Exogenous NAD+ Postpones the D-Gal-Induced Senescence of Bone Marrow-Derived Mesenchymal Stem Cells via Sirt1 Signaling
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Isolation and Culture
2.2. Cell Viability Assay
2.3. SA-β-Gal+ Activity Assay
2.4. Quantitative Real-Time Polymerase Chain Reaction
2.5. Western Blotting
2.6. Detection of ROS
2.7. Quantification of Intracellular NAD+ and NAD+/NADH Ratio
2.8. Small Interfering RNA Transfection
2.9. Statistical Analysis
3. Results
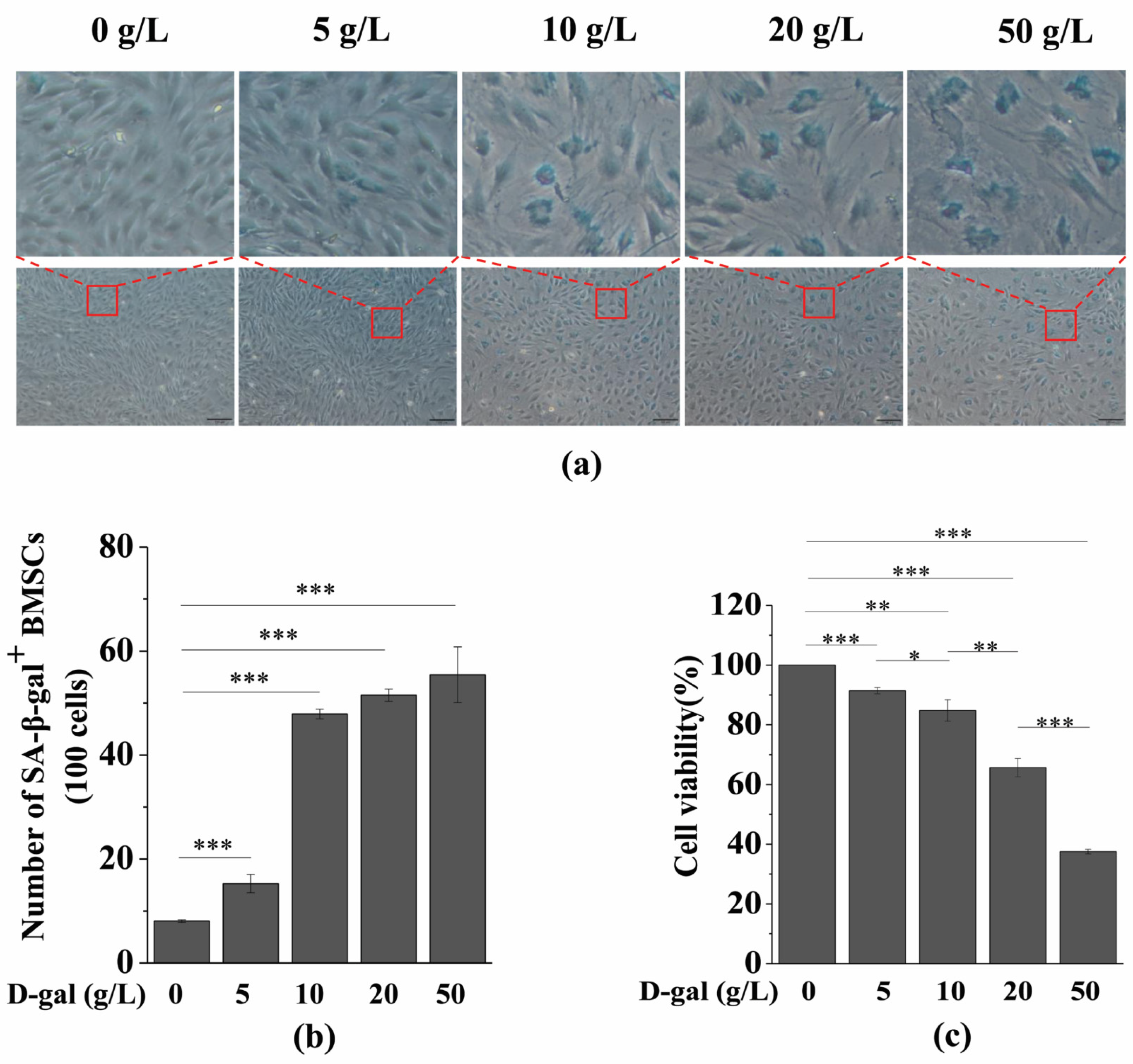
3.1. D-gal Induces BMSC Senescence
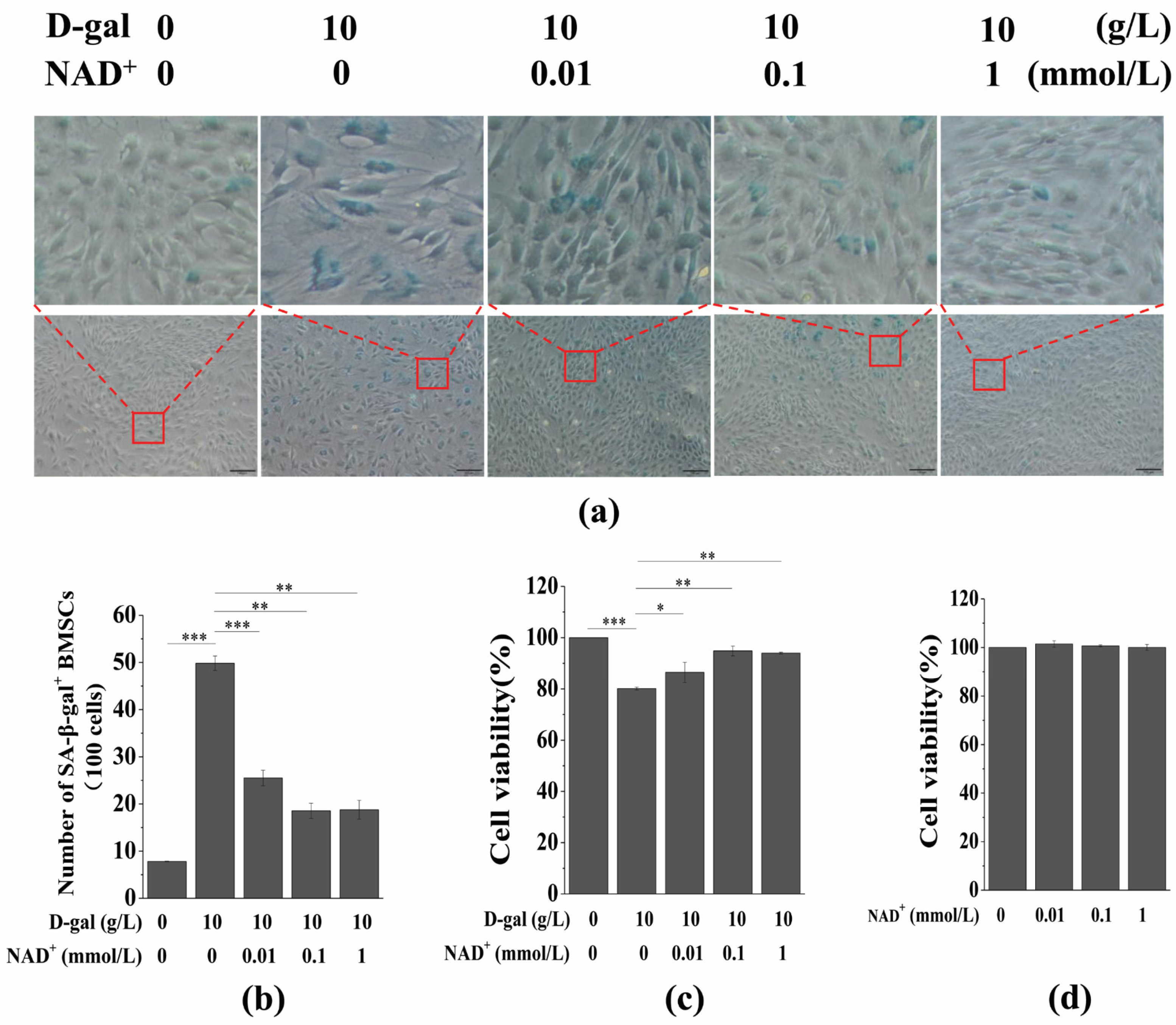
3.2. Exogenous NAD+ Postpones BMSC Senescence Induced by D-Gal
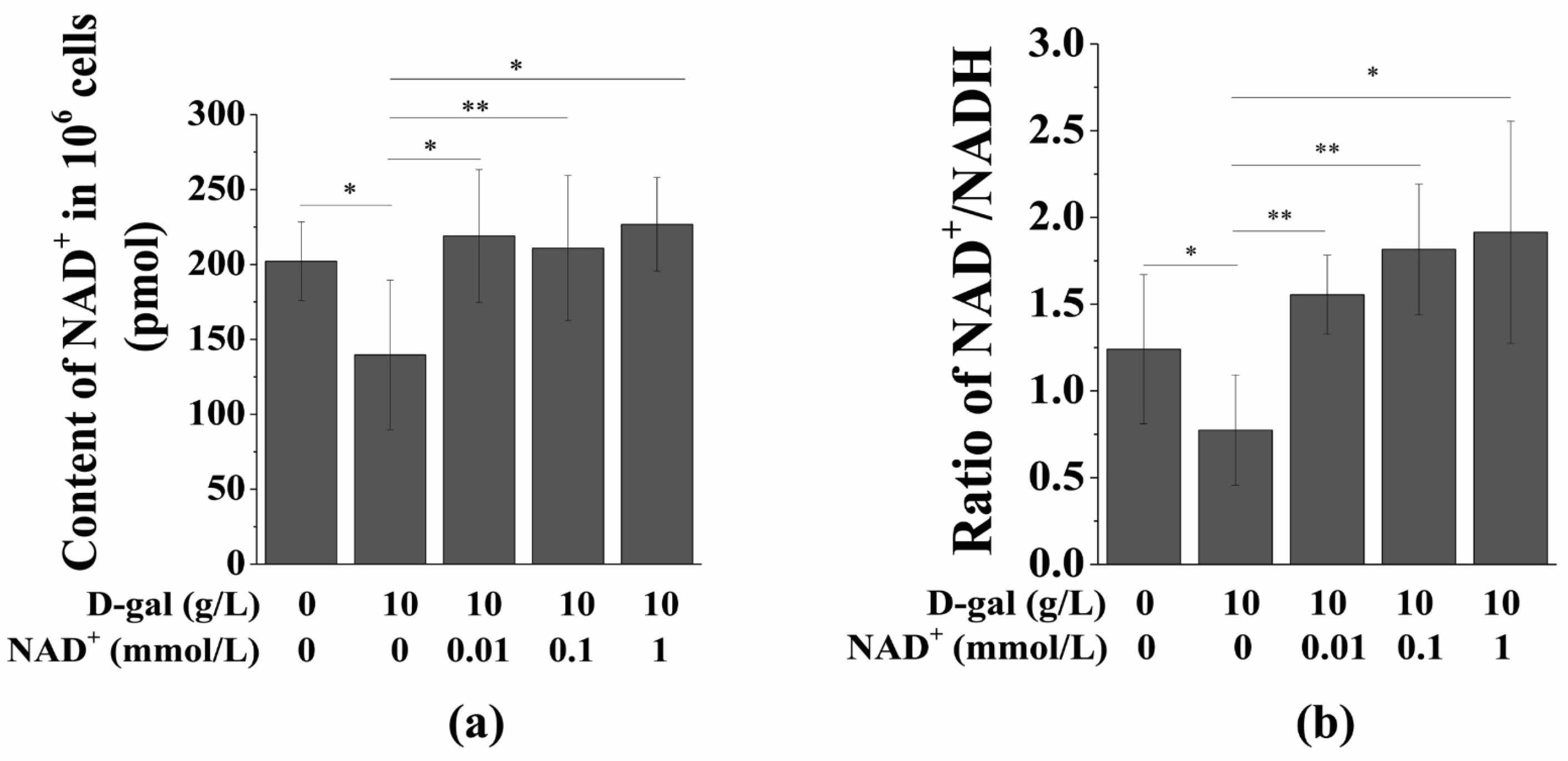
3.3. Exogenous NAD+ Increases the Intracellular NAD+ Content and NAD+/NADH Ratio
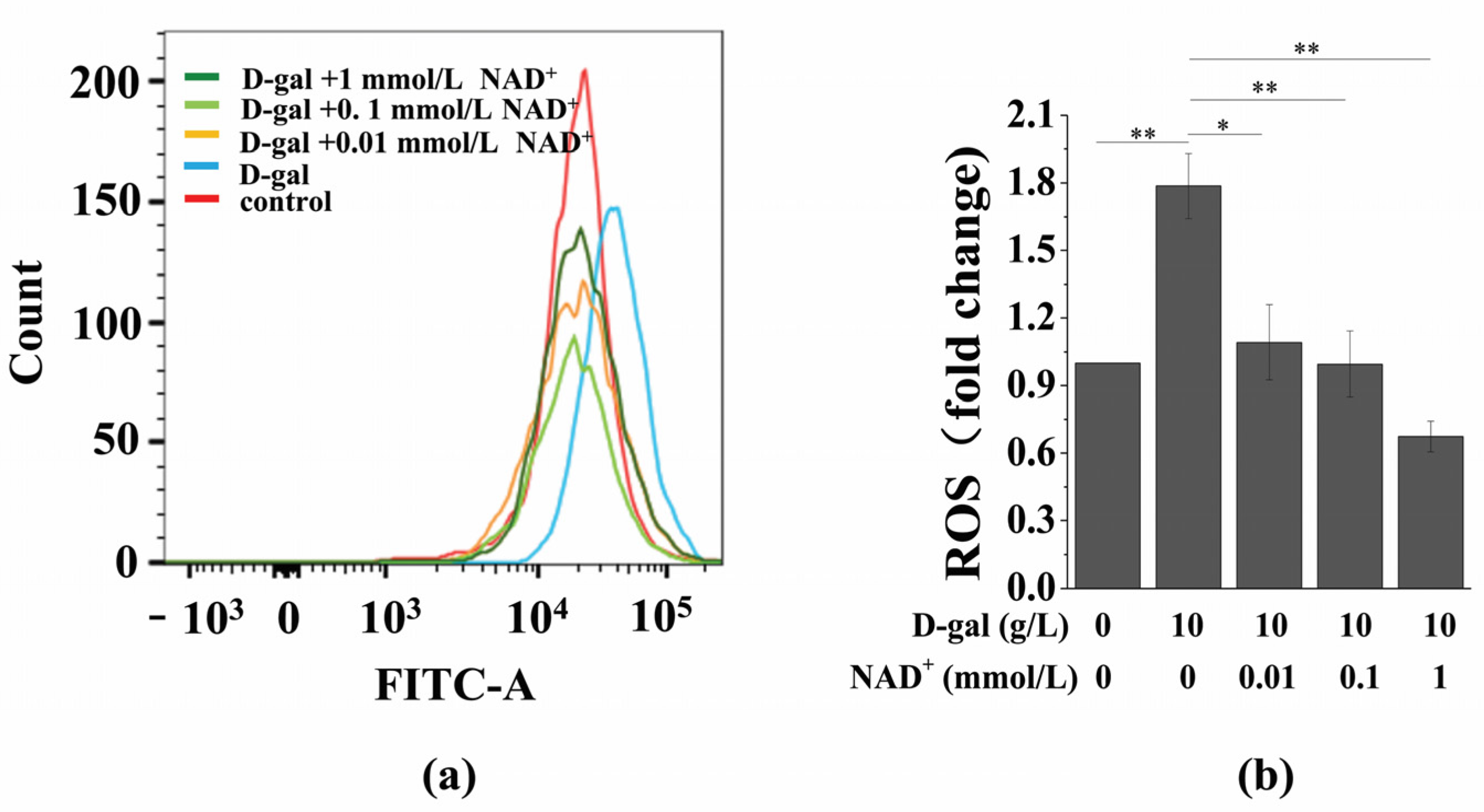
3.4. Exogenous NAD+ Inhibits Intracellular ROS Generation in Senescent BMSCs
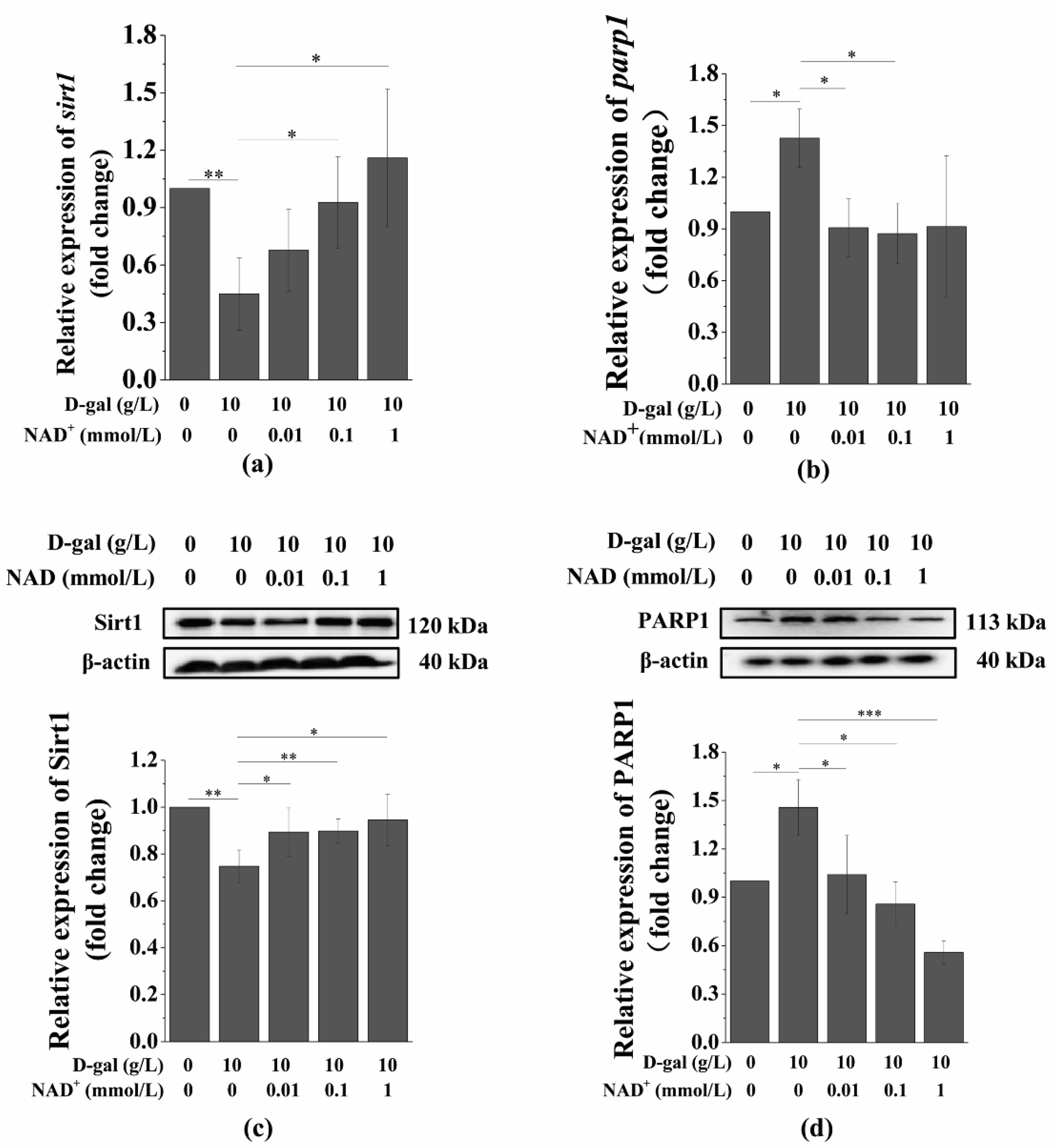
3.5. Exogenous NAD+ Regulates the Expression of Sirt1 and PARP1 in Senescent BMSCs
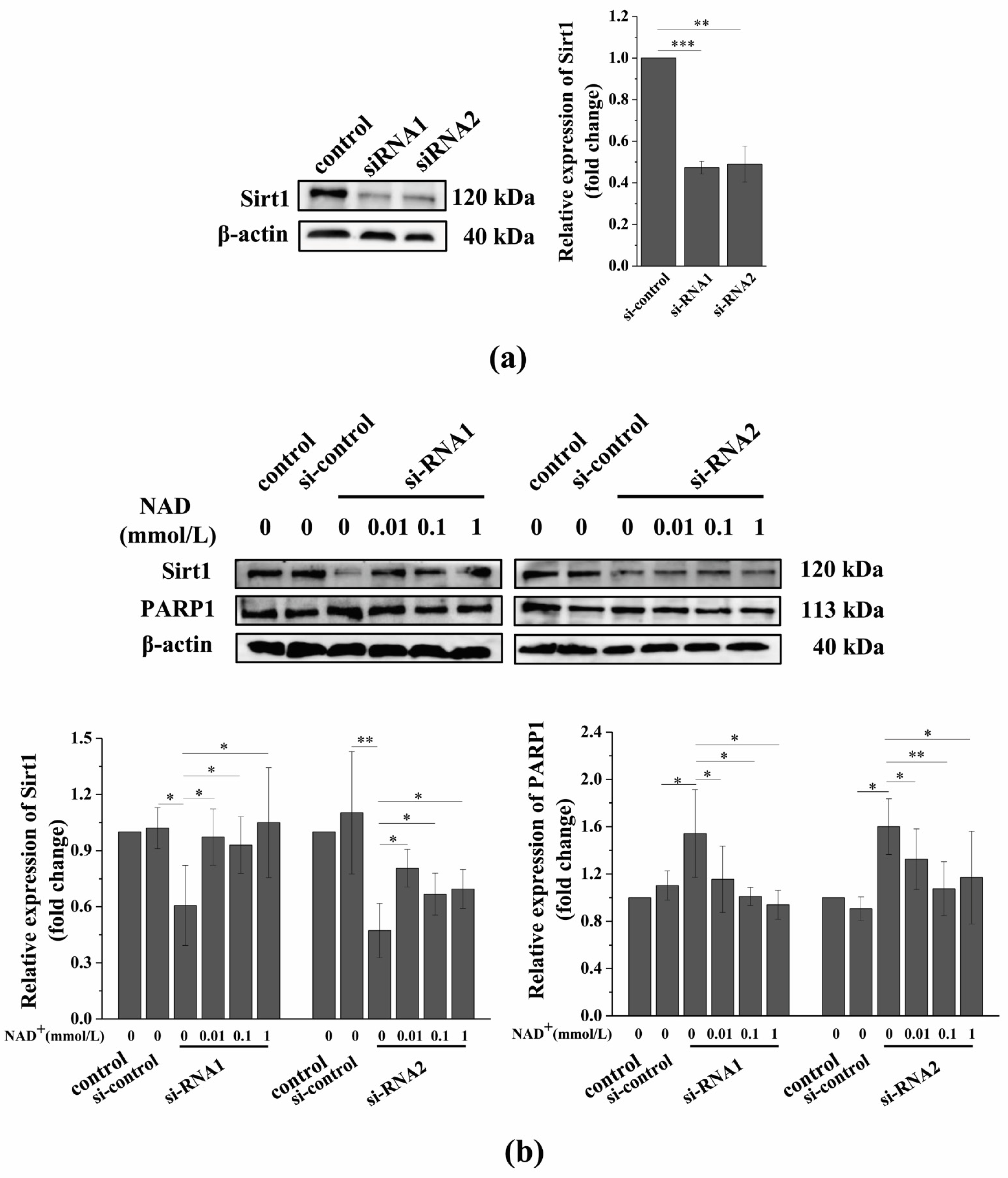
3.6. Sirt1 Knockdown Suppresses the Exogenous NAD+-Affected Expression of Sirt1 and PARP1 in Senescent BMSCs
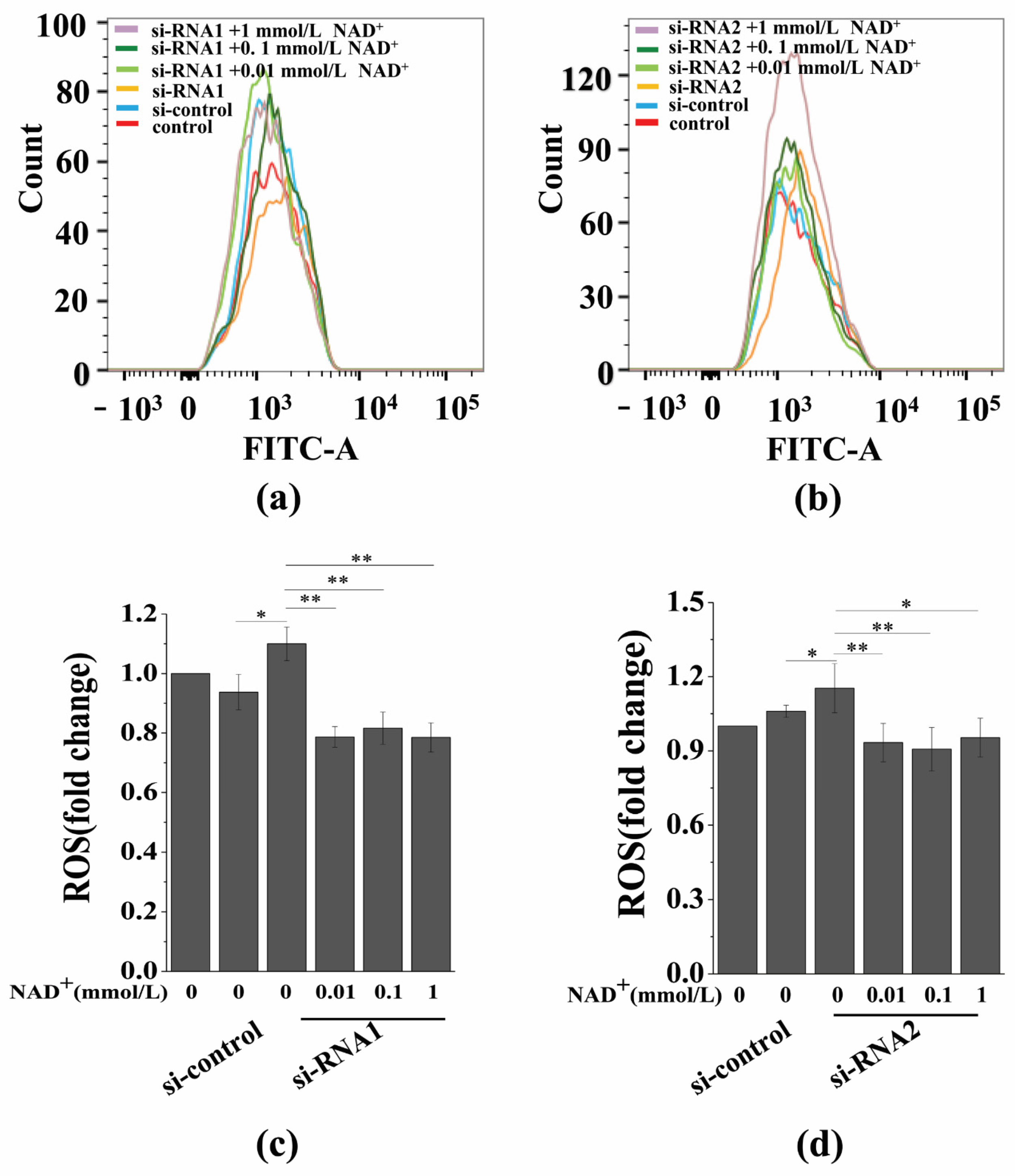
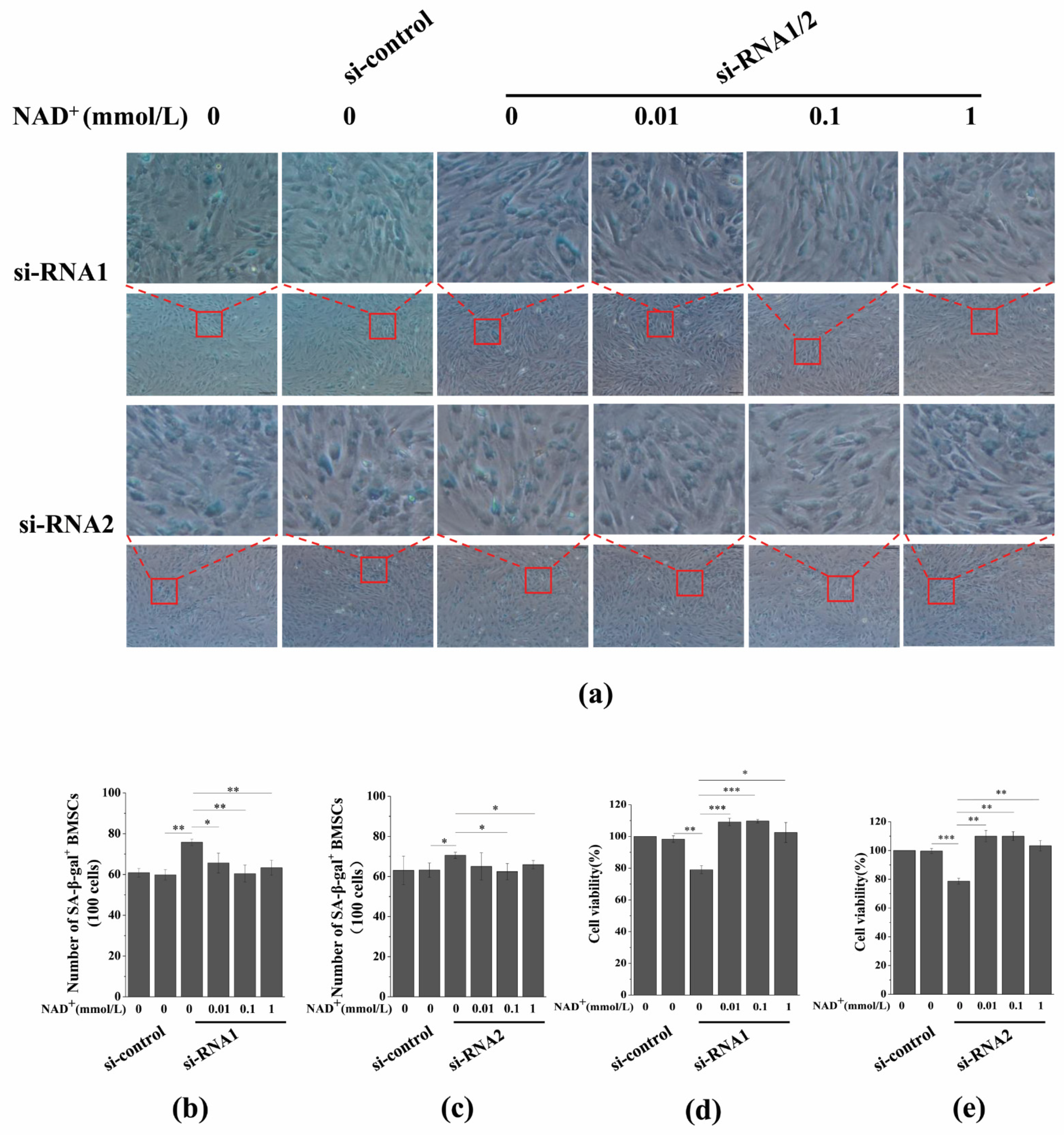
3.7. Sirt1 Knockdown Represses Exogenous NAD+-Inhibited ROS Generation and D-Gal-Induced BMSC Senescence
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jitendra, K.M.; Aurora, C.; Christine, B.; Yohei, M.; Christopher, B.; Mukesh, K.; Johann, M.K.; Michael, R.S.; Wang, Z.Q.; Hans, A.K.; et al. Telomerase abrogates aneuploidy-induced telomere replication stress, senescence and cell depletion. EMBO J. 2015, 36, 2922–2924. [Google Scholar] [CrossRef]
- Xu, Z.; Feng, W.; Shen, Q.; Yu, N.N.; Yu, K.; Wang, S.J.; Chen, Z.J.; Shioda, S.J.; Guo, Y. Rhizoma coptidis and berberine as a natural drug to combat aging and aging-related diseases via anti-oxidation and AMPK activation. Aging Dis. 2017, 8, 760–777. [Google Scholar] [CrossRef]
- Lara, A.; Allison, J.B. Stem cell DNA damage and genome mutation in the context of aging and cancer initiation. Cold. Spring Harb. Perspect. Biol. 2020, 12, a036210. [Google Scholar] [CrossRef]
- Fernandez, R.E.; Franzen, J.; Goetzke, R.; Hollmann, J.; Ostrowska, A.; Oliverio, M.; Sieben, T.; Rath, B.; Kornfeld, J.W.; Wagner, W.; et al. Senescence-associated metabolomic phenotype in primary and iPSC-derived mesenchymal stromal cells. Stem Cell Rep. 2020, 14, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Verdin, E. NAD+ in aging, metabolism, and neurodegeneration. Science 2015, 350, 1208–1213. [Google Scholar] [CrossRef] [PubMed]
- Guarente, L. Cell Metabolism. The resurgence of NAD+. Science 2016, 352, 1396–1397. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Sauve, A.A. NAD (+) metabolism: Bioenergetics, signaling and manipulation for therapy. Biochim. Biophys. Acta 2016, 1864, 1787–1800. [Google Scholar] [CrossRef]
- Chini, C.C.; Tarragó, M.G.; Chini, E.N. NAD+ and the aging process: Role in life, death and everything in between. Mol. Cell Endocrinol. 2017, 455, 62–74. [Google Scholar] [CrossRef]
- Camacho-Pereira, J.; Tarragó, M.G.; Chini, C.C.S.; Nin, V.; Escande, C.; Warner, G.M.; Puranik, A.S.; Schoon, R.A.; Reid, G.M.; Galina, A.; et al. CD38 dictates age-related NAD decline and mitochondrial dysfunction through an SIRT3-dependent mechanism. Cell Metab. 2016, 23, 1127–1139. [Google Scholar] [CrossRef]
- Chini, C.C.; Guerrico, A.M.; Nin, V.; Camacho-Pereira, J.; Escande, C.; Barbosa, M.T.; Chini, E.N. Targeting of NAD metabolism in pancreatic cancer cells: Potential novel therapy for pancreatic tumors. Clin. Cancer Res. 2014, 20, 120–130. [Google Scholar] [CrossRef]
- Alano, C.C.; Ying, W.; Swanson, R.A. Poly (ADP-ribose) polymerase-1-mediated cell death in astrocytes requires NAD+ depletion and mitochondrial permeability transition. J. Biol. Chem. 2014, 279, 18895–18902. [Google Scholar] [CrossRef]
- Wu, M.F.; Yin, J.H.; Hwang, C.S.; Tang, C.M.; Yang, D.I. NAD attenuates oxidative DNA damages induced by amyloid beta-peptide in primary rat cortical neurons. Free Radic. Res. 2014, 48, 794–805. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhao, K.K.; Tong, Y.; Zhou, Y.L.; Wang, Y.X.; Zhao, P.Q.; Wang, Z.Y. Exogenous NAD+ decreases oxidative stress and protects H2O2-treated RPE cells against necrotic death through the up-regulation of autophagy. Sci. Rep. 2016, 6, 26322. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wang, P.; Liu, X.; He, D.W.; Liang, C.X.; Yu, Y. Exogenous NAD(+) supplementation protects H9c2 cardiac myoblasts against hypoxia/reoxygenation injury via Sirt1-p53 pathway. Fundam. Clin. Pharmacol. 2014, 28, 180–189. [Google Scholar] [CrossRef]
- Zhang, H.; Ryu, D.; Wu, Y.; Gariani, K.; Wang, X.; Luan, P.L.; D’Amico, D.; Ropelle, E.R.; Lutolf, M.P.; Aebersold, R.; et al. NAD+ repletion improves mitochondrial and stem cell function and enhances life span in mice. Science 2016, 352, 1436–1443. [Google Scholar] [CrossRef]
- Gomes, A.P.; Price, N.L.; Ling, A.J.; Moslehi, J.J.; Montgomery, M.K.; Rajman, L.; White, J.P.; Teodoro, J.S.; Wrann, C.D.; Hubbard, B.P.; et al. Declining NAD+ induces a pseudohypoxic state disrupting nuclear-mitochondrial communication during aging. Cell 2013, 155, 1624–1638. [Google Scholar] [CrossRef] [PubMed]
- Pajk, M.; Cselko, A.; Varga, C.; Posa, A.; Tokodi, M.; Boldogh, I.; Goto, S.; Radak, Z. Exogenous nicotinamide supplementation and moderate physical exercise can attenuate the aging process in skeletal muscle of rats. Biogerontology 2017, 18, 593–600. [Google Scholar] [CrossRef]
- Katsyuba, E.; Mottis, A.; Zietak, M.; Franco, F.D.; Velpen, V.; Gariani, K.; Ryu, D.; Cialabrini, L.; Matilainen, O.; Liscio, P.; et al. De novo NAD+ synthesis enhances mitochondrial function and improves health. Nature 2018, 563, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Kanfi, Y.; Naiman, S.; Amir, G.; Peshti, V.; Zinman, G.; Nahum, L.; Bar-Joseph, Z.; Cohen, H.Y. The sirtuin SIRT6 regulates lifespan in male mice. Nature 2012, 483, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Alageel, A.; Tomasi, J.; Tersigni, C.; Brietzke, E.; Zuckerman, H.; Subramaniapillai, M.; Lee, Y.; Iacobucci, M.; Rosenblat, J.D.; Mansur, R.B.; et al. Evidence supporting a mechanistic role of sirtuins in mood and metabolic disorders. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 86, 95–101. [Google Scholar] [CrossRef]
- Caron, A.Z.; He, X.; Mottawea, W.; Seifert, E.L.; Jardine, K.; Dewar-Darch, D.; Cron, G.O.; Harper, M.; Stintzi, A.; McBurney, M.W. The SIRT1 deacetylase protects mice against the symptoms of metabolic syndrome. FASEB J. 2014, 28, 1306. [Google Scholar] [CrossRef]
- Perico, L.; Morigi, M.; Rota, C.; Breno, M.; Mele, C.; Noris, M.; Introna, M.; Capelli, C.; Longaretti, L.; Rottoli, D.; et al. Human mesenchymal stromal cells transplanted into mice stimulate renal tubular cells and enhance mitochondrial function. Nat. Commun. 2017, 8, 983. [Google Scholar] [CrossRef]
- Das, A.; Huang, G.X.; Bonkowski, M.S.; Longchamp, A.; Li, C.; Schultz, M.B.; Kim, L.; Osborne, B.; Joshi, S.; Lu, Y.C.; et al. Impairment of an endothelial NAD+-H2S signaling network is a reversible cause of vascular aging. Cell 2019, 173, 74–89. [Google Scholar] [CrossRef]
- Michael, B.S.; David, A.S. Why NAD+ declines during aging: It’s destroyed. Cell Metab. 2016, 23, 965–966. [Google Scholar] [CrossRef]
- Junaith, S.; Joseph, C.; Matthew, J.; Kayla, J.S.; Stephen, E.A. Dysregulation of SIRT-1 in aging mice increases skeletal muscle fatigue by a PARP-1-dependent mechanism. Aging (Albany NY) 2014, 6, 820–834. [Google Scholar] [CrossRef]
- Jiang, Y.X.; Luo, W.Q.; Wang, B.; Wang, X.Y.; Gong, P.; Xiong, Y. Resveratrol promotes osteogenesis via activating SIRT1/FoxO1 pathway in osteoporosis mice. Life Sci. 2020, 246, 117422. [Google Scholar] [CrossRef]
- Fang, E.F.; Kassahun, H.; Croteau, D.L.; Scheibye-Knudsen, M.; Marosi, K.; Lu, H.M.; Shamanna, R.A.; Kalyanasundaram, S.; Bollineni, R.C.; Wilson, M.A.; et al. NAD+ Replenishment Improves Lifespan and Healthspan in Ataxia Telangiectasia Models via Mitophagy and DNA Repair. Cell Metab. 2016, 24, 566–581. [Google Scholar] [CrossRef]
- Yuan, Y.; Shi, M.; Li, L.; Liu, J.; Chen, B.; Chen, Y.N.; An, X.X.; Liu, S.Y.; Luo, R.X.; Long, D.; et al. Mesenchymal stem cell-conditioned media ameliorate diabetic endothelial dysfunction by improving mitochondrial bioenergetics via the Sirt1/AMPK/PGC-1α pathway. Clin. Sci. (Lond.) 2016, 130, 2181–2198. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef]
- Liu, X.L.; Ding, J.; Meng, L.H. Oncogene-induced senescence: A double edged sword in cancer. Acta Pharmacol. Sin. 2018, 39, 1553–1558. [Google Scholar] [CrossRef] [PubMed]
- Kuzmin, V.S.; Pustovit, K.B.; Abramochkin, D.V. Effects of exogenous nicotinamide adenine dinucleotide (NAD+) in the rat heart are mediated by P2 purine receptors. J. Biomed. Sci. 2016, 23, 50. [Google Scholar] [CrossRef] [PubMed]
- Spagnuolo, G.; Codispoti, B.; Marrelli, M.; Rengo, C.; Rengo, S.; Tatullo, M. Commitment of oral-derived stem cells in dental and maxillofacial applications. Dent. J. 2018, 6, 72. [Google Scholar] [CrossRef]
- Barry, E.K.; Tanveer, S.; Emma, M.; Cathleen, D.; Youra, K.; Patrick, W.K.L.; Shashi, A.G. NAD+ salvage pathway in cancer metabolism and therapy. Pharmacol. Res. 2016, 114, 274–283. [Google Scholar] [CrossRef]
- Lunt, S.Y.; Vander Heiden, M.G. Aerobic glycolysis: Meeting the metabolic requirements of cell proliferation. Annu. Rev. Cell Dev. Biol. 2011, 27, 441–464. [Google Scholar] [CrossRef]
- Cantó, C.; Menzies, K.J.; Auwerx, J. NAD(+) metabolism and the control of energy homeostasis: A balancing act between mitochondria and the nucleus. Cell Metab. 2015, 22, 31–53. [Google Scholar] [CrossRef]
- Tateishi, K.; Wakimoto, H.; Iafrate, A.J.; Tanaka, S.; Loebel, F.; Lelic, N.; Wiederschain, D.; Bedel, O.; Deng, G.J.; Zhang, B.L.; et al. Extreme vulnerability of IDH1 mutant cancers to NAD+ depletion. Cancer Cell 2015, 28, 773–784. [Google Scholar] [CrossRef]
- Pillai, V.B.; Sundaresan, N.R.; Kim, G.; Gupta, M.; Rajamohan, S.B.; Pillai, J.B.; Samant, S.; Ravindra, P.V.; Isbatan, A.; Gupta, M.P.; et al. Exogenous NAD blocks cardiac hypertrophic response via activation of the SIRT3-LKB1-AMP-activated kinase pathway. J. Biol. Chem. 2010, 285, 3133–3144. [Google Scholar] [CrossRef]
- Wu, M.; Ma, L.; Xue, L.; Ye, W.L.; Lu, Z.Y.; Li, X.; Jin, Y.; Qin, X.; Chen, D.; Tang, W.C.; et al. Resveratrol alleviates chemotherapy-induced oogonial stem cell apoptosis and ovarian aging in mice. Aging (Albany NY) 2019, 11, 1030–1044. [Google Scholar] [CrossRef]
- Mi, R.C.; Han, D.M.R.; Sun, H.K. Resveratrol relieves hydrogen peroxide-induced premature senescence associated with SIRT1 in human mesenchymal stem cells. Mol. Cell. Toxicolol. 2014, 10, 29–39. [Google Scholar] [CrossRef]
- Zhou, T.; Yan, Y.; Zhao, C.; Xu, Y.; Wang, Q.; Xu, N. Resveratrol improves osteogenic differentiation of senescent bone mesenchymal stem cells through inhibiting endogenous reactive oxygen species production via AMPK activation. Redox. Rep. 2019, 24, 62–69. [Google Scholar] [CrossRef]
- Yang, X.; Jiang, T.; Wang, Y.; Guo, L. The role and mechanism of SIRT1 in resveratrol-regulated osteoblast autophagy in osteoporosis rats. Sci. Rep. 2019, 9, 18424. [Google Scholar] [CrossRef] [PubMed]
- Wahl, D.; Gokarn, R.; Mitchell, S.J.; Solon-Biet, S.M.; Cogger, V.C.; Simpson, S.J.; Couteur, D.G.L.; Cabo, R.D. Central nervous system SIRT1 expression is required for cued and contextual fear conditioning memory responses in aging mice. Nutr. Healthy Aging 2019, 5, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Hu, K.; Cao, J.; Wang, P.; Li, J.; Zeng, K.W.; He, X.D.; Tu, P.T.; Tong, T.J.; Han, L.M. lncRNA miat functions as a ceRNA to upregulate Sirt1 by sponging miR-22-3p in HCC cellular senescence. Aging (Albany NY) 2019, 11, 7098–7122. [Google Scholar] [CrossRef] [PubMed]
- Pi, C.C.; Yang, Y.; Sun, Y.N.; Wang, H.; Sun, H.; Ma, M.; Lin, L.; Shi, Y.G.; Li, Y.; Li, Y.L.; et al. Nicotinamide phosphoribosyltransferase postpones rat bone marrow mesenchymal stem cell senescence by mediating NAD+–Sirt1 signaling. Aging (Albany NY) 2019, 11, 3505–3522. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Liu, L.; Ding, Z.; Luo, Q.; Ju, Y.; Song, G. Exogenous NAD+ Postpones the D-Gal-Induced Senescence of Bone Marrow-Derived Mesenchymal Stem Cells via Sirt1 Signaling. Antioxidants 2021, 10, 254. https://doi.org/10.3390/antiox10020254
Wang J, Liu L, Ding Z, Luo Q, Ju Y, Song G. Exogenous NAD+ Postpones the D-Gal-Induced Senescence of Bone Marrow-Derived Mesenchymal Stem Cells via Sirt1 Signaling. Antioxidants. 2021; 10(2):254. https://doi.org/10.3390/antiox10020254
Chicago/Turabian StyleWang, Jie, Lin Liu, Zhongjie Ding, Qing Luo, Yang Ju, and Guanbin Song. 2021. "Exogenous NAD+ Postpones the D-Gal-Induced Senescence of Bone Marrow-Derived Mesenchymal Stem Cells via Sirt1 Signaling" Antioxidants 10, no. 2: 254. https://doi.org/10.3390/antiox10020254
APA StyleWang, J., Liu, L., Ding, Z., Luo, Q., Ju, Y., & Song, G. (2021). Exogenous NAD+ Postpones the D-Gal-Induced Senescence of Bone Marrow-Derived Mesenchymal Stem Cells via Sirt1 Signaling. Antioxidants, 10(2), 254. https://doi.org/10.3390/antiox10020254





