Ultrasonographic Assessment of Achilles Tendon Thickness in Pediatric Patients with Suspected Familial Hypercholesterolemia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Ethical Considerations
2.3. Study Population and Selection Criteria
2.4. Genetic Testing
2.5. Measurement of AT Thickness by Ultrasonography
2.6. Treatment Information
2.7. Statistical Analysis
3. Results
3.1. Study Population and Patient Selection
3.2. Comparison of AT Thickness Between FH and Non-FH Groups
3.3. Association Between Cumulative LDL-C and AT Thickness
4. Discussion
4.1. Interpretation of Main Findings
4.2. Population-Specific Considerations
4.3. Study Limitations
4.3.1. Single-Center Design and Selection Bias
4.3.2. Statistical Power and Sample Size
4.3.3. Variability in Statin Treatment
4.3.4. Pediatric Population Heterogeneity
4.4. Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FH | Familial Hypercholesterolemia |
| AT | Achilles Tendon |
| LDL-C | Low-Density Lipoprotein Cholesterol |
| HDL-C | High-Density Lipoprotein Cholesterol |
| TG | Triglycerides |
| TC | Total Cholesterol |
| IQR | Interquartile Range |
| NGS | Next Generation Sequencing |
| MLPA | Multiplex Ligation-dependent Probe Amplification |
| JAS | Japan Atherosclerosis Society |
| BMI | Body Mass Index |
References
- Fourgeaud, M.; Lebreton, L.; Belabbas, K.; Zaouiche, S.; Fouache, E.; Carreau, V.; Antigny, F.; Benlian, P.; Bruckert, E.; Farnier, M.; et al. Phenotypic and genotypic characterization of familial hypercholesterolemia in French adult and pediatric populations. J. Clin. Lipidol. 2022, 16, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Dharmayat, K.I.; Stevens, C.A.T.; Sharabiani, M.T.A.; Jones, R.S.; Watts, G.F.; Genest, J.; Ray, K.K.; Vallejo-Vaz, A.J. Prevalence of Familial Hypercholesterolemia among the General Population and Patients with Atherosclerotic Cardiovascular Disease: A Systematic Review and Meta-Analysis. Circulation 2020, 141, 1742–1759. [Google Scholar] [CrossRef] [PubMed]
- Wiegman, A.; Gidding, S.S.; Watts, G.F.; Chapman, M.J.; Ginsberg, H.N.; Cuchel, M.; Ose, L.; Averna, M.; Boileau, C.; Borén, J.; et al. Familial hypercholesterolaemia in children and adolescents: Gaining decades of life by optimizing detection and treatment. Eur. Heart J. 2015, 36, 2425–2437. [Google Scholar] [CrossRef] [PubMed]
- Harada-Shiba, M.; Arai, H.; Ishigaki, Y.; Ishibashi, S.; Okamura, T.; Ogura, M.; Dobashi, K.; Nohara, A.; Bujo, H.; Miyauchi, K.; et al. Guidelines for diagnosis and treatment of familial hypercholesterolemia 2017. J. Atheroscler. Thromb. 2018, 25, 751–770. [Google Scholar] [CrossRef] [PubMed]
- Tsouli, S.G.; Kiortsis, D.N.; Argyropoulou, M.I.; Mikhailidis, D.P.; Elisaf, M.S. Pathogenesis, detection and treatment of Achilles tendon xanthomas. Eur. J. Clin. Investig. 2005, 35, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Harada-Shiba, M.; Arai, H.; Ohmura, H.; Okazaki, H.; Sugiyama, D.; Tada, H.; Dobashi, K.; Matsuki, K.; Minamino, T.; Yamashita, S.; et al. Guidelines for the diagnosis and treatment of adult familial hypercholesterolemia 2022. J. Atheroscler. Thromb. 2023, 30, 558–586. [Google Scholar] [CrossRef]
- Michikura, M.; Ogura, M.; Hori, M.; Matsuki, K.; Makino, H.; Fujioka, S.; Shishikura, D.; Hoshiga, M.; Harada-Shiba, M.; Ogawa, M.; et al. Association of Achilles tendon thickness with lipid profile and carotid IMT in patients with familial hypercholesterolemia. Atherosclerosis 2025, 403, 119173. [Google Scholar] [CrossRef]
- Nordestgaard, B.G.; Chapman, M.J.; Humphries, S.E.; Ginsberg, H.N.; Masana, L.; Descamps, O.S.; Wiklund, O.; Hegele, R.A.; Raal, F.J.; Defesche, J.C.; et al. Familial hypercholesterolaemia is underdiagnosed and undertreated in the general population: Guidance for clinicians to prevent coronary heart disease: Consensus statement of the European Atherosclerosis Society. Eur. Heart J. 2013, 34, 3478–3490. [Google Scholar] [CrossRef] [PubMed]
- Harada-Shiba, M.; Ohtake, A.; Sugiyama, D.; Tada, H.; Dobashi, K.; Matsuki, K.; Minamino, T.; Yamashita, S.; Yamamoto, Y. Guidelines for the diagnosis and treatment of pediatric familial hypercholesterolemia 2022. J. Atheroscler. Thromb. 2023, 30, 531–557. [Google Scholar] [CrossRef] [PubMed]
- Cuchel, M.; Raal, F.J.; Hegele, R.A.; Al-Rasadi, K.; Arca, M.; Averna, M.; Bruckert, E.; Freiberger, T.; Gaudet, D.; Harada-Shiba, M.; et al. 2023 Update on European Atherosclerosis Society Consensus Statement on Homozygous Familial Hypercholesterolaemia: New treatments and clinical guidance. Eur. Heart J. 2023, 44, 2277–2291. [Google Scholar] [CrossRef] [PubMed]
- Cuchel, M.; Bruckert, E.; Ginsberg, H.N.; Raal, F.J.; Santos, R.D.; Hegele, R.A.; Kuivenhoven, J.A.; Nordestgaard, B.G.; Descamps, O.S.; Steinhagen-Thiessen, E.; et al. Homozygous familial hypercholesterolaemia: New insights and guidance for clinicians to improve detection and clinical management. Eur. Heart J. 2014, 35, 2146–2157. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.Y.; Matsunaga, K.; Inoue, T.; Tani, R.; Funatsuki, K.; Iwase, T.; Kondo, S.; Nishioka, K.; Ito, S.; Sasaki, T.; et al. Improved efficiency of the clinical diagnostic criteria for familial hypercholesterolemia in children: A comparison of the Japan Atherosclerosis Society guidelines of 2017 and 2022. J. Atheroscler. Thromb. 2024, 31, 1048–1057. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, K.; Mizobuchi, A.; Fu, H.Y.; Ishikawa, S.; Tada, H.; Kawashiri, M.-A.; Yokota, I.; Sasaki, T.; Ito, S.; Kunikata, J.; et al. Universal screening for familial hypercholesterolemia in children in Kagawa, Japan. J. Atheroscler. Thromb. 2022, 29, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Strouse, P.J.; Trout, A.T.; Offiah, A.C. Editors’ notebook: What is ‘pediatric’? Pediatr. Radiol. 2022, 52, 2241–2242. [Google Scholar] [CrossRef] [PubMed]
- Bujo, H.; Takahashi, K.; Saito, Y.; Maruyama, T.; Yamashita, S.; Matsuzawa, Y.; Ishibashi, S.; Shionoiri, F.; Yamada, N.; Kita, T. Multicenter study to determine the diagnostic criteria for heterozygous familial hypercholesterolemia in Japan. J. Atheroscler. Thromb. 2004, 11, 146–151. [Google Scholar] [CrossRef]
- Kagawa Prefecture Health and Welfare Department. Kagawa Health Checkups for the Prevention of Lifestyle-Related Diseases in Children: Manual. Revised March 2021. Available online: https://www.pref.kagawa.lg.jp/kenkosomu/seikatsushukanbyo/manual2021.html (accessed on 26 August 2025).
- Mabuchi, H.; Nohara, A.; Noguchi, T.; Kobayashi, J.; Kawashiri, M.A.; Tada, H.; Nakanishi, C.; Mori, M.; Yamagishi, M.; Inazu, A.; et al. Molecular genetic epidemiology of homozygous familial hypercholesterolemia in the Hokuriku district of Japan. Atherosclerosis 2011, 214, 404–407. [Google Scholar] [CrossRef]
- Tada, H.; Kawashiri, M.-A.; Nomura, A.; Teramoto, R.; Hosomichi, K.; Nohara, A.; Inazu, A.; Mabuchi, H.; Tajima, A.; Yamagishi, M.; et al. Oligogenic familial hypercholesterolemia, LDL cholesterol, and coronary artery disease. J. Clin. Lipidol. 2018, 12, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Shimojima, K.; Ondo, Y.; Imai, K.; Chong, P.F.; Kira, R.; Amemiya, M.; Saito, A.; Okamoto, N. Challenges in detecting genomic copy number aberrations using next-generation sequencing data and the eXome Hidden Markov Model: A clinical exome-first diagnostic approach. Hum. Genome Var. 2016, 3, 16025. [Google Scholar] [CrossRef] [PubMed]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and Guidelines for the Interpretation of Sequence Variants: A Joint Consensus Recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 2015, 17, 405–424. [Google Scholar] [CrossRef]
- Tada, H.; Nomura, A.; Yoshimura, K.; Kawashiri, M.-A.; Takamura, M.; Nohara, A.; Harada-Shiba, M.; Yamagishi, M. Genetic Testing for Familial Hypercholesterolemia in the General Population: A Nationwide Study in Japan. J. Clin. Lipidol. 2024, 18, 123–132. [Google Scholar] [CrossRef]
- Benn, M.; Watts, G.F.; Tybjærg-Hansen, A.; Nordestgaard, B.G. Mutations Causative of Familial Hypercholesterolaemia: Screening of 98,098 Individuals from the Copenhagen General Population Study Estimated a Prevalence of 1 in 217. Eur. Heart J. 2016, 37, 1384–1394. [Google Scholar] [CrossRef]
- Michikura, M.; Ogura, M.; Yamamoto, M.; Sekimoto, M.; Fuke, C.; Hori, M.; Arai, K.; Kihara, S.; Hosoda, K.; Yanagi, K.; et al. Achilles tendon ultrasonography for diagnosis of familial hypercholesterolemia among Japanese subjects. Circ. J. 2017, 81, 1879–1885. [Google Scholar] [CrossRef] [PubMed]
- Michikura, M.; Ogura, M.; Matsuki, K.; Yamaoka, M.; Makino, H.; Harada-Shiba, M. Risk assessment for cardiovascular events using Achilles tendon thickness and softness and intima-media thickness in familial hypercholesterolemia. J. Atheroscler. Thromb. 2024, 31, 1607–1619. [Google Scholar] [CrossRef] [PubMed]
- Dobashi, K. Changes in serum cholesterol in childhood and its tracking to adulthood. J. Atheroscler. Thromb. 2022, 29, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Banderali, G.; Capra, M.E.; Biasucci, G.; Stracquadaino, R.; Viggiano, C.; Pederiva, C. Detecting familial hypercholesterolemia in children and adolescents: Potential and challenges. Ital. J. Pediatr. 2022, 48, 115. [Google Scholar] [CrossRef] [PubMed]
- Gidding, S.S.; Champagne, M.A.; de Ferranti, S.D.; Defesche, J.; Ito, M.K.; Knowles, J.W.; McCrindle, B.; Raal, F.; Rader, D.; Santos, R.D.; et al. The Agenda for Familial Hypercholesterolemia: A Scientific Statement From the American Heart Association. J. Am. Coll. Cardiol. 2020, 76, 2431–2455. [Google Scholar] [CrossRef] [PubMed]
- Horton, A.E.; Martin, A.C.; Srinivasan, S.; Justo, R.N.; Poplawski, N.K.; Sullivan, D.; Brett, T.; Chow, C.K.; Nicholls, S.J.; Pang, J.; et al. Integrated guidance to enhance the care of children and adolescents with familial hypercholesterolaemia: Practical advice for the community clinician. J. Paediatr. Child Health 2022, 58, 1297–1312. [Google Scholar] [CrossRef] [PubMed]
- Cassel, M.; Intziegianni, K.; Risch, L.; Müller, S.; Engel, T.; Mayer, F. Physiological tendon thickness adaptation in adolescent elite athletes: A longitudinal study. Front. Physiol. 2017, 8, 795. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, M.D.; Bhatt, D.L. “Cholesterol-Years” for ASCVD Risk Prediction and Treatment. J. Am. Coll. Cardiol. 2020, 76, 1517–1520. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Liang, J.; Zeng, D.; Tan, W.; Yang, L.; Lu, S.; Yao, W.; Yang, Y.; Liu, L. Association of body composition with pubertal timing in children and adolescents from Guangzhou, China. Front. Public Health 2022, 10, 943886. [Google Scholar] [CrossRef] [PubMed]
- Drole Torkar, A.; Plesnik, E.; Groselj, U.; Battelino, T.; Kotnik, P. Carotid intima-media thickness in healthy children and adolescents: Normative data and systematic literature review. Front. Cardiovasc. Med. 2020, 7, 597768. [Google Scholar] [CrossRef] [PubMed]
- Hodges, J.L.; Lehmann, E.L. The efficiency of some nonparametric competitors of the t-test. Ann. Math. Stat. 1956, 27, 324–335. [Google Scholar] [CrossRef]
- Chen, Z.; Lu, G. Lowering low-density lipoprotein cholesterol targets to below 1.0 mmol/L in acute coronary syndrome patients: A potential new standard. Cardiol. Plus 2024, 9, 10159. [Google Scholar] [CrossRef]

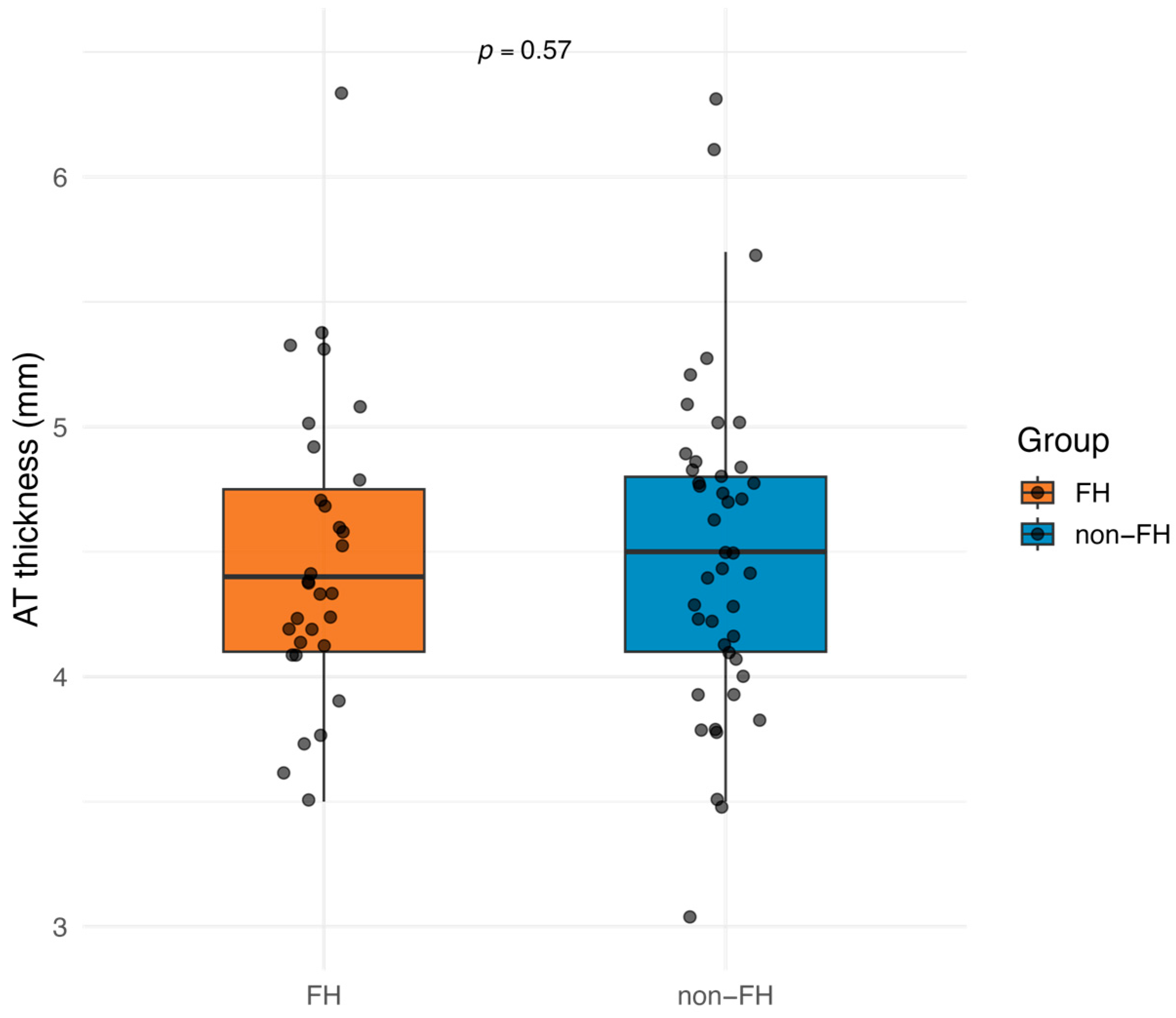
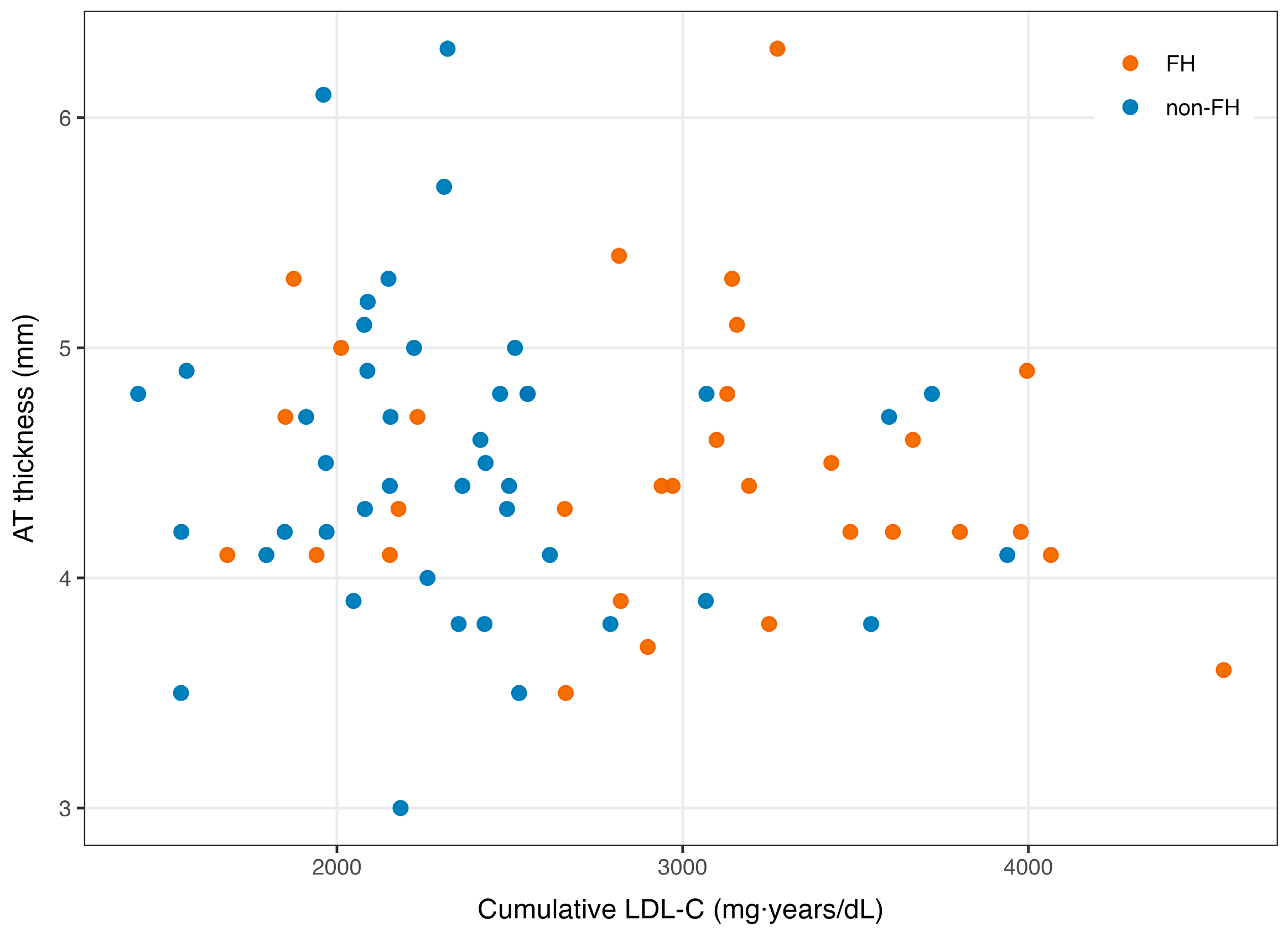
| All, n = 74 | FH, n = 31 | non-FH, n = 43 | p-Value | |
|---|---|---|---|---|
| Age (measured AT thickness), IQR | 13 (12–16) | 14 (12–16) | 13 (12–16) | 0.748 |
| Sex, Male (%) | 38 (51) | 18 (58) | 20 (47) | 0.327 |
| Height (cm), IQR | 155.3 (148.8–165.5) | 159 (150.2–168.9) | 153.0 (148.0–161.0) | 0.124 |
| Weight (kg), IQR | 48.7 (41.3–54.4) | 48.2 (39.7–56.8) | 48.8 (41.6–54.0) | 0.852 |
| BMI, IQR | 19.5 (17.6–21.7) | 18.5 (16.7–20.7) | 20.2 (18.4–22.0) | 0.017 |
| Lipids Profile | ||||
| Max LDL-C (mg/dL), IQR | 185 (164–208) | 202 (180–228) | 173 (160–192) | <0.001 |
| Cumulative LDL-C (mg·years/dL), IQR | 2482 (2088–3121) | 3098 (2446–3457) | 2310 (2064–2521) | <0.001 |
| HDL-C (mg/dL), IQR | 55 (46–65) | 58 (48–67) | 53 (44–62) | 0.313 |
| Triglyceride (mg/dL), IQR | 93 (57–141) | 91 (49–133) | 94 (52–136) | 0.634 |
| TC (mg/dL), IQR | 234 (216–258) | 239 (215–263) | 231 (209–253) | 0.237 |
| Statin treatment, n (%) | 35 (47) | 24 (77) | 11 (26) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inoue, T.; Matsunaga, K.; Tani, R.; Nishioka, K.; Kondo, S.; Iwase, T.; Fu, H.Y.; Tada, H.; Takamura, M.; Kusaka, T.; et al. Ultrasonographic Assessment of Achilles Tendon Thickness in Pediatric Patients with Suspected Familial Hypercholesterolemia. Lipidology 2025, 2, 15. https://doi.org/10.3390/lipidology2030015
Inoue T, Matsunaga K, Tani R, Nishioka K, Kondo S, Iwase T, Fu HY, Tada H, Takamura M, Kusaka T, et al. Ultrasonographic Assessment of Achilles Tendon Thickness in Pediatric Patients with Suspected Familial Hypercholesterolemia. Lipidology. 2025; 2(3):15. https://doi.org/10.3390/lipidology2030015
Chicago/Turabian StyleInoue, Tomoko, Keiji Matsunaga, Ryosuke Tani, Katsufumi Nishioka, Sonoko Kondo, Takashi Iwase, Hai Ying Fu, Hayato Tada, Masayuki Takamura, Takashi Kusaka, and et al. 2025. "Ultrasonographic Assessment of Achilles Tendon Thickness in Pediatric Patients with Suspected Familial Hypercholesterolemia" Lipidology 2, no. 3: 15. https://doi.org/10.3390/lipidology2030015
APA StyleInoue, T., Matsunaga, K., Tani, R., Nishioka, K., Kondo, S., Iwase, T., Fu, H. Y., Tada, H., Takamura, M., Kusaka, T., & Minamino, T. (2025). Ultrasonographic Assessment of Achilles Tendon Thickness in Pediatric Patients with Suspected Familial Hypercholesterolemia. Lipidology, 2(3), 15. https://doi.org/10.3390/lipidology2030015







