Non-Classical HLA Determinants of the Clinical Response after Autologous Stem Cell Transplantation for Systemic Sclerosis
Abstract
1. Introduction
2. Results
2.1. Socio-Demographical Characteristics and Response to AHSCT
2.2. Non Classical and Classical HLA Class I Genetics and Response to AHSCT
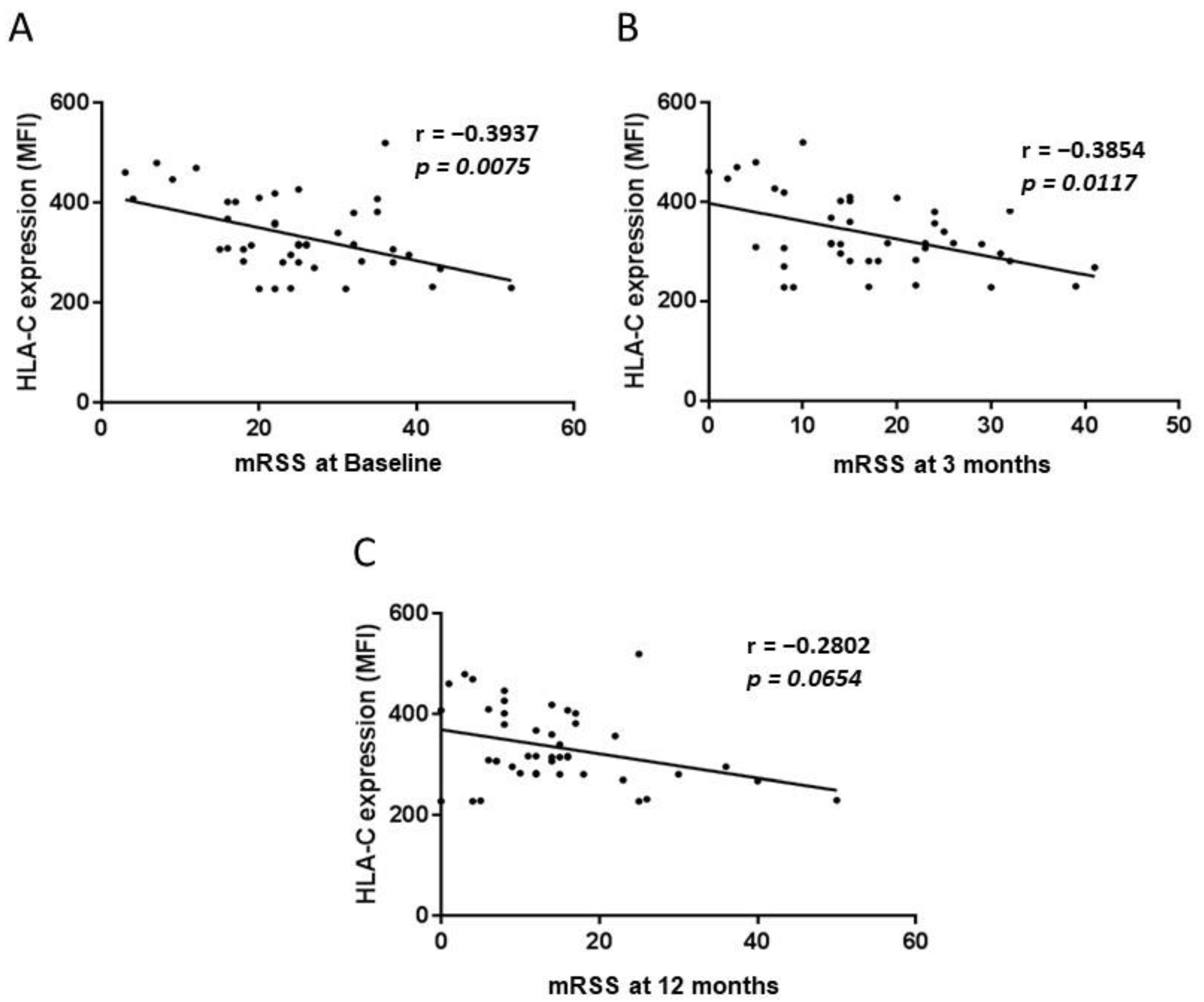
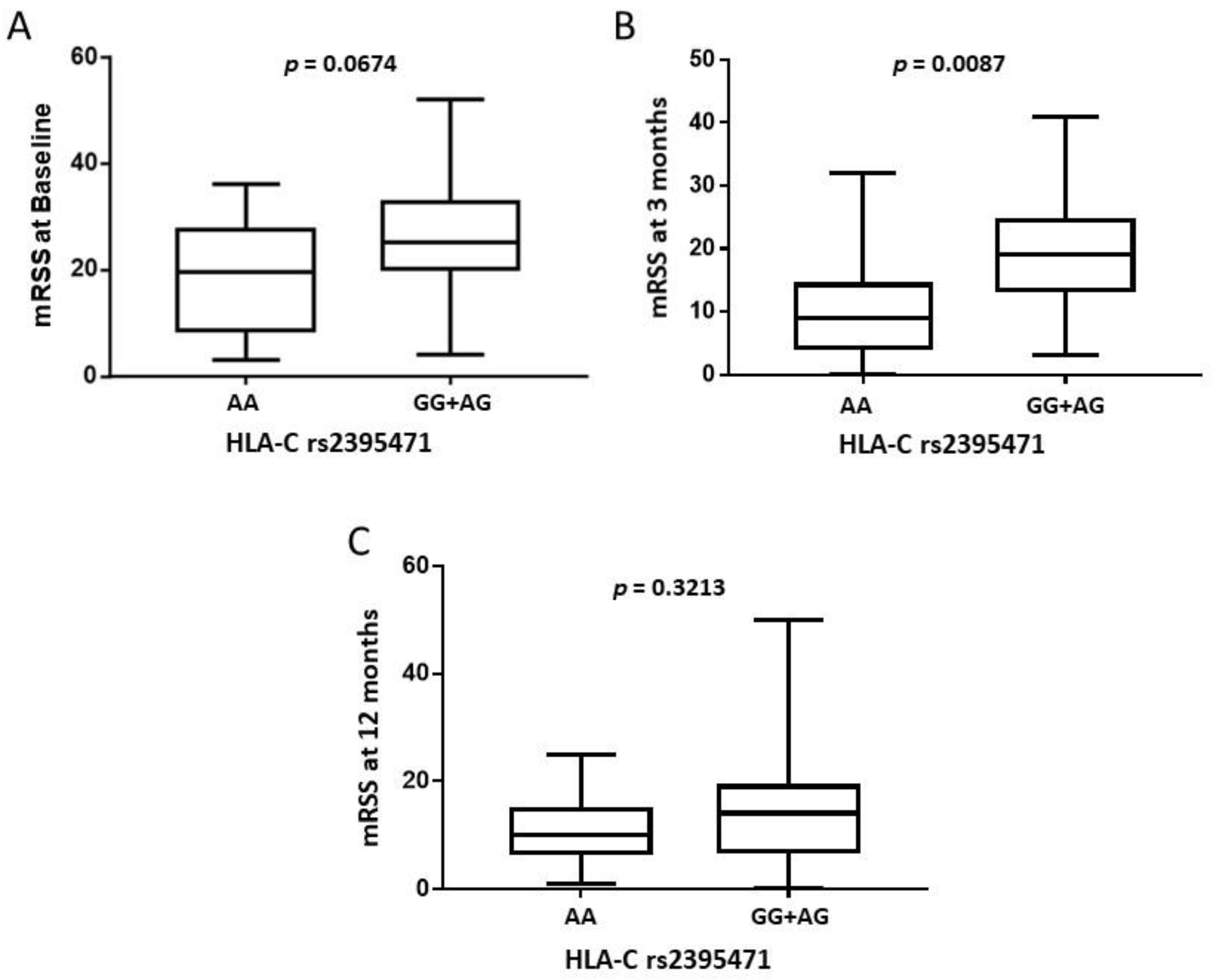
2.3. HLA-A and C and the Modified Rodnan Skin Score (mRSS)
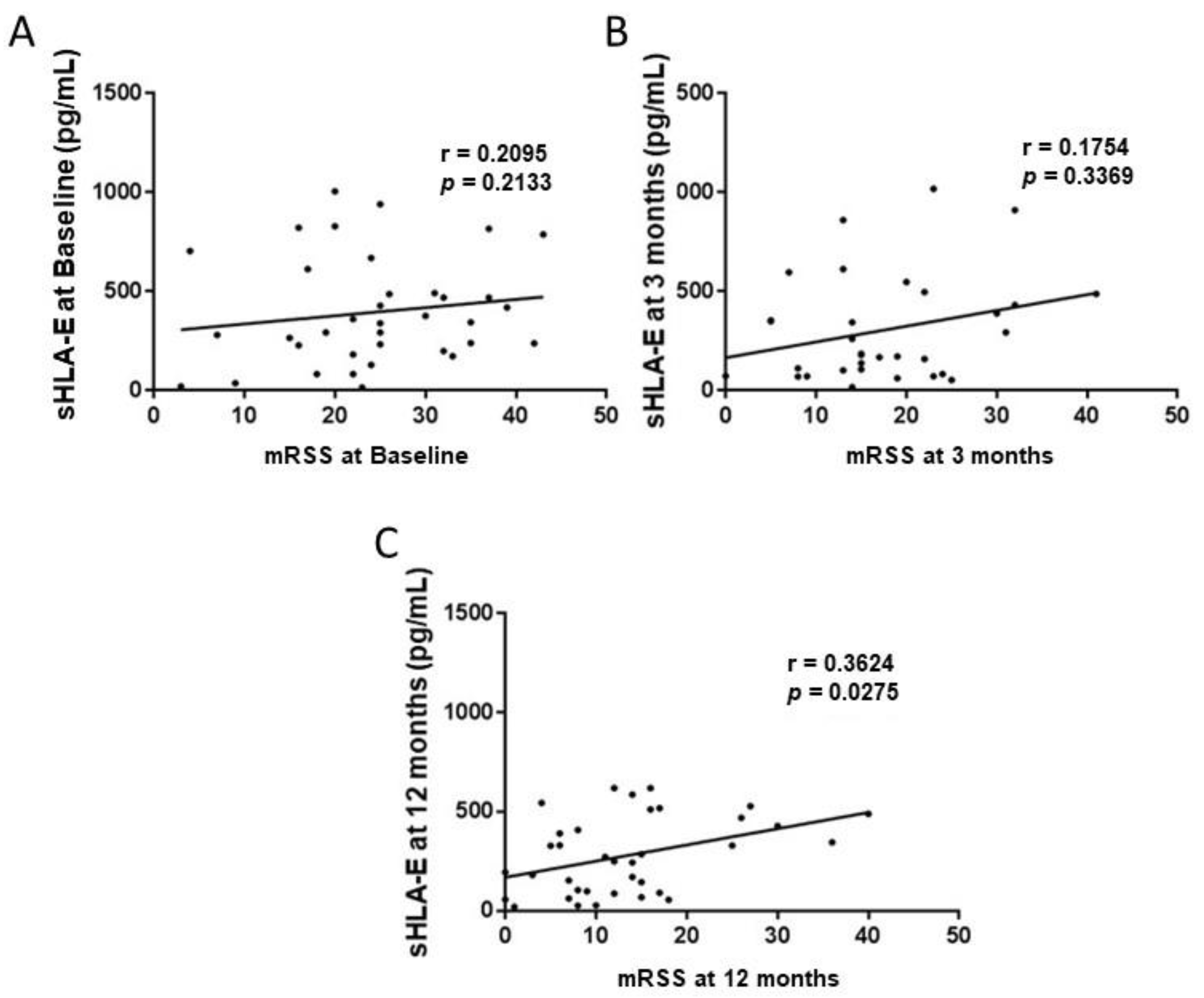
2.4. Circulating Levels of Soluble HLA-G and E Molecules
3. Discussion
4. Material and Methods
4.1. Study Subjects
4.2. HLA Genotyping
4.3. Soluble HLA-G and E Measurements
4.4. Imputation of Classical HLA Determinants of HLA-E Expression
4.5. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gabrielli, A.; Avvedimento, E.V.; Krieg, T. Scleroderma. N. Engl. J. Med. 2009, 360, 1989–2003. [Google Scholar] [CrossRef] [PubMed]
- Fuschiotti, P. Current perspectives on the immunopathogenesis of systemic sclerosis. Immunotargets Ther. 2016, 5, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Georges, C.; Chassany, O.; Toledano, C.; Mouthon, L.; Tiev, K.; Meyer, O.; Ilie, D.; Rambeloarisoa, J.; Marjanovic, Z.; Cabane, J.; et al. Impact of pain in health related quality of life of patients with systemic sclerosis. Rheumatology 2006, 45, 1298–1302. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hudson, M.; Thombs, B.D.; Steele, R.; Panopalis, P.; Newton, E.; Baron, M.; Canadian Scleroderma Research, G. Health-related quality of life in systemic sclerosis: A systematic review. Arthritis Rheumatol. 2009, 61, 1112–1120. [Google Scholar] [CrossRef]
- Rubio-Rivas, M.; Royo, C.; Simeón-Aznar, C.P.; Corbella, X.; Fonollosa, V. Mortality and survival in systemic sclerosis: Systematic review and meta-analysis. Semin. Arthritis Rheumatol. 2014, 44, 208–219. [Google Scholar] [CrossRef]
- Hao, Y.; Hudson, M.; Baron, M.; Carreira, P.; Stevens, W.; Rabusa, C.; Tatibouet, S.; Carmona, L.; Joven, B.E.; Huq, M.; et al. Early Mortality in a Multinational Systemic Sclerosis Inception Cohort. Arthritis Rheumatol. 2017, 69, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- Fransen, J.; Popa-Diaconu, D.; Hesselstrand, R.; Carreira, P.; Valentini, G.; Beretta, L.; Airo, P.; Inanc, M.; Ullman, S.; Balbir-Gurman, A.; et al. Clinical prediction of 5-year survival in systemic sclerosis: Validation of a simple prognostic model in EUSTAR centres. Ann. Rheum. Dis. 2011, 70, 1788–1792. [Google Scholar] [CrossRef] [PubMed]
- Steen, V.D.; Medsger, T.A.; Rodnan, G.P. D-Penicillamine therapy in progressive systemic sclerosis (scleroderma): A retrospective analysis. Ann. Intern. Med. 1982, 97, 652–659. [Google Scholar] [CrossRef]
- Elhai, M.; Meune, C.; Boubaya, M.; Avouac, J.; Hachulla, E.; Balbir-Gurman, A.; Riemekasten, G.; Airã, P.; Joven, B.; Vettori, S.; et al. Mapping and predicting mortality from systemic sclerosis. Ann. Rheum. Dis. 2017, 76, 1897–1905. [Google Scholar] [CrossRef]
- Mousseaux, E.; Agoston-Coldea, L.; Marjanovic, Z.; Stanciu, R.; Deligny, C.; Perdrix, L.; Boutouyrie, P.; Azarine, A.; Soulat, G.; Farge, D. Left Ventricle Replacement Fibrosis Detected by CMR Associated With Cardiovascular Events in Systemic Sclerosis Patients. J. Am. Coll. Cardiol. 2018, 71, 703–705. [Google Scholar] [CrossRef]
- Goh, N.S.; Hoyles, R.K.; Denton, C.P.; Hansell, D.M.; Renzoni, E.; Maher, T.M.; Nicholson, A.G.; Wells, A.U. Short-Term Pulmonary Function Trends Are Predictive of Mortality in Interstitial Lung Disease Associated With Systemic Sclerosis. Arthritis Rheumatol. 2017, 69, 1670–1678. [Google Scholar] [CrossRef] [PubMed]
- Poole, J.L.; Steen, V.D. The use of the health assessment questionnaire (HAQ) to determine physical disability in systemic sclerosis. Arthritis Care Res. 1991, 4, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Tyndall, A.J.; Bannert, B.; Vonk, M.; Airò, P.; Cozzi, F.; Carreira, P.E.; Bancel, D.F.; Allanore, Y.; Müller-Ladner, U.; Distler, O.; et al. Causes and risk factors for death in systemic sclerosis: A study from the EULAR Scleroderma Trials and Research (EUSTAR) database. Ann. Rheum. Dis. 2010, 69, 1809–1815. [Google Scholar] [CrossRef] [PubMed]
- Burt, R.K.; Shah, S.J.; Dill, K.; Grant, T.; Gheorghiade, M.; Schroeder, J.; Craig, R.; Hirano, I.; Marshall, K.; Ruderman, E.; et al. Autologous non-myeloablative haemopoietic stem-cell transplantation compared with pulse cyclophosphamide once per month for systemic sclerosis (ASSIST): An open-label, randomised phase 2 trial. Lancet 2011, 378, 498–506. [Google Scholar] [CrossRef]
- Van Laar, J.M.; Farge, D.; Sont, J.; Naraghi, K.; Marjanovic, Z.; Larghero, J.; Schuerwegh, A.J.; Marijt, E.W.A.; Vonk, M.C.; Schattenberg, A.V.; et al. Autologous hematopoietic stem cell transplantation vs. intravenous pulse cyclophosphamide in diffuse cutaneous systemic sclerosis: A randomized clinical trial. JAMA 2014, 311, 2490–2498. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.M.; Majhail, N.S.; Bredeson, C.; Carpenter, P.A.; Chatterjee, S.; Crofford, L.J.; Georges, G.E.; Nash, R.A.; Pasquini, M.C.; Sarantopoulos, S.; et al. Systemic Sclerosis as an Indication for Autologous Hematopoietic Cell Transplantation: Position Statement from the American Society for Blood and Marrow Transplantation. Biol. Blood Marrow Transplant. 2018, 24, 1961–1964. [Google Scholar] [CrossRef]
- Kowal-Bielecka, O.; Fransen, J.; Avouac, J.; Becker, M.; Kulak, A.; Allanore, Y.; Distler, O.; Clements, P.; Cutolo, M.; Czirjak, L.; et al. Update of EULAR recommendations for the treatment of systemic sclerosis. Ann. Rheum. Dis. 2017, 76, 1327–1339. [Google Scholar] [CrossRef]
- Kanate, A.S.; Majhail, N.S.; Savani, B.N.; Bredeson, C.; Champlin, R.E.; Crawford, S.; Giralt, S.A.; Lemaistre, C.F.; Marks, D.I.; Omel, J.L.; et al. Indications for Hematopoietic Cell Transplantation and Immune Effector Cell Therapy: Guidelines from the American Society for Transplantation and Cellular Therapy. Biol. Blood Marrow Transplant. 2020, 26, 1247–1256. [Google Scholar] [CrossRef]
- Duarte, R.F.; Labopin, M.; Bader, P.; Basak, G.W.; Bonini, C.; Chabannon, C.; Corbacioglu, S.; Dreger, P.; Dufour, C.; Gennery, A.R.; et al. Indications for haematopoietic stem cell transplantation for haematological diseases, solid tumours and immune disorders: Current practice in Europe, 2019. Bone Marrow Transplant. 2019, 54, 1525–1552. [Google Scholar] [CrossRef]
- Farge, D.; Marolleau, J.P.; Zohar, S.; Marjanovic, Z.; Cabane, J.; Mounier, N.; Hachulla, E.; Philippe, P.; Sibilia, J.; Rabian, C.; et al. Autologous bone marrow transplantation in the treatment of refractory systemic sclerosis: Early results from a French multicentre phase I-II study. Br. J. Haematol. 2002, 119, 726–739. [Google Scholar] [CrossRef]
- Burt, R.K.; Oliveira, M.C.; Shah, S.J.; Moraes, D.; Simoes, B.; Gheorghiade, M.; Schroeder, J.; Ruderman, E.; Farge, D.; Chai, Z.J.; et al. Cardiac involvement and treatment-related mortality after non-myeloablative haemopoietic stem-cell transplantation with unselected autologous peripheral blood for patients with systemic sclerosis: A retrospective analysis. Lancet 2013, 381, 1116–1124. [Google Scholar] [CrossRef]
- Farge, D.; Arruda, L.C.; Brigant, F.; Clave, E.; Douay, C.; Marjanovic, Z.; Deligny, C.; Maki, G.; Gluckman, E.; Toubert, A.; et al. Long-term immune reconstitution and T cell repertoire analysis after autologous hematopoietic stem cell transplantation in systemic sclerosis patients. J. Hematol. Oncol. 2017, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Arruda, L.C.M.; Malmegrim, K.C.R.; Lima-Júnior, J.R.; Clave, E.; Dias, J.B.E.; Moraes, D.A.; Douay, C.; Fournier, I.; Moins-Teisserenc, H.; Alberdi, A.J.; et al. Immune rebound associates with a favorable clinical response to autologous HSCT in systemic sclerosis patients. Blood Adv. 2018, 2, 126–141. [Google Scholar] [CrossRef] [PubMed]
- Trowsdale, J.; Knight, J.C. Major Histocompatibility Complex Genomics and Human Disease. Annu. Rev. Genom. Hum. Genet. 2013, 14, 301–323. [Google Scholar] [CrossRef]
- López-Botet, M.; Bellón, T.; Llano, M.; Navarro, F.; García, P.; de Miguel, M. Paired inhibitory and triggering NK cell receptors for HLA class I molecules. Hum. Immunol. 2000, 61, 7–17. [Google Scholar] [CrossRef]
- Ferreira, L.M.; Meissner, T.B.; Tilburgs, T.; Strominger, J.L. HLA-G: At the Interface of Maternal–Fetal Tolerance. Trends Immunol. 2017, 38, 272–286. [Google Scholar] [CrossRef] [PubMed]
- Braud, V.M.; Allan, D.S.J.; O’Callaghan, C.A.; Söderström, K.; D’Andrea, A.; Ogg, G.S.; Lazetic, S.; Young, N.T.; Bell, J.I.; Phillips, J.H.; et al. HLA-E binds to natural killer cell receptors CD94/NKG2A, B and C. Nature 1998, 391, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Strong, R.K.; Holmes, M.A.; Li, P.; Braun, L.; Lee, N.; Geraghty, D.E. HLA-E allelic variants. Correlating differential expression, peptide affinities, crystal structures, and thermal stabilities. J. Biol. Chem. 2003, 278, 5082–5090. [Google Scholar] [CrossRef] [PubMed]
- Vince, N.; Li, H.; Ramsuran, V.; Naranbhai, V.; Duh, F.-M.; Fairfax, B.P.; Saleh, B.; Knight, J.; Anderson, S.; Carrington, M. HLA-C Level Is Regulated by a Polymorphic Oct1 Binding Site in the HLA-C Promoter Region. Am. J. Hum. Genet. 2016, 99, 1353–1358. [Google Scholar] [CrossRef]
- Ramsuran, V.; Naranbhai, V.; Horowitz, A.; Qi, Y.; Martin, M.P.; Yuki, Y.; Gao, X.; Walker-Sperling, V.; Del Prete, G.Q.; Schneider, D.K.; et al. Elevated HLA-A expression impairs HIV control through inhibition of NKG2A-expressing cells. Science 2018, 359, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-Y.; Yan, W.-H.; Lin, A.; Xu, H.-H.; Zhang, J.-G.; Wang, X.-X. The 14 bp deletion polymorphisms in HLA-G gene play an important role in the expression of soluble HLA-G in plasma. Tissue Antigens 2008, 72, 335–341. [Google Scholar] [CrossRef] [PubMed]
- López-Botet, M.; Bellón, T. Natural killer cell activation and inhibition by receptors for MHC class I. Curr. Opin. Immunol. 1999, 11, 301–307. [Google Scholar] [CrossRef]
- Borrego, F.; Kabat, J.; Sanni, T.B.; Coligan, J.E. NK cell CD94/NKG2A inhibitory receptors are internalized and recycle independently of inhibitory signaling processes. J. Immunol. 2002, 169, 6102–6111. [Google Scholar] [CrossRef] [PubMed]
- Morandi, F.; Rizzo, R.; Fainardi, E.; Rouas-Freiss, N.; Pistoia, V. Recent Advances in Our Understanding of HLA-G Biology: Lessons from a Wide Spectrum of Human Diseases. J. Immunol. Res. 2016, 2016, 4326495. [Google Scholar] [CrossRef]
- Amodio, G.; Gregori, S. HLA-G Genotype/Expression/Disease Association Studies: Success, Hurdles, and Perspectives. Front. Immunol. 2020, 11, 1178. [Google Scholar] [CrossRef]
- Favoino, E.; Favia, I.E.; Vettori, S.; Vicenti, C.; Prete, M.; Valentini, G.; Perosa, F. Clinical correlates of human leucocyte antigen (HLA)-G in systemic sclerosis. Clin. Exp. Immunol. 2015, 181, 100–109. [Google Scholar] [CrossRef]
- Negrini, S.; Contini, P.; Pupo, F.; Greco, M.; Murdaca, G.; Puppo, F. Expression of membrane-bound human leucocyte antigen-G in systemic sclerosis and systemic lupus erythematosus. Hum. Immunol. 2020, 81, 162–167. [Google Scholar] [CrossRef]
- Pende, D.; Falco, M.; Vitale, M.; Cantoni, C.; Vitale, C.; Munari, E.; Bertaina, A.; Moretta, F.; Del Zotto, G.; Pietra, G.; et al. Killer Ig-Like Receptors (KIRs): Their Role in NK Cell Modulation and Developments Leading to Their Clinical Exploitation. Front. Immunol. 2019, 10, 1179. [Google Scholar] [CrossRef]
- Goodson-Gregg, F.J.; Krepel, S.A.; Anderson, S.K. Tuning of human NK cells by endogenous HLA-C expression. Immunogenetics 2020, 72, 205–215. [Google Scholar] [CrossRef]
- Petersdorf, E.W.; Carrington, M.; O’Huigin, C.; Bengtsson, M.; De Santis, D.; Dubois, V.; Gooley, T.; Horowitz, M.; Hsu, K.; Madrigal, J.A.; et al. Role of HLA-B exon 1 in graft-versus-host disease after unrelated haemopoietic cell transplantation: A retrospective cohort study. Lancet Haematol. 2020, 7, e50–e60. [Google Scholar] [CrossRef]
- Petersdorf, E.W.; Gooley, T.; Volt, F.; Kenzey, C.; Madrigal, A.; McKallor, C.; Querol, S.; Rafii, H.; Rocha, V.; Tamouza, R.; et al. Use of the HLA-B leader to optimize cord-blood transplantation. Haematologica 2020. [Google Scholar] [CrossRef] [PubMed]
- Tamouza, R.; Busson, M.; Rocha, V.; Fortier, C.; Haddad, Y.; Brun, M.; Boukouaci, W.; Bleux, H.; Socié, G.; Krishnamoorthy, R.; et al. Homozygous status for HLA-E*0103 confers protection from acute graft-versus-host disease and transplant-related mortality in HLA-matched sibling hematopoietic stem cell transplantation. Transplantation 2006, 82, 1436–1440. [Google Scholar] [CrossRef] [PubMed]
- Boukouaci, W.; Busson, M.; Fortier, C.; Amokrane, K.; de Latour, R.P.; Robin, M.; Krishnamoorthy, R.; Toubert, A.; Charron, D.; Socié, G.; et al. Association of HLA-G Low Expressor Genotype with Severe Acute Graft-Versus-Host Disease after Sibling Bone Marrow Transplantation. Front. Immunol. 2011, 2, 74. [Google Scholar] [CrossRef] [PubMed]
- Kordelas, L.; Schwich, E.; Lindemann, M.; Heinemann, F.M.; Buttkereit, U.; Horn, P.A.; Beelen, D.W.; Rebmann, V. Decreased Soluble Human Leukocyte Antigen E Levels in Patients After Allogeneic Hematopoietic Stem Cell Transplantation Are Associated with Severe Acute and Extended Chronic Graft-versus-Host Disease and Inferior Overall Survival. Front. Immunol. 2019, 10, 3027. [Google Scholar] [CrossRef] [PubMed]
- Morandi, F.; Cangemi, G.; Barco, S.; Amoroso, L.; Giuliano, M.; Gigliotti, A.R.; Pistoia, V.; Corrias, M.V. Plasma levels of soluble HLA-E and HLA-F at diagnosis may predict overall survival of neuroblastoma patients. BioMed Res. Int. 2013, 2013, 956878. [Google Scholar] [CrossRef]
- Derré, L.; Corvaisier, M.; Charreau, B.; Moreau, A.; Godefroy, E.; Moreau-Aubry, A.; Jotereau, F.; Gervois, N. Expression and release of HLA-E by melanoma cells and melanocytes: Potential impact on the response of cytotoxic effector cells. J. Immunol. 2006, 177, 3100–3107. [Google Scholar] [CrossRef]
- Van Hall, T.; André, P.; Horowitz, A.; Ruan, D.F.; Borst, L.; Zerbib, R.; Narni-Mancinelli, E.; Van Der Burg, S.H.; Vivier, E. Monalizumab: Inhibiting the novel immune checkpoint NKG2A. J. Immunother. Cancer 2019, 7, 263. [Google Scholar] [CrossRef]
- Carosella, E.D.; Rouas-Freiss, N.; Roux, D.T.-L.; Moreau, P.; LeMaoult, J. HLA-G: An Immune Checkpoint Molecule. Adv. Immunol. 2015, 127, 33–144. [Google Scholar] [CrossRef]
- Contini, P.; Negrini, S.; Murdaca, G.; Borro, M.; Puppo, F. Evaluation of membrane-bound and soluble forms of human leucocyte antigen-G in systemic sclerosis. Clin. Exp. Immunol. 2018, 193, 152–159. [Google Scholar] [CrossRef]
- Contini, P.; Murdaca, G.; Puppo, F.; Negrini, S. HLA-G Expressing Immune Cells in Immune Mediated Diseases. Front. Immunol. 2020, 11, 1613. [Google Scholar] [CrossRef]
- van den Hoogen, F.; Khanna, D.; Fransen, J.; Johnson, S.R.; Baron, M.; Tyndall, A.; Matucci-Cerinic, M.; Naden, R.P.; Medsger, T.A., Jr.; Carreira, P.E.; et al. 2013 classification criteria for systemic sclerosis: An American college of rheumatology/European league against rheumatism collaborative initiative. Ann. Rheum. Dis. 2013, 72, 1747–1755. [Google Scholar] [CrossRef] [PubMed]
- Snowden, J.A.; Saccardi, R.; Allez, M.; Ardizzone, S.; Arnold, R.; Cervera, R.; Denton, C.; Hawkey, C.; Labopin, M.; Mancardi, G.; et al. Haematopoietic SCT in severe autoimmune diseases: Updated guidelines of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant. 2012, 47, 770–790. [Google Scholar] [CrossRef] [PubMed]
- Pugnet, G.; Castilla-Llorente, C.; Puyade, M.; Terriou, L.; Badoglio, M.; Deligny, C.; Guillaume-Jugnot, P.; Labeyrie, C.; Benzidia, I.; Faivre, H.; et al. Indications et suivi des autogreffes de cellules souches hématopoïétiques dans les maladies auto-immunes et auto-inflammatoires: Recommandations de la Société francophone de greffe de moelle et de thérapie cellulaire (SFGM-TC). Bull. Cancer 2017, 104, S169–S180. [Google Scholar] [CrossRef] [PubMed]
- Alexander, T.; Bondanza, A.; Muraro, P.; Greco, R.; Saccardi, R.; Daikeler, T.; Kazmi, M.; Hawkey, C.; Simoes, B.P.; Leblanc, K.; et al. SCT for severe autoimmune diseases: Consensus guidelines of the European Society for Blood and Marrow Transplantation for immune monitoring and biobanking. Bone Marrow Transplant. 2015, 50, 173–180. [Google Scholar] [CrossRef]
- Lansiaux, P.; Loisel, S.; Castilla-Llorente, C.; Fontenille, C.; Kabdani, S.; Marjanovic, Z.; Pugnet, G.; Puyade, M.; Robert, E.; Terriou, L.; et al. Autologous hematopoietic cells for severe autoimmune diseases: Guidelines of the Francophone Society of Bone Marrow Transplantation and Cellular Therapy (SFGM-TC) for immune monitoring and biobanking. Bull. Cancer 2021, 108, S72–S81. [Google Scholar] [CrossRef]
- Andersen, M.H.; Fensterle, J.; Ugurel, S.; Reker, S.; Houben, R.; Guldberg, P.; Berger, T.G.; Schadendorf, D.; Trefzer, U.; Bröcker, E.-B.; et al. Immunogenicity of Constitutively Active V599EBRaf. Cancer Res. 2004, 64, 5456–5460. [Google Scholar] [CrossRef]
- Di Cristofaro, J.; El Moujally, D.; Agnel, A.; Mazieres, S.; Cortey, M.; Basire, A.; Chiaroni, J.; Picard, C. HLA-G haplotype structure shows good conservation between different populations and good correlation with high, normal and low soluble HLA-G expression. Hum. Immunol. 2013, 74, 203–206. [Google Scholar] [CrossRef]
- Di Cristofaro, J.; Reynaud-Gaubert, M.; Carlini, F.; Roubertoux, P.; Loundou, A.; Basire, A.; Frassati, C.; Thomas, P.; Gomez, C.; Picard, C. HLA-G*01:04∼UTR3 Recipient Correlates With Lower Survival and Higher Frequency of Chronic Rejection After Lung Transplantation. Am. J. Transplant. 2015, 15, 2413–2420. [Google Scholar] [CrossRef]
- Rebmann, V.; LeMaoult, J.; Rouas-Freiss, N.; Carosella, E.D.; Grosse-Wilde, H. Report of the Wet Workshop for Quantification of Soluble HLA-G in Essen, 2004. Hum. Immunol. 2005, 66, 853–863. [Google Scholar] [CrossRef]
| n (%)/Mean (SD) [Min–Max] | Missing Values (n) | |
|---|---|---|
| Age, years | 45.6 (12.8) [17–66] | |
| Sex | ||
| Male | 17 (37.0) | |
| Female | 29 (63.0) | |
| Geographic origin | ||
| European | 27 (58.7) | |
| Afro-American | 8 (17.4) | |
| North African | 7 (15.2) | |
| Asian | 4 (8.7) | |
| Disease duration, years | 2.3 (1.5) [0.2–6.2] | |
| modified Rodnan Skin Score (mRSS) | 24.9 (10.3) [3–51] | |
| Pulmonary involvement | ||
| FVC, % predicted | 78.0 (17.2) [52–130] | 2 |
| DLCO, % predicted | 52.6 (15.7) [26–90] | |
| Auto-antibodies | ||
| Antinuclear positive | 44 (95.7) | 1 |
| Anti-Scl 70 positive | 29 (63.0) | 1 |
| Anti-centromere positive | 1 (2.2) | 2 |
| 12 Months after AHSCT | 24 Months after AHSCT | |||||||
|---|---|---|---|---|---|---|---|---|
| R, n (%) | NR, n (%) | p-Value R vs. NR | Global p-Value | R, n (%) | NR, n (%) | p-Value R vs. N | Global p-Value | |
| n = 32 | n = 14 | n = 33 | n = 13 | |||||
| Geographic origin | ||||||||
| European | 20 (62.5) | 7 (50) | 0.43 | 20 (60.6) | 7 (53.8) | 0.68 | ||
| Afro-American | 4 (12.5) | 4 (28.6) | 0.19 | 4 (12.1) | 4 (30.8) | 0.20 | ||
| North African | 6 (18.8) | 1 (7.1) | 0.41 | 6 (18.2) | 1 (7.7) | 0.65 | ||
| Asian | 2 (6.2) | 2 (14.3) | 0.37 | 3 (9.1) | 1 (7.7) | 1.00 | ||
| European and North African | 26 (81.3) | 8 (57.1) | 0.16 | 26 (78.8) | 8 (61.5) | 0.23 | ||
| Afro-American and Asian | 6 (18.8) | 6 (42.9) | 7 (21.2) | 5 (38.5) | ||||
| Age at transplantation | ||||||||
| Mean (SD), | 45.6 (12.4) | 45.6 (14.4) | 0.44 | 45.4 (13.0) | 45.9 (12.9) | 0.86 | ||
| Median | 45.8 | 50.6 | 46.8 | 48.9 | ||||
| Min–Max | 20.1–66.4 | 16.7–61.5 | 20.1–66.4 | 16.7–61.1 | ||||
| Sex | ||||||||
| Male | 14 (43.8) | 3 (21.4) | 0.15 | 14 (42.4) | 3 (23.1) | 0.32 | ||
| Female | 18 (56.3) | 11 (78.5) | 19 (57.6) | 10 (76.9) | ||||
| 12 Months after AHSCT | 24 Months after AHSCT | |||||||
|---|---|---|---|---|---|---|---|---|
| R, n (%) | NR, n (%) | p-Value R vs. NR | Global p-Value | R, n (%) | NR, n (%) | p-Value R vs. NR | Global p-Value | |
| HLA-E genotype | n = 32 | n = 12 | n = 32 | n = 12 | ||||
| E*01:01/E*01:01 | 7 (21.9) | 3 (25) | 1.00 | 7 (21.9) | 3 (25) | 1.00 | ||
| E*01:01/E*01:03 | 18 (56.2) | 9 (75) | 0.26 | 18 (56.2) | 9 (75) | 0.26 | ||
| E*01:03/E*01:03 | 7 (21.9) | 0 (0) | 0.16 | 7 (21.9) | 0 (0) | 0.16 | ||
| HLA-E allele | n = 64 | n = 24 | n = 64 | n = 24 | ||||
| E*01:01 | 32 (50) | 15 (62.5) | 0.30 | 32 (50) | 15 (62.5) | 0.30 | ||
| E*01:03 | 32 (50) | 9 (37.5) | 32 (50) | 9 (37.5) | ||||
| HLA-G 14 bp genotype | n = 32 | n = 13 | n = 33 | n = 12 | ||||
| DEL/DEL | 13 (40.1) | 3 (23.1) | 0.32 | 14 (42.4) | 2 (16.7) | 0.16 | ||
| INS/DEL | 13 (40.1) | 7 (53.8) | 0.42 | 13 (39.4) | 7 (58.3) | 0.26 | ||
| INS/INS | 6 (18.8) | 3 (23.1) | 0.70 | 6 (18.2) | 3 (25) | 0.68 | ||
| HLA-G 14 bp allele | n = 64 | n = 26 | n = 66 | n = 24 | ||||
| DEL | 39 (60.9) | 13 (50) | 0.34 | 41 (62.1) | 11 (45.8) | 0.17 | ||
| INS | 25 (39.1) | 13 (50) | 25 (37.9) | 13 (54.2) | ||||
| HLA-C rs2395471 genotype | n = 32 | n = 13 | n = 33 | n = 12 | ||||
| AA | 6 (18.8) | 4 (30.8) | 0.44 | 6 (18.2) | 4 (33.3) | 0.42 | ||
| AG | 21 (65.6) | 6 (46.2) | 0.23 | 23 (69.7) | 4 (33.3) | 0.04 | ||
| GG | 5 (15.6) | 3 (23.1) | 0.67 | 4 (12.1) | 4 (33.3) | 0.18 | ||
| HLA-C rs2395471 allele | n = 64 | n = 26 | n = 66 | n = 24 | ||||
| A | 33 (51.6) | 14 (53.8) | 0.84 | 35 (53) | 12 (50) | 0.80 | ||
| G | 31 (48.4) | 12 (46.2) | 31 (47) | 12 (50) | ||||
| HLA-B peptide leader genotype | n = 32 | n = 13 | n = 33 | n = 12 | ||||
| MM | 3 (9.4) | 0 (0) | 0.19 | 3 (9.1) | 0 (0) | 0.53 | ||
| TM | 8 (25.0) | 8 (61.5) | 10 (30.3) | 6 (50) | ||||
| TT | 21 (65.6) | 5 (38.5) | 20 (60.6) | 6 (50) | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boukouaci, W.; Lansiaux, P.; Lambert, N.C.; Picard, C.; Clave, E.; Cras, A.; Marjanovic, Z.; Farge, D.; Tamouza, R. Non-Classical HLA Determinants of the Clinical Response after Autologous Stem Cell Transplantation for Systemic Sclerosis. Int. J. Mol. Sci. 2022, 23, 7223. https://doi.org/10.3390/ijms23137223
Boukouaci W, Lansiaux P, Lambert NC, Picard C, Clave E, Cras A, Marjanovic Z, Farge D, Tamouza R. Non-Classical HLA Determinants of the Clinical Response after Autologous Stem Cell Transplantation for Systemic Sclerosis. International Journal of Molecular Sciences. 2022; 23(13):7223. https://doi.org/10.3390/ijms23137223
Chicago/Turabian StyleBoukouaci, Wahid, Pauline Lansiaux, Nathalie C. Lambert, Christophe Picard, Emmanuel Clave, Audrey Cras, Zora Marjanovic, Dominique Farge, and Ryad Tamouza. 2022. "Non-Classical HLA Determinants of the Clinical Response after Autologous Stem Cell Transplantation for Systemic Sclerosis" International Journal of Molecular Sciences 23, no. 13: 7223. https://doi.org/10.3390/ijms23137223
APA StyleBoukouaci, W., Lansiaux, P., Lambert, N. C., Picard, C., Clave, E., Cras, A., Marjanovic, Z., Farge, D., & Tamouza, R. (2022). Non-Classical HLA Determinants of the Clinical Response after Autologous Stem Cell Transplantation for Systemic Sclerosis. International Journal of Molecular Sciences, 23(13), 7223. https://doi.org/10.3390/ijms23137223







