Carbon-Based Composites as Electrocatalysts for Oxygen Evolution Reaction in Alkaline Media
Abstract
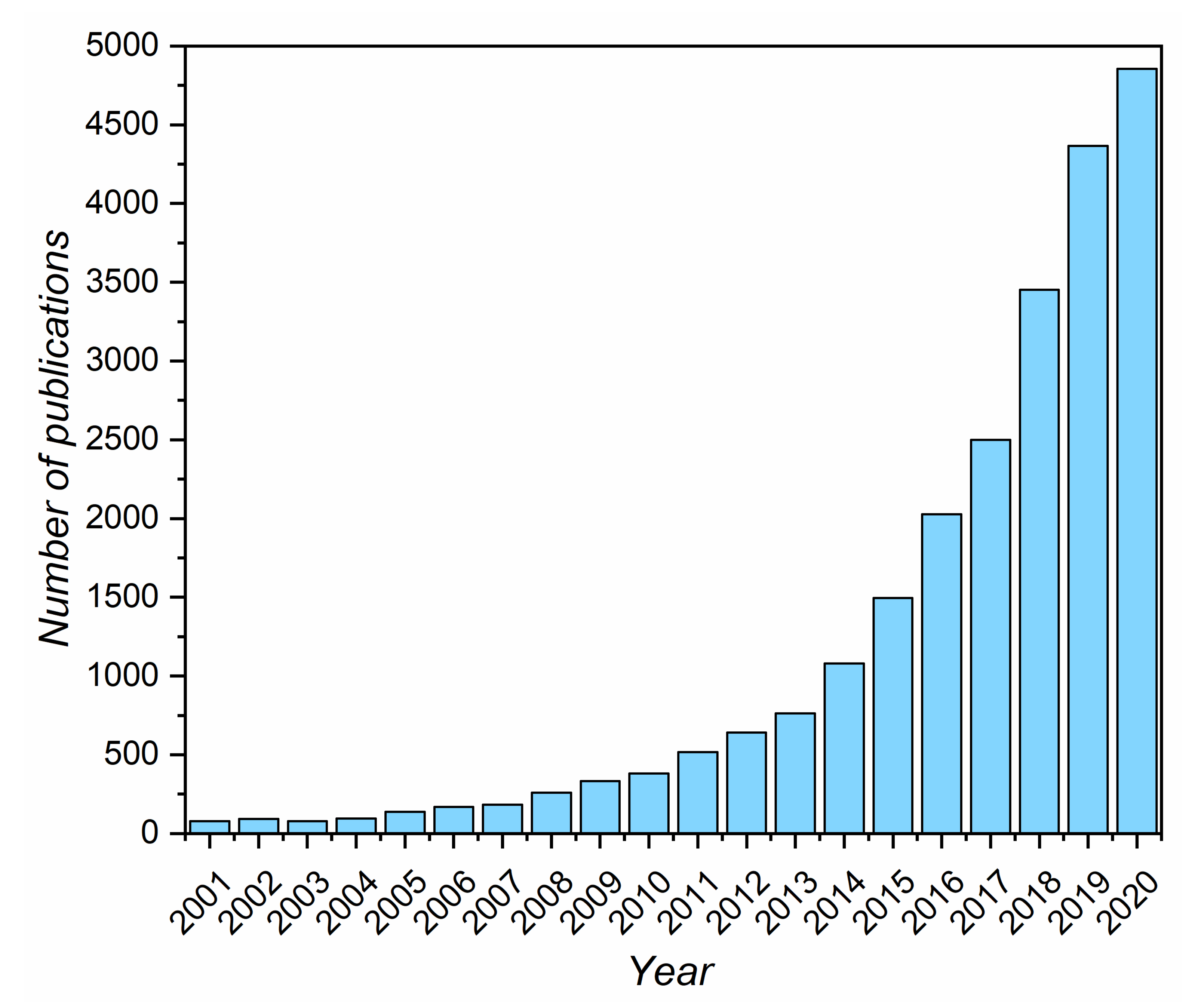
:1. Introduction
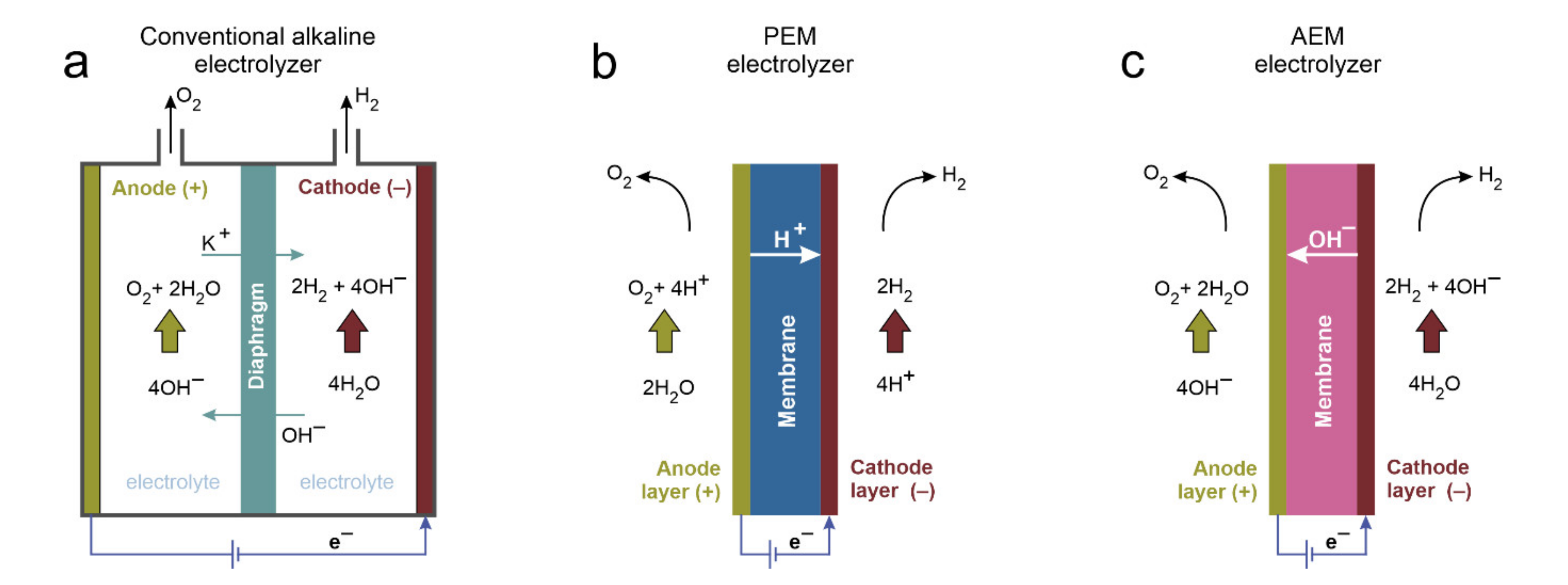
2. Water Splitting Systems
2.1. Proton Exchange Membrane Electrolyzers
2.2. Alkaline Exchange Membrane Electrolyzers
2.3. A Brief Overview of Carbon-Free Catalytic Materials for Oxygen Evolution Reaction in Alkaline Media
2.3.1. Metal Hydroxides/Oxyhydroxides
2.3.2. Metal Oxides
2.3.3. Metal Chalcogenides
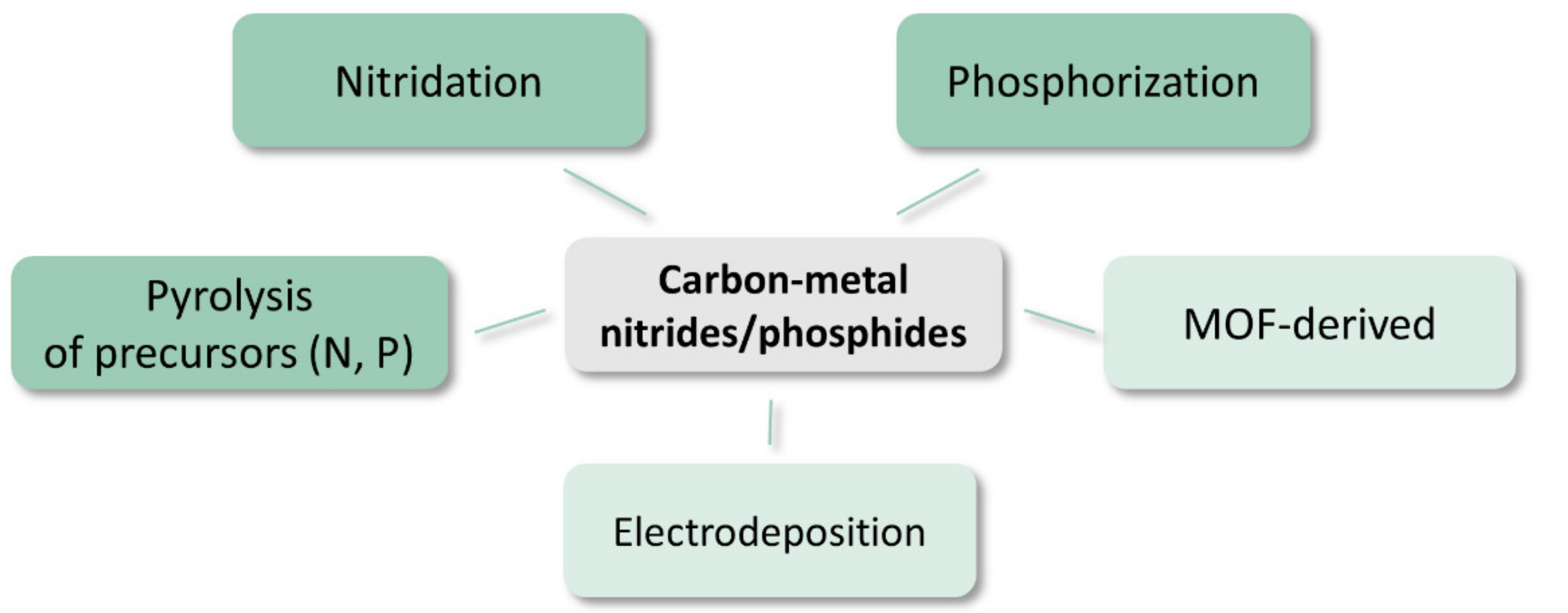
2.3.4. Metal Nitrides and Phosphides
3. Introduction to Carbon-Based Composite Materials
4. Synthesis of Carbon-Based Nanocomposites
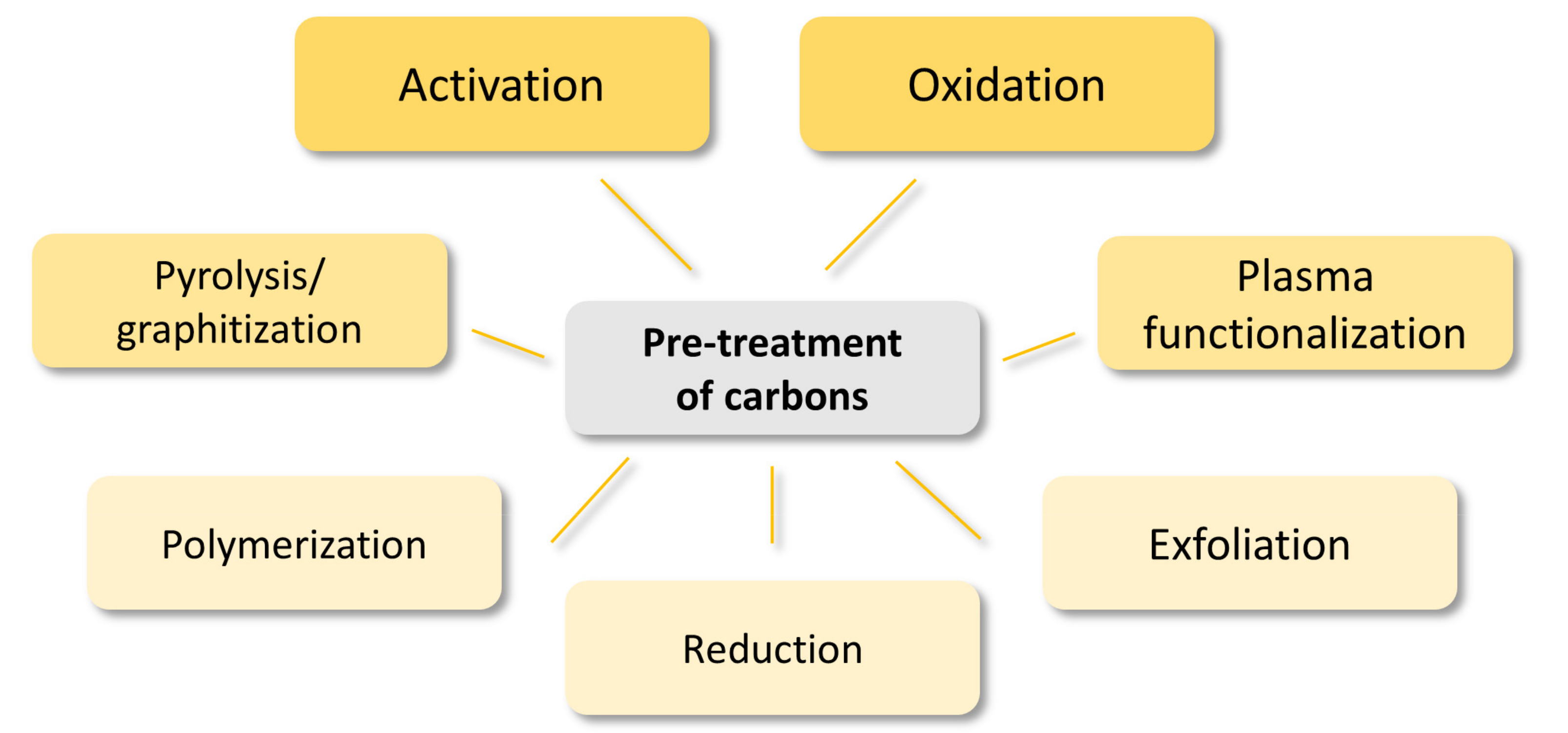
4.1. Pre-Treatment of Carbons
- Pyrolysis/graphitization
- Polymerization
- Plasma functionalization
- Exfoliation
- Reduction
- Oxidation
- Activation
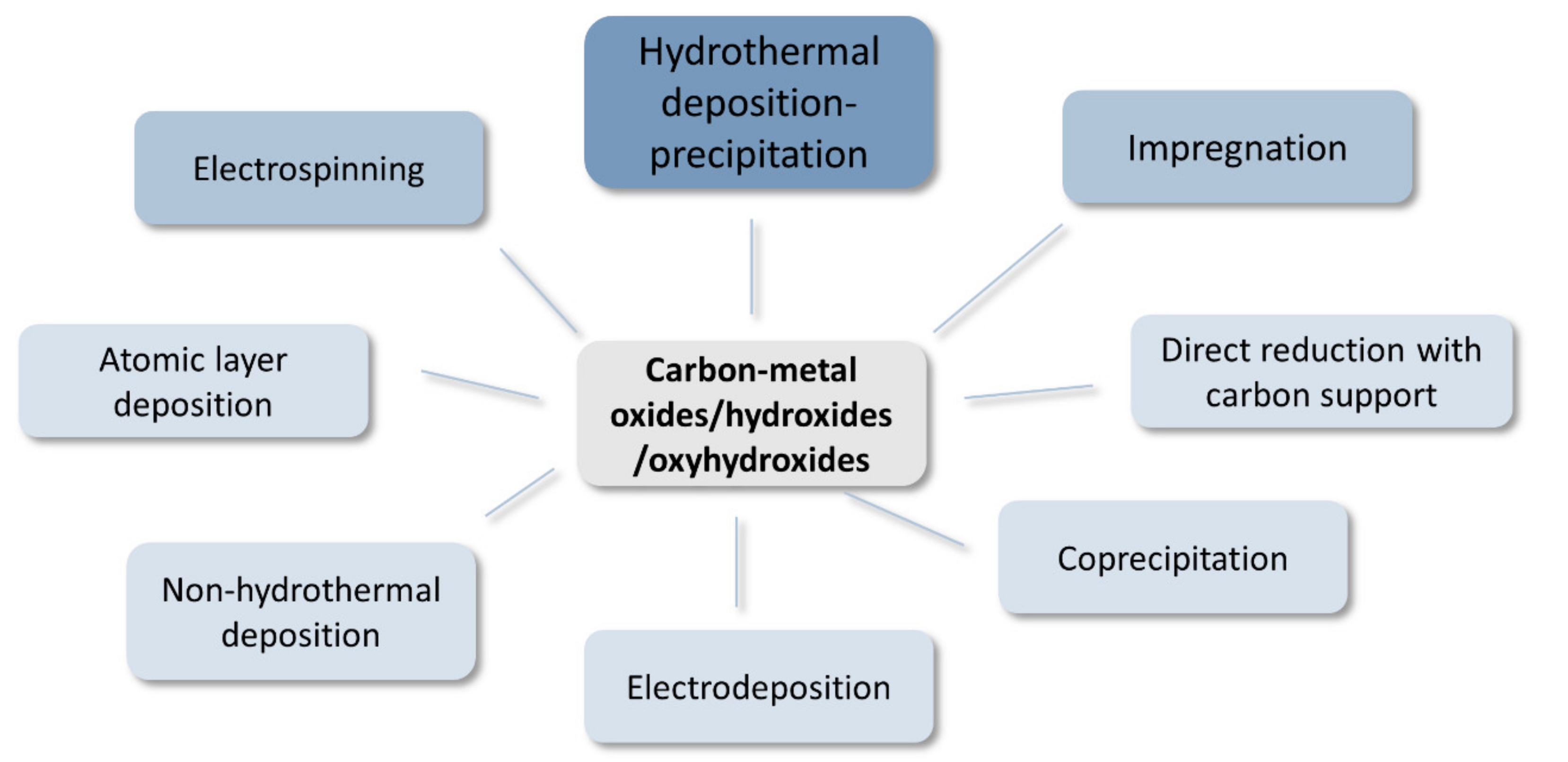
4.2. Synthesis of Carbon–Metal Hydroxides/Oxyhydroxides/Oxides
- Hydrothermal treatment of the oxide hydroxide precursors with carbon (hydrothermal deposition–precipitation)
- Non-hydrothermal deposition
- Electrospinning of precursors
- Impregnation
- Direct reduction with carbon support
- Electrodeposition
- Coprecipitation
- Atomic layer deposition
4.3. Synthesis of Carbon–Metal Chalcogenides
- Hydrothermal transformation
- High temperature selenization
- Other methods
4.4. Synthesis of Carbon-Nitrides and -Phosphides
- Pyrolysis of precursors
- Nitridation of metal precursors deposited on the carbon matrix
- Phosphorization of metal precursors with NaH2PO2
- Electrodeposition of phosphide phase
- Metal–organic framework-derived
- Phosphides and nitrides with protective carbon shell
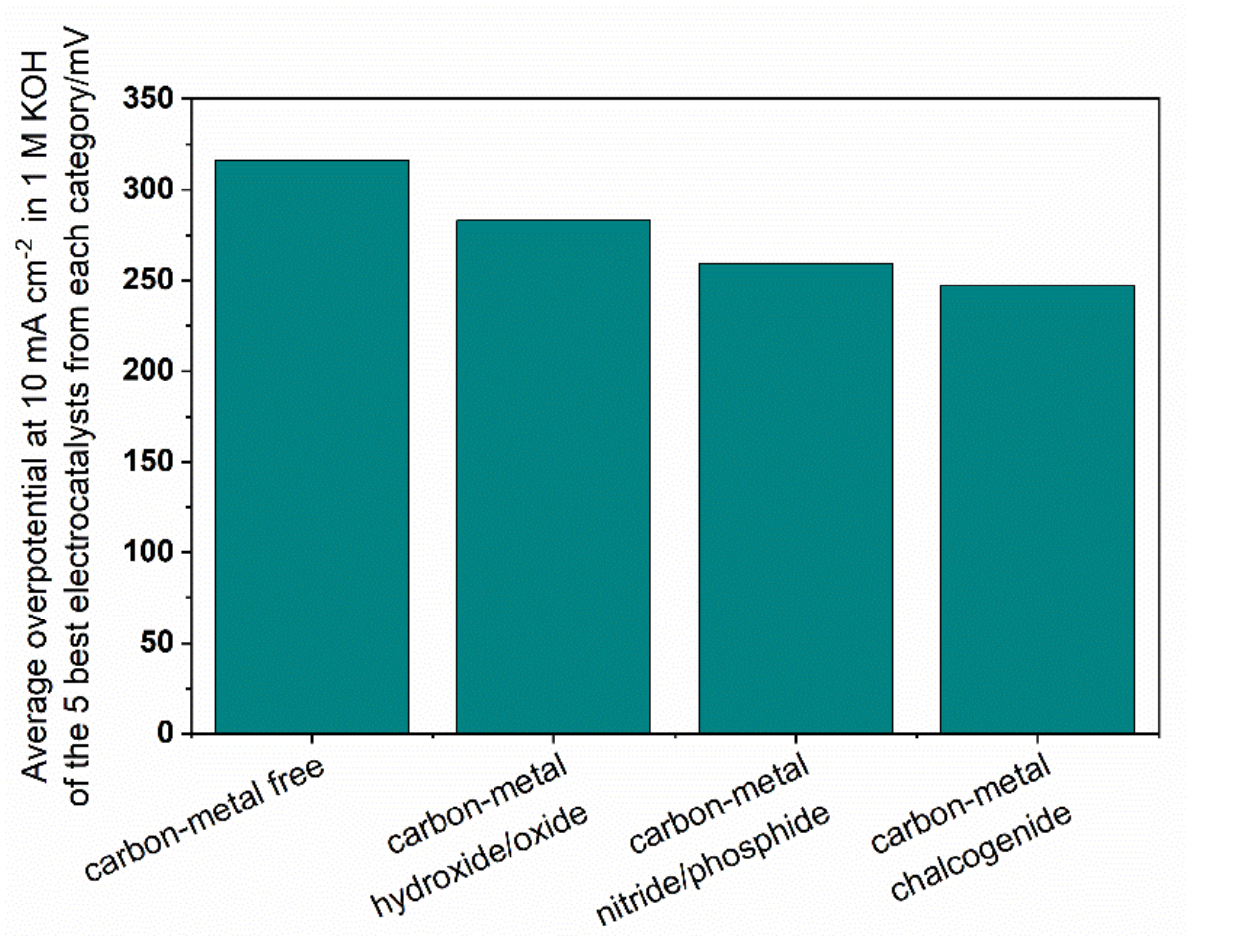
5. Reactivity of Carbon-Based Composite Materials
5.1. Metal-Free Carbon Composite Electrocatalysts
5.2. Carbon—Metal Hydroxides/Oxyhydroxides/Oxides
5.3. Carbon—Metal Chalcogenides
5.4. Carbon—Metal Nitrides and Phosphides
6. Interfaces in OER Electrocatalysts
7. Conclusions and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Lewis, N.S.; Nocera, D.G. Powering the planet: Chemical challenges in solar energy utilization. Proc. Natl. Acad. Sci. USA 2006, 103, 15729–15735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gray, H.B. Powering the planet with solar fuel. Nat. Chem. 2009, 1, 7. [Google Scholar] [CrossRef] [PubMed]
- Goodenough, J.B. Electrochemical energy storage in a sustainable modern society. Energy Environ. Sci. 2014, 7, 14–18. [Google Scholar] [CrossRef]
- David, M.; Ocampo-Martínez, C.; Sánchez-Peña, R. Advances in alkaline water electrolyzers: A review. J. Energy Storage 2019, 23, 392–403. [Google Scholar] [CrossRef] [Green Version]
- Shen, P.K.; Wang, C.-Y.; Jiang, S.P.; Sun, X.J.Z. Electrochemical Energy: Advanced Materials and Technologies; CRC Press: Boca Raton, FL, USA, 2015; ISBN 1482227282. [Google Scholar]
- Nadeem, F.; Hussain, S.M.S.; Tiwari, P.K.; Goswami, A.K.; Ustun, T.S. Comparative review of energy storage systems, their roles, and impacts on future power systems. IEEE Access 2019, 7, 4555–4585. [Google Scholar] [CrossRef]
- Bodner, M.; Hofer, A.; Hacker, V. H2 generation from alkaline electrolyzer. Wiley Interdiscip. Rev. Energy Environ. 2015, 4, 365–381. [Google Scholar] [CrossRef]
- Marchenko, O.V.; Solomin, S.V. The future energy: Hydrogen versus electricity. Int. J. Hydrogen Energy 2015, 40, 3801–3805. [Google Scholar] [CrossRef]
- Ayers, K.; Danilovic, N.; Ouimet, R.; Carmo, M.; Pivovar, B.; Bornstein, M. Perspectives on Low-Temperature Electrolysis and Potential for Renewable Hydrogen at Scale. Annu. Rev. Chem. Biomol. Eng. 2019, 10, 219–239. [Google Scholar] [CrossRef]
- Wang, Y.; Zou, Y.; Tao, L.; Wang, Y.; Huang, G.; Du, S.; Wang, S. Rational design of three-phase interfaces for electrocatalysis. Nano Res. 2019, 12, 2055–2066. [Google Scholar] [CrossRef]
- Reier, T.; Nong, H.N.; Teschner, D.; Schlögl, R.; Strasser, P. Electrocatalytic Oxygen Evolution Reaction in Acidic Environments–Reaction Mechanisms and Catalysts. Adv. Energy Mater. 2017, 7, 1601275. [Google Scholar] [CrossRef]
- Rossmeisl, J.; Qu, Z.W.; Zhu, H.; Kroes, G.J.; Nørskov, J.K. Electrolysis of water on oxide surfaces. J. Electroanal. Chem. 2007, 607, 83–89. [Google Scholar] [CrossRef]
- Pletcher, D.; Li, X. Prospects for alkaline zero gap water electrolysers for hydrogen production. Int. J. Hydrogen Energy 2011, 36, 15089–15104. [Google Scholar] [CrossRef] [Green Version]
- Chen, K.; Jiang, S.P. Review—Materials degradation of solid oxide electrolysis cells. J. Electrochem. Soc. 2016, 163, F3070–F3083. [Google Scholar] [CrossRef]
- Ding, C.; Shi, J.; Wang, Z.; Li, C. Photoelectrocatalytic Water Splitting: Significance of Cocatalysts, Electrolyte, and Interfaces. ACS Catal. 2017, 7, 675–688. [Google Scholar] [CrossRef]
- Tahir, M.; Pan, L.; Idrees, F.; Zhang, X.; Wang, L.; Zou, J.J.; Wang, Z.L. Electrocatalytic oxygen evolution reaction for energy conversion and storage: A comprehensive review; Elsevier: Amsterdam, The Netherlands, 2017; Volume 37, pp. 136–157. [Google Scholar]
- Vincent, I.; Bessarabov, D. Low cost hydrogen production by anion exchange membrane electrolysis: A review. Renew. Sustain. Energy Rev. 2018, 81, 1690–1704. [Google Scholar] [CrossRef]
- Abbasi, R.; Setzler, B.P.; Lin, S.; Wang, J.; Zhao, Y.; Xu, H.; Pivovar, B.; Tian, B.; Chen, X.; Wu, G.; et al. A Roadmap to Low-Cost Hydrogen with Hydroxide Exchange Membrane Electrolyzers. Adv. Mater. 2019, 31, 1805876. [Google Scholar] [CrossRef]
- Qi, J.; Zhang, W.; Cao, R. Solar-to-Hydrogen Energy Conversion Based on Water Splitting. Adv. Energy Mater. 2018, 8, 1701620. [Google Scholar] [CrossRef]
- Li, A.; Ooka, H.; Bonnet, N.; Hayashi, T.; Sun, Y.; Jiang, Q.; Li, C.; Han, H.; Nakamura, R. Stable Potential Windows for Long-Term Electrocatalysis by Manganese Oxides Under Acidic Conditions. Angew. Chem. Int. Ed. 2019, 58, 5054–5058. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Bonnet-Mercier, N.; Yamaguchi, A.; Suetsugu, K.; Nakamura, R. Electrochemical characterization of manganese oxides as a water oxidation catalyst in proton exchange membrane electrolysers. R. Soc. Open Sci. 2019, 6, 190122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, Y.S.; Jeong, J.; Noh, Y.; Jang, M.J.; Lee, J.; Lee, K.H.; Lim, D.C.; Seo, M.H.; Kim, W.B.; Yang, J.; et al. Commercial anion exchange membrane water electrolyzer stack through non-precious metal electrocatalysts. Appl. Catal. B Environ. 2021, 292, 120170. [Google Scholar] [CrossRef]
- Lindquist, G.A.; Oener, S.Z.; Krivina, R.; Motz, A.R.; Keane, A.; Capuano, C.; Ayers, K.E.; Boettcher, S.W. Performance and Durability of Pure-Water-Fed Anion Exchange Membrane Electrolyzers Using Baseline Materials and Operation. ACS Appl. Mater. Interfaces 2021. [Google Scholar] [CrossRef]
- Serov, A.; Kovnir, K.; Shatruk, M.; Kolen’ko, Y.V. Critical review of platinum group metal-free materials for water electrolysis: Transition from the laboratory to the market: Earth-abundant borides and phosphides as catalysts for sustainable hydrogen production. Johnson Matthey Technol. Rev. 2021, 65, 207–226. [Google Scholar] [CrossRef]
- Miller, H.A.; Bouzek, K.; Hnat, J.; Loos, S.; Bernäcker, C.I.; Weißgärber, T.; Röntzsch, L.; Meier-Haack, J. Green hydrogen from anion exchange membrane water electrolysis: A review of recent developments in critical materials and operating conditions. Sustain. Energy Fuels 2020, 4, 2114–2133. [Google Scholar] [CrossRef]
- Dekel, D.R. Review of Cell Performance in Anion Exchange Membrane Fuel Cells. J. Power Sources 2018, 375, 158–169. [Google Scholar] [CrossRef]
- Enapter electrolyser. Available online: https://www.enapter.com (accessed on 21 August 2021).
- Xu, D.; Stevens, M.B.; Cosby, M.R.; Oener, S.Z.; Smith, A.M.; Enman, L.J.; Ayers, K.E.; Capuano, C.B.; Renner, J.N.; Danilovic, N.; et al. Earth-Abundant Oxygen Electrocatalysts for Alkaline Anion-Exchange-Membrane Water Electrolysis: Effects of Catalyst Conductivity and Comparison with Performance in Three-Electrode Cells. ACS Catal. 2019, 9, 7–15. [Google Scholar] [CrossRef]
- Zhang, B.; Zheng, X.; Voznyy, O.; Comin, R.; Bajdich, M.; García-Melchor, M.; Han, L.; Xu, J.; Liu, M.; Zheng, L.; et al. Homogeneously dispersed multimetal oxygen-evolving catalysts. Science 2016, 352, 333–337. [Google Scholar] [CrossRef] [Green Version]
- Jin, H.; Mao, S.; Zhan, G.; Xu, F.; Bao, X.; Wang, Y. Fe incorporated α-Co(OH)2 nanosheets with remarkably improved activity towards the oxygen evolution reaction. J. Mater. Chem. A 2017, 5, 1078–1084. [Google Scholar] [CrossRef]
- Zhou, H.; Yu, F.; Zhu, Q.; Sun, J.; Qin, F.; Yu, L.; Bao, J.; Yu, Y.; Chen, S.; Ren, Z. Water splitting by electrolysis at high current densities under 1.6 volts. Energy Environ. Sci. 2018, 11, 2858–2864. [Google Scholar] [CrossRef]
- Ma, T.Y.; Dai, S.; Jaroniec, M.; Qiao, S.Z. Metal-organic framework derived hybrid Co3O4-carbon porous nanowire arrays as reversible oxygen evolution electrodes. J. Am. Chem. Soc. 2014, 136, 13925–13931. [Google Scholar] [CrossRef] [Green Version]
- Li, B.B.; Liang, Y.Q.; Yang, X.J.; Cui, Z.D.; Qiao, S.Z.; Zhu, S.L.; Li, Z.Y.; Yin, K. MoO2–CoO coupled with a macroporous carbon hybrid electrocatalyst for highly efficient oxygen evolution. Nanoscale 2015, 7, 16704–16714. [Google Scholar] [CrossRef]
- Zhang, P.; Li, L.; Nordlund, D.; Chen, H.; Fan, L.; Zhang, B.; Sheng, X.; Daniel, Q.; Sun, L. Dendritic core-shell nickel-iron-copper metal/metal oxide electrode for efficient electrocatalytic water oxidation. Nat. Commun. 2018, 9, 381. [Google Scholar] [CrossRef] [Green Version]
- Zhou, W.; Wu, X.J.; Cao, X.; Huang, X.; Tan, C.; Tian, J.; Liu, H.; Wang, J.; Zhang, H. Ni3S2 nanorods/Ni foam composite electrode with low overpotential for electrocatalytic oxygen evolution. Energy Environ. Sci. 2013, 6, 2921–2924. [Google Scholar] [CrossRef]
- Chen, S.; Kang, Z.; Zhang, X.; Xie, J.; Wang, H.; Shao, W.; Zheng, X.; Yan, W.; Pan, B.; Xie, Y. Highly Active Fe Sites in Ultrathin Pyrrhotite Fe7S8 Nanosheets Realizing Efficient Electrocatalytic Oxygen Evolution. ACS Cent. Sci. 2017, 3, 1221–1227. [Google Scholar] [CrossRef]
- Liu, Y.; Cheng, H.; Lyu, M.; Fan, S.; Liu, Q.; Zhang, W.; Zhi, Y.; Wang, C.; Xiao, C.; Wei, S.; et al. Low overpotential in vacancy-rich ultrathin CoSe2 nanosheets for water oxidation. J. Am. Chem. Soc. 2014, 136, 15670–15675. [Google Scholar] [CrossRef]
- Gao, Q.; Huang, C.Q.; Ju, Y.M.; Gao, M.R.; Liu, J.W.; An, D.; Cui, C.H.; Zheng, Y.R.; Li, W.X.; Yu, S.H. Phase-Selective Syntheses of Cobalt Telluride Nanofleeces for Efficient Oxygen Evolution Catalysts. Angew. Chem. Int. Ed. 2017, 56, 7769–7773. [Google Scholar] [CrossRef]
- Chen, P.; Xu, K.; Fang, Z.; Tong, Y.; Wu, J.; Lu, X.; Peng, X.; Ding, H.; Wu, C.; Xie, Y. Metallic Co4N porous nanowire arrays activated by surface oxidation as electrocatalysts for the oxygen evolution reaction. Angew. Chem. Int. Ed. 2015, 54, 14710–14714. [Google Scholar] [CrossRef]
- Yu, F.; Zhou, H.; Huang, Y.; Sun, J.; Qin, F.; Bao, J.; Goddard, W.A.; Chen, S.; Ren, Z. High-performance bifunctional porous non-noble metal phosphide catalyst for overall water splitting. Nat. Commun. 2018, 9, 2551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shinagawa, T.; Garcia-Esparza, A.T.; Takanabe, K. Insight on Tafel slopes from a microkinetic analysis of aqueous electrocatalysis for energy conversion. Sci. Rep. 2015, 5, 13801. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lyu, F.; Wang, Q.; Choi, S.M.; Yin, Y. Noble-Metal-Free Electrocatalysts for Oxygen Evolution. Small 2019, 15, 1804201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eftekhari, A. Tuning the electrocatalysts for oxygen evolution reaction. Mater. Today Energy 2017, 5, 37–57. [Google Scholar] [CrossRef]
- Gorlin, M.; De Araujo, J.F.; Schmies, H.; Bernsmeier, D.; Dresp, S.; Gliech, M.; Jusys, Z.; Chernev, P.; Kraehnert, R.; Dau, H.; et al. Tracking catalyst redox states and reaction dynamics in ni-fe oxyhydroxide oxygen evolution reaction electrocatalysts: The role of catalyst support and electrolyte pH. J. Am. Chem. Soc. 2017, 139, 2070–2082. [Google Scholar] [CrossRef]
- Song, F.; Bai, L.; Moysiadou, A.; Lee, S.; Hu, C.; Liardet, L.; Hu, X. Transition Metal Oxides as Electrocatalysts for the Oxygen Evolution Reaction in Alkaline Solutions: An Application-Inspired Renaissance. J. Am. Chem. Soc. 2018, 140, 7748–7759. [Google Scholar] [CrossRef]
- Dette, C.; Hurst, M.R.; Deng, J.; Nellist, M.R.; Boettcher, S.W. Structural Evolution of Metal (Oxy)hydroxide Nanosheets during the Oxygen Evolution Reaction. ACS Appl. Mater. Interfaces 2019, 11, 5590–5594. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhou, W.; Shao, Z. Perovskite/Carbon Composites: Applications in Oxygen Electrocatalysis. Small 2017, 13, 1603793. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Yan, Z.; Chen, C.; Chen, J. Spinels: Controlled Preparation, Oxygen Reduction/Evolution Reaction Application, and beyond. Chem. Rev. 2017, 117, 10121–10211. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.; Feng, Z.; Scherer, G.G.; Barber, J.; Shao-Horn, Y.; Xu, Z.J. Cations in Octahedral Sites: A Descriptor for Oxygen Electrocatalysis on Transition-Metal Spinels. Adv. Mater. 2017, 29, 1–8. [Google Scholar] [CrossRef]
- Jung, J.I.; Risch, M.; Park, S.; Kim, M.G.; Nam, G.; Jeong, H.Y.; Shao-Horn, Y.; Cho, J. Optimizing nanoparticle perovskite for bifunctional oxygen electrocatalysis. Energy Environ. Sci. 2016, 9, 176–183. [Google Scholar] [CrossRef] [Green Version]
- Monteverde Videla, A.H.A.; Stelmachowski, P.; Ercolino, G.; Specchia, S. Benchmark comparison of Co3O4 spinel-structured oxides with different morphologies for oxygen evolution reaction under alkaline conditions. J. Appl. Electrochem. 2017, 47, 295–304. [Google Scholar] [CrossRef]
- Stelmachowski, P.; Monteverde Videla, A.H.A.; Ciura, K.; Specchia, S. Oxygen evolution catalysis in alkaline conditions over hard templated nickel-cobalt based spinel oxides. Int. J. Hydrogen Energy 2017, 42, 27910–27918. [Google Scholar] [CrossRef]
- Stelmachowski, P.; Monteverde Videla, A.H.A.; Jakubek, T.; Kotarba, A.; Specchia, S. The Effect of Fe, Co, and Ni Structural Promotion of Cryptomelane (KMn8O16) on the Catalytic Activity in Oxygen Evolution Reaction. Electrocatalysis 2018, 9, 762–769. [Google Scholar] [CrossRef] [Green Version]
- Moon, G.H.; Yu, M.; Chan, C.K.; Tüysüz, H. Highly Active Cobalt-Based Electrocatalysts with Facile Incorporation of Dopants for the Oxygen Evolution Reaction. Angew. Chem. Int. Ed. 2019, 58, 3491–3495. [Google Scholar] [CrossRef] [PubMed]
- Rana, M.; Mondal, S.; Sahoo, L.; Chatterjee, K.; Karthik, P.E.; Gautam, U.K. Emerging Materials in Heterogeneous Electrocatalysis Involving Oxygen for Energy Harvesting. ACS Appl. Mater. Interfaces 2018, 10, 33737–33767. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, A.; Jones, T.E.; Martinez Moreno, E.; Teschner, D.; Chernev, P.; Gliech, M.; Reier, T.; Dau, H.; Strasser, P. Unified structural motifs of the catalytically active state of Co(oxyhydr)oxides during the electrochemical oxygen evolution reaction. Nat. Catal. 2018, 1, 711–719. [Google Scholar] [CrossRef]
- Suen, N.T.; Hung, S.F.; Quan, Q.; Zhang, N.; Xu, Y.J.; Chen, H.M. Electrocatalysis for the oxygen evolution reaction: Recent development and future perspectives. Chem. Soc. Rev. 2017, 46, 337–365. [Google Scholar] [CrossRef] [PubMed]
- Shirotani, I.; Adachi, T.; Tachi, K.; Todo, S.; Nozawa, K.; Yagi, T.; Kinoshita, M. Electrical conductivity and superconductivity of metal phosphides with skutterudite-type structure prepared at high pressure. J. Phys. Chem. Solids 1996, 57, 211–216. [Google Scholar] [CrossRef]
- Karaca, E.; Tütüncü, H.M.; Srivastava, G.P.; Uur, S. Electron-phonon superconductivity in the ternary phosphides BaM2P2 (M=Ni,Rh,and Ir). Phys. Rev. B 2016, 94, 054507. [Google Scholar] [CrossRef] [Green Version]
- Shi, Y.; Zhang, B. Recent advances in transition metal phosphide nanomaterials: Synthesis and applications in hydrogen evolution reaction. Chem. Soc. Rev. 2016, 45, 1529–1541. [Google Scholar] [CrossRef] [PubMed]
- Jin, S. Are Metal Chalcogenides, Nitrides, and Phosphides Oxygen Evolution Catalysts or Bifunctional Catalysts? ACS Energy Lett. 2017, 2, 1937–1938. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Kong, B.; Zhao, D.; Wang, H.; Selomulya, C. Strategies for developing transition metal phosphides as heterogeneous electrocatalysts for water splitting. Nano Today 2017, 15, 26–55. [Google Scholar] [CrossRef]
- Xu, K.; Cheng, H.; Liu, L.; Lv, H.; Wu, X.; Wu, C.; Xie, Y. Promoting Active Species Generation by Electrochemical Activation in Alkaline Media for Efficient Electrocatalytic Oxygen Evolution in Neutral Media. Nano Lett. 2017, 17, 578–583. [Google Scholar] [CrossRef]
- Tang, C.; Titirici, M.M.; Zhang, Q. A review of nanocarbons in energy electrocatalysis: Multifunctional substrates and highly active sites. J. Energy Chem. 2017, 26, 1077–1093. [Google Scholar] [CrossRef] [Green Version]
- Dionigi, F.; Strasser, P. NiFe-Based (Oxy)hydroxide Catalysts for Oxygen Evolution Reaction in Non-Acidic Electrolytes. Adv. Energy Mater. 2016, 6, 1600621. [Google Scholar] [CrossRef]
- Hu, Q.; Li, G.; Han, Z.; Wang, Z.; Huang, X.; Yang, H.; Zhang, Q.; Liu, J.; He, C. Recent progress in the hybrids of transition metals/carbon for electrochemical water splitting. J. Mater. Chem. A 2019, 7, 14380–14390. [Google Scholar] [CrossRef]
- Liang, Y.; Zhang, W.; Wu, D.; Ni, Q.Q.; Zhang, M.Q. Interface Engineering of Carbon-Based Nanocomposites for Advanced Electrochemical Energy Storage. Adv. Mater. Interfaces 2018, 5, 1800430. [Google Scholar] [CrossRef]
- Wang, L.; Fu, L.; Li, J.; Zeng, X.; Xie, H.; Huang, X.; Wang, H.; Tang, Y. On an easy way to prepare highly efficient Fe/N-co-doped carbon nanotube/nanoparticle composite for oxygen reduction reaction in Al–air batteries. J. Mater. Sci. 2018, 53, 10280–10291. [Google Scholar] [CrossRef]
- Leonard, D.P.; Stickle, W.F.; Ji, X. Carbon-Supported Iron Phosphides: Highest Intrinsic Oxygen Evolution Activity of the Iron Triad. ACS Appl. Energy Mater. 2018, 1, 3593–3597. [Google Scholar] [CrossRef]
- Chung, H.T.; Won, J.H.; Zelenay, P. Active and stable carbon nanotube/nanoparticle composite electrocatalyst for oxygen reduction. Nat. Commun. 2013, 4, 1922. [Google Scholar] [CrossRef] [Green Version]
- Youn, D.H.; Han, S.; Kim, J.Y.; Kim, J.Y.; Park, H.; Choi, S.H.; Lee, J.S. Highly active and stable hydrogen evolution electrocatalysts based on molybdenum compounds on carbon nanotube-graphene hybrid support. ACS Nano 2014, 8, 5164–5173. [Google Scholar] [CrossRef]
- Ma, T.Y.; Dai, S.; Jaroniec, M.; Qiao, S.Z. Graphitic Carbon Nitride Nanosheet–Carbon Nanotube Three-Dimensional Porous Composites as High-Performance Oxygen Evolution Electrocatalysts. Angew. Chem. 2014, 126, 7409–7413. [Google Scholar] [CrossRef]
- Ji, D.; Fan, L.; Li, L.; Mao, N.; Qin, X.; Peng, S.; Ramakrishna, S. Hierarchical catalytic electrodes of cobalt-embedded carbon nanotube/carbon flakes array for flexible solid-state zinc-air batteries. Carbon 2019, 142, 379–387. [Google Scholar] [CrossRef]
- Khalid, M.; Honorato, A.M.B.; Pasa, A.A.; Varela, H. A sugar derived carbon-red phosphorus composite for oxygen evolution reaction and supercapacitor activities. Mater. Sci. Energy Technol. 2020, 3, 508–514. [Google Scholar] [CrossRef]
- Pérez-Rodríguez, S.; Sebastián, D.; Lázaro, M.J. Electrochemical oxidation of ordered mesoporous carbons and the influence of graphitization. Electrochim. Acta 2019, 303, 167–175. [Google Scholar] [CrossRef] [Green Version]
- Pérez-Rodríguez, S.; Sebastián, D.; Lázaro, M.J. Insights on the Electrochemical Oxidation of Ordered Mesoporous Carbons. J. Electrochem. Soc. 2020, 167, 024511. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, S.; Song, H.; Guo, H.; Zhang, K.A.I.; Zhang, C.; Liu, T. Nitrogen-doped hollow carbon nanoflowers from a preformed covalent triazine framework for metal-free bifunctional electrocatalysis. Nanoscale 2020, 12, 14441–14447. [Google Scholar] [CrossRef]
- Wahab, M.A.; Joseph, J.; Atanda, L.; Sultana, U.K.; Beltramini, J.N.; Ostrikov, K.; Will, G.; O’Mullane, A.P.; Abdala, A. Nanoconfined Synthesis of Nitrogen-Rich Metal-Free Mesoporous Carbon Nitride Electrocatalyst for the Oxygen Evolution Reaction. ACS Appl. Energy Mater. 2020, 3, 1439–1447. [Google Scholar] [CrossRef]
- Li, O.L.; Pham, N.N.T.; Kim, J.; Choi, H.; Lee, D.H.; Yang, Y.; Yao, W.; Cho, Y.R.; Lee, S.G. Insights on boosting oxygen evolution reaction performance via boron incorporation into nitrogen-doped carbon electrocatalysts. Appl. Surf. Sci. 2020, 528, 146979. [Google Scholar] [CrossRef]
- Tsounis, C.; Lu, X.; Bedford, N.M.; Subhash, B.; Thomsen, L.; Zhang, Q.; Ma, Z.; Ostrikov, K.; Bendavid, A.; Scott, J.A.; et al. Valence Alignment of Mixed Ni-Fe Hydroxide Electrocatalysts through Preferential Templating on Graphene Edges for Enhanced Oxygen Evolution. ACS Nano 2020, 14, 11327–11340. [Google Scholar] [CrossRef]
- Li, R.; Xu, J.; Lu, C.; Huang, Z.; Wu, Q.; Ba, J.; Tang, T.; Meng, D.; Luo, W. Amorphous NiFe phosphides supported on nanoarray-structured nitrogen-doped carbon paper for high-performance overall water splitting. Electrochim. Acta 2020, 357, 136873. [Google Scholar] [CrossRef]
- Hummers, W.S. Preparation of Graphitic Acid. U.S. Patent 2,798,878, 9 July 1957. [Google Scholar]
- Hummers, W.S.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- He, J.Z.; Niu, W.J.; Wang, Y.P.; Sun, Q.Q.; Liu, M.J.; Wang, K.; Liu, W.W.; Liu, M.C.; Yu, F.C.; Chueh, Y.L. In-situ synthesis of hybrid nickel cobalt sulfide/carbon nitrogen nanosheet composites as highly efficient bifunctional oxygen electrocatalyst for rechargeable Zn-air batteries. Electrochim. Acta 2020, 362, 136968. [Google Scholar] [CrossRef]
- Lei, C.; Chen, H.; Cao, J.; Yang, J.; Qiu, M.; Xia, Y.; Yuan, C.; Yang, B.; Li, Z.; Zhang, X.; et al. Fe–N4 Sites Embedded into Carbon Nanofiber Integrated with Electrochemically Exfoliated Graphene for Oxygen Evolution in Acidic Medium. Adv. Energy Mater. 2018, 8, 1801912. [Google Scholar] [CrossRef]
- Rivera-Gavidia, L.M.; Fernández de la Puente, I.; Hernández-Rodríguez, M.A.; Celorrio, V.; Sebastián, D.; Lázaro, M.J.; Pastor, E.; García, G. Bi-functional carbon-based catalysts for unitized regenerative fuel cells. J. Catal. 2020, 387, 138–144. [Google Scholar] [CrossRef]
- Morales, D.M.; Kazakova, M.A.; Dieckhöfer, S.; Selyutin, A.G.; Golubtsov, G.V.; Schuhmann, W.; Masa, J. Trimetallic Mn-Fe-Ni Oxide Nanoparticles Supported on Multi-Walled Carbon Nanotubes as High-Performance Bifunctional ORR/OER Electrocatalyst in Alkaline Media. Adv. Funct. Mater. 2020, 30. [Google Scholar] [CrossRef]
- Li, L.; Yang, J.; Yang, H.; Zhang, L.; Shao, J.; Huang, W.; Liu, B.; Dong, X. Anchoring Mn3O4 Nanoparticles on Oxygen Functionalized Carbon Nanotubes as Bifunctional Catalyst for Rechargeable Zinc-Air Battery. ACS Appl. Energy Mater. 2018, 1, 963–969. [Google Scholar] [CrossRef]
- Zhao, T.; Gadipelli, S.; He, G.; Ward, M.J.; Do, D.; Zhang, P.; Guo, Z. Tunable Bifunctional Activity of MnxCo3−xO4 Nanocrystals Decorated on Carbon Nanotubes for Oxygen Electrocatalysis. ChemSusChem 2018, 11, 1248. [Google Scholar] [CrossRef]
- Li, B.; Song, F.; Qian, Y.; Shaw, J.; Rao, Y. Boron-doped graphene oxide-supported nickel nitride nanoparticles for electrocatalytic oxygen evolution in alkaline electrolytes. ACS Appl. Nano Mater. 2020, 3, 9924–9930. [Google Scholar] [CrossRef]
- Han, X.; Yu, C.; Yang, J.; Song, X.; Zhao, C.; Li, S.; Zhang, Y.; Huang, H.; Liu, Z.; Huang, H.; et al. Electrochemically Driven Coordination Tuning of FeOOH Integrated on Carbon Fiber Paper for Enhanced Oxygen Evolution. Small 2019, 15, 1901015. [Google Scholar] [CrossRef]
- Hoang, V.C.; Dinh, K.N.; Gomes, V.G. Hybrid Ni/NiO composite with N-doped activated carbon from waste cauliflower leaves: A sustainable bifunctional electrocatalyst for efficient water splitting. Carbon 2020, 157, 515–524. [Google Scholar] [CrossRef]
- Prabu, N.; Kesavan, T.; Maduraiveeran, G.; Sasidharan, M. Bio-derived nanoporous activated carbon sheets as electrocatalyst for enhanced electrochemical water splitting. Int. J. Hydrogen Energy 2019, 44, 19995–20006. [Google Scholar] [CrossRef]
- Kordek, K.; Yin, H.; Rutkowski, P.; Zhao, H. Cobalt-based composite films on electrochemically activated carbon cloth as high performance overall water splitting electrodes. Int. J. Hydrogen Energy 2019, 44, 23–33. [Google Scholar] [CrossRef]
- Kordek, K.; Jiang, L.; Fan, K.; Zhu, Z.; Xu, L.; Al-Mamun, M.; Dou, Y.; Chen, S.; Liu, P.; Yin, H.; et al. Two-Step Activated Carbon Cloth with Oxygen-Rich Functional Groups as a High-Performance Additive-Free Air Electrode for Flexible Zinc–Air Batteries. Adv. Energy Mater. 2019, 9, 1802936. [Google Scholar] [CrossRef]
- Wu, Z.; Li, P.; Qin, Q.; Li, Z.; Liu, X. N-doped graphene combined with alloys (NiCo, CoFe) and their oxides as multifunctional electrocatalysts for oxygen and hydrogen electrode reactions. Carbon 2018, 139, 35–44. [Google Scholar] [CrossRef]
- Luo, W.; Wang, J.; Hu, J.; Ji, Y.; Streb, C.; Song, Y.F. Composite Metal Oxide-Carbon Nanotube Electrocatalysts for the Oxygen Evolution and Oxygen Reduction Reactions. ChemElectroChem 2018, 5, 2850–2856. [Google Scholar] [CrossRef]
- Oh, T.; Ryu, S.; Oh, H.; Kim, J. MnCo2O4 nanoparticles supported on nitrogen and sulfur co-doped mesoporous carbon spheres as efficient electrocatalysts for oxygen catalytic reactions. Dalt. Trans. 2019, 48, 945–953. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Fan, M.; Tu, W.; Chen, K.; Shen, Y.; Zhang, H. In situ growth of Co3O4 on nitrogen-doped hollow carbon nanospheres as air electrode for lithium-air batteries. J. Alloys Compd. 2019, 777, 944–953. [Google Scholar] [CrossRef]
- Wang, F.; Chen, G.; Liu, X.; Chen, F.; Wan, H.; Ni, L.; Zhang, N.; Ma, R.; Qiu, G. Advanced Electrocatalytic Performance of Ni-Based Materials for Oxygen Evolution Reaction. ACS Sustain. Chem. Eng. 2019, 7, 341–349. [Google Scholar] [CrossRef]
- Qin, Q.; Chen, L.; Wei, T.; Wang, Y.; Liu, X. Ni/NiM2O4 (M = Mn or Fe) supported on N-doped carbon nanotubes as trifunctional electrocatalysts for ORR, OER and HER. Catal. Sci. Technol. 2019, 9, 1595–1601. [Google Scholar] [CrossRef]
- Kim, J.E.; Lim, J.; Lee, G.Y.; Choi, S.H.; Maiti, U.N.; Lee, W.J.; Lee, H.J.; Kim, S.O. Subnanometer Cobalt-Hydroxide-Anchored N-Doped Carbon Nanotube Forest for Bifunctional Oxygen Catalyst. ACS Appl. Mater. Interfaces 2016, 8, 1571–1577. [Google Scholar] [CrossRef]
- Jia, H.; Wang, Z.; Li, C.; Si, X.; Zheng, X.; Cai, Y.; Lin, J.; Liang, H.; Qi, J.; Cao, J.; et al. Designing oxygen bonding between reduced graphene oxide and multishelled Mn3O4 hollow spheres for enhanced performance of supercapacitors. J. Mater. Chem. A 2019, 7, 6686–6694. [Google Scholar] [CrossRef]
- Busacca, C.; Zignani, S.C.; Di Blasi, A.; Di Blasi, O.; Lo Faro, M.; Antonucci, V.; Aricò, A.S. Electrospun NiMn2O4 and NiCo2O4 spinel oxides supported on carbon nanofibers as electrocatalysts for the oxygen evolution reaction in an anion exchange membrane-based electrolysis cell. Int. J. Hydrogen Energy 2019. [Google Scholar] [CrossRef]
- Li, T.; Li, S.; Liu, Q.; Tian, Y.; Zhang, Y.; Fu, G.; Tang, Y. Hollow Co3O4/CeO2 Heterostructures in Situ Embedded in N-Doped Carbon Nanofibers Enable Outstanding Oxygen Evolution. ACS Sustain. Chem. Eng. 2019, 7, 17950–17957. [Google Scholar] [CrossRef]
- Kazakova, M.A.; Morales, D.M.; Andronescu, C.; Elumeeva, K.; Selyutin, A.G.; Ishchenko, A.V.; Golubtsov, G.V.; Dieckhöfer, S.; Schuhmann, W.; Masa, J. Fe/Co/Ni mixed oxide nanoparticles supported on oxidized multi-walled carbon nanotubes as electrocatalysts for the oxygen reduction and the oxygen evolution reactions in alkaline media. Catal. Today 2020, 357, 259–268. [Google Scholar] [CrossRef]
- Hof, F.; Liu, M.; Valenti, G.; Picheau, E.; Paolucci, F.; Pénicaud, A. Size Control of Nanographene Supported Iron Oxide Nanoparticles Enhances Their Electrocatalytic Performance for the Oxygen Reduction and Oxygen Evolution Reactions. J. Phys. Chem. C 2019, 123, 20774–20780. [Google Scholar] [CrossRef]
- Hu, Q.; Liu, X.; Tang, C.; Fan, L.; Chai, X.; Zhang, Q.; Liu, J.; He, C. High efficiency oxygen evolution reaction enabled by 3D network composed of nitrogen-doped graphitic carbon-coated metal/metal oxide heterojunctions. Electrochim. Acta 2018, 265, 620–628. [Google Scholar] [CrossRef]
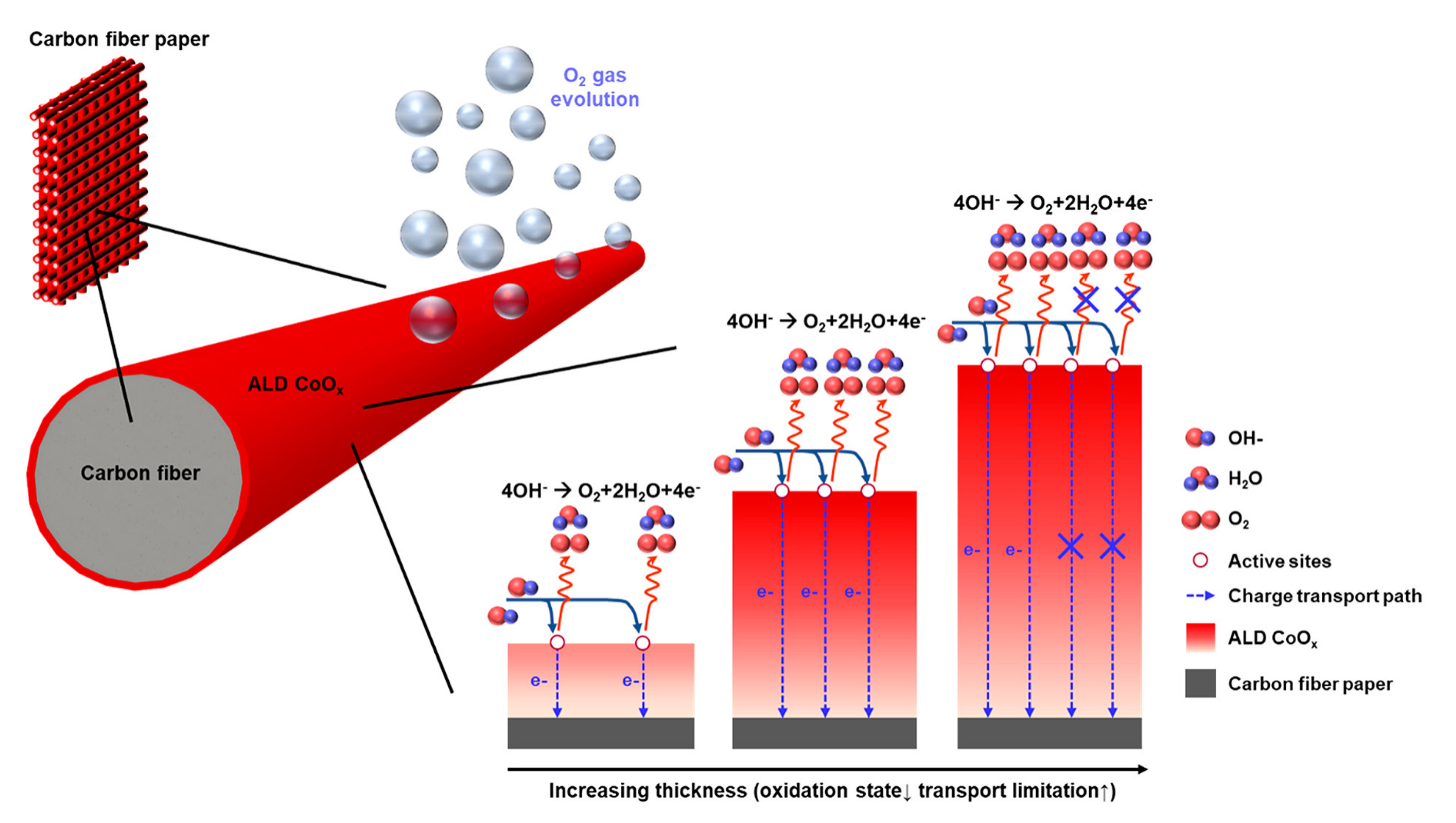
- Choi, H.J.; Han, G.D.; Bae, K.; Shim, J.H. Highly Active Oxygen Evolution on Carbon Fiber Paper Coated with Atomic-Layer-Deposited Cobalt Oxide. ACS Appl. Mater. Interfaces 2019, 11, 10608–10615. [Google Scholar] [CrossRef] [PubMed]
- Gaur, A.; Sachdeva, P.K.; Kumar, R.; Maruyama, T.; Bera, C.; Bagchi, V. Ultrathin MoS2 wrapped N-doped carbon-coated cobalt nanospheres for OER applications. Sustain. Energy Fuels 2021, 5, 801–807. [Google Scholar] [CrossRef]
- Dou, S.; Tao, L.; Huo, J.; Wang, S.; Dai, L. Etched and doped Co9S8/graphene hybrid for oxygen electrocatalysis. Energy Environ. Sci. 2016, 9, 1320–1326. [Google Scholar] [CrossRef]
- Han, X.; Wu, X.; Zhong, C.; Deng, Y.; Zhao, N.; Hu, W. NiCo2S4 nanocrystals anchored on nitrogen-doped carbon nanotubes as a highly efficient bifunctional electrocatalyst for rechargeable zinc-air batteries. Nano Energy 2017, 31, 541–550. [Google Scholar] [CrossRef]
- Wang, S.; He, P.; Jia, L.; He, M.; Zhang, T.; Dong, F.; Liu, M.; Liu, H.; Zhang, Y.; Li, C.; et al. Nanocoral-like composite of nickel selenide nanoparticles anchored on two-dimensional multi-layered graphitic carbon nitride: A highly efficient electrocatalyst for oxygen evolution reaction. Appl. Catal. B Environ. 2019, 243, 463–469. [Google Scholar] [CrossRef]
- Liu, G.; Zhang, Z.; Li, J.; Liu, J.; Long, X.; Chen, H.; Tong, H.; Liu, Z.; Qian, D. Constructing heterostructured CoSe2/Ni3Se4@N-doped carbon nanosheets/ketjen black carbon as a robust oxygen evolution electrocatalyst. Int. J. Hydrogen Energy 2020, 45, 30666–30672. [Google Scholar] [CrossRef]
- Ding, J.; Wang, P.; Ji, S.; Wang, H.; Brett, D.J.L.; Wang, R. Mesoporous nickel selenide N-doped carbon as a robust electrocatalyst for overall water splitting. Electrochim. Acta 2019, 300, 93–101. [Google Scholar] [CrossRef]
- Sun, J.; Li, G.; Zheng, K.; He, Y.; Guo, Z.; Xu, C. Surface Co3+-rich engineering of Co(SxSe1–x)2 nanocrystals coated with ultrathin carbon layer for efficient OER/HER. Appl. Surf. Sci. 2020, 517, 146183. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, S.; Wang, H.; Zhong, Y.; Hu, Y. An efficient and stable Ni–Fe selenides/nitrogen-doped carbon nanotubes in situ-derived electrocatalyst for oxygen evolution reaction. J. Mater. Sci. 2020, 55, 13927–13937. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, C.; Zhang, J.; Dastafkan, K.; Wang, K.; Zhao, C.; Shi, Z. Metal-Organic Framework-Derived Bimetallic NiFe Selenide Electrocatalysts with Multiple Phases for Efficient Oxygen Evolution Reaction. ACS Sustain. Chem. Eng. 2021, 9, 2047–2056. [Google Scholar] [CrossRef]
- Zou, J.; Luo, Q.; Wu, H.; Liu, S.; Lan, T.; Yao, Y.; Sial, M.A.Z.G.; Zhao, F.; Zhang, Q.; Zeng, X. CoSe2/Co nanoheteroparticles embedded in Co, N co-doped carbon nanopolyhedra/nanotubes as an efficient oxygen bifunctional electrocatalyst for Zn-air batteries. Sustain. Energy Fuels 2020, 4, 4722–4732. [Google Scholar] [CrossRef]
- Yuan, M.; Wang, M.; Lu, P.; Sun, Y.; Dipazir, S.; Zhang, J.; Li, S.; Zhang, G. Tuning carbon nanotube-grafted core-shell-structured cobalt selenide@carbon hybrids for efficient oxygen evolution reaction. J. Colloid Interface Sci. 2019, 533, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Dai, P.; Liu, H.; Yan, L.; Song, H.; Liu, D.; Zhao, X. Metal-organic framework derived porous flakes of cobalt chalcogenides (CoX, X = O, S, Se and Te) rooted in carbon fibers as flexible electrode materials for pseudocapacitive energy storage. Electrochim. Acta 2021, 369, 137681. [Google Scholar] [CrossRef]
- Li, W.; Niu, Y.; Wu, X.; Wu, F.; Li, T.; Hu, W. Heterostructured CoSe2/FeSe2 Nanoparticles with Abundant Vacancies and Strong Electronic Coupling Supported on Carbon Nanorods for Oxygen Evolution Electrocatalysis. ACS Sustain. Chem. Eng. 2020, 8, 4658–4666. [Google Scholar] [CrossRef]
- Ashok, A.; Kumar, A.; Ponraj, J.; Mansour, S.A. Development of Co/Co9S8 metallic nanowire anchored on N-doped CNTs through the pyrolysis of melamine for overall water splitting. Electrochim. Acta 2021, 368, 137642. [Google Scholar] [CrossRef]
- Sivanantham, A.; Hyun, S.; Son, M.; Shanmugam, S. Nanostructured core-shell cobalt chalcogenides for efficient water oxidation in alkaline electrolyte. Electrochim. Acta 2019, 312, 234–241. [Google Scholar] [CrossRef]
- Han, X.; Tong, X.; Wu, G.; Yang, N.; Guo, X.Y. Carbon fibers supported NiSe nanowire arrays as efficient and flexible electrocatalysts for the oxygen evolution reaction. Carbon 2018, 129, 245–251. [Google Scholar] [CrossRef]
- Chi, J.Q.; Yan, K.L.; Xiao, Z.; Dong, B.; Shang, X.; Gao, W.K.; Li, X.; Chai, Y.M.; Liu, C.G. Trimetallic Ni–Fe–Co selenides nanoparticles supported on carbon fiber cloth as efficient electrocatalyst for oxygen evolution reaction. Int. J. Hydrogen Energy 2017, 42, 20599–20607. [Google Scholar] [CrossRef]
- Wang, J.; Zhong, H.X.; Wang, Z.L.; Meng, F.L.; Zhang, X.B. Integrated Three-Dimensional Carbon Paper/Carbon Tubes/Cobalt-Sulfide Sheets as an Efficient Electrode for Overall Water Splitting. ACS Nano 2016, 10, 2342–2348. [Google Scholar] [CrossRef] [PubMed]
- Callejas, J.F.; Read, C.G.; Roske, C.W.; Lewis, N.S.; Schaak, R.E. Synthesis, Characterization, and Properties of Metal Phosphide Catalysts for the Hydrogen-Evolution Reaction. Chem. Mater. 2016, 28, 6017–6044. [Google Scholar] [CrossRef]
- Idrees, M.; Mukhtar, A.; Ata-ur-Rehman; Abbas, S.M.; Zhang, Q.; Li, X. Transition metal nitride electrodes as future energy storage devices: A review. Mater. Today Commun. 2021, 27, 102363. [Google Scholar] [CrossRef]
- Zheng, J.; Zhang, W.; Zhang, J.; Lv, M.; Li, S.; Song, H.; Cui, Z.; Du, L.; Liao, S. Recent advances in nanostructured transition metal nitrides for fuel cells. J. Mater. Chem. A 2020, 8, 20803–20818. [Google Scholar] [CrossRef]
- Aziz, S.T.; Malik, B.; Sadhanala, H.K.; Gedanken, A.; Noked, M.; Nessim, G.D. Nickel-Rich Phosphide (Ni12P5) Nanosheets Coupled with Oxidized Multiwalled Carbon Nanotubes for Oxygen Evolution. ACS Appl. Nano Mater. 2020, 3, 10914–10921. [Google Scholar] [CrossRef]
- Kang, Q.; Li, M.; Shi, J.; Lu, Q.; Gao, F. A Universal Strategy for Carbon-Supported Transition Metal Phosphides as High-Performance Bifunctional Electrocatalysts towards Efficient Overall Water Splitting. ACS Appl. Mater. Interfaces 2020, 12, 19447–19456. [Google Scholar] [CrossRef]
- Wu, J.; Li, P.; Parra-Puerto, A.; Wu, S.; Lin, X.; Kramer, D.; Chen, S.; Kucernak, A. Controllable Heteroatom Doping Effects of CrxCo2-xP Nanoparticles: A Robust Electrocatalyst for Overall Water Splitting in Alkaline Solutions. ACS Appl. Mater. Interfaces 2020, 12, 47397–47407. [Google Scholar] [CrossRef]
- Li, H.; Kong, X.; Geng, X.; Gu, C.; Liu, Z.; Wang, J. DNA as template and P-source for synthesis of Co2P/Co2N core–shell nanostructure embedded in N-doped carbon nanofiber derived from electrospun precursor for oxygen evolution reaction. Electrochim. Acta 2021, 367, 137562. [Google Scholar] [CrossRef]
- Liu, T.; Li, M.; Su, Z.; Bo, X.; Guan, W.; Zhou, M. Monodisperse and Tiny Co2N0.67 Nanocrystals Uniformly Embedded over Two Curving Surfaces of Hollow Carbon Microfibers as Efficient Electrocatalyst for Oxygen Evolution Reaction. ACS Appl. Nano Mater. 2018, 1, 4461–4473. [Google Scholar] [CrossRef]
- He, G.; Han, X.; Moss, B.; Weng, Z.; Gadipelli, S.; Lai, F.; Kafizas, A.G.; Brett, D.J.L.; Guo, Z.X.; Wang, H.; et al. Solid solution nitride/carbon nanotube hybrids enhance electrocatalysis of oxygen in zinc-air batteries. Energy Storage Mater. 2018, 15, 380–387. [Google Scholar] [CrossRef]
- Chen, X.; Gao, P.; Liu, H.; Xu, J.; Zhang, B.; Zhang, Y.; Tang, Y.; Xiao, C. In situ growth of iron-nickel nitrides on carbon nanotubes with enhanced stability and activity for oxygen evolution reaction. Electrochim. Acta 2018, 267, 8–14. [Google Scholar] [CrossRef]
- Shu, X.; Chen, S.; Chen, S.; Pan, W.; Zhang, J. Cobalt nitride embedded holey N-doped graphene as advanced bifunctional electrocatalysts for Zn-Air batteries and overall water splitting. Carbon 2020, 157, 234–243. [Google Scholar] [CrossRef]
- Hei, J.; Xu, G.; Wei, B.; Zhang, L.; Ding, H.; Liu, D. NiFeP nanosheets on N-doped carbon sponge as a hierarchically structured bifunctional electrocatalyst for efficient overall water splitting. Appl. Surf. Sci. 2021, 549, 149297. [Google Scholar] [CrossRef]
- Huang, J.; Li, F.; Liu, B.; Zhang, P. Ni2P/rGO/NF nanosheets as a bifunctional high-performance electrocatalyst for water splitting. Materials 2020, 13, 744. [Google Scholar] [CrossRef] [Green Version]
- Bian, J.; Song, Z.; Li, X.; Zhang, Y.; Cheng, C. Nickel iron phosphide ultrathin nanosheets anchored on nitrogen-doped carbon nanoflake arrays as a bifunctional catalyst for efficient overall water splitting. Nanoscale 2020, 12, 8443–8452. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Duan, X.; Han, W.; Fan, X.; Li, Y.; Zhang, F.; Zhang, G.; Peng, W. Decorated nickel phosphide nanoparticles with nitrogen and phosphorus co-doped porous carbon for enhanced electrochemical water splitting. J. Colloid Interface Sci. 2020, 567, 393–401. [Google Scholar] [CrossRef]
- Li, X.; Qian, X.; Xu, Y.; Duan, F.; Yu, Q.; Wang, J.; Chen, L.; Dan, Y.; Cheng, X. Electrodeposited cobalt phosphides with hierarchical nanostructure on biomass carbon for bifunctional water splitting in alkaline solution. J. Alloys Compd. 2020, 829, 154535. [Google Scholar] [CrossRef]
- Zhu, J.; Liu, C.; Sun, J.; Xing, Y.; Quan, B.; Li, D.; Jiang, D. Interfacial engineering of Co3FeNx embedded N-doped carbon nanoarray derived from metal–organic frameworks for enhanced oxygen evolution reaction. Electrochim. Acta 2020, 354, 136629. [Google Scholar] [CrossRef]
- Guan, C.; Sumboja, A.; Zang, W.; Qian, Y.; Zhang, H.; Liu, X.; Liu, Z.; Zhao, D.; Pennycook, S.J.; Wang, J. Decorating Co/CoNx nanoparticles in nitrogen-doped carbon nanoarrays for flexible and rechargeable zinc-air batteries. Energy Storage Mater. 2019, 16, 243–250. [Google Scholar] [CrossRef]
- Hu, L.; Hu, Y.; Liu, R.; Mao, Y.; Balogun, M.S.; Jie, T.; Tong, Y. Co-based MOF-derived Co/CoN/Co2P ternary composite embedded in N- and P-doped carbon as bifunctional nanocatalysts for efficient overall water splitting. Int. J. Hydrogen Energy 2019, 44, 11402–11410. [Google Scholar] [CrossRef]
- Kang, Y.; Wang, S.; Zhu, S.; Gao, H.; Hui, K.S.; Yuan, C.Z.; Yin, H.; Bin, F.; Wu, X.L.; Mai, W.; et al. Iron-modulated nickel cobalt phosphide embedded in carbon to boost power density of hybrid sodium–air battery. Appl. Catal. B Environ. 2021, 285, 119786. [Google Scholar] [CrossRef]
- Huo, D.; Song, F.; Hu, J.; Yuan, J.; Niu, L.; Wang, A.-J. One-step synthesis of carbon-encapsulated nickel phosphide nanoparticles with efficient bifunctional catalysis on oxygen evolution and reduction. Int. J. Hydrogen Energy 2021, 46, 8519–8530. [Google Scholar] [CrossRef]
- Choi, H.W.; Jeong, D.I.; Woo, S.; Kwon, S.B.; Wu, S.; Kim, J.H.; Yang, W.S.; Kang, B.K.; Lim, B.; Yoon, D.H. Morphology adjustable CoxN with 3D mesoporous structure and amorphous N-doped carbon for overall water splitting. Appl. Surf. Sci. 2020, 529, 147177. [Google Scholar] [CrossRef]
- Li, X.; Xu, Y.; Li, Y.; Fan, X.; Zhang, G.; Zhang, F.; Peng, W. Increasing the heteroatoms doping percentages of graphene by porous engineering for enhanced electrocatalytic activities. J. Colloid Interface Sci. 2020, 577, 101–108. [Google Scholar] [CrossRef]
- Guo, X.; Duan, X.; Ji, J.; Fan, X.; Li, Y.; Zhang, F.; Zhang, G.; Zhu, Y.A.; Peng, W.; Wang, S. Synthesis of nitrogen and sulfur doped graphene on graphite foam for electro-catalytic phenol degradation and water splitting. J. Colloid Interface Sci. 2021, 583, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.; Gong, M.; Zhou, Y.; Fang, J.; Huang, L.; Deng, Z.; Liu, X.; Zhao, T.; Lin, R.; Wang, K.; et al. Sulphur modulated Ni3FeN supported on N/S co-doped graphene boosts rechargeable/flexible Zn-air battery performance. Appl. Catal. B Environ. 2020, 274, 119086. [Google Scholar] [CrossRef]
- Yang, X.; Xie, H.; Zou, Y.; Huang, Y.; Jiang, L.W.; Wang, X.; Tang, D.H.; Si, C.; Wang, J.J. Cost-effective and efficient plum-pudding-like FexNi1-xS2/C composite electrocatalysts for oxygen evolution reaction. Renew. Energy 2021, 168, 416–423. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, M.; Luo, X.; Li, S.; Li, S.; Zhou, Q.; Xu, W.; Wu, R. N-, P-, and O-doped porous carbon: A trifunctional metal-free electrocatalyst. Appl. Surf. Sci. 2021, 544, 148912. [Google Scholar] [CrossRef]
- Cheng, Y.; Xu, C.; Jia, L.; Gale, J.D.; Zhang, L.; Liu, C.; Shen, P.K.; Jiang, S.P. Pristine carbon nanotubes as non-metal electrocatalysts for oxygen evolution reaction of water splitting. Appl. Catal. B Environ. 2015, 163, 96–104. [Google Scholar] [CrossRef]
- Yang, H.B.; Miao, J.; Hung, S.F.; Chen, J.; Tao, H.B.; Wang, X.; Zhang, L.; Chen, R.; Gao, J.; Chen, H.M.; et al. Identification of catalytic sites for oxygen reduction and oxygen evolution in N-doped graphene materials: Development of highly efficient metal-free bifunctional electrocatalyst. Sci. Adv. 2016, 2, e1501122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, C.; Bhoyate, S.; Hyatt, M.; Neria, B.L.; Siam, K.; Kahol, P.K.; Ghimire, M.; Mishra, S.R.; Perez, F.; Gupta, R.K. Nitrogen-doped flexible carbon cloth for durable metal free electrocatalyst for overall water splitting. Surf. Coat. Technol. 2018, 347, 407–413. [Google Scholar] [CrossRef]
- Davodi, F.; Tavakkoli, M.; Lahtinen, J.; Kallio, T. Straightforward synthesis of nitrogen-doped carbon nanotubes as highly active bifunctional electrocatalysts for full water splitting. J. Catal. 2017, 353, 19–27. [Google Scholar] [CrossRef]
- Paul, R.; Wang, M.; Roy, A. Transparent graphene/BN-graphene stacked nanofilms for electrocatalytic oxygen evolution. ACS Appl. Nano Mater. 2020, 3, 10418–10426. [Google Scholar] [CrossRef]
- Cheng, N.; Liu, Q.; Tian, J.; Xue, Y.; Asiri, A.M.; Jiang, H.; He, Y.; Sun, X. Acidically oxidized carbon cloth: A novel metal-free oxygen evolution electrode with high catalytic activity. Chem. Commun. 2015, 51, 1616–1619. [Google Scholar] [CrossRef]
- Ali, A.; Akyüz, D.; Asghar, M.A.; Koca, A.; Keskin, B. Free-standing carbon nanotubes as non-metal electrocatalyst for oxygen evolution reaction in water splitting. Int. J. Hydrogen Energy 2018, 43, 1123–1128. [Google Scholar] [CrossRef]
- Jiang, H.; Gu, J.; Zheng, X.; Liu, M.; Qiu, X.; Wang, L.; Li, W.; Chen, Z.; Ji, X.; Li, J. Defect-rich and ultrathin N doped carbon nanosheets as advanced trifunctional metal-free electrocatalysts for the ORR, OER and HER. Energy Environ. Sci. 2019, 12, 322–333. [Google Scholar] [CrossRef]
- Lu, X.; Yim, W.L.; Suryanto, B.H.R.; Zhao, C. Electrocatalytic oxygen evolution at surface-oxidized multiwall carbon nanotubes. J. Am. Chem. Soc. 2015, 137, 2901–2907. [Google Scholar] [CrossRef]
- Li, R.; Wei, Z.; Gou, X. Nitrogen and Phosphorus Dual-Doped Graphene/Carbon Nanosheets as Bifunctional Electrocatalysts for Oxygen Reduction and Evolution. ACS Catal. 2015, 5, 4133–4142. [Google Scholar] [CrossRef]
- Zhao, F.; Bae, J.; Zhou, X.; Guo, Y.; Yu, G. Nanostructured Functional Hydrogels as an Emerging Platform for Advanced Energy Technologies. Adv. Mater. 2018, 30, 1801796. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, H.; Wang, H.; Li, D.; Xuan, J.; Zhang, L. Hybrid Manufacturing of 3D Hierarchical Porous Carbons for Electrochemical Storage. Adv. Mater. Technol. 2020, 5, 1901030. [Google Scholar] [CrossRef]
- Zhu, Y.P.; Jing, Y.; Vasileff, A.; Heine, T.; Qiao, S.Z. 3D Synergistically Active Carbon Nanofibers for Improved Oxygen Evolution. Adv. Energy Mater. 2017, 7, 1602928. [Google Scholar] [CrossRef]
- Desalegn, B.Z.; Jadhav, H.S.; Seo, J.G. Highly Efficient g-C3N4 Nanorods with Dual Active Sites as an Electrocatalyst for the Oxygen Evolution Reaction. ChemCatChem 2019, 11, 2870–2878. [Google Scholar] [CrossRef]
- Peng, Z.; Yang, S.; Jia, D.; Da, P.; He, P.; Al-Enizi, A.M.; Ding, G.; Xie, X.; Zheng, G. Homologous metal-free electrocatalysts grown on three-dimensional carbon networks for overall water splitting in acidic and alkaline media. J. Mater. Chem. A 2016, 4, 12878–12883. [Google Scholar] [CrossRef]
- Ma, T.Y.; Ran, J.; Dai, S.; Jaroniec, M.; Qiao, S.Z. Phosphorus-Doped Graphitic Carbon Nitrides Grown In Situ on Carbon-Fiber Paper: Flexible and Reversible Oxygen Electrodes. Angew. Chem. Int. Ed. 2015, 54, 4646–4650. [Google Scholar] [CrossRef] [Green Version]
- Luque-Centeno, J.M.; Martínez-Huerta, M.V.; Sebastián, D.; Lemes, G.; Pastor, E.; Lázaro, M.J. Bifunctional N-doped graphene Ti and Co nanocomposites for the oxygen reduction and evolution reactions. Renew. Energy 2018, 125, 182–192. [Google Scholar] [CrossRef] [Green Version]
- Ruiz-Cornejo, J.C.; Vivo-Vilches, J.F.; Sebastián, D.; Martínez-Huerta, M.V.; Lázaro, M.J. Carbon nanofiber-supported tantalum oxides as durable catalyst for the oxygen evolution reaction in alkaline media. Renew. Energy 2021, 178, 307–317. [Google Scholar] [CrossRef]
- Alegre, C.; Busacca, C.; Di Blasi, O.; Antonucci, V.; Aricò, A.S.; Di Blasi, A.; Baglio, V. A combination of CoO and Co nanoparticles supported on electrospun carbon nanofibers as highly stable air electrodes. J. Power Sources 2017, 364, 101–109. [Google Scholar] [CrossRef]
- Alegre, C.; Modica, E.; Di Blasi, A.; Di Blasi, O.; Busacca, C.; Ferraro, M.; Aricò, A.S.; Antonucci, V.; Baglio, V. NiCo-loaded carbon nanofibers obtained by electrospinning: Bifunctional behavior as air electrodes. Renew. Energy 2018, 125, 250–259. [Google Scholar] [CrossRef]
- Alegre, C.; Busacca, C.; Di Blasi, A.; Di Blasi, O.; Aricò, A.S.; Antonucci, V.; Modica, E.; Baglio, V. Electrospun carbon nanofibers loaded with spinel-type cobalt oxide as bifunctional catalysts for enhanced oxygen electrocatalysis. J. Energy Storage 2019, 23, 269–277. [Google Scholar] [CrossRef]
- Alegre, C.; Busacca, C.; Di Blasi, A.; Di Blasi, O.; Aricò, A.S.; Antonucci, V.; Baglio, V. Electrocatalysis of Oxygen on Bifunctional Nickel-Cobaltite Spinel. ChemElectroChem 2020, 7, 124–130. [Google Scholar] [CrossRef]
- Alegre, C.; Busacca, C.; Di Blasi, A.; Di Blasi, O.; Aricò, A.S.; Antonucci, V.; Baglio, V. Toward more efficient and stable bifunctional electrocatalysts for oxygen electrodes using FeCo2O4/carbon nanofiber prepared by electrospinning. Mater. Today Energy 2020, 18, 100508. [Google Scholar] [CrossRef]
- Moon, G.D.; Ko, S.; Xia, Y.; Jeong, U. Chemical transformations in ultrathin chalcogenide nanowires. ACS Nano 2010, 4, 2307–2319. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wu, R.; Liu, M.; Liu, Y.; Xu, S.; Ha, Y.; Guo, Y.; Yu, X.; Sun, D.; Fang, F. Tunable electronic coupling of cobalt sulfide/carbon composites for optimizing oxygen evolution reaction activity. J. Mater. Chem. A 2018, 6, 10304–10312. [Google Scholar] [CrossRef]
- Ding, H.; Xu, G.; Zhang, L.; Wei, B.; Hei, J.; Chen, L. A highly effective bifunctional catalyst of cobalt selenide nanoparticles embedded nitrogen-doped bamboo-like carbon nanotubes toward hydrogen and oxygen evolution reactions based on metal-organic framework. J. Colloid Interface Sci. 2020, 566, 296–303. [Google Scholar] [CrossRef] [PubMed]
- He, B.; Wang, X.C.; Xia, L.X.; Guo, Y.Q.; Tang, Y.W.; Zhao, Y.; Hao, Q.L.; Yu, T.; Liu, H.K.; Su, Z. Metal-Organic Framework-Derived Fe-Doped Co1.11Te2 Embedded in Nitrogen-Doped Carbon Nanotube for Water Splitting. ChemSusChem 2020, 13, 5239–5247. [Google Scholar] [CrossRef]
- Oyetade, O.A.; Kriek, R.J. NiSe-Ni3Se2/Multiwalled Carbon Nanotube Composites as Efficient Electrocatalysts for the Oxygen Evolution Reaction in Alkaline Media. Electrocatalysis 2020, 11, 35–45. [Google Scholar] [CrossRef]
- Wang, X.; Huang, X.; Gao, W.; Tang, Y.; Jiang, P.; Lan, K.; Yang, R.; Wang, B.; Li, R. Metal-organic framework derived CoTe2 encapsulated in nitrogen-doped carbon nanotube frameworks: A high-efficiency bifunctional electrocatalyst for overall water splitting. J. Mater. Chem. A 2018, 6, 3684–3691. [Google Scholar] [CrossRef]
- Yang, L.; Gao, M.; Dai, B.; Guo, X.; Liu, Z.; Peng, B. An efficient NiS@N/S-C hybrid oxygen evolution electrocatalyst derived from metal-organic framework. Electrochim. Acta 2016, 191, 813–820. [Google Scholar] [CrossRef]
- Wu, L.L.; Wang, Q.S.; Li, J.; Long, Y.; Liu, Y.; Song, S.Y.; Zhang, H.J. Co9S8 Nanoparticles-Embedded N/S-Codoped Carbon Nanofibers Derived from Metal–Organic Framework-Wrapped CdS Nanowires for Efficient Oxygen Evolution Reaction. Small 2018, 14, 1704035. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Li, R.; Ma, G.; Gou, X.; Zhu, Y.; Xia, Y. Cobalt sulfide/N,S codoped porous carbon core-shell nanocomposites as superior bifunctional electrocatalysts for oxygen reduction and evolution reactions. Nanoscale 2015, 7, 20674–20684. [Google Scholar] [CrossRef] [Green Version]
- Cao, X.; Zheng, X.; Tian, J.; Jin, C.; Ke, K.; Yang, R. Cobalt Sulfide Embedded in Porous Nitrogen-doped Carbon as a Bifunctional Electrocatalyst for Oxygen Reduction and Evolution Reactions. Electrochim. Acta 2016, 191, 776–783. [Google Scholar] [CrossRef]
- Jayaramulu, K.; Masa, J.; Tomanec, O.; Peeters, D.; Ranc, V.; Schneemann, A.; Zboril, R.; Schuhmann, W.; Fischer, R.A. Nanoporous Nitrogen-Doped Graphene Oxide/Nickel Sulfide Composite Sheets Derived from a Metal-Organic Framework as an Efficient Electrocatalyst for Hydrogen and Oxygen Evolution. Adv. Funct. Mater. 2017, 27, 1700451. [Google Scholar] [CrossRef]
- Le, T.T.; Liu, X.; Xin, P.; Wang, Q.; Gao, C.; Wu, Y.; Jiang, Y.; Hu, Z.; Huang, S.; Chen, Z. Phosphorus-doped Fe7S8@C nanowires for efficient electrochemical hydrogen and oxygen evolutions: Controlled synthesis and electronic modulation on active sites. J. Mater. Sci. Technol. 2021, 74, 168–175. [Google Scholar] [CrossRef]
- Ganesan, P.; Staykov, A.; Shu, H.; Uejima, M.; Nakashima, N. Designing an FeIII-doped nickel sulfide/carbon nanotube hybrid catalyst for alkaline electrolyte membrane water electrolyzers and Zn−air battery performances. ACS Appl. Energy Mater. 2020, 3, 10961–10975. [Google Scholar] [CrossRef]
- Shen, M.; Ruan, C.; Chen, Y.; Jiang, C.; Ai, K.; Lu, L. Covalent entrapment of cobalt-iron sulfides in N-doped mesoporous carbon: Extraordinary bifunctional electrocatalysts for oxygen reduction and evolution reactions. ACS Appl. Mater. Interfaces 2015, 7, 1207–1218. [Google Scholar] [CrossRef]
- Jiang, J.; Lu, S.; Wang, W.K.; Huang, G.X.; Huang, B.C.; Zhang, F.; Zhang, Y.J.; Yu, H.Q. Ultrahigh electrocatalytic oxygen evolution by iron-nickel sulfide nanosheets/reduced graphene oxide nanohybrids with an optimized autoxidation process. Nano Energy 2018, 43, 300–309. [Google Scholar] [CrossRef]
- Cao, J.; Wang, K.; Chen, J.; Lei, C.; Yang, B.; Li, Z.; Lei, L.; Hou, Y.; Ostrikov, K. Nitrogen-Doped Carbon-Encased Bimetallic Selenide for High-Performance Water Electrolysis. Nano-Micro Lett. 2019, 11, 67. [Google Scholar] [CrossRef] [Green Version]
- Hosseini, H.; Roushani, M. Rational design of hollow core-double shells hybrid nanoboxes and nanopipes composed of hierarchical Cu-Ni-Co selenides anchored on nitrogen-doped carbon skeletons as efficient and stable bifunctional electrocatalysts for overall water splitting. Chem. Eng. J. 2020, 402, 126174. [Google Scholar] [CrossRef]
- Liu, M.; Lu, X.; Guo, C.; Wang, Z.; Li, Y.; Lin, Y.; Zhou, Y.; Wang, S.; Zhang, J. Architecting a Mesoporous N-Doped Graphitic Carbon Framework Encapsulating CoTe2 as an Efficient Oxygen Evolution Electrocatalyst. ACS Appl. Mater. Interfaces 2017, 9, 36146–36153. [Google Scholar] [CrossRef]
- Li, G.; Yu, J.; Yu, W.; Yang, L.; Zhang, X.; Liu, X.; Liu, H.; Zhou, W. Phosphorus-Doped Iron Nitride Nanoparticles Encapsulated by Nitrogen-Doped Carbon Nanosheets on Iron Foam In Situ Derived from Saccharomycetes Cerevisiae for Electrocatalytic Overall Water Splitting. Small 2020, 16, 2001980. [Google Scholar] [CrossRef]
- Yang, H.; Guo, H.; Pang, K.; Fan, P.; Li, X.; Ren, W.; Song, R. An amorphous carbon nitride/NiO/CoN-based composite: A highly efficient nonprecious electrode for supercapacitors and the oxygen evolution reaction. Nanoscale 2020, 12, 7024–7034. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Jiang, Y.; Chen, X.; Wang, L.; Zhuang, G.; Li, X.; Wang, J.G. Integrating cobalt phosphide and cobalt nitride-embedded nitrogen-rich nanocarbons: High-performance bifunctional electrocatalysts for oxygen reduction and evolution. J. Mater. Chem. A 2016, 4, 10575–10584. [Google Scholar] [CrossRef]
- Liu, T.; Li, M.; Jiao, C.; Hassan, M.; Bo, X.; Zhou, M.; Wang, H.L. Design and synthesis of integrally structured Ni3N nanosheets/carbon microfibers/Ni3N nanosheets for efficient full water splitting catalysis. J. Mater. Chem. A 2017, 5, 9377–9390. [Google Scholar] [CrossRef]
- Huang, C.; Ouyang, T.; Zou, Y.; Li, N.; Liu, Z.Q. Ultrathin NiCo2P: X nanosheets strongly coupled with CNTs as efficient and robust electrocatalysts for overall water splitting. J. Mater. Chem. A 2018, 6, 7420–7427. [Google Scholar] [CrossRef]
- Yang, D.; Hou, W.; Lu, Y.; Zhang, W.; Chen, Y. Cobalt phosphide nanoparticles supported within network of N-doped carbon nanotubes as a multifunctional and scalable electrocatalyst for water splitting. J. Energy Chem. 2021, 52, 130–138. [Google Scholar] [CrossRef]
- Chen, D.; Zhu, J.; Mu, X.; Cheng, R.; Li, W.; Liu, S.; Pu, Z.; Lin, C.; Mu, S. Nitrogen-Doped carbon coupled FeNi3 intermetallic compound as advanced bifunctional electrocatalyst for OER, ORR and zn-air batteries. Appl. Catal. B Environ. 2020, 268, 118729. [Google Scholar] [CrossRef]
- Shi, J.; Qiu, F.; Yuan, W.; Guo, M.; Lu, Z.H. Nitrogen-doped carbon-decorated yolk-shell CoP@FeCoP micro-polyhedra derived from MOF for efficient overall water splitting. Chem. Eng. J. 2021, 403, 126312. [Google Scholar] [CrossRef]
- Wang, Z.; Wei, C.; Zhu, X.; Wang, X.; He, J.; Zhao, Y. A hierarchical carbon nanotube forest supported metal phosphide electrode for efficient overall water splitting. J. Mater. Chem. A 2021, 9, 1150–1158. [Google Scholar] [CrossRef]
- Wu, Y.; Xiao, Z.; Jin, Z.; Li, X.; Chen, Y. The cobalt carbide/bimetallic CoFe phosphide dispersed on carbon nanospheres as advanced bifunctional electrocatalysts for the ORR, OER, and rechargeable Zn–air batteries. J. Colloid Interface Sci. 2021, 590, 321–329. [Google Scholar] [CrossRef]
- Liu, H.; Guan, J.; Yang, S.; Yu, Y.; Shao, R.; Zhang, Z.; Dou, M.; Wang, F.; Xu, Q. Metal–Organic-Framework-Derived Co2P Nanoparticle/Multi-Doped Porous Carbon as a Trifunctional Electrocatalyst. Adv. Mater. 2020, 32, 2003649. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Zheng, Z.; Song, Z.; Zhang, X.; Han, X.; Chen, H.; Xie, Z.; Kuang, Q.; Zheng, L. In situ construction and post-electrolysis structural study of porous Ni2P@C nanosheet arrays for efficient water splitting. Inorg. Chem. Front. 2020, 7, 2960–2968. [Google Scholar] [CrossRef]
- Yang, D.; Su, Z.; Chen, Y.; Lu, Y.; Yu, B.; Srinivas, K.; Wang, B.; Zhang, W. Double-shelled hollow bimetallic phosphide nanospheres anchored on nitrogen-doped graphene for boosting water electrolysis. J. Mater. Chem. A 2020, 8, 22222–22229. [Google Scholar] [CrossRef]
- Tang, Y.J.; You, L.; Zhou, K.; Zhou, K. Enhanced Oxygen Evolution Reaction Activity of a Co2P@NC-Fe2P Composite Boosted by Interfaces between a N-Doped Carbon Matrix and Fe2P Microspheres. ACS Appl. Mater. Interfaces 2020, 12, 25884–25894. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Yu, X.Y.; Lou, X.W.D. Carbon-Incorporated Nickel–Cobalt Mixed Metal Phosphide Nanoboxes with Enhanced Electrocatalytic Activity for Oxygen Evolution. Angew. Chem. Int. Ed. 2017, 56, 3897–3900. [Google Scholar] [CrossRef] [PubMed]
- Pu, Z.; Liu, T.; Amiinu, I.S.; Cheng, R.; Wang, P.; Zhang, C.; Ji, P.; Hu, W.; Liu, J.; Mu, S. Transition-Metal Phosphides: Activity Origin, Energy-Related Electrocatalysis Applications, and Synthetic Strategies. Adv. Funct. Mater. 2020, 30, 2004009. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Q.; Feng, X. Support and Interface Effects in Water-Splitting Electrocatalysts. Adv. Mater. 2019, 31, 1808167. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.P.; Guo, C.; Zheng, Y.; Qiao, S.Z. Surface and Interface Engineering of Noble-Metal-Free Electrocatalysts for Efficient Energy Conversion Processes. Acc. Chem. Res. 2017, 50, 915–923. [Google Scholar] [CrossRef]
- Zhao, D.; Pi, Y.; Shao, Q.; Feng, Y.; Zhang, Y.; Huang, X. Enhancing Oxygen Evolution Electrocatalysis via the Intimate Hydroxide-Oxide Interface. ACS Nano 2018, 12, 6245–6251. [Google Scholar] [CrossRef]
- Xu, J.; Wei, X.K.; Costa, J.D.; Lado, J.L.; Owens-Baird, B.; Gonçalves, L.P.L.; Fernandes, S.P.S.; Heggen, M.; Petrovykh, D.Y.; Dunin-Borkowski, R.E.; et al. Interface Engineering in Nanostructured Nickel Phosphide Catalyst for Efficient and Stable Water Oxidation. ACS Catal. 2017, 7, 5450–5455. [Google Scholar] [CrossRef]
- Liu, P.F.; Li, X.; Yang, S.; Zu, M.Y.; Liu, P.; Zhang, B.; Zheng, L.R.; Zhao, H.; Yang, H.G. Ni2P(O)/Fe2P(O) Interface Can Boost Oxygen Evolution Electrocatalysis. ACS Energy Lett. 2017, 2, 2257–2263. [Google Scholar] [CrossRef]
- Niu, Z.; Qiu, C.; Jiang, J.; Ai, L. Hierarchical CoP-FeP Branched Heterostructures for Highly Efficient Electrocatalytic Water Splitting. ACS Sustain. Chem. Eng. 2019, 7, 2335–2342. [Google Scholar] [CrossRef]
- Ding, X.; Xia, Y.; Li, Q.; Dong, S.; Jiao, X.; Chen, D. Interface Engineering of Co(OH)2/Ag/FeP Hierarchical Superstructure as Efficient and Robust Electrocatalyst for Overall Water Splitting. ACS Appl. Mater. Interfaces 2019, 11, 7936–7945. [Google Scholar] [CrossRef]
- Li, M.; Pan, X.; Jiang, M.; Zhang, Y.; Tang, Y.; Fu, G. Interface engineering of oxygen-vacancy-rich CoP/CeO2 heterostructure boosts oxygen evolution reaction. Chem. Eng. J. 2020, 395, 125160. [Google Scholar] [CrossRef]
- Li, Y.; Liao, C.; Tang, K.; Zhang, N.; Qi, W.; Xie, H.; He, J.; Yin, K.; Gao, Y.; Wang, C. Cobalt hydroxide-black phosphorus nanosheets: A superior electrocatalyst for electrochemical oxygen evolution. Electrochim. Acta 2019, 297, 40–45. [Google Scholar] [CrossRef]
- Xu, Q.; Jiang, H.; Zhang, H.; Hu, Y.; Li, C. Heterogeneous interface engineered atomic configuration on ultrathin Ni(OH)2/Ni3S2 nanoforests for efficient water splitting. Appl. Catal. B Environ. 2019, 242, 60–66. [Google Scholar] [CrossRef]
- Li, J.; Xu, X.; Zhang, B.; Hou, W.; Lv, S.; Shi, Y. Controlled synthesis and fine-tuned interface of NiS nanoparticles/Bi2WO6 nanosheets heterogeneous as electrocatalyst for oxygen evolution reaction. Appl. Surf. Sci. 2020, 526, 146718. [Google Scholar] [CrossRef]
- Guan, X.; Sun, X.; Feng, H.; Zhang, J.; Wen, H.; Tian, W.; Zheng, D.; Yao, Y. Rational interface engineering of Cu2S-CoOx/CF enhances oxygen evolution reaction activity. Chem. Commun. 2020, 56, 13571–13574. [Google Scholar] [CrossRef]
- Xia, J.; Zhao, H.; Huang, B.; Xu, L.; Luo, M.; Wang, J.; Luo, F.; Du, Y.; Yan, C.H. Efficient Optimization of Electron/Oxygen Pathway by Constructing Ceria/Hydroxide Interface for Highly Active Oxygen Evolution Reaction. Adv. Funct. Mater. 2020, 30, 1908367. [Google Scholar] [CrossRef]
- Bao, W.; Xiao, L.; Zhang, J.; Deng, Z.; Yang, C.; Ai, T.; Wei, X. Interface engineering of NiV-LDH@FeOOH heterostructures as high-performance electrocatalysts for oxygen evolution reaction in alkaline conditions. Chem. Commun. 2020, 56, 9360–9363. [Google Scholar] [CrossRef] [PubMed]
- An, L.; Feng, J.; Zhang, Y.; Wang, R.; Liu, H.; Wang, G.C.; Cheng, F.; Xi, P. Epitaxial Heterogeneous Interfaces on N-NiMoO4/NiS2 Nanowires/Nanosheets to Boost Hydrogen and Oxygen Production for Overall Water Splitting. Adv. Funct. Mater. 2019, 29, 1805298. [Google Scholar] [CrossRef] [Green Version]


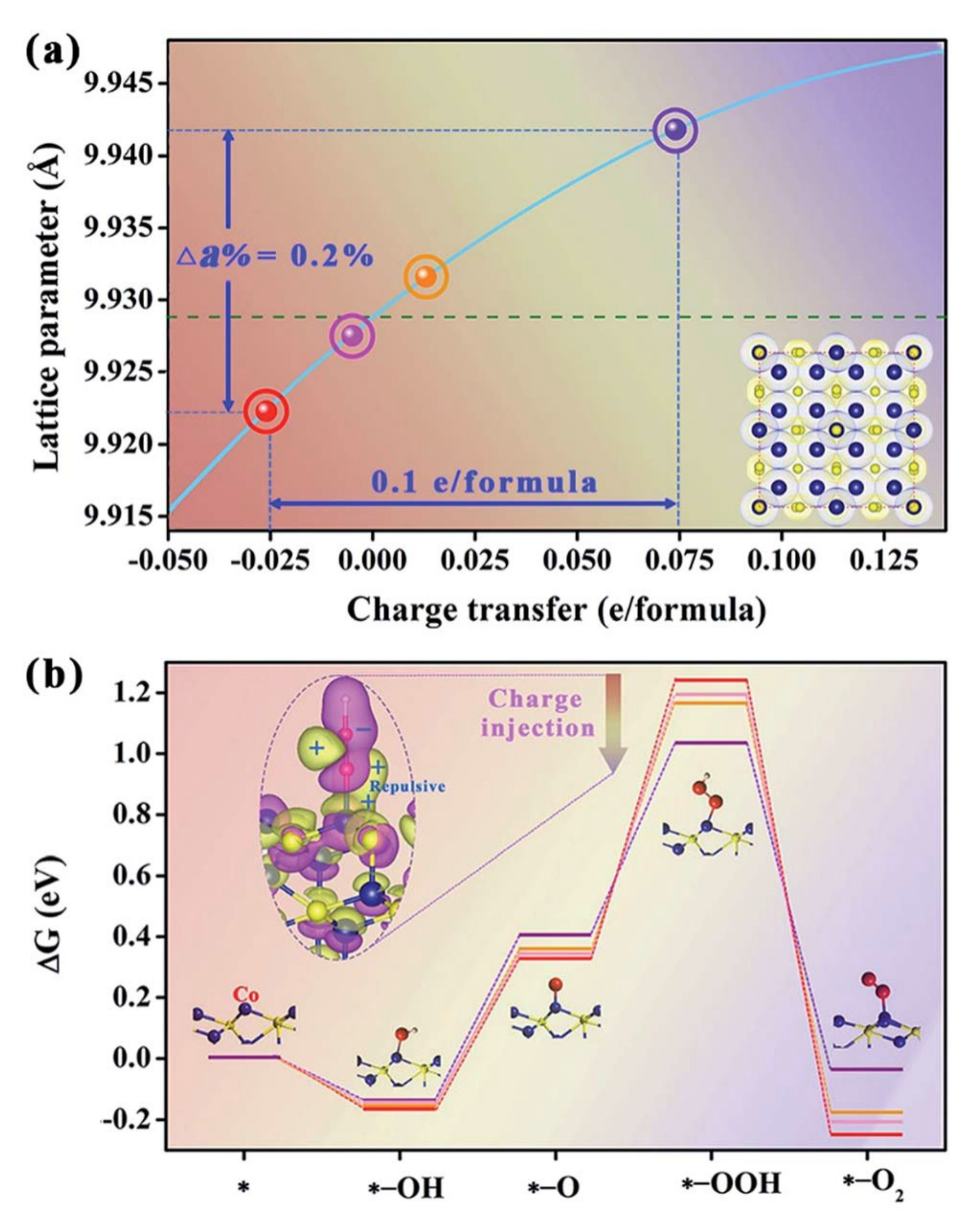
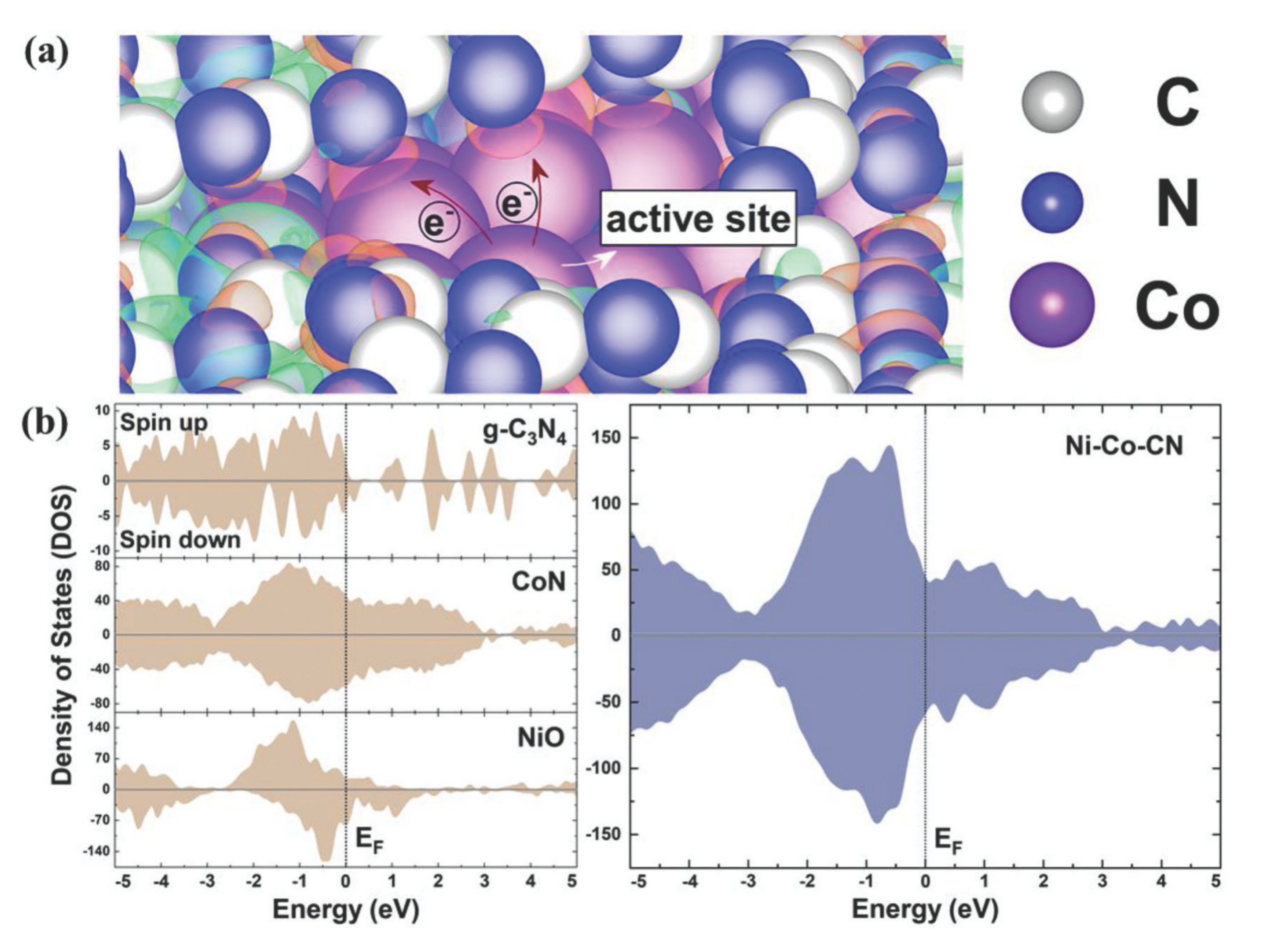
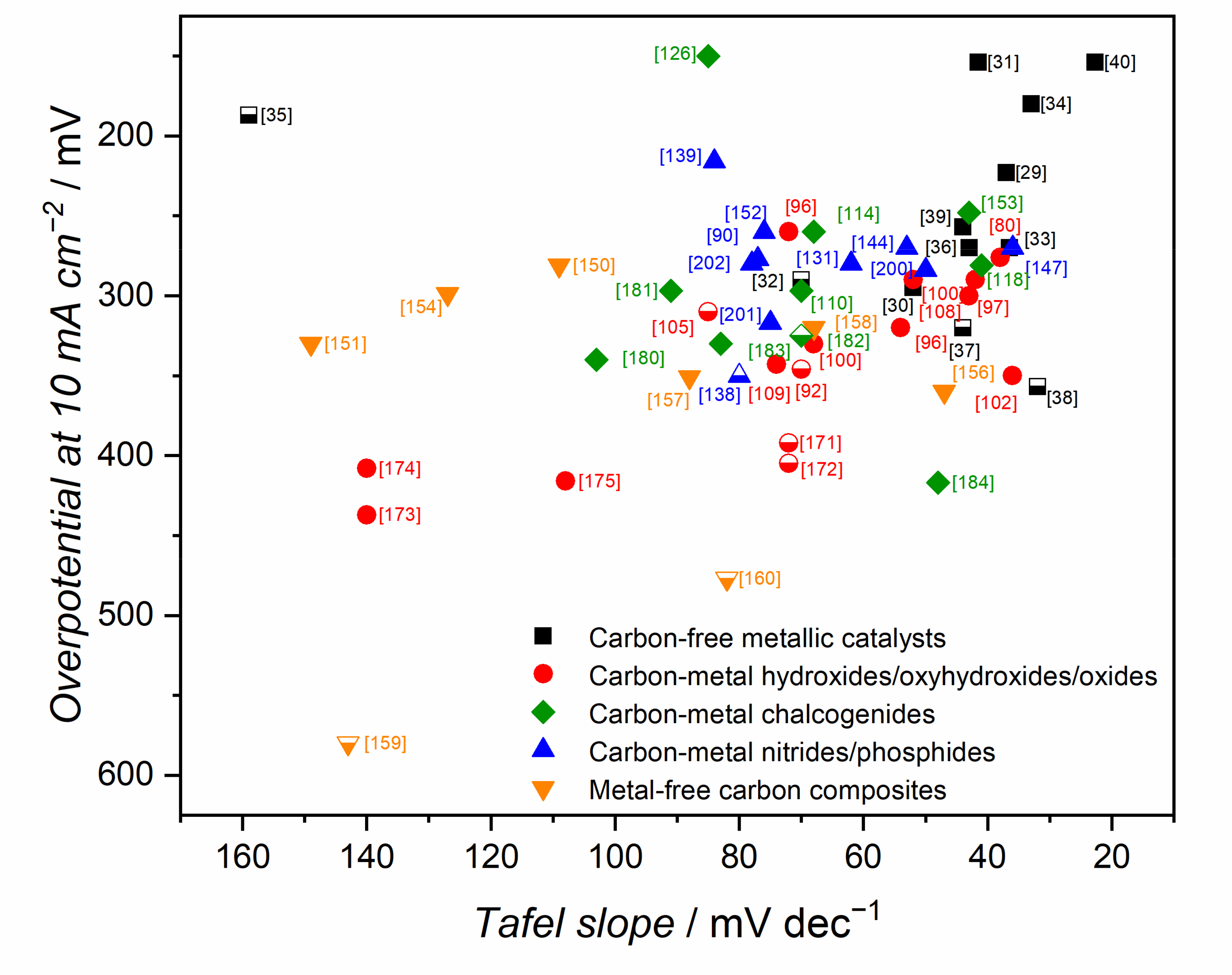
| Electrocatalyst | KOH Concentration | Substrate | Loading/mg cm−2 | Overpotential at 10 mA cm−2/mV | Tafel Slope/mV dec−1 | Ref. |
|---|---|---|---|---|---|---|
| G-FeCoW | 1 M | GCE * | 0.21 | 223 | 37 | [29] |
| α-Co4Fe(OH)x | 1 M | GCE | 0.28 | 295 | 52 | [30] |
| (Ni,Fe)OOH | 1 M | NF * | 4.0 | 154 | 41.5 | [31] |
| Co3O4C-NA | 0.1 M | Cu foil | 0.2 | 290 | 70 | [32] |
| CoO-MoO2 | 1 M | NF | N/A | 270 | 36.5 | [33] |
| Core–shell NiFeCu | 1 M | NF | 10.2 | 180 | 33 | [34] |
| Ni3S2 nanorods | 0.1 M | NF | 37 | 187 | 159 | [35] |
| Fe7S8 nanosheets | 1 M | GCE | 0.143 | 270 | 43 | [36] |
| CoSe2 nanosheet | 0.1 M | GCE | 0.142 | 320 | 44 | [37] |
| CoTe2 nanofleeces | 0.1 M | GCE | 0.25 | 357 | 32 | [38] |
| Co4N nanowire | 1 M | carbon cloth | 0.82 | 257 | 44 | [39] |
| FeP/Ni2P | 1 M | NF | 8 | 154 | 22.7 | [40] |
| Catalyst | Electrolyte | Overpotential at 10 mA cm−2/mV | Tafel Slope/mV dec−1 | Ref. |
|---|---|---|---|---|
| ANGS (activated S, N co-doped graphene) | 1 M KOH | 281 | 109 | [150] |
| SNG@GF (N and S-doped graphene on graphite foam) | 1 M KOH | 330 | 149 | [151] |
| GNP (N, P, and O-doped carbon) | 1 M KOH | 299 | 127 | [154] |
| N-GRW (N-doped graphene nanoribbons) | 1 M KOH | 360 | 47 | [156] |
| PAN-CCC (N-doped cotton cloth) | 1 M NaOH | 351 | 88 | [157] |
| NMWNT (N-doped multi-walled carbon nanotubes) | 1 M NaOH | 320 | 68 | [158] |
| G-BNG (stacked nanofilm of graphene on B,N-codoped graphene) | 0.1 M KOH | 580 | 143 | [159] |
| OCC (oxidized carbon cloth) | 0.1 M KOH | 477 | 82 | [160] |
| Catalyst | KOH Electrolyte Concentration | Overpotential at 10 mA cm−2/mV | Tafel Slope/mV dec−1 | Ref. |
|---|---|---|---|---|
| Ni-Fe Hydroxide/edge-rich vertical graphene | 1 M | 276 | 38 | [80] |
| Ni/NiO/N-doped activated carbon | 0.1 M | 346 | 70 | [92] |
| N-rGO/NiCo-NiO-CoO | 1 M | 260 | 72 | [96] |
| N-rGO/CoFe-CoFe2O4 | 1 M | 320 | 54 | [96] |
| Co0.5Fe0.5WO4/CNT | 1 M | 290 | 42 | [97] |
| Ni nanoplates/rGO | 1 M | 330 | 68 | [100] |
| Ni@Pt core–shell nanoplates/rGO | 1 M | 290 | 52 | [100] |
| Ni-NiFe2O4/N-CNT | 1 M | 340 | 51 * | [101] |
| Co(OH)x/N-CNT | 1 M | 350 | 36 | [102] |
| Hollow Co3O4/CeO2-heterostructure/N-doped carbon nanofibers | 0.1 M | 310 | 85 | [105] |
| Ni0.36Fe0.64/MnOx/N-doped graphitic carbon, Mn/Ni = 0.2 | 1 M | 300 | 43 | [108] |
| CoOx/carbon fiber paper | 1 M | 343 | 74 | [109] |
| CoOx/NrGO | 0.1 M | 392 | 72 | [171] |
| TaOx/CNF | 0.1 M | 405 | 72 | [172] |
| CoO-Co/CNF | 1 M | 437 | 140 | [173] |
| NiCo-loaded CNF | 1 M | 408 | 140 | [174] |
| Co3O4/CNF | 1 M | 416 | 108 | [175] |
| NiCo2O4/CNF | 6 M | 223 | 174 | [176] |
| FeCo2O4/CNF | 6 M | 130 | 100 | [177] |
| Catalyst | KOH Electrolyte Concentration | Overpotential at 10 mA cm−2/mV | Tafel Slope/mV dec−1 | Ref. |
|---|---|---|---|---|
| MoS2 wrapped N-doped carbon-coated Co nanospheres | 1 M | 297 | 70 | [110] |
| CoSe2/Ni3Se4@N-doped carbon nanosheets/ketjen black carbon | 1 M | 260 | 68 | [114] |
| NiFe-Se/carbon fiber paper | 1 M | 281 | 41 | [118] |
| NiFeCoSex/carbon fiber cloth | 1 M | 150 | 85 | [126] |
| FexNi1−xS2/C | 1 M | 248 | 43 | [153] |
| CoSe2@N-doped bamboo-like carbon nanotubes | 1 M | 340 | 103 | [180] |
| Fe-Co1.11Te2@N-doped carbon nanotube | 1 M | 297 | 91 | [181] |
| NiSe-Ni3Se2/MWCNT | 0.1 M | 325 | 70 | [182] |
| CoTe2 encapsulated in N-doped carbon nanotube frameworks | 1 M | 330 | 83 | [183] |
| NiS@N/S-C | 1 M | 417 | 48 | [184] |
| Catalyst | KOH Electrolyte Concentration | Overpotential at 10 mA cm−2/mV | Tafel Slope/mV dec−1 | Ref. |
|---|---|---|---|---|
| Ni3N/B-doped graphene oxide | 1 M | 280 | 78 | [90] |
| Ni12P5 nanosheets coupled with oxidized MWCNTs | 1 M | 280 | 62 | [131] |
| Co5.47N@N-doped rGO | 0.1 M | 350 | 80 | [138] |
| NiFeP@ N-doped carbon sponge | 1 M | 216 | 84 | [139] |
| Co3FeNx/N-doped carbon nanoleaf arrays@carbon cloth | 1 M | 270 | 53 | [144] |
| Fe–NiCoP embedded in the amorphous carbon layer | 1 M | 270 | 36 | [147] |
| S-Ni3FeN/N,S co-doped grephene | 1 M | 260 | 76 | [152] |
| NiCo2Px/CNTs | 1 M | 284 | 50 | [200] |
| CoP@N-doped carbon nanotube network | 1 M | 317 | 75 | [201] |
| FeNi3@N-doped carbon | 1 M | 277 | 77 | [202] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stelmachowski, P.; Duch, J.; Sebastián, D.; Lázaro, M.J.; Kotarba, A. Carbon-Based Composites as Electrocatalysts for Oxygen Evolution Reaction in Alkaline Media. Materials 2021, 14, 4984. https://doi.org/10.3390/ma14174984
Stelmachowski P, Duch J, Sebastián D, Lázaro MJ, Kotarba A. Carbon-Based Composites as Electrocatalysts for Oxygen Evolution Reaction in Alkaline Media. Materials. 2021; 14(17):4984. https://doi.org/10.3390/ma14174984
Chicago/Turabian StyleStelmachowski, Paweł, Joanna Duch, David Sebastián, María Jesús Lázaro, and Andrzej Kotarba. 2021. "Carbon-Based Composites as Electrocatalysts for Oxygen Evolution Reaction in Alkaline Media" Materials 14, no. 17: 4984. https://doi.org/10.3390/ma14174984








