Abstract
Arterial dysfunction is major risk factor for cardiovascular complications, and arterial stiffness is an independent risk factor in end-stage renal disease patients. As the distance from the heart increases, arterial stiffness (pulse wave velocity) becomes progressively more marked. This generates a centrifugal stiffness gradient, which leads to partial, continuous local wave reflections, which in turn attenuate the transmission of pulsatile pressure into the microcirculation, thus limiting the potentially deleterious outcomes both upstream (on the heart: left-ventricular hypertrophy and coronary perfusion) and downstream (on the renal and cerebral microcirculation: reduced glomerular filtration and impaired cognitive functions). The impact of arterial aging is greater on the aorta and central capacitive arteries, and it is characterized by a loss or reversal of the physiological stiffness gradient between central and peripheral arteries. Recently, however, in contrast to observations on the aorta, several studies have shown less pronounced, absent, or even negative associations between muscular peripheral arteries and age–stiffness relationships, which may be associated with a decrease in or reversal of the stiffness gradient. These findings point to a potential benefit of assessing the muscular peripheral arteries to predict the risk of cardiovascular disease and suggest that reversal of the stiffness gradient may be an independent risk factor for all-cause mortality.
1. Introduction
Arterial aging is a major risk factor for cardiovascular morbidity and mortality [1]. The main age-related changes that occur within the vascular system are arterial stiffening and endothelial dysfunction [1,2,3,4]. Arterial stiffness is typically assessed from measurements of pulse wave velocity (PWV) in different arterial segments [4,5]. Epidemiological studies have highlighted the role of carotid–femoral (cf) (aortic) PWV measurements in determining the risk of all-cause and cardiovascular mortality [6,7,8,9,10]. The morphometry and biostructure of the different arterial segments are heterogeneous with different age-related consequences [4,11]. In younger populations, arterial stiffening increases progressively from the ascending aorta to the muscular peripheral conduit arteries. There is, therefore, an arterial stiffness gradient [12,13,14,15,16] that exerts a marked effect on blood flow, pressure wave propagation and reflection along the arterial tree, and ultimately, the degree of pulsatile pressure reaching the microcirculation [17,18,19]. Arterial aging is characterized by a steeper increase in aortic stiffness—the main factor influencing the stiffness gradient—with a subsequent reduction in or reversal of the stiffness gradient [12,14,15,16,20,21,22,23,24,25,26]. One study showed that the arterial stiffness gradient is a more reliable predictor of cardiovascular mortality than cfPWV alone in dialysis patients [25], although these findings were not replicated in a community-based sample [23]. However, another study comparing diabetic and nondiabetic individuals reported that the age-related decline in the stiffness gradient was observed with no significant age-related increases in cfPWV [26]. These observations suggest a potential direct pathophysiological role for peripheral arteries in reducing or reversing the physiological stiffness gradient in certain clinical conditions. In the study by Fortier et al., 43% of the study population of dialysis patients were diabetic [25]. Their findings led the authors of the present review to hypothesize that uremia and diabetes may each play specific roles in the process of arterial aging in ESRD populations.
In this review, we compare the age–stiffness relationships of central elastic arteries on one hand and peripheral muscular arteries on the other, in both end-stage renal disease (ESRD) patients and control populations. Assessments are based on data from our arterial stiffness registry data base [6,19,20,22,24,27,28,29,30] with two particular focuses: first, on 938 subjects (412 dialysis patients and 526 controls); second, data from ESRD patients both with and without diabetes. Individuals in the control group had normal kidney function (serum creatinine < 110 µmol/L, eGFR > 60 mL/min/1.73 m2, and absence of albuminuria or microalbuminuria), and no history of cardiovascular disease. This group also included individuals addressed for diagnosis, and, in some cases, initiation of treatment of essential, uncomplicated hypertension.
2. Arterial Stiffness and How It Affects Arterial Functions
The two main functions of the arterial tree are to deliver a continuous adequate supply of oxygenated blood from the heart to the peripheral tissues and organs (conduit/distribution function), and to serve as a buffer, transforming high-flow pressure oscillations within the aorta into a non-pulsatile, low-pressure capillary flow within the microcirculation (dampening or cushioning function) [13,31]. The conduit function is an essential action of medium-sized distributing arteries; the media of these muscular arteries comprises multiple layers of vascular smooth muscle cells (VSMCs), which give these vessels their elastic properties, i.e., the ability to stretch, contract, and relax, thus modifying their diameter and ability to regulate the distribution of blood. The extent of the cushioning function is contingent on the morphometry and viscoelastic properties of the arteries, i.e., arterial compliance/stiffness. Stiffness determines the ability of the vessel walls to resist deformation (strain) when subjected to pressure (stress); stiffening is, therefore, a representation of the stress–strain relationship. Elastic arteries dilate and stiffen with age. These changes are most marked in the aorta and central arteries. They are the largest vessels in the body and have a high elastin and collagen content in the tunica media and fewer smooth muscle cells. Arterial stiffness is characterized by a steep pressure–volume relationship, and high pulsatile pressure.
The stiffness of the arteries also determines the velocity of propagation of the pressure wave along the arterial tree from the proximal aorta toward the peripheral vessels, i.e., PWV [4,13,31]. Measuring PWV, therefore, provides an assessment of the stiffness of an artery. The stiffness of a hollow structure depends on its morphometry (wall thickness, h; radius, r), the intrinsic elastic properties of the wall (incremental elastic modulus [Einc]), and its density (ρ) as represented by the Moens–Korteweg equation: PWV2 = Einc × h/2r × ρ [13]. As the distance from the heart increases, so does PWV, thus creating an arterial stiffness gradient between the ascending aorta and the muscular peripheral conduit arteries [4,12,13,14,15,16].
PWV is not to be confused with blood flow velocity. While PWV is measured in meters per second, blood flow velocity is expressed in centimeters per second. PWV represents the transmission of energy and, thus, the speed with which the pressure wave propagates along the arterial wall while blood flow velocity corresponds to the displacement of a mass throughout the incompressible blood column. Given that conduit and cushioning functions are closely interrelated and synchronized throughout the cardiac cycle, the difference in propagation speed is physiologically beneficial for left-ventricular work and arterial blood flow [13,31]. With each systolic contraction, the volume of blood ejected from the left ventricle encounters a column of blood within the aorta and the arterial tree. To make room for this newly ejected stroke volume, some of this blood flows directly to the peripheral tissues, and some remains momentarily within the aorta and central arteries, causing the walls to stretch (expand), which increases local blood pressure and tensile stress [13,15,31]. To propel the entire arterial column of blood by relying solely on the ‘thrusting’ forces of blood entering the proximal aorta would require notable cardiac energy expenditure to counteract the inertia of the blood column. However, when the stroke volume enters the blood column in the proximal ascending aorta, it generates a pressure wave that moves downstream, propagating the pressure gradient along the length of the arterial tree. Because PWV increases progressively from the aorta to the distal peripheral arteries, the blood column is rapidly (milliseconds) moved from the heart to the most distant sites of the arterial system [31] during ventricular ejection, and, as this arterial blood moves downstream, it ‘frees up’ space for the stroke volume. At the end of ventricular ejection, the length of the stroke volume (stroke distance) is measured in centimeters, i.e., mean blood velocity in cm/s. If PWV did not largely exceed peak aortic blood flow velocity, there would be a notable risk of generating longitudinal shock waves with the subsequent potential risk of arterial damage [13,31].
The second way to accommodate part of the stroke volume is through distension of the aorta; this increases both aortic pressure and tensile stress. However, the stiffer the arterial walls are, the greater the energy required to ensure aortic distension is; therefore, the greater the demands on the heart are (cardiac work). The pressure–volume relationship is nonlinear; when distension pressure is low, the strain is borne by elastin fibers that are distensible. However, when distension pressure is high, the more rigid collagen fibers come into play, making the arterial wall ‘resistant’ to distension, and limiting aortic blood pooling during ventricular ejection [13,31]. Some of the energy produced by the heart is temporarily stored within the vessel walls to enable distension. In physiological conditions, the elastic fibers give the vessel walls their resilience and the energy required to resume their initial size and shape once the stress has ceased [13]. When the heart rests during diastole, the energy stored enables the aorta to recoil, pumping blood into the peripheral tissues to ensure a continuous flow of blood, and, in so doing, serving as an ‘additional ventricle’ [13,28,31]. However, one major shortcoming of this description of the aorta as the global instrument of compliance is that it fails to take into consideration the heterogeneity of aortic characteristics, i.e., that stiffening is progressively greater along the vessel, from the ascending to the abdominal aorta, that the diameter tapers (decreases) in the same centrifugal direction, and that the different segments of the aorta react differently to hemodynamic and metabolic risk factors [4,11,13,14,15,16,20,22,32]. These attributes can be explained by the embryonic origins of the different aortic segments, with their specific morphometry and VSMC phenotypes [4,11].
Together with morphometric changes (tapering) that can occur, the stiffness gradient determines the strength (impedance gradient) with which the arterial pressure wave, generated in the aorta (forward or incident wave), is transmitted along the arterial tree to the microcirculation. Any change in impedance generates unique local partial reflections of forward pressure waves that travel back to the central aorta (reflected waves) [13,15,18,24,31]. When PWV is low, the backward wave returns to the aorta at end-systole and early diastole, thereby increasing late diastolic pressure and coronary perfusion [13,31]. The sum of the continuous partial reflections—expressed as the systemic reflection coefficient—is in the region of 0.80 to 0.85, meaning that 80–85% of forward arterial pressure is reflected [24]. This limits the transmission of pulsatile pressure waves into the microcirculation and, consequently, ensures the protection of low-resistance peripheral organs such as the kidney and the brain [13,15,16,17,18,19,23]. Mechanisms of autoregulation add to this protective effect, while impaired autoregulation and a less pronounced stiffness gradient favor the transmission of pulsatile pressure [33]. Pressure transmission and its impact on the microcirculation have typically been assessed in terms of forward/centrifugal propagation in humans. However, reflected waves traveling back toward the heart face a reverse impedance gradient, whereby partial re-reflections occur at the site of each impedance mismatch. This re-reflection is said to ‘trap’ part of the systemic reflected wave and reduce the magnitude and impact of the final reflection, i.e., reflected wave (augmentation index) [13,15,18,19,24]. Attenuation of forward/incident pressure propagation affects the transmission of pulsatile pressure to the peripheral microvascular vessels, whereas attenuation of the backward reflected pressure can affect heart function and structure.
Arterial aging or arteriosclerosis (arterial stiffening)—characterized by fibroelastic thickening of the intima, elastolysis, calcification of elastic lamellae, increased collagen content, and apoptosis of VSMC—is a frequent cause of left-ventricular (and atrial) hypertrophy, arrhythmias, and congestive heart failure. Atherosclerosis—the primary pathological outcome of conduit dysfunction and characterized by atheromatous plaques, inflammation, patchy intimal calcification, ischemia, or infarction of downstream tissues—is a frequent cause of ischemic heart disease, stroke, and peripheral artery disease [4,12,14,18,31,32,33].
3. Aging and Arterial Stiffness
The characteristic physiologic, histologic, and molecular changes to aging arteries have been widely described and reviewed elsewhere [2,3,4,14,18,32,34,35,36,37,38,39,40,41]. The vessel wall is subjected to lifelong exposure to biomechanical and biochemical stressors. Biomechanical stress is, therefore, the consequence of a lifetime of continuous cycles of stretching and recoiling, during which shear stress and changes in wall tension lead to biomaterial fatigue, arterial remodeling, and deterioration of the vascular extracellular matrix (fragmentation and loss of elastin, accumulation and crosslinking of collagen, and an increased collagen/elastin ratio) [4,5,14,18,34,36]. VSMCs exhibit a high degree of plasticity, are prone to phenotype switching, and play a key role in arterial remodeling [4,5,18,35,37]. In physiologic conditions, VSMCs have a contractile phenotype that, with time, shifts toward a senescence-associated secretory phenotype. This activates microinflammation and oxidative stress, renews collagen synthesis, and triggers production of metalloproteinases, collagenases, and elastases, which in turn instigates an osteogenic program resulting in arterial wall calcification, as well as a decrease in VSMC division and numbers [34,35,36,37,38,39,40,41].
The age-related changes that come about in both elastic aortic segments and in muscular peripheral arteries are contingent on the specific properties of the arterial wall. Aortic stiffness increases steadily with age [12,13,14,15,16,20,21,22,25]. In younger individuals, stiffening of the aorta is significantly lower than that of peripheral arteries, thus explaining the significant ‘stiffness gradient’. However, with aging, stiffness of the aorta increases, while stiffening in the peripheral arteries remains unchanged or diminishes. This reduces the normal gradient—increasing stiffness as the distance from the heart increases—and leads to a progressively reduced or inversed stiffness gradient and, consequently, potential damage to the microcirculation (decline in kidney function, cognitive dysfunction, and vascular dementia) [15,16,17,18,19,22,23,24,25].
The progression of age-related stiffness is not uniform and varies with different clinical conditions, as well as environmental and genetic factors [4,11,23,25,26]. Certain individuals exhibit ‘accelerated arterial aging’ or ‘early vascular aging’, defined as arterial stiffness that is abnormally high for a given chronological age [42]. This is calculated from the intercept and slope of the age/arterial stiffness (typically cfPWV) correlation. Accelerated arterial aging is observed in several disease states (arterial hypertension, chronic kidney disease/ESRD, diabetes mellitus, and inflammatory diseases), and it is a reliable predictor of cardiovascular complications.
ESRD is probably the most characteristic clinical hallmark of accelerated vascular aging [6,19,20,21,22,25,30]. This is illustrated by the significantly steeper slope (β coefficient) of the non-adjusted age–cfPWV correlation (Figure 1A) and confirmed by multiple stepwise regression (Table 1).
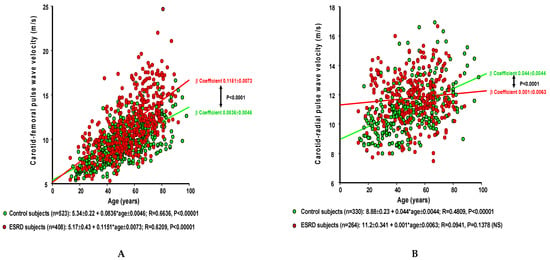
Figure 1.
(A) The age–carotid–femoral pulse wave velocity linear regression in the general population (green circles) and in dialyzed ESRD patients (red circles). (B) The age–carotid–radial pulse wave velocity linear regression in the general population (green circles) and in dialyzed ESRD patients (red circles). *: refer to multiplication sign in the formula.

Table 1.
Multiple regression reports concerning factors associated with pulse wave velocity in central elastic and peripheral muscular arterial segments.
In the control group, the factors documented as significant (after univariate analyses) accounted for 62% of cfPVW variance, compared to only 43% in ESRD patients. The burden of atherosclerotic disease increases in the early stages of chronic kidney disease, and the burden of arteriosclerotic cardiovascular complications increases with progression to ESRD [43]. To date, the majority of the data on aortic stiffness are based on the thoracic aorta, the abdominal aorta, and the femoral artery. However, understanding the role of age-related stiffness along the full length of the arterial tree is a complex undertaking given the heterogeneity of the structure, dimensions, and biomechanical composition of the different segments of the aorta [4,11,34].
While the relationships between aging and cfPWV are well documented, those between age and peripheral muscular conduit arteries are inconsistent [20,26,31]. When we compared data from a control population (normal kidney function and no history of cardiovascular disease), the carotid–radial (cr)PWV and finger–toe PWV were seen to be higher in younger individuals, and to progress more slowly with age (Figure 1B).
In ESRD patients, age–stiffness correlations were inconsistent in muscular arteries, varying between moderate and absent, and were seen to increase [20,31] or decrease with age [25]. In ESRD, crPWV did not increase with age in non-adjusted analyses (Figure 1B). After adjustment in multiple regression analysis, crPWV increased moderately with age, although the rise was significantly less steep than in control subjects (p = 0.0389) (Table 1). The consequence of the different age-related changes to cfPWV and regional PWV is a reduction in or reversal of the stiffness gradient [31]. Certain characteristics are now recognized as having an influence on the age-stiffness relationships. Specifically, one recent study showed that the correlations between age and stiffness on one hand and pressure and stiffness on the other vary between diabetics and nondiabetics [26]. Given that the proportion of diabetics in dialysis populations is on the increase, we studied the same parameters (within the same age range) but while making the distinction between ESRD patients with and those without diabetes (Figure 2).
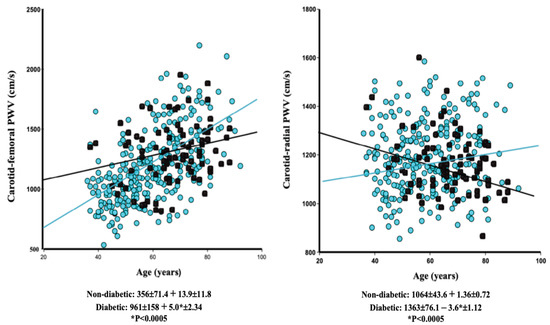
Figure 2.
The age–carotid–femoral pulse wave velocity (left) and the age–carotid–radial pulse wave velocity (right) linear regression in nondiabetic (blue circles) and diabetic (black circles) end-stage renal disease patients on dialysis.
In line with the observations made in the general population of diabetics [26], the non-adjusted correlations in ESRD diabetics and confirmed by multiple regression analysis (Table 2A) were characterized by higher cfPWV in younger individuals and a lower progression of age-related stiffness (Figure 2, left panel). The unadjusted age-related changes in crPWV showed that the main difference was a negative correlation between age and crPWV in diabetics (Figure 2, right panel).

Table 2.
Multiple regression report of factors associated with pulse wave velocity in central elastic and peripheral muscular arterial segments in nondiabetic and diabetic end-stage renal disease patients.
While the differences were only marginally significant in multiple regression analysis (Table 2B), the proportion of diabetics in the study population was relatively low (24%). Fortier et al. reported a negative correlation between age and crPWV in a study of ESRD patients of whom 43% were diabetic [25]. While age-related changes in the stiffness gradient are typically considered a consequence of aortic stiffening, a direct effect of the age-related decrease in the stiffening of muscular arteries cannot be excluded (crPWV). The notable differences in the age–stiffness relationships—and notably the rate of deterioration of vascular stiffness with advancing age—in diabetics and in the general population have led to the suggestion that vascular stiffening is more likely a characteristic of diabetes than of aging [26]. Although the assessment of arterial stiffness from PWV measurements has proven invaluable in improving our understanding of arterial pathophysiology, epidemiology, and clinical outcomes, this technique does have a number of limitations. Most clinical studies have looked at the arterial tree as a whole, from the carotid to the femoral arteries, with cfPWV measurements being used to estimate overall aortic stiffness. However, this approach can no longer be considered appropriate since the various segments are now known to have different properties, different embryologic origins, different sensitivity to risk factors, different progression of the stiffening process, and evolving morphometric properties (e.g., aortic tapering) [4,11,13,14,32]. Given the changing biomechanical characteristics that are observed between the brachial and radial arteries, the same concerns can now be applied to assessments of crPWV [44].
4. Conclusions
The aorta and the arterial tree as a whole are heterogenous structures that are susceptible to the varying degrees of development of arteriosclerosis. As the distance from the heart increases from the ascending aorta to the peripheral arteries, so does the degree of vessel wall stiffening. The ensuing physiological stiffness gradient has a fundamental impact on the cushioning function that transforms high-flow pressure oscillations within the aorta—resulting from cyclic cardiac contractions—into a non-pulsatile, low-pressure capillary flow within the microcirculation. Arterial aging is characterized by a more marked increase in aortic stiffness with a subsequent reduction in or reversal of the stiffness gradient. However, in contrast to observations on the aorta, several studies have shown less notable, absent, or even negative associations between muscular peripheral arteries and age–stiffness relationships, which may also be associated with a decrease in or reversal of the stiffness gradient. While aortic stiffening is recognized as the main factor influencing the stiffness gradient, recent studies—mainly among diabetic and dialysis patients—have shown evidence of reductions in the stiffness gradient even in the absence of notable age-related aortic stiffening, suggesting that reversal of the stiffness gradient may be a risk factor for all-cause mortality that is independent of aortic stiffness. Therefore, while stiffening of the aorta with advancing age remains a reliable indicator of cardiovascular risk, there is emerging evidence of a similar relationship between age and stiffness in the peripheral conduit arteries. Further investigations are required to confirm these observations in wider populations.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors thank Moyra Barbier for editorial assistance.
Conflicts of Interest
The authors declare no conflict of interest.
References
- North, B.J.; Sinclair, D.A. The intersection between aging and cardiovascular disease. Circ. Res. 2012, 110, 1097–1108. [Google Scholar] [CrossRef] [PubMed]
- Donato, A.J.; Machin, D.R.; Lesniewski, L.A. Mechanisms of dysfunction in the aging vasculature and role in age-related diseases. Circ. Res. 2018, 123, 825–848. [Google Scholar] [CrossRef] [PubMed]
- Ungvari, Z.; Tarantini, S.; Donato, A.J.; Galvan, V.; Csiszar, A. Mechanisms of vascular aging. Circ. Res. 2018, 123, 849–867. [Google Scholar] [CrossRef] [PubMed]
- Vatner, S.F.; Zhang, J.; Vyzas, C.; Mishra, K.; Graham, R.M.; Vatner, D.E. Vascular stiffness in aging and disease. Front. Physiol. 2021, 12, 762437. [Google Scholar] [CrossRef] [PubMed]
- The Reference Values for Arterial Stiffness’ Collaboration. Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: ‘Establishing normal and reference values’. Eur. Heart J. 2010, 31, 2338–2350. [Google Scholar] [CrossRef]
- Blacher, J.; Guérin, A.P.; Pannier, B.; Marchais, S.J.; Safar, M.E.; London, G.M. Impact of aortic stiffness on survival in end-stage renal disease. Circulation 1999, 99, 2434–2439. [Google Scholar] [CrossRef]
- Shoji, T.; Emoto, M.; Shinohara, K.; Kakiya, R.; Tsujimoto, Y.; Kishimoto, H.; Ishimura, E.; Tabata, T.; Nishizawa, Y. Diabetes mellitus, aortic stiffness, and cardiovascular mortality in end-stage renal disease. J. Am. Soc. Nephrol. 2001, 12, 2117–2124. [Google Scholar] [CrossRef]
- Laurent, S.; Boutouyrie, P.; Asmar, R.; Gautier, I.; Laloux, B.; Guize, L.; Ducimetiere, P.; Benetos, A. Aortic stiffness is an independent predictor of all-cause and cardiovascular mortality in hypertensive patients. Hypertension 2001, 37, 1236–1241. [Google Scholar] [CrossRef]
- Cruickshank, K.; Riste, L.; Anderson, S.G.; Wright, J.S.; Dunn, G.; Gosling, R.G. Aortic pulse-wave velocity and its relationship to mortality in diabetes and glucose intolerance: An index of vascular function. Circulation 2002, 106, 2085–2090. [Google Scholar] [CrossRef]
- Boutouyrie, P.; Tropeano, A.I.; Asmar, R.; Gautier, I.; Benetos, A.; Lacolley, P.; Laurent, S. Aortic stiffness is an independent predictor of primary coronary events in hypertensive patients: A longitudinal study. Hypertension 2002, 39, 10–15. [Google Scholar] [CrossRef]
- Majesky, M.W. Developmental basis of vascular smooth muscle diversity. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1248–1258. [Google Scholar] [CrossRef]
- Avolio, A.P.; Chen, S.G.; Wang, R.P.; Zhang, C.L.; Li, M.F.; O’Rourke, M.F. Effects of aging on changing arterial compliance and left ventricular load in a northern Chinese urban community. Circulation 1983, 68, 50–58. [Google Scholar] [CrossRef]
- Nichols, W.W.; O’Rourke, M.F. McDonald’s Blood Flow in Arteries: Theoretical, Experimental and Clinical Principles, 5th ed.; Hodder Arnold Publisher: London, UK, 2005; pp. 193–233, 233–267, 299–337. [Google Scholar]
- O’Rourke, M.F. Arterial aging: Pathophysiological principles. Vasc. Med. 2007, 12, 329–341. [Google Scholar] [CrossRef]
- Mitchell, G.F. Effects of central arterial aging on the structure and function of the peripheral vasculature: Implications for end-organ damage. J. Appl. Physiol. 2008, 105, 1652–1660. [Google Scholar] [CrossRef]
- Bortolotto, L.A.; Hanon, O.; Franconi, G.; Boutouyrie, P.; Legrain, S.; Girerd, X. The aging process modifies the distensibility of elastic but not muscular arteries. Hypertension 1999, 34, 889–892. [Google Scholar] [CrossRef]
- Mitchell, G.F.; van Buchem, M.A.; Sigurdsson, S.; Gotal, J.D.; Jonsdottir, M.K.; Kjartansson, Ó.; Garcia, M.; Aspelund, T.; Harris, T.B.; Gudnason, V.; et al. Arterial stiffness, pressure and flow pulsatility and brain structure and function: The Age, Gene/Environment Susceptibility–Reykjavik study. Brain 2011, 134, 3398–3407. [Google Scholar] [CrossRef]
- Mitchell, G.F. Aortic stiffness, pressure and flow pulsatility, and target organ damage. J. Appl. Physiol. 2018, 125, 1871–1880. [Google Scholar] [CrossRef]
- London, G.M.; Safar, M.E.; Pannier, B. Aortic aging in ESRD: Structural, hemodynamic, and mortality implications. J. Am. Soc. Nephrol. 2016, 27, 1837–1846. [Google Scholar] [CrossRef]
- Pannier, B.; Guérin, A.P.; Marchais, S.J.; Safar, M.E.; London, G.M. Stiffness of capacitive and conduit arteries. Prognostic significance for end-stage renal disease patients. Hypertension 2005, 45, 592–596. [Google Scholar] [CrossRef]
- Kimoto, E.; Shoji, T.; Shinohara, K.; Inaba, M.; Okuno, Y.; Miki, T.; Koyama, H.; Emoto, M.; Nishizawa, Y. Preferential stiffening of central over peripheral arteries in type 2 diabetes. Diabetes 2003, 52, 448–452. [Google Scholar] [CrossRef]
- Safar, M.E.; Asmar, R.; Benetos, A.; Blacher, J.; Boutouyrie, P.; Lacolley, P.; Laurent, S.; London, G.; Pannier, B.; Protogerou, A.; et al. French Study Group on Arterial Stiffness. Interaction between hypertension and arterial stiffness. Hypertension 2018, 72, 796–805. [Google Scholar] [CrossRef] [PubMed]
- Niiranen, T.J.; Kalesan, B.; Larson, M.G.; Hamburg, N.M.; Benjamin, E.J.; Mitchell, G.F.; Vasan, R.S. Aortic-brachial arterial stiffness gradient and cardiovascular risk in the community: The Framingham heart study. Hypertension 2017, 69, 1022–1028. [Google Scholar] [CrossRef] [PubMed]
- London, G.M.; Pannier, B.; Safar, M.E. Arterial stiffness gradient, systemic reflection coefficient, and pulsatile pressure wave transmission in essential hypertension. Hypertension 2019, 74, 1366–1372. [Google Scholar] [CrossRef] [PubMed]
- Fortier, C.; Mac-Way, F.; Desmeules, S.; Marquis, K.; De Serres, S.A.; Lebel, M.; Boutouyrie, P.; Agharazii, M. Aortic-brachial stiffness mismatch and mortality in dialysis population. Hypertension 2015, 65, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Cameron, J.D.; Bulpitt, C.J.; Pinto, E.S.; Rajkumar, C. The aging of elastic and muscular arteries: A comparison of diabetic and nondiabetic subjects. Diabetes Care 2003, 26, 2133–2138. [Google Scholar] [CrossRef]
- London, G.M.; Marchais, S.J.; Safar, M.E.; Genest, A.F.; Guerin, A.P.; Metivier, F.; Chedid, K.; London, A.M. Aortic and large artery compliance in end-stage renal failure. Kidney Int. 1990, 37, 137–142. [Google Scholar] [CrossRef]
- London, G.M.; Guérin, A.P.; Marchais, S.J.; Pannier, B.; Safar, M.E.; Day, M.; Metivier, F. Cardiac and arterial interactions in end-stage renal disease. Kidney Int. 1996, 50, 600–608. [Google Scholar] [CrossRef]
- Guérin, A.P.; Blacher, J.; Pannier, B.; Marchais, S.J.; Safar, M.E.; London, G.M. Impact of aortic stiffness attenuation on survival of patients in end-stage renal disease. Circulation 2001, 103, 987–992. [Google Scholar] [CrossRef]
- London, G.M.; Blacher, J.; Pannier, B.; Guérin, A.P.; Marchais, S.J.; Safar, M.E. Arterial wave reflections and survival in end-stage renal failure. Hypertension 2001, 38, 434–438. [Google Scholar] [CrossRef]
- Briet, M.; Boutouyrie, P.; Laurent, S.; London, G.M. Arterial stiffness and pulse pressure in CKD and ESRD. Kidney Int. 2012, 82, 388–400. [Google Scholar] [CrossRef]
- DeBakey, M.E.; Glaeser, D.H. Patterns of atherosclerosis: Effect of risk factors on recurrence and survival—Analysis of 11,890 cases with more than 25-year follow-up. Am. J. Cardiol. 2000, 85, 1045–1053. [Google Scholar] [CrossRef]
- Bidani, A.K.; Griffin, K.A.; Williamson, G.; Wang, X.; Loutzenhiser, R. Protective importance of the myogenic response in the renal circulation. Hypertension 2009, 54, 393–398. [Google Scholar] [CrossRef]
- Tsamis, A.; Krawiec, J.T.; Vorp, D.A. Elastin and collagen fibre microstructure of the human aorta in ageing and disease: A review. J. R. Soc. Interface 2013, 10, 20121004. [Google Scholar] [CrossRef]
- Ribeiro-Silva, J.C.; Nolasco, P.; Krieger, J.E.; Miyakawa, A.A. Dynamic crosstalk between vascular smooth muscle cells and the aged extracellular matrix. Int. J. Mol. Sci. 2021, 22, 10175. [Google Scholar] [CrossRef]
- Cocciolone, A.J.; Hawes, J.Z.; Staiculescu, M.C.; Johnson, E.O.; Murshed, M.; Wagenseil. J.E. Elastin, arterial mechanics, and cardiovascular disease. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H189–H205. [Google Scholar] [CrossRef]
- Lacolley, P.; Regnault, V.; Segers, P.; Laurent, S. Vascular smooth muscle cells and arterial stiffening: Relevance in development, aging, and disease. Physiol. Rev. 2017, 97, 1555–1617. [Google Scholar] [CrossRef]
- Shao, J.-S.; Cheng, S.-L.; Sadhu, J.; Towler, D.A. Inflammation and the osteogenic regulation of vascular calcification. A review and perspective. Hypertension 2010, 55, 579–592. [Google Scholar] [CrossRef]
- Yasmin; McEniery, C.M.; O’Shaughnessy, K.M.; Harnett, P.; Arshad, A.; Wallace, S.; Maki-Petaja, K.; McDonnell, B.; Ashby, M.J.; Brown, J.; et al. Variation in the human matrix metalloproteinase-9 gene is associated with arterial stiffness in healthy individuals. Arterioscler. Thomb. Vasc. Biol. 2006, 26, 1799–1805. [Google Scholar] [CrossRef]
- Shroff, R.C.; McNair, R.; Figg, N.; Skepper, J.N.; Schurgers, L.; Gupta, A.; Hiorns, M.; Donald, A.E.; Deanfield, J.; Rees, L.; et al. Dialysis accelerates medial vascular calcification in part by triggering smooth muscle cell apoptosis. Circulation 2008, 118, 1748–1757. [Google Scholar] [CrossRef]
- London, G.M.; Guérin, A.P.; Marchais, S.J.; Métivier, F.; Pannier, B.; Adda, H. Arterial media calcification in end-stage renal disease: Impact on all-cause and cardiovascular mortality. Nephrol. Dial. Transplant. 2003, 18, 1731–1740. [Google Scholar] [CrossRef]
- Laurent, S.; Boutouyrie, P.; Cunha, P.G.; Lacolley, P.; Nilsson, P.M. Concept of extremes in vascular aging. From early vascular aging to supernormal vascular aging. Hypertension 2019, 74, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Wanner, C.; Amann, K.; Shoji, T. The heart and vascular system in dialysis. Lancet 2016, 388, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Hayoz, D.; Rutschmann, B.; Perret, F.; Niederberger, M.; Tardy, Y.; Mooser, V.; Nussberger, J.; Waeber, B.; Brunner, H.R. Conduit artery compliance and distensibility are not necessarily reduced in hypertension. Hypertension 1992, 20, 1–6. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).