Abstract
The sarco(endo)plasmic reticulum Ca2+ ATPase (SERCA) actively transports Ca2+ into the sarcoplasmic reticulum to facilitate cardiac muscle relaxation. Phospholamban (PLN) allosterically inhibits SERCA, and an imbalance of SERCA2a, dominant cardiac isoform, and PLN content disrupts Ca2+ homeostasis and cardiac contractility. A previous study has shown that ovariectomized (OVX) rats have reduced SERCA activity due to lowered SERCA2a and increased PLN content. Furthermore, it was found that forced treadmill running in OVX rats restored SERCA activity and protein content levels. Here, we investigated whether voluntary wheel running (VWR) would produce similar effects on cardiac SERCA function in OVX mice. Female mice were divided into the following groups for 8 weeks: SHAM; OVX; SHAM + VWR; and OVX + VWR (n = 10/group). SERCA activity and Ca2+ uptake assays were performed in cardiac muscle homogenates. Protein levels of SERCA2, PLN, and pPLN were determined via Western blot analysis. We found statistical interactions for Ca2+ uptake, maximal SERCA activity, and SERCA2a content where VWR increased these parameters in SHAM mice but not in OVX mice. We detected a main effect of OVX on PLN content, and main effects of OVX and VWR on pPLN content. The OVX mice ran significantly less than the SHAM mice, suggesting that estrogen deprivation and lack of regular exercise may blunt the effects of voluntary aerobic exercise on cardiac SERCA function.
1. Introduction
Cardiovascular disease (CVD) is the leading cause of morbidity and mortality in women and is responsible for approximately one in three female deaths per year [1,2,3]. Previous studies suggest that during their reproductive years, females have a lower risk for CVD compared to age-matched males and typically develop CVD 7–10 years later than males [4,5,6]. However, this advantage disappears following menopause as the risk for CVD increases substantially [1,2,4,6]. Menopause and the deprivation of sex hormones such as estrogen is associated with the presentation of CVD [1]. The dramatic decline in estrogen levels during the menopausal transition coincides with increased CVD incidence and risk for disease [1,7,8]. Previous studies have reported significantly more cardiovascular events in postmenopausal women compared to age-matched premenopausal women, further suggesting that estrogen deficiency is a leading determinant of increased CVD risk [2,5,7,9].
Results from previous studies suggest that long-term estrogen deficiency damages cardiac Ca2+-handling proteins and intracellular Ca2+ homeostasis, leading to systolic and diastolic dysfunction [2,10]. The sarco(endo)plasmic reticulum Ca2+-ATPase (SERCA) pump is the primary enzyme involved in regulating intracellular Ca2+ homeostasis and cardiac contractility [1,11,12,13]. SERCA is a P-type ATPase that catalyzes the active transport of Ca2+ ions from the cytosol into the sarcoplasmic reticulum (SR), which initiates muscle relaxation and ensures a sufficient Ca2+ load for subsequent contractions [10]. In the heart, SERCA2a is the most abundant isoform and is responsible for 70% of cytosolic Ca2+ removal in human hearts and 92% in rodent hearts [12,13,14]. Phospholamban (PLN) is a key regulator of SERCA function and cardiac contractility [9]. This 52 amino-acid protein allosterically inhibits SERCA activity, which reduces the rate of Ca2+ uptake into the SR [9,15,16]. Phosphorylation of PLN at Ser16/Thr17 by protein kinase A (PKA) and Ca2+/calmodulin-dependent protein kinase II (CAMKII), respectively, relieves SERCA inhibition by PLN and restores Ca2+ uptake [15,16]. Previous studies have demonstrated that failing hearts have reduced SERCA2a content and activity levels as well as increased inhibition of SERCA activity by PLN [16,17,18]. Thus, an imbalance between SERCA2a and PLN content in cardiac muscle can disrupt Ca2+ homeostasis and cardiac contractility, ultimately leading to cardiomyopathies and heart failure.
It has been well documented that ovariectomized (OVX) rodents, an experimental model for studying the effects of estrogen deficiency, have significantly reduced cardiac SR-Ca2+ uptake, SERCA activity, and SERCA2a content levels compared to controls [19,20,21,22,23]. Additionally, a previous study reported that total PLN and phosphorylated PLN (pPLN) content levels were elevated and reduced, respectively, in the hearts of OVX rats compared with controls, suggestive of increased SERCA inhibition mediated by PLN [23]. Estrogen supplementation was found to restore cardiac SERCA activity, as well as SERCA2a, PLN and pPLN content in OVX rats, suggesting that estrogen may play a critical role in the regulation of Ca2+ homeostasis and SR-Ca2+ uptake [23]. Additionally, Bupha-Intr et al. [24] showed that regular exercise through forced treadmill running restored the OVX-induced effects on SR-Ca2+ uptake, SERCA activity, SERCA2a content, and PLN content. In the present study, we sought to investigate the effects of voluntary wheel running (VWR), a less stressful form of exercise, on cardiac SR-Ca2+ uptake and SERCA function in OVX mice. We hypothesized that VWR would produce similar beneficial effects as forced treadmill running on cardiac SERCA function in OVX mice by increasing SR-Ca2+ uptake, SERCA activity, and SERCA2a content and reducing total PLN content.
2. Results
2.1. Animal Characteristics
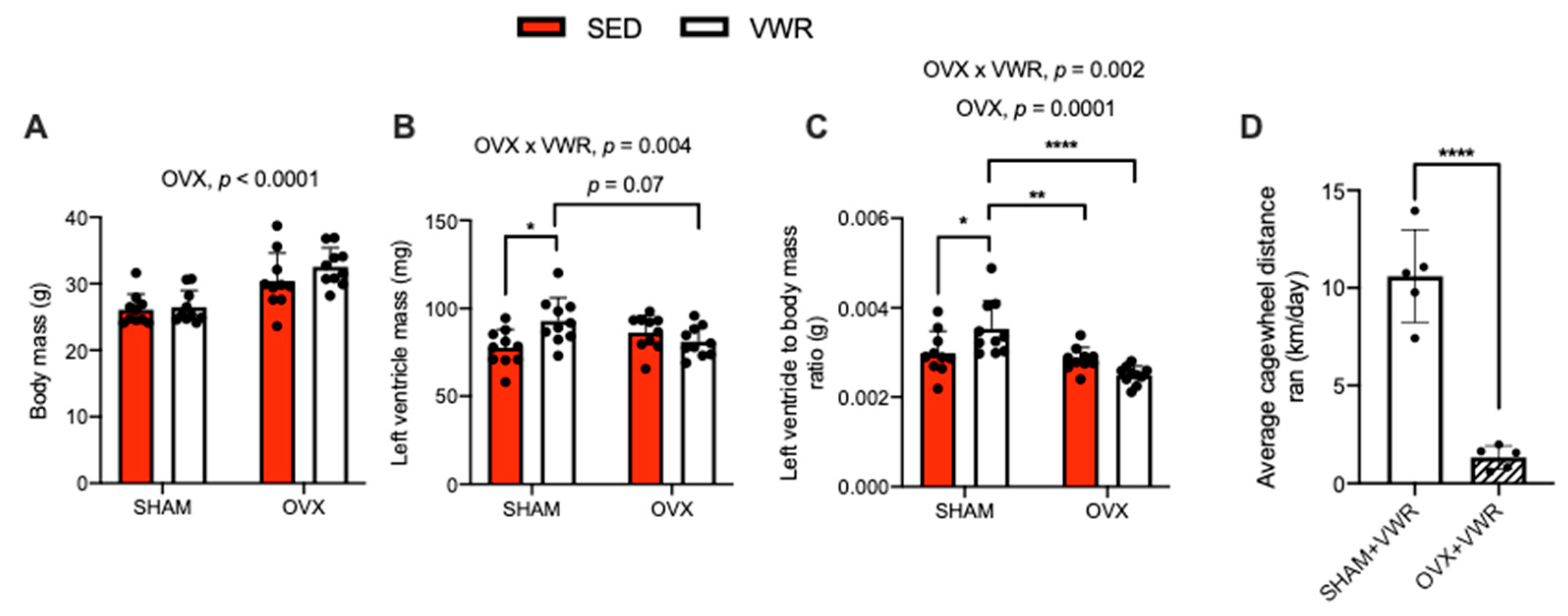
The effects of OVX and VWR on body mass, left ventricle mass (absolute and relative to body mass), and cagewheel running distance are shown in Figure 1. A main effect of OVX was found on body mass (g) (Figure 1A, p < 0.0001). We found a significant interaction between the effects of OVX and VWR on left ventricle mass (Figure 1B, p = 0.004). In the SHAM condition, the VWR mice had greater left ventricle mass compared with SED mice (Figure 1B, p = 0.01). Left ventricle mass was greater in SHAM + VWR mice compared with OVX + VWR mice, though this approached statistical significance (Figure 1B, p = 0.07). We also detected a main effect of OVX and a significant interaction between the effects of OVX and VWR on the ratio of left ventricle mass to total body mass (Figure 1C, p = 0.0001; p = 0.002). SHAM + VWR mice had a greater ratio of left ventricle mass to total body mass compared with SHAM + SED mice, OVX + SED mice, and OVX + VWR mice (Figure 1C, p = 0.04; p = 0.006; p < 0.0001). The SHAM + VWR mice ran a greater average cagewheel distance (km/day) compared with OVX + VWR mice, which was calculated over the 8-week exercise protocol (Figure 1D, p < 0.0001) [25].
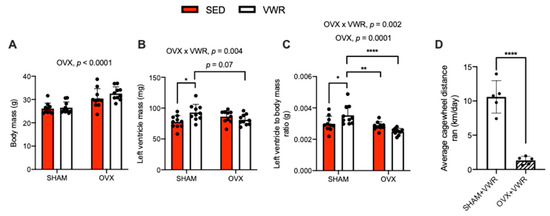
Figure 1.
Animal characteristics. (A) Body mass (g). (B) Left ventricle mass (mg). (C) Left ventricle to body mass ratio (g). (D) Average cagewheel distance ran (km/day) collected over the 8-week study. For (A–C) a two-way ANOVA with OVX and VWR as main effects and Fisher’s Least Significant Difference test were used. For (D) an unpaired t-test was used. For (A–C) a main effect of OVX was found. For (B,C) a significant interaction between OVX and VWR was detected. For (D) a significant difference between SHAM + VWR mice and OVX + VWR mice on average daily wheel running distance was found. *,**,****—p < 0.05, p < 0.01, p < 0.0001; p = 0.05–0.10; n = 10 per group.
2.2. Ca2+ Uptake
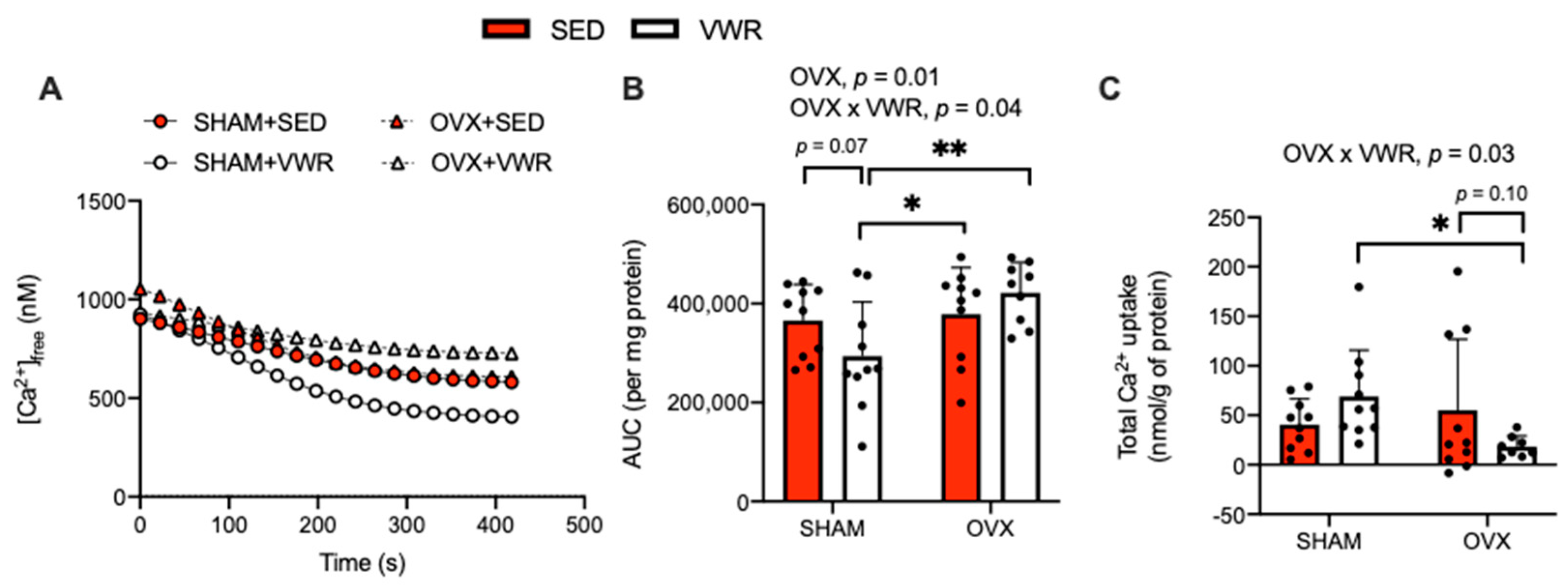
The effects of OVX and VWR on [Ca2+]free, Ca2+ uptake over time, and total Ca2+ uptake in LV homogenates are shown in Figure 2. We found that SHAM + VWR mice had the greatest amount of SR-Ca2+ uptake and OVX + VWR mice had the least amount of SR-Ca2+ uptake (Figure 2A–C). When analyzing the AUC, we detected a main effect of OVX (p = 0.01) and a significant interaction between the effects of OVX and VWR (Figure 2B, p = 0.04). In the SHAM condition, VWR decreased the AUC, though this only approached statistical significance (Figure 2B, p = 0.07). Conversely, in the OVX condition, VWR had no effect on the AUC (Figure 2B). We also conducted a fold-change analysis on the AUC in the SHAM + VWR group and OVX + VWR group relative to their sedentary counterparts and found that the AUC was significantly higher in OVX + VWR mice compared with SHAM + VWR mice (Supplemental Figure S1A, p = 0.01). Likewise, a significant interaction between the effects of OVX and VWR on total Ca2+ uptake was found (Figure 2C, p = 0.03). In this case, we found a reduction that approached statistical significance in the OVX + VWR group compared with OVX + SED group; and the SHAM + VWR group had significantly higher levels of total Ca2+ uptake compared with the OVX + VWR group (Figure 2C, p = 0.10; p = 0.03). Further, using a fold-change analysis relative to the sedentary groups we found that total Ca2+ uptake was significantly reduced in OVX + VWR mice compared with SHAM + VWR mice (Supplemental Figure S1B, p = 0.0003).
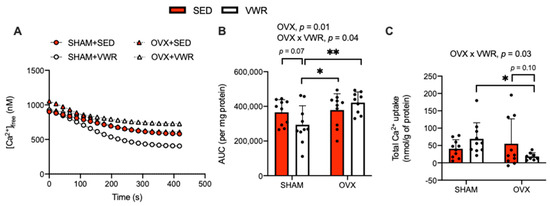
Figure 2.
VWR reduced SR-Ca2+ uptake in LV homogenates of OVX mice. (A) [Ca2+]free (nM) over time. (B) AUC (per mg of protein). (C) Total Ca2+ uptake (nmol/g of protein). For (A–C) a two-way ANOVA with OVX and VWR as main effects and Fisher’s Least Significant Difference test were used. For (B) a main effect of OVX was detected. For (B,C) a significant interaction was found between OVX and VWR. *,**—p < 0.05 and p < 0.01; p = 0.05–0.10; n = 10 per group.
2.3. SERCA ATPase Activity
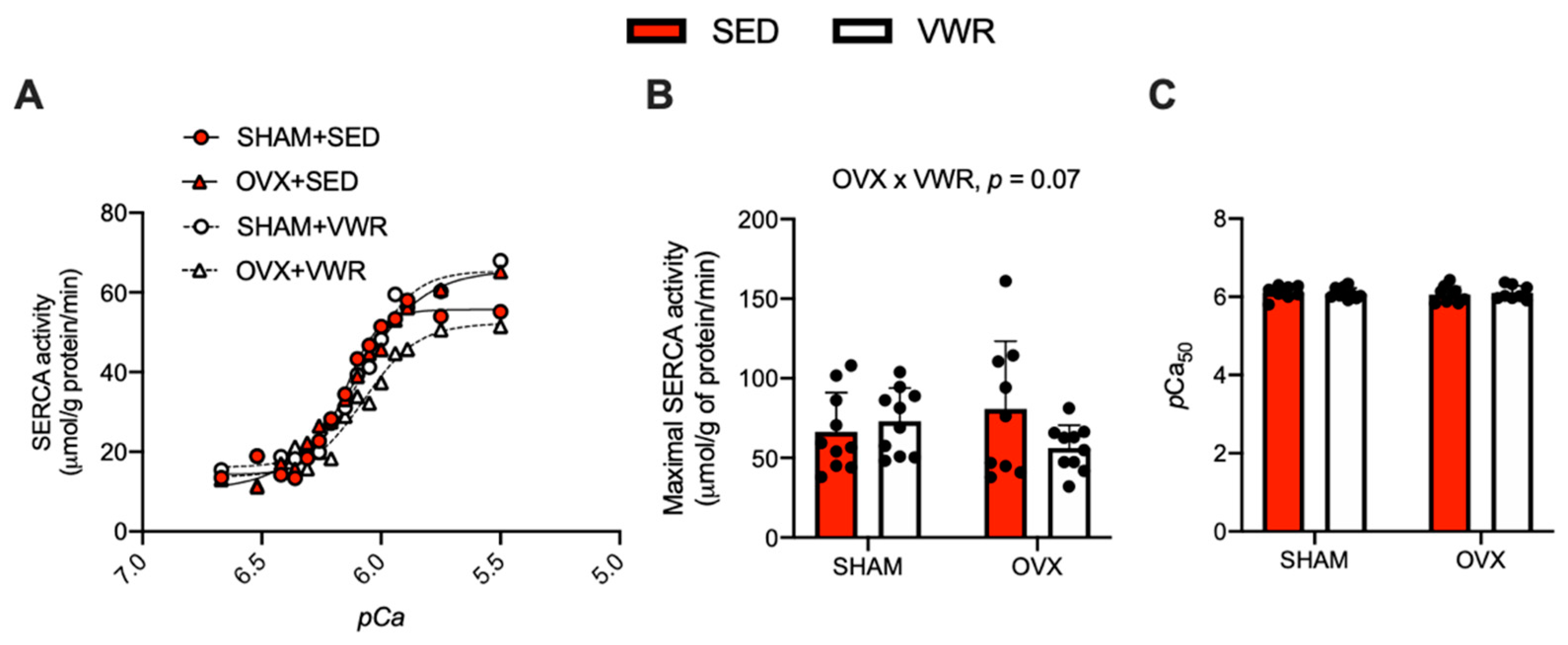
The effects of OVX and VWR on Ca2+-dependent SERCA activity, maximal SERCA activity, and pCa50 in LV homogenates are shown in Figure 3. Ca2+-dependent SERCA activity was measured at both submaximal and maximal Ca2+ concentrations ranging from a pCa of 6.8 to 5.5 (Figure 3A). Similar to our Ca2+ uptake experiments, we found an interaction between the effects of OVX and VWR on maximal SERCA activity to be approaching significance (Figure 3B, p = 0.07). According to the results of the fold-change analysis on maximal SERCA activity in the SHAM + VWR group and OVX + VWR group relative to their respective sedentary groups, we found that SHAM + VWR mice had higher maximal SERCA activity compared with OVX + VWR mice (Supplemental Figure S1C, p = 0.002). There were no differences in pCa50 between groups (Figure 3C).
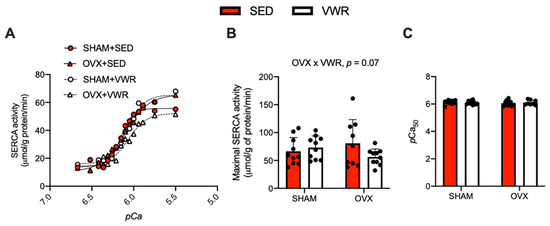
Figure 3.
Maximal SERCA activity is reduced in LV homogenates of OVX + VWR mice. (A) SERCA activity (μmol/g of protein/min) over pCa. (B) Maximal SERCA activity (μmol/g of protein/min). (C) Measure of pCa50 among the groups. For (A–C) a two-way ANOVA with OVX and VWR as main effects and Fisher’s Least Significant Difference test were used. For (B) a trending interaction between OVX and VWR was detected. p = 0.05–0.10; n = 10 per group.
2.4. SERCA and PLN Protein Content
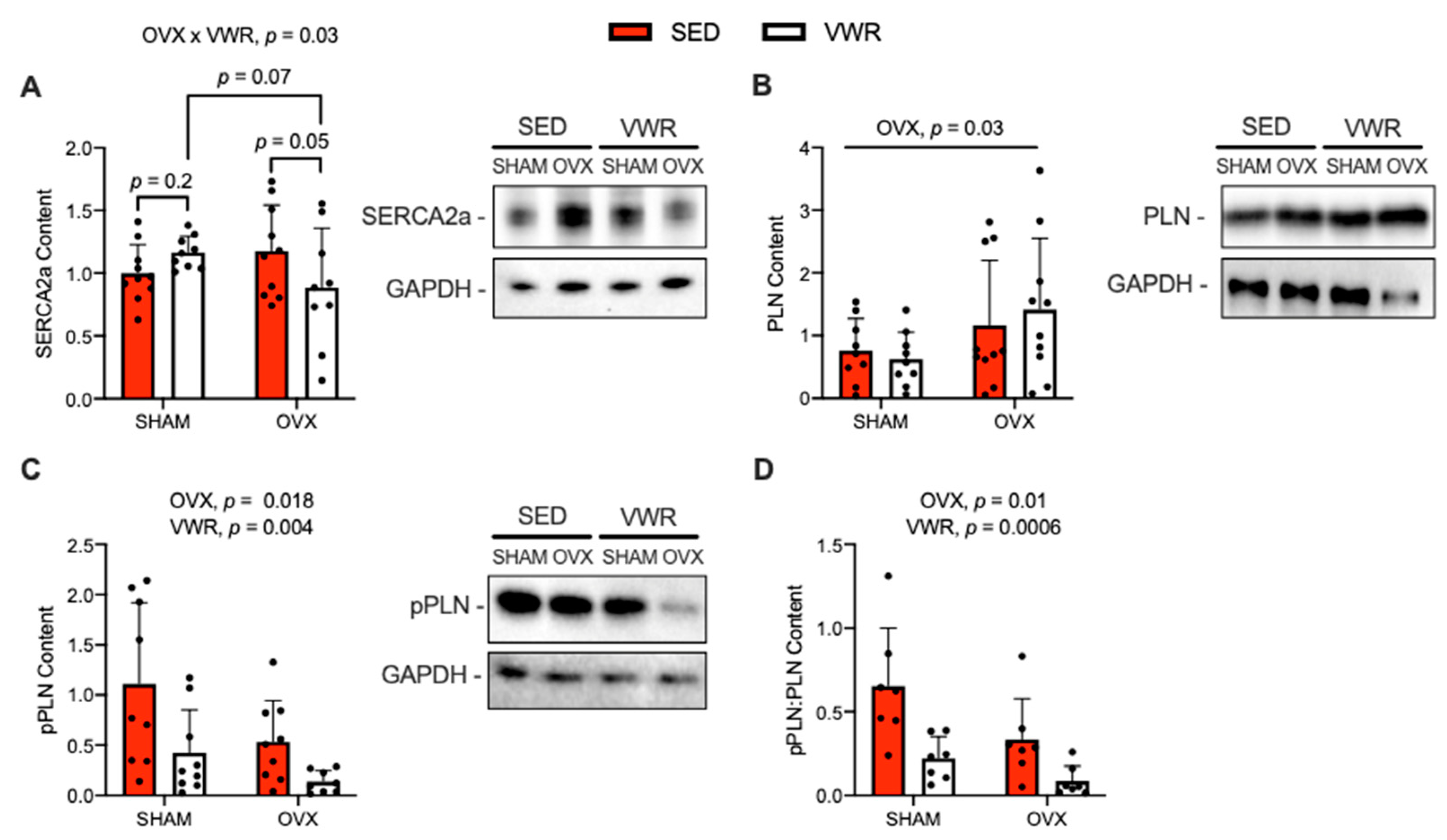
SERCA2a, PLN and pPLN protein content are shown in Figure 4. A significant interaction was found between the effects of OVX and VWR on SERCA2a content (Figure 4A, p = 0.03). The OVX + VWR group had lower content of SERCA2a compared with the OVX + SED group and SHAM + VWR group, though only approaching statistical significance (Figure 4A, p = 0.05; p = 0.07). Additionally, with a fold-change analysis relative to sedentary groups, we found that OVX + VWR mice had significantly lower content levels of SERCA2a compared with SHAM + VWR mice (Supplemental Figure S1D, p = 0.01). A main effect of OVX was found on PLN content, where PLN content was elevated in OVX mice compared with SHAM mice (Figure 4B, p = 0.03). A main effect of OVX was detected on absolute and relative (to total) pPLN, in which pPLN content was lower in OVX mice compared with SHAM mice (Figure 4C, p = 0.018; Figure 4D, p = 0.01). Further, a main effect of VWR was found on absolute and relative (to total) pPLN, where pPLN content was lower in VWR mice compared with SED mice (Figure 4C, p = 0.004; Figure 4D, p = 0.0006).
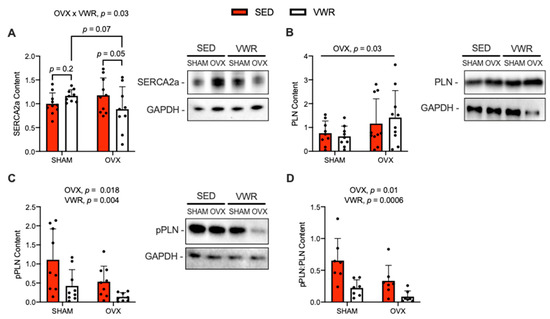
Figure 4.
Effects of OVX and VWR on SERCA2a, PLN, and pPLN (Ser16/Thr17) protein content. (A) SERCA2a content. (B) PLN content. (C) pPLN content. (D) Ratio of pPLN:PLN content. For (A–D) a two-way ANOVA with OVX and VWR as main effects and Fisher’s Least Significant Difference test were used. For (A) a significant interaction between OVX and VWR on SERCA2a content was detected. For (B) a main effect of OVX on PLN content was found. For (C,D) main effects of OVX and VWR were detected. p = 0.05–0.10; n = 10 per group.
3. Discussion
In the present study, we tested the hypothesis that VWR would produce beneficial effects on cardiac SERCA function in OVX mice with a specific increase in SR-Ca2+ uptake and SERCA activity caused by an elevation in SERCA2a and a reduction in PLN content. Our findings were inconsistent with our hypothesis as various statistical interactions detected across our analyses showed that these expected benefits of VWR on cardiac SERCA function were only apparent in SHAM mice and not in OVX mice. That is, Ca2+ uptake trended to be higher in SHAM + VWR mice compared with SHAM + SED mice, whereas we observed the opposite effect in OVX mice. Furthermore, the OVX + VWR group had significantly less Ca2+ uptake compared with SHAM + VWR mice, which was associated with lower SERCA2a content; however, the latter effect only approached statistical significance. Finally, PLN content and pPLN (Ser16/Thr17) content were higher and lower, respectively in OVX mice vs. SHAM mice regardless of exercise condition. Notably, we also found that OVX mice ran less than SHAM mice when given access to the cagewheel. Collectively, our findings show that in the absence of estrogen and with significantly reduced levels of VWR, OVX mice do not experience improvements in cardiac SERCA function.
As expected, we found a main effect of OVX on the AUC where OVX mice had reduced Ca2+ uptake over time compared with SHAM mice. These findings are consistent with previous literature that found higher cytosolic [Ca2+] and impaired SR-Ca2+ uptake in the hearts of OVX rodents [23,26]. However, we did not observe a reduction in SERCA2 content levels in OVX rodents vs. SHAM rodents as found previously [24]. Though we do not know the exact cause for this discrepancy, this could present a species or age-related difference, as the study done by Bupha-Intr et al. [24] used young rats (8–9 weeks) rather than adult-aged mice (26 weeks), which were used in the present study. We also identified a main effect of OVX on total PLN content, where OVX mice had elevated PLN content compared with SHAM mice. This finding is in accordance with previous findings that detected higher PLN content in OVX mice [23]. PLN reduces the affinity of SERCA for Ca2+ by binding to the transmembrane domain of the pump [27]. Phosphorylation of PLN at Ser16/Thr17 inhibits PLN activity and restores SERCA activity [15,16]. We found main effects of OVX on absolute and relative (to total) pPLN content where OVX mice had reduced pPLN content, which is indicative of overactive PLN. Though we did not find any differences in pCa50 which provides an indirect measure of SERCA’s apparent affinity for Ca2+, the overall increase in dephosphorylated PLN could contribute to the impairments found on Ca2+ uptake over time in OVX mice as previously shown in PLN overexpressing mice [28,29].
Regular exercise has been found to benefit cardiac Ca2+-handling in OVX rodents. Bupha-Intr et al. [24] used a 9-week five times per week forced treadmill running protocol, which restored levels of SR-Ca2+ uptake, SERCA activity, and SERCA2a, PLN, and pPLN content levels in OVX rats to be the same as in healthy SHAM rats [24]. We sought to recapitulate the same results using a voluntary exercise protocol and found improvements in cardiac SERCA function in SHAM + VWR mice and impairments in OVX + VWR mice. We detected significant interactions between the effects of OVX and VWR on the AUC and total Ca2+ uptake, where VWR increased SR-Ca2+ uptake in SHAM mice and decreased SR-Ca2+ uptake in OVX mice. In fact, the OVX + VWR group had the lowest level of Ca2+ uptake. In accordance with this finding, SERCA ATPase activity was also assessed and an interaction that approached significance was found between the effects of OVX and VWR. Based on this trend, it can be suggested that maximal SERCA activity was reduced in the OVX + VWR group compared with the OVX + SED group. We speculate that an increase in sample size would be sufficient to detect a significant interaction. Nonetheless, the trending interaction in SERCA activity corresponds well and likely contributes to the significant interactions found in our measures of Ca2+ uptake, as SERCA must first hydrolyze ATP to drive Ca2+ movement into the SR [11,12,13]. Moreover and consistent with these findings, a significant interaction between the effects of OVX and VWR was found on SERCA2a protein content. Indeed, the OVX + VWR group had the lowest levels of SERCA2a. Previous literature suggests that SERCA pump density is the main determinant of maximal SERCA activity and SR-Ca2+ uptake [30]. Thus, the decreased SR-Ca2+ uptake and maximal SERCA activity in OVX + VWR mice may be attributed to reduced SERCA2a density [30]. As SERCA2a content is reduced and PLN content is elevated in OVX + VWR mice, this suggests that the ratio of pump-to-inhibitor may be negatively altered, further contributing to impairments in SERCA function as seen in previous studies that found overexpression of PLN to reduce SERCA activity and SR-Ca2+ uptake [31,32,33]. Interestingly, we also found a main effect of VWR on absolute and relative (to total) pPLN content, which indicates that in response to VWR there was a decline in PLN phosphorylation and highlights the potential for increased inhibitory action of PLN. This is inconsistent with a recent study that showed VWR increased Ser16 phosphorylation of PLN in male and female wildtype mice [34]. However, it is important to consider the duration of the exercise protocol as Morissette et al. [34] conducted a 5-month training protocol and our study was a 6-week protocol. Furthermore, this apparent activation of PLN observed with VWR in the present study did not lead to any changes in SERCA’s apparent affinity for Ca2+ with VWR.
Our findings provide novel insight into how low and irregular amounts of voluntary exercise in OVX mice may have prevented any improvements in cardiac SERCA function. VWR is not a controlled form of exercise and the OVX + VWR mice ran six times less than the SHAM + VWR mice. This limitation was expected as numerous studies have reported significant reductions in running distance and spontaneous daily physical activity in rodents following ovariectomy [35,36,37]. In humans, previous literature has characterized menopause as a barrier of exercise for women and found that during the menopausal transition women spend more time being sedentary and less time being active [38,39]. A recent study that surveyed postmenopausal women found that 64% have a sedentary lifestyle, which was associated with severe menopausal symptoms such as weight gain and obesity, depression, fatigue, and hot flushes; and was negatively associated with hormone replacement therapy [40]. A study in rodents found that estrogen regulates motivation to exercise [41,42]. Importantly, this reveals another avenue for future research that can investigate ways to amplify the effects of seemingly limited VWR in OVX mice such that these mice will receive the benefits of VWR while performing lower exercise volume. Studies that match exercise, use estrogen supplementation, or other alternative treatments combined with VWR will be important for investigating the benefits of VWR in murine cardiac muscle and in finding viable preventative strategies against CVD in postmenopausal women.
4. Materials and Methods
4.1. Animals
Forty female C57BL6/J mice at 26 weeks of age were used in this study (Jackson Laboratory, Bar Harbor, ME). Before arriving to Brock University’s Animal Facility, half of the mice underwent a bilateral ovariectomy (n = 20) and the other half underwent a sham ovariectomy (n = 20) at 24 weeks of age at the Jackson Laboratory. The mice were housed in pairs and acclimated to the animal facility for 1-week before the exercise intervention commenced. Following acclimation, the mice were assigned to a sedentary (SED; n = 10 for SHAM and OVX) or voluntary wheel running (VWR; n = 10 for SHAM and OVX) group for 8 weeks. The VWR groups were housed in the Techniplast DVC80 caging system throughout the entire protocol which has GYM500 software to track daily distance travelled. The mice had ad libitum access to food and water and followed a standard 12-h light and dark schedule. Following the 8-week VWR intervention, the mice were anesthetized with a weight-adjusted bolus intraperitoneal injection of sodium pentobarbital (5 mg/100 g body mass) before being euthanized via exsanguination. The left ventricles were then dissected, weighed, homogenized (250 mM sucrose, 5 mM HEPES, 0.2 mM PMSF, 0.2% NaN3; pH 7.5), and frozen in liquid nitrogen to be stored at −80 °C. All experimental procedures were preapproved by the Brock University Animal Care Committee (AUP: No. 17-12-01) and followed the guidelines of the Canadian Council on Animal Care.
4.2. Ca2+ Uptake Assay
A Ca2+ uptake assay with Indo-1 was performed to quantify SR-Ca2+ uptake in LV homogenates. SR-Ca2+ uptake measurements are based on the difference in Ca2+-bound Indo-1 and Ca2+-free Indo-1 [43]. Indo-1 is impermeable to the SR membrane, thus as Ca2+ uptake occurs, the amount of Ca2+-bound Indo-1 will decrease and the amount of Ca2+-free Indo-1 will increase [43]. A M2 Molecular Devices MultiMode plate reader measured the fluorescence of Ca2+-bound Indo-1 at 450 nm and Ca2+-free Indo-1 at 485 nm at 37 °C [44]. To first measure the amount of starting [Ca2+]free, LV muscle homogenate and 2 mM Indo-1 (57180, Sigma-Aldrich, Burlington, MA, USA) were added to Ca2+ uptake buffer (200 mM KCl, 20 mM HEPES, 10 mM NaN3, 5 μM TPEN, 5 mM Oxalate, 15 mM MgCl2). Each sample was plated in duplicate on an all-black 96-well plate and placed into the plate reader. The plate was then removed and 250 mM ATP was added to initiate the uptake reaction, and was placed in the plate reader for 15–30 min. The plate was removed again and 50 mM EGTA and 100 mM CaCl were added to each well and placed back in the plate reader to measure intracellular [Ca2+]f. Total Ca2+ uptake along with area under the curve (AUC) analysis was completed to assess the amount of Ca2+ uptake over a period of time using GraphPad Prism 8 software (GraphPad Software Inc., San Diego, CA, USA). All measures were normalized to protein content using a bicinchoninic acid (BCA) protein assay.
4.3. SERCA Activity Assay
An enzyme-linked spectrophotometric assay was used to measure SERCA activity in LV homogenates over various [Ca2+] ranging from pCa 6.8 to 5.5 using ionophore (A23187, C7522, Sigma-Aldrich, Burlington, MA, USA). The ATPase activity of SERCA was linked to the rate of disappearance of NADH. 10 mM CaCl2 was added to a master mix in varying amounts of [Ca2+] and added to a 96-well plate. The master mix contained ATPase reaction buffer (100 mM KCl, 20 mM HEPES, 10 mM NaN3, 1 mM EGTA, 10 mM MgCl2, 5 mM ATP and 10 mM phosphoenolpyruvate; pH 7.0), 18 U• mL−1 LDH, 18 U• mL−1 PK, 1 mg•mL−1 ionophore and LV homogenate sample. An amount of 1.9% NADH was then added to each well and placed in the M2 Molecular Devices Spectrophotometric Plate Reader. The plate was read for 30 min at a wavelength of 340 nm and a temperature of 37 °C. The data was then fit onto a sigmoidal dose-response curve to calculate the pCa50 (concentration of Ca2+ required to elicit 1⁄2Vmax) using GraphPad Prism 8 software (GraphPad Software Inc., San Diego, CA, USA). All rates of SERCA activity across the various Ca2+ concentrations were normalized to protein content using a BCA assay.
4.4. Western Blotting
Western blots were performed for SERCA2a and total PLN using TGX BioRad PreCast 4–15% gradient gels (4561086; BioRad, Hercules, CA, USA). LV homogenate protein samples were solubilized in 4× in Laemelli buffer (#161-0747, BioRad, Hercules, CA, USA), loaded for gel electrophoresis, and then transferred to polyvinylidene difluoride (PVDF) membranes using the BioRad Transblot Turbo. The membranes were then blocked with milk (5% (w/v) in tris-buffered saline tween [TBST]) at room temperature for 1 h with their respective primary antibodies (1:2000 dilution). The primary antibodies for SERCA2a (MA3-919) and PLN (MA3-922) were obtained from ThermoFisher Scientific (Walktham, MA, USA). The primary antibody for pPLN (Ser16/Thr17; 8496) was obtained from Cell Signaling Technology (Danvers, MA, USA), and for panMHC (MF 20-s) was obtained from Developmental Studies Hydrobioma Bank (Iowa City, IA, USA). After incubation was complete, membranes were washed 3 times in 5 min intervals with TBST. The membranes were then incubated with anti-mouse secondary antibody for 1 h at room temperature (1:2000 dilution). After the secondary incubation, membranes were washed again 3 times in 5 min intervals with TBST. Millipore Immobilon (WBKLS0500) or ThermoFisher Supersignal Femto West (PI34096) chemiluminescent substrate was added for detection, and then visualized using a BioRad ChemiDoc Imager. Optical densities were normalized to total protein visualized on PVDF membranes with GAPDH (#2118S, Cell Signaling Technology)
4.5. Statistical Analysis
All data are expressed as mean ± SD, with significance reported as p ≤ 0.05, and values approaching significance were reported as p = 0.06–0.10. All comparisons between SHAM + SED, OVX + SED, SHAM + VWR, and OVX + VWR were conducted using a two-way ANOVA with VWR and OVX as main effects and a Fisher’s Least Significant Difference test. Comparisons between SHAM + VWR and OVX + VWR were performed using an unpaired t-test. All statistical analysis was performed using GraphPad Prism 8 software (GraphPad Software Inc., San Diego, CA, USA).
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/muscles1030016/s1, Figure S1: Fold-change analysis for SR-Ca2+ uptake, SERCA activity, and SERCA2a content.
Author Contributions
Conceptualization, V.A.F.; methodology, V.A.F.; formal analysis, V.A.F., B.M.M. and M.S.G.; investigation, V.A.F., B.M.M. and M.S.G.; writing—original draft preparation, B.M.M.; writing—review and editing, V.A.F., B.M.M., M.S.G., A.M., J.S.-M. and R.E.K.M.; supervision, R.E.K.M. and V.A.F. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by a Canada Research Chair (Tier II) to VAF and an award from the Scottish Rite Charitable Foundation of Canada and The Alzheimer’s Society of Brant, Haldimand Norfolk, Hamilton, Halton to REKM. A. Mohammad was supported by a Brock University Match of Minds as well as an NSERC USRA. J. Sweezey-Munroe was supported by a Brock University Match of Minds.
Institutional Review Board Statement
All experimental procedures were preapproved by the Brock University Animal Care Committee (AUP: No. 17-12-01) and followed the guidelines of the Canadian Council on Animal Care.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data can be made available upon request to the corresponding author.
Acknowledgments
We are grateful for the help of our Animal Care Staff at Brock University.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Vogel, B.; Acevedo, M.; Appelman, Y.; Merz, C.N.B.; Chieffo, A.; Figtree, G.A.; Guerrero, M.; Kunadian, V.; Lam, C.S.; Maas, A.H. The Lancet women and cardiovascular disease Commission: Reducing the global burden by 2030. Lancet 2021, 397, 2385–2438. [Google Scholar] [CrossRef]
- Jiao, L.; Machuki, J.O.; Wu, Q.; Shi, M.; Fu, L.; Adekunle, A.O.; Tao, X.; Xu, C.; Hu, X.; Yin, Z.; et al. Estrogen and calcium handling proteins: New discoveries and mechanisms in cardiovascular diseases. Am. J. Physiol. Heart Circ. Physiol. 2020, 318, H820–H829. [Google Scholar] [CrossRef] [PubMed]
- Bots, S.H.; Peters, S.A.; Woodward, M. Sex differences in coronary heart disease and stroke mortality: A global assessment of the effect of ageing between 1980 and 2010. BMJ Glob. Health 2017, 2, e000298. [Google Scholar] [CrossRef] [PubMed]
- Khesroh, A.A.; Al-Roumi, F.; Al-Zakwani, I.; Attur, S.; Rashed, W.; Zubaid, M. Gender differences among patients with acute coronary syndrome in the Middle East. Heart Views Off. J. Gulf Heart Assoc. 2017, 18, 77. [Google Scholar] [CrossRef]
- El Khoudary, S.R.; Aggarwal, B.; Beckie, T.M.; Hodis, H.N.; Johnson, A.E.; Langer, R.D.; Limacher, M.C.; Manson, J.E.; Stefanick, M.L.; Allison, M.A. Menopause transition and cardiovascular disease risk: Implications for timing of early prevention: A scientific statement from the American Heart Association. Circulation 2020, 142, e506–e532. [Google Scholar] [CrossRef]
- Iorga, A.; Cunningham, C.M.; Moazeni, S.; Ruffenach, G.; Umar, S.; Eghbali, M. The protective role of estrogen and estrogen receptors in cardiovascular disease and the controversial use of estrogen therapy. Biol. Sex Differ. 2017, 8, 33. [Google Scholar] [CrossRef]
- Moreau, K.L. Intersection between gonadal function and vascular aging in women. J. Appl. Physiol. 2018, 125, 1881–1887. [Google Scholar] [CrossRef]
- Roa-Díaz, Z.M.; Raguindin, P.F.; Bano, A.; Laine, J.E.; Muka, T.; Glisic, M. Menopause and cardiometabolic diseases: What we (don’t) know and why it matters. Maturitas 2021, 152, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Santoro, N. The menopausal transition. Am. J. Med. 2005, 118, 8–13. [Google Scholar] [CrossRef]
- Maslov, P.Z.; Kim, J.K.; Argulian, E.; Ahmadi, A.; Narula, N.; Singh, M.; Bax, J.; Narula, J. Is cardiac diastolic dysfunction a part of post-menopausal syndrome? JACC Heart Fail. 2019, 7, 192–203. [Google Scholar] [CrossRef]
- Hamstra, S.I.; Kurgan, N.; Baranowski, R.W.; Qiu, L.; Watson, C.J.; Messner, H.N.; MacPherson, R.E.; MacNeil, A.J.; Roy, B.D.; Fajardo, V.A. Low-dose lithium feeding increases the SERCA2a-to-phospholamban ratio, improving SERCA function in murine left ventricles. Exp. Physiol. 2020, 105, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Misquitta, C.; Mack, D.; Grover, A. Sarco/endoplasmic reticulum Ca2+ (SERCA)-pumps: Link to heart beats and calcium waves. Cell Calcium 1999, 25, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Dally, S.; Bredoux, R.; Corvazier, E.; Andersen, J.P.; Clausen, J.D.; Dode, L.; Fanchaouy, M.; Gelebart, P.; Monceau, V.; Del Monte, F. Ca2+-ATPases in non-failing and failing heart: Evidence for a novel cardiac sarco/endoplasmic reticulum Ca2+-ATPase 2 isoform (SERCA2c). Biochem. J. 2006, 395, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Stammers, A.N.; Susser, S.E.; Hamm, N.C.; Hlynsky, M.W.; Kimber, D.E.; Kehler, D.S.; Duhamel, T.A. The regulation of sarco (endo) plasmic reticulum calcium-ATPases (SERCA). Can. J. Physiol. Pharmacol. 2015, 93, 843–854. [Google Scholar] [CrossRef]
- Kranias, E.G.; Hajjar, R.J. Modulation of cardiac contractility by the phopholamban/SERCA2a regulatome. Circ. Res. 2012, 110, 1646–1660. [Google Scholar] [CrossRef]
- Hamstra, S.I.; Whitley, K.C.; Baranowski, R.W.; Kurgan, N.; Braun, J.L.; Messner, H.N.; Fajardo, V.A. The role of phospholamban and GSK3 in regulating rodent cardiac SERCA function. Am. J. Physiol.-Cell Physiol. 2020, 319, C694–C699. [Google Scholar] [CrossRef]
- Kawase, Y.; Hajjar, R.J. The cardiac sarcoplasmic/endoplasmic reticulum calcium ATPase: A potent target for cardiovascular diseases. Nat. Clin. Pract. Cardiovasc. Med. 2008, 5, 554–565. [Google Scholar] [CrossRef]
- Steenman, M.; Lande, G. Cardiac aging and heart disease in humans. Biophys. Rev. 2017, 9, 131–137. [Google Scholar] [CrossRef]
- Kravtsov, G.M.; Kam, K.W.; Liu, J.; Wu, S.; Wong, T.M. Altered Ca2+ handling by ryanodine receptor and Na+-Ca2+ exchange in the heart from ovariectomized rats: Role of protein kinase A. Am. J. Physiol.-Cell Physiol. 2007, 292, C1625–C1635. [Google Scholar] [CrossRef]
- Firth, J.M.; Yang, H.-Y.; Francis, A.J.; Islam, N.; MacLeod, K.T. The effect of estrogen on intracellular Ca2+ and Na+ regulation in heart failure. Basic Transl. Sci. 2020, 5, 901–912. [Google Scholar] [CrossRef]
- Machuki, J.O.a.; Zhang, H.-Y.; Geng, J.; Fu, L.; Adzika, G.K.; Wu, L.; Shang, W.; Wu, J.; Kexue, L.; Zhao, Z. Estrogen regulation of cardiac cAMP-L-type Ca2+ channel pathway modulates sex differences in basal contraction and responses to β2AR-mediated stress in left ventricular apical myocytes. Cell Commun. Signal. 2019, 17, 34. [Google Scholar] [CrossRef] [PubMed]
- Turdi, S.; Huff, A.F.; Pang, J.; He, E.Y.; Chen, X.; Wang, S.; Chen, Y.; Zhang, Y.; Ren, J. 17-β estradiol attenuates ovariectomy-induced changes in cardiomyocyte contractile function via activation of AMP-activated protein kinase. Toxicol. Lett. 2015, 232, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Bupha-Intr, T.; Wattanapermpool, J. Regulatory role of ovarian sex hormones in calcium uptake activity of cardiac sarcoplasmic reticulum. Am. J. Physiol.-Heart Circ. Physiol. 2006, 291, H1101–H1108. [Google Scholar] [CrossRef] [PubMed]
- Bupha-Intr, T.; Laosiripisan, J.; Wattanapermpool, J. Moderate intensity of regular exercise improves cardiac SR Ca2+ uptake activity in ovariectomized rats. J. Appl. Physiol. 2009, 107, 1105–1112. [Google Scholar] [CrossRef]
- Mohammad, A.; Finch, M.S.; Sweezey-Munroe, J.; MacPherson, R.E. Voluntary wheel running alters markers of amyloid-beta precursor protein processing in an ovarian hormone depleted model. Front. Endocrinol. 2022, 13. [Google Scholar] [CrossRef]
- Fares, E.; Pyle, W.G.; Ray, G.; Rose, R.A.; Denovan-Wright, E.M.; Chen, R.P.; Howlett, S.E. The impact of ovariectomy on calcium homeostasis and myofilament calcium sensitivity in the aging mouse heart. PLoS ONE 2013, 8, e74719. [Google Scholar] [CrossRef]
- Akin, B.L.; Hurley, T.D.; Chen, Z.; Jones, L.R. The structural basis for phospholamban inhibition of the calcium pump in sarcoplasmic reticulum. J. Biol. Chem. 2013, 288, 30181–30191. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, V.A.; Bombardier, E.; McMillan, E.; Tran, K.; Wadsworth, B.J.; Gamu, D.; Hopf, A.; Vigna, C.; Smith, I.C.; Bellissimo, C. Phospholamban overexpression in mice causes a centronuclear myopathy-like phenotype. Dis. Model. Mech. 2015, 8, 999–1009. [Google Scholar] [CrossRef]
- Chu, G.; Dorn, G.W.; Luo, W.; Harrer, J.M.; Kadambi, V.J.; Walsh, R.A.; Kranias, E.G. Monomeric phospholamban overexpression in transgenic mouse hearts. Circ. Res. 1997, 81, 485–492. [Google Scholar] [CrossRef]
- Kiliszek, M.; Mackiewicz, U.; Maczewski, M.; Burzynska, B. Molecular evidence that exercise training has beneficial effects on cardiac performance. Ann. Transl. Med. 2016, 4, 228. [Google Scholar] [CrossRef]
- Limas, C.J.; OLIVARI, M.-T.; Goldenberg, I.F.; Levine, T.B.; Benditt, D.G.; Simon, A. Calcium uptake by cardiac sarcoplasmic reticulum in human dilated cardiomyopathy. Cardiovasc. Res. 1987, 21, 601–605. [Google Scholar] [CrossRef]
- Park, W.J.; Oh, J.G. SERCA2a: A prime target for modulation of cardiac contractility during heart failure. BMB Rep. 2013, 46, 237. [Google Scholar] [CrossRef]
- Kadambi, V.J.; Ponniah, S.; Harrer, J.M.; Hoit, B.D.; Dorn, G.n.; Walsh, R.A.; Kranias, E.G. Cardiac-specific overexpression of phospholamban alters calcium kinetics and resultant cardiomyocyte mechanics in transgenic mice. J. Clin. Investig. 1996, 97, 533–539. [Google Scholar] [CrossRef]
- Morissette, M.P.; Susser, S.E.; Stammers, A.N.; Moffatt, T.L.; Wigle, J.T.; Wigle, T.J.; Netticadan, T.; Premecz, S.; Jassal, D.S.; O’Hara, K.A.; et al. Exercise-induced increases in the expression and activity of cardiac sarcoplasmic reticulum calcium ATPase 2 is attenuated in AMPKα(2) kinase-dead mice. Can J Physiol Pharm. 2019, 97, 786–795. [Google Scholar] [CrossRef]
- Cabelka, C.A.; Baumann, C.W.; Collins, B.C.; Nash, N.; Le, G.; Lindsay, A.; Spangenburg, E.E.; Lowe, D.A. Effects of ovarian hormones and estrogen receptor α on physical activity and skeletal muscle fatigue in female mice. Exp. Gerontol. 2019, 115, 155–164. [Google Scholar] [CrossRef]
- Morgan, M.; Pfaff, D. Effects of estrogen on activity and fear-related behaviors in mice. Horm. Behav. 2001, 40, 472–482. [Google Scholar] [CrossRef]
- Bondarev, D.; Finni, T.; Kokko, K.; Kujala, U.M.; Aukee, P.; Kovanen, V.; Laakkonen, E.K.; Sipilä, S. Physical performance during the menopausal transition and the role of physical activity. J. Gerontol. Ser. A 2021, 76, 1587–1590. [Google Scholar] [CrossRef]
- Tinker, A.; Molloy, L.; Monks, I.; Pennells, L.; Russell, E.; Haines, E. The benefits and barriers of exercise for the physical health of older women. J. Aging Clin. Res. 2017, 6, 73–79. [Google Scholar] [CrossRef]
- Karine, D.; Rémi, R.-L.; Irene, S.; Martin, B.; Jean-Marc, L.; Éric, D. Effects of the menopausal transition on factors related to energy balance. A MONET group study: I. Energy expenditure. Eur. J. Clin. Nutr. 2013, 67, 407. [Google Scholar]
- Blümel, J.E.; Chedraui, P.; Aedo, S.; Fica, J.; Mezones-Holguín, E.; Barón, G.; Bencosme, A.; Benítez, Z.; Bravo, L.M.; Calle, A. Obesity and its relation to depressive symptoms and sedentary lifestyle in middle-aged women. Maturitas 2015, 80, 100–105. [Google Scholar] [CrossRef]
- Krause, W.C.; Rodriguez, R.; Gegenhuber, B.; Matharu, N.; Rodriguez, A.N.; Padilla-Roger, A.M.; Toma, K.; Herber, C.B.; Correa, S.M.; Duan, X. Oestrogen engages brain MC4R signalling to drive physical activity in female mice. Nature 2021, 599, 131–135. [Google Scholar] [CrossRef]
- Lightfoot, J.T. Sex hormones’ regulation of rodent physical activity: A review. Int. J. Biol. Sci. 2008, 4, 126. [Google Scholar] [CrossRef]
- Tupling, R.; Green, H. Silver ions induce Ca2+ release from the SR in vitro by acting on the Ca2+ release channel and the Ca2+ pump. J. Appl. Physiol. 2002, 92, 1603–1610. [Google Scholar] [CrossRef]
- Grynkiewicz, G.; Poenie, M.; Tsien, R.Y. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem. 1985, 260, 3440–3450. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).