Future Therapeutics: Targeting the NLRP3 Inflammasome Pathway to Manage Diabetic Retinopathy Development and Progression
Abstract
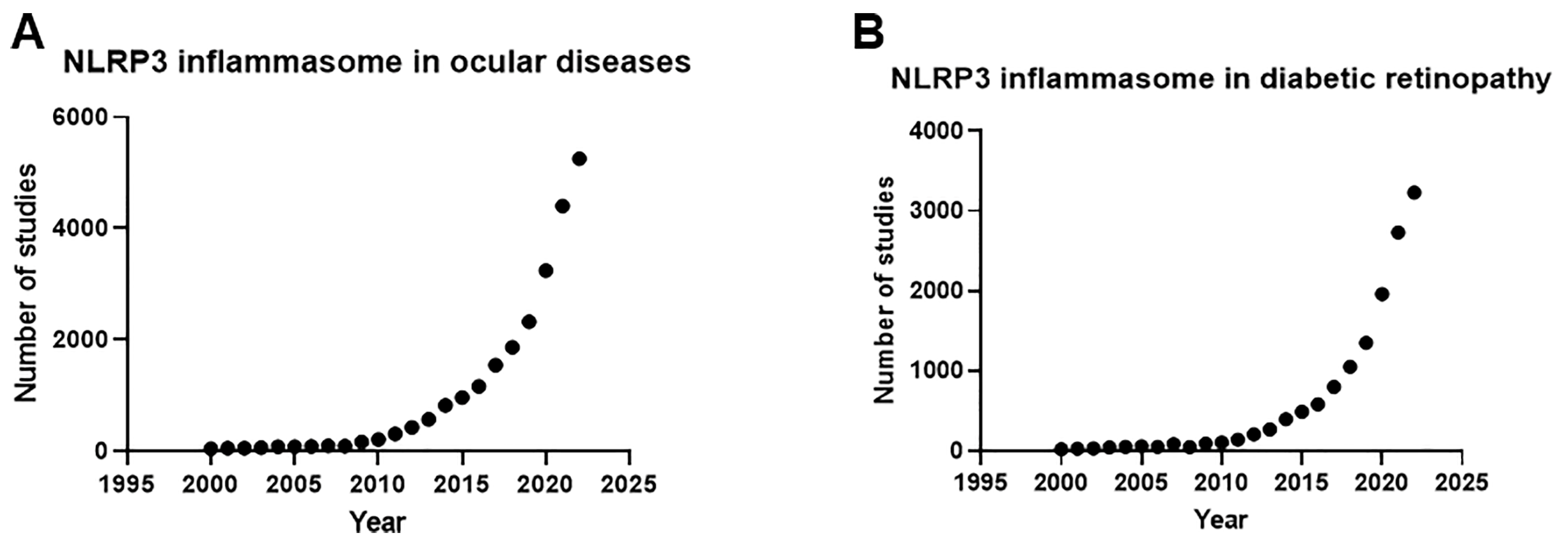
1. Introduction
2. Methods
3. Clinical Risk Factors of DR
3.1. Hyperglycemia
3.2. Hypertension
3.3. Dyslipidemia
4. The NLRP3 Inflammasome
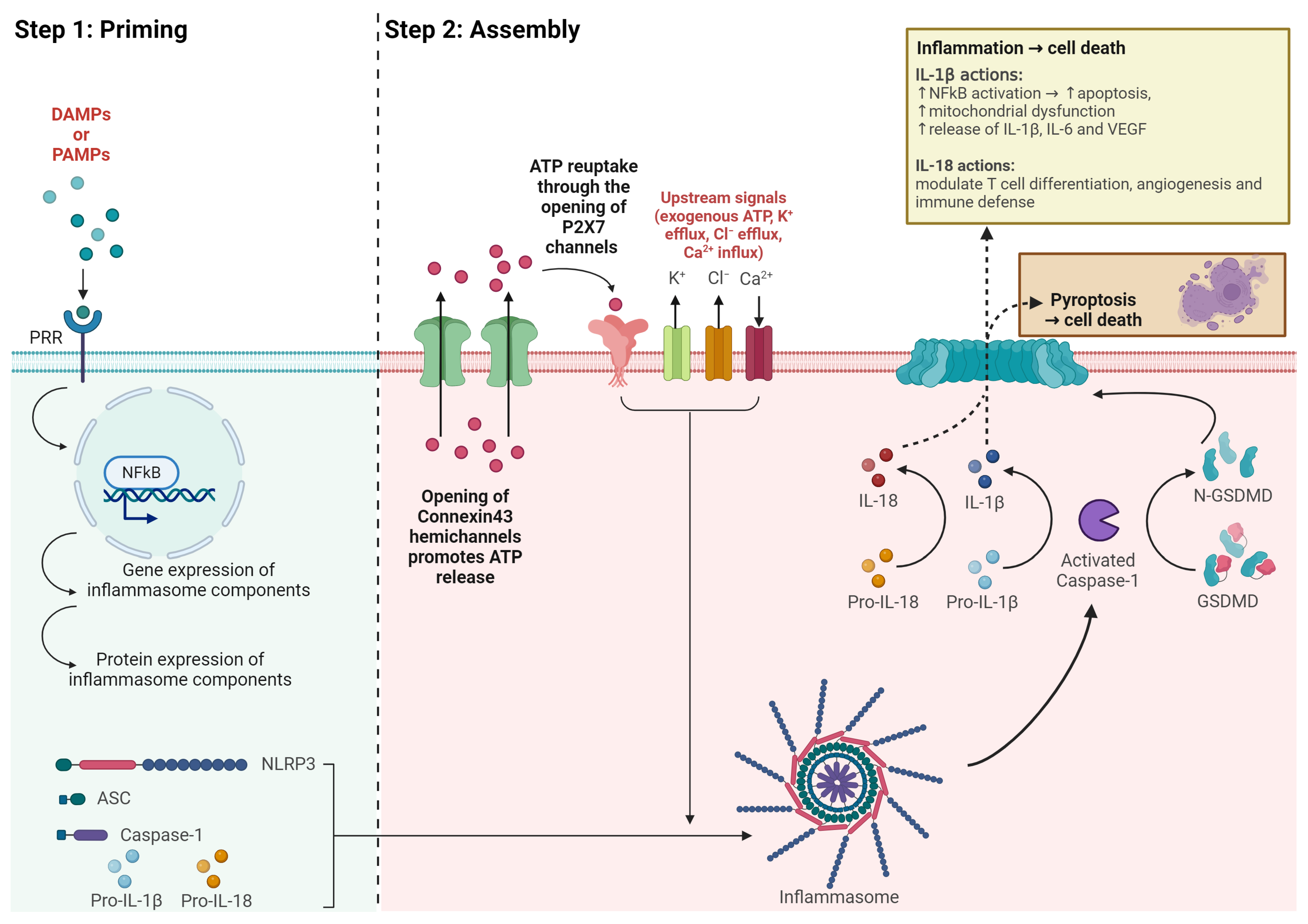
4.1. The NLRP3 Inflammasome
4.2. Structure of the NLRP3 Inflammasome
4.3. Clinical Evidence of the NLRP3 Inflammasome in DR Progression
4.4. NLRP3 Inflammasome Activation
4.5. Cytokine Release
4.6. Inflammasome-Targeting Novel Therapeutics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Teo, Z.L.; Tham, Y.-C.; Yu, M.; Chee, M.L.; Rim, T.H.; Cheung, N.; Bikbov, M.M.; Wang, Y.X.; Tang, Y.; Lu, Y. Global prevalence of diabetic retinopathy and projection of burden through 2045: Systematic review and meta-analysis. Ophthalmology 2021, 128, 1580–1591. [Google Scholar] [CrossRef]
- Steinmetz, J.D.; Bourne, R.R.; Briant, P.S.; Flaxman, S.R.; Taylor, H.R.; Jonas, J.B.; Abdoli, A.A.; Abrha, W.A.; Abualhasan, A.; Abu-Gharbieh, E.G. Causes of blindness and vision impairment in 2020 and trends over 30 years, and prevalence of avoidable blindness in relation to VISION 2020: The Right to Sight: An analysis for the Global Burden of Disease Study. Lancet Glob. Health 2021, 9, e144–e160. [Google Scholar] [CrossRef]
- Pan, W.W.; Lin, F.; Fort, P.E. The innate immune system in diabetic retinopathy. Prog. Retin. Eye Res. 2021, 84, 100940. [Google Scholar] [CrossRef]
- Abderrazak, A.; Syrovets, T.; Couchie, D.; El Hadri, K.; Friguet, B.; Simmet, T.; Rouis, M. NLRP3 inflammasome: From a danger signal sensor to a regulatory node of oxidative stress and inflammatory diseases. Redox Biol. 2015, 4, 296–307. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Hara, H.; Núñez, G. Mechanism and regulation of NLRP3 inflammasome activation. Trends Biochem. Sci. 2016, 41, 1012–1021. [Google Scholar] [CrossRef]
- Shao, B.-Z.; Xu, Z.-Q.; Han, B.-Z.; Su, D.-F.; Liu, C. NLRP3 inflammasome and its inhibitors: A review. Front. Pharmacol. 2015, 6, 262. [Google Scholar] [CrossRef] [PubMed]
- Swanson, K.V.; Deng, M.; Ting, J.P.-Y. The NLRP3 inflammasome: Molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 2019, 19, 477–489. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-M.; Kim, J.-J.; Kim, H.J.; Shong, M.; Ku, B.J.; Jo, E.-K. Upregulated NLRP3 inflammasome activation in patients with type 2 diabetes. Diabetes 2013, 62, 194–204. [Google Scholar] [CrossRef]
- Bullón, P.; Alcocer-Gómez, E.; Carrión, A.M.; Marín-Aguilar, F.; Garrido-Maraver, J.; Román-Malo, L.; Ruiz-Cabello, J.; Culic, O.; Ryffel, B.; Apetoh, L. AMPK phosphorylation modulates pain by activation of NLRP3 inflammasome. Antioxid. Redox. Signal. 2016, 24, 157–170. [Google Scholar] [CrossRef]
- Fan, Y.-P.; Wu, C.-T.; Lin, J.-L.; Hsiung, C.A.; Liu, H.Y.; Lai, J.-N.; Yang, C.-C. Metformin treatment is associated with a decreased risk of nonproliferative diabetic retinopathy in patients with type 2 diabetes mellitus: A population-based cohort study. J. Diabetes Res. 2020, 2020, 9161039. [Google Scholar] [CrossRef]
- American Diabetes Association Professional Practice Committee. 3. Prevention or delay of diabetes and associated comorbidities: Standards of care in diabetes—2024. Diabetes Care 2024, 47, S43–S51. [Google Scholar] [CrossRef] [PubMed]
- Assessment, G. 6. Glycemic targets: Standards of medical care in diabetes—2022. Diabetes Care 2022, 45, S83. [Google Scholar]
- Stratton, I.; Kohner, E.; Aldington, S.; Turner, R.; Holman, R.; Manley, S.; Matthews, D. UKPDS 50: Risk factors for incidence and progression of retinopathy in Type II diabetes over 6 years from diagnosis. Diabetologia 2001, 44, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Klonoff, D.C. United Kingdom prospective diabetes study follow-up studies establish a legacy effect of therapy for hyperglycemia but not hypertension. J. Diabetes Sci. Technol. 2008, 2, 922–924. [Google Scholar] [CrossRef] [PubMed]
- The United Kingdom Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 1998, 352, 837–853. [Google Scholar] [CrossRef]
- Action to Control Cardiovascular Risk in Diabetes Follow-On (ACCORDION) Eye Study Group. Persistent effects of intensive glycemic control on retinopathy in type 2 diabetes in the action to control cardiovascular risk in diabetes (ACCORD) follow-on study. Diabetes Care 2016, 39, 1089–1100. [Google Scholar] [CrossRef] [PubMed]
- ADVANCE Collaborative Group. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 2008, 358, 2560–2572. [Google Scholar] [CrossRef]
- Azad, N.; Agrawal, L.; Emanuele, N.V.; Klein, R.; Bahn, G.D.; Reaven, P. Association of blood glucose control and pancreatic reserve with diabetic retinopathy in the Veterans Affairs Diabetes Trial (VADT). Diabetologia 2014, 57, 1124–1131. [Google Scholar] [CrossRef] [PubMed]
- Feldman-Billard, S.; Larger, É.; Massin, P. Early worsening of diabetic retinopathy after rapid improvement of blood glucose control in patients with diabetes. Diabetes Metab. 2018, 44, 4–14. [Google Scholar] [CrossRef]
- Lim, S.W.; van Wijngaarden, P.; Harper, C.A.; Al-Qureshi, S.H. Early worsening of diabetic retinopathy due to intensive glycaemic control. Clin. Exp. Ophthalmol. 2019, 47, 265–273. [Google Scholar] [CrossRef]
- Chew, E.Y.; Davis, M.D.; Danis, R.P.; Lovato, J.F.; Perdue, L.H.; Greven, C.; Genuth, S.; Goff, D.C.; Leiter, L.A.; Ismail-Beigi, F. The effects of medical management on the progression of diabetic retinopathy in persons with type 2 diabetes: The Action to Control Cardiovascular Risk in Diabetes (ACCORD) Eye Study. Ophthalmology 2014, 121, 2443–2451. [Google Scholar] [CrossRef] [PubMed]
- The United Kingdom Prospective Diabetes Study (UKPDS) Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. Br. Med. J. 1998, 317, 703–713. [Google Scholar] [CrossRef]
- Do, D.V.; Han, G.; Abariga, S.A.; Sleilati, G.; Vedula, S.S.; Hawkins, B.S. Blood pressure control for diabetic retinopathy. Cochrane Database Syst. Rev. 2023, 3, CD006127. [Google Scholar]
- Wang, B.; Wang, F.; Zhang, Y.; Zhao, S.-H.; Zhao, W.-J.; Yan, S.-L.; Wang, Y.-G. Effects of RAS inhibitors on diabetic retinopathy: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2015, 3, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Klein, B.E.; Klein, R.; Moss, S.E. Is serum cholesterol associated with progression of diabetic retinopathy or macular edema in persons with younger-onset diabetes of long duration? Am. J. Ophthalmol. 1999, 128, 652–654. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, C.; Shi, K.; Yin, X. Relationship between dyslipidemia and diabetic retinopathy: A systematic review and meta-analysis. Medicine 2018, 97, e12283. [Google Scholar] [CrossRef]
- Cikamatana, L.; Mitchell, P.; Rochtchina, E.; Foran, S.; Wang, J. Five-year incidence and progression of diabetic retinopathy in a defined older population: The Blue Mountains Eye Study. Eye 2007, 21, 465–471. [Google Scholar] [CrossRef]
- Morisaki, N.; Watanabe, S.; Kobayashi, J.; Kanzaki, T.; Takahashi, K.; Yokote, K.; Tezuka, M.; Tashiro, J.; Inadera, H.; Saito, Y. Diabetic control and progression of retinopathy in elderly patients: Five-year follow-up study. J. Am. Geriatr. Soc. 1994, 42, 142–145. [Google Scholar] [CrossRef] [PubMed]
- Keech, A.C.; Mitchell, P.; Summanen, P.; O’Day, J.; Davis, T.M.; Moffitt, M.; Taskinen, M.-R.; Simes, R.J.; Tse, D.; Williamson, E. Effect of fenofibrate on the need for laser treatment for diabetic retinopathy (FIELD study): A randomised controlled trial. Lancet 2007, 370, 1687–1697. [Google Scholar] [CrossRef]
- Chandra, A.; Kaur, P.; Sahu, S.K.; Mittal, A. A new insight into the treatment of diabetes by means of pan PPAR agonists. Chem. Biol. Drug Des. 2022, 100, 947–967. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, F.; Zhang, X.; Cheng, R.; Ma, J.-x.; Yi, J.; Li, J. Fenofibrate ameliorates diabetic retinopathy by modulating Nrf2 signaling and NLRP3 inflammasome activation. Mol. Cell. Biochem. 2018, 445, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Han, X.; Yao, Z.; Sun, Y.; Yu, J.; Cai, J.; Ren, G.; Jiang, G.; Han, F.; Hospital, Z.X. PPARα agonist stimulated angiogenesis by improving endothelial precursor cell function via a NLRP3 inflammasome pathway. Cell. Physiol. Biochem. 2017, 42, 2255–2266. [Google Scholar] [CrossRef] [PubMed]
- Paik, S.; Kim, J.K.; Silwal, P.; Sasakawa, C.; Jo, E.-K. An update on the regulatory mechanisms of NLRP3 inflammasome activation. Cell. Mol. Immunol. 2021, 18, 1141–1160. [Google Scholar] [CrossRef]
- Jacobs, S.R.; Damania, B. NLRs, inflammasomes, and viral infection. J. Leukoc. Biol. 2012, 92, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Martinon, F.; Burns, K.; Tschopp, J. The inflammasome: A molecular platform triggering activation of inflammatory caspases and processing of proIL-β. Mol. Cell 2002, 10, 417–426. [Google Scholar] [CrossRef]
- Dinarello, C.A. A clinical perspective of IL-1β as the gatekeeper of inflammation. Eur. J. Immunol. 2011, 41, 1203–1217. [Google Scholar] [CrossRef] [PubMed]
- Velloso, F.J.; Trombetta-Lima, M.; Anschau, V.; Sogayar, M.C.; Correa, R.G. NOD-like receptors: Major players (and targets) in the interface between innate immunity and cancer. Biosci. Rep. 2019, 39, BSR20181709. [Google Scholar] [CrossRef]
- Duez, H.; Pourcet, B. Nuclear receptors in the control of the NLRP3 inflammasome pathway. Front. Endocrinol. 2021, 12, 630536. [Google Scholar] [CrossRef]
- Wang, C.; Yang, T.; Xiao, J.; Xu, C.; Alippe, Y.; Sun, K.; Kanneganti, T.-D.; Monahan, J.B.; Abu-Amer, Y.; Lieberman, J. NLRP3 inflammasome activation triggers gasdermin D–independent inflammation. Sci. Immunol. 2021, 6, eabj3859. [Google Scholar] [CrossRef]
- Loukovaara, S.; Piippo, N.; Kinnunen, K.; Hytti, M.; Kaarniranta, K.; Kauppinen, A. NLRP3 inflammasome activation is associated with proliferative diabetic retinopathy. Acta Ophthalmol. 2017, 95, 803–808. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, X.; Liao, N.; Mi, L.; Peng, Y.; Liu, B.; Zhang, S.; Wen, F. Enhanced Expression of NLRP3 Inflammasome-Related Inflammation in Diabetic Retinopathy. Investig. Ophthalmol. Vis. Sci. 2018, 59, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.Y.J.; Murphy, R.; Rupenthal, I.D.; Mugisho, O.O. Correlation between the progression of diabetic retinopathy and inflammasome biomarkers in vitreous and serum—A systematic review. BMC Ophthalmol. 2022, 22, 238. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.Y.-J.; Rupenthal, I.D.; Booth, M.; Murphy, R.; Mugisho, O.O. Systemic Inflammasome Biomarkers as Predictors of Diabetic Retinopathy Progression: Evidence from a Pilot Study. Future Pharm. 2023, 3, 612–624. [Google Scholar] [CrossRef]
- Zhu, Q.; Kanneganti, T.-D. Cutting edge: Distinct regulatory mechanisms control proinflammatory cytokines IL-18 and IL-1β. J. Immunol. 2017, 198, 4210–4215. [Google Scholar] [CrossRef] [PubMed]
- Mangan, M.S.; Olhava, E.J.; Roush, W.R.; Seidel, H.M.; Glick, G.D.; Latz, E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat. Rev. Drug Discov. 2018, 17, 588–606. [Google Scholar] [CrossRef] [PubMed]
- Mugisho, O.O.; Green, C.R.; Kho, D.T.; Zhang, J.; Graham, E.S.; Acosta, M.L.; Rupenthal, I.D. The inflammasome pathway is amplified and perpetuated in an autocrine manner through connexin43 hemichannel mediated ATP release. Biochim. Biophys. Acta 2018, 1862, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Dror, E.; Dalmas, E.; Meier, D.T.; Wueest, S.; Thévenet, J.; Thienel, C.; Timper, K.; Nordmann, T.M.; Traub, S.; Schulze, F. Postprandial macrophage-derived IL-1β stimulates insulin, and both synergistically promote glucose disposal and inflammation. Nat. Immunol. 2017, 18, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Vandanmagsar, B.; Youm, Y.-H.; Ravussin, A.; Galgani, J.E.; Stadler, K.; Mynatt, R.L.; Ravussin, E.; Stephens, J.M.; Dixit, V.D. The NLRP3 inflammasome instigates obesity-induced inflammation and insulin resistance. Nat. Med. 2011, 17, 179–188. [Google Scholar] [CrossRef]
- Maedler, K.; Sergeev, P.; Ris, F.; Oberholzer, J.; Joller-Jemelka, H.I.; Spinas, G.A.; Kaiser, N.; Halban, P.A.; Donath, M.Y. Glucose-induced β cell production of IL-1β contributes to glucotoxicity in human pancreatic islets. J. Clin. Investig. 2002, 110, 851–860. [Google Scholar] [CrossRef]
- Kowluru, R.; Odenbach, S. Role of interleukin-1β in the pathogenesis of diabetic retinopathy. Br. J. Ophthalmol. 2004, 88, 1343–1347. [Google Scholar] [CrossRef]
- Vallejo, S.; Palacios, E.; Romacho, T.; Villalobos, L.; Peiró, C.; Sánchez-Ferrer, C.F. The interleukin-1 receptor antagonist anakinra improves endothelial dysfunction in streptozotocin-induced diabetic rats. Cardiovasc. Diabetol. 2014, 13, 158. [Google Scholar] [CrossRef]
- Kowluru, R.A.; Mohammad, G.; Santos, J.M.; Tewari, S.; Zhong, Q. Interleukin-1β and mitochondria damage, and the development of diabetic retinopathy. J. Ocul. Biol. Dis. Infor. 2011, 4, 3–9. [Google Scholar] [CrossRef]
- Liu, Y.; Costa, M.B.; Gerhardinger, C. IL-1β is upregulated in the diabetic retina and retinal vessels: Cell-specific effect of high glucose and IL-1β autostimulation. PLoS ONE 2012, 7, e36949. [Google Scholar] [CrossRef] [PubMed]
- Mesquida, M.; Drawnel, F.; Fauser, S. The role of inflammation in diabetic eye disease. Semin. Immunopathol. 2019, 41, 427–445. [Google Scholar] [CrossRef]
- Vincent, J.A.; Mohr, S. Inhibition of caspase-1/interleukin-1β signaling prevents degeneration of retinal capillaries in diabetes and galactosemia. Diabetes 2007, 56, 224–230. [Google Scholar] [CrossRef]
- Keyel, P.A. How is inflammation initiated? Individual influences of IL-1, IL-18 and HMGB1. Cytokine 2014, 69, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Thomas, H.; Jäger, M.; Mauel, K.; Brandau, S.; Lask, S.; Flohé, S.B. Interaction with mesenchymal stem cells provokes natural killer cells for enhanced IL-12/IL-18-induced interferon-gamma secretion. Mediat. Inflamm. 2014, 2014, 143463. [Google Scholar] [CrossRef] [PubMed]
- Kandikattu, H.K.; Venkateshaiah, S.U.; Mishra, A. Synergy of Interleukin (IL)-5 and IL-18 in eosinophil mediated pathogenesis of allergic diseases. Cytokine Growth Factor Rev. 2019, 47, 83–98. [Google Scholar] [CrossRef]
- Hirano, Y.; Yasuma, T.; Mizutani, T.; Fowler, B.J.; Tarallo, V.; Yasuma, R.; Kim, Y.; Bastos-Carvalho, A.; Kerur, N.; Gelfand, B.D. IL-18 is not therapeutic for neovascular age-related macular degeneration. Nat. Med. 2014, 20, 1372–1375. [Google Scholar] [CrossRef]
- Doyle, S.L.; Campbell, M.; Ozaki, E.; Salomon, R.G.; Mori, A.; Kenna, P.F.; Farrar, G.J.; Kiang, A.-S.; Humphries, M.M.; Lavelle, E.C. NLRP3 has a protective role in age-related macular degeneration through the induction of IL-18 by drusen components. Nat. Med. 2012, 18, 791–798. [Google Scholar] [CrossRef]
- Doyle, S.L.; López, F.J.; Celkova, L.; Brennan, K.; Mulfaul, K.; Ozaki, E.; Kenna, P.F.; Kurali, E.; Hudson, N.; Doggett, T. IL-18 immunotherapy for neovascular AMD: Tolerability and efficacy in nonhuman primates. Investig. Ophthalmol. Vis. Sci. 2015, 56, 5424–5430. [Google Scholar] [CrossRef] [PubMed]
- Doyle, S.L.; Ozaki, E.; Brennan, K.; Humphries, M.M.; Mulfaul, K.; Keaney, J.; Kenna, P.F.; Maminishkis, A.; Kiang, A.-S.; Saunders, S.P. IL-18 attenuates experimental choroidal neovascularization as a potential therapy for wet age-related macular degeneration. Sci. Transl. Med. 2014, 6, 230ra244. [Google Scholar] [CrossRef] [PubMed]
- Satoh, M.; Tabuchi, T.; Itoh, T.; Nakamura, M. NLRP3 inflammasome activation in coronary artery disease: Results from prospective and randomized study of treatment with atorvastatin or rosuvastatin. Clin. Sci. 2014, 126, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Noonan, J.E.; Jenkins, A.J.; Ma, J.-X.; Keech, A.C.; Wang, J.J.; Lamoureux, E.L. An update on the molecular actions of fenofibrate and its clinical effects on diabetic retinopathy and other microvascular end points in patients with diabetes. Diabetes 2013, 62, 3968–3975. [Google Scholar] [CrossRef]
- Inoue, K.; Kataoka, S.Y.; Kawano, S.; Furukawa, T.A.; Lois, N.; Watanabe, N. Fenofibrate for diabetic retinopathy. Cochrane Database Syst. Rev. 2019, 16, 90. [Google Scholar] [CrossRef]
- Abcouwer, S.F. Direct effects of PPARα agonists on retinal inflammation and angiogenesis may explain how fenofibrate lowers risk of severe proliferative diabetic retinopathy. Diabetes 2013, 62, 36. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Hua, H.; Ji, Y.; Jia, Z.; Peng, M.; Huang, S. Anti-inflammatory role of fenofibrate in treating diseases. Biomol. Biomed. 2023, 23, 376. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Shao, Z.; Fu, Z.; Edin, M.L.; Sun, Y.; Liegl, R.G.; Wang, Z.; Liu, C.-H.; Burnim, S.B.; Meng, S.S. Fenofibrate inhibits cytochrome P450 epoxygenase 2C activity to suppress pathological ocular angiogenesis. EBioMedicine 2016, 13, 201–211. [Google Scholar] [CrossRef] [PubMed]
- LaMoia, T.E.; Shulman, G.I. Cellular and molecular mechanisms of metformin action. Endocr. Rev. 2021, 42, 77–96. [Google Scholar] [CrossRef]
- Shao, Y.; Wang, M.; Zhu, Y.; Li, X.; Liu, J. Association of metformin treatment with enhanced effect of anti-VEGF agents in diabetic macular edema patients. Acta Diabetol. 2022, 59, 553–559. [Google Scholar] [CrossRef]
- Yang, F.; Qin, Y.; Wang, Y.; Meng, S.; Xian, H.; Che, H.; Lv, J.; Li, Y.; Yu, Y.; Bai, Y. Metformin inhibits the NLRP3 inflammasome via AMPK/mTOR-dependent effects in diabetic cardiomyopathy. Int. J. Biol. Sci. 2019, 15, 1010. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, G.; Hashimoto-Hachiya, A.; Yen, V.H.; Takemura, M.; Yumine, A.; Furue, K.; Furue, M.; Nakahara, T. Metformin inhibits IL-1β secretion via impairment of NLRP3 inflammasome in keratinocytes: Implications for preventing the development of psoriasis. Cell Death Discov. 2020, 6, 11. [Google Scholar] [CrossRef] [PubMed]
- Xian, H.; Liu, Y.; Nilsson, A.R.; Gatchalian, R.; Crother, T.R.; Tourtellotte, W.G.; Zhang, Y.; Aleman-Muench, G.R.; Lewis, G.; Chen, W. Metformin inhibition of mitochondrial ATP and DNA synthesis abrogates NLRP3 inflammasome activation and pulmonary inflammation. Immunity 2021, 54, 1463–1477.e11. [Google Scholar] [CrossRef] [PubMed]
- Dang, K.-R.; Wu, T.; Hui, Y.-N.; Du, H.-J. Newly-found functions of metformin for the prevention and treatment of age-related macular degeneration. Int. J. Ophthalmol. 2021, 14, 1274. [Google Scholar] [CrossRef]
- Chen, W.; Zhao, M.; Zhao, S.; Lu, Q.; Ni, L.; Zou, C.; Lu, L.; Xu, X.; Guan, H.; Zheng, Z. Activation of the TXNIP/NLRP3 inflammasome pathway contributes to inflammation in diabetic retinopathy: A novel inhibitory effect of minocycline. Inflamm. Res. 2017, 66, 157–166. [Google Scholar] [CrossRef]
| Administration Route | Drug | Mechanism of Action | Phases | Study Status | Clinical Trial Registration | Sponsor |
|---|---|---|---|---|---|---|
| Intravitreal injection | OCU200 | Fusion of human transferrin and human tumstatin | 1 | Not yet recruiting | NCT05802329 | Ocugen (Malvern, PA, USA) |
| IBI324 | Anti-VEGF-A and anti-Angiotensin-2 bispecific antibody | 1 | Not yet recruiting | NCT05489718 | Innovent Biologics (Suzhou) Co., Ltd. (Suzhou, China) | |
| Foselutoclax (UBX1325) | Potent small-molecule inhibitor of Bcl-xL, member of the Bcl-2 family of apoptosis-regulating proteins | 2 | Recruiting | NCT06011798 | Unity Biotechnology, Inc. (South San Francisco, CA, USA) | |
| EYE103 (Restoret™) | Wnt signaling pathway agonist | 1|2 | Recruiting | NCT05919693 | EyeBiotech Ltd. (London, UK) | |
| Infliximab (Remicade™) | TNF-α inhibitor | 1|2 | Unknown | NCT00959725 | Icahn School of Medicine at Mount Sinai (New York, NY, USA) | |
| iCo-007 | Anti-sense drug targeting c-Raf Kinase | 2 | Terminated due to disease progression and vision reduction | NCT01565148 | Johns Hopkins University (Baltimore, MD, USA) | |
| REGN910-3 (Nesvacumab) | Immunoglobulin G1 (IgG1) monoclonal antibody targeting Ang2 | 2 | Completed | NCT02712008 | Regeneron Pharmaceuticals (Tarrytown, NY, USA) | |
| Ocriplasmin (also known as microplasmin) | Truncated recombinant form of human plasmin, used to treat vitreomacular traction with or without full-thickness macular hole | 2 | Completed | NCT00412451 | ThromboGenics (Louvain, Belgium) | |
| Risuteganib (Luminate®®) | Integrin inhibitor | 2 | Completed | NCT02348918 | Allegro Ophthalmics, LLC (San Juan Capistrano, CA, USA) | |
| GB-102 (sunitinib malate) | Multiple intracellular tyrosine kinases inhibitor | 2 | Completed | NCT04085341 | Graybug Vision (La Jolla, CA, USA) | |
| KVD001 | Targets plasma kallikrein | 2 | Completed | NCT03466099 | KalVista Pharmaceuticals, Ltd. (Cambridge, MA, USA) | |
| BI 765128 | Undisclosed | 1|2 | Completed | NCT04919499 | Boehringer Ingelheim (Ingelheim, Germany) | |
| Subcutaneous injection | AKB-9778 | Activator or Tie-2, a receptor tyrosine kinase (RTK) | 2 | Completed | NCT03197870 | EyePoint Pharmaceuticals, Inc. (Watertown, MA, USA) |
| Periocular injection | AIV007 | Broad-spectrum tyrosine kinase inhibitor | 1 | Recruiting | NCT05698329 | AiViva BioPharma, Inc. (Costa Mesa, CA, USA) |
| subconjunctival injection | Sirolimus | Inhibition of rapamycin (mTOR), protein kinase that specifically governs cell growth, replication, and viability | 1|2 | Completed | NCT00711490 | National Eye Institute (NEI) (Bethesda, MD, USA) |
| Oral | MS-553 | Selective PKC-β inhibitor | 1 | Recruiting | NCT04187443 | MingSight Pharmaceuticals Co., Ltd. (San Diego, CA, USA) |
| Tonabersat | Connexin43 hemichannel blocker, upstream inhibitor of the NLRP3 inflammasome pathway | 2 | Recruiting | NCT05727891 | Jaeb Center for Health Research (Tampa, FL, USA) | |
| RZ402 | Small-molecule selective and potent plasma kallikrein inhibitor | 2 | Recruiting | NCT05712720, NCT04527107 | Rezolute (Redwood City, CA, USA) | |
| CU06-1004 (also known as Sac-1004) | Enhances endothelial cell survival and prevents endothelial barrier disruption; inhibits ICAM-1 and VCAM-1 expression by inhibiting NF-κB activation | 2 | Active, not recruiting | NCT05573100 | Curacle Co., Ltd. (Seongnam-si, Republic of Korea) | |
| Ruboxistaurin | Active β-selective Phosphate Kinase C inhibitor | 3 | Completed | NCT00266695 | Chromaderm, Inc. (Cambridge, MA, USA) | |
| Completed | NCT00604383 | |||||
| Completed | NCT00090519 | |||||
| Completed | NCT00133952 | |||||
| PF-04634817 | Chemokine receptor (CCR2/5) antagonist | 2 | Terminated due to changes in drug development prioritization | NCT01994291 | Pfizer (New York, NY, USA) | |
| BI 1467335 (formerly PXS-4728A) | Inhibits neutrophil tethering and rolling, reduces inflammation | 2 | Completed | NCT03238963 | Boehringer Ingelheim (Ingelheim, Germany) | |
| Minocycline | Antibiotic binding to the bacterial 30S ribosomal subunit and inhibiting protein synthesis | 1|2 | Completed | NCT01120899 | National Eye Institute (NEI) (Bethesda, MD, USA) | |
| Calcium dobesilate | Reduces capillary permeability, inhibits platelet aggression and blood viscosity, inhibits apoptosis of vascular endothelial cells, inhibits the expression of inflammatory and upstream VEGF regulator, ICAM-1, and protects against reactive oxygen species | 4 | Unknown | NCT04283162 | Zhongda Hospital (Nanjing, China) | |
| Fenofibrate | Activates PPARα | 3 | Recruiting | NCT04661358 | Jaeb Center for Health Research (Tampa, FL, USA) | |
| YD312 (imatinib) | Inhibits excessive vascular angiogenesis observed in oxygen-induced retinopathy | 2 | Unknown | NCT03635814 | YD Global Life Science Co., Ltd. (Seongnam, Republic of Korea) | |
| Finerenone (BAY94-8862) | Selective mineralocorticoid receptor antagonist | NA | Completed | NCT04477707, NCT04795726 | Bayer (Leverkusen, Germany) | |
| Intravenous injection | Methotrexate | Inhibits production of nucleic acid synthesis by inhibiting the enzyme dihydrofolate reductase | NA | Terminated as Lucentis to be an effective treatment | NCT00779142 | Wake Forest University (Winston-Salem, NC, USA) |
| Ophthalmic solution (eyedrop) | OTT166 | Small-molecule arginylglycylaspartic acid integrin inhibitor | 2 | Completed | NCT05409235 | OcuTerra Therapeutics, Inc. (Boston, MA, USA) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuo, C.Y.J.; Rupenthal, I.D.; Murphy, R.; Mugisho, O.O. Future Therapeutics: Targeting the NLRP3 Inflammasome Pathway to Manage Diabetic Retinopathy Development and Progression. Int. J. Transl. Med. 2024, 4, 402-418. https://doi.org/10.3390/ijtm4030027
Kuo CYJ, Rupenthal ID, Murphy R, Mugisho OO. Future Therapeutics: Targeting the NLRP3 Inflammasome Pathway to Manage Diabetic Retinopathy Development and Progression. International Journal of Translational Medicine. 2024; 4(3):402-418. https://doi.org/10.3390/ijtm4030027
Chicago/Turabian StyleKuo, Charisse Y. J., Ilva D. Rupenthal, Rinki Murphy, and Odunayo O. Mugisho. 2024. "Future Therapeutics: Targeting the NLRP3 Inflammasome Pathway to Manage Diabetic Retinopathy Development and Progression" International Journal of Translational Medicine 4, no. 3: 402-418. https://doi.org/10.3390/ijtm4030027
APA StyleKuo, C. Y. J., Rupenthal, I. D., Murphy, R., & Mugisho, O. O. (2024). Future Therapeutics: Targeting the NLRP3 Inflammasome Pathway to Manage Diabetic Retinopathy Development and Progression. International Journal of Translational Medicine, 4(3), 402-418. https://doi.org/10.3390/ijtm4030027







