Abstract
Besides beech, oak is one of the most ecologically and economically important deciduous tree species in Central European forests. Due to progressive global warming, oaks are increasingly exposed to biotic and abiotic stress factors. In addition to drought and heat, thermophilic herbivorous insects also represent a challenge for the oak trees. During earlier investigations on Tortrix viridana in North Rhine-Westphalia, differences in the infestation intensity of individual pedunculate oaks (Quercus robur) by herbivorous insects led to a definition of tolerant (T-oaks, less infested) and susceptible (S-oaks, heavily infested) oaks. Performance studies with T. viridana and Lymantria dispar were conducted to validate the observations. Both herbivorous insects needed more time for their juvenile development when reared on T-oaks. While the larvae of L. dispar tended to eat less leaf material from T-oaks than from S-oaks, T. viridana needed to consume more T-oak leaf material to achieve approximately the same pupal weight as larvae fed with leaf material from S-oaks. The mortality rate of T. viridana was significantly higher on T-oaks than on S-oaks, whereas no significant difference was found for L. dispar. Basically, T-oaks seem to offer a poorer nutritional basis for both kinds of leaf-eating insects.
1. Introduction
Covering 32% of the national territory, forests are a major part of Germany’s landscape. They play a decisive role in climate and culture, not only as an important carbon sink [1], but also as part of the water cycle, as an oxygen and wood producer, a habitat for many animal and plant species, a recreational area for humans and much more [2,3]. Poor silvicultural practices of the past, together with adverse anthropogenic effects, have posed many challenges for forests, an effect which is likely to increase. Among these challenges are temperature changes due to global warming, increasingly frequent drought and precipitation events [4] and evolving threats from new pests and diseases [5]. After beech, oak is the second most common deciduous tree genus in European forests. The most common oak species in Central Europe are the pedunculate oak (Q. robur) and the sessile oak (Quercus petraea). They account for up to 10% of all trees in German forests [6]. Together with beech wood, oak has high value as construction timber. In addition to soft coniferous woods, straight deciduous woods are particularly valued for their robustness and hardness. For example, railroad sleepers, constructions for half-timbered houses and pile dwellings are made of oak wood.
Interactions between trees and herbivorous insects are among the driving factors of co-evolutionary processes in forest ecosystems [7,8]. Q. robur hosts a large number of herbivorous insects. Besides the gypsy moth (L. dispar) and the green oak leaf roller (T. viridana), the small (Operophtera brumata) and the large (Erannis defoliaria) winter moth as well as the oak processionary (Thaumetopoea processionea) also belong to the oak feeding society, to name just a few. In years with mass occurrence of larvae of various herbivorous caterpillars, the oak is exposed to additional stressors. During a calamity of the pest, this can cause a growth loss of up to 30% (resources are needed for the budding of new leaves), a premature St. John’s shoot and even the failure to fruit. In the worst case, oak dieback occurs [9,10], but this usually only occurs in connection with further weakening of the oak (fungal attack or severe drought) [11,12,13].
T. viridana is an oligophagous specialist. It occurs only on oak trees (usually Q. robur and sometimes Q. petraea) and cannot develop on other deciduous trees. In contrast, L. dispar, as a generalist, can develop on many different types of trees and shrubs. Both pests are common in Central Europe and infest deciduous forests.
In the present study, we compared the performance of T. vidiana and L. dispar on two different oak types defined as tolerant oaks (T-oaks: less defoliated during outbreak events) and susceptible oaks (S-oaks: heavily defoliated during outbreak events) in an earlier study [14]. In the same study, we showed that T- and S-oak leaves differ in their composition of polyphenolic constituents and their blend of VOCs (volatile organic compounds), leading us to the hypothesis that T- and S-oaks follow different strategies. Where T-oaks count on a constitutive high level of defence compounds, S-oaks react just in time, emitting typical ‘cry for help’ [15] HIPVs (herbivore-induced plant volatiles). We already showed that larvae of the green oak leaf roller needed to eat more T-oak leaf material to gain the same pupal weight as larvae fed with S-oak leaves [14,16]. Thus, here we present the comparison of performance studies with a specialist and a generalist herbivorous moth.
2. Experiments
2.1. Plant and Caterpillar Material
During an outbreak of the green oak leaf roller (2003–2005), oak trees in North Rhine-Westphalia were classified as susceptible or tolerant to the damage caused by the caterpillar of T. viridana [14,17]. In order to conduct further analysis, genetically comparable plant material was required. For this purpose, summer grafting [17] was carried out. Young shoots of the classified oak trees were taken from the original stand in 2008, 2011 and 2015 and grafted onto 2-year-old seedlings of Q. robur. The oaks were wrapped in Crispac bags in spring to prevent foreign insects from settling on the trees. The bagged trees were brought to a shady greenhouse so that a slow acclimatization to the increasing sunlight could take place.
Breeding of larvae of T. viridana was established in 2007 with caterpillars from 15 stands in North Rhine-Westphalia, and since then was continuously genetically refreshed with larvae from other oak stands. Two clutches of L. dispar eggs were kindly provided by ARätin Andrea Stradner from the University of Natural Resources and Life Sciences, Vienna.
2.2. Feeding Experiment with Tortrix viridana
A total of 307 freshly hatched larvae of T. viridana were placed on opening buds of either T-oaks (150) or S-oaks (157) with a fine brush and isolated with a Crispac bag. These bags also prevented the larvae from breaking out. The larvae were individualised by an assigned number. Under constant conditions (12–13 °C and 10 h/14 h light/darkness) in a climate chamber the larvae could develop on the trees until their pupation.
From larval stage 3 onwards, the larvae were weighed in a 4-day cycle and the leaf area eaten was determined with the help of scale paper. The larvae were checked daily in order to document the changes into the next larval stages. The fresh pupae were transferred into Petri dishes and kept under the same conditions until the adult moth hatched. A statistical analysis was performed using the t-test for developmental time and amount of eaten leaf, and the Chi2-test for overall mortality. For details, see Ghirardo et al. [14] and Schroeder et al. [16].
2.3. Feeding Experiment with Lymantria dispar
The newly hatched larvae of L. dispar were set upon a piece of wheat germ medium developed by Magnoler [18]. After reaching the 3rd instar the caterpillars were separately placed on S- or T-oak trees in the field to adapt them to leaf feeding. Once they reached the 4th instar the larvae were taken into the experiment. Under constant conditions (20 °C ± 5 and 16 h/8 h light/darkness) the larvae were fed in plastic boxes with detached leaf material. The development was documented until pupation.
A total of 160 larvae were used for the experiment. They were divided into two groups, one fed with T-oak and the other with S-oak leaves. The two groups were further divided into two subgroups. One half of each group was fed with leaves from pre-infested trees and the other half with leaves of non-infested trees. For pre-infestation at least 25 larvae of L. dispar were put on single T- or S-oaks for a minimum of two days before starting the feeding experiment. The pre-infested trees are hereafter referred to as ‘Damaged-trees’, while those that were left non-infested are referred to as ‘Control-trees’. The groups are abbreviated as follows: T_C (T-oaks control) = fed with leaves from T-oaks without pre-infestation; S_C (S-oaks control) = the same as before but fed with leaves from S-oaks; T_D (T-oaks damaged) = fed with leaves from pre-infested T-oaks; and S_D (S-oaks damaged) = the same as before but fed with leaves from pre-infested S-oaks.
The larvae were monitored daily and fed with fresh leaves at least every fourth day, always ensuring that they were adequately fed. Images of leaves were taken with a Canon Lide 210 scanner and Canon MP Navigator EX software. Leaves were scanned before and after feeding. Analysis of leaf images was performed using RStudio software [19,20] and the package ‘raster’ [21]. Comparison of images of old leaves (uneaten and lying in the plastic box for some days) and the respective image of the fresh leaf revealed a median difference in leaf area of 1% (shrinking of leaves).
For the L. dispar data, statistical analysis was performed using RStudio Software [19,20] and the packages ‘PMCMRplus’ [22] and ‘asbio’ [23]. Visualization of the results was performed using the package ‘ggplot2′ [24].
3. Results
3.1. Developmental Time
A calculation over all larval stages of T. viridana showed no significant differences in the developmental duration between larvae reared on T- or S-oaks. However, the duration time in the first two instars is significantly higher (t-test for L1 p = 0.01; for L2 p = 0.02) for larvae fed on T-oaks than on S-oaks. Interestingly, for the fourth instars it is the other way round. In this stage, larvae fed on S-oaks needed significantly more time (t-test p = 0.006) to reach the next stage than larvae reared on T-oaks (Table 1), [16].

Table 1.
Developmental time for Tortrix viridana on S- and T-oaks (time in days).
The sexes of L. dispar differ much more than those of T. viridana. Thus, for the gypsy moth, all analyses were performed separately for males and females. The number of instars until pupation differed within and between the sexes. The female larvae pupated after the fifth instar, whereas the males nearly equally pupated after the fourth or the fifth instar, respectively.
The duration of the instars and pupal stage is shown in Figure 1. Both females and males showed no significant difference in the duration of the fourth instar. The fifth instar of the females lasted significantly longer for the T_D group than those of the S-oak control group (S_C), while the males showed no significant differences. The pupal stage in males lasted significantly longer in the T_D group than in both control groups (T_C and S_C), whereas female pupae showed a significantly slower development only for T_D compared to T_C (Figure 1).
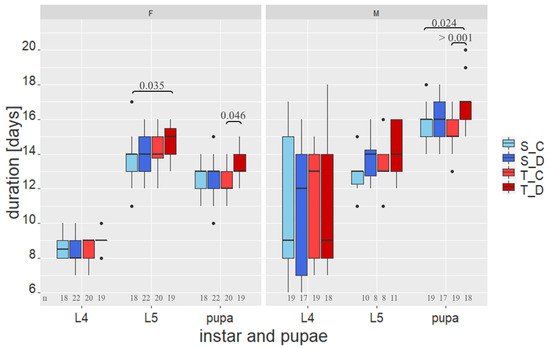
Figure 1.
Duration of the instars and the pupal stage of Lymantria dispar. Boxplot colours indicate the different groups. S_C: S-oaks control; S_D: S-oaks damaged (pre-infested), T_C: T-oaks control and T_D: T-oaks damaged. Data are grouped by sex: females (left) and males (right), and by larval stage: L4 (fourth instar), L5 (fifth instar) and pupa. Bars with numbers above boxplots indicate significant differences between groups and the p-value from Nemenyi’s non-parametric all-pairs comparison. Numbers below show the numbers of observations for each boxplot.
3.2. Leaf Area Fed
The amount of leaf area fed was observed from the third until the fifth instar for T. viridana. There were no significant differences in the leaf area fed between T- and S-oaks in the third and fourth instar. However, the leaf area fed differed significantly in the fifth instar (t-test p = 0.04) and also when looking at all instars (t-test p = 0.02). Larvae reared on T-oaks ate nearly 10% more leaves than larvae on S-oaks (Table 2) [16]. Even more important is the fact that larvae of T. viridana fed with T-oak leaves needed to consume significantly more leaf material to gain the same pupal weight as larvae fed with S-oak leaves [14].

Table 2.
Amount/area of leaves fed (in mm2) on S- and T-oaks in total and within the stages.
For L. dispar, the total leaf area fed was measured from the fourth instar until pupation. Female larvae of the S_C group ate significantly more leaves than the larvae of the S_D (posthoc.kruskal.nemenyi.test p = 0.038) and T_D (posthoc.kruskal.nemenyi.test p > 0.001) groups, while the larvae of the T_C group did not differ from the others (Figure 2).
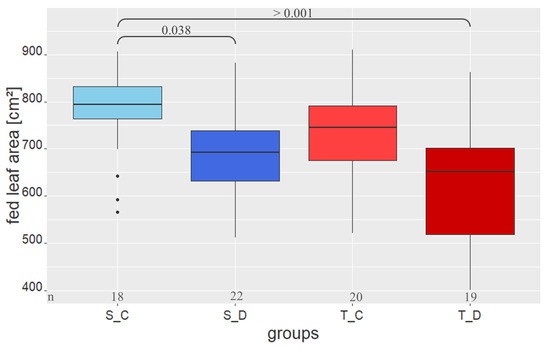
Figure 2.
Total leaf area fed by the female larvae from fourth instar until pupation. Abbreviations and explanations are as in Figure 1.
For the males, none of the groups showed significant differences, but there was a tendency for the T_D group larvae to eat slightly less than the others. Overall, the females ate much more than the males during the experiment.
3.3. Mortality Rate
From the original 157 hatched larvae of T. viridana placed on S-oaks, 59 individuals completed their development to adult moths. On T-oaks only 40 individuals out of the original 150 reached the adult stage. That means a significantly different overall mortality rate of 62.4% for larvae reared on S-oaks and 73.3% for larvae reared on T-oaks (Chi2 p < 0.05) (Table 3). On both oak types, the mortality rate was highest within the first two larval stages, followed by the pupal stage, with 10.8% in S-oaks and 14.7% in T-oaks (Table 3, [16]).

Table 3.
Mortality of Tortrix viridana (in %) on S- and T-oaks.
For L. dispar, the overall mortality rate was rather low. During the rearing of the larvae for the later experiment in the first three larval stages, only five to seven individuals per instar died. During the experiment beginning with the fourth instar, five larvae had problems pupating and died. Of these, three belonged to group S_C and one each to S-D and T_C. During the hatching to adult moth, two additional individuals died, one of the S_C group and one of the T_D group.
3.4. Comparison of the Performance of Both Moths
Both insects showed significant differences in their developmental times, mostly with an increased developmental time when fed with T-oaks. For T. viridana and the females of L. dispar, significant differences in the leaf area fed between S-oaks and T-oaks were observed but with different directions. While L. dispar larvae ate more leaves on S-oaks, the larvae of T. viridana needed to eat more T-oak leaves. Most striking is the difference between the insects in mortality rate. Where more T. viridana larvae died during their development than reached the adult stage, for L. dispar a very low overall mortality was witnessed (Table 4).

Table 4.
Comparison of the performance parameters of both moths. Differences between larvae from T- and S-oaks have been verified by significance tests: * = p < 0.05; ** = p < 0.01.
4. Discussion
For T. viridana, the supposedly worse physiological quality of leaves from T-oaks had no overall influence on the juvenile developmental duration, whereas the more detailed experiment with L. dispar implicated a negative effect of pre-infestation, especially in T-oaks. Thus, here, pre-experiment induced defence compounds elongated the developmental time when fed with leaves from T-oaks.
For both insects, the amount of leaf area fed has to be considered together with the total weight of the pupae. T. viridana engaged in compensatory feeding on T-oaks to overcome the negative effects of plant defence-related reduction in leaf quality, resulting in nearly the same pupal weight for larvae reared on S-oaks or T-oaks [14]. This could not be observed for L. dispar. The differences in pupae weight were correlated with differences in food intake for L. dispar (data not shown), which instead slowed down in development. However, both insects overall seemed to prefer the leaves of S-oaks. This could be explained by the repellent herbivore-induced plant volatiles (HIPVs) produced in T-oaks [14]. Rossiter et al. [25] found constitutive and elevated phenolic and nonphenolic constituents to be partly responsible for the reduced performance in L. dispar. This supports the hypothesis of weaker development of larvae because of strong defence signalling in pre-infested T-oaks (T_D), since T-oaks have elevated fractions of the herbivore-repellent herbivore-induced plant volatiles (HIPVs) of the sesquiterpenes α-farnesene and germacrene D [14]. An elevation in these volatile defence-associated emissions was demonstrated by Copolovici et al. [26] in feeding experiments with L. dispar on Q. robur as well. This would mean that, unlike the effects of T-oaks on T. viridana, the HIPVs, rather than the secondary metabolites, e.g., tannins, would have had the negative effect on L. dispar performance. In T. viridana, on the other hand, the tannins may be the reason for the high mortality rate, because it is proven that phenolic compounds in the guts of insects lead to the destruction of essential nutrients and subsequently to damage of the midgut [27].
5. Conclusions
In an earlier study, we hypothesized that T- and S-oaks follow different defence strategies. Where T-oaks prefer a constitutive way of defence, as shown by high amounts of sesquiterpenes and bioactive tannins, S-oaks react ‘just-in-time’ to insect attacks by increasing defensive substances only after insects start feeding [14,28]. The defence strategy of T-oaks may make them a contributor to the stronger resistance of German forests against pests such as T. viridana and L. dispar, not least in regard to climate change.
Author Contributions
H.S. conceived and designed the experiments; F.O. and C.-A.W. performed the experiments and all authors analysed the data; F.O. and H.S. wrote the paper with contributions from C.-A.W. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Ethical review and approval were waived for this study, because the insects were only used for (natural) feeding experiments and are neither endangered nor protected species.
Informed Consent Statement
Not applicable.
Acknowledgments
This project was financially supported by the ‘Deutsche Forschungsgemeinschaft’ (DFG) Bonn, Germany and the ‘Bundesministerium für Ernährung und Landwirtschaft’ (BMEL) via the ‘Fachagentur Nachwachsende Rohstoffe’ (FNR). We would like to thank Susanne Jelkmann for her technical assistance during laboratory work and all gardeners for their support.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| T-oaks | oaks classified as tolerant; less infested during outbreak of herbivorous insects |
| S-oaks | oaks classified as susceptible; heavily infested during outbreak of herbivorous insects |
| VOCs | volatile organic compounds |
| HIPVs | herbivore-induced plant volatiles |
| T_C | T-oaks control |
| S_C | S-oaks control |
| T_D | T-oaks damaged |
| S_D | S-oaks damaged |
| L (combined with a number; for example, 3) | larval stage 3 or third instar |
| C | control tree |
| D | damaged tree |
References
- Riedel, T.; Stürmer, W.; Hennig, P.; Dunger, K.; Bolte, A. Wälder in Deutschland sind eine wichtige Kohlenstoffsenke. AFZ-Der Wald 2019, 14, 14–18. Available online: www.bundeswaldinventur.de/fileadmin/SITE_MASTER/content/Downloads/CI2017/AFZ_14_19_Kohlenstoff_Artikel_2_Riedel.pdf (accessed on 5 November 2020).
- Bartsch, N.; Röhrig, E. Prozesse der Walddynamik. In Waldökologie; Springer Spektrum: Berlin/Heidelberg, Germany, 2016. [Google Scholar] [CrossRef]
- Ellenberg, H.; Dierschke, H. Vegetation Mitteleuropas mit den Alpen. In Ökologischer, Dynamischer und Historischer Sicht; 203 Tabellen with Assistance of Christoph Leuschner, 6th ed.; Verlag Eugen Ulmer: Stuttgart, Germany, 2010. [Google Scholar]
- Parry, M.L.; Canziani, O.; Palutikof, J.P.; van der Linden, P.; Hanson, C.E. Climate Change 2007: Impacts, Adaptation and Vulnerability; Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2007; 976p. [Google Scholar]
- Delb, H. Modul Wald Teilbericht Waldschutz und Klimawandel; Forstliche Versuchs- und Forschungsanstalt Baden-Württemberg: Abteilung Waldschutz, 2013; Available online: www.kwis-rlp.de (accessed on 9 November 2020).
- BMEL Bundeswaldinventur. Der Wald in Deutschland Ausgewählte Ergebnisse der Dritten Bundeswaldinventur; Bundesministerium für Ernährung und Landwirtschaft: Berlin, Germany, 2014. [Google Scholar]
- Ehrlich, P.R.; Raven, P.H. Butterflies and plants: A study in co-evolution. Evolution 1964, 18, 586–608. [Google Scholar] [CrossRef]
- Becerra, J.X. Synchronous coadaptation in an ancient case of herbivory. Proc. Natl. Acad. Sci. USA 2003, 100, 12804–12807. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dengler, A.; Röhrig, E. Waldbau. Der Wald Als Vegetationstyp Und Seine Bedeutung Für Den Menschen; Verlag Paul Parey: Berlin, Germany, 1980; 283p. [Google Scholar]
- Hunter, M.D. Differential susceptibility to variable plant phenology and its role in competition between two insect herbivores on oak. Ecol. Entomol. 1990, 15, 401–408. [Google Scholar] [CrossRef]
- Hartmann, G.; Blank, R. Winterfrost, Kahlfraß und Prachtkäferbefall als Faktoren im Ursachenkomplex des Eichensterbens in Norddeutschland. Forst Holz 1992, 15, 443–452. [Google Scholar]
- Führer, E. Oak Decline in Central Europe: A Synopsis of Hypotheses. Proceedings: Population Dynamics, Impacts and Integrated Management of Forest Defoliating Insects; USDA Forest Service Technical Report NE-247; McManus, M.L., Liebhold, A.M., Eds.; USDA Forest Service, Northeastern Research Station: Radnor, PA, USA, 1998; pp. 7–24. [Google Scholar]
- Schröder, H.; Ziegler, C. Die Situation der Eiche in NRW im Frühjahr 2005. AFZ Wald 2006, 6, 320–321. [Google Scholar]
- Ghirardo, A.; Heller, W.; Fladung, M.; Schnitzler, J.-P.; Schroeder, H. Function of defensive volatiles in pedunculate oak (Quercus robur) is tricked by the moth Tortrix viridana. Plant Cell Environ. 2012, 35, 2192–2207. [Google Scholar] [CrossRef] [PubMed]
- Dicke, M.; Baldwin, I.T. The evolutionary context for herbivore-induced plant volatiles: Beyond the ‘cry for help’. Trends Plant Sci. 2010, 15, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, H.; Orgel, F.; Fladung, M. Performance of the green oak leaf roller (Tortrix viridana L.) on leaves from resistant and susceptible oak genotypes. Mitt. Dtsch. Ges. Allg. Angew. Entomol. 2015, 20, 265–269. [Google Scholar]
- Schröder, H. Sommerveredelung bei Eichen-eine Erfolgsgeschichte. AFZ Wald 2010, 5, 16–17. [Google Scholar]
- Magnoler, A. A wheat germ medium for rearing of the gypsy moth Lymantria dispar L. Entomophaga 1970, 15, 401–406. [Google Scholar] [CrossRef]
- R Core Team. R: A language and Environment for Statistical; Version 3.6.1; R Foundation for Statistical Computing: Vienna, Austria, 2017; Available online: https://www.R-project.org/ (accessed on 3 August 2020).
- RStudio Team Integrated Development Environment for, R.; Version 1.2.5001; RStudio, Inc.: Boston, MA, USA, 2019; Available online: http://www.rstudio.com/. (accessed on 3 August 2020).
- Hijmans, R.J. Raster: Geographic Data Analysis and Modeling. R Package Version 3.0-7. 2019. Available online: https://CRAN.R-project.org/package=raster (accessed on 12 February 2020).
- Pohlert, T. PMCMRplus: Calculate Pairwise Multiple Comparisons of Mean Rank Sums. Version 1.4.2. 2019. Available online: https://CRAN.R-project.org/package=PMCMRplus (accessed on 12 February 2020).
- Aho, K. Asbio: A Collection of Statistical Tools for Biologists. Version 1.5-5. 2019. Available online: https://CRAN.R-project.org/package=asbio (accessed on 8 February 2020).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016; Available online: https://ggplot2.tidyverse.org (accessed on 21 July 2020).
- Rossiter, M.C.; Schultz, J.C.; Baldwin, I.T. Relationships among Defoliation, Red Oak Phenolics, and Gypsy Moth Growth and Reproduction. Ecology 1988, 69, 267–277. [Google Scholar] [CrossRef]
- Copolovici, L.; Pag, A.; Kännaste, A.; Bodescu, A.; Tomescu, D.; Copolovici, D. Disproportionate photosynthetic decline and inverse relationship between constitutive and induced volatile emissions upon feeding of Quercus robur leaves by large larvae of gypsy moth (Lymantria dispar). Environ. Exp. Bot. 2017, 138, 184–192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barbehenn, R.; Cheek, S.; Gasperut, A.; Lister, E.; Maben, R. Phenolic compounds in red oak and sugar maple leaves have prooxidant activities in the midgut fluids of Malacosoma disstria and Orgyia leucostigma caterpillars. J. Chem. Ecol. 2005, 31, 969–988. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kersten, B.; Ghirardo, A.; Schnitzler, J.P.; Kanawati, B.; Schmitt-Kopplin, P.; Fladung, M.; Schroeder, H. Integrated transcriptomics and metabolomics decipher differences in the resistance of pedunculate oak to the herbivore Tortrix viridana L. BMC Genom. 2013, 14, 737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).