Antoniotti, C.; Halbert, D.D.; Xiu, J.; Stafford, P.; Berry, D.A.; Oberley, M.J.; Shields, A.F.; et al. Clinical Validation of a Machine-learning-derived Signature Predictive of Outcomes from First-line Oxaliplatin-based Chemotherapy in Advanced Colorectal Cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2021, 27, 1174–1183. [Google Scholar] [CrossRef]
- Wagner, A.D.; Grothey, A.; Andre, T.; Dixon, J.G.; Wolmark, N.; Haller, D.G.; Allegra, C.J.; de Gramont, A.; VanCutsem, E.; Alberts, S.R.; et al. Sex and Adverse Events of Adjuvant Chemotherapy in Colon Cancer: An Analysis of 34640 Patients in the ACCENT Database. J. Natl. Cancer Inst. 2021, 113, 400–407. [Google Scholar] [CrossRef]
- Bano, N.; Najam, R.; Qazi, F.; Mateen, A. Gastrointestinal Adverse Effects in Advanced Colorectal Carcinoma Patients Treated with Different Schedules of FOLFOX. Asian Pac. J. Cancer Prev. 2014, 15, 8089–8093. [Google Scholar] [CrossRef]
- Maroun, J.A.; Anthony, L.B.; Blais, N.; Burkes, R.; Dowden, S.D.; Dranitsaris, G.; Samson, B.; Shah, A.; Thirlwell, M.P.; Vincent, M.D.; et al. Prevention and Management of Chemotherapy-Induced Diarrhea in Patients with Colorectal Cancer: A Consensus Statement by the Canadian Working Group on Chemotherapy-Induced Diarrhea. Curr. Oncol. 2007, 14, 13–20. [Google Scholar] [CrossRef]
- Edge, S.B.; Compton, C.C. The American Joint Committee on Cancer: The 7th Edition of the AJCC Cancer Staging Manual and the Future of TNM. Ann. Surg. Oncol. 2010, 17, 1471–1474. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New Response Evaluation Criteria in Solid Tumours: Revised RECIST Guideline (Version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines on Evaluation of Similar Biotherapeutic Products (SBPs); World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Basch, E.; Reeve, B.B.; Mitchell, S.A.; Clauser, S.B.; Minasian, L.M.; Dueck, A.C.; Mendoza, T.R.; Hay, J.; Atkinson, T.M.; Abernethy, A.P.; et al. Development of the National Cancer Institute’s Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). JNCI J. Natl. Cancer Inst. 2014, 106, dju244. [Google Scholar] [CrossRef]
- Sun, Z.; Zhu, R.-J.; Yang, G.-F.; Li, Y. Neoadjuvant Chemotherapy with FOLFOX4 Regimen to Treat Advanced Gastric Cancer Improves Survival without Increasing Adverse Events: A Retrospective Cohort Study from a Chinese Center. Sci. World J. 2014, 2014, 418694. [Google Scholar] [CrossRef]
- Salehifar, E.; Avan, R.; Janbabaei, G.; Mousavi, S.K.; Faramarzi, F. Comparison the Incidence and Severity of Side Effects Profile of FOLFOX and DCF Regimens in Gastric Cancer Patients. Iran. J. Pharm. Res. 2019, 18, 1032–1039. [Google Scholar] [CrossRef]
- Pandey, M.; Goel, R.; Gaurav, K.; Shukla, M. Glutamine: A Novel Approach to Chemotherapy-Induced Toxicity. Indian J. Med. Paediatr. Oncol. 2012, 33, 13. [Google Scholar] [CrossRef] [PubMed]
- Widjaja, N.A.; Pratama, A.; Prihaningtyas, R.; Irawan, R.; Ugrasena, I. Efficacy Oral Glutamine to Prevent Oral Mucositis and Reduce Hospital Costs During Chemotherapy in Children with Acute Lymphoblastic Leukemia. Asian Pac. J. Cancer Prev. 2020, 21, 2117–2121. [Google Scholar] [CrossRef] [PubMed]
- Altman, B.J.; Stine, Z.E.; Dang, C.V. From Krebs to Clinic: Glutamine Metabolism to Cancer Therapy. Nat. Rev. Cancer 2016, 16, 619–634. [Google Scholar] [CrossRef]
- Li, T.; Copeland, C.; Le, A. Glutamine Metabolism in Cancer. In The Heterogeneity of Cancer Metabolism; Le, A., Ed.; Springer International Publishing: Cham, Switzerland, 2021; pp. 17–38. ISBN 978-3-030-65768-0. [Google Scholar]
- Commisso, C.; Davidson, S.M.; Soydaner-Azeloglu, R.G.; Parker, S.J.; Kamphorst, J.J.; Hackett, S.; Grabocka, E.; Nofal, M.; Drebin, J.A.; Thompson, C.B.; et al. Macropinocytosis of Protein Is an Amino Acid Supply Route in Ras-Transformed Cells. Nature 2013, 497, 633–637. [Google Scholar] [CrossRef]
- Kamphorst, J.J.; Nofal, M.; Commisso, C.; Hackett, S.R.; Lu, W.; Grabocka, E.; Vander Heiden, M.G.; Miller, G.; Drebin, J.A.; Bar-Sagi, D.; et al. Human Pancreatic Cancer Tumors Are Nutrient Poor and Tumor Cells Actively Scavenge Extracellular Protein. Cancer Res. 2015, 75, 544–553. [Google Scholar] [CrossRef]
- Chiara, S.; Nobile, M.T.; Gozza, A.; Taveggia, P.; Heouaine, A.; Pastrone, I.; Percivale, P.L.; Lionetto, R.; Sanguineti, O.; Rosso, R. Phase II Study of Weekly Oxaliplatin and High-Dose Infusional 5-Fluorouracil plus Leucovorin in Pretreated Patients with Metastatic Colorectal Cancer. Anticancer Res. 2004, 24, 355–360. [Google Scholar]
- Yoshida, S.; Matsui, M.; Shirouzu, Y.; Fujita, H.; Yamana, H.; Shirouzu, K. Effects of Glutamine Supplements and Radiochemotherapy on Systemic Immune and Gut Barrier Function in Patients with Advanced Esophageal Cancer. Ann Surg. 1998, 227, 485–491. [Google Scholar] [CrossRef]
- Kim, M.-H.; Kim, H. The Roles of Glutamine in the Intestine and Its Implication in Intestinal Diseases. Int. J. Mol. Sci. 2017, 18, 1051. [Google Scholar] [CrossRef]
- Cluntun, A.A.; Lukey, M.J.; Cerione, R.A.; Locasale, J.W. Glutamine Metabolism in Cancer: Understanding the Heterogeneity. Trends Cancer 2017, 3, 169–180. [Google Scholar] [CrossRef]
- Suzannec Klimberg, V.; McClellan, J.L. Glutamine, Cancer, and Its Therapy. Am. J. Surg. 1996, 172, 418–424. [Google Scholar] [CrossRef]

| Glutamine Group (n = 22) | Placebo Group (n = 22) | p-Value | ||
|---|---|---|---|---|
| Age (years) | Mean ± SD | 45.41 ± 5.72 | 53.09 ± 7.19 | <0.001 * |
| Range | 35–61 | 44–66 | ||
| Gender | Male | 7 (31.82%) | 10 (45.45%) | 0.390 |
| Female | 15 (68.18%) | 12 (54.55%) | ||
| ECOG performance status | 0 | 8 (36.36%) | 4 (18.18%) | 0.189 |
| 1 | 9 (40.91%) | 15 (68.18%) | ||
| 2 | 5 (22.73%) | 3 (13.64%) | ||
| Site | Colon | 22 (100%) | 22 (100%) | - |
| Pathology | Adenocarcinoma | 22 (100%) | 22 (100%) | - |
| CEA | Mean ± SD | 3.54 ± 1.82 | 4.18 ± 2.06 | 0.284 |
| Range | 1–9 | 1–9 | ||
| Surgery | Yes | 22 (100%) | 22 (100%) | - |
| Stage | Stage II | 5 (22.73%) | 8 (36.36%) | 0.322 |
| Stage III | 17 (77.27%) | 14 (36.64%) | ||
| Treatment | Chemotherapy | 22 (100%) | 22 (100%) | - |
| Chemotherapy | mFOLFOX-6 | 22 (100%) | 22 (100%) | - |
| Glutamine Group (n = 22) | Placebo Group (n = 22) | p-Value | |
|---|---|---|---|
| Partial response | 7 (31.82%) | 6 (27.27%) | 0.892 |
| Complete response | 15 (68.18%) | 16 (72.73%) |
| Glutamine Group (n = 22) | Placebo Group (n = 22) | p-Value | |
|---|---|---|---|
| After Two Cycles | |||
| No diarrhea | 12 (54.55%) | 8 (36.36%) | 0.066 |
| Grade 1 | 8 (36.36%) | 5 (22.73%) | |
| Grade 2 | 2 (9.09%) | 7 (31.82%) | |
| Grade 3 | 0 (0%) | 2 (9.09%) | |
| After Four Cycles | |||
| No diarrhea | 8 (36.36%) | 1 (4.55%) | < 0.001 * |
| Grade 1 | 11 (50%) | 2 (9.09%) | |
| Grade 2 | 2 (9.09%) | 11 (50%) | |
| Grade 3 | 1 (4.55%) | 8 (36.36%) | |
| After Six Cycles | |||
| No diarrhea | 12 (54.55%) | 1 (4.55%) | < 0.001 * |
| Grade 1 | 6 (27.27%) | 3 (13.6%) | |
| Grade 2 | 4 (18.18%) | 9 (40.91%) | |
| Grade 3 | 0 (0%) | 5 (22.73%) | |
| Grade 4 | 0 (0%) | 4 (18.18%) | |
| Mean | SD | 95% CI for the Mean | Mortality | p-Value | |
|---|---|---|---|---|---|
| Glutamine group (n = 22) | 22.09 | 3.108 | 20.51–23.67 | 5 (22.7%) | 0.322 |
| Placebo group (n = 22) | 20.73 | 2.986 | 18.21–23.25 | 8 (36.4%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabry, N.M.; Naguib, T.M.; Kabel, A.M.; Khafagy, E.-S.; Arab, H.H.; Almorsy, W.A. Ameliorative Potential of L-Alanyl L-Glutamine Dipeptide in Colon Cancer Patients Receiving Modified FOLFOX-6 Regarding the Incidence of Diarrhea, the Treatment Response, and Patients’ Survival: A Randomized Controlled Trial. Medicina 2022, 58, 394. https://doi.org/10.3390/medicina58030394
Sabry NM, Naguib TM, Kabel AM, Khafagy E-S, Arab HH, Almorsy WA. Ameliorative Potential of L-Alanyl L-Glutamine Dipeptide in Colon Cancer Patients Receiving Modified FOLFOX-6 Regarding the Incidence of Diarrhea, the Treatment Response, and Patients’ Survival: A Randomized Controlled Trial. Medicina. 2022; 58(3):394. https://doi.org/10.3390/medicina58030394
Chicago/Turabian StyleSabry, Nesreen M., Tamer M. Naguib, Ahmed M. Kabel, El-Sayed Khafagy, Hany H. Arab, and Walid A. Almorsy. 2022. "Ameliorative Potential of L-Alanyl L-Glutamine Dipeptide in Colon Cancer Patients Receiving Modified FOLFOX-6 Regarding the Incidence of Diarrhea, the Treatment Response, and Patients’ Survival: A Randomized Controlled Trial" Medicina 58, no. 3: 394. https://doi.org/10.3390/medicina58030394
APA StyleSabry, N. M., Naguib, T. M., Kabel, A. M., Khafagy, E.-S., Arab, H. H., & Almorsy, W. A. (2022). Ameliorative Potential of L-Alanyl L-Glutamine Dipeptide in Colon Cancer Patients Receiving Modified FOLFOX-6 Regarding the Incidence of Diarrhea, the Treatment Response, and Patients’ Survival: A Randomized Controlled Trial. Medicina, 58(3), 394. https://doi.org/10.3390/medicina58030394






