Vascular and Neuronal Protection in the Developing Retina: Potential Therapeutic Targets for Retinopathy of Prematurity
Abstract
1. Introduction
2. Vasculature in the Retina
2.1. Normal Development of the Retinal Vasculature
2.2. Pathogenesis of Retinopathy of Prematurity
2.2.1. Vascular Endothelial Growth Factor (VEGF)
2.2.2. Insulin-Like Growth Factor-1 (IGF-1)
2.2.3. Erythropoietin (Epo)
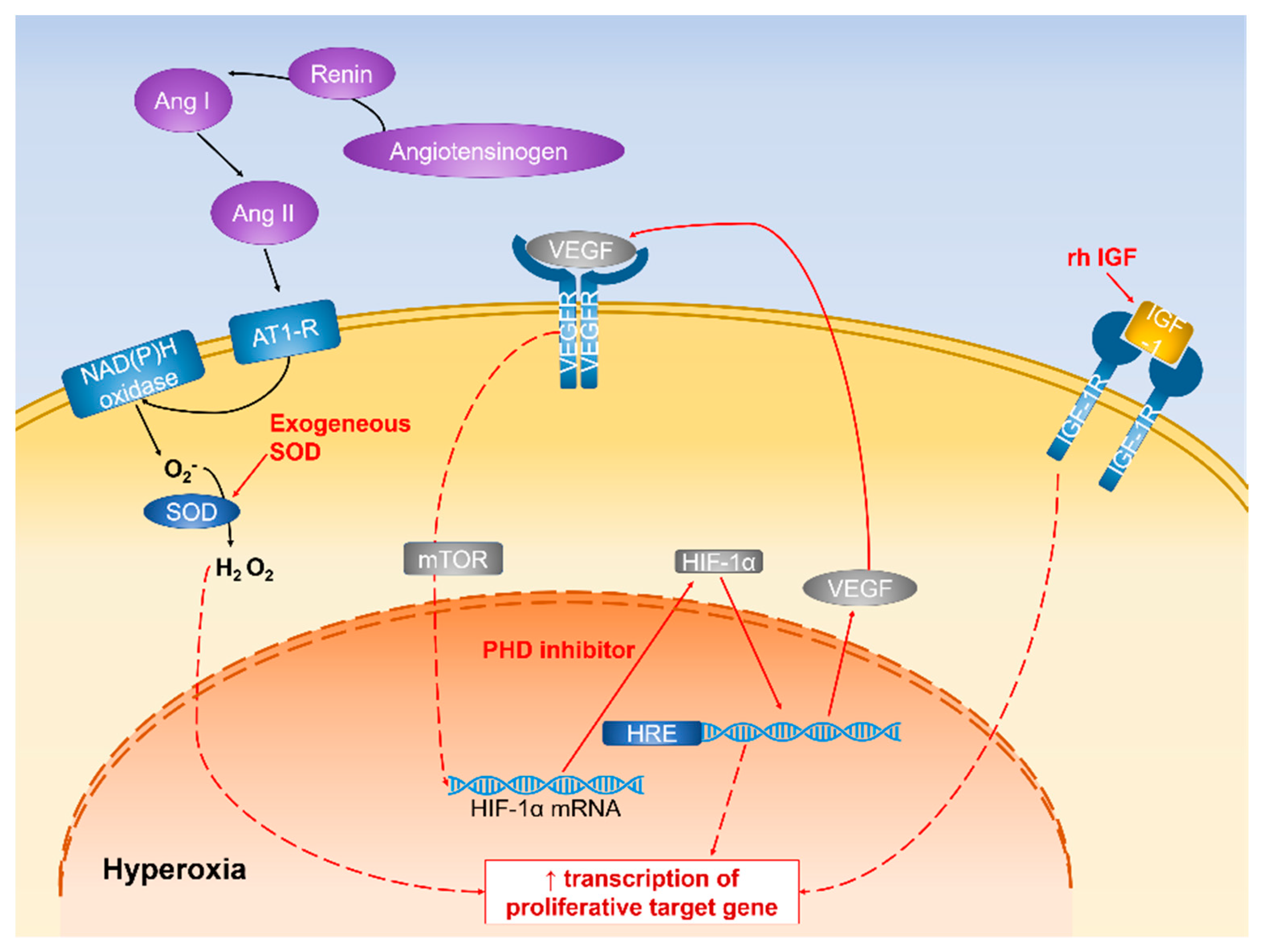
2.2.4. Hypoxia-Inducible Factor-1 (HIF-1)
2.2.5. Nitric Oxide (NO)
2.2.6. Adenosine
2.2.7. β-Adrenergic Receptor (β-AR)
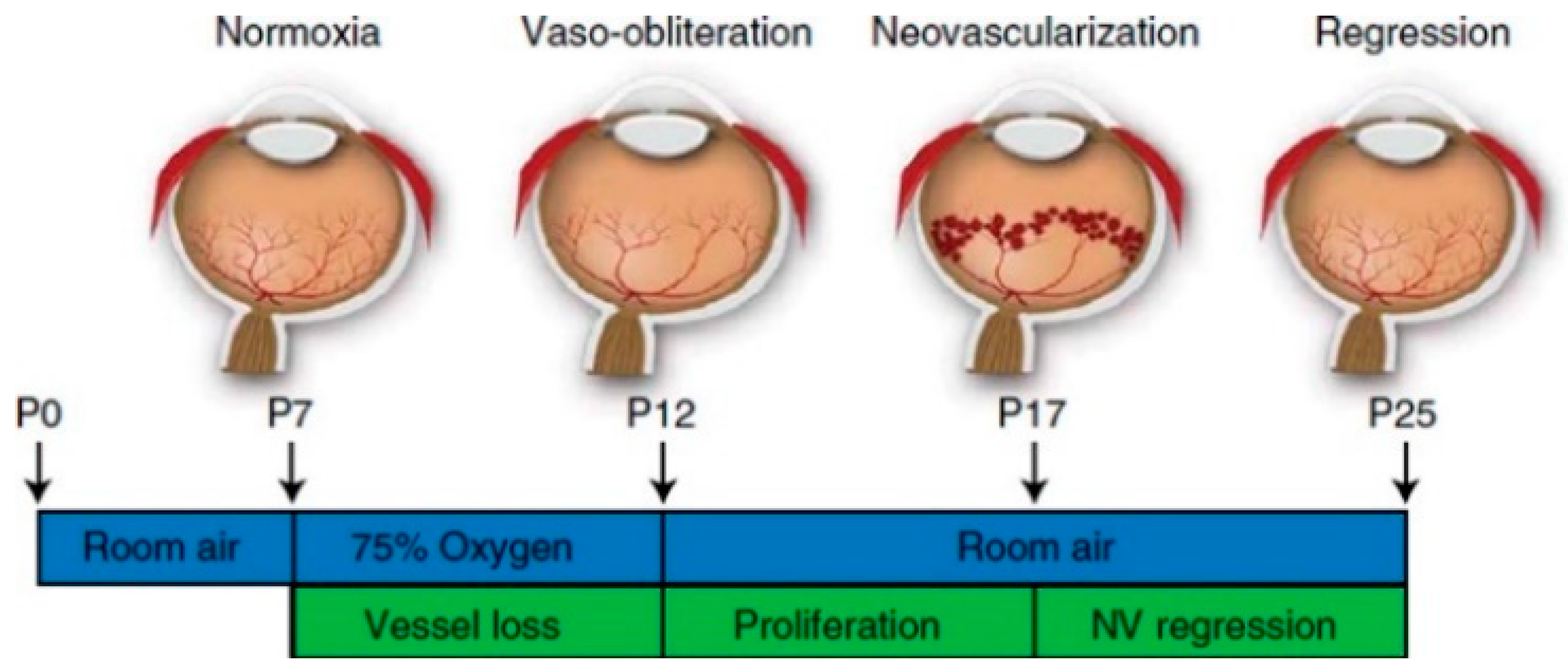
3. Animal Models for ROP—Oxygen-Induced Retinopathy (OIR)
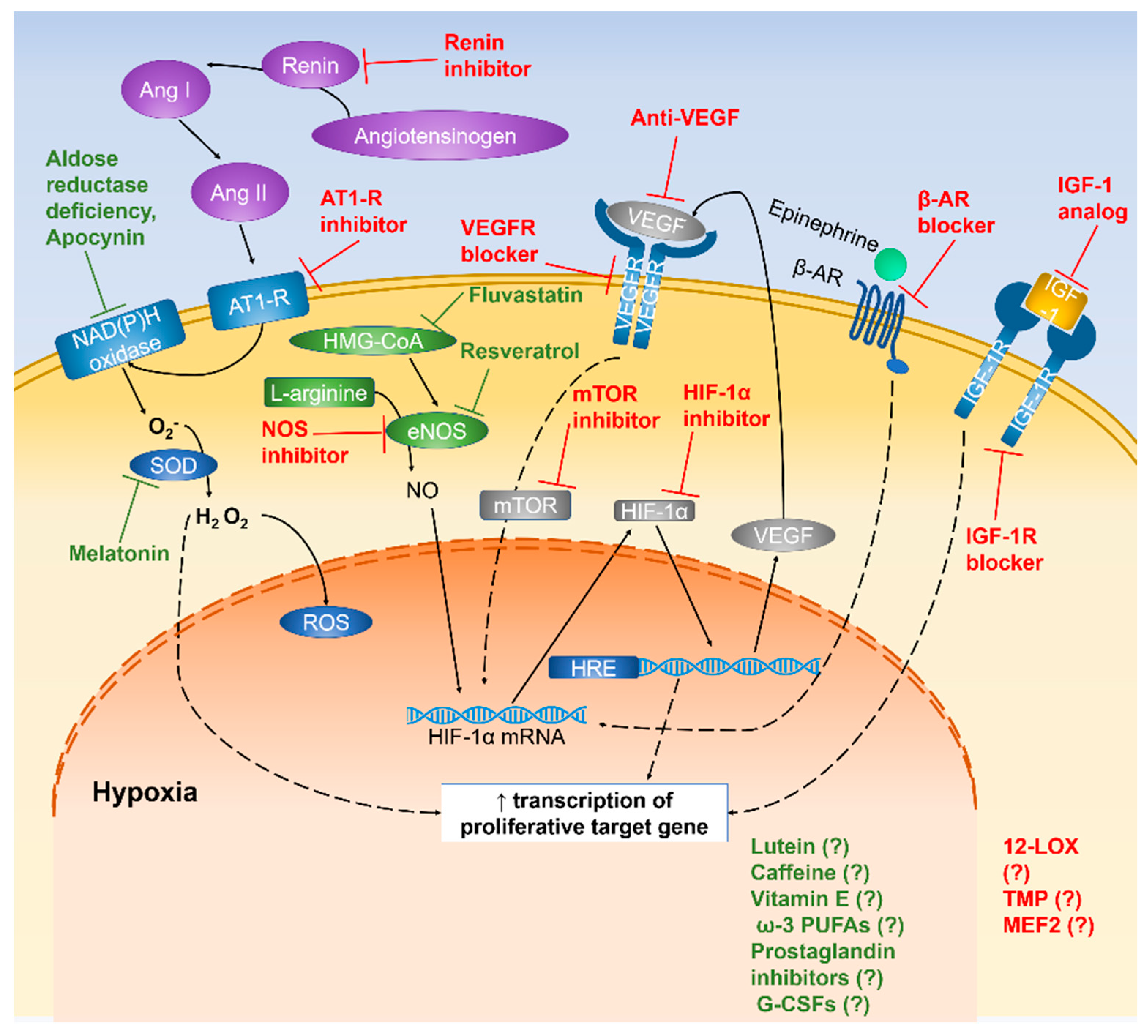
4. Vascular Protection in the ROP
4.1. Growth Factors
4.1.1. Anti-VEGF
4.1.2. IGF-Binding Protein (IGFBP)
4.2. Transcription Factors
4.2.1. Regulation of HIF-1α Expression
4.2.2. Inhibitory Effect of NOS Expression
4.2.3. Blockage of β-ARs
4.3. Anti-Angiogenesis
4.3.1. Steroid Agents
4.3.2. Other Angiogenic Inhibitors
5. Neuroprotective Agents in ROP
5.1. Antioxidants
5.1.1. Nutritional Antioxidants
Lutein
Caffeine
Omega-3 Long-Chain Polyunsaturated Fatty Acids (ω-3 PUFAs)
Resveratrol
Vitamin E
5.1.2. Endogenous Antioxidants
Suppression of Aldose Reductase
Superoxide Dismutase (SOD)
Statin
Melatonin
Apocynin
5.2. Anti-Inflammatory Agents
5.2.1. Prostaglandin Inhibitors
5.2.2. Granulocyte Colony-Stimulating Factor (G-CSF)
5.3. Others
Inhibition of Renin-Angiotensin System (RAS)
6. Stem Cell Therapy in ROP
7. Current Treatments in ROP
8. Conclusions and Future Perspectives
Funding
Conflicts of Interest
Abbreviations
| 5′ N | 5′ nucleotidase |
| AD | Alzheimer’s disease |
| AMD | Age-related macular degeneration |
| AT1-R | Angiotensin II type-1 receptor |
| β-AR | β-adrenergic receptor |
| BRB | Blood-retinal barrier |
| COX | Cyclooxygenase |
| DHA | Docosahexaenoic |
| DR | Diabetic retinopathy |
| EPA | Eicosapentaenoic |
| eEPC | Early endothelial progenitor cells |
| Epo | Erythropoietin |
| FDA | Food and Drug Administration |
| G-CSF | Granulocyte colony-stimulating factor |
| HIF | Hypoxia-inducible factor |
| HRE | Hypoxia response element |
| HSPG | Heparin sulphate proteoglycans |
| IGF-1 | Insulin-like growth factor-1 |
| KDR | Kinase insert domain-containing receptor |
| KLT-1 | Kms-related tyrosine kinase 1 |
| mTOR | Mammalian target of rapamycin |
| NOS | Nitric oxide synthetase |
| NRP | Neurophilin |
| ω-3 PUFAs | Omega-3 long-chain polyunsaturated fatty acids |
| OIR | Oxygen-induced retinopathy |
| OPC | Outgrowth endothelial cells |
| PD | Parkinson’s disease |
| PHD | Prolyl hydroxylase |
| RAS | Renin-angiotensin system |
| RGC | Retinal ganglion cells |
| ROP | Retinopathy of prematurity |
| ROS | Reactive oxygen species |
| SC | Stem cell |
| SRPK1 | Serine arginine protein kinase 1 |
| SRSF1 | Serine-rich splicing factor-1 |
| STOP-ROP | Supplemental Therapeutic Oxygen for Prethreshold Retinopathy Of Prematurity |
| VEGF | Vascular endothelial growth factor |
References
- Terry, T.L. Extreme prematurity and fibroblastic overgrowth of persistent vascular sheath behind each crystalline lens: I. Preliminary report. Am. J. Ophthalmol. 1942, 25, 203–204. [Google Scholar] [CrossRef]
- Jain, V.; Langham, M.C.; Wehrli, F.W. MRI estimation of global brain oxygen consumption rate. Br. J. Pharmacol. 2010, 30, 1987. [Google Scholar]
- Anderson, B.; Saltzman, H.A. Retinal Oxygen Utilization Measured by Hyperbaric Blackout. Arch. Ophthalmol. 1964, 72, 792–795. [Google Scholar] [CrossRef] [PubMed]
- Anderson, B., Jr. Ocular effects of changes in oxygen and carbon dioxide tension. Trans. Am. Ophthalmol. Soc. 1968, 66, 423. [Google Scholar] [PubMed]
- Nag, T.; Wadhwa, S. Morphological and Neurochemical Development of the Human Neural Retina. Neuroembryol. Aging 2006, 4, 19–30. [Google Scholar] [CrossRef]
- Weidman, T.A. Fine Structure of the Developing Retina. Int. Ophthalmol. Clin. 1975, 15, 65–84. [Google Scholar] [CrossRef] [PubMed]
- Van Cruchten, S.; Vrolyk, V.; Lepage, M.-F.P.; Baudon, M.; Voute, H.; Schoofs, S.; Haruna, J.; Benoit-Biancamano, M.-O.; Ruot, B.; Allegaert, K.; et al. Pre- and Postnatal Development of the Eye: A Species Comparison. Birth Defects Res. 2017, 109, 1540–1567. [Google Scholar] [CrossRef] [PubMed]
- Scott, A.; Fruttiger, M. Oxygen-induced retinopathy: A model for vascular pathology in the retina. Eye 2010, 24, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lai, C.H.; Lo, A.C. Therapeutic strategies for retinopathy of prematurity. Hong Kong J. Ophthalmol. 2015, 19, 8–15. [Google Scholar]
- Group, S.-R.M.S. Supplemental therapeutic oxygen for prethreshold retinopathy of prematurity (STOP-ROP), a randomized, controlled trial. I: Primary outcomes. Pediatrics 2000, 105, 295–310. [Google Scholar]
- Chen, J.; Smith, L.E. Retinopathy of prematurity. Angiogenesis 2007, 10, 133–140. [Google Scholar] [CrossRef]
- Hellström, A.; LSmith, E.; Dammann, O. Retinopathy of prematurity. Lancet 2013, 382, 1445–1457. [Google Scholar] [CrossRef]
- Sapieha, P.; Joyal, J.-S.; Rivera, J.C.; Kermorvant-Duchemin, E.; Sennlaub, F.; Hardy, P.; Lachapelle, P.; Chemtob, S. Retinopathy of prematurity: Understanding ischemic retinal vasculopathies at an extreme of life. J. Clin. Investig. 2010, 120, 3022–3032. [Google Scholar] [CrossRef]
- Tin, W.; Gupta, S. Optimum oxygen therapy in preterm babies. Arch. Dis. Child. Fetal Neonatal Ed. 2007, 92, F143–F147. [Google Scholar] [CrossRef] [PubMed]
- Connor, K.M.; Krah, N.M.; Dennison, R.J.; Aderman, C.M.; Chen, J.; Guerin, K.I.; Sapieha, P.; Stahl, A.; Willett, K.L.; Smith, L.E.H. Quantification of oxygen-induced retinopathy in the mouse: A model of vessel loss, vessel regrowth and pathological angiogenesis. Nat. Protoc. 2009, 4, 1565–1573. [Google Scholar] [CrossRef] [PubMed]
- Alon, T.; Hemo, I.; Itin, A.; Pe’Er, J.; Stone, J.; Keshet, E. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nat. Med. 1995, 1, 1024–1028. [Google Scholar] [CrossRef] [PubMed]
- Mintz-Hittner, H.A.; Kennedy, K.A.; Chuang, A.Z. Efficacy of intravitreal bevacizumab for stage 3+ retinopathy of prematurity. N. Engl. J. Med. 2011, 364, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Sato, T.; Wada, K.; Arahori, H.; Kuno, N.; Imoto, K.; Iwahashi-Shima, C.; Kusaka, S. Serum Concentrations of Bevacizumab (Avastin) and Vascular Endothelial Growth Factor in Infants with Retinopathy of Prematurity. Am. J. Ophthalmol. 2012, 153, 327–333. [Google Scholar] [CrossRef]
- Wu, W.-C.; Shih, C.-P.; Lien, R.; Wang, N.-K.; Chen, Y.-P.; Chao, A.-N.; Chen, K.-J.; Chen, T.-L.; Hwang, Y.-S.; Lai, C.-C. Serum vascular endothelial growth factor after bevacizumab or ranibizumab treatment for retinopathy of prematurity. Retina 2017, 37, 1–701. [Google Scholar] [CrossRef]
- Chung, E.J.; Kim, J.H.; Ahn, H.S.; Koh, H.J. Combination of laser photocoagulation and intravitreal bevacizumab (Avastin®) for aggressive zone I retinopathy of prematurity. Graefe’s Arch. Clin. Exp. Ophthalmol. 2007, 245, 1727–1730. [Google Scholar] [CrossRef]
- Kim, J.; Kim, S.J.; Chang, Y.S.; Park, W.S. Combined intravitreal bevacizumab injection and zone I sparing laser photocoagulation in patients with zone I retinopathy of prematurity. Retina 2014, 34, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Castellanos, M.A.; Schwartz, S.; Hernández-Rojas, M.L.; Kon-Jara, V.A.; García-Aguirre, G.; Guerrero-Naranjo, J.L.; Chan, R.V.P.; Quiroz-Mercado, H. Long-term effect of antiangiogenic therapy for retinopathy of prematurity up to 5 Years of Follow-up. Retina 2013, 33, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Morin, J.; Luu, T.M.; Superstein, R.; Ospina, L.H.; Lefebvre, F.; Simard, M.-N.; Shah, V.; Shah, P.S.; Kelly, E.N.; The Canadian Neonatal Network and the Canadian Neonatal Follow-Up Network Investigators. Neurodevelopmental Outcomes Following Bevacizumab Injections for Retinopathy of Prematurity. Pediatrics 2016, 137, e20153218. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, M.A.M.; Schwartz, S.; García-Aguirre, G.; Quiroz-Mercado, H. Short-term outcome after intravitreal ranibizumab injections for the treatment of retinopathy of prematurity. Br. J. Ophthalmol. 2013, 97, 816–819. [Google Scholar] [CrossRef] [PubMed]
- Mota, Á.; Carneiro, Â.; Breda, J.; Rosas, V.; Magalhães, A.; Silva, R.; Falcão-Reis, F. Combination of Intravitreal Ranibizumab and Laser Photocoagulation for Aggressive Posterior Retinopathy of Prematurity. Case Rep. Ophthalmol. 2012, 3, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Tokunaga, C.C.; Mitton, K.; Dailey, W.; Massoll, C.; Roumayah, K.; Guzmán, E.; Tarabishy, N.; Cheng, M.; Drenser, K.A. Effects of Anti-VEGF Treatment on the Recovery of the Developing Retina Following Oxygen-Induced Retinopathy. Investig. Opthalmology Vis. Sci. 2014, 55, 1884–1892. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, H.; Culp, D.; Yang, Z.; Fotheringham, L.; Flannery, J.; Hammond, S.; Kafri, T.; Hartnett, M.E. Targeting Müller Cell–Derived VEGF164 to Reduce Intravitreal Neovascularization in the Rat Model of Retinopathy of Prematurity. Investig. Opthalmol. Vis. Sci. 2014, 55, 824–831. [Google Scholar] [CrossRef]
- McLeod, D.S.; Taomoto, M.; Cao, J.; Zhu, Z.; Witte, L.; Lutty, G.A. Localization of VEGF receptor-2 (KDR/Flk-1) and effects of blocking it in oxygen-induced retinopathy. Investig. Ophthalmol. Vis. Sci. 2002, 43, 474–482. [Google Scholar]
- Gammons, M.V.; Dick, A.D.; Harper, S.J.; Bates, D.O. SRPK1 inhibition modulates VEGF splicing to reduce pathological neovascularization in a rat model of retinopathy of prematurity. Investig. Ophthalmol. Vis. Sci. 2013, 54, 5797–5806. [Google Scholar] [CrossRef]
- Yagasaki, R.; Nakahara, T.; Ushikubo, H.; Mori, A.; Sakamoto, K.; Ishii, K. Anti-angiogenic Effects of Mammalian Target of Rapamycin Inhibitors in a Mouse Model of Oxygen-Induced Retinopathy. Boil. Pharm. Bull. 2014, 37, 1838–1842. [Google Scholar] [CrossRef]
- Vanhaesebrouck, S.; Daniëls, H.; Moons, L.; Vanhole, C.; Carmeliet, P.; De Zegher, F. Oxygen-Induced Retinopathy in Mice: Amplification by Neonatal IGF-I Deficit and Attenuation by IGF-I Administration. Pediatr. Res. 2009, 65, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Löfqvist, C.; Chen, J.; Connor, K.M.; Smith, A.C.H.; Aderman, C.M.; Liu, N.; Pintar, J.E.; Ludwig, T.; Hellström, A.; Smith, L.E.H. IGFBP3 suppresses retinopathy through suppression of oxygen-induced vessel loss and promotion of vascular regrowth. Proc. Natl. Acad. Sci. USA 2007, 104, 10589–10594. [Google Scholar] [CrossRef] [PubMed]
- Kielczewski, J.L.; Hu, P.; Shaw, L.C.; Calzi, S.L.; Mames, R.N.; Gardiner, T.A.; McFarland, E.; Chan-Ling, T.; Grant, M.B. Novel Protective Properties of IGFBP-3 Result in Enhanced Pericyte Ensheathment, Reduced Microglial Activation, Increased Microglial Apoptosis, and Neuronal Protection after Ischemic Retinal Injury. Am. J. Pathol. 2011, 178, 1517–1528. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.E.H.; Shen, W.; Perruzzi, C.; Soker, S.; Kinose, F.; Xu, X.; Robinson, G.; Driver, S.; Bischoff, J.; Zhang, B.; et al. Regulation of vascular endothelial growth factor-dependent retinal neovascularization by insulin-like growth factor-1 receptor. Nat. Med. 1999, 5, 1390–1395. [Google Scholar] [CrossRef] [PubMed]
- Sears, J.E.; Hoppe, G.; Ebrahem, Q.; Anand-Apte, B. Prolyl hydroxylase inhibition during hyperoxia prevents oxygen-induced retinopathy. Proc. Natl. Acad. Sci. USA 2008, 105, 19898–19903. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.-J.; Takeda, K.; Fong, G.-H. Prolyl Hydroxylase Domain Protein 2 (PHD2) Mediates Oxygen-Induced Retinopathy in Neonatal Mice. Am. J. Pathol. 2011, 178, 1881–1890. [Google Scholar] [CrossRef] [PubMed]
- Brafman, A.; Mett, I.; Shafir, M.; Gottlieb, H.; Damari, G.; Gozlan-Kelner, S.; Vishnevskia-Dai, V.; Skaliter, R.; Einat, P.; Faerman, A.; et al. Inhibition of Oxygen-Induced Retinopathy in RTP801-Deficient Mice. Investig. Opthalmol. Vis. Sci. 2004, 45, 3796–3805. [Google Scholar] [CrossRef]
- Beauchamp, M.H.; Sennlaub, F.; Speranza, G.; Gobeil, F.; Checchin, D.; Kermorvant-Duchemin, E.; Abran, D.; Hardy, P.; Lachapelle, P.; Varma, D.R.; et al. Redox-dependent effects of nitric oxide on microvascular integrity in oxygen-induced retinopathy. Free. Radic. Boil. Med. 2004, 37, 1885–1894. [Google Scholar] [CrossRef]
- Brooks, S.E.; Gu, X.; Samuel, S.; Marcus, D.M.; Bartoli, M.; Huang, P.L.; Caldwell, R.B. Reduced severity of oxygen-induced retinopathy in eNOS-deficient mice. Investig. Ophthalmol. Vis. Sci. 2001, 42, 222–228. [Google Scholar]
- Zhang, Q.; Zhang, J.; Guan, Y.; Zhang, S.; Zhu, C.; Xu, G.-T.; Wang, L. Suppression of retinal neovascularization by the iNOS inhibitor aminoguanidine in mice of oxygen-induced retinopathy. Graefe’s Arch. Clin. Exp. Ophthalmol. 2009, 247, 919–927. [Google Scholar] [CrossRef]
- Ristori, C.; Filippi, L.; Dal Monte, M.; Martini, D.; Cammalleri, M.; Fortunato, P.; la Marca, G.; Fiorini, P.; Bagnoli, P. Role of the adrenergic system in a mouse model of oxygen-induced retinopathy: Antiangiogenic effects of β-adrenoreceptor blockade. Investig. Ophthalmol. Vis. Sci. 2011, 52, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Monte, M.D.; Casini, G.; La Marca, G.; Isacchi, B.; Filippi, L.; Bagnoli, P. Eye drop propranolol administration promotes the recovery of oxygen-induced retinopathy in mice. Exp. Eye Res. 2013, 111, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Martini, D.; Monte, M.D.; Ristori, C.; Cupisti, E.; Mei, S.; Fiorini, P.; Filippi, L.; Bagnoli, P. Antiangiogenic effects of β2-adrenergic receptor blockade in a mouse model of oxygen-induced retinopathy. J. Neurochem. 2011, 119, 1317–1329. [Google Scholar] [CrossRef] [PubMed]
- Rotschild, T.; Nandgaonkar, B.N.; Yu, K.; Higgins, R.D. Dexamethasone Reduces Oxygen Induced Retinopathy in a Mouse Model. Pediatr. Res. 1999, 46, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Penn, J.S.; Rajaratnam, V.S.; Collier, R.J.; Clark, A.F. The effect of an angiostatic steroid on neovascularization in a rat model of retinopathy of prematurity. Investig. Ophthalmol. Vis. Sci. 2001, 42, 283–290. [Google Scholar]
- Kim, J.H.; Yu, Y.S.; Shin, J.Y.; Lee, H.-Y.; Kim, K.-W. Deguelin inhibits retinal neovascularization by down-regulation of HIF-1α in oxygen-induced retinopathy. J. Cell. Mol. Med. 2008, 12, 2407–2415. [Google Scholar] [CrossRef]
- DeNiro, M.; Al-Halafi, A.; Al-Mohanna, F.H.; AlSmadi, O.; Al-Mohanna, F.A. Pleiotropic effects of YC-1 selectively inhibit pathological retinal neovascularization and promote physiological revascularization in a mouse model of oxygen-induced retinopathy. Mol. Pharmacol. 2010, 77, 348–367. [Google Scholar] [CrossRef]
- Park, S.W.; Kim, J.H.; Kim, K.E.; Jeong, M.H.; Park, H.; Park, B.; Suh, Y.G.; Park, W.J.; Kim, J.H. Beta-lapachone inhibits pathological retinal neovascularization in oxygen-induced retinopathy via regulation of HIF-1α. J. Cell. Mol. Med. 2014, 18, 875–884. [Google Scholar] [CrossRef]
- Pan, H.; Nguyen, N.-Q.-N.; Yoshida, H.; Bentzien, F.; Shaw, L.C.; Rentier-Delrue, F.; Martial, J.A.; Weiner, R.; Struman, I.; Grant, M.B. Molecular targeting of antiangiogenic factor 16K hPRL inhibits oxygen-induced retinopathy in mice. Investig. Opthalmol. Vis. Sci. 2004, 45, 2413–2419. [Google Scholar] [CrossRef]
- Al-Shabrawey, M.; Mussell, R.; Kahook, K.; Tawfik, A.; Eladl, M.; Sarthy, V.; Nussbaum, J.; El-Marakby, A.; Park, S.Y.; Gurel, Z. Increased expression and activity of 12-lipoxygenase in oxygen-induced ischemic retinopathy and proliferative diabetic retinopathy: Implications in retinal neovascularization. Diabetes 2011, 60, 614–624. [Google Scholar] [CrossRef]
- Liang, X.; Zhou, H.; Ding, Y.; Li, J.; Yang, C.; Luo, Y.; Li, S.; Sun, G.; Liao, X.; Min, W. TMP Prevents Retinal Neovascularization and Imparts Neuroprotection in an Oxygen-Induced Retinopathy Model. Investig. Opthalmol. Vis. Sci. 2012, 53, 2157–2169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.X.; Sima, J.; Shao, C.; Fant, J.; Chen, Y.; Rohrer, B.; Gao, G.; Ma, J.-X. Plasminogen kringle 5 reduces vascular leakage in the retina in rat models of oxygen-induced retinopathy and diabetes. Diabetologia 2004, 47, 124–131. [Google Scholar] [CrossRef]
- Xu, Z.; Gong, J.; Maiti, D.; Vong, L.; Wu, L.; Schwarz, J.J.; Duh, E.J. MEF2C Ablation in Endothelial Cells Reduces Retinal Vessel Loss and Suppresses Pathologic Retinal Neovascularization in Oxygen-Induced Retinopathy. Am. J. Pathol. 2012, 180, 2548–2560. [Google Scholar] [CrossRef]
- Ferrara, N.; Davis-Smyth, T. The Biology of Vascular Endothelial Growth Factor. Endocr. Rev. 1997, 18, 4–25. [Google Scholar] [CrossRef]
- Ferrara, N.; Gerber, H.P. Vascular endothelial growth factor molecular and biological aspects. In Advances in Organ Biology; Elsevier: Amsterdam, The Netherlands, 1999; pp. 25–57. [Google Scholar]
- Cavallaro, G.; Filippi, L.; Bagnoli, P.; La Marca, G.; Cristofori, G.; Raffaeli, G.; Padrini, L.; Araimo, G.; Fumagalli, M.; Groppo, M.; et al. The pathophysiology of retinopathy of prematurity: An update of previous and recent knowledge. Acta Ophthalmol. 2014, 92, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Abbracchio, M.; Brambilla, R.; Ceruti, S.; Kim, H.; Von Lubitz, D.; Jacobson, K.; Cattabeni, F. G-protein-dependent activation of phospholipase C by adenosine A3 receptors in rat brain. Pharmacol. Res. 1995, 31, 168. [Google Scholar] [CrossRef]
- Bellik, L.; Vinci, M.C.; Filippi, S.; Ledda, F.; Parenti, A. Intracellular pathways triggered by the selective FLT-1-agonist placental growth factor in vascular smooth muscle cells exposed to hypoxia. Br. J. Pharmacol. 2005, 146, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Terman, B.I.; Dougher-Vermazen, M.; Carrion, M.E.; Dimitrov, D.; Armellino, D.C.; Gospodarowicz, D.; Böhlen, P. Identification of the KDR tyrosine kinase as a receptor for vascular endothelial cell growth factor. Biochem. Biophys. Res. Commun. 1992, 187, 1579–1586. [Google Scholar] [CrossRef]
- Quinn, T.P.; Peters, K.G.; De Vries, C.; Ferrara, N.; Williams, L.T. Fetal liver kinase 1 is a receptor for vascular endothelial growth factor and is selectively expressed in vascular endothelium. Proc. Natl. Acad. Sci. USA 1993, 90, 7533–7537. [Google Scholar] [CrossRef] [PubMed]
- Waltenberger, J.; Claesson-Welsh, L.; Siegbahn, A.; Shibuya, M.; Heldin, C.H. Different signal transduction properties of KDR and Flt1, two receptors for vascular endothelial growth factor. J. Boil. Chem. 1994, 269, 26988–26995. [Google Scholar]
- Bernatchez, P.N.; Soker, S.; Sirois, M.G. Vascular Endothelial Growth Factor Effect on Endothelial Cell Proliferation, Migration, and Platelet-activating Factor Synthesis Is Flk-1-dependent. J. Boil. Chem. 1999, 274, 31047–31054. [Google Scholar] [CrossRef] [PubMed]
- Kaur, C.; Foulds, W.; Ling, E. Blood–retinal barrier in hypoxic ischaemic conditions: Basic concepts, clinical features and management. Prog. Retin. Eye Res. 2008, 27, 622–647. [Google Scholar] [CrossRef] [PubMed]
- Provis, J. Development of the Primate Retinal Vasculature. Prog. Retin. Eye Res. 2001, 20, 799–821. [Google Scholar] [CrossRef]
- Gariano, R.F.; Gardner, T.W. Retinal angiogenesis in development and disease. Nature 2005, 438, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Stone, J.; Maslim, J. Mechanisms of retinal angiogenesis. Prog. Retin. Eye Res. 1997, 16, 157–181. [Google Scholar] [CrossRef]
- Hellström, A. IGF-I Is Critical for Normal Vascularization of the Human Retina. J. Clin. Endocrinol. Metab. 2002, 87, 3413–3416. [Google Scholar] [CrossRef] [PubMed]
- Hellström, A.; Perruzzi, C.; Ju, M.; Engström, E.; Hård, A.-L.; Liu, J.-L.; Albertsson-Wikland, K.; Carlsson, B.; Niklasson, A.; Sjödell, L.; et al. Low IGF-I suppresses VEGF-survival signaling in retinal endothelial cells: Direct correlation with clinical retinopathy of prematurity. Proc. Natl. Acad. Sci. USA 2001, 98, 5804–5808. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, R.; Okada, T.; Moriya, M.; Naito, M.; Tsuruo, T.; Miyatake, K.; Nakano, Y. Brain Capillary Endothelial Cells Express two forms of Erythropoietin Receptor mRNA. JBIC J. Boil. Inorg. Chem. 1996, 239, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Watts, K.D.; McColley, S.A. Elevated vascular endothelial growth factor is correlated with elevated erythropoietin in stable, young cystic fibrosis patients. Pediatr. Pulmonol. 2011, 46, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Connor, K.M.; Aderman, C.M.; Smith, L.E. Erythropoietin deficiency decreases vascular stability in mice. J. Clin. Investig. 2008, 118, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Hartnett, M.E. Pathophysiology and mechanisms of severe retinopathy of prematurity. Ophthalmology 2015, 122, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Rusai, K.; Vannay, A.; Szebeni, B.; Borgulya, G.; Fekete, A.; Vásárhelyi, B.; Tulassay, T.; Szabó, A.J. Endothelial nitric oxide synthase gene T−786C and 27-bp repeat gene polymorphisms in retinopathy of prematurity. Mol. Vis. 2008, 14, 286–290. [Google Scholar] [PubMed]
- Yanamandra, K.; Napper, D.; Pramanik, A.; Bocchini, J.A.; Dhanireddy, R. Endothelial Nitric Oxide Synthase genotypes in the etiology of retinopathy of prematurity in premature infants. Ophthalmic Genet. 2010, 31, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Hashiguchi, A.; Yano, S.; Morioka, M.; Hamada, J.; Ushio, Y.; Takeuchi, Y.; Fukunaga, K. Up-Regulation of Endothelial Nitric Oxide Synthase via Phosphatidylinositol 3-Kinase Pathway Contributes to Ischemic Tolerance in the CA1 Subfield of Gerbil Hippocampus. Br. J. Pharmacol. 2004, 24, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Haskó, G.; Cronstein, B.N. Regulation of Inflammation by Adenosine. Front. Immunol. 2013, 4, 85. [Google Scholar] [CrossRef] [PubMed]
- Madeira, M.H.; Boia, R.; Elvas, F.; Martins, T.; Cunha, R.A.; Ambrósio, A.F.; Santiago, A.R.; Information, P.E.K.F.C. Selective A2A receptor antagonist prevents microglia-mediated neuroinflammation and protects retinal ganglion cells from high intraocular pressure–induced transient ischemic injury. Transl. Res. 2016, 169, 112–128. [Google Scholar] [CrossRef]
- Cerri, S.; Levandis, G.; Ambrosi, G.; Montepeloso, E.; Antoninetti, G.F.; Franco, R.; Lanciego, J.L.; Baqi, Y.; Müller, C.E.; Pinna, A. Neuroprotective potential of adenosine A2A and cannabinoid CB1 receptor antagonists in an animal model of Parkinson disease. J. Neuropathol. Exp. Neurol. 2014, 73, 414–424. [Google Scholar] [CrossRef]
- Gyoneva, S.; Shapiro, L.; Lazo, C.; Garnier-Amblard, E.; Smith, Y.; Miller, G.W.; Traynelis, S.F. Adenosine A2A receptor antagonism reverses inflammation-induced impairment of microglial process extension in a model of Parkinson’s disease. Neurobiol. Dis. 2014, 67, 191–202. [Google Scholar] [CrossRef]
- Canas, P.M.; Porciúncula, L.O.; Cunha, G.M.A.; Silva, C.G.; Machado, N.J.; Oliveira, J.M.A.; Oliveira, C.R.; Cunha, R.A. Adenosine A2A Receptor Blockade Prevents Synaptotoxicity and Memory Dysfunction Caused by β-Amyloid Peptides via p38 Mitogen-Activated Protein Kinase Pathway. J. Neurosci. 2009, 29, 14741–14751. [Google Scholar] [CrossRef]
- Chen, J.F.; Huang, Z.; Ma, J.; Zhu, J.; Moratalla, R.; Standaert, D.; Moskowitz, M.A.; Fink, J.S.; Schwarzschild, M.A. A2A adenosine receptor deficiency attenuates brain injury induced by transient focal ischemia in mice. J. Neurosci. 1999, 19, 9192–9200. [Google Scholar] [CrossRef]
- Santiago, A.R.; Baptista, F.I.; Santos, P.F.; Cristóvão, G.; Ambrósio, A.F.; Cunha, R.A.; Gomes, C.A. Role of microglia adenosine A2A receptors in retinal and brain neurodegenerative diseases. Mediat. Inflamm. 2014, 2014, 465694. [Google Scholar] [CrossRef]
- Fischer, S.; Sharma, H.; Karliczek, G.; Schaper, W. Expression of vascular permeability factor/vascular endothelial growth factor in pig cerebral microvascular endothelial cells and its upregulation by adenosine. Mol. Brain Res. 1995, 28, 141–148. [Google Scholar] [CrossRef]
- Takagi, H.; King, G.L.; Ferrara, N.; Aiello, L.P. Hypoxia regulates vascular endothelial growth factor receptor KDR/Flk gene expression through adenosine A2 receptors in retinal capillary endothelial cells. Investig. Ophthalmol. Vis. Sci. 1996, 37, 1311–1321. [Google Scholar]
- Grant, M.B.; Davis, M.I.; Caballero, S.; Feoktistov, I.; Biaggioni, I.; Belardinelli, L. Proliferation, migration, and ERK activation in human retinal endothelial cells through A2B adenosine receptor stimulation. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2068–2073. [Google Scholar]
- De Hoz, R.; Gallego, B.I.; Ramírez, A.I.; Rojas, B.; Salazar, J.J.; Valiente-Soriano, F.J.; Avilés-Trigueros, M.; Villegas-Perez, M.P.; Vidal-Sanz, M.; Triviño, A. Rod-like microglia are restricted to eyes with laser-induced ocular hypertension but absent from the microglial changes in the contralateral untreated eye. PLoS ONE 2013, 8, e83733. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.; Calder, C.J.; Albon, J.; Erichsen, J.T.; Boulton, M.E.; Morgan, J.E. Involvement of the CD200 receptor complex in microglia activation in experimental glaucoma. Exp. Eye Res. 2011, 92, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.P.; Sharma, S.; Steinle, J.J. Age-related changes in sympathetic neurotransmission in rat retina and choroid. Exp. Eye Res. 2007, 84, 75–81. [Google Scholar] [CrossRef]
- Steinle, J.J.; Smith, P.G. Role of adrenergic receptors in vascular remodelling of the rat choroid. Br. J. Pharmacol. 2002, 136, 730–734. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, S.; Moura, D. Vascular adrenoceptors: An update. Pharmacol. Rev. 2001, 53, 319–356. [Google Scholar]
- Smith, L.; Wesolowski, E.; McLellan, A.; Kostyk, S.K.; D’Amato, R.; Sullivan, R.; D’Amore, P.A. Oxygen-induced retinopathy in the mouse. Investig. Ophthalmol. Vis. Sci. 1994, 35, 101–111. [Google Scholar]
- Grossniklaus, H.E.; Kang, S.J.; Berglin, L. Animal Models of Choroidal and Retinal Neovascularization. Prog. Retin. Eye Res. 2010, 29, 500–519. [Google Scholar] [CrossRef] [PubMed]
- Penn, J.S.; Tolman, B.L.; Lowery, L.A. Variable oxygen exposure causes preretinal neovascularization in the newborn rat. Investig. Ophthalmol. Vis. Sci. 1993, 34, 576–585. [Google Scholar]
- Rapisarda, A.; Uranchimeg, B.; Sordet, O.; Pommier, Y.; Shoemaker, R.H.; Melillo, G. Topoisomerase I-mediated inhibition of hypoxia-inducible factor 1: Mechanism and therapeutic implications. Cancer Res. 2004, 64, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Tang, B.; Sun, X. Development of Inhibitors Targeting Hypoxia-Inducible Factor 1 and 2 for Cancer Therapy. Yonsei Med. J. 2017, 58, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Miwa, Y.; Hoshino, Y.; Shoda, C.; Jiang, X.; Tsubota, K.; Kurihara, T. Pharmacological HIF inhibition prevents retinal neovascularization with improved visual function in a murine oxygen-induced retinopathy model. Neurochem. Int. 2019, 128, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Joyal, J.-S.; Hatton, C.J.; Juan, A.M.; Pei, D.T.; Hurst, C.G.; Xu, D.; Stahl, A.; Hellström, A.; Smith, L.E.H. Propranolol Inhibition of β-Adrenergic Receptor Does Not Suppress Pathologic Neovascularization in Oxygen-Induced Retinopathy. Investig. Opthalmol. Vis. Sci. 2012, 53, 2968–2977. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.-H.; Koh, Y.J.; Jeong, H.-S.; Lee, D.-H.; Lee, E.H.; Cho, C.-H. Propranolol increases vascular permeability through pericyte apoptosis and exacerbates oxygen-induced retinopathy. Biochem. Biophys. Res. Commun. 2018, 503, 2792–2799. [Google Scholar] [CrossRef] [PubMed]
- Parupia, M.H.; Dhanireddy, R. Association of Postnatal Dexamethasone Use and Fungal Sepsis in the Development of Severe Retinopathy of Prematurity and Progression to Laser Therapy in Extremely Low-Birth-Weight Infants. J. Perinatol. 2001, 21, 242–247. [Google Scholar] [CrossRef]
- Pisani, F.; Cammalleri, M.; Dal Monte, M.; Locri, F.; Mola, M.G.; Nicchia, G.P.; Frigeri, A.; Bagnoli, P.; Svelto, M. Potential role of the methylation of VEGF gene promoter in response to hypoxia in oxygen-induced retinopathy: Beneficial effect of the absence of AQP4. J. Cell. Mol. Med. 2018, 22, 613–627. [Google Scholar] [CrossRef]
- Kolibabka, M.; Dietrich, N.; Klein, T.; Hammes, H.-P. Anti-angiogenic effects of the DPP-4 inhibitor linagliptin via inhibition of VEGFR signalling in the mouse model of oxygen-induced retinopathy. Diabetologia 2018, 61, 2412–2421. [Google Scholar] [CrossRef]
- Vähätupa, M.; Cordova, Z.M.; Barker, H.; Aittomäki, S.; Uusitalo, H.; Järvinen, T.A.; Pesu, M.; Uusitalo-Järvinen, H. Furin deficiency in myeloid cells leads to attenuated revascularization in a mouse-model of oxygen-induced retinopathy. Exp. Eye Res. 2018, 166, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.-S.; Lee, Y.-N.; Wang, S.-W.; Wu, Y.-J.; Su, C.-H.; Hsieh, C.-L.; Tien, T.Y.; Wang, B.-J.; Chen, M.-C.; Chen, C.-W.; et al. KC21 Peptide Inhibits Angiogenesis and Attenuates Hypoxia-Induced Retinopathy. J. Cardiovasc. Transl. Res. 2019, 12, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.; Leblanc, M.E.; Wang, W.; Liang, D.; Chen, P.; Chou, T.-H.; Tian, H.; Li, W. Anti-secretogranin III therapy of oxygen-induced retinopathy with optimal safety. Angiogenesis 2019, 22, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Geng, W.; Qin, F.; Ren, J.; Xiao, S.; Wang, A. Mini-peptide RPL41 attenuated retinal neovascularization by inducing degradation of ATF4 in oxygen-induced retinopathy mice. Exp. Cell Res. 2018, 369, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Iizuka, N.; Morita, A.; Kawano, C.; Mori, A.; Sakamoto, K.; Kuroyama, M.; Ishii, K.; Nakahara, T. Anti-angiogenic effects of valproic acid in a mouse model of oxygen-induced retinopathy. J. Pharmacol. Sci. 2018, 138, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Lalkovičová, M.; Danielisová, V. Neuroprotection and antioxidants. Neural Regen. Res. 2016, 11, 865. [Google Scholar] [CrossRef] [PubMed]
- Sohn, E.H.; Van Dijk, H.W.; Jiao, C.; Kok, P.H.B.; Jeong, W.; Demirkaya, N.; Garmager, A.; Wit, F.; Kucukevcilioglu, M.; Van Velthoven, M.E.J.; et al. Retinal neurodegeneration may precede microvascular changes characteristic of diabetic retinopathy in diabetes mellitus. Proc. Natl. Acad. Sci. USA 2016, 113, E2655–E2664. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Hu, Y.; Zhou, T.; Zhou, K.K.; Mott, R.; Wu, M.; Boulton, M.; Lyons, T.J.; Gao, G.; Ma, J.-X. Activation of the Wnt Pathway Plays a Pathogenic Role in Diabetic Retinopathy in Humans and Animal Models. Am. J. Pathol. 2009, 175, 2676–2685. [Google Scholar] [CrossRef]
- Simó, R.; Carrasco, E.; García-Ramírez, M.; Hernández, C. Angiogenic and antiangiogenic factors in proliferative diabetic retinopathy. Curr. Diabetes Rev. 2006, 2, 71–98. [Google Scholar] [CrossRef]
- Vessey, K.; Wilkinson-Berka, J.; Fletcher, E. Characterization of retinal function and glial cell response in a mouse model of oxygen-induced retinopathy. J. Comp. Neurol. 2011, 519, 506–527. [Google Scholar] [CrossRef]
- Obrosova, I.G. Increased Sorbitol Pathway Activity Generates Oxidative Stress in Tissue Sites for Diabetic Complications. Antioxid. Redox Signal. 2005, 7, 1543–1552. [Google Scholar] [CrossRef] [PubMed]
- Meneses, P.; Hajjar, K.; Berns, K.; Duvoisin, R. Recombinant angiostatin prevents retinal neovascularization in a murine proliferative retinopathy model. Gene Ther. 2001, 8, 646. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kermorvant-Duchemin, E.; Sapieha, P.; Sirinyan, M.; Beauchamp, M.; Checchin, D.; Hardy, P.; Sennlaub, F.; Lachapelle, P.; Chemtob, S. Understanding ischemic retinopathies: Emerging concepts from oxygen-induced retinopathy. Doc. Ophthalmol. 2010, 120, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Tsao, R.; Zhang, S.; Dong, Z.; Yang, R.; Gong, J.; Pei, Y. Antioxidant activity, mutagenicity/anti-mutagenicity, and clastogenicity/anti-clastogenicity of lutein from marigold flowers. Food Chem. Toxicol. 2006, 44, 1522–1529. [Google Scholar] [CrossRef] [PubMed]
- Maoka, T.; Tokuda, H.; Suzuki, N.; Kato, H.; Etoh, H. Anti-Oxidative, Anti-Tumor-Promoting, and Anti-Carcinogensis Activities of Nitroastaxanthin and Nitrolutein, the Reaction Products of Astaxanthin and Lutein with Peroxynitrite. Mar. Drugs 2012, 10, 1391–1399. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-Y.; Fu, Z.-J.; Ma, H.; Jang, W.-C.; So, K.-F.; Wong, D.; Lo, A.C.Y. Effect of Lutein on Retinal Neurons and Oxidative Stress in a Model of Acute Retinal Ischemia/Reperfusion. Investig. Opthalmol. Vis. Sci. 2009, 50, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-Y.; Yang, D.; Fu, Z.J.; Woo, T.; Wong, D.; Lo, A.C.Y. Lutein enhances survival and reduces neuronal damage in a mouse model of ischemic stroke. Neurobiol. Dis. 2012, 45, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Ozawa, Y.; Kurihara, T.; Noda, K.; Imamura, Y.; Kobayashi, S.; Ishida, S.; Tsubota, K. Neuroprotective Effect of an Antioxidant, Lutein, during Retinal Inflammation. Investig. Opthalmol. Vis. Sci. 2009, 50, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Alves-Rodrigues, A.; Shao, A. The science behind lutein. Toxicol. Lett. 2004, 150, 57–83. [Google Scholar] [CrossRef] [PubMed]
- Snodderly, D.M. Evidence for protection against age-related macular degeneration by carotenoids and antioxidant vitamins. Am. J. Clin. Nutr. 1995, 62, 1448S–1461S. [Google Scholar] [CrossRef]
- Bone, R.A.; Landrum, J.T.; Friedes, L.M.; Gomez, C.M.; Kilburn, M.D.; Menendez, E.; Vidal, I.; Wang, W. Distribution of Lutein and Zeaxanthin Stereoisomers in the Human Retina. Exp. Eye Res. 1997, 64, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Sommerburg, O.; Keunen, J.E.E.; Bird, A.C.; Van Kuijk, F.J.G.M. Fruits and vegetables that are sources for lutein and zeaxanthin: The macular pigment in human eyes. Br. J. Ophthalmol. 1998, 82, 907–910. [Google Scholar] [CrossRef] [PubMed]
- Fung, F.K.C.; Wong, D.; Chan, H.H.L.; Lo, A.C.Y.; Li, S.-Y.; Fu, Z.J. Anti-Inflammatory Effects of Lutein in Retinal Ischemic/Hypoxic Injury: In Vivo and In Vitro Studies. Investig. Opthalmol. Vis. Sci. 2012, 53, 5976–5984. [Google Scholar]
- Li, S.-Y.; Lo, A.C.Y. Lutein Protects RGC-5 Cells Against Hypoxia and Oxidative Stress. Int. J. Mol. Sci. 2010, 11, 2109–2117. [Google Scholar] [CrossRef] [PubMed]
- Woo, T.T.; Li, S.-Y.; Lai, W.W.; Wong, D.; Lo, A.C. Neuroprotective effects of lutein in a rat model of retinal detachment. Graefe’s Archive for Clin. Exp. Ophthalmol. 2013, 251, 41–51. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, Z.; Zhao, J.; Li, Q.; Huang, C.; Zhu, L.; Lu, D. Neuroprotective effect of lutein on NMDA-induced retinal ganglion cell injury in rat retina. Cell. Mol. Neurobiol. 2016, 36, 531–540. [Google Scholar] [CrossRef]
- Fu, Z.; Meng, S.S.; Burnim, S.B.; Smith, L.E.; Lo, A.C. Lutein facilitates physiological revascularization in a mouse model of retinopathy of prematurity. Clin. Exp. Ophthalmol. 2017, 45, 529–538. [Google Scholar] [CrossRef]
- Shi, X.; Dalal, N.; Jain, A. Antioxidant behaviour of caffeine: Efficient scavenging of hydroxyl radicals. Food Chem. Toxicol. 1991, 29, 1–6. [Google Scholar] [CrossRef]
- Barcelos, R.P.; Souza, M.A.; Amaral, G.P.; Stefanello, S.T.; Bresciani, G.; Fighera, M.R.; Soares, F.A.A.; Barbosa, N.V. Caffeine supplementation modulates oxidative stress markers in the liver of trained rats. Life Sci. 2014, 96, 40–45. [Google Scholar] [CrossRef]
- Devasagayam, T.; Kamat, J.; Mohan, H.; Kesavan, P. Caffeine as an antioxidant: Inhibition of lipid peroxidation induced by reactive oxygen species. Biochim. Biophys. Acta (BBA) Biomembr. 1996, 1282, 63–70. [Google Scholar] [CrossRef]
- Chavez-Valdez, R.; Wills-Karp, M.; Ahlawat, R.; Cristofalo, E.A.; Nathan, A.; Gauda, E.B. Caffeine Modulates TNF-α Production by Cord Blood Monocytes: The Role of Adenosine Receptors. Pediatr. Res. 2009, 65, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, G.; Hu, J.-L.; Fu, X.-H.; Zeng, Y.-J.; Zhou, Y.-G.; Xiong, G.; Yang, N.; Dai, S.-S.; He, F.-T. Chronic or high dose acute caffeine treatment protects mice against oleic acid-induced acute lung injury via an adenosine A2A receptor-independent mechanism. Eur. J. Pharmacol. 2011, 654, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Dall’Lgna, O.P.; Porciúncula, L.O.; Souza, D.O.; Cunha, R.A.; Lara, D.R. Neuroprotection by caffeine and adenosine A2A receptor blockade of β-amyloid neurotoxicity. Br. J. Pharmacol. 2003, 138, 1207–1209. [Google Scholar] [CrossRef] [PubMed]
- Endesfelder, S.; Zaak, I.; Weichelt, U.; Bührer, C.; Schmitz, T. Caffeine protects neuronal cells against injury caused by hyperoxia in the immature brain. Free. Radic. Boil. Med. 2014, 67, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhou, R.; Li, B.; Li, H.; Wang, Y.; Gu, X.; Tang, L.; Wang, C.; Zhong, D.; Ge, Y.; et al. Caffeine preferentially protects against oxygen-induced retinopathy. FASEB J. 2017, 31, 3334–3348. [Google Scholar] [CrossRef] [PubMed]
- Barberger-Gateau, P.; Letenneur, L.; Deschamps, V.; Pérès, K.; Dartigues, J.-F.; Renaud, S. Fish, meat, and risk of dementia: Cohort study. BMJ 2002, 325, 932–933. [Google Scholar] [CrossRef]
- Morris, M.C.; Evans, D.A.; Bienias, J.L.; Tangney, C.C.; Bennett, D.A.; Wilson, R.S.; Aggarwal, N.; Schneider, J. Consumption of Fish and n-3 Fatty Acids and Risk of Incident Alzheimer Disease. Arch. Neurol. 2003, 60, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Calon, F.; Cole, G. Neuroprotective action of omega-3 polyunsaturated fatty acids against neurodegenerative diseases: Evidence from animal studies. Prostaglandins Leukot. Essent. Fat. Acids 2007, 77, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Calon, F.; Lim, G.P.; Yang, F.; Morihara, T.; Teter, B.; Ubeda, O.; Rostaing, P.; Triller, A.; Salem, N.; Ashe, K.H.; et al. Docosahexaenoic Acid Protects from Dendritic Pathology in an Alzheimer’s Disease Mouse Model. Neuron 2004, 43, 633–645. [Google Scholar] [CrossRef]
- Shimazawa, M.; Nakajima, Y.; Mashima, Y.; Hara, H. Docosahexaenoic acid (DHA) has neuroprotective effects against oxidative stress in retinal ganglion cells. Brain Res. 2009, 1251, 269–275. [Google Scholar] [CrossRef]
- Sapieha, P.; Stahl, A.; Chen, J.; Seaward, M.R.; Willett, K.L.; Krah, N.M.; Dennison, R.J.; Connor, K.M.; Aderman, C.M.; Liclican, E.; et al. 5-Lipoxygenase Metabolite 4-HDHA Is a Mediator of the Antiangiogenic Effect of ω-3 Polyunsaturated Fatty Acids. Sci. Transl. Med. 2011, 3, 69ra12. [Google Scholar] [CrossRef] [PubMed]
- Stahl, A.; Sapieha, P.; Connor, K.M.; SanGiovanni, J.P.; Chen, J.; Aderman, C.M.; Willett, K.L.; Krah, N.M.; Dennison, R.J.; Seaward, M.R.; et al. PPARγ mediates a direct anti-angiogenic effect of ω3-PUFAs in proliferative retinopathy. Circ. Res. 2010, 107, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Beharry, K.D.; Cai, C.L.; Siddiqui, F.; Chowdhury, S.; D’Agrosa, C.; Valencia, G.B.; Aranda, J.V. Comparative Effects of Coenzyme Q10 or n-3 Polyunsaturated Fatty Acid Supplementation on Retinal Angiogenesis in a Rat Model of Oxygen-Induced Retinopathy. Antioxidants 2018, 7, 160. [Google Scholar] [CrossRef] [PubMed]
- De La Lastra, C.A.; Villegas, I. Resveratrol as an antioxidant and pro-oxidant agent: Mechanisms and clinical implications. Biochem. Soc. Trans. 2007, 35, 1156–1160. [Google Scholar] [CrossRef] [PubMed]
- Zini, R.; Morin, C.; Bertelli, A.A.; Tillement, J.P. Effects of resveratrol on the rat brain respiratory chain. Drugs Under Exp. Clin. Res. 1999, 25, 87–97. [Google Scholar]
- Sinha, K.; Chaudhary, G.; Gupta, Y.K. Protective effect of resveratrol against oxidative stress in middle cerebral artery occlusion model of stroke in rats. Life Sci. 2002, 71, 655–665. [Google Scholar] [CrossRef]
- Martín, A.R.; Villegas, I.; La Casa, C.; de la Lastra, C.A. Resveratrol, a polyphenol found in grapes, suppresses oxidative damage and stimulates apoptosis during early colonic inflammation in rats. Biochem. Pharmacol. 2004, 67, 1399–1410. [Google Scholar] [PubMed]
- Martín, A.R.; Villegas, I.; Sánchez-Hidalgo, M.; De La Lastra, C.A. The effects of resveratrol, a phytoalexin derived from red wines, on chronic inflammation induced in an experimentally induced colitis model. Br. J. Pharmacol. 2006, 147, 873–885. [Google Scholar] [CrossRef]
- Jin, F.; Wu, Q.; Lu, Y.-F.; Gong, Q.-H.; Shi, J.-S. Neuroprotective effect of resveratrol on 6-OHDA-induced Parkinson’s disease in rats. Eur. J. Pharmacol. 2008, 600, 78–82. [Google Scholar] [CrossRef]
- Ates, O.; Cayli, S.; Altinoz, E.; Gurses, I.; Yucel, N.; Sener, M.; Kocak, A.; Yologlu, S. Neuroprotection by resveratrol against traumatic brain injury in rats. Mol. Cell. Biochem. 2007, 294, 137–144. [Google Scholar] [CrossRef]
- Lopez, M.S.; Dempsey, R.J.; Vemuganti, R. Resveratrol neuroprotection in stroke and traumatic CNS injury. Neurochem. Int. 2015, 89, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Gong, Q.; Dong, H.; Shi, J. Resveratrol, a neuroprotective supplement for Alzheimer’s disease. Curr. Pharm. Des. 2012, 18, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Bastianetto, S.; Ménard, C.; Quirion, R. Neuroprotective action of resveratrol. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2015, 1852, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Lançon, A.; Frazzi, R.; Latruffe, N. Anti-Oxidant, Anti-Inflammatory and Anti-Angiogenic Properties of Resveratrol in Ocular Diseases. Molecules 2016, 21, 304. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.T.; Suh, E.S. Retinal Protective Effects of Resveratrol via Modulation of Nitric Oxide Synthase on Oxygen-induced Retinopathy. Korean J. Ophthalmol. 2010, 24, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Jiang, D. Effect of resveratrol on Bcl-2 and VEGF expression in oxygen-induced retinopathy of prematurity. J. Pediatr. Ophthalmol. Strabismus 2012, 49, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Akama, K.T.; Albanese, C.; Pestell, R.G.; Van Eldik, L.J. Amyloid β-peptide stimulates nitric oxide production in astrocytes through an NFκB-dependent mechanism. Proc. Natl. Acad. Sci. USA 1998, 95, 5795–5800. [Google Scholar] [CrossRef]
- Sano, M.; Ernesto, C.; Thomas, R.G.; Klauber, M.R.; Schafer, K.; Grundman, M.; Woodbury, P.; Growdon, J.; Cotman, C.W.; Pfeiffer, E. A controlled trial of selegiline, alpha-tocopherol, or both as treatment for Alzheimer’s disease. N. Engl. J. Med. 1997, 336, 1216–1222. [Google Scholar] [CrossRef]
- Penn, J.S.; Tolman, B.L.; Bullard, L.E. Effect of a Water-Soluble Vitamin E Analog, Trolox C, on Retinal Vascular Development in an Animal Model of Retinopathy of Prematurity. Free. Radic. Boil. Med. 1997, 22, 977–984. [Google Scholar] [CrossRef]
- Penn, J.S.; Thum, L.A.; Naash, M.I. Oxygen-induced retinopathy in the rat. Vitamins C and E as potential therapies. Investig. Ophthalmol. Vis. Sci. 1992, 33, 1836–1845. [Google Scholar]
- Johnson, L.; Quinn, G.E.; Abbasi, S.; Otis, C.; Goldstein, D.; Sacks, L.; Porat, R.; Fong, E.; Delivoria-Papadopoulos, M.; Peckham, G.; et al. Effect of sustained pharmacologic vitamin E levels on incidence and severity of retinopathy of prematurity: A controlled clinical trial. J. Pediatr. 1989, 114, 827–838. [Google Scholar] [CrossRef]
- Raju, T.N.; Langenberg, P.; Bhutani, V.; Quinn, G.E. Vitamin E prophylaxis to reduce retinopathy of prematurity: A reappraisal of published trials. J. Pediatr. 1997, 131, 844–850. [Google Scholar] [CrossRef]
- Finer, N.N.; Schindler, R.F.; Grant, G.; Hill, G.B.; Peters, K. Effect of intramuscular vitamin E on frequency and severity of retrolental fibroplasia. A controlled trial. Lancet 1982, 1, 1087–1091. [Google Scholar] [CrossRef]
- Phelps, D.L.; Rosenbaum, A.L.; Isenberg, S.J.; Leake, R.D.; Dorey, F.J. Tocopherol efficacy and safety for preventing retinopathy of prematurity: A randomized, controlled, double-masked trial. Pediatrics 1987, 79, 489–500. [Google Scholar] [PubMed]
- Obrosova, I.G.; Pacher, P.; Szabó, C.; Zsengeller, Z.; Hirooka, H.; Stevens, M.J.; Yorek, M.A. Aldose Reductase Inhibition Counteracts Oxidative-Nitrosative Stress and Poly(ADP-Ribose) Polymerase Activation in Tissue Sites for Diabetes Complications. Diabetes 2005, 54, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.S.; Ho, E.C.; Lam, K.S. Contribution of Polyol Pathway to Diabetes-Induced Oxidative Stress. J. Am. Soc. Nephrol. 2003, 14, 233–236. [Google Scholar] [CrossRef]
- Lee, A.Y.W.; Chung, S.S.M. Contributions of polyol pathway to oxidative stress in diabetic cataract. FASEB J. 1999, 13, 23–30. [Google Scholar] [CrossRef]
- Lou, M.F.; Dickerson, J.E.; Garadi, R.; York, B.M. Glutathione depletion in the lens of galactosemic and diabetic rats. Exp. Eye Res. 1988, 46, 517–530. [Google Scholar] [CrossRef]
- Song, Z.; Fu, D.T.; Chan, Y.-S.; Leung, S.; Chung, S.S.; Chung, S.K. Transgenic mice overexpressing aldose reductase in Schwann cells show more severe nerve conduction velocity deficit and oxidative stress under hyperglycemic stress. Mol. Cell. Neurosci. 2003, 23, 638–647. [Google Scholar] [CrossRef]
- Cheung, A.K.; Lo, A.C.; So, K.F.; Chung, S.S.; Chung, S.K.; Lo, A.C.Y. Gene deletion and pharmacological inhibition of aldose reductase protect against retinal ischemic injury. Exp. Eye Res. 2007, 85, 608–616. [Google Scholar] [CrossRef]
- Obrosova, I.G.; Minchenko, A.G.; Vasupuram, R.; White, L.; Abatan, O.I.; Kumagai, A.K.; Frank, R.N.; Stevens, M.J.; Minchenko, O. Aldose Reductase Inhibitor Fidarestat Prevents Retinal Oxidative Stress and Vascular Endothelial Growth Factor Overexpression in Streptozotocin-Diabetic Rats. Diabetes 2003, 52, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.K.; Fung, M.K.; Lo, A.C.; Lam, T.T.; So, K.F.; Chung, S.S.; Chung, S.K. Aldose reductase deficiency prevents diabetes-induced blood-retinal barrier breakdown, apoptosis, and glial reactivation in the retina of db/db mice. Diabetes 2005, 54, 3119–3125. [Google Scholar] [CrossRef] [PubMed]
- Thomas, T.; Rauscher, F.; Sanders, R.; Veltman, J.; Watkins, J.B. Effects of Aldose Reductase Inhibitors on Antioxidant Defense in Rat and Rabbit Liver. Toxicol. Sci. 2000, 53, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.J.; Li, S.-Y.; Kociok, N.; Wong, D.; Chung, S.K.; Lo, A.C.Y. Aldose Reductase Deficiency Reduced Vascular Changes in Neonatal Mouse Retina in Oxygen-Induced Retinopathy. Investig. Opthalmol. Vis. Sci. 2012, 53, 5698–5712. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Nian, S.; Li, S.-Y.; Wong, D.; Chung, S.K.; Lo, A.C.Y. Deficiency of aldose reductase attenuates inner retinal neuronal changes in a mouse model of retinopathy of prematurity. Graefe’s Arch. Clin. Exp. Ophthalmol. 2015, 253, 1503–1513. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Rajapakse, N.; Horiguchi, T.; Payne, R.; Busija, D.W. Neuroprotection against hypoxia-ischemia in neonatal rat brain by novel superoxide dismutase mimetics. Neurosci. Lett. 2003, 346, 41–44. [Google Scholar] [CrossRef]
- Spierer, A.; Rabinowitz, R.; Pri-Chen, S.; Rosner, M. An increase in superoxide dismutase ameliorates oxygen-induced retinopathy in transgenic mice. Eye 2005, 19, 86. [Google Scholar] [CrossRef][Green Version]
- Huang, H.F.; Guo, F.; Cao, Y.Z.; Shi, W.; Xia, Q. Neuroprotection by Manganese Superoxide Dismutase (M n SOD) Mimics: Antioxidant Effect and Oxidative Stress Regulation in Acute Experimental Stroke. CNS Neurosci. Ther. 2012, 18, 811–818. [Google Scholar] [CrossRef]
- Niesman, M.R.; Johnson, K.A.; Penn, J.S. Therapeutic Effect of Liposomal Superoxide Dismutase in an Animal Model of Retinopathy of Prematurity. Neurochem. Res. 1997, 22, 597–605. [Google Scholar] [CrossRef]
- Dohare, P.; Hyzinski-García, M.C.; Vipani, A.; Bowens, N.H.; Nalwalk, J.W.; Feustel, P.J.; Keller, R.W., Jr.; Jourd’Heuil, D.; Mongin, A.A. The neuroprotective properties of the superoxide dismutase mimetic tempol correlate with its ability to reduce pathological glutamate release in a rodent model of stroke. Free. Radic. Boil. Med. 2014, 77, 168–182. [Google Scholar] [CrossRef]
- Paraskevas, K.I.; Tzovaras, A.S.; Briana, D.D.; Mikhailidis, D.P. Emerging indications for statins: A pluripotent family of agents with several potential applications. Curr. Pharm. Des. 2007, 13, 3622–3636. [Google Scholar] [CrossRef] [PubMed]
- Bonetti, P.; Lerman, L.; Napoli, C. Statin effects beyond lipid lowering—Are they clinically relevant? Eur. Hear. J. 2003, 24, 225–248. [Google Scholar] [CrossRef]
- Wood, W.G.; Eckert, G.P.; Igbavboa, U.; Müller, W.E. Statins and neuroprotection: a prescription to move the field forward. Ann. N. Y. Acad. Sci. 2010, 1199, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Sigurdsson, G.; Haraldsdottir, S.O.; Melberg, T.H.; Tikkanen, M.J.; Miettinen, T.E.; Kristianson, K.J. Simvastatin compared to fluvastatin in the reduction of serum lipids and apolipoproteins in patients with ischaemic heart disease and moderate hypercholesterolaemia. Acta Cardiol. 1998, 53, 7–14. [Google Scholar] [PubMed]
- Johnson-Anuna, L.N.; Eckert, G.P.; Keller, J.H.; Igbavboa, U.; Franke, C.; Fechner, T.; Schubert-Zsilavecz, M.; Karas, M.; Müller, W.E.; Wood, W.G. Chronic administration of statins alters multiple gene expression patterns in mouse cerebral cortex. J. Pharmacol. Exp. Ther. 2005, 312, 786–793. [Google Scholar] [CrossRef]
- Franke, C.; Nöldner, M.; Abdel-Kader, R.; Johnson-Anuna, L.N.; Wood, W.G.; Müller, W.E.; Eckert, G.P. Bcl-2 upregulation and neuroprotection in guinea pig brain following chronic simvastatin treatment. Neurobiol. Dis. 2007, 25, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Bartoli, M.; Al-Shabrawey, M.; Labazi, M.; Behzadian, M.A.; Istanboli, M.; El-Remessy, A.B.; Caldwell, R.W.; Marcus, D.M.; Caldwell, R.B. HMG-CoA reductase inhibitors (statin) prevents retinal neovascularization in a model of oxygen-induced retinopathy. Investig. Ophthalmol. Vis. Sci. 2009, 50, 4934–4940. [Google Scholar] [CrossRef]
- Lee, M.-Y.; Kuan, Y.-H.; Chen, H.-Y.; Chen, T.-Y.; Chen, S.-T.; Huang, C.-C.; Yang, I.-P.; Hsu, Y.-S.; Wu, T.-S.; Lee, E.-J. Intravenous administration of melatonin reduces the intracerebral cellular inflammatory response following transient focal cerebral ischemia in rats. J. Pineal Res. 2007, 42, 297–309. [Google Scholar] [CrossRef]
- Chen, T.-Y.; Lee, M.-Y.; Chen, H.-Y.; Kuo, Y.-L.; Lin, S.-C.; Wu, T.-S.; Lee, E.-J. Melatonin attenuates the postischemic increase in blood-brain barrier permeability and decreases hemorrhagic transformation of tissue-plasminogen activator therapy following ischemic stroke in mice. J. Pineal Res. 2006, 40, 242–250. [Google Scholar] [CrossRef]
- Kondoh, T.; Uneyama, H.; Nishino, H.; Torii, K. Melatonin reduces cerebral edema formation caused by transient forebrain ischemia in rats. Life Sci. 2002, 72, 583–590. [Google Scholar] [CrossRef]
- Watson, N.; Diamandis, T.; Gonzales-Portillo, C.; Reyes, S.; Borlongan, C.V. Melatonin as an Antioxidant for Stroke Neuroprotection. Cell Transplant. 2016, 25, 883–891. [Google Scholar] [CrossRef] [PubMed]
- Alghamdi, B.S. The neuroprotective role of melatonin in neurological disorders. J. Neurosci. Res. 2018, 96, 1136–1149. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Alconada, D.; Álvarez, A.; Arteaga, O.; Martínez-Ibargüen, A.; Hilario, E. Neuroprotective Effect of Melatonin: A Novel Therapy against Perinatal Hypoxia-Ischemia. Int. J. Mol. Sci. 2013, 14, 9379–9395. [Google Scholar] [CrossRef]
- Tomás-Zapico, C.; Coto-Montes, A.; Tomás-Zapico, C.; Coto-Montes, A.; Tomás-Zapico, C.; Coto-Montes, A. A proposed mechanism to explain the stimulatory effect of melatonin on antioxidative enzymes. J. Pineal Res. 2005, 39, 99–104. [Google Scholar] [CrossRef]
- Kaur, C.; Sivakumar, V.; Robinson, R.; Foulds, W.S.; Luu, C.D.; Ling, E.A. Neuroprotective effect of melatonin against hypoxia-induced retinal ganglion cell death in neonatal rats. J. Pineal Res. 2013, 54, 190–206. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Lu, X.; Hu, Y.; Yang, B.; Tsui, C.-K.; Yu, S.; Lu, L.; Liang, X. Melatonin attenuated retinal neovascularization and neuroglial dysfunction by inhibition of HIF-1α-VEGF pathway in oxygen-induced retinopathy mice. J. Pineal Res. 2018, 64, e12473. [Google Scholar] [CrossRef] [PubMed]
- Simonyi, A. The neuroprotective effects of apocynin. Front. Biosci. 2012, 4, 2183. [Google Scholar] [CrossRef]
- Jackman, K.; Miller, A.; De Silva, T.; Crack, P.J.; Drummond, G.; Sobey, C. Reduction of cerebral infarct volume by apocynin requires pretreatment and is absent in Nox2-deficient mice. Br. J. Pharmacol. 2009, 156, 680–688. [Google Scholar] [CrossRef]
- Chen, H.; Song, Y.S.; Chan, P.H. Inhibition of NADPH oxidase is neuroprotective after ischemia-reperfusion. Br. J. Pharmacol. 2009, 29, 1262–1272. [Google Scholar] [CrossRef]
- Kelly, K.A.; Li, X.; Tan, Z.; Vangilder, R.L.; Rosen, C.L.; Huber, J.D. NOX2 inhibition with apocynin worsens stroke outcome in aged rats. Brain Res. 2009, 1292, 165–172. [Google Scholar] [CrossRef]
- Impellizzeri, D.; Mazzon, E.; Esposito, E.; Paterniti, I.; Bramanti, P.; Cuzzocrea, S. Effect of Apocynin, an inhibitor of NADPH oxidase, in the inflammatory process induced by an experimental model of spinal cord injury. Free Radic. Res. 2011, 45, 221–236. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Pallares, J.; Parga, J.A.; Muñoz, A.; Rey, P.; Guerra, M.J.; Labandeira-Garcia, J.L. Mechanism of 6-hydroxydopamine neurotoxicity: The role of NADPH oxidase and microglial activation in 6-hydroxydopamine-induced degeneration of dopaminergic neurons. J. Neurochem. 2007, 103, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Rey, P.; Lopez-Real, A.; Sánchez-Iglesias, S.; Muñoz, A.; Soto-Otero, R.; Labandeira-Garcia, J. Angiotensin type-1-receptor antagonists reduce 6-hydroxydopamine toxicity for dopaminergic neurons. Neurobiol. Aging 2007, 28, 555–567. [Google Scholar] [CrossRef] [PubMed]
- Al-Shabrawey, M.; Bartoli, M.; El-Remessy, A.B.; Platt, D.H.; Matragoon, S.; Behzadian, M.A.; Caldwell, R.W.; Caldwell, R.B. Inhibition of NAD(P)H Oxidase Activity Blocks Vascular Endothelial Growth Factor Overexpression and Neovascularization during Ischemic Retinopathy. Am. J. Pathol. 2005, 167, 599–607. [Google Scholar] [CrossRef]
- Saito, Y.; Uppal, A.; Byfield, G.; Budd, S.; Hartnett, M.E. Activated NAD(P)H Oxidase from Supplemental Oxygen Induces Neovascularization Independent of VEGF in Retinopathy of Prematurity Model. Investig. Opthalmol. Vis. Sci. 2008, 49, 1591–1598. [Google Scholar] [CrossRef] [PubMed]
- Johnston, P.G.; Gillam-Krakauer, M.; Fuller, M.P.; Reese, J. Evidence-Based Use of Indomethacin and Ibuprofen in the Neonatal Intensive Care Unit. Clin. Perinatol. 2012, 39, 111–136. [Google Scholar] [CrossRef]
- Parikh, P.; Juul, S.E. Neuroprotective Strategies in Neonatal Brain Injury. J. Pediatr. 2018, 192, 22–32. [Google Scholar] [CrossRef]
- Tutak, E.; Satar, M.; Zorludemir, S.; Erdogan, S.; Yapıcıoğlu, H.; Narlı, N. Neuroprotective Effects of Indomethacin and Aminoguanidine in the Newborn Rats with Hypoxic-Ischemic Cerebral Injury. Neurochem. Res. 2005, 30, 937–942. [Google Scholar] [CrossRef]
- Lambat, Z.; Conrad, N.; Anoopkumar-Dukie, S.; Walker, R.B.; Daya, S. An Investigation into the Neuroprotective Properties of Ibuprofen. Metab. Brain Dis. 2000, 15, 249–256. [Google Scholar] [CrossRef]
- Iwata, Y.; Nicole, O.; Zurakowski, D.; Okamura, T.; Jonas, R.A. Ibuprofen for neuroprotection after cerebral ischemia. J. Thorac. Cardiovasc. Surg. 2010, 139, 489–493. [Google Scholar] [CrossRef]
- Świątkiewicz, M.; Zaremba, M.; Joniec, I.; Członkowski, A.; Kurkowska-Jastrzębska, I. Potential neuroprotective effect of ibuprofen, insights from the mice model of Parkinson’s disease. Pharmacol. Rep. 2013, 65, 1227–1236. [Google Scholar] [CrossRef]
- Nandgaonkar, B.N.; Rotschild, T.; Yu, K.; Higgins, R.D. Indomethacin Improves Oxygen-Induced Retinopathy in the Mouse. Pediatr. Res. 1999, 46, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.; Barr, S.M.; Geng, Y.; Yun, Y.; Higgins, R.D. Ibuprofen improves oxygen-induced retinopathy in a mouse model. Curr. Eye Res. 2003, 27, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Solaroglu, I. Neuroprotective Effect of Granulocyte-Colony Stimulating Factor. Front. Biosci. 2007, 12, 712. [Google Scholar] [CrossRef]
- Schäbitz, W.-R.; Kollmar, R.; Schwaninger, M.; Juettler, E.; Bardutzky, J.; Schölzke, M.N.; Sommer, C.; Schwab, S. Neuroprotective Effect of Granulocyte Colony–Stimulating Factor After Focal Cerebral Ischemia. Stroke 2003, 34, 745–751. [Google Scholar]
- Komine-Kobayashi, M.; Zhang, N.; Liu, M.; Tanaka, R.; Hara, H.; Osaka, A.; Mochizuki, H.; Mizuno, Y.; Urabe, T. Neuroprotective effect of recombinant human granulocyte colony-stimulating factor in transient focal ischemia of mice. J. Cereb. Blood Flow Metab. 2006, 26, 402–413. [Google Scholar] [CrossRef] [PubMed]
- Meuer, K.; Pitzer, C.; Teismann, P.; Krüger, C.; Göricke, B.; Laage, R.; Lingor, P.; Peters, K.; Schlachetzki, J.C.; Kobayashi, K. Granulocyte-colony stimulating factor is neuroprotective in a model of Parkinson’s disease. J. Neurochem. 2006, 97, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Yanqing, Z.; Yu-Min, L.; Jian, Q.; Bao-Guo, X.; Chuan-Zhen, L. Fibronectin and neuroprotective effect of granulocyte colony-stimulating factor in focal cerebral ischemia. Brain Res. 2006, 1098, 161–169. [Google Scholar] [CrossRef]
- Hartung, T. Anti-inflammatory effects of granulocyte colony-stimulating factor. Curr. Opin. Hematol. 1998, 5, 221–225. [Google Scholar] [CrossRef]
- Kojima, H.; Otani, A.; Oishi, A.; Makiyama, Y.; Nakagawa, S.; Yoshimura, N. Granulocyte colony-stimulating factor attenuates oxidative stress–induced apoptosis in vascular endothelial cells and exhibits functional and morphologic protective effect in oxygen-induced retinopathy. Blood 2011, 117, 1091–1100. [Google Scholar] [CrossRef]
- Wilkinson-Berka, J.L.; Tan, G.; Binger, K.J.; Sutton, L.; McMaster, K.; Deliyanti, D.; Perera, G.; Campbell, D.J.; Miller, A.G.; Wilkinson-Berka, J.; et al. Aliskiren reduces vascular pathology in diabetic retinopathy and oxygen-induced retinopathy in the transgenic (mRen-2)27 rat. Diabetologia 2011, 54, 2724–2735. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.E.; Pianta, M.J.; Vingrys, A.J.; Wilkinson-Berka, J.L.; Fletcher, E.L.; Wilkinson-Berka, J.L.; Wilkinson-Berka, J.L. AT1 receptor inhibition prevents astrocyte degeneration and restores vascular growth in oxygen-induced retinopathy. Glia 2008, 56, 1076–1090. [Google Scholar] [CrossRef] [PubMed]
- Downie, L.E.; Hatzopoulos, K.M.; Pianta, M.J.; Vingrys, A.J.; Wilkinson-Berka, J.L.; Kalloniatis, M.; Fletcher, E.L.; Wilkinson-Berka, J.L.; Wilkinson-Berka, J.L. Angiotensin type-1 receptor inhibition is neuroprotective to amacrine cells in a rat model of retinopathy of prematurity. J. Comp. Neurol. 2010, 518, 41–63. [Google Scholar] [CrossRef] [PubMed]
- Biehl, J.K.; Russell, B. Introduction to stem cell therapy. J. Cardiovasc. Nurs. 2009, 24, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Lai, A.; Fu, Z.; Lo, A. Stem cell therapy for retinopathy of prematurity. Anat. Physiol. 2013. [Google Scholar] [CrossRef]
- Machalińska, A.; Modrzejewska, M.; Kotowski, M.; Dziedziejko, V.; Kucia, M.; Kawa, M.; Safranow, K.; Baśkiewicz-Masiuk, M.; Modrzejewska, A.; Karczewicz, D. Circulating stem cell populations in preterm infants: Implications for the development of retinopathy of prematurity. Arch. Ophthalmol. 2010, 128, 1311–1319. [Google Scholar] [CrossRef] [PubMed]
- Paczkowska, E.; Kucia, M.; Koziarska, D.; Halasa, M.; Safranow, K.; Masiuk, M.; Karbicka, A.; Nowik, M.; Nowacki, P.; Ratajczak, M.Z.; et al. Clinical Evidence That Very Small Embryonic-Like Stem Cells Are Mobilized into Peripheral Blood in Patients After Stroke. Stroke 2009, 40, 1237–1244. [Google Scholar] [CrossRef]
- Ratajczak, M.; Machalinski, B.; Wojakowski, W.; Ratajczak, J.; Kucia, M. A hypothesis for an embryonic origin of pluripotent Oct-4+ stem cells in adult bone marrow and other tissues. Leukemia 2007, 21, 860. [Google Scholar] [CrossRef]
- Ritter, M.R.; Banin, E.; Moreno, S.K.; Aguilar, E.; Dorrell, M.I.; Friedlander, M. Myeloid progenitors differentiate into microglia and promote vascular repair in a model of ischemic retinopathy. J. Clin. Investig. 2006, 116, 3266–3276. [Google Scholar] [CrossRef]
- Medina, R.J.; O’Neill, C.L.; Humphreys, M.W.; Gardiner, T.A.; Stitt, A.W. Outgrowth Endothelial Cells: Characterization and Their Potential for Reversing Ischemic Retinopathy. Investig. Opthalmol. Vis. Sci. 2010, 51, 5906–5913. [Google Scholar] [CrossRef]
- Noueihed, B.; Rivera, J.C.; Chemtob, S. AB028. Mesenchymal stem cells repair retinal vascular damage in retinopathy of prematurity mouse model. Ann. Eye Sci. 2018, 3, AB028. [Google Scholar] [CrossRef]
- Moisseiev, E.; Anderson, J.D.; Oltjen, S.; Goswami, M.; Zawadzki, R.J.; Nolta, J.A.; Park, S.S. Protective Effect of Intravitreal Administration of Exosomes Derived from Mesenchymal Stem Cells on Retinal Ischemia. Curr. Eye Res. 2017, 42, 1358–1367. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Liu, F.; Shi, M.; Sun, C.; Tan, Z.; Chang, X.; Li, Q.; Feng, Z. Bone marrow mesenchymal stem cells modified by angiogenin-1 promotes tissue repair in mice with oxygen-induced retinopathy of prematurity by promoting retinal stem cell proliferation and differentiation. J. Cell. Physiol. 2019, 234, 21027–21038. [Google Scholar] [CrossRef] [PubMed]
- Mendel, T.A.; Clabough, E.B.D.; Kao, D.S.; Demidova-Rice, T.N.; Durham, J.T.; Zotter, B.C.; Seaman, S.A.; Cronk, S.M.; Rakoczy, E.P.; Katz, A.J.; et al. Correction: Pericytes Derived from Adipose-Derived Stem Cells Protect against Retinal Vasculopathy. PLoS ONE 2013, 8, 65691. [Google Scholar] [CrossRef]
- Dogra, M.R.; Katoch, D. An Update on Retinopathy of Prematurity (ROP). Indian J. Pediatr. 2017, 84, 930–936. [Google Scholar] [CrossRef] [PubMed]
- Broxterman, E.C.; Hug, D.A. Retinopathy of Prematurity: A Review of Current Screening Guidelines and Treatment Options. Mo. Med. 2016, 113, 187–190. [Google Scholar] [PubMed]
- Cryotherapy for Retinopathy of Prematurity Cooperative Group. Multicenter trial of cryotherapy for retinopathy of prematurity: Preliminary results. Pediatrics 1988, 81, 697–706. [Google Scholar]
- Elsas, F.; Collins, M.; Jones, J.; Kimble, J.; Kline, L.; Witherspoon, D.; Roth, A.; Demorest, B.; Gilbert, W.; Plotsky, D. Multicenter trial of cryotherapy for retinopathy of prematurity: Ophthalmological outcomes at 10 years. Arch. Ophthalmol. 2001, 119, 1110–1118. [Google Scholar]
- Palmer, E.; Hardy, R.; Dobson, V.; Phelps, D.; Quinn, G.; Summers, C.; Krom, C.; Tung, B. 15-year outcomes following threshold retinopathy of prematurity: Final results from the multicenter trial of cryotherapy for retinopathy of prematurity. Arch. Ophthalmol. 2005, 123, 311–318. [Google Scholar] [PubMed]
- Clark, D.; Mandal, K. Treatment of retinopathy of prematurity. Early Hum. Dev. 2008, 84, 95–99. [Google Scholar] [CrossRef] [PubMed]
| Pathogenic Agents in ROP | Phase 1 in ROP Development | Phase 2 in ROP Development | Relevant Vascular Protective Agents in ROP Development (Phase of ROP) | Intervention | Animal Model | Beneficial Effect | Adverse Effect | Reference |
|---|---|---|---|---|---|---|---|---|
| VEGF | ↓ | ↑ | VEGF (Phase 1) | Intraocular injection | Rat OIR model |
| / | [16] |
| Bevacizumab (Phase 2) | Intravitreal injection | (Clinical study) |
|
| [17,18,19,20,21,22,23] | |||
| Ranibizumab (Phase 2) | Intravitreal injection | (Clinical study) | / | [19,24,25] | ||||
| Aflibercept (Phase 2) | Intravitreal injection | Mouse OIR model | / | [26] | ||||
| VEGFA shRNA (Phase 2) | Subretinal injection | Rat OIR model |
| / | [27] | |||
| Anti-KDR (Phase 2) | Surgical implantation | Dog OIR model |
| / | [28] | |||
| SRPIN340 (Phase 2) | Intraocular injection | Rat OIR model |
| / | [29] | |||
| Rapamycin (Phase 2) | Subcutaneous injection | Mouse OIR model |
| / | [30] | |||
| IGF-1 | ↓ | ↑ | rhIGF-1 (Phase 1) | Intraperitoneal injection | Mouse OIR model |
| / | [31] |
| IGFBP3 (Phase 1 and 2) | Knockout mouse | Mouse OIR model |
| / | [32,33] | |||
| Jb3 (Phase 2) | Subcutaneous injection | Mouse OIR model |
| / | [34] | |||
| Epo | ↓ | ↑ | / | / | / |
| / | / |
| HIF-1 | ↓ | ↑ | DMOG (Phase 1) | Intraperitoneal injection | Mouse OIR model |
| / | [35] |
| PHD2 (Phase 1 and 2) | Knockout mouse | Mouse OIR model | / | [36] | ||||
| RTP801 (Phase 1 and 2) | Knockout mouse | Mouse OIR model |
| / | [37] | |||
| NO | ↓ | ↑ | l-NA (Phase 2) | Intraperitoneal injection | Rat OIR model |
| / | [38] |
| l-NNA (Phase 2) | Intraperitoneal injection | Mouse OIR model | / | [39] | ||||
| AG (Phase 2) | Intravitreal injection | Mouse OIR model | / | [40] | ||||
| Adenosine | ↓ | ↑ | / | / | / | / | / | / |
| β-AR | ? | ↑ | Propranolol (Phase 2) | Subcutaneous injection | Mouse OIR model |
| / | [41] |
| Topical administration | Mouse OIR model | / | [42] | |||||
| Atenolol (Phase 2) | Subcutaneous injection | Mouse OIR model |
| / | [43] | |||
| ICI 118,551 (Phase 2) | / | |||||||
| SR59230A (Phase 2) | / | |||||||
| Other angiogenic agents | ? | ↑ | Dexamethasone (Phase 2) | Subcutaneous injection | Mouse OIR model |
|
| [44] |
| Anecortave acetate (Phase 2) | Intravitreal injection | Rat OIR model | [45] | |||||
| Degulin (Phase 2) | Intravitreal injection | Mouse OIR model |
| / | [46] | |||
| YC-1 (Phase 2) | Intravitreal injection | Mouse OIR model | / | [47] | ||||
| β-lapachone (Phase 2) | Intravitreal injection | Mouse OIR model | / | [48] | ||||
| 16K HPRL (Phase 2) | Intravitreal injection | Mouse OIR model |
| / | [49] | |||
| 12-LOX (Phase 2) | Intraperitoneal injection | Mouse OIR model | / | [50] | ||||
| TMP (Phase 2) | Intraperitoneal injection | Mouse OIR model |
| / | [51] | |||
| K5 (Phase 2) | Intravitreal injection | Rat OIR model | / | [52] | ||||
| MEF2C (Phase 1 and 2) | Knockout mouse | Mouse OIR model | / | [53] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsang, J.K.W.; Liu, J.; Lo, A.C.Y. Vascular and Neuronal Protection in the Developing Retina: Potential Therapeutic Targets for Retinopathy of Prematurity. Int. J. Mol. Sci. 2019, 20, 4321. https://doi.org/10.3390/ijms20174321
Tsang JKW, Liu J, Lo ACY. Vascular and Neuronal Protection in the Developing Retina: Potential Therapeutic Targets for Retinopathy of Prematurity. International Journal of Molecular Sciences. 2019; 20(17):4321. https://doi.org/10.3390/ijms20174321
Chicago/Turabian StyleTsang, Jessica K. W., Jin Liu, and Amy C. Y. Lo. 2019. "Vascular and Neuronal Protection in the Developing Retina: Potential Therapeutic Targets for Retinopathy of Prematurity" International Journal of Molecular Sciences 20, no. 17: 4321. https://doi.org/10.3390/ijms20174321
APA StyleTsang, J. K. W., Liu, J., & Lo, A. C. Y. (2019). Vascular and Neuronal Protection in the Developing Retina: Potential Therapeutic Targets for Retinopathy of Prematurity. International Journal of Molecular Sciences, 20(17), 4321. https://doi.org/10.3390/ijms20174321





