Abstract
Algal biomass is a prospective feedstock for the eco-sustainable production of many different products with added value, such as meals, feeds, and fuels. The remaining biomass from the algae can be used as raw material and can be transformed into useful secondary products after the important macromolecules have been removed. By optimizing algal biomass hydrolysate utilizing microbial fermentation, several studies demonstrated the generation of bioenergy (bioalcohol, biogas, and biohydrogen) and biochemicals (organic acids and biopolymers). Since the harvest and maintenance of sustainable algal cultivation incur considerable energy and economical prowess, developing products from algae remains a challenge to be countered in commercial applications. This is a typical bottleneck issue when processing algae for fuels or chemicals at the pilot scale. Implementation of integrated algae biorefinery methods can substantially reduce the cost of production and energy consumption. An algae-based green economy can be financially more viable and utilizable, especially for countries with weaker economies. This review’s goal is to examine the implementation of integrated biorefineries for the recovery of bioproducts generated from algae and potential applications. In this context, the life cycle analysis and business elements of a unified algal biorefinery are also addressed.
1. Introduction
Microalgae are adaptable unicellular/multicellular organisms that are common and widely distributed, having considerable significance in biotechnological and environmental research. Algae are photosynthetic organisms, capable of converting solar energy into biomass and oxygen [1].
Algal biomass typically consists of the three main macromolecules—carbohydrates, lipids, and proteins with a trace amount of minerals [2]. The remaining biomass can be utilized for the production of secondary products like biogas, butanol, ethanol, etc., by fermentation, once the important macromolecules have been removed from the algae [3]. By valorization of algal biomass hydrolysate employing microbial fermentation, several studies demonstrated the generation of bioenergy (biogas, biohydrogen, and bioalcohol) and biochemicals (biopolymers and organic acids) [4,5].
The two main issues that civilization is currently experiencing on a global scale are environmental sustainability and the energy crisis brought on by an excessive reliance on fossil fuels. Global warming is happening due to the enormous quantities of greenhouse gases (GHGs) emitted into the atmosphere because of fossil fuel usage. As a result, many scientists are working to identify additional renewable energy sources. The conflict between using food and fuel has been sparked by the application of first-generation lignocellulosic biomass feedstocks (LBF) for biofuel production. Food prices may continue to rise as a result of the growth of biofuels [6]. Therefore, second-generation biofuels have been made using LBF obtained from forestry and agronomic residues. Due to this complex structure, the viability of lignocellulosic biomass for the production of biofuels is constrained by the time-consuming and expensive pretreatment required [7]. To process third-generation biofuels, which could be a solution to these issues, algae can be considered as a potential feedstock.
Biorefineries aim to switch from petroleum to locally available renewable raw materials in order to create a robust and sustainable bio-based industry. There are two categories for the development of biorefineries [8]: the first is the energy goal, and the second one is the economic goal in this regard [9]. Basic criteria for a successful biorefinery include, among other things, providing food and feedstock, maintaining biomass regeneration and diversity, limiting ecological effect, responding to market swings, and converting various feeds and multi-product conversion processes [10].
The demand for electricity has increased tenfold globally because of the growth of human resources and economic expansion. One resource that might help the energy sector balance supply and demand is bioenergy [11]. Unconventional energy sources, like bioenergy, perform better than fossil fuels in practically every consumption metric [12]. The efficiency and effectiveness of the entire process could be increased by implementing nanotechnologies in bioenergy refineries. An appropriate nanotechnology-based method could reduce feedstock availability limitations and could increase the collection of biomass after harvesting by several notches. Additionally, it suggests generating extra bioenergy from different sources to supplement the bioenergy produced by certain algae species thriving on underutilized water resources [13]. Special focus is given on microbial biomass for the production of bioenergy due to some of its beneficial properties [14]. Microalgae provide a number of benefits, including quick development and a brief harvesting cycle. This renders them an interesting source of biomass for biofuel production [15]. It is the need of the hour to find a solution to address the issue of commercial-scale feedstocks availability for the production of bioenergy [13].
In aquatic environments, green algae can survive and grow into the main facilitator. The lowest amount of nutrients is used by algal biomass relative to its production. According to [16], nitrogen consumption is approximately 40, 45, and 0.003 mg/hectare for sunflower, rapeseed, and algae cultivation, respectively, to produce 1 kg. biomass/hectare, which is remarkably low for algae cultivation. The phosphorus requirement for rapeseed, sunflower, and algae is approximately 25, 56, and 0.002 mg/hectare, respectively. Due to its diverse biochemical composition (carbohydrates, lipids, and proteins), algal biomass can be used to produce a variety of economically significant bioproducts. In recent years, downstream biorefineries have been integrated to simultaneously produce algae-derived biofuels and biochemicals. Due to the limited supply of agricultural land, cultivating algae in marine ecosystems could potentially help to meet the world’s rising energy demand [17].
Microalgae biofuel production has blossomed and become more economically viable because of recent advancements in technologies in photobioreactor design via integrated algae farming and biorefinery strategies [18]. Algal biomass can be utilized for the production of different macromolecules, such as lipids, fatty acids, and essential amines, before being transformed to biofuel. High-value products produced by an algae biorefinery system include biomaterials, biofertilizers, and pharmaceuticals, among others [19]. Algae biorefineries have grown closer to becoming a commercial reality by incorporating biofuels and natural processing. Micro algae can be utilized to make goods with a high economic value that are used in a variety of sectors, such as food, nutraceuticals, cosmetics, and pharmaceuticals. An integrated bio refinery, which generates most of the products from a single biological feedstock, can be used to achieve this. The idea of a biorefinery was initially developed by petroleum refineries. Fuels, oils, and other goods produced by biorefineries are useful for the chemical industries [20]. To make the most of all the raw resources and increase the output of the finished products, a number of techniques can be used. It is a huge problem to find a method to produce these products in an algae-based biorefinery that is both ecologically sound and sustainable.
This review focuses on existing methods for effective and profitable utilization of microalgae using a biorefinery concept on the basis of this issue and its constraints. A microalgal biorefinery approach that is sufficiently executed to produce biofuels and multiple product recovery has also been described, concentrating on technical and financial issues. In addition to this debate on life cycle evaluation and techno-economic analysis of integrated algal biorefinery, this review provides an exhaustive note on current methodologies of algae pretreatments, biofuel generation, and value-added product recovery as a part of an integrated concept.
2. Technologies for Algal Biomass Conversion
Proteins, lipids, and carbohydrates are important biochemical components of algal biomass and can be utilized to produce biofuel. Algal biomass has received much attention recently as a potential source for biorefineries. Algal biorefineries use algae biomass as a feedstock to make biofuels and chemicals with higher value [21]. Carbohydrates, lipids, and proteins are among the biochemical elements of algal biomass that are converted into biofuels. Algae can be used to make biofuels and bioproducts, as shown in Table 1 [22]. In terms of commercialization, pigments from microalgae have a high revenue generation > USD 1 billion (selling price—USD 400/kg) and the global carotenoid market is expected to be USD 1.5 billion in 2019 to USD 2.0 billion by 2022 [23]. Over the past 20 years, the commercial manufacture of omega-3 polyunsaturated fatty acids (PUFAs) using biotechnology has replaced fish oil in many applications. Due to its excellent product safety and prospective sustainability, as well as growing awareness in the developing vegan market, the microorganism production of PUFAs has increased in comparison to that of fatty fishes. The majority of the microbial production of omega-3 is now conducted under heterotrophic conditions utilizing standard fermentation technologies, despite the fact that autotrophic production by microalgae appears to be more long-term sustainable [24]. Several researchers have reported the production of essential omega fatty acids from strains D. tertiolecta and N. oculata. Palmitic, linoleic, oleic, palmitoleic, and eicosanoic acids are among the fatty acids produced from Nannocholoropsis sp. as well as Dunaliella sp. [25]. However, a diverse microalgal germplasm still exists and should be fully explored for carotenoid productions. The diatom Phaeodactylum tricornutum, a promising source of polyunsaturated eicosapentaenoic acid (EPA), and the carotenoid fucoxanthin were grown using three food wastes in a study reported by Russo et al. (2023). According to the experimental findings presented in this work, some food by-products could be effectively used in biorefineries to produce fucoxanthin and other high-value chemicals like eicosapentaenoic acid (EPA) [26]. To obtain a sustainable omega-3 supply that can compete with fish oil, it is imperative to lower the cost of DHA algal oil manufacturing. For the purpose of producing a DHA-rich thraustochytrid from food industry waste and byproducts, a techno-economic analysis was conducted by Russo et al., 2022 [27]; the wastes were cheese whey, beetroot molasses, and corn steep liquor. Algal biomass is processed upstream, in bioreactions, and downstream to develop the final products. The goals of biomass-based biorefineries are to maximize profitability, minimize waste, and optimize resource utilization. Algal biomass can be recovered using three different processing methods, and algae can be grown using recycled wastewater streams and waste carbon dioxide. According to extensive research, the type of algae grown upstream, which tends to favor accumulating biomass quickly and at high density, determines the efficiency of biochemical reactions and downstream processes. An appropriate process-oriented algae strain should be chosen in addition to taking the mechanical and physicochemical properties of algae cells into account. As a result of the algae’s resistance to shear stress and contamination brought on by the cultivation system, they can be kept in the medium for repeated cycles without being significantly affected by the environmental conditions [28,29]. The various useful bioproducts that might be generated at the algal biorefinery are expanded by biomass productivity volume. Additionally, it is crucial to choose algae that can separate well and that do not necessitate a significant amount of energy to destroy cells [30].

Table 1.
Bioproducts and biofuels from algae.
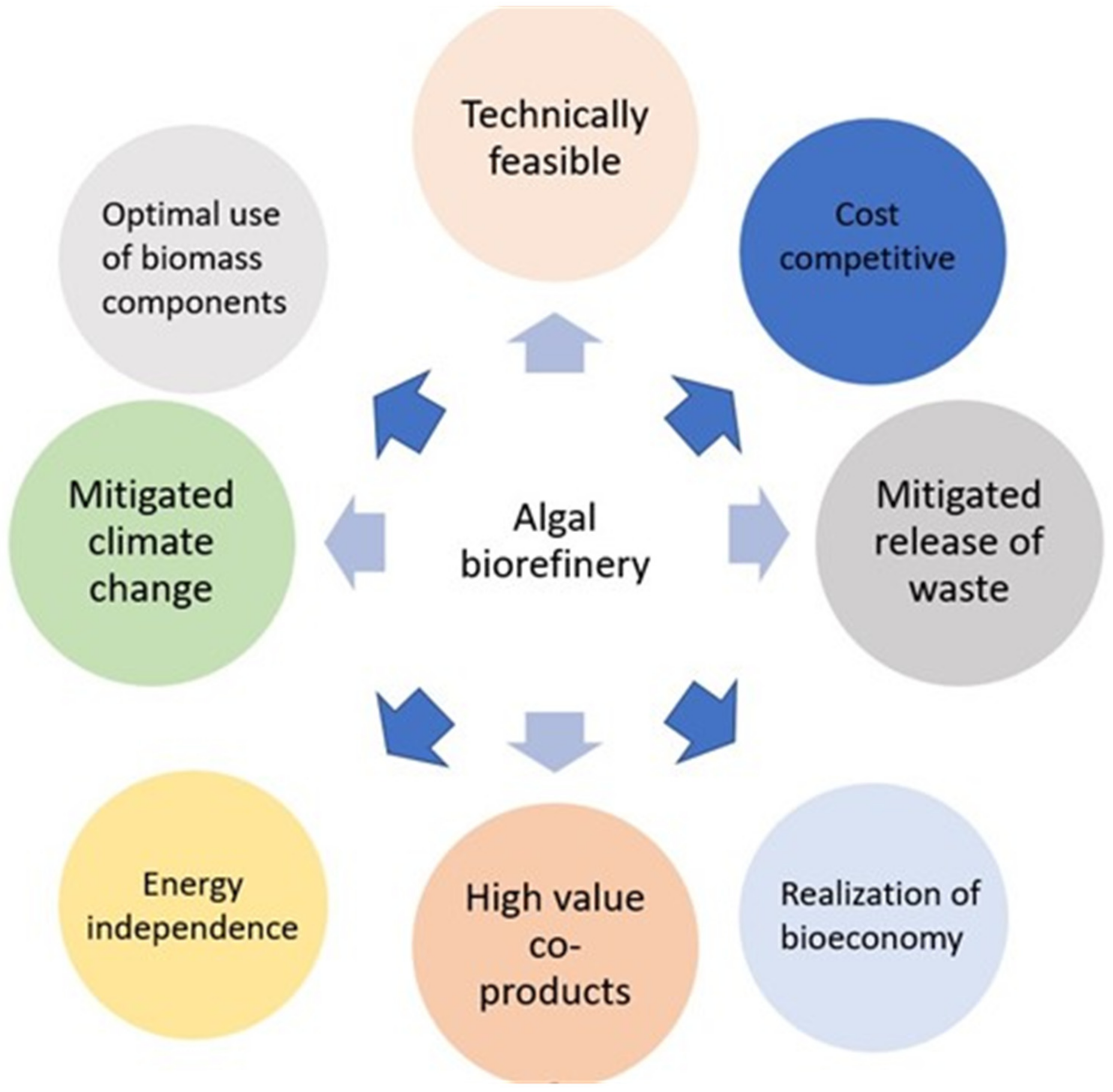
In addition, the selection of appropriate algal strains can be influenced by the agronomy types. Algae are grown using photobioreactors (PBRs) and open raceway ponds (ORPs). There are specific targeted parameters for each system. Competition in the ORP cultivation conditions and other micro-orgasmic contaminants can have an impact on the persistence rate of algal cells [35]; the use of an algae strain that is resistant to bacteria and predators is therefore advised. Algal adhesion to PBR walls is the primary challenge in PBR cultivation [36]. Choosing algae species with highly suspended characteristics is one approach in this regard. It has been clear from the beginning that the bioprocesses upstream in the cultivation of algae play a crucial role in determining the downstream outcomes. Depending on the cultivation conditions, the biochemical characteristics of an algal biomass define it. By adjusting the parameters of algae cultivation, it is possible to manage the allocation of biochemical properties and meet consumer demands. Based on all the aforementioned factors, pre-screening is currently being conducted to choose appropriate algae strains. The aimed biorefinery pathways will then be used to genetically modify these algae strains. Using an algal biorefinery as fully as possible, as shown in Figure 1, could lead to the development of value-added products by the methods of fermentative valorization, biochemical conversion, thermochemical conversion, thermochemical treatment, etc.
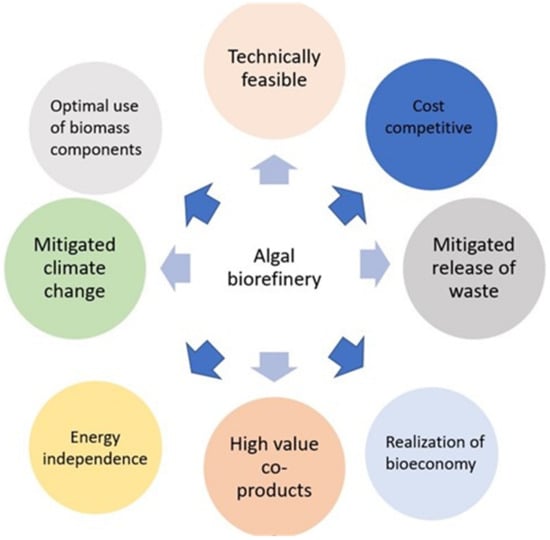
Figure 1.
Potential route map of algae biorefinery.
2.1. Fermentative Valorization
In order to maximize the recovery of algal resources as a variety of biofuels (like bioethanol) and value-added compounds, fermentation—a productive valorization method—uses algae as a promising renewable feedstock. The appropriate biomass pre-treatment and valorization techniques are essential for maximizing the hydrolysis of sugar, carbohydrates, and biofuels. To expedite the high-throughput fermentation process, pre-treatment and hydrolysis of the biomass must be coupled. The fourth generation of biofuels, known as next-generation biofuels, are produced through biorefining and rely on cutting-edge technological processes like photofermentation or combined algal biomass processes using genetically modified microalgae [37]. Due to its zero-waste approach, high-throughput fermentation is a novel bioethanol production technique. Chlamydomonas mexicana was subjected to successive fermentation processes by El-Dalatony et al. (2019), and the leftover biomass was transesterified, leading to the simultaneous extraction of carbohydrate, protein, and lipid [38]. In addition to producing bioethanol, high-throughput fermentation can also be utilized to manufacture various types of biofuels while minimizing expenses and waste generation. The fermentative byproducts of serialized valorization exhibited a profile resembling that of methods for isolating whole cells [1]. The different types of high-throughput fermentation, such as dark fermentation combined with photo-fermentation, anaerobic digestion to produce biomethane, and the production of hydrogen from algal biomass, were reviewed by Xia et al. (2015). After dark fermentation, the organic waste (acetates, proteins, and acetate) can be further processed through a photofermentation to produce usable fatty acids [39].
2.2. Biochemical Conversion Technologies
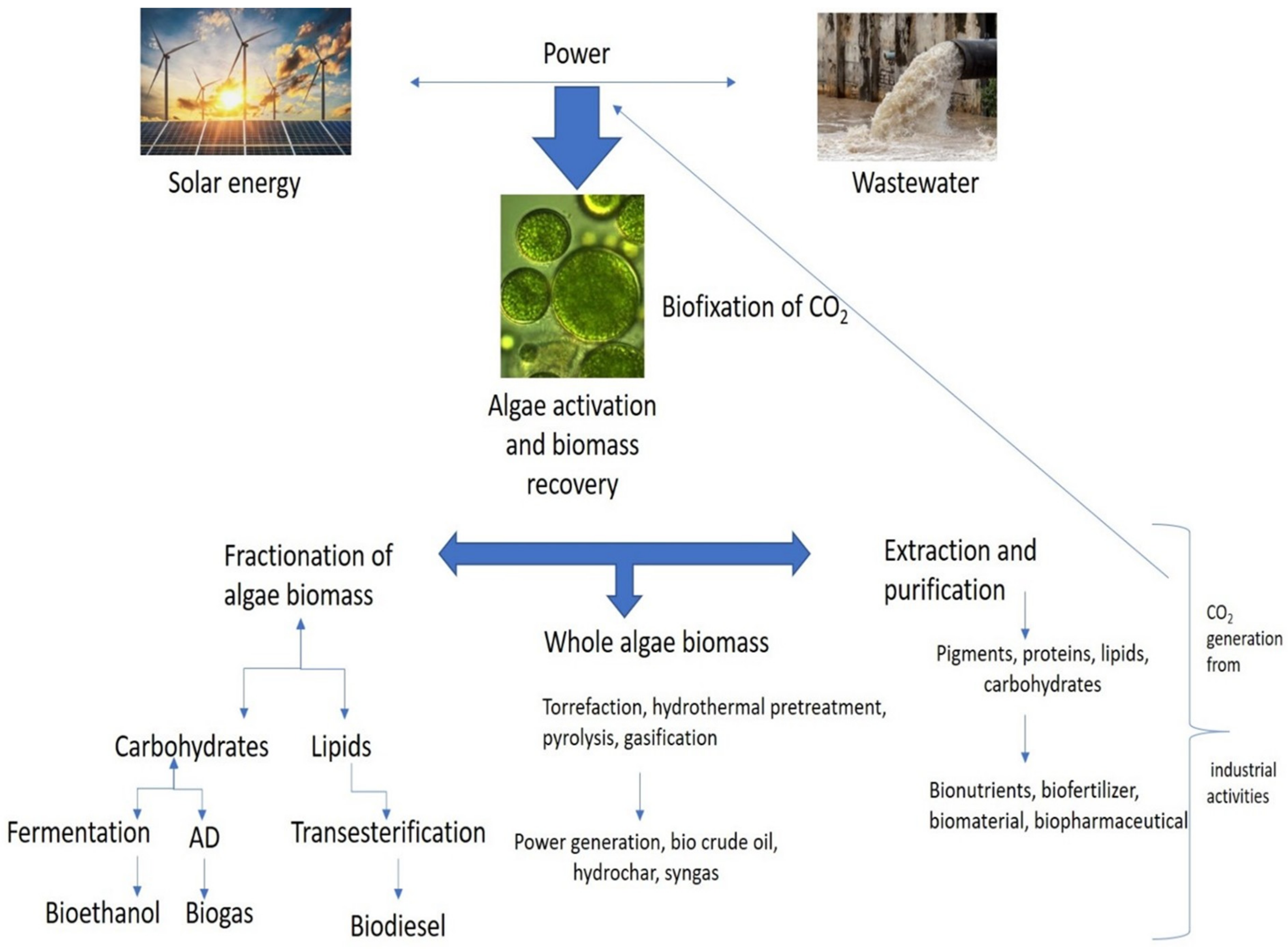
Important steps in the biochemical conversion process include selecting algae species with anaerobic digestion of organic feedstock and fermentation of bioethanol, biohydrogen, and bioplastics using bacteria and other microorganisms. Due to the complexity of algae cell walls, enzymes find it challenging to hydrolyze or produce anaerobic substrates, which results in very low yields. Numerous pretreatments and disintegration techniques have been employed to address this issue [40]. Algal biomass has been converted using a variety of biochemical processes to produce biofuels. According to Bhatia et al., 2020, microalgae cultivated on a mixed substrate produced approximately 17 mL/g COD during a process of anaerobic digestion (municipal wastewater) [12]. The biohydrogen was produced by Kumar et al. at laboratory scale using diverse microalgal species with concentration of 2.50 g/L of algal biomass [41]. Some scientists have used pilot-scale reactors to boost productivity and yield. For instance, in high-rate algal ponds, Passos et al. (2015) observed that microalgal biomass produced about 0.3 L of methane/gm. of VS of biomethane [42]. Depending on the substrate, the cultivation conditions, and concentration of biomass, different microalgal species will produce biofuel to varying degrees. According to Ho et al., the laboratory-scale reactor of 1 L was used to grow the microalga Chlorella vulgaris in synthetic growth media, which produced approximately 1.35 g/L of biomass and 3.50 g/L of bioethanol [43]. Scenedesmus dimorphus produced 0.26 g/g bioethanol in a 2 L laboratory scale bioreactor when cultivated on the same cultivation medium. Algae biorefineries incorporate both upstream and downstream bioreactions, as depicted in Figure 2. They integrate upstream and downstream processing. With the development of algae cultivation, it is possible to revive waste CO2 and wastewater streams through three different processing routes [44].
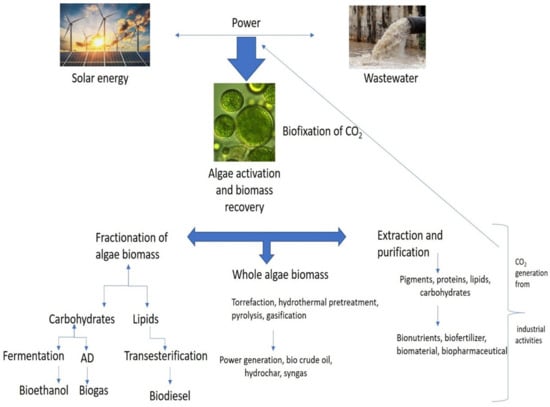
Figure 2.
Algae biorefineries.
2.3. Anaerobic Digestion (AD)
Anaerobic digestion techniques use the microalgae’s rich organic load to produce biofuels (Figure 2). Algae’s biochemical makeup makes them a valuable anaerobic digestate for use with bioenergy technologies and waste management [45]. Algal digestate is a highly helpful source of nutrients for large-scale algal farming to assist the growth of the microalgae Chlorella vulgaris (nitrogen, phosphorus, and potassium) [46]. Since AD uses renewable substrates for production of bioenergy, biodiesel, green diesel, bioethanol, and hydrogen, the AD of microalgae has recently been looked at as a prospective substitute. To develop biogas production systems competently and affordably, numerous techno-economic constraints must be overcome. These bottlenecks include higher expenses for biomass production, a slower conversion of biomass to biogas, limited microalgal biodegradability, and increased sensitivity of the methanogens used [47]. Nevertheless, the biogas generation yield can be improved by applying metabolic and genetic modification methods. The hydrogen yield during the acidogenesis step can be increased by using the proper pretreatment procedures to remove H2 overpowering homoacetogens from the inoculum and fermentation media.
Co-digestion of microalgal biomass with energy-dense waste resources, such as forest litter and agricultural waste, could increase methane yield and combat ammonia inhibition by achieving a balance in the C/N ratio [48]. Reviews that focus on microalgae’s AD using the biorefinery technique have become scarce in recent years. The AD of prokaryotic and eukaryotic microalgae, as well as strategies for boosting biogas and bioenergy output, are thoroughly covered in this paper. The co-digestion of microalgal biomass by microorganisms and the AD of microalgae’s removal of nutrients and microcystins have also been investigated.
In anaerobic co-digestion, two or more feedstocks are broken down. Financial viability, increased bioenergy output, and fewer bottlenecks in the mono-digestion process have all been demonstrated by technology. Anaerobic co-digestion, for example, enhances conventional AD technology by addressing the related issues, such as nutrient overload, retention of dangerous compounds, or resistant molecules in the feedstock. Microalgal biomass could be digested along with other agricultural wastes to boost the production of biogas. Microalgae can be co-digested with wheat straw in the ratio of 1:1, boosting the methane generation by 77% as compared to single digestion of microalgae biomass. Moreover, methane production was increased by 15% as a result of pretreating the biomass [49].
AD is used for anaerobic degradation of algal biomass [50]. Algal biomass can also be digested during anaerobic digestion in addition to biogas (e.g., biomethane and biohydrogen) [51]. Various microbial species engage in interactions with substrates (like lipid-rich algae biomass) during AD [52,53]. Yang et al. (2011) used the lipid-extracted Scenedesmus biomass to produce biohydrogen and biomethane in two steps [54]. A thermally treated algal sludge was utilized for biohydrogen production, and the process’s effluent was used in biomethane production. In order to produce biofuels and treat wastewater, Deng et al. (2017) also evidenced an algae biorefinery using Chlorella vulgaris grown under thermophilic conditions in pretreated AD swine manure. Algae produced 1.0 g/L of biomass while removing nitrogen and carbohydrates with 99% and 54% efficiency, respectively [55]. There are, however, always inhibitors that prevent AD from working effectively. Lakaniemi et al. investigated the ability of fish algae in anaerobic biohydrogen production and biomethane in freshwater and marine environments [56]. According to a group of researchers, long-chain fatty acids (LCFAs) are the primary inhibitors of the AD process [57]. In the biomass of Nannochloropsis salina, there was a negative association between lipid concentration and microbiological activity. LCFAs limit the production of biomethane by bacteria and methanogens by accumulating volatile fatty acids (VFAs).
2.4. Thermochemical Conversion Methods
Instead of using biological or chemical processes, biorefinery processing with thermochemical upgrading converted bioenergy from whole biomass. Examples of thermochemical technologies include pyrolysis, gasification, and hydrothermal treatment [58]. In addition to achieving good energy efficiency, hydrothermal processing of wet biomass performs remarkably well in a number of processes, including enhanced decarboxylation, hydrodeoxygenation, and fast hydrolysis [59]. To enhance the energy efficiency of hydrothermal processing by up to 85%, the warm effluents from the reactor can also be utilized to heat the feed stream [60]. This contrasts with the traditional conversion method that uses dry biomass and conducts the reaction using steam at a lower temperature. As a result, the procedure and the evaluation of thermal performance are impacted by the limited possibilities for thermal energy recovery.
2.5. Hydrothermal Treatment
Hydrothermal processing is a feasible option for treating wet organic waste, where elimination of the energy-intensive pre-drying stage (required for conventional treatments) is possible and can yield a variety of goods. Other advantages noted in the literature include low activation energy requirements and flexibility in producing high-quality char, oil, and gas [61,62]. This is due to the fact that water can function in this process as a reactant medium and aid in the reaction’s hydrolysis [63]. Scenedesmus and Spirulina algal biomass were subjected to slow pyrolysis and hydrothermal liquefaction, and the results revealed that the bio-oils produced by these two procedures have similar elemental compositions [64].
However, in the case of wet biomass with 80% moisture, it was found that hydrothermal treatment was more advantageous energetically [65]. Hydrotreatment required less energy than pyrolysis for wet biomass since pre-drying was required to eliminate the water in the latter process. Moreover, hydrothermal treatment eliminates all biological issues—including pathogens—from the waste, due to natural autoclaving at temperatures above 121 °C [66]. The hydrothermal treatment of biomass wastes has been the subject of several review publications [67,68]. In the most recent comprehensive reviews on the hydrothermal processing of waste biomass, it is discussed how biomass constituents react in hydrothermal media and how process conditions affect it [69].
2.6. Gasification and Pyrolysis
Gasification is a process that turns carbon-rich feedstocks into syngas by partially oxidizing them at temperatures between 100 and 1000 °C in the presence of a finite amount of oxygen or air or steam. The main gases that make up syngas are CO2, CO, H2, and CH4 [70]. Additional syngas produced in this way can be converted to hydrogen through the water–gas shift reaction or lower-range liquid hydrocarbons through Fisher–Tropsch synthesis [71,72]. After the algal species were completely gasified, the syngas was created at 700 °C. Hydrogen content in Chlorella vulgaris was found to be high when Ru/TiO2 was used as a catalyst [73]. After gasifying the biomass of N. occulata, [74] obtained syngas with the following composition: 40–53% H2, 30–40% CO2, 10% CH4, and 6% CO. The catalyst is crucial for biomass-to-syngas conversion because it speeds up tar degradation and makes hydrogen production easier. According to [75], the complete gasification of the algal species Spirulina platensis in the presence of Ru as catalyst resulted in syngas that was primarily made up of CO2, CH4, and H2. According to [76]’s comparative studies of Saccharina latissimi, C. vulgaris, and S. platensis for the production of hydrogen, Saccharina latissimi produced a higher yield of hydrogen during the gasification process.
Pyrolysis is a process where the biomass is heated without air in a specific temperature range (400 to 600 °C) in order to produce energy [77]. Algal biomass pyrolysis has received much attention in recent times because of its high efficacy and environmental advantages. In a manner similar to the hydrothermal processes, the end-products of pyrolysis are divided into three categories in addition to the breakdown of large molecules into smaller molecules through the pyrolysis reaction [58]. These include biochar made at slower heating rates (5–10 °C per minute) and longer reaction times (10–30 s), as well as bio-crude oil with tars and related products made at a more moderate pyrolytic temperature (400–500 °C for 2–3 s reactions). In addition, they comprise biogases produced at advanced heating rates (10–600 °C/s) and in shorter reaction times of 1–2 s. Using macroalgae, having high nitrogen and ash contents of about 6.10 wt%, a group of authors were able to create pyrolytic bio oil [78]. Between 37% and 47% of the bio-oil was produced at approximately 500 °C. In addition, the bio-oil production’s general advanced heating value (HHV) was comparable to that of traditional terrestrial biomass, which is approximately 25 MJ/kg for Laminaria, 22 MJ/kg for Porphyra, and 20 MJ/kg for Undaria. In terms of quality, pyrolytic bio-oil containing nitrogen could be used as a chemical feedstock for high-value chemical components or as a biofertilizer [79]. Additionally, an efficient method of slow pyrolysis was used to combine biochar and bio-oil from Spirulina species (0.49). This provided more information about the potential for pyrolysis-based algal biorefineries.
2.7. Direct Combustion Techniques
Most studies on dry extraction use combustion techniques and pyrolysis [80,81]. Compared to wet-extraction techniques, these methods produce less energy recovery and have a lower ecological viability. The assessment study carried out by Bennion et al. in 2015 revealed that dehydrating microalgae before the pyrolysis step and heating during the pyrolysis reaction significantly increase the demand for fossil fuels [82]. This led to an increase in the dry (pyrolysis) extraction pathway’s GHG emissions. According to Sun et al., 2019, the drying techniques applied in pyrolysis methods lead to considerable emission of GHGs [83]. Furthermore, drying microalgae with a moisture content below 50% is necessary for the indirect combustion process to achieve high heat conversion efficiency, which is a labor- and energy-intensive process. Despite having a lower global warming potential than conventional diesel fuel, biomass has a lower calorific value (14–16 MJ/kg), which makes it less competitive [84]. The selection of an efficient and suitable drying technology is necessary to produce microalgal biofuels by dry extraction process.
3. Algal Biomass Derived Value-Added Products
3.1. Production of Biodiesel
According to the biorefinery route for typical chemical conversion, algal lipids can be converted into biofuel (biodiesel) [85]. Algal crude lipids include free fatty acids (FFAs) and triacylglycerol (TAGs), which can be transesterified with alcohol to produce fatty acid alkyl esters [86]. After the algal-lipids were retrieved using chemical solvents, but before the traditional transesterification method was used, an optimization study was carried out [87].
With the aid of catalysts such as acids and catalysts (HCl, H2SO4, NaOH, KOH), among other inorganic heterogeneous catalysts, transesterification can be accomplished (ZrO2, TiO2). A study compared the effectiveness of transesterification on 22% lipid of Chlorella sp. using homogeneous acid (H2SO4) and alkaline (NaOH) as catalysts. According to the findings, acid catalysts had a yield of 60% or higher for converting biodiesel [88]. In order to increase biodiesel production, the acid catalyst first converted free fatty acids into esters and then converted them again [43]. An unwanted side reaction that slowed transesterification was the formation of soap as a result of the alkaline catalyst [57]. According to Rahman et al., 2017, Spirulina maxima is normally transesterified and esterified during homogeneous acid–alkali catalysis [89]. FFA content in algal oil was initially reduced using a catalyst (H2SO4). The transesterification was then followed by another procedure with KOH serving as the catalyst. In this study, we were able to demonstrate that the dual-step process increased the yield of biodiesel by 87% when compared to 90% biodiesel from rapeseed oil.
In addition to a catalytic process, transesterification using supercritical fluids such as water or CO2 may be another approach to turning lipids into biodiesel [90]. Unlike catalytic transesterification, the uncatalyzed method of conversion necessitates high temperatures and pressures. In comparison to traditional processes, supercritical transesterification has a number of benefits, including the absence of a catalyst, faster reaction times, purer biodiesel, and reduced environmental impact [91]. Nan et al. (2015) studied biodiesel production by utilizing Chlorella protothecoides, using a noncatalytic transesterification process [92]. The optimal biodiesel yields ranged from 87–90%, according to the findings. A uninterrupted method for non-catalytic transesterification with supercritical CO2 was also recently described [93]. As examples of algae with various lipid contents, Chrysophyta microalgae (11 wt% lipid) and Chlorella sp. were studied. The effectiveness of continuous transesterification was evaluated against oil samples with less than 19% lipid. The highest yields of biodiesel were produced by Chrysophyta and Chlorella species.
Additionally, in-situ transesterification is considered to be an alternative to the conventional approaches, which simultaneously involves lipid mining and catalytic transesterification [94]. Using water as the reacting medium, supercritical in-situ transesterification (SC-IST) can make biodiesel directly from wet algal biomass. Levine et al. (2010) produced 79% of fatty-acid ethyl ester (FAEE) from Chlorella vulgaris (53.3% lipids) using supercritical water and ethanol in place [95]. Research on the microalga Nannochloropsis oceanica has shown that biodiesel can be produced, with a final yield of 90% fatty acid methyl ester (FAME) [96]. Additionally, it has been claimed that chlorella can indeed be transesterified dry in the presence of both methanol and sulphuric acid as catalysts, yielding FAME yields of more than 90% [97].
Biorefineries use algae biomass as a feedstock to produce bioethanol, animal feed, as well as pharmaceuticals after extracting the lipids from it. A conclusion can be drawn that the wet method of in-situ transesterification is better for algae biorefineries because it avoids the drying process based on a comparison of the dry and wet methods. However, because supercritical conditions call for high pressures and temperatures, they rendered this less necessary than the catalytic process. As a result, it is anticipated that the production of algae biodiesel could be aided by a catalytic transesterification procedure in algae biorefineries.
3.2. Production of Biohydrogen
Over the past few decades, focus has been placed on the production of hydrogen by photosynthetic algae under oxygen-rich, hypoxic, or anoxic conditions. The commercialization of biohydrogen from algae has been initiated and implemented in several regions of the world [1]. In a 2011 study, Biller and Ross reported that algal biomass was hydrothermally pressed to make biofuels in the forms of liquid bio-oil, solid hydrochar, and gaseous forms of biohydrogen/biomethane [31]. Hydrothermal processes are further classified according to their operating conditions into liquefaction, gasification, and carbonization, where the proportions of the biofuel end products vary [98]. Temperature and reaction time are believed to be factors influencing the physicochemical properties of algal biofuels [99]. Thermal carbonization (HTC) is a benign process that produces solid hydrochar as the main byproduct under light pressures and temperatures (190–275 °C) [100]. In most cases, hydrothermal liquefaction (HTL) occurs at pressures between 45 and 160 bar and temperatures between 245 and 460 °C to produce viscous bio-crude oil. [101]. In contrast, hydrothermal gasification (HTG) needs higher and severe thermal conditions at temperatures range of approximately 400–800 °C in order to produce biogas, beyond the critical point of water. Dark fermentation (without a light source) and photofermentation (with light source) are additional categories for biological hydrogen fermentation. Typically, dark fermentation is used to produce biological hydrogen. However, some researchers advise pretreating algae before dark fermentation to promote carbohydrate polymer hydrolysis. Ruiz-Marin et al. implemented a number of color light energies for the cultivation of immobilized algal biomass and the production of biohydrogen in 2020 [102]. By using municipal wastewater as a growth medium, Scenedesmus obliquus and Chlorella vulgaris produce more biomass. The researchers looked at the connection between algal growth and biohydrogen output using two different light colors. In comparison to algae produced under purple light, blue light-grown algae have larger biomass concentrations. The amount of biohydrogen produced by algae grown under purple light was roughly 130 mL/L as opposed to blue light’s 60 mL/L. In Table 2, a compilation of previous hydrothermal studies of algae biomass has been summarized.
Similarly, in order to produce biohydrogen, Kumar et al. (2018) used diluted swine manure to ferment mixed algae. The maximum biomass concentration was obtained at a dilution ratio of 5 g/L of swine manure, or about 2.57 g/L [41]. In addition, for algal cultivation, the biomass concentration fell as the dilution ratio rose. The harvested biomass was then fermented in the dark to produce biohydrogen. Using a loading rate of 5 g/L of swine manure, the maximum hydrogen yield was around 13 mL/g of algal biomass. Scenedesmus obliquus and Chlorella vulgaris were cultured in tubular photobioreactors using urban wastewater in a 2015 study by Batista et al. to produce biohydrogen. The highest yields of biohydrogen per gram of VS were reported to be produced by Scenedesmus obliquus and Chlorella vulgaris, at 41 mL and 57 mL, respectively [103].

Table 2.
Hydrothermal treatment technologies of algal biomass in absence of water.
Table 2.
Hydrothermal treatment technologies of algal biomass in absence of water.
| Hydrothermal Treatment | Algae Type | Temperature (°C) | Residence Time (min) | Products | Yield (%) | Reference |
|---|---|---|---|---|---|---|
| HTL | Chlorella vulgaris | 350 | 60 | Biocrude oil | 30–35 | [31] |
| Nannochloropsis occulata | 225–300 | 30–90 | Biocrude oil | 30–55 | [104] | |
| Dunaliella tertiolecta | 280–400 | 90 | Biocrude oil | 50–90 | [105] | |
| Botryococcus braunii | - | - | - | 40 | [106] | |
| Neochloris oleoabundans | - | - | - | 40 | [100] | |
| HTC | Nannochloropsis oculata | 190–210 | 20–40 | Hydrochar | 50 | [107] |
| Chlamydomonas reinhardtii | 190–220 | 30–120 | Hydrochar | 25 | [108] | |
| Chlorella vulgaris | 150–250 | 60 | Hydrochar | 70 | [109] | |
| HTG | Chlorella vulgaris | 500 | 30 | Biogas | 40 | [110] |
| Chlorella vulgaris | 500 | 30 | Biohydrogen | 65 | [111] | |
| Nannochloropsis occulata | 500 | 60 | Syngas | 40 | [31] |
3.3. Production of Bioethanol
The most popular methods for converting algae biomass into bioethanol and biogas in algae biorefineries are fermentation and anaerobic digestion. To make the carbohydrates metabolizable by the microorganisms performing the fermentation, the carbohydrates must be extracted and saccharified to produce bioethanol. For the recovery of intracellular products of microalgae, such as oil and starch for the production of biodiesel and ethanol, respectively, as well as added-value compounds, cell disruption is frequently necessary [112]. Algal biomass contains carbohydrates that can be successfully hydrolyzed to create simple sugars [113], which will then be fermented for bioethanol production [114]. On the other hand, AD is the anoxic degradation of decomposable chemicals. Anaerobic digestion is the breakdown of biodegradable chemicals, while fermentation is another way for cell metabolism to obtain energy. According to Rempel et al. report’s from 2019, Spirulina platensis was fermented to produce bioethanol, and the fermentation’s leftover residue was then digested to produce biomethane [115]. An analysis of energy potential showed that fermentation coupled with anaerobic digestion produced approximately 13,900 kJ/kg as opposed to approximately 16,700 kJ/kg by direct anaerobic digestion [115]. Spirulina is a fascinating renewable energy source as a result for a range of goods. Bioethanol can be used as a substitute for fuels like gasoline because its physicochemical properties are comparable. The chemical makeup of the feedstock is more important in determining bioethanol yield. Most of the bioethanol is produced from substrates high in carbohydrates. Starch, sugar, and cellulose are a few of the polysaccharides found in algal biomass. Algal biomass can therefore be used as a feedstock to produce bioethanol [116].
Onay et al., 2019, studied different saccharification processes by using microalgal biomass from benthic wastewater (Hindakia tetrachotoma) [117]. A 1 L flat airlift photobioreactor is used to cultivate microalgae, and the media concentration is adjusted from 25% to 100% using diluted distilled water. In the case of the lowest (25%) concentration of growth media, 0.78 g of biomass is generated per liter of medium. When the growth media concentration is increased to 50%, the biomass yield decreases to about 0.6 g/L. Higher concentrations of this trend were still present. The microalgae were then hydrolyzed using enzyme, acidic, and alkaline methods. Enzymatic hydrolysis produces more bioethanol with a yield and substrate conversion of about 11.2 grammes per liter. El-Mekkawi et al., 2019, described a similar pilot-scale cultivation of algae for the production of bioethanol in a nearby wastewater treatment facility [116]. According to estimates, carbohydrates make up 45% of the harvested algal biomass (Microcystis sp.). Prior to yeast fermentation, this algal biomass was acid hydrolyzed, yielding about 19 grammes of bioethanol per liter. Phwan et al., 2019, tested various acid concentrations and levels with microalgae to produce bioethanol [118]. Their study focused on sulfuric acid and acetic acid, both of which ranged in concentration from 1–9%. The highest yields were measured at 0.2 and 0.281 g/g, respectively, at 5% sulfuric and acetic acid concentrations. When acetic acid and sulfuric acid were compared for the purpose of enhancing the production of bioethanol from microalgae, it was found in their study that sulfuric acid provided the best bioethanol yield in all concentrations. According to the study’s findings, using strong acids increased bioethanol production while requiring less chemical input and energy.
Figure 3 shows various applications of value-added biofuels production from various algal biomasses [119].
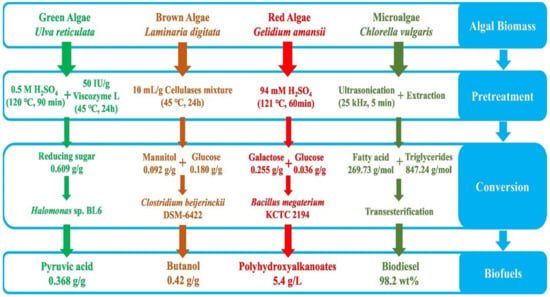
Figure 3.
Application of value-added biofuels production from various algal biomass (wt: weight). Reprinted with permission from Elsevier [119]. License number 5490240705329.
3.4. Production of Biochar (BC)
BC is a carbonaceous substance created when biomass is thermally treated at a moderate temperature with a constrained amount of oxygen present. By using the HTL process, wet algal biomass is converted into algal BC for a brief period of time at a moderate temperature [120]. According to several studies, the relative yield of algal BC is higher than that of other feedstocks per unit algal biomass. The range from 8.1–62.4% represents the yield of BC per unit dry weight of the macroalgal biomass [121]. When the pyrolysis temperature was raised from 300 to 750 °C and the pyrolysis time was extended from 10 to 60 min, the yield decreased [120]. The synthetic magnetic BC is well suited for use as an electrode in Li-ion batteries due to its large surface area (296.4 m2 g−1) and enriched carbonaceous structure. The synthesized magnetic electrode had better cyclic stability and a higher initial specific discharge capacity (740 mAh g−1) than the BC electrode, according to the electrochemical analysis [122]. This process is intriguing for the future expansion and investigation of algal biomass as a feedstock because it produces biofuels and BC simultaneously using biorefinery methods. However, the preparation of BC from algal biomass and its use are the subject of surprisingly few studies and reports, providing new research opportunities.
3.5. Production of Biomethane
Anaerobic digestion of algal residual biomass produces a mixture of gases, with CO2 making up between 30 and 50% of the mixture and CH4 making up between 50 and 70%. This process is well known for producing biomethane (CH4) [120]. Depending on the species of algae used and the experimental conditions, the methane production yield from algal biomass generally ranges from 0.2 to 0.4 m3 CH4/kg or 0.024 to 0.6 L CH4/g VS (volatile solid) [123]. For the production of biomethane from Dunaliella sp., the yield of CH4 even varies within the same species, with values between 0.063 L CH4/g VS and 0.323 L CH4/g VS [124]. Temperature, biomass loading volume and rate, time, bacterial strains, and algal cell wall composition are some of the factors that affect the biomethane production yield from algae [125]. The C:N ratio is another crucial element that significantly affects the yield of biomethane. Algal residual biomass is thought to have a low C:N ratio (6–9); however, mixing algal biomass with higher carbonaceous feedstocks has been suggested for improved biomethane yields [126]. This process must be combined with some other biofuel and biomaterial production processes using a biorefinery approach in order to make the process high yielding and cost-effective [127]. Algal biomass can be used to make a variety of biofuels, including biodiesel, bioethanol, biomethane, and biohydrogen, among others, of which former two are considered primary products and the latter two co-products in biorefinery production model [120].
4. Techno Economic Analysis (TEA) of Algae Based Biorefinery
Deemed to be an important aspect of biotechnology, biorefinery involves the significant production of bio-enriched products from biomass treatment hubs by using a combination of contemporary, feasible, and viable processes used in industries at a large scale and are of tremendous significance to society [128,129]. A TEA is required to launch a biotechnological industry so that logistics can work resourcefully and the biorefinery can be founded following the most economical setup methodologies at the disposal [130,131]. By generating concepts with longstanding sustainability outlines, bioeconomy seeks to substitute energy sources such as fossil fuels. Technology is essential for the industrial application of many biotechnological applications because it allows efficient use of inexpensive, easy-to-obtain chemicals with high added value. Bio-economy is defined as the process of creating bio-based products from renewable resources such as biological feedstock and biomass [132,133]. Presently, TEA is defined in terms of the economic and technological feasibility pertaining to commercial approaches [134,135]. At first look, a biorefinery presents two directives, i.e., a direct cascade in which the main objective is to produce high-value compounds while also manufacturing biofuels from the leftovers, and an inverse cascade in which the main goal is to produce biofuels followed by the reverse formation of compounds [136,137]. Nevertheless, new procedures have been put forth to improve the bio-economy by implementing technology suited to the valorization of macroalgae [138]. The most recent research trend in the previous decade has been the employment of newer pretreatment technologies of biomass [139]. Researchers are integrating algal feedstock into the preparation, estimate, economic, social, and conservational features of 3rd generation biofuels and bioproducts, as well as analyzing the feedstock’s life cycle and feasibility [137,140]. To achieve an effective macroalgae biorefinery development, techno economic viability for an industrial plan and scale must be evaluated. These may be systematically analyzed and enhanced by producing significant goods, such as biofuel, that fulfill market quality and price-norms [141,142].
5. A Feasible Biorefinery Template: Techno Economic Analysis
According to recent research, the logistics of biomass are thought to be the most important aspect for the development of the bioeconomy. More research is required before macroalgae biorefineries can be economically viable. It may be possible to improve the design economics and pricing of various bioproducts from marine biomass by designing biorefineries based on the technical method of deriving compounds from macroalgae [143,144].
The creation, commercialization, and operation of a biorefinery facility are all difficult tasks. The cost of the manufacturing process, harvesting, and biomass collection systems for raw materials that must be obtained at a specific time, which affects transportation costs, is the current challenge for feedstock. Thus, it has become crucial to improve the technical, economic, and environmental aspects of developing a biorefinery in a pilot plant. As a general guideline, it is essential to create and estimate scenarios when conducting research and designing a biorefinery plant [145]. Platforms such as the Aspen Plus, Simulator process, and SuperPro Designer software simplify the modeling simulations [146]. The software application computes variables and operating costs for the upstream and downstream processes of producing and distilling biofuels based on both raw material and energy balance calculations from process modelling using simulations [147]. A techno-economic analysis of the current operational conditions is displayed in Table 3.

Table 3.
Operational conditions and techno-economic analysis studies in algal biorefinery.
6. Challenges and Constraints
Algae biorefineries have attracted much attention nowadays because of their efficiency in treating wastewater and generating biofuels. They can also be used as a raw material for a variety of industries, including the cosmetics and pharmaceutical industries. Despite this, there are still many problems that need to be resolved, such as ways to increase the production of biomass and strategies to reduce the high energy needs for growing and harvesting algae. Some researchers have suggested using syngas as a carbon source and wastewater as a fertilizer source in algae culture to address these issues. Algal biorefinery can reduce cultivation costs in half, but its commercial applicability is subject to some limitations. Harvesting and biomass productivity (quantity and quality) problems should be addressed in this regard. The genetic elements and metabolic pathways in algal biomass may be changed or modified through genetic engineering to increase biomass production. Another option for a culture medium is fermented wastewater with a high nitrogen content. Algal biomass productivity will increase due to using this technique to produce biofuels and extract bioactive substances for use in commercial biofuel and bioactive chemical applications. On the other hand, the negative effects of the preferred component extraction will be mitigated by a particular algae species that has been discovered and chosen for growth. To reduce the high energy requirement for ABF production, it would be advantageous to link upstream and downstream processes to create a low-cost, value-added product manufacturing system.
7. Conclusions
Algae are ideally suited for multi-product biorefineries due to the wide range of biochemical compositions present in algae biomass. Several key points are being summarized herewith for an optimally functioning algal biorefinery.
- (a)
- Biorefineries need to be more cost- and sustainably effective.
- (b)
- Based on the current lab-scale LCA evaluation, algal biorefinery with minimal environmental impact can be put forth.
- (c)
- A critical evaluation of the function of catalysts and their synthesis via hydrothermal treatment of biomass needs to be conducted to consolidate data on modelling projects as well as techno-economic assessments.
The study’s important findings, knowledge gaps, and recommendations for the future are presented at the study’s conclusion to help enhance the commercial feasibility and environmental sustainability of hydrothermal treatment of wet wastes.
Author Contributions
Conceptualization: N.D. and S.B.; validation, N.D., P.K., J.T.E.L. and S.B.; formal analysis, N.D. and S.B.; investigation, N.D., P.K. and S.B; resources, N.D and S.B.; data curation, N.D., P.K., J.T.E.L. and S.B.; writing—original draft preparation, N.D.; writing—review and editing, N.D., P.K. and S.B.; visualization, N.D., P.K. and S.B.; supervision, N.D., and S.B.; project administration, N.D and J.T.E.L.; funding acquisition: N.D. and J.T.E.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Marie Curie Research fellowship grant awarded to Nalok Dutta from UKRI Engineering and Physical Sciences research council, EP/X032078/1 and the National Research Foundation, Prime Minister’s Office, Singapore under its Campus for Research Excellence and Technological Enterprise (CREATE) program.
Data Availability Statement
The data supporting the reported review was procured from the research articles enlisted in the references section.
Conflicts of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Uma, V.; Usmani, Z.; Sharma, M.; Diwan, D.; Sharma, M.; Guo, M.; Tuohy, M.G.; Makatsoris, C.; Zhao, X.; Thakur, V.K. Valorisation of algal biomass to value-added metabolites: Emerging trends and opportunities. Phytochem. Rev. 2022, 1–26. [Google Scholar] [CrossRef]
- Kumaresan, V.; Nizam, F.; Ravichandran, G.; Viswanathan, K.; Palanisamy, R.; Bhatt, P.; Arasu, M.V.; Al-Dhabi, N.A.; Mala, K. Transcriptome changes of blue-green algae, Arthrospira sp. in response to sulfate stress. Algal Res. 2017, 23, 96–103. [Google Scholar]
- Scarcelli, P.G.; Ruas, G.; Lopez-Serna, R.; Serejo, M.L.; Blanco, S.; Boncz, M.Á.; Muñoz, R. Integration of algae-based sewage treatment with anaerobic digestion of the bacterial-algal biomass and biogas upgrading. Bioresour. Technol. 2021, 340, 125552. [Google Scholar] [CrossRef]
- Ganesan, R.; Manigandan, S.; Samuel, M.S.; Shanmuganathan, R.; Brindhadevi, K.; Chi, N.T.; Duc, P.A.; Pugazhendhi, A. A review on prospective production of biofuel from microalgae. Biotechnol. Rep. 2020, 27, e00509. [Google Scholar] [CrossRef] [PubMed]
- Khammee, P.; Ramaraj, R.; Whangchai, N.; Bhuyar, P.; Unpaprom, Y. The immobilization of yeast for fermentation of macroalgae Rhizoclonium sp. for efficient conversion into bioethanol. Biomass Convers. Biorefinery 2021, 11, 827–835. [Google Scholar] [CrossRef]
- Gui, M.M.; Lee, K.; Bhatia, S.J. Feasibility of edible oil vs. non-edible oil vs. waste edible oil as biodiesel feedstock. Energy 2008, 33, 1646–1653. [Google Scholar] [CrossRef]
- Limayem, A.; Ricke, S.C. Lignocellulosic biomass for bioethanol production: Current perspectives, potential issues and future prospects. Prog. Energy Combust. Sci. 2012, 38, 449–467. [Google Scholar] [CrossRef]
- Xia, F.; Liu, H.; Lu, J.; Lv, Y.; Zhai, S.; An, Q.; Cheng, Y.; Wang, H. An integrated biorefinery process to produce butanol and pulp from corn straw. Ind. Crop. Prod. 2019, 140, 111648. [Google Scholar] [CrossRef]
- Bozell, J.J.; Astner, A.; Baker, D.; Biannic, B.; Cedeno, D.; Elder, T.; Hosseinaei, O.; Delbeck, L.; Kim, J.-W.; O’lenick, C.J.; et al. Integrating separation and conversion—Conversion of biorefinery process streams to biobased chemicals and fuels. BioEnergy Res. 2014, 7, 856–866. [Google Scholar] [CrossRef]
- Bozell, J.J.; Petersen, G.R. Technology development for the production of biobased products from biorefinery carbohydrates—The US Department of Energy’s “Top 10” revisited. Green Chem. 2010, 12, 539–554. [Google Scholar] [CrossRef]
- Low, S.S.; Bong, K.X.; Mubashir, M.; Cheng, C.K.; Lam, M.K.; Lim, J.W.; Ho, Y.C.; Lee, K.T.; Munawaroh, H.S.H.; Show, P.L. Microalgae cultivation in palm oil mill effluent (POME) treatment and biofuel production. Sustainability 2021, 13, 3247. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Jagtap, S.S.; Bedekar, A.A.; Bhatia, R.K.; Patel, A.K.; Pant, D.; Banu, J.R.; Rao, C.V.; Kim, Y.-G.; Yang, Y.-H. Recent developments in pretreatment technologies on lignocellulosic biomass: Effect of key parameters, technological improvements, and challenges. Bioresour. Technol. 2020, 300, 122724. [Google Scholar] [CrossRef] [PubMed]
- Scheuer, C.; Boot, E.; Carse, N.; Clardy, A.; Gallagher, J.; Heck, S.; Marron, S.; Martinez-Alvarez, L.; Masarykova, D.; McMillan, P.; et al. Disentangling Inclusion in Physical Education Lessons: Developing a Resource Toolkit for Teachers; Slovak Scientific Society for Physical Education and Sport and FIEP: Bratislava, Slovakia, 2021; pp. 343–354. [Google Scholar]
- Chozhavendhan, S.; Rajamehala, M.; Karthigadevi, G.; Kumar, R.P.; Bharathiraja, B.; Jayakumar, M. Application of nanotechnology for the sustainable development of algal biofuel industries. In Nanomaterials; Academic Press: Cambridge, MA, USA, 2021; pp. 401–410. [Google Scholar] [CrossRef]
- Yadav, G.; Sen, R. Sustainability of microalgal biorefinery: Scope, challenges, and opportunities. In Sustainable Energy Technology and Policies; Springer: Singapore, 2018; pp. 335–351. [Google Scholar] [CrossRef]
- Jez, S.; Spinelli, D.; Fierro, A.; Dibenedetto, A.; Aresta, M.; Busi, E.; Basosi, R.J. Comparative life cycle assessment study on environmental impact of oil production from micro-algae and terrestrial oilseed crops. Bioresour. Technol. 2017, 239, 266–275. [Google Scholar] [CrossRef]
- Li, Y.; Horsman, M.; Wu, N.; Lan, C.Q.; Dubois-Calero, N.J. Biofuels from microalgae. Biotechnol. Prog. 2008, 24, 815–820. [Google Scholar] [CrossRef] [PubMed]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Budzianowski, W.M. High-value low-volume bioproducts coupled to bioenergies with potential to enhance business development of sustainable biorefineries. Renew. Sustain. Energy Rev. 2017, 70, 793–804. [Google Scholar] [CrossRef]
- Özdenkçi, K.; Sarwar, G. The Role of Biomass Conversion Processes on Circular Economy. In Constructing A Green Circular Society; Unigrafia Oy: Helsinki, Finland, 2017; pp. 101–120. [Google Scholar]
- Trivedi, J.; Aila, M.; Bangwal, D.; Kaul, S.; Garg, M. Algae based biorefinery—How to make sense? Renew. Sustain. Energy Rev. 2015, 47, 295–307. [Google Scholar] [CrossRef]
- Dutta, N.; Mahmood, F.; Sarwar, A.; Sultan, M.; Ahmad, M.; Khan, M.U.; Omar, M.M. Algae Biomass Conversion Technologies. In Handbook of Research on Algae as a Sustainable Solution for Food, Energy, and the Environment; IGI Global: Hershey, PA, USA, 2022; pp. 524–546. [Google Scholar]
- Thoré, E.S.; Muylaert, K.; Bertram, M.G.; Brodin, T. Microalgae. Curr. Biol. 2023, 33, R91–R95. [Google Scholar] [CrossRef]
- Russo, G.L.; Langellotti, A.L.; Oliviero, M.; Sacchi, R.; Masi, P. Sustainable production of food grade omega-3 oil using aquatic protists: Reliability and future horizons. New Biotechnol. 2021, 62, 32–39. [Google Scholar] [CrossRef]
- Bhuyar, P.; Rahim, M.H.A.; Yusoff, M.M.; Maniam, G.P.; Govindan, N. A selective microalgae strain for biodiesel production in relation to higher lipid profile. Maejo Int. J. Energy Environ. Commun. 2019, 1, 8–14. [Google Scholar] [CrossRef]
- Russo, G.L.; Langellotti, A.L.; Verardo, V.; Martín-García, B.; Oliviero, M.; Baselice, M.; Di Pierro, P.; Sorrentino, A.; Viscardi, S.; Marileo, L.; et al. Bioconversion of Cheese Whey and Food By-Products by Phaeodactylum tricornutum into Fucoxanthin and n-3 Lc-PUFA through a Biorefinery Approach. Mar. Drugs 2023, 21, 190. [Google Scholar] [CrossRef]
- Russo, G.L.; Langellotti, A.L.; Sacchi, R.; Masi, P. Techno-economic assessment of DHA-rich Aurantiochytrium sp. production using food industry by-products and waste streams as alternative growth media. Bioresour. Technol. Rep. 2022, 18, 100997. [Google Scholar] [CrossRef]
- Rodolfi, L.; Chini Zittelli, G.; Bassi, N.; Padovani, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar] [CrossRef]
- Subhash, G.V.; Rajvanshi, M.; Kumar, G.R.K.; Sagaram, U.S.; Prasad, V.; Govindachary, S.; Dasgupta, S. Challenges in microalgal biofuel production: A perspective on techno economic feasibility under biorefinery stratagem. Bioresour. Technol. 2021, 343, 126155. [Google Scholar] [CrossRef]
- Günerken, E.; D’Hondt, E.; Eppink, M.; Garcia-Gonzalez, L.; Elst, K.; Wijffels, R.H. Cell disruption for microalgae biorefineries. Biotechnol. Adv. 2015, 33, 243–260. [Google Scholar] [CrossRef]
- Biller, P.; Ross, A. Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content. Bioresour. Technol. 2011, 102, 215–225. [Google Scholar] [CrossRef]
- Ma, J.; Zhao, Q.-B.; Laurens, L.L.; Jarvis, E.E.; Nagle, N.J.; Chen, S.; Frear, C.S. Mechanism, kinetics and microbiology of inhibition caused by long-chain fatty acids in anaerobic digestion of algal biomass. Biotechnol. Biofuels 2015, 8, 141. [Google Scholar] [CrossRef] [PubMed]
- John, R.P.; Anisha, G.; Nampoothiri, K.M.; Pandey, A.J. Micro and macroalgal biomass: A renewable source for bioethanol. Bioresour. Technol. 2011, 102, 186–193. [Google Scholar] [CrossRef]
- Sun, H.; Zhao, W.; Mao, X.; Li, Y.; Wu, T.; Chen, F. High-value biomass from microalgae production platforms: Strategies and progress based on carbon metabolism and energy conversion. Biotechnol. Biofuels 2018, 11, 227. [Google Scholar] [CrossRef] [PubMed]
- Cairns, J., Jr.; Lanza, G.R.; Parker, B.C. Pollution related structural and functional changes in aquatic communities with emphasis on freshwater algae and protozoa. Proc. Acad. Nat. Sci. Phila. 1972, 124, 79–127. [Google Scholar]
- Zeriouh, O.; Reinoso-Moreno, J.V.; López-Rosales, L.; Cerón-García, M.C.; Sánchez-Mirón, A.; García-Camacho, F.; Molina-Grima, E. Biofouling in photobioreactors for marine microalgae. Crit. Rev. Biotechnol. 2017, 37, 1006–1023. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.E.F.; Bertucco, A. Bioethanol from microalgal biomass: A promising approach in biorefinery. Braz. Arch. Biol. Technol. 2019, 62, e19160816. [Google Scholar] [CrossRef]
- El-Dalatony, M.M.; Salama, E.-S.; Kurade, M.B.; Kim, K.-Y.; Govindwar, S.P.; Kim, J.R.; Kwon, E.E.; Min, B.; Jang, M.; Oh, S.-E. Whole conversion of microalgal biomass into biofuels through successive high-throughput fermentation. Chem. Eng. J. 2019, 360, 797–805. [Google Scholar] [CrossRef]
- Xia, A.; Cheng, J.; Song, W.; Su, H.; Ding, L.; Lin, R.; Lu, H.; Liu, J.; Zhou, J.; Cen, K. Fermentative hydrogen production using algal biomass as feedstock. Renew. Sustain. Energy Rev. 2015, 51, 209–230. [Google Scholar] [CrossRef]
- Chen, Z.; Rao, Y.; Usman, M.; Chen, H.; Białowiec, A.; Zhang, S.; Luo, G. Anaerobic fermentation of hydrothermal liquefaction wastewater of dewatered sewage sludge for volatile fatty acids production with focuses on the degradation of organic components and microbial community compositions. Sci. Total Environ. 2021, 777, 146077. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.; Nguyen, D.D.; Sivagurunathan, P.; Kobayashi, T.; Xu, K.; Chang, S.W. Cultivation of microalgal biomass using swine manure for biohydrogen production: Impact of dilution ratio and pretreatment. Bioresour. Technol. 2018, 260, 16–22. [Google Scholar] [CrossRef]
- Passos, F.; Ferrer, I. Influence of hydrothermal pretreatment on microalgal biomass anaerobic digestion and bioenergy production. Water Res. 2015, 68, 364–373. [Google Scholar] [CrossRef]
- Ho, S.-H.; Huang, S.-W.; Chen, C.-Y.; Hasunuma, T.; Kondo, A.; Chang, J.S. Bioethanol production using carbohydrate-rich microalgae biomass as feedstock. Bioresour. Technol. 2013, 135, 191–198. [Google Scholar] [CrossRef]
- Cheng, C.-L.; Che, P.-Y.; Chen, B.-Y.; Lee, W.-J.; Lin, C.-Y.; Chang, J.-S. Biobutanol production from agricultural waste by an acclimated mixed bacterial microflora. Appl. Energy 2012, 100, 3–9. [Google Scholar] [CrossRef]
- Stiles, W.A.; Styles, D.; Chapman, S.P.; Esteves, S.; Bywater, A.; Melville, L.; Silkina, A.; Lupatsch, I.; Grünewald, C.F.; Lovitt, R. Using microalgae in the circular economy to valorise anaerobic digestate: Challenges and opportunities. Bioresour. Technol. 2018, 267, 732–742. [Google Scholar] [CrossRef]
- Ras, M.; Lardon, L.; Bruno, S.; Bernet, N.; Steyer, J.-P. Experimental study on a coupled process of production and anaerobic digestion of Chlorella vulgaris. Bioresour. Technol. 2011, 102, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Bohutskyi, P.; Bouwer, E. Advanced biofuels and bioproducts. In Chapter 36. Biogas Production from Algae and Cyanobacteria Through Anaerobic Digestion. A Review, Analysis, and Research Needs; Springer: New York, NY, USA, 2013. [Google Scholar]
- Krishnan, S.; Din, M.; Taib, S.M.; Ling, Y.; Aminuddin, E.; Chelliapan, S.; Mishra, P.; Rana, S.J. Utilization of micro-algal biomass residues (MABRS) for bio-hythane production—A perspective. J. Appl. Biotechnol. Bioeng. 2018, 5, 162–165. [Google Scholar] [CrossRef]
- Solé-Bundó, M.; Eskicioglu, C.; Garfí, M.; Carrère, H.; Ferrer, I. Anaerobic co-digestion of microalgal biomass and wheat straw with and without thermo-alkaline pretreatment. Bioresour. Technol. 2017, 237, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Ward, A.; Lewis, D.; Green, F. Anaerobic digestion of algae biomass: A review. Algal Res. 2014, 5, 204–214. [Google Scholar] [CrossRef]
- Ji, M.; Wang, X.; Usman, M.; Liu, F.; Dan, Y.; Zhou, L.; Campanaro, S.; Luo, G.; Sang, W. Effects of different feedstocks-based biochar on soil remediation: A review. Environ. Pollut. 2022, 294, 118655. [Google Scholar] [CrossRef]
- Gonzalez-Fernandez, C.; Sialve, B.; Molinuevo-Salces, B. Anaerobic digestion of microalgal biomass: Challenges, opportunities and research needs. Bioresour. Technol. 2015, 198, 896–906. [Google Scholar] [CrossRef]
- Ji, M.; Sang, W.; Tsang, D.C.; Usman, M.; Zhang, S.; Luo, G. Molecular and microbial insights towards understanding the effects of hydrochar on methane emission from paddy soil. Sci. Total Environ. 2020, 714, 136769. [Google Scholar] [CrossRef]
- Yang, Z.; Guo, R.; Xu, X.; Fan, X.; Luo, S. Hydrogen and methane production from lipid-extracted microalgal biomass residues. Int. J. Hydrogen Energy 2011, 36, 3465–3470. [Google Scholar] [CrossRef]
- Deng, X.-Y.; Gao, K.; Zhang, R.-C.; Addy, M.; Lu, Q.; Ren, H.-Y.; Chen, P.; Liu, Y.-H.; Ruan, R. Growing Chlorella vulgaris on thermophilic anaerobic digestion swine manure for nutrient removal and biomass production. Bioresour. Technol. 2017, 243, 417–425. [Google Scholar] [CrossRef]
- Lakaniemi, A.-M.; Hulatt, C.J.; Thomas, D.N.; Tuovinen, O.H.; Puhakka, J.A. Biogenic hydrogen and methane production from Chlorella vulgaris and Dunaliella tertiolectabiomass. Biotechnol. Biofuels 2011, 4, 34. [Google Scholar] [CrossRef]
- Park, J.-Y.; Park, M.S.; Lee, Y.-C.; Yang, J.-W. Advances in direct transesterification of algal oils from wet biomass. Bioresour. Technol. 2015, 184, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-H.; Lin, B.-J.; Huang, M.-Y.; Chang, J.-S. Thermochemical conversion of microalgal biomass into biofuels: A review. Bioresour. Technol. 2015, 184, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Tekin, K.; Karagöz, S.; Bektaş, S. A review of hydrothermal biomass processing. Renew. Sustain. Energy Rev. 2014, 40, 673–687. [Google Scholar] [CrossRef]
- Magdeldin, M.; Kohl, T.; Järvinen, M.J. Techno-economic assessment of the by-products contribution from non-catalytic hydrothermal liquefaction of lignocellulose residues. Energy 2017, 137, 679–695. [Google Scholar] [CrossRef]
- Libra, J.A.; Ro, K.S.; Kammann, C.; Funke, A.; Berge, N.D.; Neubauer, Y.; Titirici, M.-M.; Fühner, C.; Bens, O.; Kern, J.; et al. Hydrothermal carbonization of biomass residuals: A comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels 2011, 2, 71–106. [Google Scholar] [CrossRef]
- Liu, Z.; Balasubramanian, R. Upgrading of waste biomass by hydrothermal carbonization (HTC) and low temperature pyrolysis (LTP): A comparative evaluation. Appl. Energy 2014, 114, 857–864. [Google Scholar] [CrossRef]
- Kruse, A.; Dinjus, E. Hot compressed water as reaction medium and reactant: Properties and synthesis reactions. J. Supercrit. Fluids 2007, 39, 362–380. [Google Scholar] [CrossRef]
- Anbu, S.; Padma, J.; Punithavalli, K.; Saranraj, P. Fruits peel waste as a novel media for the growth of economically important fungi. Pharmacogn. Phytochem. 2017, 6, 426–428. [Google Scholar]
- Vardon, D.R.; Sharma, B.K.; Blazina, G.V.; Rajagopalan, K.; Strathmann, T.J. Thermochemical conversion of raw and defatted algal biomass via hydrothermal liquefaction and slow pyrolysis. Bioresour. Technol. 2012, 109, 178–187. [Google Scholar] [CrossRef]
- Hossain, M.S.; Balakrishnan, V.; Rahman, N.N.N.A.; Sarker, M.Z.I.; Kadir, M.O.A. Treatment of clinical solid waste using a steam autoclave as a possible alternative technology to incineration. Int. J. Environ. Res. Public Health 2012, 9, 855–867. [Google Scholar] [CrossRef]
- Pavlovic, I.; Knez, Z.; Skerget, M. Hydrothermal reactions of agricultural and food processing wastes in sub-and supercritical water: A review of fundamentals, mechanisms, and state of research. J. Agric. Food Chem. 2013, 61, 8003–8025. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Oyedun, A.O.; Kumar, A.J.R.; Reviews, S.E. A review on the current status of various hydrothermal technologies on biomass feedstock. Renew. Sustain. Energy Rev. 2018, 81, 1742–1770. [Google Scholar] [CrossRef]
- Djandja, O.S.; Wang, Z.; Chen, L.; Qin, L.; Wang, F.; Xu, Y.; Duan, P. Progress in hydrothermal liquefaction of algal biomass and hydrothermal upgrading of the subsequent crude bio-oil: A mini review. Energy Fuels 2020, 34, 11723–11751. [Google Scholar] [CrossRef]
- Raheem, A.; Prinsen, P.; Vuppaladadiyam, A.K.; Zhao, M.; Luque, R. A review on sustainable microalgae based biofuel and bioenergy production: Recent developments. J. Clean. Prod. 2018, 181, 42–59. [Google Scholar] [CrossRef]
- Raheem, A.; Dupont, V.; Channa, A.Q.; Zhao, X.; Vuppaladadiyam, A.K.; Taufiq-Yap, Y.-H.; Zhao, M.; Harun, R. Parametric characterization of air gasification of Chlorella vulgaris biomass. Energy Fuels 2017, 31, 2959–2969. [Google Scholar] [CrossRef]
- Sanchez-Silva, L.; López-González, D.; Garcia-Minguillan, A.; Valverde, J. Pyrolysis, combustion and gasification characteristics of Nannochloropsis gaditana microalgae. Bioresour. Technol. 2013, 130, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Chakinala, A.G.; Brilman, D.W.; van Swaaij, W.P.; Kersten, S.R. Catalytic and non-catalytic supercritical water gasification of microalgae and glycerol. Ind. Eng. Chem. Res. 2010, 49, 1113–1122. [Google Scholar] [CrossRef]
- Duman, G.; Uddin, M.A.; Yanik, J. Hydrogen production from algal biomass via steam gasification. Bioresour. Technol. 2014, 166, 24–30. [Google Scholar] [CrossRef]
- Rizwan, M.; Lee, J.H.; Gani, R. Optimal design of microalgae-based biorefinery: Economics, opportunities and challenges. Appl. Energy 2015, 150, 69–79. [Google Scholar] [CrossRef]
- Stucki, S.; Vogel, F.; Ludwig, C.; Haiduc, A.G.; Brandenberger, M. Catalytic gasification of algae in supercritical water for biofuel production and carbon capture. Energy Environ. Sci. 2009, 2, 535–541. [Google Scholar] [CrossRef]
- Mathimani, T.; Baldinelli, A.; Rajendran, K.; Prabakar, D.; Matheswaran, M.; van Leeuwen, R.P.; Pugazhendhi, A. Review on cultivation and thermochemical conversion of microalgae to fuels and chemicals: Process evaluation and knowledge gaps. J. Clean. Prod. 2019, 208, 1053–1064. [Google Scholar] [CrossRef]
- Bae, Y.J.; Ryu, C.; Jeon, J.-K.; Park, J.; Suh, D.J.; Suh, Y.-W.; Chang, D.; Park, Y.-K. The characteristics of bio-oil produced from the pyrolysis of three marine macroalgae. Bioresour. Technol. 2011, 102, 3512–3520. [Google Scholar] [CrossRef] [PubMed]
- Chaiwong, K.; Kiatsiriroat, T.; Vorayos, N.; Thararax, C. Study of bio-oil and bio-char production from algae by slow pyrolysis. Bioenergy 2013, 56, 600–606. [Google Scholar] [CrossRef]
- Pragya, N.; Pandey, K.K. Life cycle assessment of green diesel production from microalgae. Renew. Energy 2016, 86, 623–632. [Google Scholar] [CrossRef]
- Xu, L.; Brilman, D.W.W.; Withag, J.A.; Brem, G.; Kersten, S. Assessment of a dry and a wet route for the production of biofuels from microalgae: Energy balance analysis. Bioresour. Technol. 2011, 102, 5113–5122. [Google Scholar] [CrossRef] [PubMed]
- Bennion, E.P.; Ginosar, D.M.; Moses, J.; Agblevor, F.; Quinn, J.C. Lifecycle assessment of microalgae to biofuel: Comparison of thermochemical processing pathways. Appl. Energy 2015, 154, 1062–1071. [Google Scholar] [CrossRef]
- de la Hidalga, A.N.; Owen, D.; Spasic, I.; Rosin, P.; Sun, X. Use of semantic segmentation for increasing the throughput of digitisation workflows for natural history collections. Biodivers. Inf. Sci. Stand. 2019, 3, e37161. [Google Scholar] [CrossRef]
- Milledge, J.J.; Smith, B.; Dyer, P.W.; Harvey, P.J.E. Macroalgae-derived biofuel: A review of methods of energy extraction from seaweed biomass. Energies 2014, 7, 7194–7222. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef]
- Demirbas, A. Biorefineries: Current activities and future developments. Energy Convers. Manag. 2009, 50, 2782–2801. [Google Scholar] [CrossRef]
- Patil, P.D.; Gude, V.G.; Mannarswamy, A.; Cooke, P.; Nirmalakhandan, N.; Lammers, P.; Deng, S. Comparison of direct transesterification of algal biomass under supercritical methanol and microwave irradiation conditions. Fuel 2012, 97, 822–831. [Google Scholar] [CrossRef]
- Mathimani, T.; Uma, L.; Prabaharan, D.J. Homogeneous acid catalysed transesterification of marine microalga Chlorella sp. BDUG 91771 lipid–an efficient biodiesel yield and its characterization. Renew. Energy 2015, 81, 523–533. [Google Scholar] [CrossRef]
- Rahman, M.; Aziz, M.; Al-Khulaidi, R.A.; Sakib, N.; Islam, M. Biodiesel production from microalgae Spirulina maxima by two step process: Optimization of process variable. J. Radiat. Res. Appl. Sci. 2017, 10, 140–147. [Google Scholar] [CrossRef]
- Salam, K.A.; Velasquez-Orta, S.B.; Harvey, A.P. A sustainable integrated in situ transesterification of microalgae for biodiesel production and associated co-product—A review. Renew. Sustain. Energy Rev. 2016, 65, 1179–1198. [Google Scholar] [CrossRef]
- Deshpande, S.R.; Sunol, A.K.; Philippidis, G. Status and prospects of supercritical alcohol transesterification for biodiesel production. WIREs Energy Environ. 2017, 6, e252. [Google Scholar] [CrossRef]
- Nan, Y.; Liu, J.; Lin, R.; Tavlarides, L.L. Production of biodiesel from microalgae oil (Chlorella protothecoides) by non-catalytic transesterification in supercritical methanol and ethanol: Process optimization. J. Supercrit. Fluids 2015, 97, 174–182. [Google Scholar] [CrossRef]
- Zhou, D.; Qiao, B.; Li, G.; Xue, S.; Yin, J. Continuous production of biodiesel from microalgae by extraction coupling with transesterification under supercritical conditions. Bioresour. Technol. 2017, 238, 609–615. [Google Scholar] [CrossRef]
- Ehimen, E.; Sun, Z.; Carrington, C.J. Variables affecting the in situ transesterification of microalgae lipids. Fuel 2010, 89, 677–684. [Google Scholar] [CrossRef]
- Levine, R.B.; Pinnarat, T.; Savage, P.E. Biodiesel production from wet algal biomass through in situ lipid hydrolysis and supercritical transesterification. Energy Fuels 2010, 24, 5235–5243. [Google Scholar] [CrossRef]
- Im, H.; Lee, H.; Park, M.S.; Yang, J.-W.; Lee, J.W. Concurrent extraction and reaction for the production of biodiesel from wet microalgae. Bioresour. Technol. 2014, 152, 534–537. [Google Scholar] [CrossRef]
- Viêgas, C.V.; Hachemi, I.; Freitas, S.P.; Mäki-Arvela, P.; Aho, A.; Hemming, J.; Smeds, A.; Heinmaa, I.; Fontes, F.B.; da Silva Pereira, D.C.; et al. A route to produce renewable diesel from algae: Synthesis and characterization of biodiesel via in situ transesterification of Chlorella alga and its catalytic deoxygenation to renewable diesel. Fuel 2015, 155, 144–154. [Google Scholar] [CrossRef]
- Dutta, N.; Usman, M.; Luo, G.; Zhang, S. An insight into valorization of lignocellulosic biomass by optimization with the combination of hydrothermal (HT) and biological techniques: A review. Sustain. Chem. 2022, 3, 35–55. [Google Scholar] [CrossRef]
- Hrnčič, M.K.; Kravanja, G.; Knez, Ž.J.E. Hydrothermal treatment of biomass for energy and chemicals. Energy 2016, 116, 1312–1322. [Google Scholar] [CrossRef]
- Heilmann, S.M.; Davis, H.T.; Jader, L.R.; Lefebvre, P.A.; Sadowsky, M.J.; Schendel, F.J.; Von Keitz, M.G.; Valentas, K.J. Hydrothermal carbonization of microalgae. Biomass Bioenergy 2010, 34, 875–882. [Google Scholar] [CrossRef]
- Barreiro, D.L.; Prins, W.; Ronsse, F.; Brilman, W.J.B. Hydrothermal liquefaction (HTL) of microalgae for biofuel production: State of the art review and future prospects. Bioenergy 2013, 53, 113–127. [Google Scholar]
- Ruiz-Marin, A.; Canedo-López, Y.; Chávez-Fuentes, P. Biohydrogen production by Chlorella vulgaris and Scenedesmus obliquus immobilized cultivated in artificial wastewater under different light quality. AMB Express 2020, 10, 191. [Google Scholar] [CrossRef] [PubMed]
- Batista, A.P.; Ambrosano, L.; Graça, S.; Sousa, C.; Marques, P.A.; Ribeiro, B.; Botrel, E.P.; Neto, P.C.; Gouveia, L. Combining urban wastewater treatment with biohydrogen production–an integrated microalgae-based approach. Bioresour. Technol. 2015, 184, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.M.; Duan, P.; Savage, P.E. Hydrothermal liquefaction and gasification of Nannochloropsis sp. Fuels 2010, 24, 3639–3646. [Google Scholar] [CrossRef]
- Ekpo, U.; Ross, A.; Camargo-Valero, M.A.; Williams, P. A comparison of product yields and inorganic content in process streams following thermal hydrolysis and hydrothermal processing of microalgae, manure and digestate. Bioresour. Technol. 2016, 200, 951–960. [Google Scholar] [CrossRef]
- Faeth, J.L.; Savage, P.E. Effects of processing conditions on biocrude yields from fast hydrothermal liquefaction of microalgae. Bioresour. Technol. 2016, 206, 290–293. [Google Scholar] [CrossRef]
- Levine, R.B.; Sierra, C.O.S.; Hockstad, R.; Obeid, W.; Hatcher, P.G.; Savage, P.E. The use of hydrothermal carbonization to recycle nutrients in algal biofuel production. Environ. Prog. Sustain. Energy 2013, 32, 962–975. [Google Scholar] [CrossRef]
- Li, H.; Liu, Z.; Zhang, Y.; Li, B.; Lu, H.; Duan, N.; Liu, M.; Zhu, Z.; Si, B. Conversion efficiency and oil quality of low-lipid high-protein and high-lipid low-protein microalgae via hydrothermal liquefaction. Bioresour. Technol. 2014, 154, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Onwudili, J.A.; Lea-Langton, A.R.; Ross, A.B.; Williams, P.T. Catalytic hydrothermal gasification of algae for hydrogen production: Composition of reaction products and potential for nutrient recycling. Bioresour. Technol. 2013, 127, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Park, K.Y.; Lee, K.; Kim, D. Characterized hydrochar of algal biomass for producing solid fuel through hydrothermal carbonization. Bioresour. Technol. 2018, 258, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Shuping, Z.; Yulong, W.; Mingde, Y.; Kaleem, I.; Chun, L.; Tong, J. Production and characterization of bio-oil from hydrothermal liquefaction of microalgae Dunaliella tertiolecta cake. Energy 2010, 35, 5406–5411. [Google Scholar] [CrossRef]
- Miranda, J.; Passarinho, P.C.; Gouveia, L. Pre-treatment optimization of Scenedesmus obliquus microalga for bioethanol production. Bioresour. Technol. 2012, 104, 342–348. [Google Scholar] [CrossRef]
- Chen, C.-Y.; Zhao, X.-Q.; Yen, H.-W.; Ho, S.-H.; Cheng, C.-L.; Lee, D.-J.; Bai, F.-W.; Chang, J.-S. Microalgae-based carbohydrates for biofuel production. Biochem. Eng. J. 2013, 78, 1–10. [Google Scholar] [CrossRef]
- Zhao, B.; Su, Y. Process effect of microalgal-carbon dioxide fixation and biomass production: A review. Renew. Sustain. Energy Rev. 2014, 31, 121–132. [Google Scholar] [CrossRef]
- Rempel, A.; de Souza Sossella, F.; Margarites, A.C.; Astolfi, A.L.; Steinmetz, R.L.R.; Kunz, A.; Treichel, H.; Colla, L.M. Bioethanol from Spirulina platensis biomass and the use of residuals to produce biomethane: An energy efficient approach. Bioresour. Technol. 2019, 288, 121588. [Google Scholar] [CrossRef]
- El-Mekkawi, S.A.; Abdo, S.M.; Samhan, F.A.; Ali, G.H. Optimization of some fermentation conditions for bioethanol production from microalgae using response surface method. Bull. Natl. Res. Cent. 2019, 43, 164. [Google Scholar] [CrossRef]
- Onay, M. Bioethanol production via different saccharification strategies from H. tetrachotoma ME03 grown at various concentrations of municipal wastewater in a flat-photobioreactor. Fuel 2018, 239, 1315–1323. [Google Scholar] [CrossRef]
- Phwan, C.K.; Chew, K.W.; Sebayang, A.H.; Ong, H.C.; Ling, T.C.; Malek, M.A.; Ho, Y.-C.; Show, P.L. Effects of acids pre-treatment on the microbial fermentation process for bioethanol production from microalgae. Biotechnol. Biofuels 2019, 12, 191. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Zhang, F.; Wu, Y.-R. Emerging technologies for conversion of sustainable algal biomass into value-added products: A state-of-the-art review. Sci. Total Environ. 2021, 784, 147024. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Sun, Y.; Rathour, R.; Pandey, A.; Thakur, I.S.; Tsang, D.C. Algae as potential feedstock for the production of biofuels and value-added products: Opportunities and challenges. Sci. Total Environ. 2020, 716, 137116. [Google Scholar] [CrossRef] [PubMed]
- Michalak, I.; Baśladyńska, S.; Mokrzycki, J.; Rutkowski, P. Biochar from a freshwater macroalga as a potential biosorbent for wastewater treatment. Water 2019, 11, 1390. [Google Scholar] [CrossRef]
- De Bhowmick, G.; Sarmah, A.K.; Sen, R. Zero-waste algal biorefinery for bioenergy and biochar: A green leap towards achieving energy and environmental sustainability. Sci. Total Environ. 2019, 650, 2467–2482. [Google Scholar] [CrossRef]
- Rabii, A.; Aldin, S.; Dahman, Y.; Elbeshbishy, E. A review on anaerobic co-digestion with a focus on the microbial populations and the effect of multi-stage digester configuration. Energies 2019, 12, 1106. [Google Scholar] [CrossRef]
- Milledge, J.J.; Nielsen, B.V.; Maneein, S.; Harvey, P.J. A brief review of anaerobic digestion of algae for bioenergy. Energies 2019, 12, 1166. [Google Scholar] [CrossRef]
- Barbot, Y.N.; Al-Ghaili, H.; Benz, R. A review on the valorization of macroalgal wastes for biomethane production. Mar. Drugs 2016, 14, 120. [Google Scholar] [CrossRef]
- McKennedy, J.; Sherlock, O. Anaerobic digestion of marine macroalgae: A review. Renew. Sustain. Energy Rev. 2015, 52, 1781–1790. [Google Scholar] [CrossRef]
- Mussgnug, J.H.; Klassen, V.; Schlüter, A.; Kruse, O. Microalgae as substrates for fermentative biogas production in a combined biorefinery concept. J. Biotechnol. 2010, 150, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Zollmann, M.; Robin, A.; Prabhu, M.; Polikovsky, M.; Gillis, A.; Greiserman, S.; Golberg, A. Green technology in green macroalgal biorefineries. Phycologia 2019, 58, 516–534. [Google Scholar] [CrossRef]
- Ubando, A.T.; Felix, C.B.; Chen, W.-H. Biorefineries in circular bioeconomy: A comprehensive review. Bioresour. Technol. 2020, 299, 122585. [Google Scholar] [CrossRef]
- Ramachandra, T.; Hebbale, D. Bioethanol from macroalgae: Prospects and challenges. Renew. Sustain. Energy Rev. 2019, 117, 109479. [Google Scholar] [CrossRef]
- Manhongo, T.; Chimphango, A.; Thornley, P.; Röder, M. An economic viability and environmental impact assessment of mango processing waste-based biorefineries for co-producing bioenergy and bioactive compounds. Renew. Sustain. Energy Rev. 2021, 148, 111216. [Google Scholar] [CrossRef]
- Gajaria, T.K.; Suthar, P.; Baghel, R.S.; Balar, N.B.; Sharnagat, P.; Mantri, V.A.; Reddy, C. Integration of protein extraction with a stream of byproducts from marine macroalgae: A model forms the basis for marine bioeconomy. Bioresour. Technol. 2017, 243, 867–873. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.D.; Kavitha, S.; Tyagi, V.K.; Rajkumar, M.; Bhatia, S.K.; Kumar, G.; Banu, J.R. Macroalgae-derived biohydrogen production: Biorefinery and circular bioeconomy. Biomass Convers. Biorefinery 2022, 12, 769–791. [Google Scholar] [CrossRef]
- DeRose, K.; DeMill, C.; Davis, R.W.; Quinn, J.C. Integrated techno economic and life cycle assessment of the conversion of high productivity, low lipid algae to renewable fuels. Algal Res. 2019, 38, 101412. [Google Scholar] [CrossRef]
- Gu, X.; Yu, L.; Pang, N.; Martinez-Fernandez, J.S.; Fu, X.; Chen, S.J. Comparative techno-economic analysis of algal biofuel production via hydrothermal liquefaction: One stage versus two stages. Appl. Energy 2020, 259, 114115. [Google Scholar] [CrossRef]
- Lara, A.; Rodríguez-Jasso, R.M.; Loredo-Treviño, A.; Aguilar, C.N.; Meyer, A.S.; Ruiz, H.A. Enzymes in the third generation biorefinery for macroalgae biomass. In Biomass, Biofuels, Biochemicals; Elsevier: Amsterdam, The Netherlands, 2020; pp. 363–396. [Google Scholar] [CrossRef]
- Pinales-Márquez, C.D.; Rodríguez-Jasso, R.M.; Araújo, R.G.; Loredo-Treviño, A.; Nabarlatz, D.; Gullón, B.; Ruiz, H.A. Circular bioeconomy and integrated biorefinery in the production of xylooligosaccharides from lignocellulosic biomass: A review. Ind. Crop. Prod. 2021, 162, 113274. [Google Scholar] [CrossRef]
- Lynd, L.R.; Liang, X.; Biddy, M.J.; Allee, A.; Cai, H.; Foust, T.; Himmel, M.E.; Laser, M.S.; Wang, M.; Wyman, C.E. Cellulosic ethanol: Status and innovation. Curr. Opin. Biotechnol. 2017, 45, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Ingle, K.N.; Traugott, H.; Golberg, A. Challenges for marine macroalgal biomass production in Indian coastal waters. Bot. Mar. 2020, 63, 327–340. [Google Scholar] [CrossRef]
- Olofsson, J.; Barta, Z.; Börjesson, P.; Wallberg, O. Integrating enzyme fermentation in lignocellulosic ethanol production: Life-cycle assessment and techno-economic analysis. Biotechnol. Biofuels 2017, 10, 51. [Google Scholar] [CrossRef] [PubMed]
- Konda, N.; Singh, S.; Simmons, B.A.; Klein-Marcuschamer, D. An investigation on the economic feasibility of macroalgae as a potential feedstock for biorefineries. BioEnergy Res. 2015, 8, 1046–1056. [Google Scholar] [CrossRef]
- Ansari, F.A.; Nasr, M.; Guldhe, A.; Gupta, S.K.; Rawat, I.; Bux, F. Techno-economic feasibility of algal aquaculture via fish and biodiesel production pathways: A commercial-scale application. Sci. Total Environ. 2020, 704, 135259. [Google Scholar] [CrossRef]
- Chemodanov, A.; Robin, A.; Golberg, A. Design of marine macroalgae photobioreactor integrated into building to support seagriculture for biorefinery and bioeconomy. Bioresour. Technol. 2017, 241, 1084–1093. [Google Scholar] [CrossRef]
- Chong, T.Y.; Cheah, S.A.; Ong, C.T.; Wong, L.Y.; Goh, C.R.; Tan, I.S.; Foo, H.C.Y.; Lam, M.K.; Lim, S.J.E. Techno-economic evaluation of third-generation bioethanol production utilizing the macroalgae waste: A case study in Malaysia. Energy 2020, 210, 118491. [Google Scholar] [CrossRef]
- Rogers, J.N.; Stokes, B.; Dunn, J.; Cai, H.; Wu, M.; Haq, Z.; Baumes, H. An assessment of the potential products and economic and environmental impacts resulting from a billion ton bioeconomy. Biofuels Bioprod. Biorefin. 2017, 11, 110–128. [Google Scholar] [CrossRef]
- Aziz, M.; Zaini, I.N. Production of hydrogen from algae: Integrated gasification and chemical looping. Energy Procedia 2017, 142, 210–215. [Google Scholar] [CrossRef]
- Hasanly, A.; Khajeh Talkhoncheh, M.; Karimi Alavijeh, M.J. Techno-economic assessment of bioethanol production from wheat straw: A case study of Iran. Clean Technol. Environ. Policy 2018, 20, 357–377. [Google Scholar] [CrossRef]
- Soleymani, M.; Rosentrater, K.A. Techno-economic analysis of biofuel production from macroalgae (seaweed). Bioengineering 2017, 4, 92. [Google Scholar] [CrossRef] [PubMed]
- Brigljević, B.; Fasahati, P.; Liu, J.J. Integrated Bio-refinery Utilizing Brown Macroalgae: Process Design, Simulation and Techno-economical Assessment. Comput. Aided Chem. Eng. 2018, 44, 337–342. [Google Scholar]
- Fasahati, P.; Saffron, C.M.; Woo, H.C.; Liu, J.J. Potential of brown algae for sustainable electricity production through anaerobic digestion. Energy Convers. Manag. 2017, 135, 297–307. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).