Gastro-Protective Effects of Albizia anthelmintica Leaf Extract on Indomethacin-Induced Gastric Ulcer in Wistar Rats: In Silico and In Vivo Studies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Drugs and Chemicals
2.2. Plant Material and Extraction
2.3. Ethical Statement
2.4. Animals
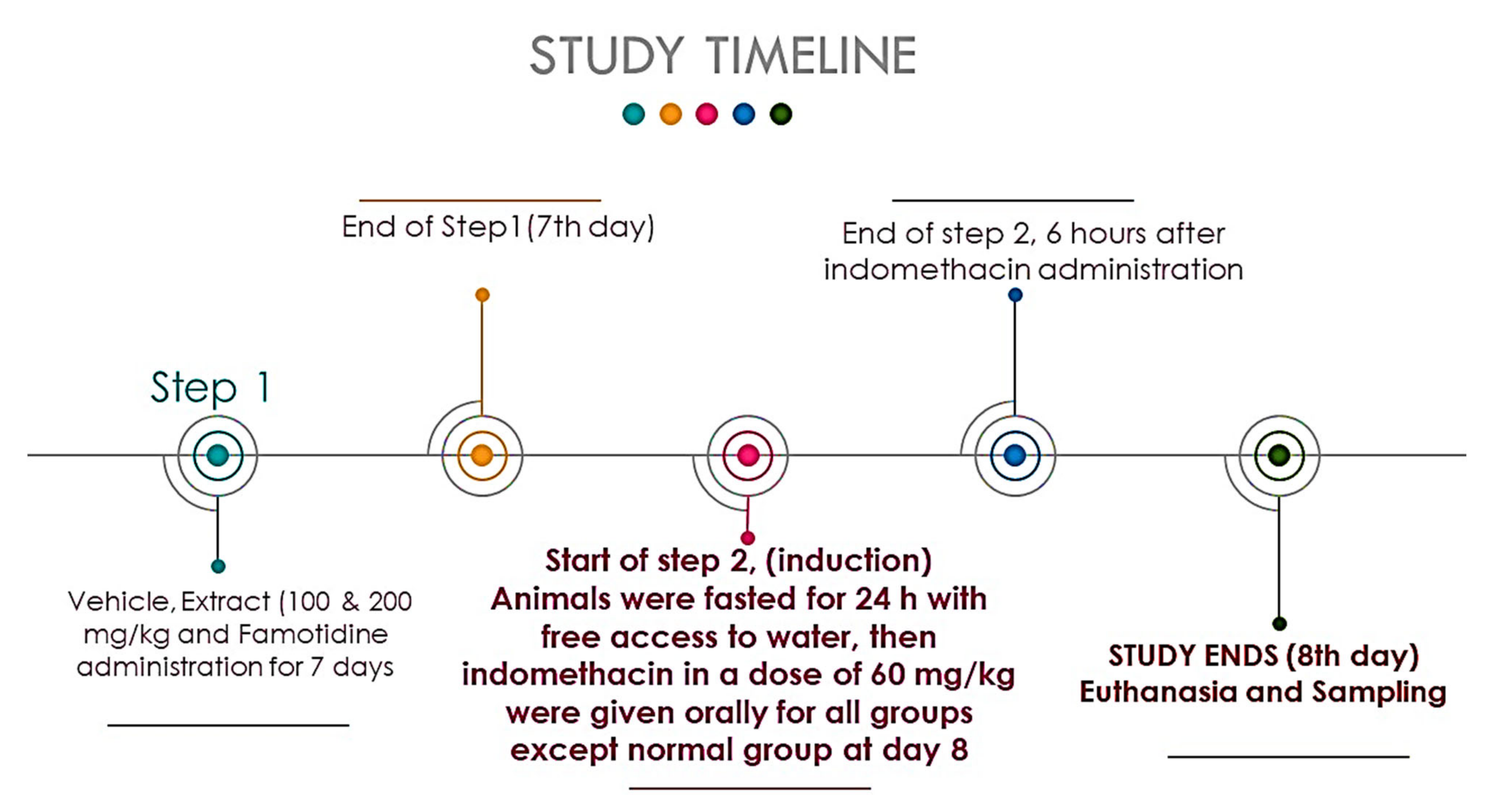
2.5. Experimental Design
2.6. Blood and Tissue Sampling
2.7. Assessment of Gross Mucosal Damage
2.8. Measurement of Gastric Acidity
2.9. Assessment of Gastric Tissue Oxidative Stress Markers
2.10. Determination of Gastric Tissue Inflammatory Markers IL-6 and TNF-α
2.11. Determination of Gastric Tissue Protein Expression of NF-κB and IKκB
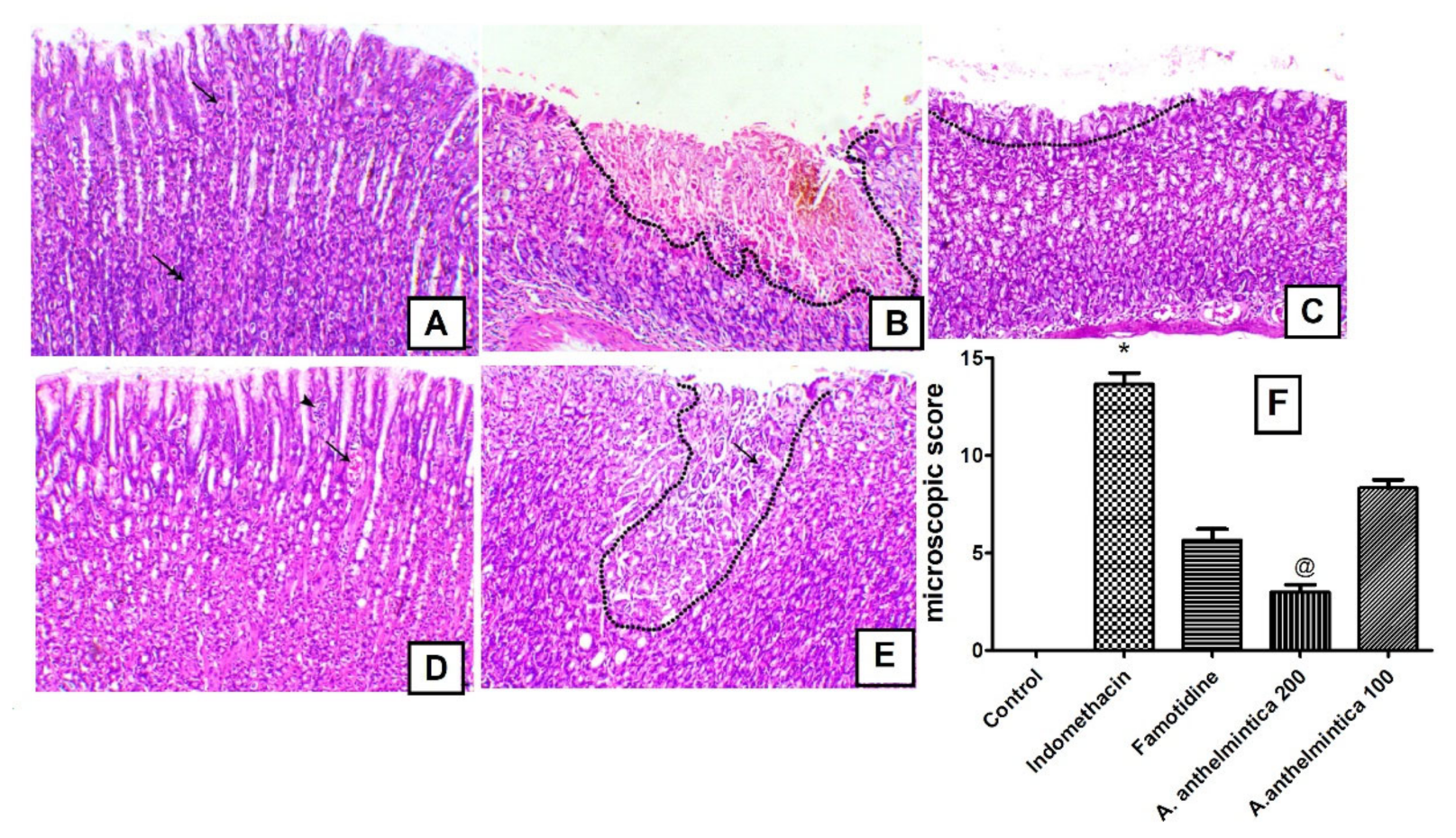
2.12. Microscopic Assessment of Gastric Ulcers
2.13. Immunohistochemical Evaluation of Gastric Inducible Nitric Oxide Synthase (iNOS)
2.14. Molecular Modelling
2.15. Statistical Analysis
3. Results
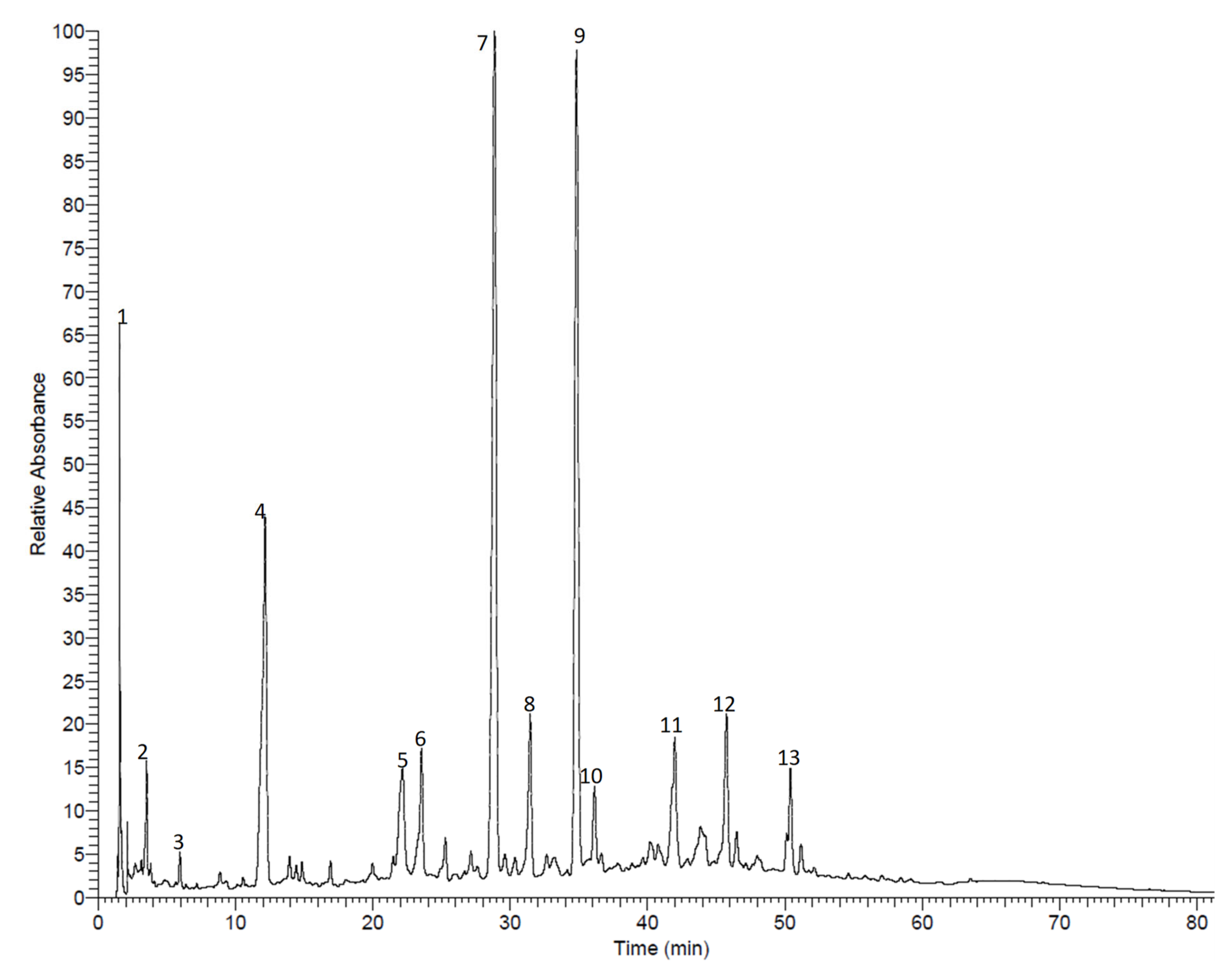
3.1. Compound Isolation and Phytochemical Profiling
3.2. Effect of A. anthelmintica Extract on Ulcer Index of Indomethacin-Treated Rats
3.3. Effect of A. anthelmintica Extract on Gastric Damage Alterations Induced by Indomethacin in Rats
3.4. Effect of A. anthelmintica Extract on Gastric Acidity Alterations Induced by Indomethacin in Rats
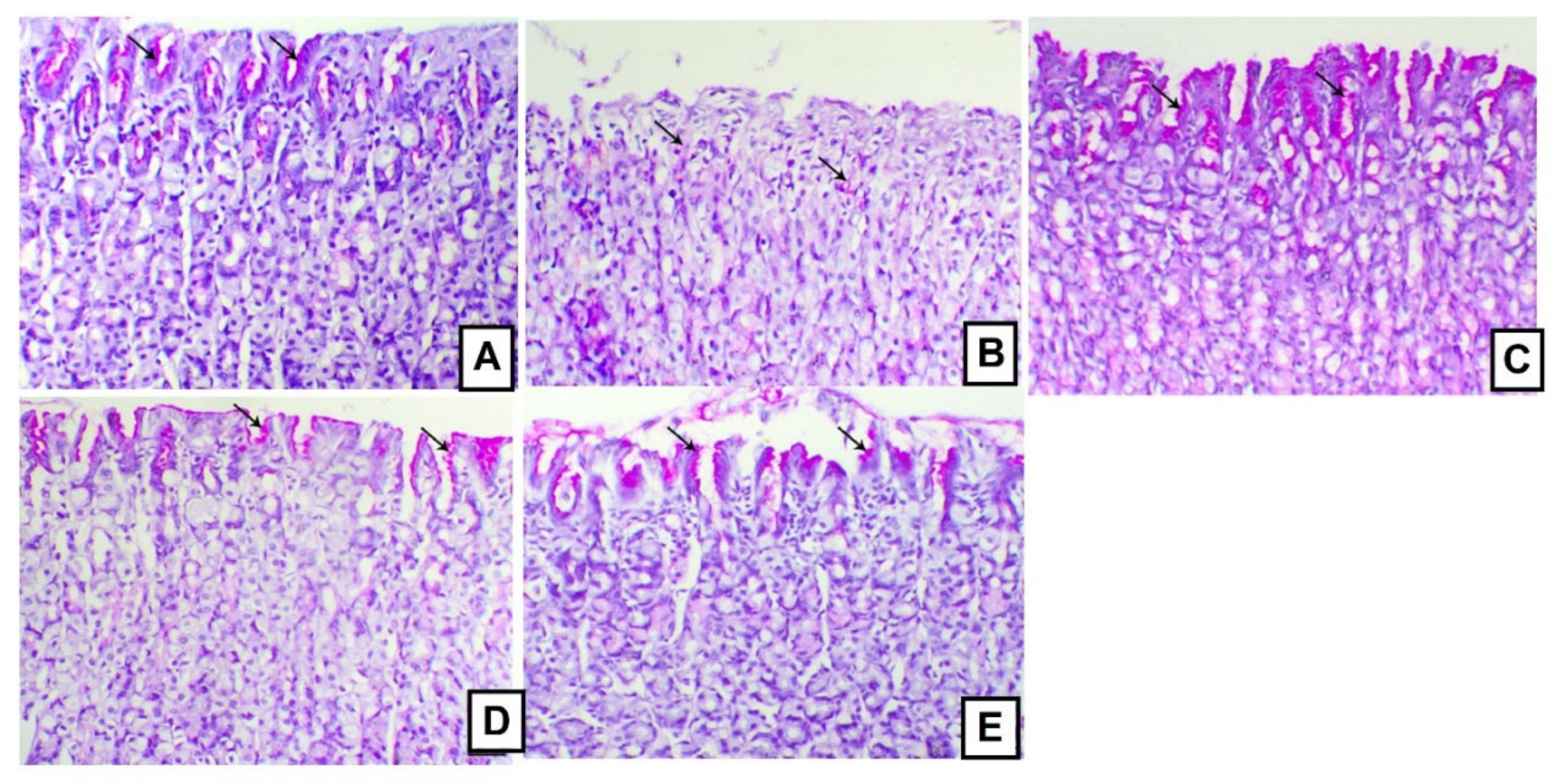
3.5. Effects of A. anthelmintica Extract on Gastric Mucin Alteration Induced by Indomethacin in Rats
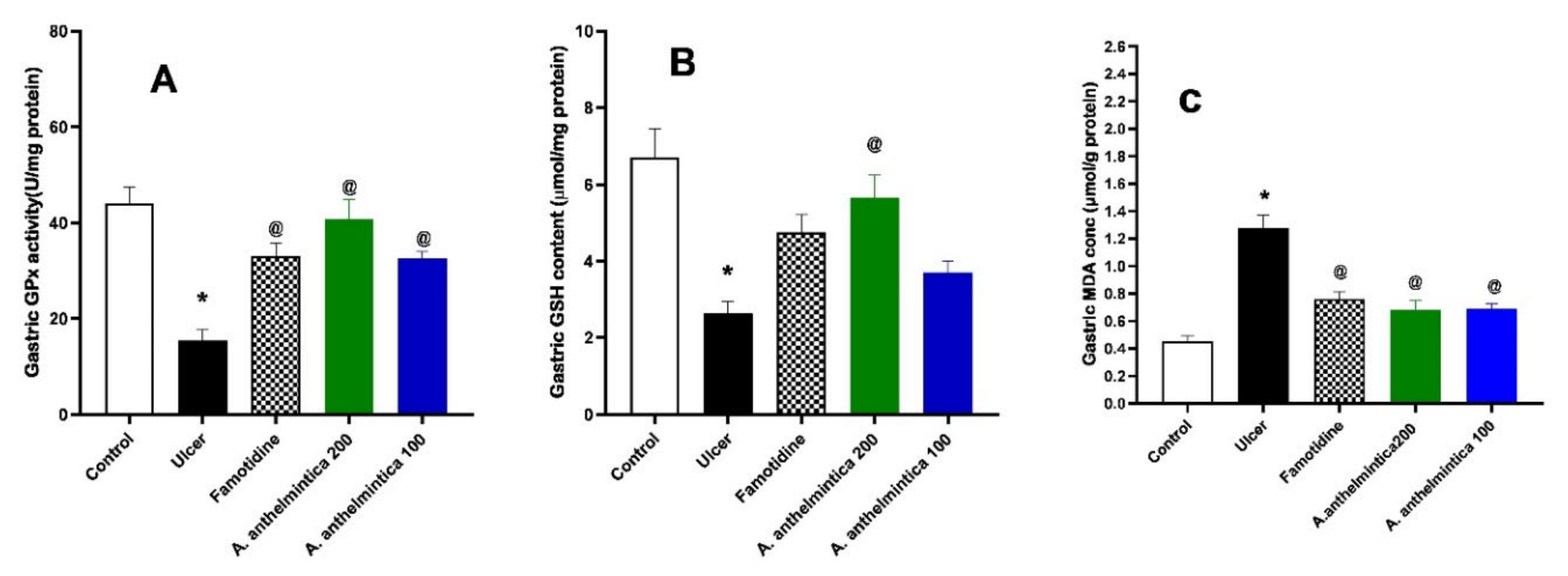
3.6. Effects of A. anthelmintica Extract on Changes in Oxidative Stress Markers Induced by Indomethacin in Rat Gastric Mucosa
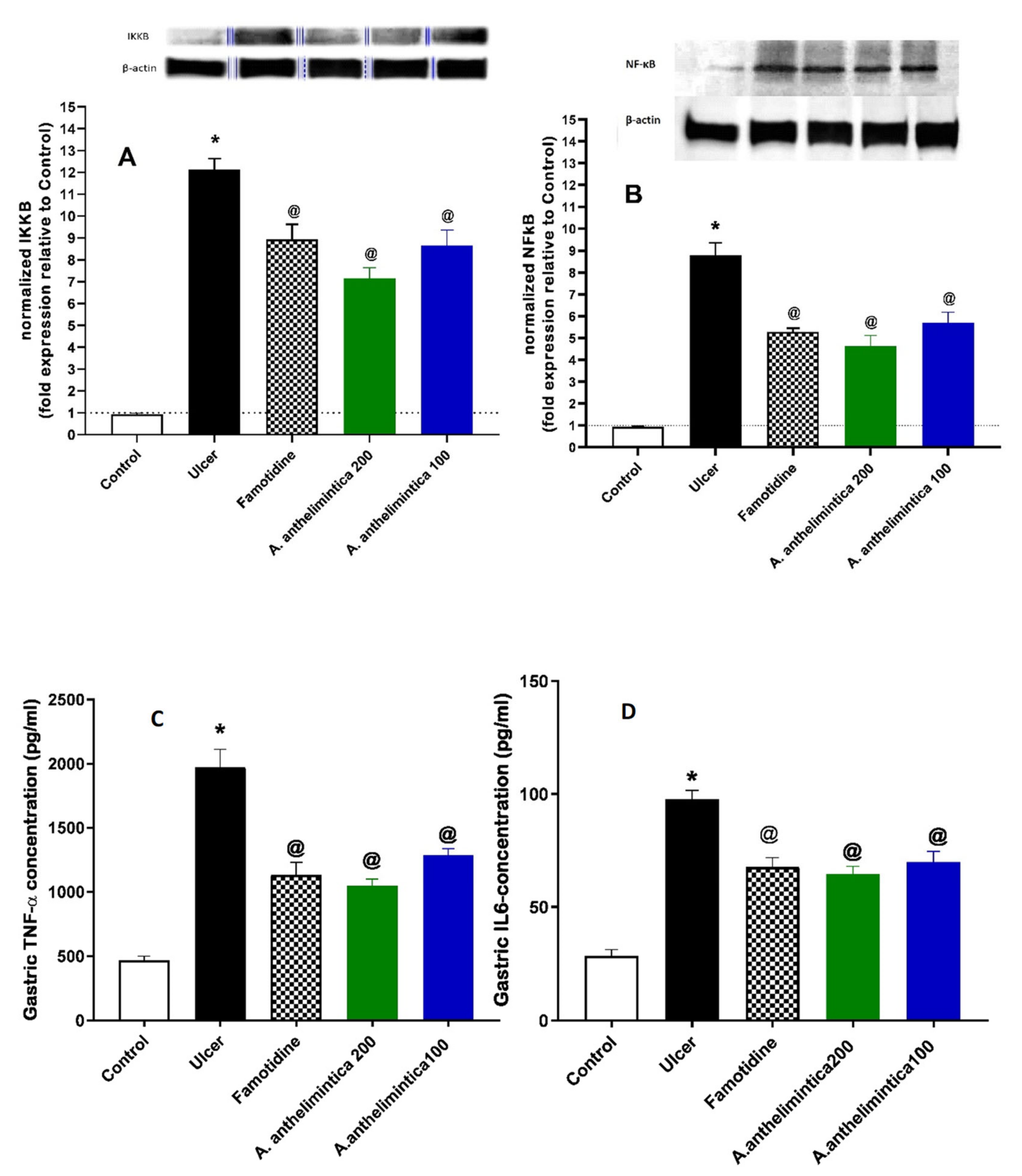
3.7. Effect of Albizia anthelmintica on Inflammatory Changes Induced by Indomethacin in Rat Gastric Mucosa
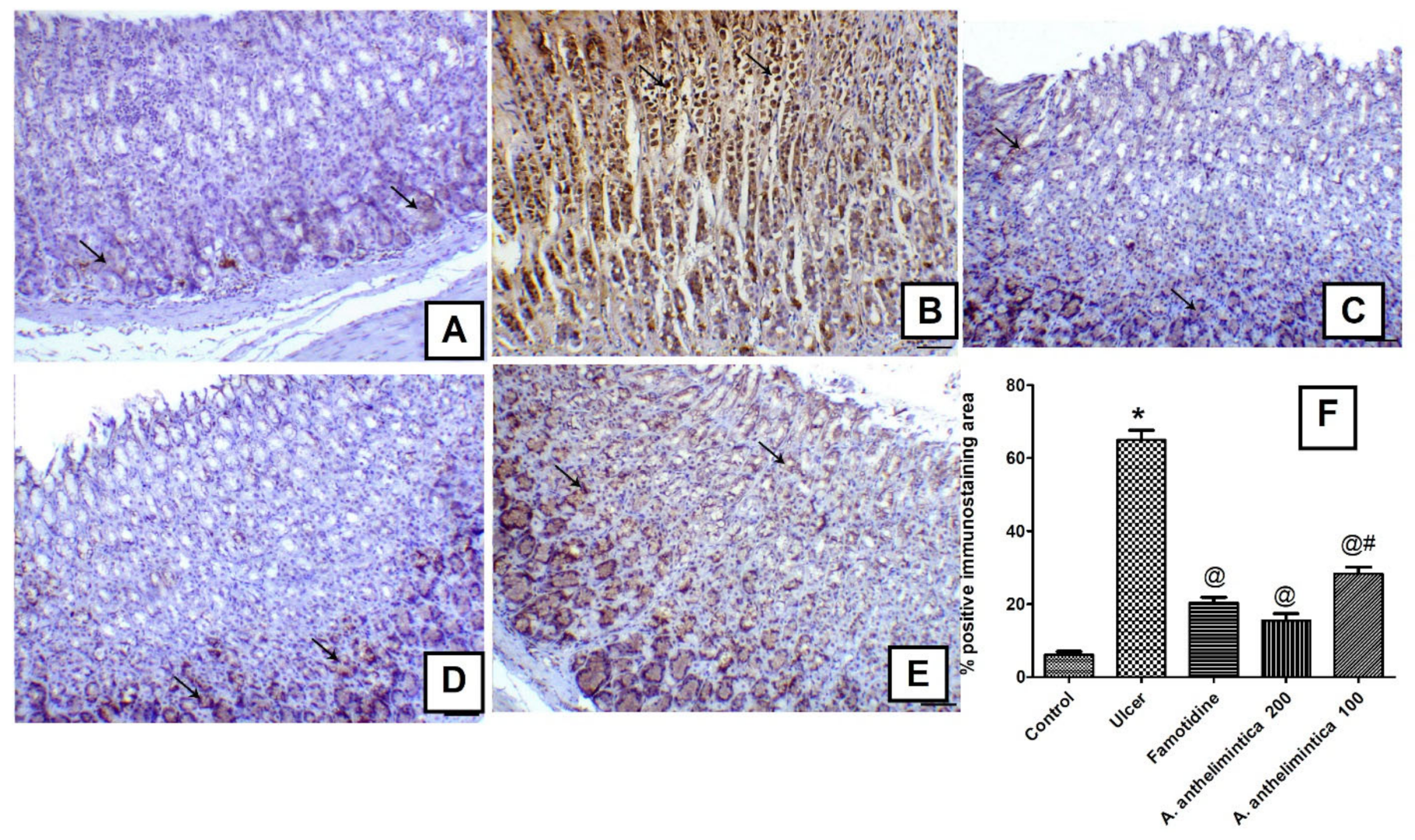
3.8. Effects of A. anthelmintica Extract on Changes in Gastric iNOS Immunoexpression Induced by Indomethacin in Rat Gastric Mucosa
3.9. Molecular Docking Studies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Sung, J.J.Y.; Kuipers, E.J.; El-Serag, H.B. Systematic review: The global incidence and prevalence of peptic ulcer disease. Aliment. Pharmacol. Ther. 2009, 29, 938–946. [Google Scholar] [CrossRef] [PubMed]
- Azhari, H.; Underwood, F.; King, J.; Coward, S.; Shah, S.; Ng, S.; Ho, G.; Chan, C.; Tang, W.; Kaplan, G.G. A36 the global incidence of peptic ulcer disease and its complications at the turn of the 21st century: A systematic review. J. Can. Assoc. Gastroenterol. 2018, 1, 61–62. [Google Scholar] [CrossRef] [Green Version]
- Ramakrishnan, K.; Salinas, R.C. Peptic ulcer disease. Am. Fam. Physician 2007, 76, 1005–1012. [Google Scholar] [PubMed]
- Karbalaei, M.; Keikha, M. Potential association between the hopQ alleles of Helicobacter pylori and gastrointestinal diseases: A systematic review and meta-analysis. Meta Gene 2020, 26, 100816. [Google Scholar] [CrossRef]
- Peura, D.A. Prevention of nonsteroidal anti-inflammatory drug–associated gastrointestinal symptoms and ulcer complications. Am. J. Med. Suppl. 2004, 117, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, D.; Chattopadhyay, A. Reactive oxygen species-induced gastric ulceration: Protection by melatonin. Current Med. Chem. 2006, 13, 1187–1202. [Google Scholar] [CrossRef] [PubMed]
- Lau, J.Y.; Sung, J.J.; Metz, D.C.; Howden, C.W. 187 Systematic Review of the Epidemiology of Complicated Peptic Ulcer: Incidence, Recurrence, Risk Factors and Mortality. Gastroenterology 2008, 134, A-32. [Google Scholar] [CrossRef]
- Yuan, Y.; Padol, I.T.; Hunt, R.H. Peptic ulcer disease today. Nat. Clin. Pract. Gastroenterol. Hepatol. 2006, 3, 80–89. [Google Scholar] [CrossRef]
- van Soest, E.M.; Siersema, P.D.; Dieleman, J.P.; Sturkenboom, M.; Kuipers, E.J. Persistence and adherence to proton pump inhibitors in daily clinical practice. Aliment. Pharmacol. Ther. 2006, 24, 377–385. [Google Scholar] [CrossRef]
- Sivri, B. Trends in peptic ulcer pharmacotherapy. Fundam. Clin. Pharmacol. 2004, 18, 23–31. [Google Scholar] [CrossRef]
- Zaghlool, S.S.; Abo-Seif, A.A.; Rabeh, M.A.; Abdelmohsen, U.R.; Messiha, B.A.S. Gastro-protective and anti-oxidant potential of Althaea officinalis and solanum nigrum on pyloric ligation/indomethacin-induced ulceration in rats. Antioxidants 2019, 8, 512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campos-Vidal, Y.; Herrera-Ruiz, M.; Trejo-Tapia, G.; Gonzalez-Cortazar, M.; Aparicio, A.J.; Zamilpa, A. Gastroprotective activity of kaempferol glycosides from Malvaviscus arboreus Cav. J. Ethnopharmacol. 2021, 268, 113633. [Google Scholar] [CrossRef] [PubMed]
- Sobeh, M.; Rezq, S.; Sabry, O.M.; Abdelfattah, M.A.O.; El Raey, M.A.; El-Kashak, W.A.; El-Shazly, A.M.; Mahmoud, M.F.; Wink, M. Albizia anthelmintica: HPLC-MS/MS profiling and in vivo anti-inflammatory, pain killing and antipyretic activities of its leaf extract. Biomed. Pharmacother. 2019, 115, 108882. [Google Scholar] [CrossRef] [PubMed]
- Slomiany, B.L.; Slomiany, A. Ghrelin suppression of Helicobacter pylori-induced gastric mucosal expression of iNOS is mediated through the inhibition of IKK-β activation by cNOS-dependent S-nitrosylation. Open J. Cell Biol. 2011, 1, 1–10. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Tappel, A.L.; Zalkin, H. Lipide peroxidation in isolated mitochondria. Arch. Biochem. Biophys. 1959, 80, 326–332. [Google Scholar] [CrossRef]
- Ellman, M. A spectrophotometric method for determination of reduced glutathione in tissues. Anal. Biochem. 1959, 74, 214–226. [Google Scholar]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar]
- Laemmli, U. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Nabil, M.; El Demellawy, M.A.; Mahmoud, M.F.; Mahmoud, A.A. Prolonged overnutrition with fructose or fat induces metabolic derangements in rats by disrupting the crosstalk between the hypothalamus and periphery: Possible amelioration with fenofibrate. Eur. J. Pharmacol. 2020, 879, 173136. [Google Scholar] [CrossRef]
- Bancroft, J.C.J.; Stevens, A. Pigments and Minerals. In Theory and practice of Histological Techniques, 3rd ed.; Churchill Livingstone: New York, NY, USA, 1990. [Google Scholar]
- Ortaç, D.; Cemek, M.; Karaca, T.; Büyükokuroğlu, M.E.; Özdemir, Z.Ö.; Kocaman, A.T.; Göneş, S. In vivo anti-ulcerogenic effect of okra (Abelmoschus esculentus) on ethanol-induced acute gastric mucosal lesions. Pharm. Biol. 2018, 56, 165–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McManus, J.F.A. Histological and histochemical uses of periodic acid. Stain Technol. 1948, 23, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Saber, S.; Khalil, R.M.; Abdo, W.S.; Nassif, D.; El-Ahwany, E. Olmesartan ameliorates chemically-induced ulcerative colitis in rats via modulating NFκB and Nrf-2/HO-1 signaling crosstalk. Toxicol. Appl. Pharmacol. 2019, 364, 120–132. [Google Scholar] [CrossRef] [PubMed]
- El-Hawary, S.S.; Sobeh, M.; Badr, W.K.; Abdelfattah, M.A.; Ali, Z.Y.; El-Tantawy, M.E.; Rabeh, M.A.; Wink, M. HPLC-PDA-MS/MS profiling of secondary metabolites from Opuntia ficus-indica cladode, peel and fruit pulp extracts and their antioxidant, neuroprotective effect in rats with aluminum chloride induced neurotoxicity. Saudi J. Biol. Sci. 2020, 27, 2829–2838. [Google Scholar] [CrossRef] [PubMed]
- Parmar, N.S. The gastric anti-ulcer activity of naringenin, a specific histidine decarboxylase inhibitor. Int. J. Tissue React. 1983, 5, 415–420. [Google Scholar]
- Miyazaki, Y.; Ichimura, A.; Sato, S.; Fujii, T.; Oishi, S.; Sakai, H.; Takeshima, H. The natural flavonoid myricetin inhibits gastric H+, K+-ATPase. Eur. J. Pharmacol. 2018, 820, 217–221. [Google Scholar] [CrossRef]
- Kiss, A.K.; Naruszewicz, M. Polyphenolic compounds characterization and reactive nitrogen species scavenging capacity of Oenothera paradoxa defatted seed extracts. Food Chem. 2012, 131, 485–492. [Google Scholar] [CrossRef]
- Al-Rehaily, A.J.; Al-Howiriny, T.S.; Al-Sohaibani, M.O.; Rafatullah, S. Gastroprotective effects of ‘Amla’ Emblica officinalis on in vivo test models in rats. Phytomedicine 2002, 9, 515–522. [Google Scholar] [CrossRef]
- Lee, H.; Håkanson, R.; Karlsson, A.; Mattsson, H.; Sundler, F. Lansoprazole and Omeprazole Have Similar Effects on Plasma Gastrin Levels, Enterochromaffin-Like Cells, Gastrin Cells and Somatostatin Cells in the Rat Stomach. Digestion 1992, 51, 125–132. [Google Scholar] [CrossRef]
- Fossmark, R.; Martinsen, T.C.; Waldum, H.L. Adverse Effects of Proton Pump Inhibitors-Evidence and Plausibility. Int. J. Mol. Sci. 2019, 20, 5203. [Google Scholar] [CrossRef] [Green Version]
- Cheng, Y.-T.; Lu, C.-C.; Yen, G.-C. Phytochemicals enhance antioxidant enzyme expression to protect against NSAID-induced oxidative damage of the gastrointestinal mucosa. Mol. Nutr. Food Res. 2017, 61, 1600659. [Google Scholar]
- Cheng, Y.-T.; Wu, C.-H.; Ho, C.-Y.; Yen, G.-C. Catechin protects against ketoprofen-induced oxidative damage of the gastric mucosa by up-regulating Nrf2 in vitro and in vivo. J. Nutr. Biochem. 2013, 24, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Iloki-Assanga, S.B.; Lewis-Luján, L.M.; Fernández-Angulo, D.; Gil-Salido, A.A.; Lara-Espinoza, C.L.; Rubio-Pino, J.L. Retino-protective effect of Bucida buceras against oxidative stress induced by H2O2 in human retinal pigment epithelial cells line. BMC Complementary Altern. Med. 2015, 15, 254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banning, A.; Deubel, S.; Kluth, D.; Zhou, Z.; Brigelius-Flohé, R. The GI-GPx Gene Is a Target for Nrf2. Mol. Cell. Biol. 2005, 25, 4914–4923. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ukil, A.; Maity, S.; Das, P.K. Protection from experimental colitis by theaflavin-3,3′-digallate correlates with inhibition of IKK and NF- κ B activation: Protection of colitis by TFDG via NF- κ B pathway. Br. J. Pharmacol. 2006, 149, 121–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, J.-M.; Han, Y.-M.; Kangwan, N.; Lee, S.-Y.; Jung, M.-K.; Kim, E.-H.; Hahm, K.-B. S-allyl cysteine alleviates nonsteroidal anti-inflammatory drug-induced gastric mucosal damages by increasing cyclooxygenase-2 inhibition, heme oxygenase-1 induction, and histone deacetylation inhibition: S-allyl cysteine prevent gastric damage. J. Gastroenterol. Hepatol. 2014, 29, 80–92. [Google Scholar] [CrossRef]
- Santucci, L.; Fiorucci, S.; Di Matteo, F.M.; Morelli, A. Role of tumor necrosis factor α release and leukocyte margination in indomethacin-induced gastric injury in rats. Gastroenterology 1995, 108, 393–401. [Google Scholar] [CrossRef]
- Suzuki, T.; Grand, E.; Bowman, C.; Merchant, J.L.; Todisco, A.; Wang, L.; Del Valle, J. TNF-α and interleukin 1 activate gastrin gene expression via MAPK- and PKC-dependent mechanisms. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 281, G1405–G1412. [Google Scholar] [CrossRef]
- Nandi, J.; Saud, B.; Zinkievich, J.M.; Yang, Z.; Levine, R.A. TNF-α modulates iNOS expression in an experimental rat model of indomethacin-induced jejunoileitis. Mol. Cell. Biochem. 2010, 336, 17–24. [Google Scholar] [CrossRef]
- Salami, A.T.; Adebimpe, M.A.; Olagoke, O.C.; Iyiola, T.O.; Olaleye, S.B. Potassium bromate cytotoxicity in the Wister rat model of chronic gastric ulcers: Possible reversal by protocatechuic acid. J. Food Biochem. 2020, 44, e13501. [Google Scholar] [CrossRef]


| Groups | Necrosis | Hemorrhage | Edema | Inflammation |
|---|---|---|---|---|
| Control | 0 ± 0 | 0 ± 0 | 0 ± 0 | 0 ± 0 |
| Indomethacin | 2.67 * ± 0.21 | 3.67 * ± 0.21 | 4.00 * ± 0 | 3.33 * ± 0.21 |
| Famotidine | 1.00 ± 0.0 | 1.67 ± 0.21 | 1.67 ± 0.21 | 1.33 ± 0.21 |
| Extract 200 mg/kg | 0.33 @ ± 0.21 | 0.33 @# ± 0.21 | 1.33 @ ± 0.21 | 1.00 @ ± 0 |
| Extract 100 mg/kg | 1.67 ± 0.21 | 2.00 ± 0 | 2.67 ± 0.21 | 2.00 ± 0 |
| Compounds # | Histidine Decarboxylase (4E1O) | Gastric Proton Pump (5YLV) | ||
|---|---|---|---|---|
| Scoring Function | Amino Acid Residues Interactions | Scoring Function | Amino Acid Residues Interactions | |
| Galloyglucose | −18.15 | SER 196 (H-bonding) HIS 194 (H-bonding, Hydrophobic) | −12.27 | THR 134 (H-bonding) GLU 900 (H-bonding) LEU 811 (H-bonding) |
| Hydroxybenzyl-O-galloyl-glucose | −24.86 | GLY 246 (H-bonding) ASP 273 (H-bonding) SER 151 (H-bonding) | −16.81 | LEU 811 (H-bonding) TYR 799 (Hydrophobic) |
| Pyrogallol-O-methylgalloyl-glucose | −24.53 | SER 196 (H-bonding) ASP 273 (H-bonding) TYR 80 (Hydrophobic) | −18.06 | GLU 900 (H-bonding) ASP 137 (H-bonding) ASN 138 (H-bonding) |
| Glyceric acid-O-galloy glucose | −20.42 | PRO 82 (H-bonding) SER 151 (H-bonding) SER 196 (H-bonding) HIS 194 (H-bonding, Hydrophobic) | −15.08 | THR 134 (H-bonding) GLU 900 (H-bonding) LEU 811 (H-bonding) ASN 138 (H-bonding) |
| Coumaroyl-O-galloyl-glucose | −23.13 | SER 151 (H-bonding) ASP 273 (H-bonding) GLY 246 (H-bonding) | −13.52 | GLU 900 (H-bonding) LEU 811 (H-bonding) GLN 127 (H-bonding) TYR 799 (Hydrophobic) |
| Benzyl-O-galloyl-glucose | −22.01 | SER 151 (H-bonding) ASP 273 (H-bonding) | −17.11 | LEU 811 (H-bonding) GLN 127 (H-bonding) |
| Quercetin glucoside | −24.05 | SER 196 (H-bonding) HIS 194 (Hydrophobic) TYR 81 (H-bonding) | −16.78 | LEU 811 (H-bonding) ASP 137 (H-bonding) THR 134 (H-bonding) |
| Quercetin-O-galloyl-glucoside | −21.03 | GLY 246 (H-bonding) ASP 273 (H-bonding) TYR 81 (H-bonding) | −20.59 | LEU 811 (H-bonding) ASP 137 (H-bonding) GLN 127 (H-bonding) ASN 138 (H-bonding) |
| Quercetin benzoyl-glucoside | −20.79 | GLY 246 (H-bonding) ASP 273 (H-bonding) TYR 81 (H-bonding) HIS 194 (Hydrophobic) | −18.51 | GLU 900 (H-bonding) LEU 811 (H-bonding) GLN 127 (H-bonding) ASN 138 (H-bonding) THR 134 (H-bonding) |
| Quercetin coumaroyl-glucoside | −19.59 | GLY 246 (H-bonding) TYR 81 (H-bonding) ASP 273 (H-bonding) HIS 194 (H-bonding, Hydrophobic) | −17.83 | LEU 811 (H-bonding) GLN 127 (H-bonding) LEU 133 (H-bonding) |
| Quercetin caffeoyl-glucoside | −24.41 | SER 151 (H-bonding) TYR 81 (H-bonding) GLY 246 (H-bonding) | −21.58 | GLU 900 (H-bonding) LEU 811 (H-bonding) LEU 811 (H-bonding) |
| Kaempferol glucoside | −24.06 | PRO 82 (H-bonding) GLY 246 (H-bonding) ASP 273 (H-bonding) HIS 194 (Hydrophobic) | −16.44 | LEU 811 (H-bonding) GLU 343 (H-bonding) GLU 795 (H-bonding) CYS 813 (H-bonding) |
| Kaempferol galloyl-glucoside | −25.21 | SER 196 (H-bonding) THR 248 (H-bonding) TYR 81 (H-bonding) ASP 273 (H-bonding) | −19.67 | LEU 811 (H-bonding) GLU 900 (H-bonding) GLN 127 (H-bonding) LEU 133 (H-bonding) ASP 132 (H-bonding) |
| Kaempferol coumaroyl-glucoside | −16.40 | GLY 246 (H-bonding) ASP 273 (H-bonding) TYR 81 (H-bonding) HIS 194 (H-bonding) SER 151 (Hydrophobic) HIS 194 (Hydrophobic) | −18.30 | GLU 900 (H-bonding) GLN 127 (H-bonding) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nabil, M.; El Raey, M.A.; Abdo, W.; Abdelfattah, M.A.O.; El-Shazly, A.M.; Sobeh, M.; Mahmoud, M.F. Gastro-Protective Effects of Albizia anthelmintica Leaf Extract on Indomethacin-Induced Gastric Ulcer in Wistar Rats: In Silico and In Vivo Studies. Antioxidants 2021, 10, 176. https://doi.org/10.3390/antiox10020176
Nabil M, El Raey MA, Abdo W, Abdelfattah MAO, El-Shazly AM, Sobeh M, Mahmoud MF. Gastro-Protective Effects of Albizia anthelmintica Leaf Extract on Indomethacin-Induced Gastric Ulcer in Wistar Rats: In Silico and In Vivo Studies. Antioxidants. 2021; 10(2):176. https://doi.org/10.3390/antiox10020176
Chicago/Turabian StyleNabil, Mohamed, Mohamed A. El Raey, Walied Abdo, Mohamed A. O. Abdelfattah, Assem M. El-Shazly, Mansour Sobeh, and Mona F. Mahmoud. 2021. "Gastro-Protective Effects of Albizia anthelmintica Leaf Extract on Indomethacin-Induced Gastric Ulcer in Wistar Rats: In Silico and In Vivo Studies" Antioxidants 10, no. 2: 176. https://doi.org/10.3390/antiox10020176
APA StyleNabil, M., El Raey, M. A., Abdo, W., Abdelfattah, M. A. O., El-Shazly, A. M., Sobeh, M., & Mahmoud, M. F. (2021). Gastro-Protective Effects of Albizia anthelmintica Leaf Extract on Indomethacin-Induced Gastric Ulcer in Wistar Rats: In Silico and In Vivo Studies. Antioxidants, 10(2), 176. https://doi.org/10.3390/antiox10020176






