Abstract
A chemical study of the ethyl acetate (EtOAc) extract from the deep-sea-derived fungus Penicillium thomii YPGA3 led to the isolation of a new austalide meroterpenoid (1) and seven known analogues (2−8), two new labdane-type diterpenoids (9 and 10) and a known derivative (11). The structures of new compounds 1, 9, and 10 were determined by comprehensive analyses via nuclear magnetic resonance (NMR) and mass spectroscopy (MS) data. The absolute configurations of 1, 9, and 10 were determined by comparisons of experimental electronic circular dichroism (ECD) with the calculated ECD spectra. Compound 1 represented the third example of austalides bearing a hydroxyl group at C-5 instead of the conserved methoxy in other known analogues. To our knowledge, diterpenoids belonging to the labdane-type were discovered from species of Penicillium for the first time. Compound 1 showed cytotoxicity toward MDA-MB-468 cells with an IC50 value of 38.9 μM. Compounds 2 and 11 exhibited inhibition against α-glucosidase with IC50 values of 910 and 525 μM, respectively, being more active than the positive control acarbose (1.33 mM).
1. Introduction
Austalides are a class of natural meroterpenoids with attractive scaffolds. Previous biosynthetic studies revealed that they are biosynthesized by cyclization and oxidative modification of 6-[(2E, 6E)farnesyl]-5,7-dihydroxy-4-methylphthalide [1]. These meroterpenoids were mainly produced by the species of the fungal genera Aspergillus and Penicillium, especially those from marine environments. Since austalides A−E were first reported in 1981, a total of 36 analogues have been identified [2,3,4,5,6,7,8,9,10,11]. The structural variations of austalides are attributed to oxidation occurring at C-13, C-14, C-17, and the isopropyl (C-15, C-25, C-26) to generate alcohol, isopropenyl, lactone, ester, or ether functionalities. Some of the analogues exhibited significant bioactivities, such as cytotoxicity [6,10], antibacterial activity [7], anti-influenza A virus (H1N1) activity [8], endo-1,3-D-glucanase inhibition [9], and osteoclast differentiation inhibitory effects [11].
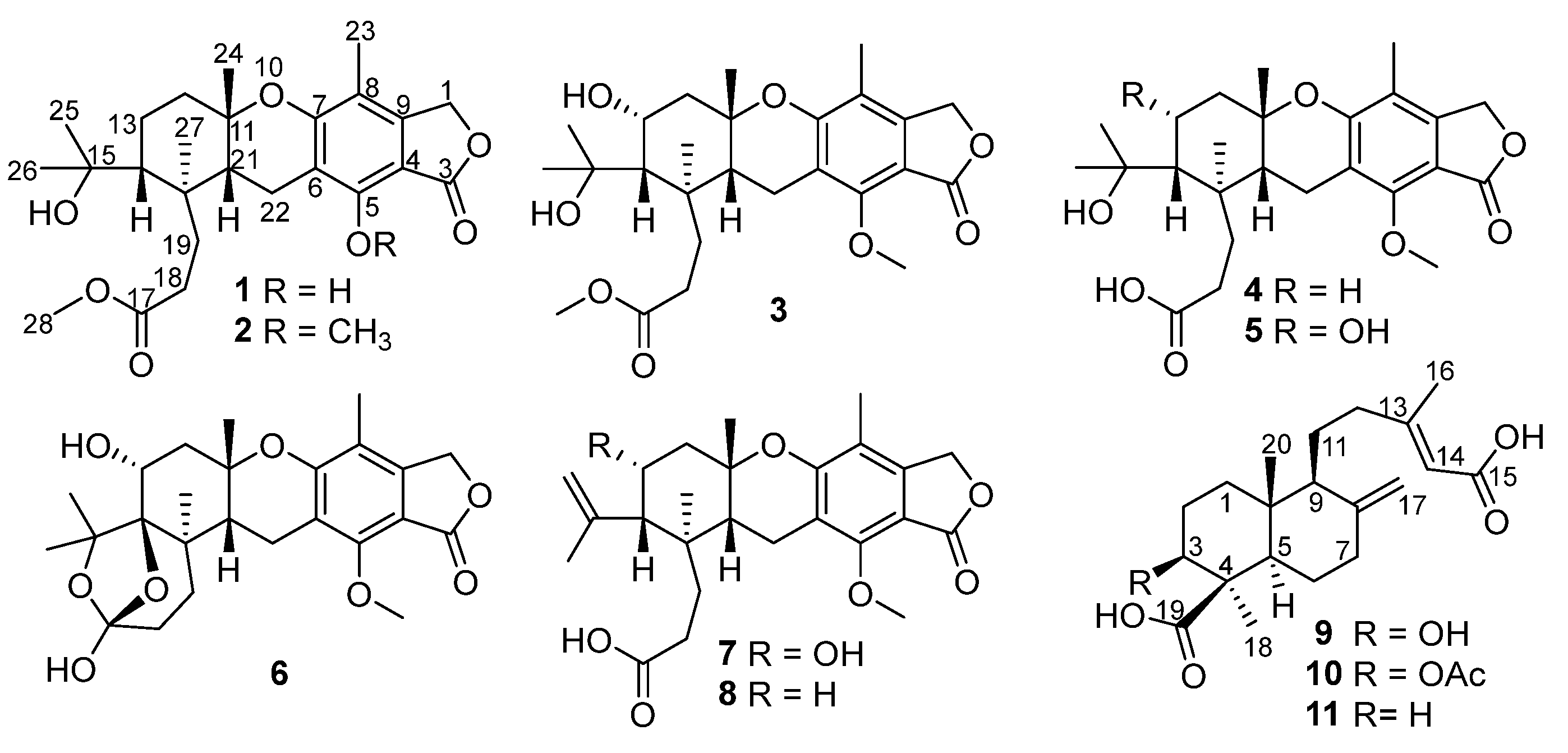
In our efforts to search for new or bioactive molecules from deep-sea-derived Penicillium or Aspergillus strains [12,13,14,15], chemical examination of Penicillium thomii YPGA3 afforded a new austalide meroterpenoid (1) and seven known analogues (2−8) (Figure 1). Compound 1 represented the third example of austalides bearing a phenol hydroxy at C-5 instead of the conserved methoxy in other known analogues. Additionally, two new labdane-type diterpenoids (9 and 10) and a known analogue (11) were also obtained (Figure 1). The labdane-type diterpenoids are mainly produced by plants and are rarely found in fungus. To our knowledge, diterpenoids belonging to the labdane-type were discovered from species of Penicillium for the first time. All compounds were evaluated for their inhibitions against α-glucosidase and NO (nitric oxide) production in lipopolysaccharide (LPS)-activated RAW 264.7 macrophages, and cytotoxicity toward two types of human breast carcinoma cells. Herein, the isolation, structural elucidation, and the bioactivity of compounds 1–11 are described.
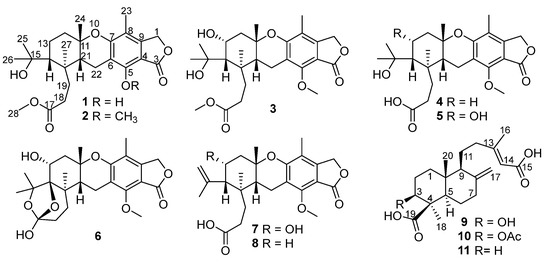
Figure 1.
Structures of compounds 1–11 from Penicillium thomii YPGA3.
2. Results
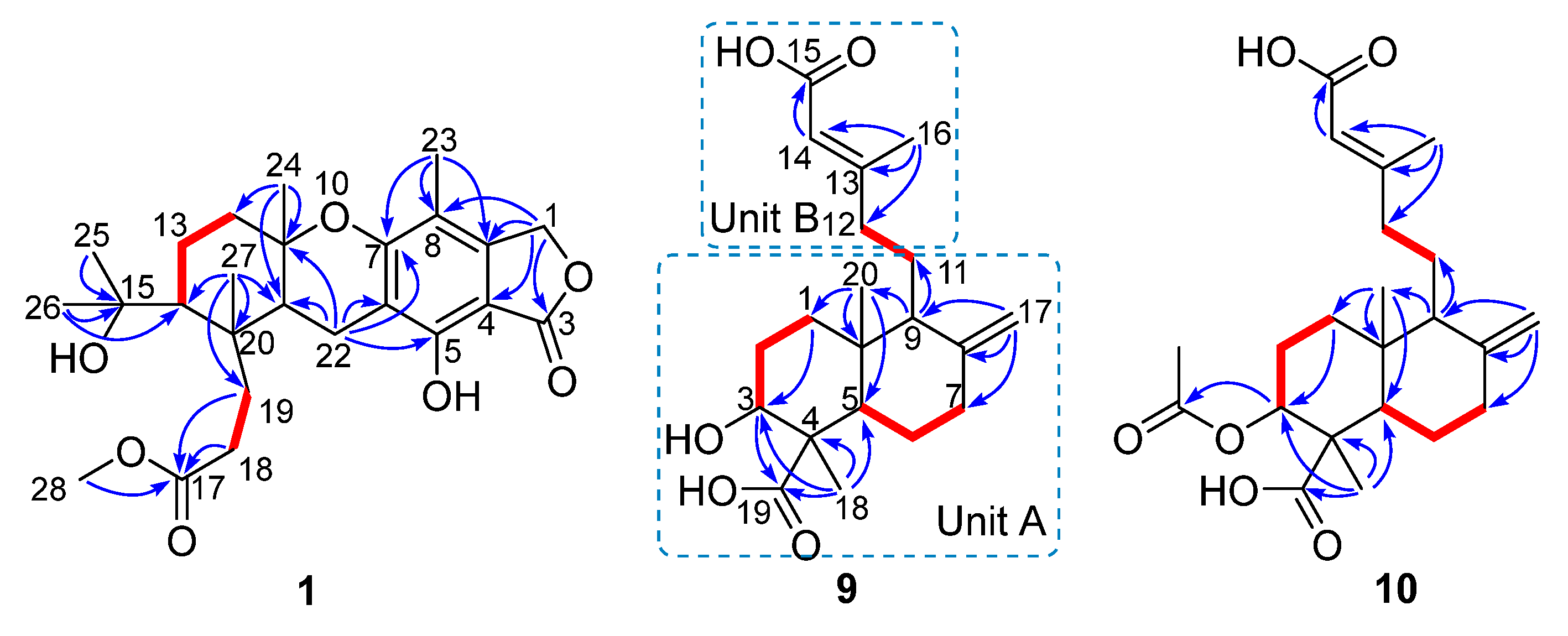
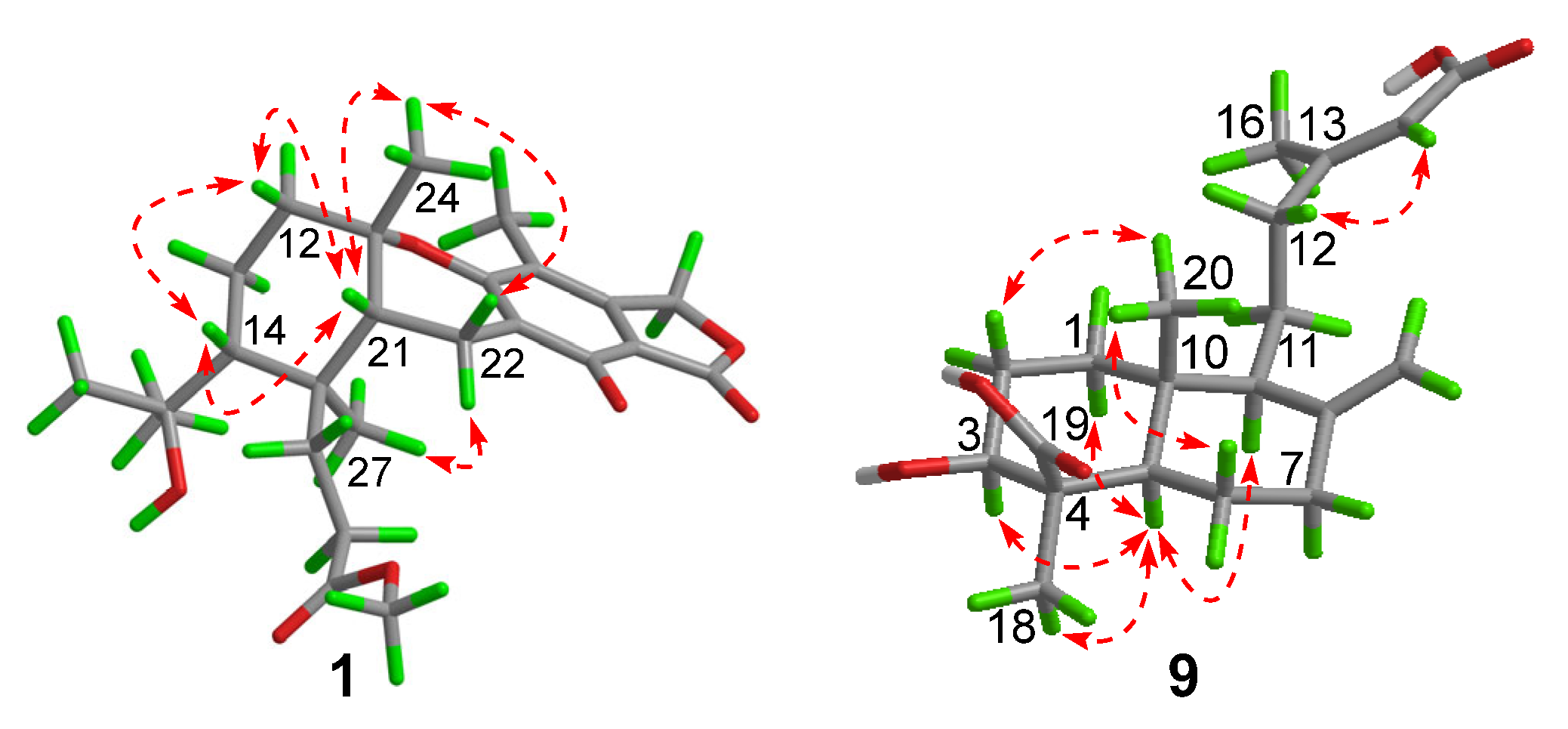
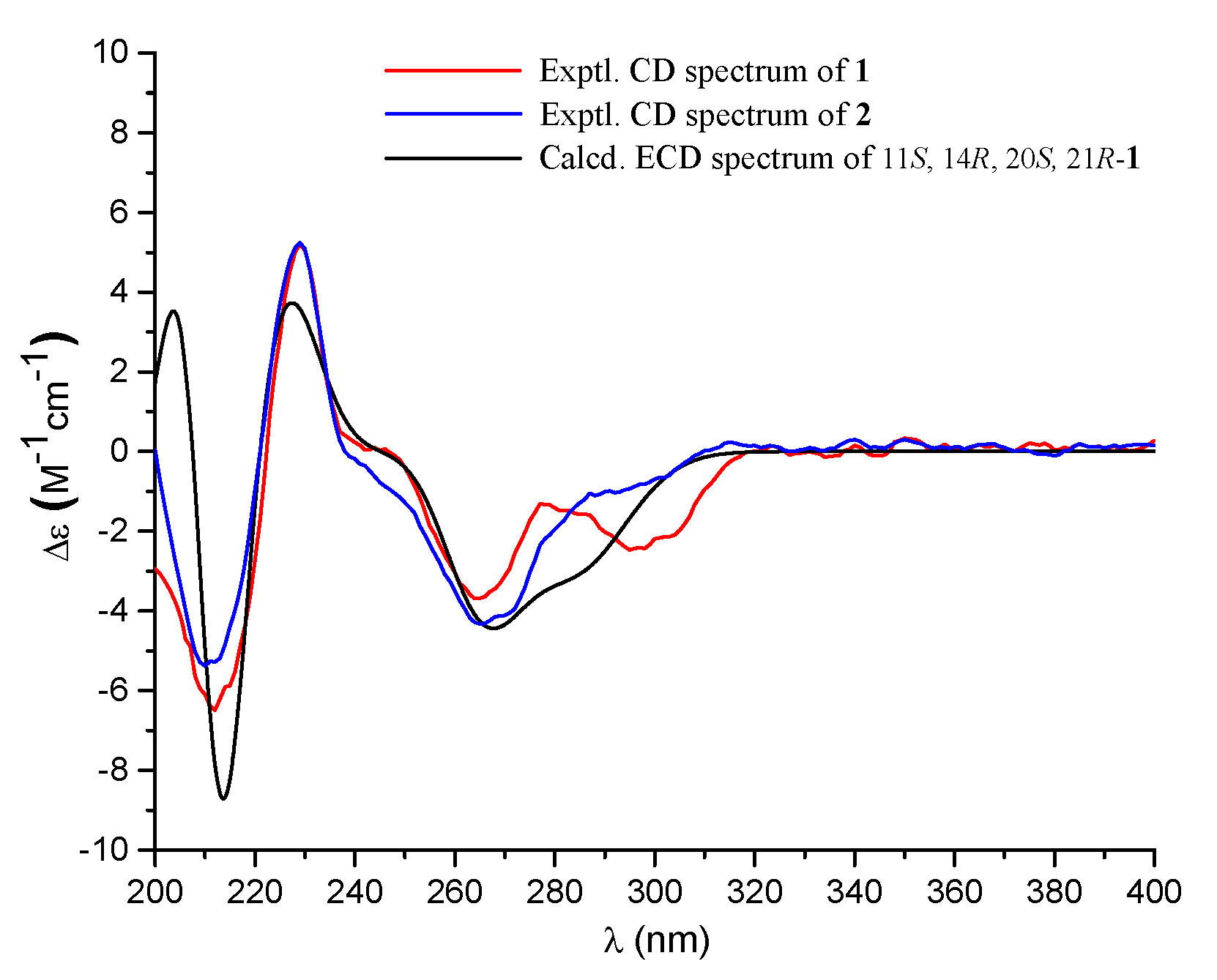
Compound 1 had a molecular formula of C25H34O7, as established by the high-resolution electrospray ionization mass spectroscopy (HRESIMS) (469.2202 [M + Na]+, calcd. for 469.2197) (see Figure S34 in the Supplementary Materials), requiring nine degrees of unsaturation. The 1H nuclear magnetic resonance (NMR) spectrum exhibited signals for five methyl singlets (δH 2.02, 1.28, 1.21 × 2, 0.71), a methoxy (δH 3.67), an oxygenated methylene (δH 5.22), and a series of alkyl protons (see Figure S1). The 13C NMR and the heteronuclear single-quantum coherence (HSQC) spectra exhibited 25 carbon resonances attributable to a benzene ring (δc 160.8, 153.8, 145.4, 112.3, 103.0, 111.2), five methyls (δc 33.2, 28.2, 27.7, 19.4, 10.6), a methoxy (δc 52.0), six methylenes (δc 70.6, 40.5, 22.6, 30.1, 34.9, 18.4) including an oxygenated one, two methines (δc 41.5, 52.1), two ester carbons (δc 174.2, 177.0), one sp3 quaternary carbon (δc 42.8), and two oxygenated carbons (δc 78.2, 75.8) (see Figures S2 and S3). As six degrees of unsaturation were accounted for by the benzene ring and two carbonyl carbons, the remaining three degrees of unsaturation required that compound 1 contained three additional rings. The aforementioned information was very similar to that of austalide P (2) [6], a co-isolated analogue first isolated from a sponge-associated fungus Aspergillus sp., with the only difference owing to the absence of the aromatic methoxy group (δH 4.04, δC 62.2) in 2. This indicated that 1 was the 5-demethoxylated derivative of 2. The structure of 1 was further certified as correct by detailed interpretation of 2D NMR data (Figure 2). The relative configuration of 1 was assigned by a nuclear Overhauser effect spectroscopy (NOESY) experiment (Figure 3). The NOE correlations of H-21 (δH 1.68)/H3-24 (δH 1.21), H-14 (δH 1.55), H3-24/H-22β (δH 2.69), and H3-27 (δH 0.71)/H-22α (δH 2.94) clarified the same orientation of H3-24, H-21, and H-14, while H3-27 was in the opposite orientation. Thus, the relative configuration of 1 was assigned as 11S∗, 14R∗, 20S∗, and 21R∗. In order to resolve its absolute configuration, the theoretical electronic circular dichroism (ECD) data of 11S, 14R, 20S, 21R-1 were calculated by the time-dependent density-functional theory (TDDFT) method and showed an ECD curve with Cotton effects at 265 (−), 227 (+), and 214 (−) nm, which were in good agreement with the experimental cotton effects at 264 (−), 229 (+), and 212 (−) nm (Figure 4), suggesting that compound 1 has the 11S, 14R, 20S, 21R configuration. Compound 1 was given the trivial name austalide Y and represented the third example of an austalide meroterpenoid without the 5-methoxy group.
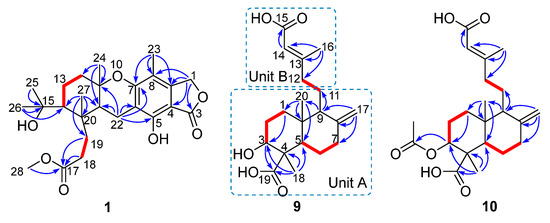
Figure 2.
Key correlation spectroscopy (COSY,  ) and heteronuclear multiple-bond correlations (HMBC,
) and heteronuclear multiple-bond correlations (HMBC,  ) of compounds 1, 9, and 10.
) of compounds 1, 9, and 10.
 ) and heteronuclear multiple-bond correlations (HMBC,
) and heteronuclear multiple-bond correlations (HMBC,  ) of compounds 1, 9, and 10.
) of compounds 1, 9, and 10.
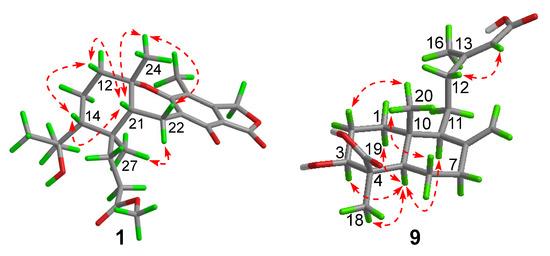
Figure 3.
Key nuclear Overhauser effect spectroscopy (NOESY) correlations ( ) of compounds 1 and 9.
) of compounds 1 and 9.
 ) of compounds 1 and 9.
) of compounds 1 and 9.
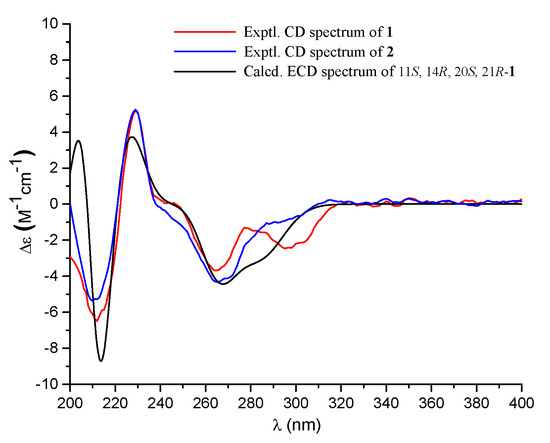
Figure 4.
Experimental electronic circular dichroism (ECD) spectra (200–400 nm) of compounds 1 and 2 in methanol and the calculated ECD spectrum of 11S, 14R, 20S, 21R-1 at the B3LYP/6-31+G(d,p) level in methanol.
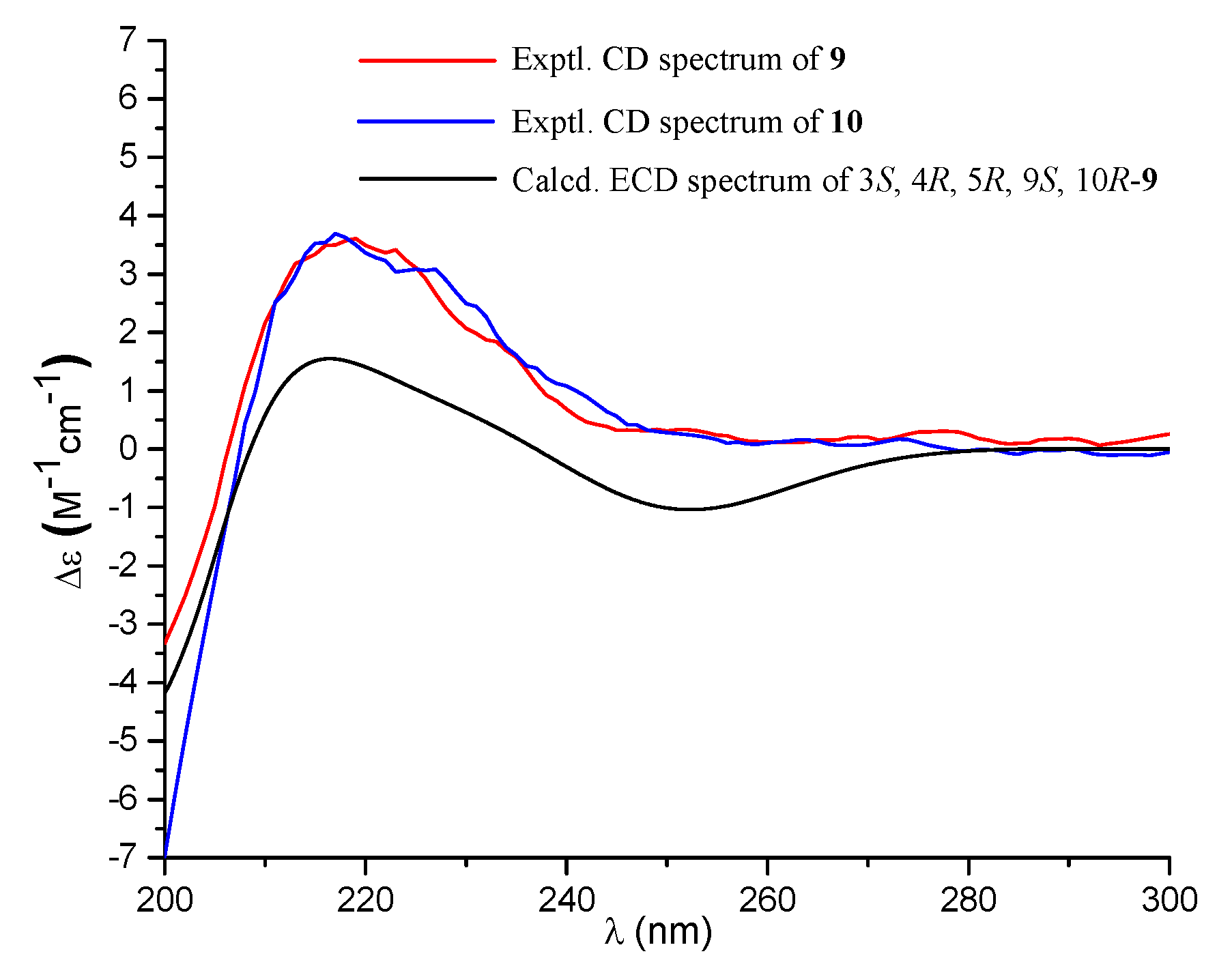
Compound 9 was isolated as a colorless oil with the molecular formula of C20H30O5 as determined by the HRESIMS at m/z 373.1991 [M + Na]+ (calcd. 373.1985), requiring six degrees of unsaturation. The 1H NMR spectrum exhibited signals for an olefinic methyl (δH 2.13), two methyl singlets (δH 1.38 and 0.69), an olefinic methylene (δH 4.56, 4.90), an oxygenated proton (δH 3.18), and several aliphatic protons. The 13C NMR spectra exhibited 20 carbon resonances, which were classified by an HSQC experiment as three methyls (δC 24.8, 18.9, 13.5), six sp3 methylenes (δC 40.7, 39.6, 38.7, 29.7, 27.2, 23.0), three methines (δC 79.0, 56.5 × 2) including one oxygenated methine, two sp3 quaternary carbons (δC 50.3, 41.0), two carboxylic acid groups (δC 170.4, 180.5), and two double bonds (δC 161.7, 149.0, 107.3, 116.9). As four of the six degrees of unsaturation were covered by two carboxylic acid groups and two double bonds, the remaining two degrees of unsaturation required that 9 was bicyclic. The gross structure was further established by detailed analyses of the 2D NMR data (Figure 2). The correlation spectroscopy (COSY) relationships from H2-1 to H-3 and H-5 to H2-7 coupled with the heteronuclear multiple-bond correlations (HMBCs) of H3-20 (δH 0.69) to C-1 (δC 38.7), C-5 (δC 56.5), C-10 (δC 41.0), H3-18 (δH 1.38) to C-3 (δC 79.0), C-4 (δC 50.3), C-5, C-19 (δC 180.5), the olefinic methylene protons H2-17 (δH 4.56, 4.90) to C-7 (δC 39.6), C-8 (δC 149.0), C-9 (δC 56.5), and H-9 (δH 1.61) to C-11 (δC 23.0), C-20 (δC 13.5) established a bicyclic unit (unit A). Additional HMBCs from H3-16 (δH 2.13) to C-12 (δC 40.7), C-13 (δC 161.7), C-14 (δC 116.9), and H-14 (δH 5.61) to C-15 (δH 170.4) established a senecioic acid moiety (unit B), which was linked to unit A at C-11 by the 1H-1H COSY relationship between H2-11 and H2-12. Thus, the gross structure of 9 was established as depicted. The relative configuration of 9 was determined by an NOESY experiment (Figure 3) and coupling constants. The coupling constants of JH-3/H-2α (4.4 Hz) and JH-3/H-2β (12.1 Hz) suggested that OH-3 was β-orientated. The NOE correlations of H-5 with H-3, H-9 (δH 1.61), H3-18 (δH 1.38), H3-18 with H-6α (δH 2.03), and H3-20 (δH 0.69) with H-6β (δH 1.93) indicated that H-3, H-5, and H-9 were α-orientated, while H3-20 was β-orientated. The NOE correlation between H-14 and H2-12 (δH 2.30, 2.02) was indicative of an E configuration of the double bond Δ13. Comparison of the experimental ECD data with those of the calculated ECD data at the B3LYP/6-31+G(d,p) level for 3S, 4R, 5R, 9S, 10R-9 allowed the assignment of the 3S, 4R, 5R, 9S, 10R configuration for 9 (Figure 5). As the structure of 9 was 3-hydroxylated derivative of agathic acid (10) [16], it was named 3β-hydroxy-agathic acid.
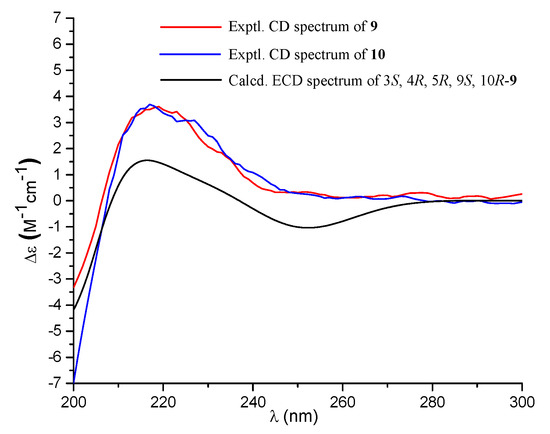
Figure 5.
Experimental ECD spectra (200–300 nm) of 9 and 10 in methanol and the calculated ECD spectrum of 3S, 4R, 5R, 9S, 10R-9 at the B3LYP/6-31+G(d,p) level in methanol.
Compound 10 had a molecular formula of C22H32O6, as determined by the HRESIMS at m/z 415.2096 [M + H]+ (calcd. 415.2091), requiring seven degrees of unsaturation. The NMR data of 10 were similar to those of 9 with the obvious distinction due to the presence of an acetyl group (δH 2.04, δC 172.8), suggesting that 10 was an acetylated derivative of 9. The downfield-shifted H-3 (δH 4.55) showed an HMBC with the acetyl carbonyl carbon (δC 172.8), locating the acetyl group at C-3 (Figure 2). The relative configuration of 10 was determined to be the same as that of 9 based on their similar NOESY data. The structure of 10 was determined as depicted and is a C-3 epimer of a known analogue mumic acid A [17]. The similar specific rotations and circular dichroism (CD) spectra of 9 and 10 confirmed the same absolute configuration of both 9 and 10 (Figure 5), and compound 10 was named 3β-acetoxy-agathic acid.
In addition, eight additional known compounds were identical to austalide P (2) [6], austalide H (3) [5], austalide P acid (4) [9], austalide H acid (5) [9], 17-O-demethylaustalide B (6) [9], austalide Q acid (7) [9], 13-deoxyaustalide Q acid (8) [9], and agathic acid (10) [16] based on comparisons of their NMR data and specific rotations with those reported in the literature. Furthermore, the 13C NMR data of austalide H (3) and 13C NMR data of compounds 3, 4, and 7 in methanol-d6 were reported for the first time.
All compounds were screened for their inhibitory activities against α-glucosidase at the initial concentration of 1 mM. Compounds 2 and 11 exhibited inhibition by more than 50% and were further evaluated to calculate the IC50 values. The results showed that compounds 2 and 11 inhibited α-glucosidase with IC50 values of 910 ± 4 and 525 ± 2 μM, being more active than the positive control acarbose (1.33 mM). Other compounds, on the other hand, showed inhibition less than 40% at the concentration of 1 mM. As for the labdane-type diterpenoids 9−11, the introduction of hydroxy and acetoxy groups at C-3 may lead to a sharp decrease in activity, since compounds 9 and 10 showed low inhibition when compared with that of 11.
The isolated compounds were also evaluated for their inhibitory effects against NO production in LPS-activated RAW 264.7 macrophages at the concentration of 50 μM following the same procedures in our previous study [10]. The cell viability was further determined by the MTT assay to evaluate whether the inhibition on NO production was owing to the cytotoxicity. As results (see Table S1 in the Supplementary Materials), compounds 1, 2, and 10, possessing inhibition rates of more than 50% on NO production, showed obvious cytotoxic effects, which suggested that the inhibitory effects of NO production were due to the cytotoxicity. All compounds were further evaluated for their cytotoxicity toward two types of human breast carcinoma cells (MCF-7, MDA-MB-468) [13], and the results showed that only compound 1 showed a weak inhibitory effect toward MDA-MB-468 cells with an IC50 value of 38.9 ± 1.83 μM.
3. Experimental Section
3.1. General Experimental Procedure
Specific rotations were recorded by an SGW®-1 automatic polarimeter (Shanghai Jing Ke Industrial Co., Ltd., Shanghai, China). The NMR spectra were measured on a Bruker Avance III HD-400 spectrometer (Bruker, Fällanden, Switzerland). HRESIMS spectra were obtained on a Waters Xevo G2 Q-TOF spectrometer (Waters Corporation, Milford, MA, USA). Semi-preparative high-performance liquid chromatography (HPLC) was undertaken on a Shimadzu LC-6AD pump (Shimadzu Co., Kyoto, Japan) using a UV detector, and a YMC-Pack ODS-A HPLC column (semipreparative, 250 × 10 mm, S-5 μm, 12 nm, YMC Co., Ltd, Kyoto, Japan) was used for separation.
3.2. Fungal Strain and Identification
Fungus YPGA3 was isolated from deep sea water at a depth of 4500 m in the Yap Trench (West Pacific Ocean). The strain was identified as Penicillium thomii based on microscopic examination and by internal transcribed spacer (ITS) sequencing. The ITS sequence was deposited in GenBank (http://www.ncbi.nlm.nih.gov) with accession number MG835903. The strain YPGA3 (MCCC 3A01052) was deposited at the Marine Culture Collection of China.
3.3. Fermentation
The fermentation was carried out in 30 Fernbach flasks (500 mL), each containing 70 g of rice. Artificial seawater (90 mL) was added to each flask, and the contents were soaked for three hours before autoclaving at 15 psi for 30 min. After cooling to room temperature, each flask was inoculated with 3.0 mL of the spore inoculum and incubated at room temperature for 30 days.
3.4. Extraction and Isolation
The fermented materials were extracted with ethyl acetate (EtOAc) (3 × 5000 mL) in an ultrasonic bath at 30 °C for 20 min. After evaporation under vacuum, the EtOAc extract (3.1g) was subjected to ODS silica gel column chromatography (CC) eluting with MeOH/H2O (20:80→100:0) to afford ten fractions (F1–F10). F5 was further chromatographed over C-18 silica gel CC eluted with MeOH/H2O (65:35) to afford seven subfractions (F5a–F5g). F5d was further purified by HPLC on a semi-preparative YMC-pack ODS-A column using CH3CN/H2O (61:39, 3 mL/min) to afford 5 (31 mg, tR 40 min). F6 was further chromatographed over ODS silica gel CC eluted with MeOH/H2O (20:80→100:0) to afford fourteen subfractions (F6a–F6n). F6d was separated by HPLC CH3CN/H2O (54:46, 3 mL/min) to yield 3 (14 mg, tR 19.9 min). Purification of F6e by HPLC using CH3CN/H2O (54:46, 3 mL/min) gave 7 (10 mg, tR 21.9 min). F6h was purified by HPLC using CH3CN/H2O (52:48, 3 mL/min) to give 9 (3 mg, tR 23.9 min). F6i was purified by HPLC using CH3CN/H2O (53:47, 3 mL/min) to afford 4 (5 mg, tR 49.6 min) and 10 (2 mg, tR 53.6 min). F6j was purified by HPLC using CH3CN/H2O (50:50, 3 mL/min) to afford 6 (2 mg, tR 22.5 min). F6k was purified by HPLC using CH3CN/H2O (65:35, 3 mL/min) to afford 1 (1 mg, tR 36.2 min) and 2 (6 mg, tR 39.0 min). F6l was purified by HPLC using CH3CN/H2O (68:32, 3 mL/min) to afford 11 (20 mg, tR 24.4 min). F6m was purified by HPLC using CH3CN/H2O (70:30, 3 mL/min) to afford 8 (1.5 mg, tR 48.9 min).
Austalide Y (1): colorless oil; −33 (c 0.05, MeOH); UV (MeOH) λmax 222, 270 nm; ECD (c 2.24 × 10−4 M, MeOH) λmax (Δε) 264 (−3.69), 229 (+5.21), 212 (−6.49); 1H and 13C NMR data, see Table 1; HRESIMS m/z 469.2202 [M + Na]+ (calcd. for C25H34O7Na+, 469.2197).

Table 1.
Nuclear magnetic resonance (NMR) data of 1, 3, 4, and 7 in Methanol-d4 a (δ in ppm).
3β-Hydroxy-agathic acid (9): colorless oil; +66 (c 0.06, MeOH); ECD (c 5.7 × 10−4 M, MeOH) λmax 217 (4.05); 1H and 13C NMR data, see Table 2; HRESIMS m/z 373.1991 [M + Na]+ (calcd. for C20H30O5Na+, 373.1985).

Table 2.
1H and 13C NMR Data of 9 and 10 in Methanol-d4 a (δ in ppm).
3β-Acetoxy-agathic acid (10): colorless oil; +58 (c 0.03, MeOH); ECD (c 5.1 × 10−4 M, MeOH) λmax 225 (3.78); 1H and 13C NMR data, see Table 2; HRESIMS m/z 415.2096 [M + Na]+ (calcd. for C22H32O6Na+, 415.2091).
3.5. α-Glucosidase Assay
The α-glucosidase inhibitory effect was assessed as follows. First, 0.2 U of α-glucosidase from Saccharomyces cerevisiae was purchased from Sigma-Aldrich (St. Louis, MO, USA), and was diluted in a 0.067 M phosphate buffer consisting of Na2HPO4·12H2O and KH2PO4 (pH 6.8). The assay was conducted in a 60 μL reaction system containing 20 μL of diluted enzyme solution, and 20μL of dimethyl sulfoxide (DMSO) or sample (dissolved in DMSO). After 10 min of incubation in 96-well plates at 37 °C, a 20 μL portion of 4 mM 4-nitrophenyl-α-d-glucopyranoside (PNPG) (Aladdin, Shanghai, China) was added as a substrate to start the enzymatic reaction. The plate was incubated for an additional 20 min at 37 °C, and the reaction was quenched by adding 60 μL of 0.2 M Na2CO3. The final concentrations of tested compounds were between 0.2 and 2 mM. The optical density (OD) was measured at an absorbance wavelength of 405 nm using a Microplate Reader (Tecan, Mannedorf, Switzerland). All assays were performed in three replicates, and acarbose (Aladdin, Shanghai, China) was used as the positive control.
4. Conclusions
In conclusion, a new austalide meroterpenoid (1) and seven known austalide analogues (2−8), two new labdane-type diterpenes (9 and 10) and one known derivative (11) were isolated from a fraction of the EtOAc extract of the deep-sea derived strain Penicillium thomii YPGA3. The structures of compounds 1, 9, and 10 were determined by comprehensive analyses of the NMR and mass spectroscopy (MS) data, the absolute configurations of 1 and 9 were determined by ECD calculations. Compound 1 showed weak inhibition toward MDA-MB-468 cells with an IC50 value of 38.9 μM. Compounds 2 and 11 exhibited inhibitory effects against α-glucosidase with IC50 values of 910 and 525 μM, respectively, being more active than the positive control acarbose (1.33 mM).
Supplementary Materials
The following are available online at https://www.mdpi.com/1660-3397/18/3/164/s1, Figures S1–S37: 1H, 13C NMR, HSQC, 1H-1H COSY, HMBC, HRESIMS spectra of the new compounds 1, 9, and 10, 1H and 13C NMR of known compounds 2−8, and 11, and details for ECD calculations.
Author Contributions
Z.C., W.L., Y.L., and S.H. isolated the compounds and elucidated the structures. W.L., X.C., and J.Z. tested the inhibition against α-glucosidase. R.F., Y.W., and Y.Z. tested the inhibition against LPS-induced NO production in RAW 264.7 macrophage cells. R.F. conducted the MTT assay. X.L., S.A.A., W.X., and Z.L. isolated and identified the fungus, and conducted the solid fermentation of the fungus. Z.C. and Q.L. designed the study and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81903536), the National Key R&D Program of China (2017YFD0600702-2), the China Ocean Mineral Resources R&D Association Program (DY135-B2-01 and DY135-B2-09), the Youth Talent Promotion Project of Henan Province (2020HYTP059), the First-Class Discipline Construction Project of Henan University (2018YLZDCG03), China-ASEAN Maritime Cooperation Fund Project “Monitoring and conservation of the coastal ecosystem in the South China Sea”, and Bilateral Cooperation of Maritime Affairs.
Acknowledgments
The authors thank Jianxun Kang at Zhengzhou University for NMR measurements.
Conflicts of Interest
The authors declare no conflict of interest.
References
- de Jesus, A.E.; Horak, R.M.; Steyn, P.S.; Vleggaar, R. Biosynthesis of austalide D, a meroterpenoid mycotoxin from Aspergillus ustus. J. Chem. Soc. Chem. Commun. 1983, 13, 716–718. [Google Scholar] [CrossRef]
- Horak, R.M.; Steyn, P.S.; Van Rooyen, P.H.; Vleggaar, R.; Rabie, C.J. Structures of the austalides A–E, five noval toxic metabolites from Aspergillus ustus. J. Chem. Soc. Chem. Commun. 1981, 24, 1265–1267. [Google Scholar] [CrossRef]
- Horak, R.M.; Steyn, P.S.; Vleggaar, R.; Rabie, C.J. Metabolites of Aspergillus ustus. Part 1. A of the heteronuclear selective population inversion (SPI) n.m.r. technique to the structure elucidation of the austalides A–F, novel ortho ester meroterpenoids. J. Chem. Soc.-Perkin Trans. 1 1985, 345–356. [Google Scholar] [CrossRef]
- Horak, R.M.; Steyn, P.S.; Vleggaar, R. Metabolites of Aspergillus ustus. Part 2. Stereoelectronic control in the acid-catalysed hydrolysis of the ortho ester moiety in austalides A–F. J. Chem. Soc.-Perkin Trans. 1 1985, 357–361. [Google Scholar] [CrossRef]
- Horak, R.M.; Steyn, P.S.; Vleggaar, R.; Rabie, C.J. Metabolites of Aspergillus ustus. Part 3. Structure elucidation of austalides G–L. J. Chem. Soc.-Perkin Trans. 1 1985, 363–367. [Google Scholar] [CrossRef]
- Zhou, Y.; Mándi, A.; Debbab, A.; Wray, V.; Schulz, B.; Müller, W.E.G.; Lin, W.; Proksch, P.; Kurtán, T.; Aly, A.H. New austalides from the sponge-associated fungus Aspergillus sp. Eur. J. Org. Chem. 2011, 2011, 6009–6019. [Google Scholar] [CrossRef]
- Zhou, Y.; Debbab, A.; Wray, V.; Lin, W.; Schulz, B.; Trepos, R.; Pile, C.; Hellio, C.; Proksch, P.; Aly, A.H. Marine bacterial inhibitors from the sponge-derived fungus Aspergillus sp. Tetrahedron Lett. 2014, 55, 2789–2792. [Google Scholar] [CrossRef]
- Peng, J.; Zhang, X.; Wang, W.; Zhu, T.; Gu, Q.; Li, D. Austalides S–U, New meroterpenoids from the sponge-derived fungus Aspergillus aureolatus HDN14-107. Mar. Drugs 2016, 14, 131. [Google Scholar] [CrossRef] [PubMed]
- Zhuravleva, O.I.; Sobolevskaya, M.P.; Leshchenko, E.V.; Kirichuk, N.N.; Denisenko, V.A.; Dmitrenok, P.S.; Dyshlovoy, S.A.; Zakharenko, A.M.; Kim, N.Y.; Afiyatullov, S.S. Meroterpenoids from the alga-derived fungi Penicillium thomii Maire and Penicillium lividum Westling. J. Nat. Prod. 2014, 77, 1390–1395. [Google Scholar] [CrossRef] [PubMed]
- Antipova, T.V.; Zaitsev, K.V.; Oprunenko, Y.F.; Zherebker, A.Y.; Rystsov, G.K.; Zemskova, M.Y.; Zhelifonova, V.P.; Ivanushkina, N.E.; Kozlovsky, A.G. Austalides V and W, new meroterpenoids from the fungus Aspergillus ustus and their antitumor activities. Bioorg. Med. Chem. Lett. 2019, 29, 126708. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Lee, J.; Kim, K.-J.; Sung, Y.; Park, K.-H.; Oh, E.; Park, C.; Son, Y.-J.; Kang, H. Austalides, osteoclast differentiation inhibitors from a marine-derived strain of the fungus Penicillium rudallense. J. Nat. Prod. 2019, 82, 3083–3088. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Xu, W.; Wang, Y.; Bai, S.; Liu, L.; Luo, Z.; Yuan, W.; Li, Q. Two new meroterpenoids and two new monoterpenoids from the deep sea-derived fungus Penicillium sp. YPGA11. Fitoterapia 2019, 133, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Li, Y.; Xu, W.; Liu, W.; Liu, L.; Zhu, D.; Kang, Y.; Luo, Z.; Li, Q. Three new cyclopiane-type diterpenes from a deep-sea derived fungus Penicillium sp. YPGA11 and their effects against human esophageal carcinoma cells. Bioorganic Chem. 2019, 91, 103129. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Li, Y.; Liu, W.; Liu, L.; Liu, J.; Yuan, W.; Luo, Z.; Xu, W.; Li, Q. Butenolide Derivatives with α-glucosidase inhibitions from the deep-sea-derived fungus Aspergillus terreus YPGA10. Mar. Drugs 2019, 17, 332. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Xu, W.; Liu, L.; Li, S.; Yuan, W.; Luo, Z.; Zhang, J.; Cheng, Y.; Li, Q. Peniginsengins B–E, New farnesylcyclohexenones from the deep sea-derived fungus Penicillium sp. YPGA11. Mar. Drugs 2018, 16, 358. [Google Scholar] [CrossRef] [PubMed]
- Jim-Min, F.; Yi-Chien, S.; Yu-Hung, C.; Yu-Shia, C. Diterpenes from the bark of Juniperus chinensis. Phytochemistry 1993, 34, 1581–1584. [Google Scholar] [CrossRef]
- Kiren, Y.; Nugroho, A.E.; Hirasawa, Y.; Shirota, O.; Bekenova, M.; Narbekovich, N.O.; Shapilova, M.; Maeno, H.; Morita, H. Mumic acids A–E: New diterpenoids from mumiyo. J. Nat. Med. 2014, 68, 199–205. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).