A Major Facilitator Superfamily Transporter Contributes to Ergot Alkaloid Accumulation but Not Secretion in Aspergillus leporis
Abstract
1. Introduction
2. Materials and Methods
2.1. Heterologous Expression of easT in A. fumigatus Strain LA
2.2. Localization of easT in A. fumigatus
2.3. Knockout of easT in A. leporis
3. Results and Discussion
3.1. Analysis of easT by Heterologous Expression in A. fumigatus
3.2. Localization of the Product of easT Heterologously Expressed in A. fumigatus
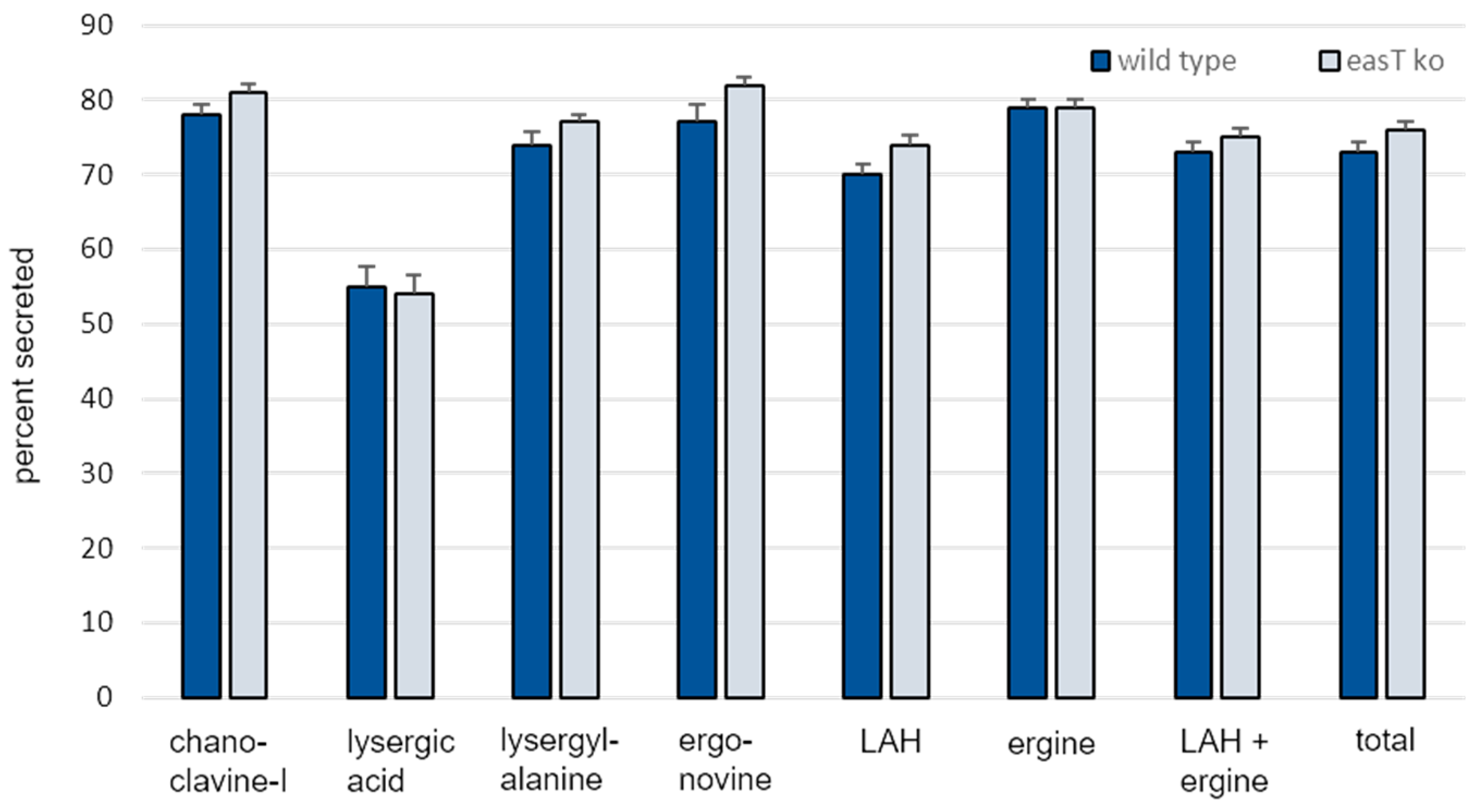
3.3. Analysis of easT by Gene Knockout in A. leporis
3.4. Differences in Ergot Alkaloid Accumulation in Engineered A. fumigatus Compared to A. leporis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Matossian, M.K. Poisons of the Past: Molds, Epidemics, and History; Yale University Press: New Haven, CT, USA, 1989. [Google Scholar]
- Schiff, P. Ergot and its alkaloids. Am. J. Pharm. Educ. 2006, 70, 98. [Google Scholar] [CrossRef]
- Haarmann, T.; Rolke, Y.; Giesbart, S.; Tudzynski, P. Ergot: From witchcraft to biotechnology. Mol. Plant Pathol. 2009, 10, 563–577. [Google Scholar] [CrossRef]
- Tasker, N.R.; Wipf, P. Biosynthesis, total synthesis, and biological profiles of Ergot alkaloids. Alkaloids 2021, 85, 1–112. [Google Scholar]
- Leadmon, C.E.; Sampson, J.K.; Maust, M.D.; Macias, A.M.; Rehner, S.A.; Kasson, M.T.; Panaccione, D.G. Several Metarhizium species produce ergot alkaloids in a condition-specific manner. Appl. Environ. Microbiol. 2020, 86, e00373-20. [Google Scholar] [CrossRef]
- Jones, A.M.; Steen, C.R.; Panaccione, D.G. Independent evolution of a lysergic acid amide in Aspergillus species. Appl. Environ. Microbiol. 2021, 87, e01801-21. [Google Scholar] [CrossRef]
- Mulinti, P.; Allen, N.A.; Gravelat, F.N.; Sheppard, D.C.; Panaccione, D.G. Accumulation of ergot alkaloids during conidiophore development in Aspergillus fumigatus. Curr. Microbiol. 2014, 68, 1–5. [Google Scholar] [CrossRef]
- Davis, K.A.; Sampson, J.K.; Panaccione, D.G. Genetic reprogramming of the ergot alkaloid pathway of Metarhizium brunneum. Appl. Environ. Microbiol. 2020, 86, e01251-20. [Google Scholar] [CrossRef]
- Nielsen, C.A.F.; Folly, C.; Hatsch, A.; Molt, A.; Schröder, H.; O’Connor, S.E.; Naesby, M. The important ergot alkaloid intermediate chanoclavine-I produced in the yeast Saccharomyces cerevisiae by the combined action of EasC and EasE from Aspergillus japonicus. Microb. Cell Factor. 2014, 13, 95. [Google Scholar] [CrossRef]
- Wu, N.; Yao, M.; Xiao, W.; Dong, T.; Ma, H.; Du, X.; Wang, Y.; Yuan, Y. Modular pathway compartmentalization for agroclavine overproduction in Saccharomyces cerevisiae. ACS Synth. Biol. 2023, 12, 1133–1145. [Google Scholar] [CrossRef]
- Chanda, A.; Roze, L.V.; Kang, S.; Artymovich, K.A.; Hicks, G.R.; Raikhel, N.V.; Calvo, A.M.; Linz, J.E. A key role for vesicles in fungal secondary metabolism. Proc. Natl. Acad. Sci. USA 2009, 106, 19533–19538. [Google Scholar] [CrossRef]
- Menke, J.; Weber, J.; Broz, K.; Kistler, H.C. Cellular development associated with induced mycotoxin synthesis in the filamentous fungus Fusarium graminearum. PLoS ONE 2013, 8, e63077. [Google Scholar] [CrossRef]
- Boenisch, M.J.; Broz, K.L.; Purvine, S.O.; Chrisler, W.B.; Nicora, C.D.; Connolly, L.R.; Freitag, M.; Baker, S.E.; Kistler, H.C. Structural reorganization of the fungal endoplasmic reticulum upon induction of mycotoxin biosynthesis. Sci. Rep. 2017, 7, 44296. [Google Scholar] [CrossRef] [PubMed]
- Kistler, H.C.; Broz, K. Cellular compartmentalization of secondary metabolism. Front. Microbiol. 2015, 6, 68. [Google Scholar] [CrossRef] [PubMed]
- Arnold, S.L.; Panaccione, D.G. Biosynthesis of the pharmaceutically important fungal ergot alkaloid dihydrolysergic acid requires a specialized allele of cloA. Appl. Environ. Microbiol. 2017, 83, e00805-17. [Google Scholar] [CrossRef]
- States, J.S.; Christensen, M. Aspergillus leporis, a new species related to A. flavus. Mycologia 1966, 58, 738–742. [Google Scholar] [CrossRef]
- Wangeline, A.L.; Valdez, J.R.; Lindblom, S.D.; Bowling, K.L.; Reeves, F.B.; Pilon-Smits, E.A. Characterization of rhizosphere fungi from selenium hyperaccumulator and nonhyperaccumulator plants along the eastern Rocky Mountain Front Range. Am. J. Bot. 2011, 98, 1139–1147. [Google Scholar] [CrossRef]
- Jones, A.M.; Panaccione, D.G. Ergot alkaloids contribute to the pathogenic potential of the fungus Aspergillus leporis. Appl. Environ. Microbiol. 2023, 89, e00415-23. [Google Scholar] [CrossRef]
- Beliveau, J.; Ramstad, E. 8-Hydroxylation of agroclavine and elymoclavine by fungi. Lloydia 1967, 29, 234–238. [Google Scholar]
- Coyle, C.M.; Cheng, J.Z.; O’Connor, S.E.; Panaccione, D.G. An old yellow enzyme gene controls the branch point between Aspergillus fumigatus and Claviceps purpurea ergot alkaloid pathways. Appl. Environ. Microbiol. 2010, 76, 3898–3903. [Google Scholar] [CrossRef]
- Robinson, S.L.; Panaccione, D.G. Heterologous expression of lysergic acid and novel ergot alkaloids in Aspergillus fumigatus. Appl. Environ. Microbiol. 2014, 80, 6465–6472. [Google Scholar] [CrossRef]
- Bilovol, Y.; Panaccione, D.G. Functional analysis of the gene controlling hydroxylation of festuclavine in the ergot alkaloid pathway of Neosartorya fumigata. Curr. Genet. 2016, 62, 853–860. [Google Scholar] [CrossRef]
- Silar, P. Two new easy to use vectors for transformations. Fungal Genet. Rep. 1995, 42, 23. [Google Scholar] [CrossRef]
- Strickland, D.; Lin, Y.; Wagner, E.; Hope, C.M.; Zayner, J.; Antoniou, C.; Sosnick, T.R.; Weiss, E.L.; Glotzer, M. TULIPs: Tunable, light-controlled interacting protein tags for cell biology. Nat. Methods 2012, 4, 379–384. [Google Scholar] [CrossRef]
- Ulrich, A.; Andersen, K.R.; Schwartz, T.U. Exponential megapriming PCR (EMP) cloning—Seamless DNA insertion into any target plasmid without sequence constraints. PLoS ONE 2012, 7, e53360. [Google Scholar] [CrossRef]
- Lund, B.A.; Leiros, H.-K.S.; Bjerga, G.E.K. A high-throughput, restriction-free cloning and screening strategy based on ccdB-gene replacement. Microb. Cell Factor. 2014, 13, 38. [Google Scholar] [CrossRef]
- Reddy, V.S.; Shlykov, M.A.; Castillo, R.; Sun, E.I.; Saler, M.H. The major facilitator superfamily (MFS) revisited. FEBS J. 2012, 279, 2022–2035. [Google Scholar] [CrossRef]
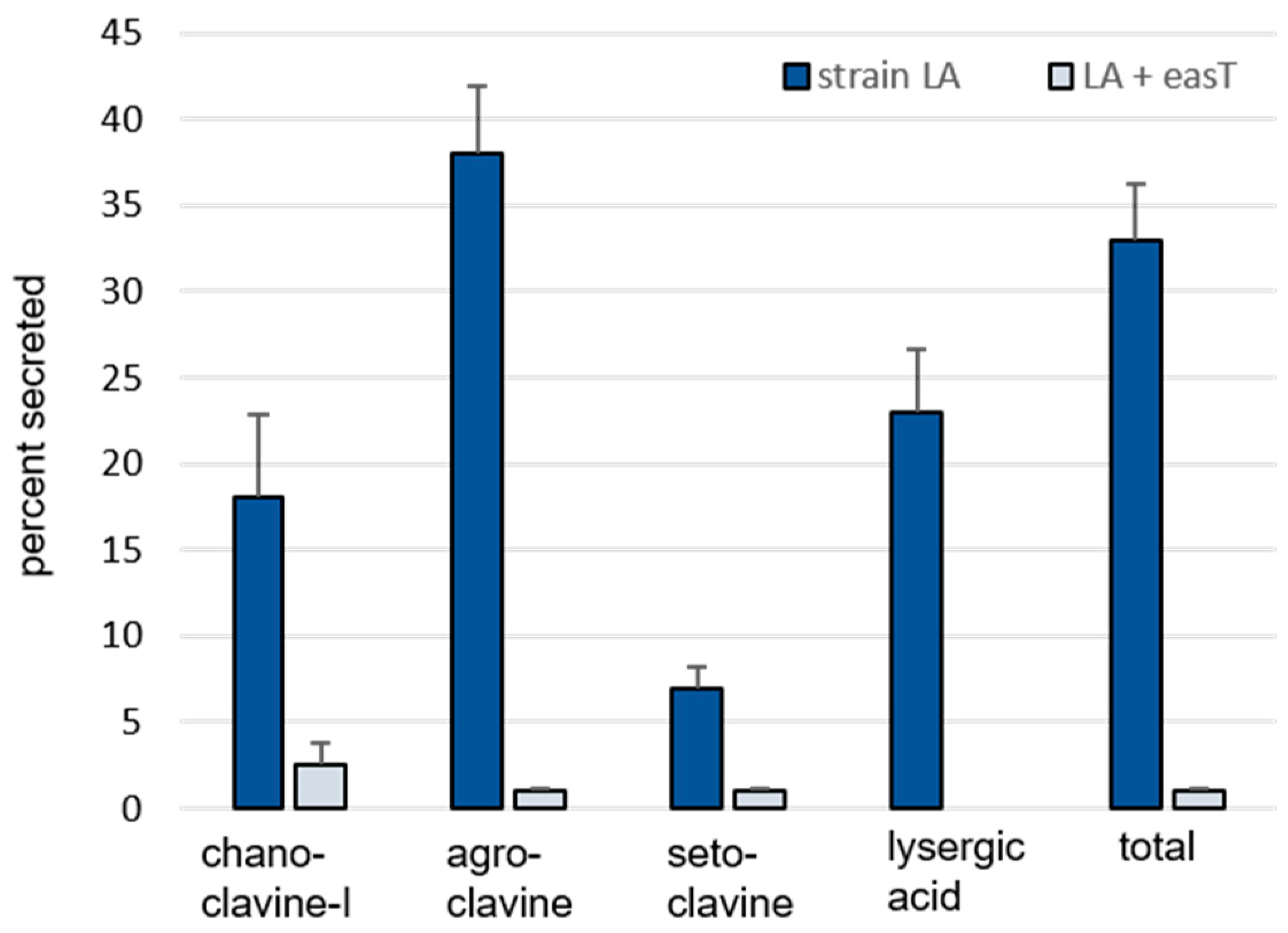

| nmol Alkaloid/g Dry Weight Fungus (Mean ± SE) | ||||
|---|---|---|---|---|
| Solid Phase | Liquid Phase | |||
| Alkaloid | A. fumigatus Strain LA | Strain LA + easT | A. fumigatus Strain LA | Strain LA + easT |
| Chanoclavine-I | 66 ± 9 A 1 | 174 ± 18 B | 14 ± 1 a | 4 ± 0.4 b |
| Agroclavine | 302 ± 51 A | 4891 ± 634 B | 171 ± 11 a | 37 ± 8 b |
| Setoclavine | 37 ± 4 A | 303 ± 43 B | 3 ± 0.3 a | 2 ± 0.2 b |
| Lysergic acid | 19 ± 4 | n.d. 2 | 5 ± 0.5 | n.d. |
| Total | 424 ± 67 A | 5367 ± 690 B | 192 ± 12 a | 43 ± 9 b |
| nmol Alkaloid/g Dry Weight Fungus (Mean ± SE) | ||||
|---|---|---|---|---|
| Solid Phase | Liquid Phase | |||
| Alkaloid | Wild Type | easT Knockout | Wild Type | easT Knockout |
| Chanoclavine-I | 6 ± 0.7 A 1 | 4 ± 0.4 A | 20 ± 2 a | 19 ± 0.7 a |
| Lysergic acid | 2 ± 0.2 A | 2 ± 0.2 A | 3 ± 0.3 a | 2 ± 0.2 a |
| Lysergyl-alanine | 4 ± 0.3 A | 3 ± 0.2 B | 11 ± 0.9 a | 9 ± 0.4 a |
| Ergonovine | 2 ± 0.1 A | 1 ± 0.1 B | 6 ± 0.7 a | 6 ± 0.3 a |
| LAH | 185 ± 17 A | 144 ± 35 B | 427 ± 40 a | 401 ± 21 a |
| Ergine | 61 ± 6 A | 46 ± 4 B | 237 ± 20 a | 174 ± 6 b |
| LAH + ergine | 246 ± 22 A | 189 ± 13 B | 665 ± 55 a | 575 ± 23 a |
| Total | 259 ± 23 A | 200 ± 14 B | 705 ± 59 a | 611 ± 25 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, A.M.; Davis, K.A.; Panaccione, D.G. A Major Facilitator Superfamily Transporter Contributes to Ergot Alkaloid Accumulation but Not Secretion in Aspergillus leporis. Appl. Microbiol. 2024, 4, 406-417. https://doi.org/10.3390/applmicrobiol4010028
Jones AM, Davis KA, Panaccione DG. A Major Facilitator Superfamily Transporter Contributes to Ergot Alkaloid Accumulation but Not Secretion in Aspergillus leporis. Applied Microbiology. 2024; 4(1):406-417. https://doi.org/10.3390/applmicrobiol4010028
Chicago/Turabian StyleJones, Abigail M., Kyle A. Davis, and Daniel G. Panaccione. 2024. "A Major Facilitator Superfamily Transporter Contributes to Ergot Alkaloid Accumulation but Not Secretion in Aspergillus leporis" Applied Microbiology 4, no. 1: 406-417. https://doi.org/10.3390/applmicrobiol4010028
APA StyleJones, A. M., Davis, K. A., & Panaccione, D. G. (2024). A Major Facilitator Superfamily Transporter Contributes to Ergot Alkaloid Accumulation but Not Secretion in Aspergillus leporis. Applied Microbiology, 4(1), 406-417. https://doi.org/10.3390/applmicrobiol4010028







