Synergizing Immune Balance: Curcumin Gold Nanoparticles and Ultrasound Irradiation for Macrophage Down-Regulation
Abstract
1. Introduction
2. Materials and Methods
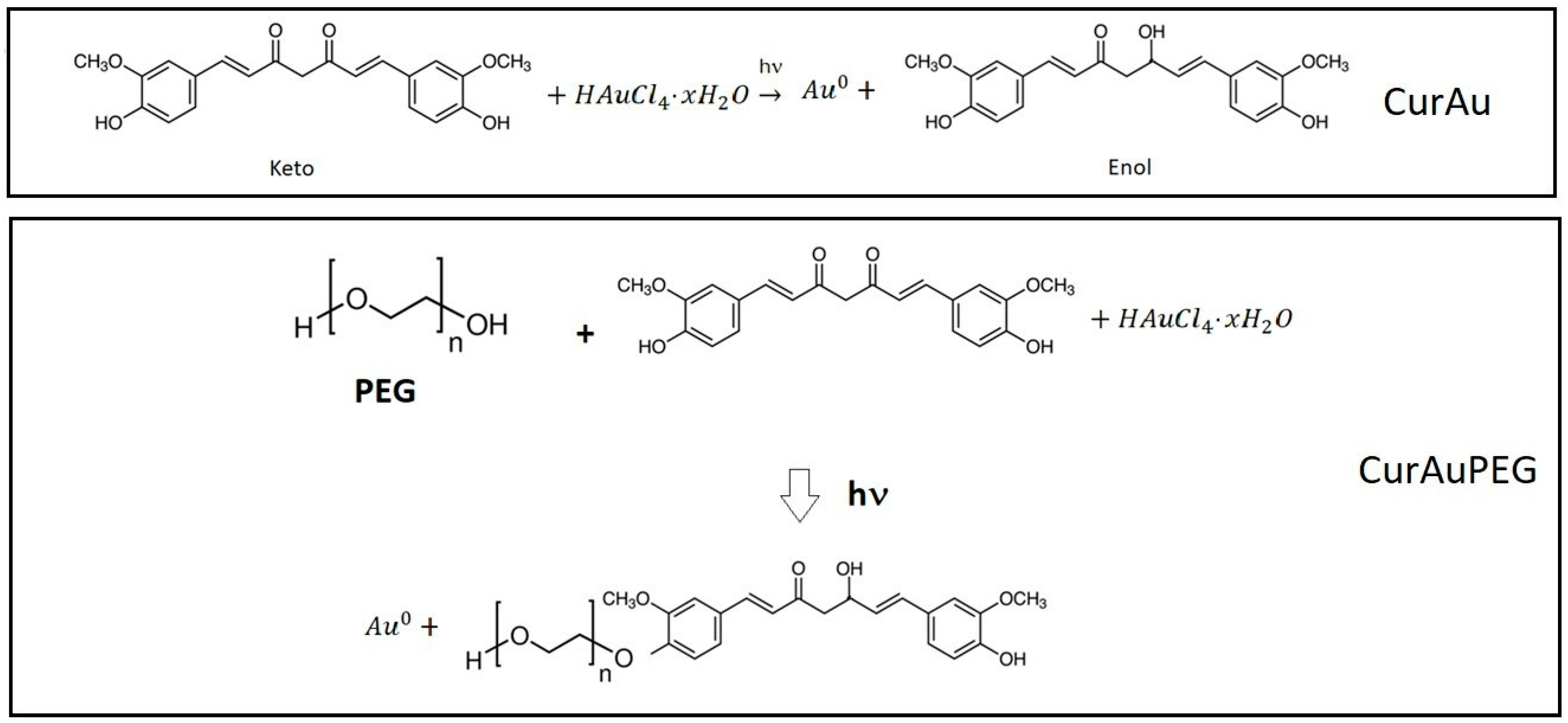
2.1. Synthesis of Gold Nanoparticles Associated with Curcumin [CurAuNPs]
2.2. Nanoparticles Characterization
2.3. Study of Singlet Oxygen Released by Ultrasound Irradiation
2.4. Cell Culture
2.5. Cytotoxicity of Nanoparticles
2.6. Ultrasound Irradiation
2.7. Statistical Analysis
3. Results
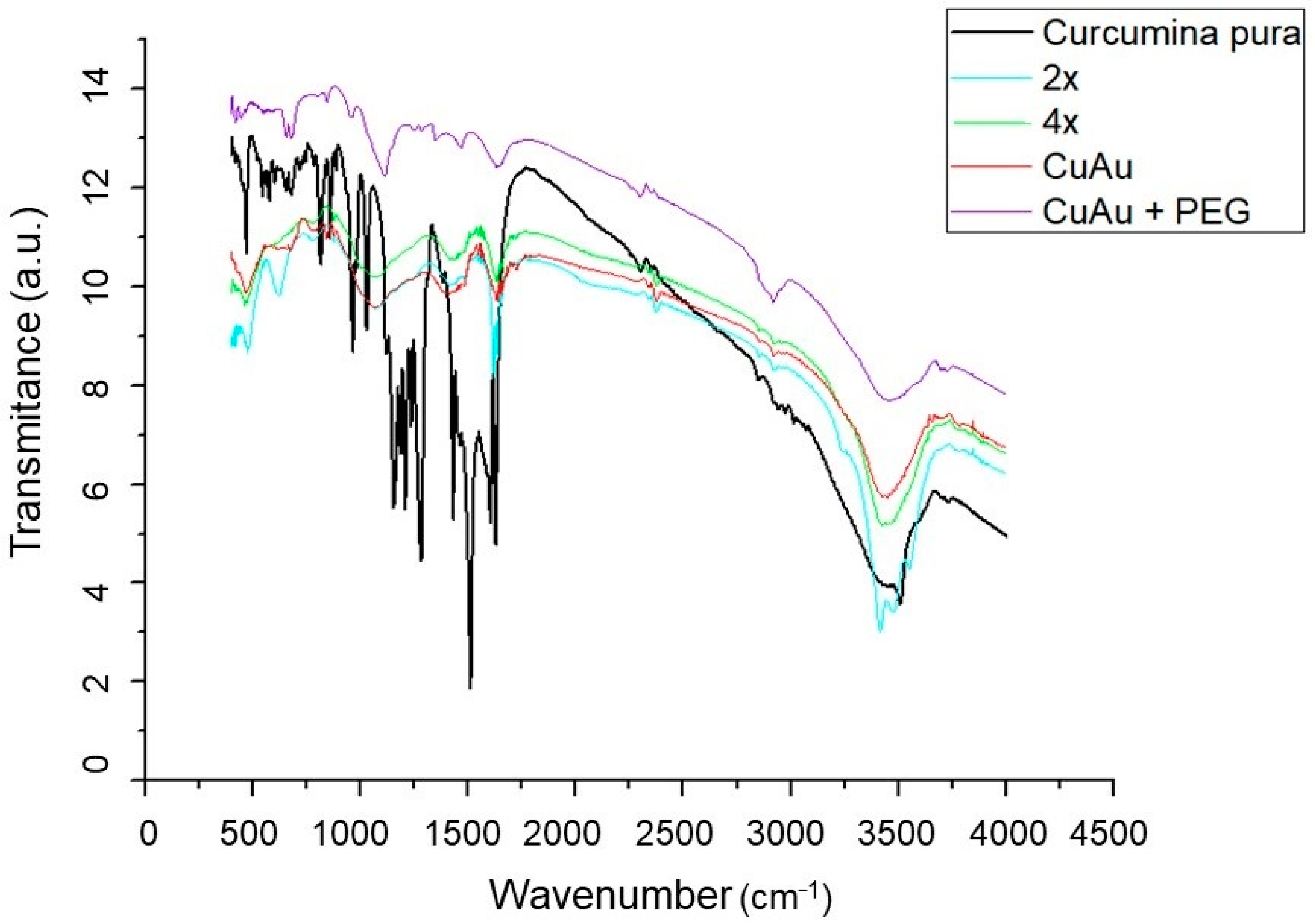
3.1. Synthesis and Characterization of CurAuNPs
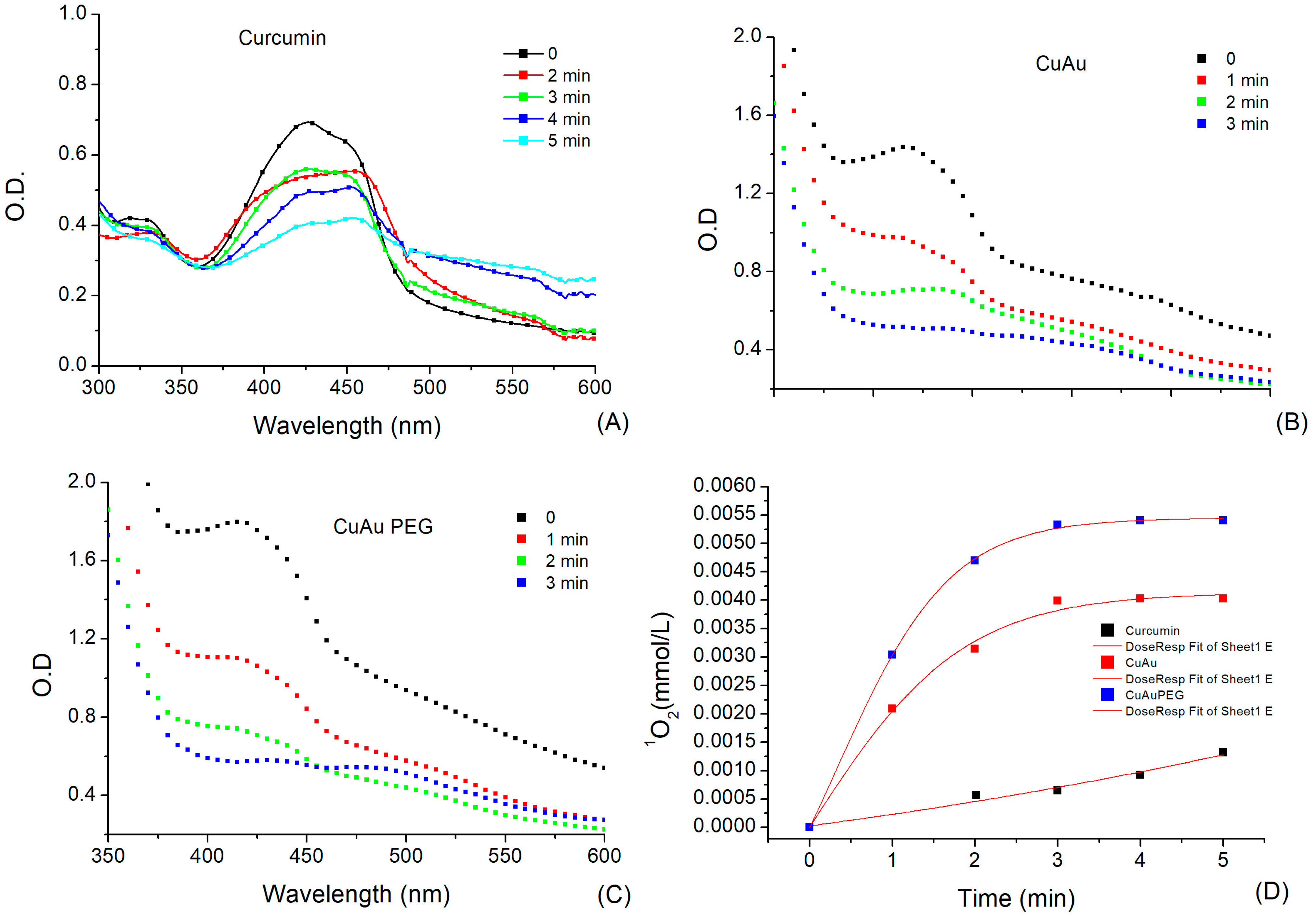
3.2. Singlet Oxygen [1O2] Release from CurAuNPs under Ultrasound Irradiation
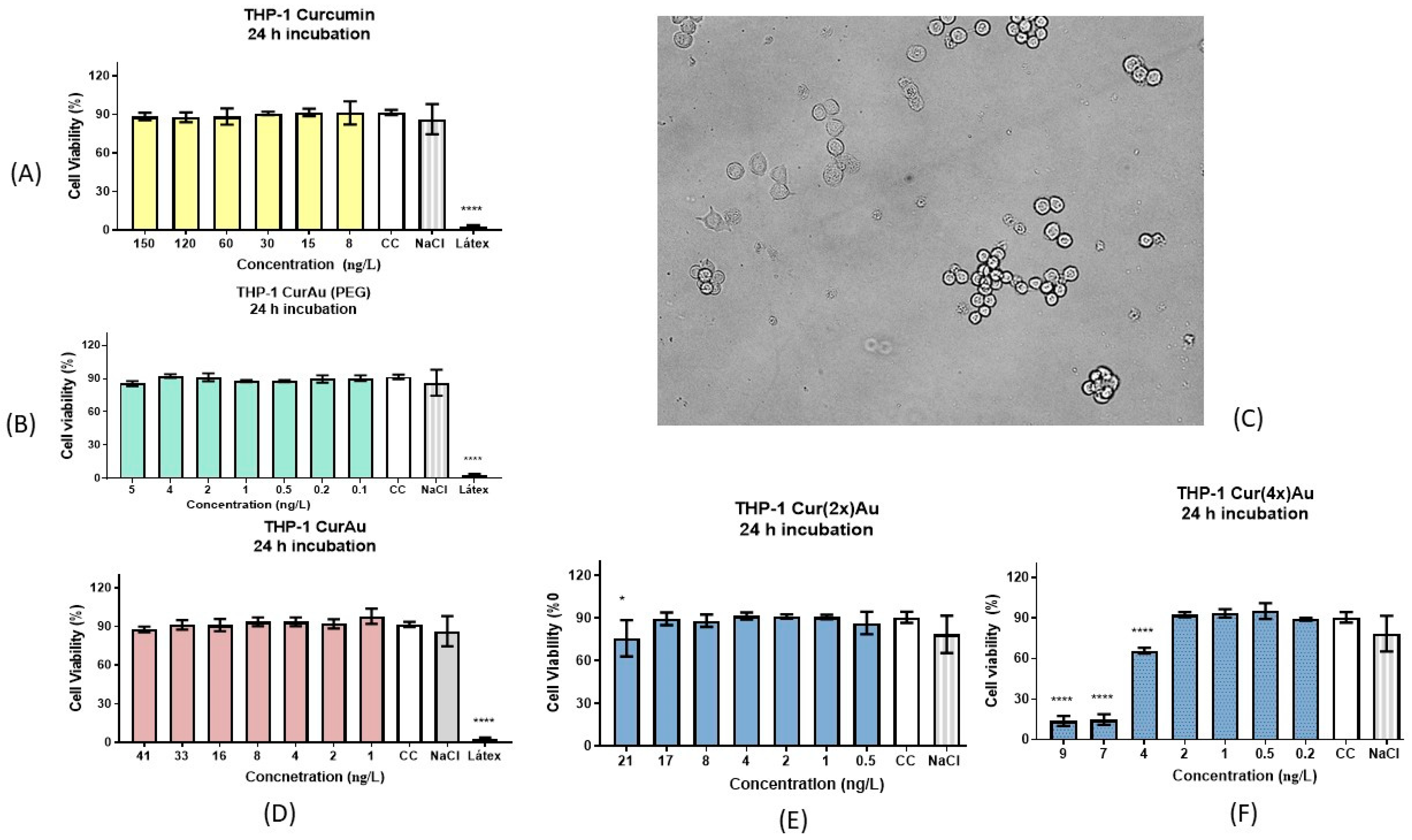
3.3. Cell Viability Study
3.4. Ultrasound Irradiation
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aggarwal, B.B.; Sundaram, C.; Malani, N.; Ichikawa, H. Curcumin: The Indian solid gold. In The Molecular Targets and Therapeutic Uses of Curcumin in Health and Disease; Aggarwal, B.B., Surh, Y.J., Shishodia, S., Eds.; Advances in Experimental Medicine and Biology; Springer: Boston, MA, USA, 2007; Volume 595, pp. 1–75. [Google Scholar]
- Aggarwal, B.B.; Harikumar, K.B. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int. J. Biochem. Cell Biol. 2009, 41, 40–59. [Google Scholar] [CrossRef]
- Anand, P.; Sundaram, C.; Jhurani, S.; Kunnumakkara, A.B.; Aggarwal, B.B. Curcumin and cancer: An “old-age” disease with an “age-old” solution. Cancer Lett. 2008, 267, 133–164. [Google Scholar] [CrossRef]
- Kazantzis, K.T.; Koutsonikoli, K.; Mavroidi, B.; Zachariadis, M.; Alexiou, P.; Pelecanou, M.; Politopoulos, K.; Alexandratou, E.; Sagnou, M. Curcumin derivatives as photosensitizers in photodynamic therapy: Photophysical properties and in vitro studies with prostate cancer cells. Photochem. Photobiol. Sci. 2020, 19, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Tomeh, M.A.; Hadianamrei, R.; Zhao, X.B. A Review of Curcumin and Its Derivatives as Anticancer Agents. Int. J. Mol. Sci. 2019, 20, 1033. [Google Scholar] [CrossRef]
- Momtazi-Borojeni, A.A.; Abdollahi, E.; Nikfar, B.; Chaichian, S.; Ekhlasi-Hundrieser, M. Curcumin as a potential modulator of M1 and M2 macrophages: New insights in atherosclerosis therapy. Heart Fail. Rev. 2019, 24, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.Y.; Geng, X.F.; Hou, J.X.; Wu, G.S. New insights into M1/M2 macrophages: Key modulators in cancer progression. Cancer Cell Int. 2021, 21, 389. [Google Scholar] [CrossRef]
- De Paoli, F.; Staels, B.; Chinetti-Gbaguidi, G. Macrophage Phenotypes and Their Modulation in Atherosclerosis. Circ. J. 2014, 78, 1775–1781. [Google Scholar] [CrossRef]
- Murray, P.J.; Wynn, T.A. Protective and pathogenic functions of macrophage subsets. Nat. Rev. Immunol. 2011, 11, 723–737. [Google Scholar] [CrossRef]
- Italiani, P.; Boraschi, D. From monocytes to M1/M2 macrophages: Phenotypical vs. functional differentiation. Front. Immunol. 2014, 5, 514. [Google Scholar] [CrossRef]
- Zhou, Y.Y.; Zhang, T.T.; Wang, X.F.; Wei, X.W.; Chen, Y.Z.; Guo, L.Y.; Zhang, J.F.; Wang, C.Q. Curcumin Modulates Macrophage Polarization through the Inhibition of the Toll-like Receptor 4 Expression and Its Signaling Pathways. Cell. Physiol. Biochem. 2015, 36, 631–641. [Google Scholar] [CrossRef]
- Yan, S.X.; Zhou, M.; Zheng, X.Y.; Xing, Y.Y.; Dong, J.; Yan, M.W.; Li, R. Anti-Inflammatory Effect of Curcumin on the Mouse Model of Myocardial Infarction through Regulating Macrophage Polarization. Mediat. Inflamm. 2021, 2021. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.K.; Sun, X.; Sang, L.M.; Zhang, M.; Yan, X.B.; Ju, Q.; Ma, X.F.; Xu, M. Curcumin inhibits malignant behavior of colorectal cancer cells by regulating M2 polarization of tumor-associated macrophages and metastasis associated in colon cancer 1 (MACC1) expression. Chem. Biol. Drug Des. 2023, 102, 1202–1212. [Google Scholar] [CrossRef] [PubMed]
- Abdollahi, E.; Johnston, T.P.; Ghaneifar, Z.; Vahedi, P.; Goleij, P.; Azhdari, S.; Moghaddam, A.S. Immunomodulatory Therapeutic Effects of Curcumin on M1/M2 Macrophage Polarization in Inflammatory Diseases. Curr. Mol. Pharmacol. 2023, 16, 2–14. [Google Scholar] [CrossRef] [PubMed]
- Noorafshan, A.; Ashkani-Esfahani, S. A Review of Therapeutic Effects of Curcumin. Curr. Pharm. Des. 2013, 19, 2032–2046. [Google Scholar] [PubMed]
- Jiang, L.; Wang, J.H.; Jiang, J.Q.; Zhang, C.M.; Zhao, M.; Chen, Z.; Wang, N.; Hu, D.D.; Liu, X.Y.; Peng, H.S.; et al. Sonodynamic therapy in atherosclerosis by curcumin nanosuspensions: Preparation design, efficacy evaluation, and mechanisms analysis. Eur. J. Pharm. Biopharm. 2020, 146, 101–110. [Google Scholar] [CrossRef]
- Wang, F.P.; Gao, Q.P.; Guo, S.Y.; Cheng, J.L.; Sun, X.; Li, Q.N.; Wang, T.Y.; Zhang, Z.G.; Cao, W.W.; Tian, Y. The Sonodynamic Effect of Curcumin on THP-1 Cell-Derived Macrophages. Biomed. Res. Int. 2013, 2013, 737264. [Google Scholar] [CrossRef]
- Rosenthal, I.; Sostaric, J.Z.; Riesz, P. Sonodynamic therapy—A review of the synergistic effects of drugs and ultrasound. Ultrason. Sonochem. 2004, 11, 349–363. [Google Scholar] [CrossRef]
- Canaparo, R.; Foglietta, F.; Barbero, N.; Serpe, L. The promising interplay between sonodynamic therapy and nanomedicine. Adv. Drug Deliv. Rev. 2022, 189, 114495. [Google Scholar] [CrossRef]
- Costley, D.; Mc Ewan, C.; Fowley, C.; McHale, A.P.; Atchison, J.; Nomikou, N.; Callan, J.F. Treating cancer with sonodynamic therapy: A review. Int. J. Hyperth. 2015, 31, 107–117. [Google Scholar] [CrossRef]
- Geng, C.; Zhang, Y.L.; Hidru, T.H.; Zhi, L.Y.; Tao, M.X.; Zou, L.X.; Chen, C.; Li, H.H.; Liu, Y. Sonodynamic therapy: A potential treatment for atherosclerosis. Life Sci. 2018, 207, 304–313. [Google Scholar] [CrossRef]
- Bai, W.K.; Shen, E.; Hu, B. Induction of the apoptosis of cancer cell by sonodynamic therapy: A review. Chin. J. Cancer Res. 2012, 24, 368–373. [Google Scholar] [CrossRef]
- Gong, Z.R.; Dai, Z.F. Design and Challenges of Sonodynamic Therapy System for Cancer Theranostics: From Equipment to Sensitizers. Adv. Sci. 2021, 8, 2002178. [Google Scholar] [CrossRef]
- Kayani, Z.; Vais, R.D.; Soratijahromi, E.; Mohammadi, S.; Sattarahmady, N. Curcumin-gold-polyethylene glycol nanoparticles as a nanosensitizer for photothermal and sonodynamic therapies: In vitro and animal model studies. Photodiagn Photodyn. Ther. 2021, 33, 102139. [Google Scholar] [CrossRef] [PubMed]
- Xing, X.J.; Zhao, S.J.; Xu, T.; Huang, L.; Zhang, Y.; Lan, M.H.; Lin, C.W.; Zheng, X.L.; Wang, P.F. Advances and perspectives in organic sonosensitizers for sonodynamic therapy. Coord. Chem. Rev. 2021, 445, 214087. [Google Scholar] [CrossRef]
- Zheng, L.B.; Sun, X.Y.; Zhu, X.; Lv, F.X.; Zhong, Z.Y.; Zhang, F.; Guo, W.H.; Cao, W.W.; Yang, L.M.; Tian, Y. Apoptosis of THP-1 Derived Macrophages Induced by Sonodynamic Therapy Using a New Sonosensitizer Hydroxyl Acetylated Curcumin. PLoS ONE 2014, 9, e93133. [Google Scholar] [CrossRef]
- Obeid, M.A.; Alsaadi, M.; Aljabali, A.A. Recent updates in curcumin delivery. J. Liposome Res. 2023, 33, 53–64. [Google Scholar] [CrossRef]
- Goncalves, K.D.; Vieira, D.P.; Courrol, L.C. Synthesis and characterization of aminolevulinic acid gold nanoparticles: Photo and sonosensitizer agent for atherosclerosis. J. Lumin. 2018, 197, 317–323. [Google Scholar] [CrossRef]
- Goncalvez, K.D.; Vieira, D.P.; Courrol, L.C. Study of THP-1 Macrophage Viability after Sonodynamic Therapy Using Methyl Ester of 5-Aminolevulinic Acid Gold Nanoparticles. Ultrasound Med. Biol. 2018, 44, 2009–2017. [Google Scholar] [CrossRef] [PubMed]
- Qian, X.Q.; Zheng, Y.Y.; Chen, Y. Micro/Nanoparticle-Augmented Sonodynamic Therapy (SDT): Breaking the Depth Shallow of Photoactivation. Adv. Mater. 2016, 28, 8097–8129. [Google Scholar] [CrossRef]
- Xu, H.Y.; Zhang, X.; Han, R.B.; Yang, P.M.; Ma, H.F.; Song, Y.; Lu, Z.C.; Yin, W.D.; Wu, X.X.; Wang, H. Nanoparticles in sonodynamic therapy: State of the art review. RSC Adv. 2016, 6, 50697–50705. [Google Scholar] [CrossRef]
- Laurindo, L.F.; de Carvalho, G.M.; Zanuso, B.D.; Figueira, M.E.; Direito, R.; Goulart, R.D.; Buglio, D.S.; Barbalho, S.M. Curcumin-Based Nanomedicines in the Treatment of Inflammatory and Immunomodulated Diseases: An Evidence-Based Comprehensive Review. Pharmaceutics 2023, 15, 229. [Google Scholar] [CrossRef]
- Costantino, M.; Corno, C.; Colombo, D.; Perego, P. Curcumin and Related Compounds in Cancer Cells: New Avenues for Old Molecules. Front. Pharmacol. 2022, 13, 889816. [Google Scholar] [CrossRef] [PubMed]
- Kurapov, P.B.; Bakhtenko, E.Y. Gold Nanoparticles in the Diagnosis and Treatment of Cancer. Bull. Russ. State Med. Univ. 2018, 86–93. [Google Scholar] [CrossRef]
- Patra, D.; El Kurdi, R. Curcumin as a novel reducing and stabilizing agent for the green synthesis of metallic nanoparticles. Green. Chem. Lett. Rev. 2021, 14, 474–487. [Google Scholar] [CrossRef]
- Sreelakshmi, C.; Goel, N.; Datta, K.K.R.; Addlagatta, A.; Ummanni, R.; Reddy, B.V.S. Green Synthesis of Curcumin Capped Gold Nanoparticles and Evaluation of Their Cytotoxicity. Nanosci. Nanotechnol. Lett. 2013, 5, 1258–1265. [Google Scholar] [CrossRef]
- Singh, D.K.; Jagannathan, R.; Khandelwal, P.; Abraham, P.M.; Poddar, P. In situ synthesis and surface functionalization of gold nanoparticles with curcumin and their antioxidant properties: An experimental and density functional theory investigation. Nanoscale 2013, 5, 1882–1893. [Google Scholar] [CrossRef] [PubMed]
- El Kurdi, R.; Patra, D. Capping of supramolecular curcubit 7 uril facilitates formation of Au nanorods during pre-reduction by curcumin. Colloids Surf. A Physicochem. Eng. Asp. 2018, 553, 97–104. [Google Scholar] [CrossRef]
- dos Santos Courrol, D.; Teixeira, B.H.; Pereira, C.B.; Franzolin, M.R.; Courrol, L.C. Pegylated Curcumin with Gold Nanoparticles: Antimicrobial Agent Evaluation. J. Biomed. Eng. Biosci. 2016, 3, 43–47. [Google Scholar] [CrossRef]
- Delaney, L.J.; Isguven, S.; Eisenbrey, J.R.; Hickok, N.J.; Forsberg, F. Making waves: How ultrasound-targeted drug delivery is changing pharmaceutical approaches. Mater. Adv. 2022, 3, 3023–3040. [Google Scholar] [CrossRef] [PubMed]
- Hornsby, T.K.; Kashkooli, F.M.; Jakhmola, A.; Kolios, M.C.; Tavakkoli, J. Multiphysics Modeling of Low-Intensity Pulsed Ultrasound Induced Chemotherapeutic Drug Release from the Surface of Gold Nanoparticles. Cancers 2023, 15, 523. [Google Scholar] [CrossRef]
- Ilbeigi, S.; Ranjbar, A.; Zahraie, N.; Vais, R.D.; Monjezi, M.R.; Sattarahmady, N. Sonodynamic therapy of pancreatic cancer cells based on synergistic chemotherapeutic effects of selenium-PEG-curcumin nanoparticles and gemcitabine. Appl. Phys. A 2023, 129, 82. [Google Scholar] [CrossRef]
- Belfield, K.D.; Bondar, M.V.; Przhonska, O.V. Singlet oxygen quantum yield determination for a fluorene-based two-photon photosensitizer. J. Fluoresc. 2006, 16, 111–117. [Google Scholar] [CrossRef]
- Carloni, P.; Damiani, E.; Greci, L.; Stipa, P.; Tanfani, F.; Tartaglini, E.; Wozniak, M. On the use of 1,3-diphenylisobenzofuran (dpbf)—Reactions with carbon and oxygen-centered radicals in model and natural systems. Res. Chem. Intermed. 1993, 19, 395–405. [Google Scholar] [CrossRef]
- Fudimura, K.A.; Seabra, A.B.; Santos, M.C.; Haddad, P.S. Synthesis and Characterization of Methylene Blue-Containing Silica-Coated Magnetic Nanoparticles for Photodynamic Therapy. J. Nanosci. Nanotechnol. 2017, 17, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Ogunsipe, A.; Maree, D.; Nyokong, T. Solvent effects on the photochemical and fluorescence properties of zinc phthalocyanine derivatives. J. Mol. Struct. 2003, 650, 131–140. [Google Scholar] [CrossRef]
- Mohan, P.R.K.; Sreelakshmi, G.; Muraleedharan, C.V.; Joseph, R. Water soluble complexes of curcumin with cyclodextrins: Characterization by FT-Raman spectroscopy. Vibrat. Spectrosc. 2012, 62, 77–84. [Google Scholar] [CrossRef]
- Helson, L. Curcumin (diferuloylmethane) delivery methods: A review. Biofactors 2013, 39, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Yakub, G.; Manolova, N.E.; Rashkov, I.B.; Markova, N.; Toshkova, R.; Georgieva, A.; Mincheva, R.; Toncheva, A.; Raquez, J.M.; Dubois, P. Pegylated Curcumin Derivative: Water-Soluble Conjugates with Antitumor and Antibacterial Activity. ACS Omega 2022, 7, 36403–36414. [Google Scholar] [CrossRef] [PubMed]
- Sazgarnia, A.; Shanei, A.; Eshghi, H.; Hassanzadeh-Khayyat, M.; Esmaily, H.; Shanei, M.M. Detection of sonoluminescence signals in a gel phantom in the presence of Protoporphyrin IX conjugated to gold nanoparticles. Ultrasonics 2013, 53, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Shanei, A.; Shanei, M.M. Effect of gold nanoparticle size on acoustic cavitation using chemical dosimetry method. Ultrason. Sonochem. 2017, 34, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Daneshvar, F.; Salehi, F.; Kayani, Z.; Sattarahmady, N.; Vais, R.D.; Azarpira, N. Fractionated Sonodynamic Therapy Using Gold@Poly(ortho-aminophenol) Nanoparticles and Multistep Low-Intensity Ultrasound Irradiation to Treat Melanoma Cancer: In Vitro and In Vivo Studies. Ultrasound Med. Biol. 2023, 49, 1299–1308. [Google Scholar] [CrossRef]
- Ozlem Caliskan, S.; Ozen, H.; Kaya, I.; Ilikci Sagkan, R.; Ertabaklar, H. An In Vitro Study on Sonodynamic Therapy of Leishmania tropica Using curcumin. Mikrobiyol. Bul. 2022, 56, 706–721. [Google Scholar] [CrossRef]
- Abrahamse, H.; Hamblin, M.R. New photosensitizers for photodynamic therapy. Biochem. J. 2016, 473, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Therapeutic Roles of Curcumin: Lessons Learned from Clinical Trials. AAPS J. 2013, 15, 195–218. [Google Scholar] [CrossRef] [PubMed]
- McNally, S.J.; Harrison, E.M.; Ross, J.A.; Garden, O.J.; Wigmore, S.J. Curcumin induces heme oxygenase 1 through generation of reactive oxygen species, p38 activation and phosphatase inhibition. Int. J. Mol. Med. 2007, 19, 165–172. [Google Scholar] [CrossRef]
- Kunwar, A.; Barik, A.; Mishra, B.; Rathinasamy, K.; Pandey, R.; Priyadarsini, K.I. Quantitative cellular uptake, localization and cytotoxicity of curcumin in normal and tumor cells. Biochim. Biophys. Acta Gen. Subj. 2008, 1780, 673–679. [Google Scholar] [CrossRef]
- Andrade, R.G.D.; Reis, B.; Costas, B.; Lima, S.A.C.; Reis, S. Modulation of Macrophages M1/M2 Polarization Using Carbohydrate-Functionalized Polymeric Nanoparticles. Polymers 2021, 13, 88. [Google Scholar] [CrossRef]
- Lomovskaya, Y.V.; Kobyakova, M.I.; Senotov, A.S.; Lomovsky, A.I.; Minaychev, V.V.; Fadeeva, I.S.; Shtatnova, D.Y.; Krasnov, K.S.; Zvyagina, A.I.; Akatov, V.S.; et al. Macrophage-like THP-1 Cells Derived from High-Density Cell Culture Are Resistant to TRAIL-Induced Cell Death via Down-Regulation of Death-Receptors DR4 and DR5. Biomolecules 2022, 12, 150. [Google Scholar] [CrossRef] [PubMed]
- Virag, L.; Jaen, R.I.; Regdon, Z.; Bosca, L.; Prieto, P. Self-defense of macrophages against oxidative injury: Fighting for their own survival. Redox Biol. 2019, 26, 101261. [Google Scholar] [CrossRef]
- Perez, S.; Rius-Perez, S. Macrophage Polarization and Reprogramming in Acute Inflammation: A Redox Perspective. Antioxidants 2022, 11, 1394. [Google Scholar] [CrossRef]
- Tu, J.J.; Huang, W.; Zhang, W.W.; Mei, J.W.; Zhu, C. A Tale of Two Immune Cells in Rheumatoid Arthritis: The Crosstalk Between Macrophages and T Cells in the Synovium. Front. Immunol. 2021, 12, 655477. [Google Scholar] [CrossRef] [PubMed]


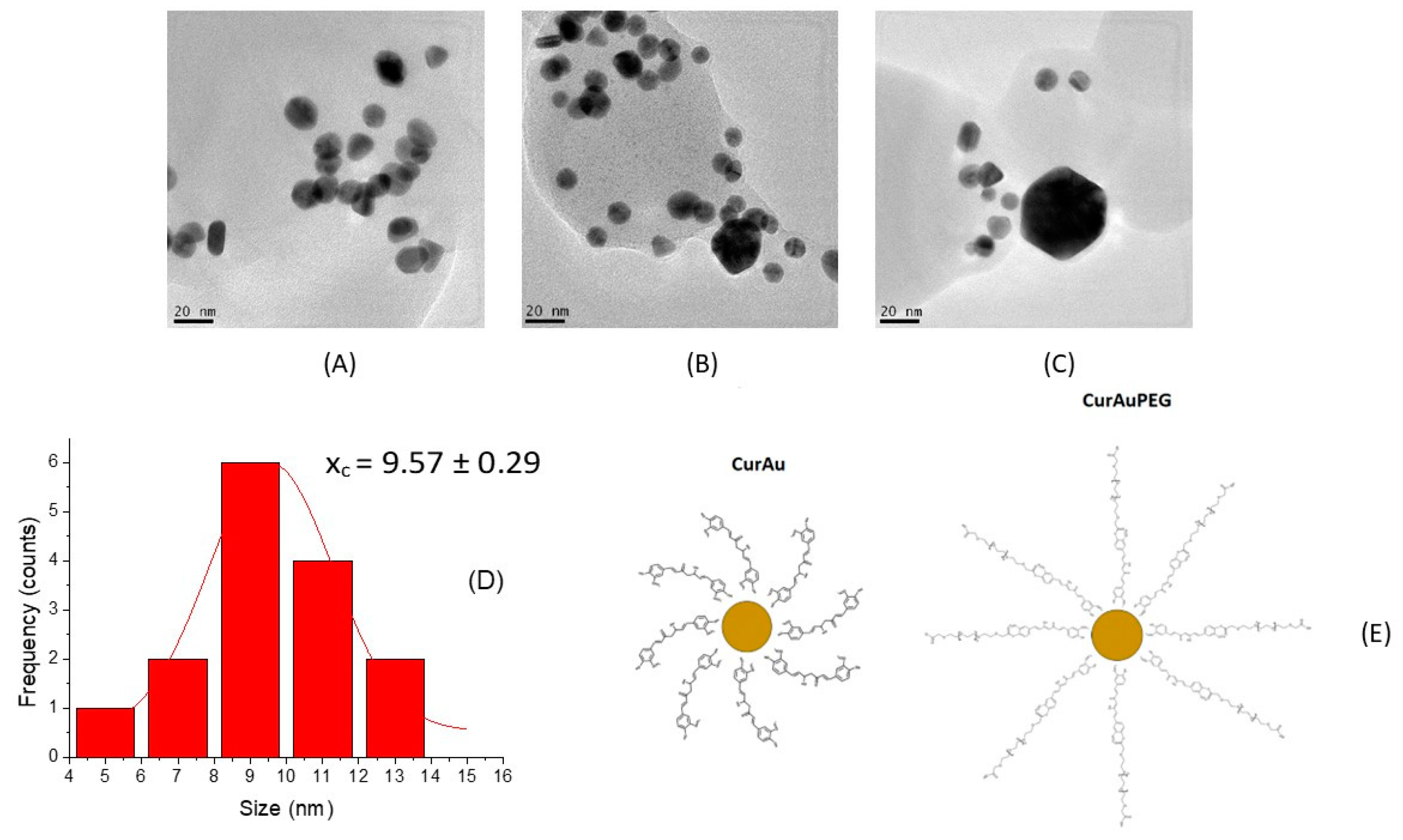


| Zeta Potential (mV) | Size (nm) | |
|---|---|---|
| CurAu | −19.16 | 9.6 nm |
| CurAu PEG | −23.00 | 20.0 nm |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teixeira, B.H.; Gonçalves, K.d.O.; Vieira, D.P.; Coronato Courrol, L. Synergizing Immune Balance: Curcumin Gold Nanoparticles and Ultrasound Irradiation for Macrophage Down-Regulation. AppliedChem 2024, 4, 70-85. https://doi.org/10.3390/appliedchem4010006
Teixeira BH, Gonçalves KdO, Vieira DP, Coronato Courrol L. Synergizing Immune Balance: Curcumin Gold Nanoparticles and Ultrasound Irradiation for Macrophage Down-Regulation. AppliedChem. 2024; 4(1):70-85. https://doi.org/10.3390/appliedchem4010006
Chicago/Turabian StyleTeixeira, Bruna Henrique, Karina de Oliveira Gonçalves, Daniel Perez Vieira, and Lilia Coronato Courrol. 2024. "Synergizing Immune Balance: Curcumin Gold Nanoparticles and Ultrasound Irradiation for Macrophage Down-Regulation" AppliedChem 4, no. 1: 70-85. https://doi.org/10.3390/appliedchem4010006
APA StyleTeixeira, B. H., Gonçalves, K. d. O., Vieira, D. P., & Coronato Courrol, L. (2024). Synergizing Immune Balance: Curcumin Gold Nanoparticles and Ultrasound Irradiation for Macrophage Down-Regulation. AppliedChem, 4(1), 70-85. https://doi.org/10.3390/appliedchem4010006









