Effect of Magnetopriming on Photosynthetic Performance of Plants
Abstract
:1. Introduction
2. Effect of a MF on Photosynthetic Pigments
3. Effect of MF on Chlorophyll Fluorescence
4. Effect of MFs on Photosynthesis
5. Effect of MFs on Photosynthetic Enzymes
6. Effects of Magnetic Field Treatment on Leaf Features
7. Effect of MFs on the Yield of Plants
8. Conclusions and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kanervo, E.; Tasaka, Y.; Murata, N.; Aro, E.-M. Membrane lipid unsaturation modulates processing of the photosystem II reaction-center protein D1 at low temperatures. Plant Physiol. 1997, 114, 841–849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quiles, M.J. Stimulation of chlororespiration by heat and high light intensity in oat plants. Plant Cell Environ. 2006, 29, 1463–1470. [Google Scholar] [CrossRef] [PubMed]
- Battisti, D.S.; Naylor, R.L. Historical warnings of future food insecurity with unprecedented seasonal heat. Science 2009, 323, 240–244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lawlor, D.W.; Tezara, W. Causes of decreased photosynthetic rate and metabolic capacity in water-deficient leaf cells: A critical evaluation of mechanisms and integration of processes. Ann. Bot. 2009, 103, 561–579. [Google Scholar] [CrossRef] [Green Version]
- Kalaji, H.M.; Carpentier, R.; Allakhverdiev, S.I.; Bosa, K. Fluorescence parameters as early indicators of light stress in barley. J. Photochem. Photobiol. B Biol. 2012, 112, 1–6. [Google Scholar] [CrossRef]
- Murata, N.; Allakhverdiev, S.I.; Nishiyama, Y. The mechanism of photoinhibition in vivo: Re-evaluation of the roles of catalase, α-tocopherol, non-photochemical quenching, and electron transport. Biochim. Biophys. Acta (BBA)-Bioenerg. 2012, 1817, 1127–1133. [Google Scholar] [CrossRef] [Green Version]
- Urban, J.; Ingwers, M.W.; McGuire, M.A.; Teskey, R.O. Increase in leaf temperature opens stomata and decouples net photosynthesis from stomatal conductance in Pinus taeda and Populus deltoides x nigra. J. Exp. Bot. 2017, 68, 1757–1767. [Google Scholar] [CrossRef] [PubMed]
- Kataria, S.; Jain, M.; Rastogi, A.; Brestic, M. Static magnetic field treatment enhanced photosynthetic performance in soybean under supplemental ultraviolet-B radiation. Photosynth. Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- Zivcak, M.; Brestic, M.; Balatova, Z.; Drevenakova, P.; Olsovska, K.; Kalaji, H.M.; Yang, X.; Allakhverdiev, S.I. Photosynthetic electron transport and specific photoprotective responses in wheat leaves under drought stress. Photosynth. Res. 2013, 117, 529–546. [Google Scholar] [CrossRef] [PubMed]
- Alte, F.; Stengel, A.; Benz, J.P.; Petersen, E.; Soll, J.; Groll, M.; Bölter, B. Ferredoxin: NADPH oxidoreductase is recruited to thylakoids by binding to a polyproline type II helix in a pH-dependent manner. Proc. Natl. Acad. Sci. USA 2010, 107, 19260–19265. [Google Scholar] [CrossRef] [Green Version]
- Benz, J.P.; Lintala, M.; Soll, J.; Mulo, P.; Bölter, B. A new concept for ferredoxin–NADP (H) oxidoreductase binding to plant thylakoids. Trends Plant Sci. 2010, 15, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Kalaji, H.M.; Jajoo, A.; Oukarroum, A.; Brestic, M.; Zivcak, M.; Samborska, I.A.; Cetner, M.D.; Łukasik, I.; Goltsev, V.; Ladle, R.J. Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol. Plant. 2016, 38, 102. [Google Scholar] [CrossRef] [Green Version]
- Cruz, J.A.; Avenson, T.J.; Kanazawa, A.; Takizawa, K.; Edwards, G.E.; Kramer, D.M. Plasticity in light reactions of photosynthesis for energy production and photoprotection. J. Exp. Bot. 2005, 56, 395–406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Price, C.; Williams, E.; Elhalel, G.; Sentman, D. Natural ELF fields in the atmosphere and in living organisms. Int. J. Biometeorol. 2021, 65, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Sarraf, M.; Kataria, S.; Taimourya, H.; Santos, L.O.; Menegatti, R.D.; Jain, M.; Ihtisham, M.; Liu, S. Magnetic field (MF) applications in plants: An overview. Plants 2020, 9, 1139. [Google Scholar] [CrossRef]
- Maffei, M.E. Magnetic field effects on plant growth, development, and evolution. Front. Plant Sci. 2014, 5, 445. [Google Scholar] [CrossRef] [Green Version]
- Islam, M.; Vigani, G.; Maffei, M.E. The Geomagnetic Field (GMF) Modulates Nutrient Status and Lipid Metabolism during Arabidopsis thaliana Plant Development. Plants 2020, 9, 1729. [Google Scholar] [CrossRef]
- Islam, M.; Maffei, M.E.; Vigani, G. The geomagnetic field is a contributing factor for an efficient iron uptake in Arabidopsis thaliana. Front. Plant Sci. 2020, 11, 325. [Google Scholar] [CrossRef] [Green Version]
- Shine, M.; Guruprasad, K.; Anand, A. Enhancement of germination, growth, and photosynthesis in soybean by pre-treatment of seeds with magnetic field. Bioelectromagnetics 2011, 32, 474–484. [Google Scholar] [CrossRef]
- Agliassa, C.; Narayana, R.; Christie, J.M.; Maffei, M.E. Geomagnetic field impacts on cryptochrome and phytochrome signaling. J. Photochem. Photobiol. B Biol. 2018, 185, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Baghel, L.; Kataria, S.; Guruprasad, K. Effect of static magnetic field pretreatment on growth, photosynthetic performance and yield of soybean under water stress. Photosynthetica 2018, 56, 718–730. [Google Scholar] [CrossRef]
- Baghel, L.; Kataria, S.; Jain, M. Mitigation of adverse effects of salt stress on germination, growth, photosynthetic efficiency and yield in maize (Zea mays L.) through magnetopriming. Acta Agrobot. 2019, 72, 1757. [Google Scholar] [CrossRef] [Green Version]
- Kataria, S.; Baghel, L.; Jain, M.; Guruprasad, K. Magnetopriming regulates antioxidant defense system in soybean against salt stress. Biocatal. Agric. Biotechnol. 2019, 18, 101090. [Google Scholar] [CrossRef]
- Fatima, A.; Kataria, S.; Prajapati, R.; Jain, M.; Agrawal, A.K.; Singh, B.; Kashyap, Y.; Tripathi, D.K.; Singh, V.P.; Gadre, R. Magnetopriming effects on arsenic stress-induced morphological and physiological variations in soybean involving synchrotron imaging. Physiol. Plant. 2020, 173, 88–99. [Google Scholar] [CrossRef]
- Rochalska, M. Influence of frequent magnetic field on chlorophyll content in leaves of sugar beet plants. Nukleonika 2005, 50, 25–28. [Google Scholar]
- Kataria, S.; Baghel, L.; Guruprasad, K. Pre-treatment of seeds with static magnetic field improves germination and early growth characteristics under salt stress in maize and soybean. Biocatal. Agric. Biotechnol. 2017, 10, 83–90. [Google Scholar] [CrossRef]
- Baghel, L.; Kataria, S.; Guruprasad, K.N. Static magnetic field treatment of seeds improves carbon and nitrogen metabolism under salinity stress in soybean. Bioelectromagnetics 2016, 37, 455–470. [Google Scholar] [CrossRef] [PubMed]
- Taimourya, H.; Oussible, M.; Baamal, L.; Bourarach, E.; Hassanain, N.; Masmoudi, L.; El-Harif, A. Effect of magnetically treated water on strawberry plants (Fragaria× ananassa Duch.) in the northwest of Morocco. In Proceedings of the VIII International Scientific Agriculture Symposium, “Agrosym 2017”, Jahorina, Bosnia and Herzegovina, 5–8 October 2017; pp. 382–387. [Google Scholar]
- Carbonell, M.V.; Martinez, E.; Amaya, J.M. Stimulation of germination in rice (Oryza sativa L.) by a static magnetic field. Electro-Magn. 2000, 19, 121–128. [Google Scholar] [CrossRef]
- Taimourya, H.; Oussible, M.; Baamal, L.; Bourarach, E.H.; Hassanain, N.; Masmoudi, L.; El Harif, A. Magnetically treated irrigation water improves the production and the fruit quality of strawberry plants (Fragaria× ananassa Duch.) in the northwest of Morocco. J. Agric. Sci. Technol. 2018, 8, 145–156. [Google Scholar] [CrossRef] [Green Version]
- Dhawi, F.; Al-Khayri, J.M. Magnetic fields induce changes in photosynthetic pigments content in date palm (Phoenix dactylifera L.) seedlings. Open Agric. J. 2009, 3, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Taimourya, H.; Oussible, M.; Baamal, L.; Harif, A.; Zaid, E.; Guedira, A.; Smouni, A. Magnetic Treatment of Culture Medium Enhance Growth and Minerals Uptake of Strawberry (Fragaria× ananassa Duch.) and Tomato (Solanum lycopersicum) in Fe Deficiency Conditions. Int. J. Sci. Eng. Res. 2017, 8, 1414–1436. [Google Scholar]
- Anand, A.; Nagarajan, S.; Verma, A.; Joshi, D.; Pathak, P.; Bhardwaj, J. Pre-treatment of seeds with static magnetic field ameliorates soil water stress in seedlings of maize (Zea mays L.). Indian J. Biochem. Biol. 2012, 49, 63–70. Available online: http://nopr.niscair.res.in/handle/123456789/13593 (accessed on 1 February 2021).
- Abdul Qados, A.; Hozayn, M. Response of growth, yield, yield components and some chemical constituents of flax for irrigation with magnetized and tap water. World Appl. Sci. J. 2010, 8, 630–634. [Google Scholar]
- De Souza, A.; Garcí, D.; Sueiro, L.; Gilart, F.; Porras, E.; Licea, L. Pre-sowing magnetic treatments of tomato seeds increase the growth and yield of plants. Bioelectromagnetics 2006, 27, 247–257. [Google Scholar] [CrossRef]
- El Sayed, H.E.S.A. Impact of magnetic water irrigation for improve the growth, chemical composition and yield production of broad bean (Vicia faba L.) plant. J. Exp. Agric. Int. 2014, 4, 476–496. [Google Scholar] [CrossRef]
- Oldacay, S.; Erdem, G. Evaluation of Chlorophyll Contents and Peroxidase Activities in Helianthus annuus Genotypes Exposed to Radiation and Magnetic Fields. J. Appl. Sci. 2002, 2, 934–937. [Google Scholar] [CrossRef] [Green Version]
- Racuciu, M.; Miclaus, S.; Creanga, D. The response of plant tissues to magnetic fluid and electromagnetic exposure. Rom. J. Biophys. 2009, 19, 73–83. Available online: https://www.rjb.ro/articles/229/art07Racuciu.pdf (accessed on 1 March 2021).
- Rakosy-Tican, L.; Aurori, C.; Morariu, V. Influence of near null magnetic field on in vitro growth of potato and wild Solanum species. Bioelectromagnetics 2005, 26, 548–557. [Google Scholar] [CrossRef]
- Tahir, N.A.; Karim, H.F.H. Impact of magnetic application on the parameters related to growth of chickpea (Cicer arietinum L.). Jordan J. Biol. Sci. 2010, 3, 175–184. [Google Scholar]
- Voica, C.; Polescu, L.; Lazar, D. The influence of the magnetic fluids on some physiological processes in Phaseolus vulgaris. Rev. Roum. Biol. 2003, 48, 9–15. [Google Scholar]
- Çelik, Ö.; Atak, Ç.; Rzakulieva, A. Stimulation of rapid regeneration by a magnetic field in paulownia node cultures. J. Cent. Eur. Agric. 2008, 9, 297–304. Available online: https://jcea.agr.hr/en/issues/article/525 (accessed on 20 March 2021).
- Atak, Ç.; Yurttaş, B.; Yalçın, S.; Mutlu, D.; Rzakoulieva, A.; Danilov, V. Effects of Magnetic Field on soybean (Glycine max. L. Merrill) seeds. Com. JINR. Dubna 1997. 1–13. Available online: https://hdl.handle.net/11413/2851 (accessed on 20 March 2021).
- Shine, M.; Guruprasad, K. Impact of pre-sowing magnetic field exposure of seeds to stationary magnetic field on growth, reactive oxygen species and photosynthesis of maize under field conditions. Acta Physiol. Plant. 2012, 34, 255–265. [Google Scholar] [CrossRef]
- Turker, M.; Temirci, C.; Battal, P.; Erez, M.E. The effects of an artificial and static magnetic field on plant growth, chlorophyll and phytohormone levels in maize and sunflower plants. Phyton. Ann. Rei Bot. 2007, 46, 271–284. Available online: https://www.zobodat.at/pdf/PHY_46_2_0271-0284.pdf (accessed on 2 February 2021).
- Radhakrishnan, R.; Kumari, B.D.R. Influence of pulsed magnetic field on soybean (Glycine max L.) seed germination, seedling growth and soil microbial population. Indian J. Biochem. Biophys. 2013, 50, 312–317. Available online: http://nopr.niscair.res.in/bitstream/123456789/20879/1/IJBB%2050(4)%20312-317.pdf (accessed on 2 July 2020). [PubMed]
- Asghar, T.; Iqbal, M.; Jamil, Y.; Nisar, J.; Shahid, M. Comparison of HeNe laser and sinusoidal non-uniform magnetic field seed pre-sowing treatment effect on Glycine max (Var 90-I) germination, growth and yield. J. Photochem. Photobiol. B Biol. 2017, 166, 212–219. [Google Scholar] [CrossRef]
- Chen, Y.-P.; Li, R.; He, J.-M. Magnetic field can alleviate toxicological effect induced by cadmium in mungbean seedlings. Ecotoxicology 2011, 20, 760–769. [Google Scholar] [CrossRef]
- Kataria, S.; Baghel, L.; Guruprasad, K. Alleviation of adverse effects of ambient UV stress on growth and some potential physiological attributes in soybean (Glycine max) by seed pre-treatment with static magnetic field. J. Plant Growth Regul. 2017, 36, 550–565. [Google Scholar] [CrossRef]
- Kataria, S.; Rastogi, A.; Bele, A.; Jain, M. Role of nitric oxide and reactive oxygen species in static magnetic field pre-treatment induced tolerance to ambient UV-B stress in soybean. Physiol. Mol. Biol. Plants 2020, 26, 931–945. [Google Scholar] [CrossRef] [PubMed]
- Javed, N.; Ashraf, M.; Akram, N.A.; Al-Qurainy, F. Alleviation of adverse effects of drought stress on growth and some potential physiological attributes in maize (Zea mays L.) by seed electromagnetic treatment. Photochem. Photobiol. 2011, 87, 1354–1362. [Google Scholar] [CrossRef] [PubMed]
- Aladjadjiyan, A.; Ylieva, T. Influence of stationary magnetic field on the early stages of the development of tobacco seeds (Nicotiana tabacum L.). J. Cent. Eur. Agric. 2003, 4, 131–138. Available online: https://jcea.agr.hr/en/issues/article/124 (accessed on 1 March 2021).
- Campbell, G.S.; Norman, J. An introduction to Environmental Biophysics; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar] [CrossRef]
- Strzałka, K.; Kostecka-Gugała, A.; Latowski, D. Carotenoids and environmental stress in plants: Significance of carotenoid-mediated modulation of membrane physical properties. Russ. J. Plant Physiol. 2003, 50, 168–173. [Google Scholar] [CrossRef]
- Abdolmaleki, P.; Ghanati, F.; Sahebjamei, H.; Sarvestani, A.S. Peroxidase activity, lignification and promotion of cell death in tobacco cells exposed to static magnetic field. Environmentalist 2007, 27, 435–440. [Google Scholar] [CrossRef]
- Racuciu, M.; Creanga, D.; Amoraritei, C. Biochemical changes induced by low frequency magnetic field exposure of vegetal organisms. Rom. J. Phys. 2007, 52, 601–606. Available online: https://rjp.nipne.ro/2007_52_5-6/0645_0652.pdf (accessed on 13 March 2021).
- Racuciu, M.; Creanga, D.; Galugaru, C. The influence of extremely low frequency magnetic field on tree seedlings. Rom. J. Phys. 2008, 35, 337–342. Available online: https://rjp.nipne.ro/2008_53_1-2/0361_0368.pdf (accessed on 13 March 2021).
- Taia, W.K.; Kotbi, A.M.; AlZahrani, H.S. The effect of static magnetic forces on water contents and photosynthetic pigments in sweet basil Ocimum basilicum L. (Lamiaceae). Saudi J. Biol. Sci. 2007, 14, 103–107. Available online: https://inis.iaea.org/search/searchsinglerecord.aspx?recordsFor=SingleRecord&RN=40087321 (accessed on 6 March 2021).
- Strasser, R.J.; Tsimilli-Michael, M.; Srivastava, A. Analysis of the chlorophyll a fluorescence transient. In Chlorophyll a Fluorescence; Springer: Berlin/Heidelberg, Germany, 2004; pp. 321–362. [Google Scholar] [CrossRef]
- Schreiber, U.; Bilger, W.; Neubauer, C. Chlorophyll fluorescence as a nonintrusive indicator for rapid assessment of in vivo photosynthesis. In Ecophysiology of Photosynthesis; Springer: Berlin/Heidelberg, Germany, 1995; pp. 49–70. [Google Scholar] [CrossRef]
- Tsimilli-Michael, M.; Strasser, R.J. The energy flux theory 35 years later: Formulations and applications. Photosynth. Res. 2013, 117, 289–320. [Google Scholar] [CrossRef]
- Deamici, K.M.; Cuellar-Bermudez, S.P.; Muylaert, K.; Santos, L.O.; Costa, J.A.V. Quantum yield alterations due to the static magnetic fields action on Arthrospira platensis SAG 21.99: Evaluation of photosystem activity. Bioresour. Technol. 2019, 292, 121945. [Google Scholar] [CrossRef] [PubMed]
- Markou, G.; Muylaert, K. Effect of light intensity on the degree of ammonia toxicity on PSII activity of Arthrospira platensis and Chlorella vulgaris. Bioresour. Technol. 2016, 216, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Benavides, A.M.S.; Ranglová, K.; Malapascua, J.R.; Masojídek, J.; Torzillo, G. Diurnal changes of photosynthesis and growth of Arthrospira platensis cultured in a thin-layer cascade and an open pond. Algal Res. 2017, 28, 48–56. [Google Scholar] [CrossRef]
- Voznyak, V.M.; Ganago, I.B.; Moskalenko, A.A.; Elfimov, E.I. Magnetic field-induced fluorescence changes in chlorophyll-proteins enriched with P-700. Biochim. Biophys. Acta (BBA)-Bioenerg. 1980, 592, 364–368. [Google Scholar] [CrossRef]
- Schansker, G.; Tóth, S.Z.; Strasser, R.J. Dark recovery of the Chl a fluorescence transient (OJIP) after light adaptation: The qT-component of non-photochemical quenching is related to an activated photosystem I acceptor side. Biochim. Biophys. Acta (BBA)-Bioenerg. 2006, 1757, 787–797. [Google Scholar] [CrossRef] [Green Version]
- Stirbet, A.; Lazár, D.; Kromdijk, J. Chlorophyll a fluorescence induction: Can just a one-second measurement be used to quantify abiotic stress responses? Photosynthetica 2018, 56, 86–104. [Google Scholar] [CrossRef]
- Ripley, B.S.; Redfern, S.P.; Dames, J. Quantification of the photosynthetic performance of phosphorus-deficient Sorghum by means of chlorophyll-a fluorescence kinetics. S. Afr. J. Sci. 2004, 100, 615–618. Available online: https://hdl.handle.net/10520/EJC96177 (accessed on 2 March 2021).
- Kataria, S.; Jain, M. Magnetopriming Alleviates Adverse Effects of Abiotic Stresses in Plants. In Plant Tolerance to Environmental Stress; CRC Press: Boca Raton, FL, USA, 2019; pp. 427–442. [Google Scholar] [CrossRef]
- Maurino, V.G.; Weber, A.P. Engineering photosynthesis in plants and synthetic microorganisms. J. Exp. Bot. 2013, 64, 743–751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, A.D.; Datta, S.P.; Smith, G.H. Oxford Dictionary of Biochemistry and Molecular Biology; Oxford University Press: Oxford, UK, 1997. [Google Scholar]
- Zhao, B.; Su, Y. Process effect of microalgal-carbon dioxide fixation and biomass production: A review. Renew. Sustain. Energy Rev. 2014, 31, 121–132. [Google Scholar] [CrossRef]
- Calvin, M. Forty years of photosynthesis and related activities. Photosynth. Res. 1989, 21, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Iverson, T.M. Evolution and unique bioenergetic mechanisms in oxygenic photosynthesis. Curr. Opin. Chem. Biol. 2006, 10, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Červený, J.; Šetlík, I.; Trtílek, M.; Nedbal, L. Photobioreactor for cultivation and real-time, in-situ measurement of O2 and CO2 exchange rates, growth dynamics, and of chlorophyll fluorescence emission of photoautotrophic microorganisms. Eng. Life Sci. 2009, 9, 247–253. [Google Scholar] [CrossRef]
- Vian, A.; Davies, E.; Gendraud, M.; Bonnet, P. Plant responses to high frequency electromagnetic fields. BioMed Res. Int. 2016, 2016, 1830262. [Google Scholar] [CrossRef] [Green Version]
- Deamici, K.M.; Santos, L.O.; Costa, J.A.V. Magnetic field as promoter of growth in outdoor and indoor assays of Chlorella fusca. Bioprocess Biosyst. Eng. 2021, 44, 1453–1460. [Google Scholar] [CrossRef]
- Krylov, A.; Tarakonova, G.A. Plant physiology. Fiziol. Rost. 1960, 7, 156. [Google Scholar]
- Martinez, E.; Carbonell, M.V.; Amaya, J.M. A static magnetic field of 125 mT stimulates the initial growth stages of barley (Hordeum vulgare L.). Electro- Magn. 2000, 19, 271–277. [Google Scholar] [CrossRef]
- Kataria, S.; Baghel, L.; Guruprasad, K.N. Effect of seed pretreatment by magnetic field on the sensitivity of maize seedlings to ambient ultraviolet radiation (280–400 nm). Int. J. Trop. Agric. 2015, 33, 3645–3652. Available online: https://www.cabdirect.org/cabdirect/abstract/20173067937 (accessed on 2 March 2020).
- Pittman, U. Magnetism and plant growth: I effect on germination and early growth of cereal seeds. Can. J. Plant Sci. 1963, 43, 513–518. [Google Scholar] [CrossRef]
- Galland, P.; Pazur, A. Magnetoreception in plants. J. Plant Res. 2005, 118, 371–389. [Google Scholar] [CrossRef]
- Rathod, G.R.; Anand, A. Effect of seed magneto-priming on growth, yield and Na/K ratio in wheat (Triticum aestivum L.) under salt stress. Indian J. Plant Physiol. 2016, 21, 15–22. [Google Scholar] [CrossRef]
- Long, S.P.; Zhu, X.G.; Naidu, S.L.; Ort, D.R. Can improvement in photosynthesis increase crop yields? Plant Cell Environ. 2006, 29, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Patel, P. Impact of Magneto-Priming of Seeds with Special Reference to Photosynthesis and Yield of Soybean and Maize. Ph.D Thesis, School of Life Sciences, Devi Ahilya Vishwavidyalaya, Indore, India, 2020. [Google Scholar]
- Mathan, J.; Bhattacharya, J.; Ranjan, A. Enhancing crop yield by optimizing plant developmental features. Development 2016, 143, 3283–3294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fatima, A.; Kataria, S.; Baghel, L.; Guruprasad, K.; Agrawal, A.; Singh, B.; Sarkar, P.; Shripathi, T.; Kashyap, Y. Synchrotron-based phase-sensitive imaging of leaves grown from magneto-primed seeds of soybean. J. Synchrotron Radiat. 2017, 24, 232–239. [Google Scholar] [CrossRef]
- Fatima, A.; Kataria, S.; Agrawal, A.K.; Singh, B.; Kashyap, Y.; Jain, M.; Brestic, M.; Allakhverdiev, S.I.; Rastogi, A. Use of Synchrotron Phase-Sensitive Imaging for the Investigation of Magnetopriming and Solar UV-Exclusion Impact on Soybean (Glycine max) Leaves. Cells 2021, 10, 1725. [Google Scholar] [CrossRef]
- Afzal, I.; Noor, M.; Bakhtavar, M.; Ahmad, A.; Haq, Z. Improvement of spring maize performance through physical and physiological seed enhancements. Seed Sci. Technol. 2015, 43, 238–249. [Google Scholar] [CrossRef]
- Gupta, M.K.; Anand, A.; Paul, V.; Dahuja, A.; Singh, A. Reactive oxygen species mediated improvement in vigour of static and pulsed magneto-primed cherry tomato seeds. Indian J. Plant Physiol. 2015, 20, 197–204. [Google Scholar] [CrossRef]
- Raipuria, R.K.; Kataria, S.; Watts, A.; Jain, M. Magneto-priming promotes nitric oxide via nitric oxide synthase to ameliorate the UV-B stress during germination of soybean seedlings. J. Photochem. Photobiol. B Biol. 2021, 220, 112211. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Ramakrishnan, R.S.; Anand, A. Growth, Na+/K+ Partitioning and Yield of Chickpea Plants Alleviated From Salt Stress by Magnetopriming. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 821–833. [Google Scholar] [CrossRef]
- Maheshwari, B.L.; Grewal, H.S. Magnetic treatment of irrigation water: Its effects on vegetable crop yield and water productivity. Agric. Water Manag. 2009, 96, 1229–1236. [Google Scholar] [CrossRef]
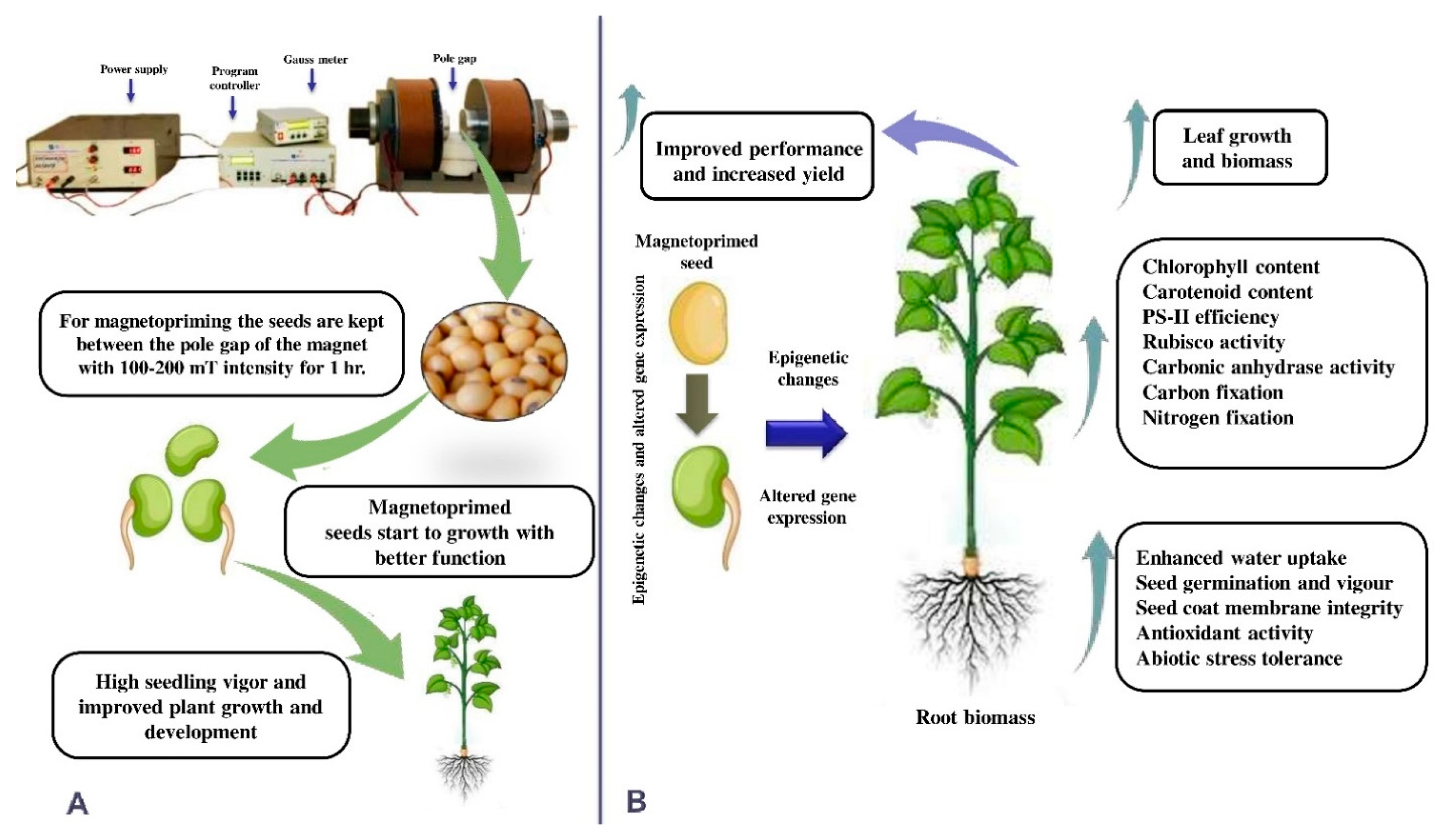
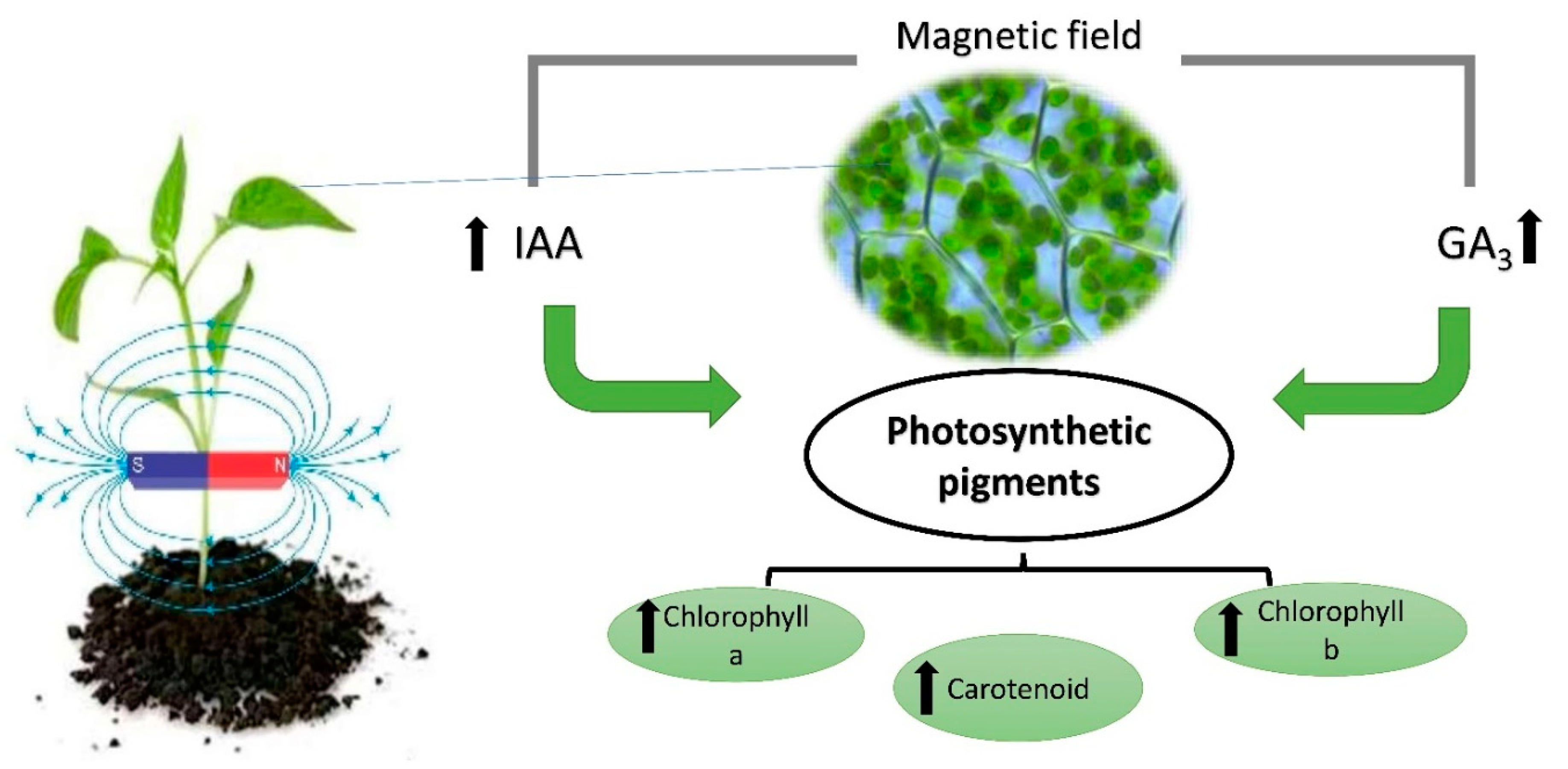
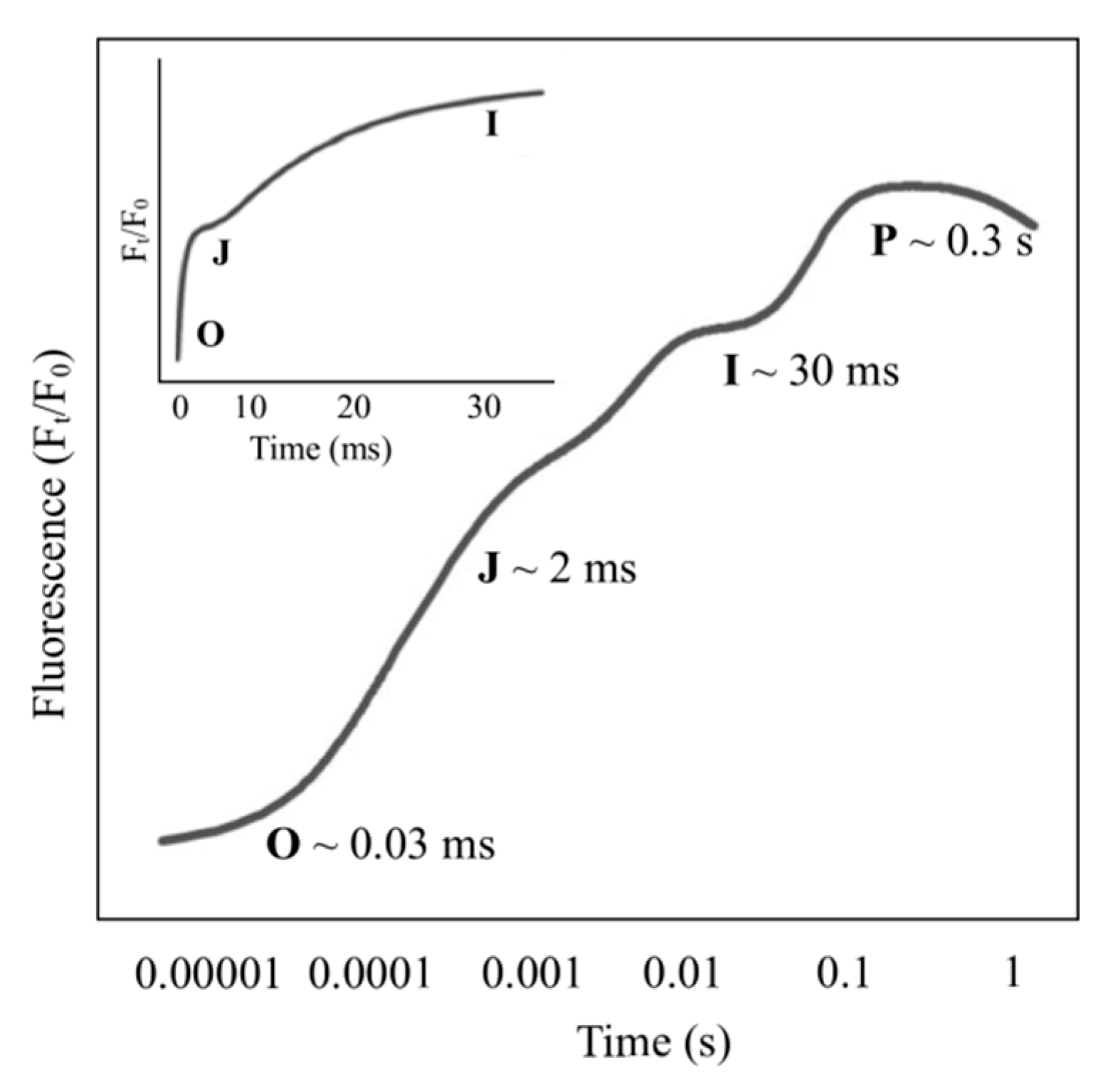

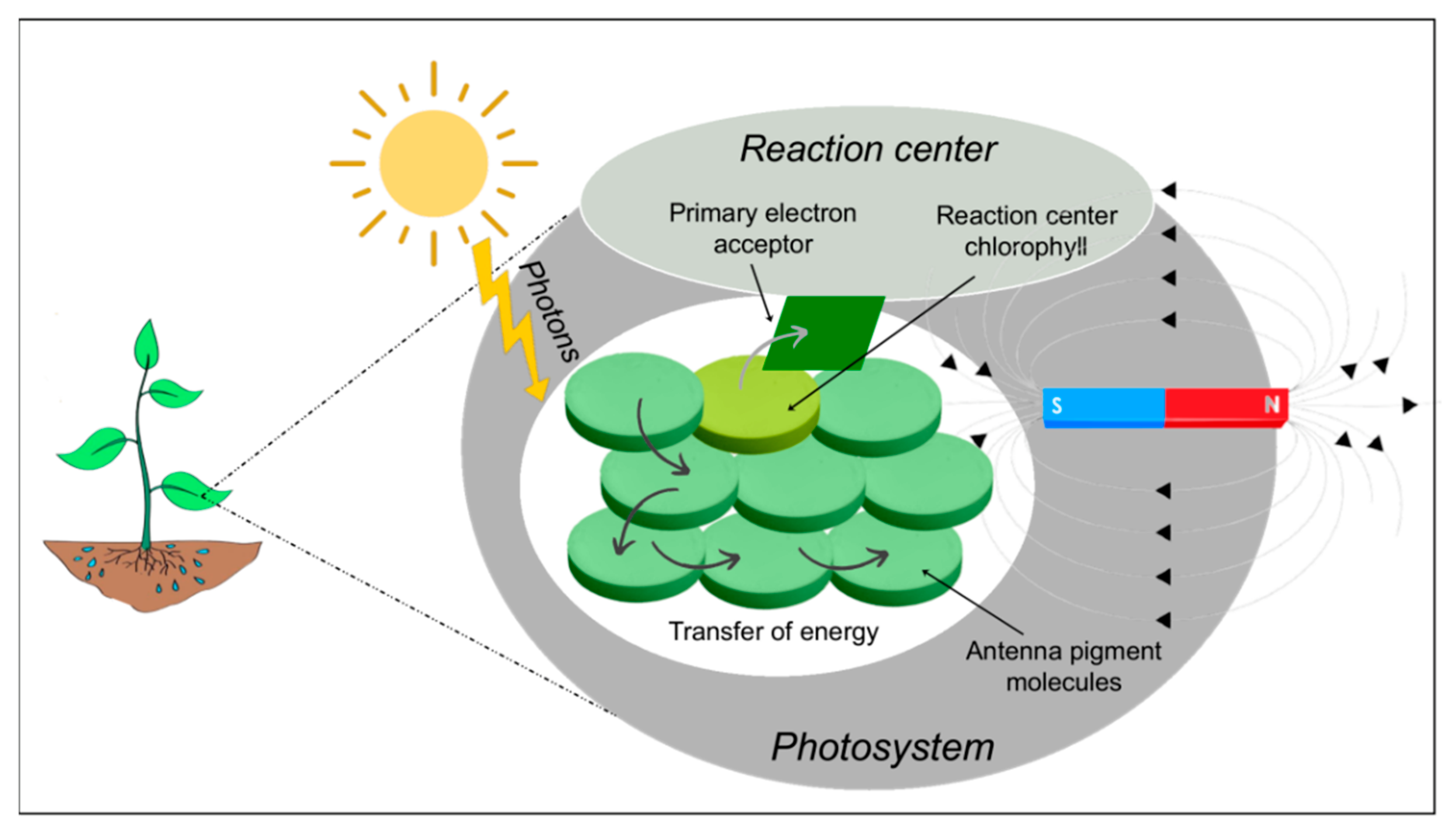
| MF Optimal Intensity | Stress Condition | Plant Species | Effects | References |
|---|---|---|---|---|
| 200 mT | Water stress | Glycine max L. | Increased the plant growth attributes, photosynthetic performance, biomass accumulation, and crop yield | [21] |
| 200 mT | Salinity stress | Glycine max L. | An enhancement of growth attributes, photosynthetic performance and crop yield | [27] |
| 200 mT | Salinity stress | Zea mays L. | Enhanced seedling vigor growth parameters, PSII photochemistry (Fv/Fm) and crop yield | [22] |
| 200 mT | Arsenic (As) toxicity tolerance | Glycine max L. | Reduced As toxicity and increased plant growth parameters with noticeable increase in water uptake, stomatal conductance, PSII performance and photosynthesis | [24] |
| 200 mT | Ultraviolet-B radiation tolerant | Glycine max L. | Increased photosynthetic performance along with higher crop yield, decreased H2O2 content and antioxidant levels | [8] |
| 200 mT | Ultraviolet-B radiation tolerant | Glycine max L. | Significant enhancement in growth parameters and higher expression of genes related to amylase, NR and NOS enzymes | [91] |
| 50 mT | Salinity stress | Triticum aestivum L. | Increased total chlorophyll contents and Na+/K+ ratio and growth attributes | [84] |
| 100 mT | Salinity stress | Cicer arietinum L. | Enhanced physiological traits, antioxidant activity and Na+/K+ ratio, overall growth attributes | [92] |
| 136 mT | Drought stress | Pisum sativum L. Apium graveolens L. | Increased yield and physiological parameters along with Na+/K+ ratio | [93] |
| 150 mT | Drought stress | Zea mays L. | An enhancement of chlorophyll a and b pigments, leaf water potential, stomatal conductance and decreased total proline contents | [51] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarraf, M.; Deamici, K.M.; Taimourya, H.; Islam, M.; Kataria, S.; Raipuria, R.K.; Abdi, G.; Brestic, M. Effect of Magnetopriming on Photosynthetic Performance of Plants. Int. J. Mol. Sci. 2021, 22, 9353. https://doi.org/10.3390/ijms22179353
Sarraf M, Deamici KM, Taimourya H, Islam M, Kataria S, Raipuria RK, Abdi G, Brestic M. Effect of Magnetopriming on Photosynthetic Performance of Plants. International Journal of Molecular Sciences. 2021; 22(17):9353. https://doi.org/10.3390/ijms22179353
Chicago/Turabian StyleSarraf, Mohammad, Kricelle Mosquera Deamici, Houda Taimourya, Monirul Islam, Sunita Kataria, Ritesh Kumar Raipuria, Gholamreza Abdi, and Marian Brestic. 2021. "Effect of Magnetopriming on Photosynthetic Performance of Plants" International Journal of Molecular Sciences 22, no. 17: 9353. https://doi.org/10.3390/ijms22179353










