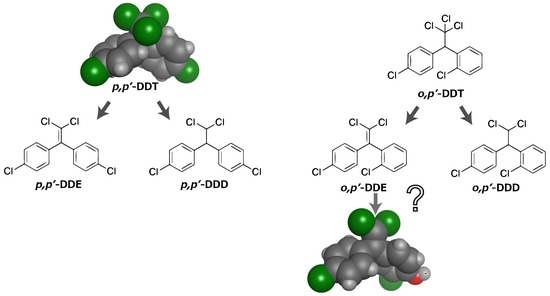
A Novel Action of Endocrine-Disrupting Chemicals on Wildlife; DDT and Its Derivatives Have Remained in the Environment
Abstract
Share and Cite
Matsushima, A. A Novel Action of Endocrine-Disrupting Chemicals on Wildlife; DDT and Its Derivatives Have Remained in the Environment. Int. J. Mol. Sci. 2018, 19, 1377. https://doi.org/10.3390/ijms19051377
Matsushima A. A Novel Action of Endocrine-Disrupting Chemicals on Wildlife; DDT and Its Derivatives Have Remained in the Environment. International Journal of Molecular Sciences. 2018; 19(5):1377. https://doi.org/10.3390/ijms19051377
Chicago/Turabian StyleMatsushima, Ayami. 2018. "A Novel Action of Endocrine-Disrupting Chemicals on Wildlife; DDT and Its Derivatives Have Remained in the Environment" International Journal of Molecular Sciences 19, no. 5: 1377. https://doi.org/10.3390/ijms19051377
APA StyleMatsushima, A. (2018). A Novel Action of Endocrine-Disrupting Chemicals on Wildlife; DDT and Its Derivatives Have Remained in the Environment. International Journal of Molecular Sciences, 19(5), 1377. https://doi.org/10.3390/ijms19051377