State of the Art on the Interaction of Entomopathogenic Nematodes and Plant Growth-Promoting Rhizobacteria to Innovate a Sustainable Plant Health Product
Abstract
1. Introduction
2. The Role of Soil Organisms in Sustainable Agriculture
2.1. The Use of EPNs in Biological Control
2.2. The Use of PGPR in Biological Control and Sustainable Agriculture
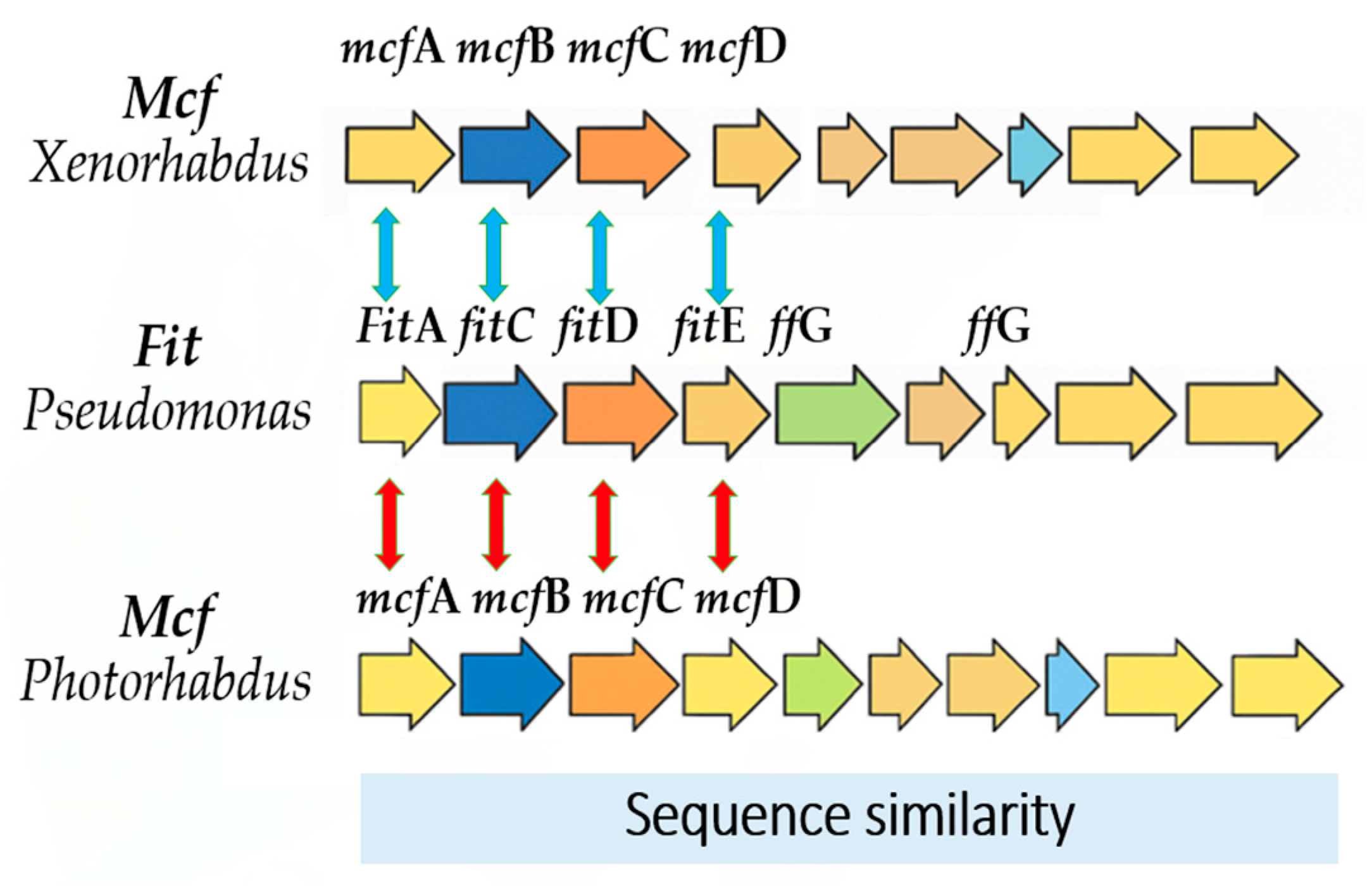
2.3. Shared Genetic Information Between EPNs and PGPR
3. EPNs and Their Microbial Interactions
3.1. Biological Interaction Between EPNs and PGPR
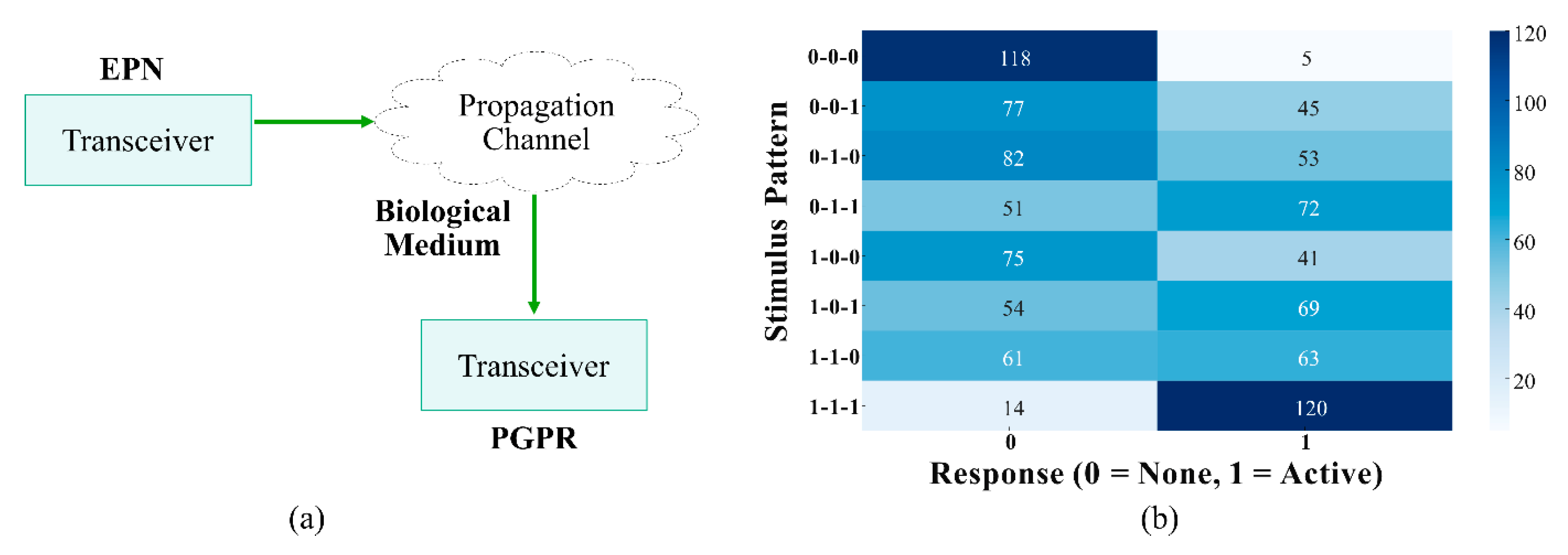
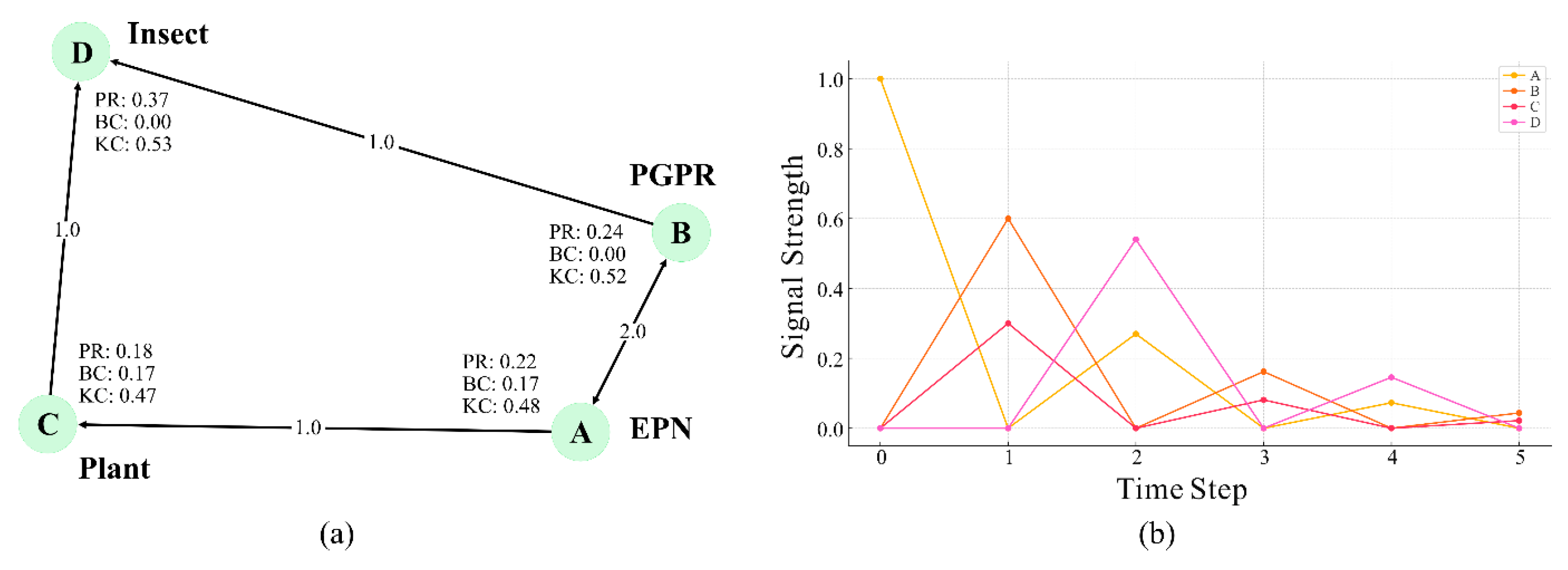
4. Molecular Communication Approaches
5. Alginate-Based Formulations
5.1. Alginate Formulation of EPNs
5.2. Alginate-Based Formulation of PGPR
6. Current Challenges, Research Barriers, and Future Directions
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| EPN | entomopathogenic nematodes |
| PGPR | plant growth-promoting rhizobacteria |
| IJs | infective juveniles |
| fit | fluorescent insecticidal toxin |
| Mcf | makes caterpillars floppy |
| DAPG | 2,4-diacetyl phloroglucinol |
| FAM | Frequently Associated Microbiota |
| ISR | inducing systemic resistance |
| SAR | systemic acquired resistance |
| UV | Ultraviolet |
| HCN | hydrogen cyanide |
| PCBs | polychlorinated biphenyls |
| CFFLs | coherent feed-forward loops |
| UNSDGs | United Nations Sustainable Development Goals |
References
- Lacey, L.A.; Georgis, R. Entomopathogenic nematodes for Control of Insect Pests Above and Below Ground with Comments on Commercial Production. J. Nematol. 2012, 44, 218–225. [Google Scholar]
- Koppenhöfer, A.M.; Shapiro-Ilan, D.I.; Hiltpold, I. Entomopathogenic nematodes in Sustainable Food Production. Front. Sustain. Food Syst. 2020, 4, 125. [Google Scholar] [CrossRef]
- Shapiro-Ilan, D.; Dolinski, C. Entomopathogenic Nematode Application Technology. In Nematode Pathogenesis of Insects and Other Pests: Ecology and Applied Technologies for Sustainable Plant and Crop Protection; Campos-Herrera, R., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 231–254. ISBN 978-3-319-18266-7. [Google Scholar]
- Massa, F.; Defez, R.; Bianco, C. Exploitation of Plant Growth Promoting Bacteria for Sustainable Agriculture: Hierarchical Approach to Link Laboratory and Field Experiments. Microorganisms 2022, 10, 865. [Google Scholar] [CrossRef] [PubMed]
- Vejan, P.; Abdullah, R.; Khadiran, T.; Ismail, S.; Nasrulhaq Boyce, A. Role of Plant Growth Promoting Rhizobacteria in Agricultural Sustainability—A Review. Molecules 2016, 21, 573. [Google Scholar] [CrossRef] [PubMed]
- Lugtenberg, B.; Kamilova, F. Plant-Growth-Promoting Rhizobacteria. Annu. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef]
- Spescha, A.; Weibel, J.; Wyser, L.; Brunner, M.; Hess Hermida, M.; Moix, A.; Scheibler, F.; Guyer, A.; Campos-Herrera, R.; Grabenweger, G.; et al. Combining entomopathogenic Pseudomonas bacteria, nematodes and fungi for biological control of a below-ground insect pest. Agric. Ecosyst. Environ. 2023, 348, 108414. [Google Scholar] [CrossRef]
- Neidig, N.; Paul, R.J.; Scheu, S.; Jousset, A. Secondary Metabolites of Pseudomonas fluorescens CHA0 Drive Complex Non-Trophic Interactions with Bacterivorous Nematodes. Microb. Ecol. 2011, 61, 853–859. [Google Scholar] [CrossRef]
- Zwyssig, M.; Spescha, A.; Patt, T.; Belosevic, A.; Machado, R.A.R.; Regaiolo, A.; Keel, C.; Maurhofer, M. Entomopathogenic pseudomonads can share an insect host with Entomopathogenic nematodes and their mutualistic bacteria. ISME J. 2024, 18, wrae028. [Google Scholar] [CrossRef] [PubMed]
- Ogier, J.-C.; Pagès, S.; Frayssinet, M.; Gaudriault, S. Entomopathogenic nematode-associated microbiota: From monoxenic paradigm to pathobiome. Microbiome 2020, 8, 25. [Google Scholar] [CrossRef]
- Ruiu, L.; Marche, M.G.; Mura, M.E.; Tarasco, E. Involvement of a novel Pseudomonas protegens strain associated with entomopathogenic nematode infective juveniles in insect pathogenesis. Pest Manag. Sci. 2022, 78, 5437–5443. [Google Scholar] [CrossRef]
- Jo Hurley, M. An Investigation on the Interactions Between Entomopathogenic nematodes and Plant Growth Promoting Bacteria. Ph.D. Thesis, South East Technological University, Carlow, Ireland, 2018. [Google Scholar]
- Ruffner, B.; Péchy-Tarr, M.; Höfte, M.; Bloemberg, G.; Grunder, J.; Keel, C.; Maurhofer, M. Evolutionary patchwork of an insecticidal toxin shared between plant-associated pseudomonads and the insect pathogens Photorhabdus and Xenorhabdus. BMC Genom. 2015, 16, 609. [Google Scholar] [CrossRef] [PubMed]
- Farsad, N.; Yilmaz, H.B.; Eckford, A.; Chae, C.-B.; Guo, W. A Comprehensive Survey of Recent Advancements in Molecular Communication. IEEE Commun. Surv. Tutor. 2016, 18, 1887–1919. [Google Scholar] [CrossRef]
- Gaffney, M.; Walsh, L.; Cucak, M.; Fealy, R.; Purvis, G.; Lambkin, K.; Griffin, D.; McNamara, L.; Thorne, F.; Fealy, R.; et al. Establishing a Platform for Integrated Pest Management in Irish Crops. EMS Annu. Meet. Abstr. 2017, 14, 1. Available online: https://meetingorganizer.copernicus.org/EMS2017/posters/25527 (accessed on 25 June 2025).
- Oerke, E.-C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Ofuya, T.I.; Okunlola, A.I.; Mbata, G.N. A Review of Insect Pest Management in Vegetabl e Crop Production in Nigeria. Insects 2023, 14, 111. [Google Scholar] [CrossRef]
- Ouaarous, M.; El Fakhouri, K.; Taarji, N.; Baouchi, A.; Amri, M.; Ramdani, C.; Sobeh, M.; Mesfioui, A.; El Bouhssini, M. Impact of Field Insect Pests on Seed and Nutritional Quality of Some Important Crops: A Comprehensive Review. ACS Omega 2025, 10, 8779–8792. [Google Scholar] [CrossRef]
- Tonnang, H.E.; Sokame, B.M.; Abdel-Rahman, E.M.; Dubois, T. Measuring and modelling crop yield losses due to invasive insect pests under climate change. Curr. Opin. Insect Sci. 2022, 50, 100873. [Google Scholar] [CrossRef]
- Usman, M.; Wakil, W.; Shapiro-Ilan, D.I. Entomopathogenic nematodes as biological control agent against Bactrocera zonata and Bactrocera dorsalis (Diptera: Tephritidae). Biol. Control 2021, 163, 104706. [Google Scholar] [CrossRef]
- FAO. Status of the World’s Soil Resources: Main Report; ITPS: Rome, Italy, 2015; ISBN 978-92-5-109004-6. [Google Scholar]
- FAO. Building Climate Resilience for Food Security and Nutrition; WFP: Rome, Italy, 2018; ISBN 978-92-5-130571-3. [Google Scholar]
- Imperiali, N.; Chiriboga, X.; Schlaeppi, K.; Fesselet, M.; Villacrés, D.; Jaffuel, G.; Bender, S.F.; Dennert, F.; Blanco-Pérez, R.; Van Der Heijden, M.G.A.; et al. Combined Field Inoculations of Pseudomonas Bacteria, Arbuscular Mycorrhizal Fungi, and Entomopathogenic nematodes and their Effects on Wheat Performance. Front. Plant Sci. 2017, 8, 1809. [Google Scholar] [CrossRef] [PubMed]
- Miles, C.; Blethen, C.; Gaugler, R.; Shapiro-Ilan, D.; Murray, T. Using Entomopathogenic nematodes for Crop Insect pest Control; A Pacific Northwest Extension Publications (PNW 544): Washington, DC, USA, 2012; pp. 1–9. Available online: https://www.researchgate.net/profile/Randy-Gaugler/publication/263444652_Using_beneficial_nematodes_for_crop_insect_pest_control/links/0046353ad890787049000000/Using-beneficial-nematodes-for-crop-insect-pest-control.pdf (accessed on 25 June 2025).
- Lacey, L.A.; Grzywacz, D.; Shapiro-Ilan, D.I.; Frutos, R.; Brownbridge, M.; Goettel, M.S. Insect pathogens as biological control agents: Back to the future. J. Invertebr. Pathol. 2015, 132, 1–41. [Google Scholar] [CrossRef] [PubMed]
- Platt, T.; Stokwe, N.F.; Malan, A.P. A Review of the Potential Use of Entomopathogenic nematodes to Control Above-Ground Insect Pests in South Africa. South Afr. J. Enol. Vitic. 2020, 41, 1–16. [Google Scholar] [CrossRef]
- Lewis, E.; Campbell, J.; Griffin, C.; Kaya, H.; Peters, A. Behavioral ecology of Entomopathogenic nematodes. Biol. Control 2006, 38, 66–79. [Google Scholar] [CrossRef]
- Devi, G. Management of Cutworm by Entomopathogenic nematodes—A Review. Int. J. Curr. Microbiol. Appl. Sci. 2020, 9, 2520–2526. [Google Scholar] [CrossRef]
- Hough-Goldstein, J.; Janis, J.A.; Ellers, C.D. Release Methods for Perillus bioculatus (F.), a Predator of the Colorado Potato Beetle. Biol. Control 1996, 6, 114–122. [Google Scholar] [CrossRef]
- Kapranas, A.; Sbaiti, I.; Degen, T.; Turlings, T.C.J. Biological control of cabbage fly Delia radicum with Entomopathogenic nematodes: Selecting the most effective nematode species and testing a novel application method. Biol. Control 2020, 144, 104212. [Google Scholar] [CrossRef]
- Palumbo, J.C.; Kerns, D.L.; Engle, C.E.; Sanchez, C.A.; Wilcox, M. lmidacloprid Formulation and Soil Placement Effects on Colonization by Sweetpotato whitefly (Homoptera: Aleyrodidae): Head Size and Incidence of Chlorosis in Lettuce. J. Econ. Entomol. 1996, 89, 735–742. [Google Scholar] [CrossRef]
- Beck, B.; Spanoghe, P.; Moens, M.; Brusselman, E.; Temmerman, F.; Pollet, S.; Nuyttens, D. Improving the biocontrol potential of Steinernema feltiae against Delia radicum through dosage, application technique and timing. Pest Manag. Sci. 2014, 70, 841–851. [Google Scholar] [CrossRef]
- Triapitsyn, S.V. A key to the Mymaridae (Hymenoptera) egg parasitoids of Proconiine sharpshooters (Hemiptera: Cicadellidae) in the Nearctic region, with description of two new species of Gonatocerus. Zootaxa 2006, 1203, 1–38. [Google Scholar] [CrossRef]
- Lankin, G.; Castaneda-Alvarez, C.; Vidal-Retes, G.; Aballay, E. Biological control of the potato cutworm Agrotis deprivata (Lepidoptera: Noctuidae) with Steinernema feltiae LR (Nematoda: Steinernematidae): Influence of the temperature, host developmental stage, and application mode on its survival and infectivity. Biol. Control 2020, 144, 104219. [Google Scholar] [CrossRef]
- Hoarau, C.; Campbell, H.; Prince, G.; Chandler, D.; Pope, T. Biological control agents against the cabbage stem flea beetle in oilseed rape crops. Biol. Control 2022, 167, 104844. [Google Scholar] [CrossRef]
- Hokkanen, H.M.T. Biological control methods of pest insects in oilseed rape. EPPO Bull. 2008, 38, 104–109. [Google Scholar] [CrossRef]
- Shapiro-Ilan, D.; Hazir, S.; Glazer, I. Chapter 6—Basic and Applied Research: Entomopathogenic nematodes. In Microbial Control of Insect and Mite Pests; Lacey, L.A., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 91–105. ISBN 978-0-12-803527-6. [Google Scholar]
- Srce, S. Računski Centar—Effectiveness of Entomopathogenic nematodes in the Control of Oilseed Rape Pests. Available online: https://www.croris.hr/crosbi/publikacija/prilog-skup/860354?utm_source=chatgpt.com (accessed on 25 June 2025).
- Xu, C.; De Clercq, P.; Moens, M.; Chen, S.; Han, R. Efficacy of Entomopathogenic nematodes (Rhabditida: Steinernematidae and Heterorhabditidae) against the striped flea beetle, Phyllotreta striolata. BioControl 2010, 55, 789–797. [Google Scholar] [CrossRef]
- Kim, W.-I.; Cho, W.K.; Kim, S.-N.; Chu, H.; Ryu, K.-Y.; Yun, J.-C.; Park, C.-S. Genetic Diversity of Cultivable Plant Growth-Promoting Rhizobacteria in Korea. J. Microbiol. Biotechnol. 2011, 21, 777–790. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, P.N.; Jha, D.K. Plant growth-promoting rhizobacteria (PGPR): Emergence in agriculture. World J. Microbiol. Biotechnol. 2012, 28, 1327–1350. [Google Scholar] [CrossRef] [PubMed]
- Jalmi, S.K.; Sinha, A.K. Ambiguities of PGPR-Induced Plant Signaling and Stress Management. Front. Microbiol. 2022, 13, 899563. [Google Scholar] [CrossRef]
- Kumari, R.; Pandey, E.; Bushra, S.; Faizan, S.; Pandey, S. Plant Growth Promoting Rhizobacteria (PGPR) induced protection: A plant immunity perspective. Physiol. Plant. 2024, 176, e14495. [Google Scholar] [CrossRef]
- Afzal, I.; Shinwari, Z.K.; Sikandar, S.; Shahzad, S. Plant beneficial endophytic bacteria: Mechanisms, diversity, host range and genetic determinants. Microbiol. Res. 2019, 221, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Ahirwar, N.K.; Gupta, G.; Singh, V.; Rawlley, R.K.; Ramana, S. Influence on growth and fruit yield of tomato (Lycopersicon esculentum Mill.) plants by inoculation with Pseudomonas fluorescence (SS5): Possible role of plant growth promotion. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 720–730. [Google Scholar]
- Andreolli, M.; Zapparoli, G.; Angelini, E.; Lucchetta, G.; Lampis, S.; Vallini, G. Pseudomonas protegens MP12: A plant growth-promoting endophytic bacterium with broad-spectrum antifungal activity against grapevine phytopathogens. Microbiol. Res. 2019, 219, 123–131. [Google Scholar] [CrossRef]
- Hol, W.H.G.; Bezemer, T.M.; Biere, A. Getting the ecology into interactions between plants and the plant growth-promoting bacterium Pseudomonas fluorescens. Front. Plant Sci. 2013, 4, 81. [Google Scholar] [CrossRef] [PubMed]
- Mhatre, P.H.; Karthik, C.; Kadirvelu, K.; Divya, K.L.; Venkatasalam, E.P.; Srinivasan, S.; Ramkumar, G.; Saranya, C.; Shanmuganathan, R. Plant growth promoting rhizobacteria (PGPR): A potential alternative tool for nematodes bio-control. Biocatal. Agric. Biotechnol. 2019, 17, 119–128. [Google Scholar] [CrossRef]
- Pereira, R.V.; Filgueiras, C.C.; Dória, J.; Peñaflor, M.F.G.V.; Willett, D.S. The Effects of Biostimulants on Induced Plant Defense. Front. Agron. 2021, 3, 630596. [Google Scholar] [CrossRef]
- Ramamoorthy, V.; Viswanathan, R.; Raguchander, T.; Prakasam, V.; Samiyappan, R. Induction of systemic resistance by plant growth promoting rhizobacteria in crop plants against pests and diseases. Crop Prot. 2001, 20, 1–11. [Google Scholar] [CrossRef]
- Ramette, A.; Frapolli, M.; Saux, M.F.-L.; Gruffaz, C.; Meyer, J.-M.; Défago, G.; Sutra, L.; Moënne-Loccoz, Y. Pseudomonas protegens sp. nov., widespread plant-protecting bacteria producing the biocontrol compounds 2,4-diacetylphloroglucinol and pyoluteorin. Syst. Appl. Microbiol. 2011, 34, 180–188. [Google Scholar] [CrossRef]
- Rasmann, S.; Bennett, A.; Biere, A.; Karley, A.; Guerrieri, E. Root symbionts: Powerful drivers of plant above- and belowground indirect defenses: Root symbionts and indirect defenses. Insect Sci. 2017, 24, 947–960. [Google Scholar] [CrossRef]
- Ruiu, L. Plant-Growth-Promoting Bacteria (PGPB) Against Insects and Other Agricultural Pests. Agronomy 2020, 10, 861. [Google Scholar] [CrossRef]
- Ryan, R.P.; Germaine, K.; Franks, A.; Ryan, D.J.; Dowling, D.N. Bacterial endophytes: Recent developments and applications. FEMS Microbiol. Lett. 2008, 278, 1–9. [Google Scholar] [CrossRef]
- Grobelak, A.; Kokot, P.; Hutchison, D.; Grosser, A.; Kacprzak, M. Plant growth-promoting rhizobacteria as an alternative to mineral fertilizers in assisted bioremediation—Sustainable land and waste management. J. Environ. Manag. 2018, 227, 1–9. [Google Scholar] [CrossRef]
- Bukhat, S.; Imran, A.; Javaid, S.; Shahid, M.; Majeed, A.; Naqqash, T. Communication of plants with microbial world: Exploring the regulatory networks for PGPR mediated defense signaling. Microbiol. Res. 2020, 238, 126486. [Google Scholar] [CrossRef]
- Majeed, A.; Kaleem Abbasi, M.; Hameed, S.; Yasmin, S.; Hanif, M.K.; Naqqash, T.; Imran, A. Pseudomonas sp. AF-54 containing multiple plant beneficial traits acts as growth enhancer of Helianthus annuus L. under reduced fertilizer input. Microbiol. Res. 2018, 216, 56–69. [Google Scholar] [CrossRef]
- Flury, P.; Aellen, N.; Ruffner, B.; Péchy-Tarr, M.; Fataar, S.; Metla, Z.; Dominguez-Ferreras, A.; Bloemberg, G.; Frey, J.; Goesmann, A.; et al. Insect pathogenicity in plant-beneficial pseudomonads: Phylogenetic distribution and comparative genomics. ISME J. 2016, 10, 2527–2542. [Google Scholar] [CrossRef]
- Ali, S.; Moon, Y.-S. Advances in PGPR-Mediated Plant-Pathogen Control for Food Security and Ecosystem Stability. Phyton 2025, 94, 1419–1451. [Google Scholar] [CrossRef]
- Garrido-Sanz, D.; Meier-Kolthoff, J.P.; Göker, M.; Martín, M.; Rivilla, R.; Redondo-Nieto, M. Genomic and Genetic Diversity Within the Pseudomonas fluorescens Complex. PLoS ONE 2016, 11, e0150183. [Google Scholar] [CrossRef]
- Tovi, N.; Frenk, S.; Hadar, Y.; Minz, D. Host Specificity and Spatial Distribution Preference of Three Pseudomonas Isolates. Front. Microbiol. 2019, 9, 3263. [Google Scholar] [CrossRef]
- Péchy-Tarr, M.; Bruck, D.J.; Maurhofer, M.; Fischer, E.; Vogne, C.; Henkels, M.D.; Donahue, K.M.; Grunder, J.; Loper, J.E.; Keel, C. Molecular analysis of a novel gene cluster encoding an insect toxin in plant-associated strains of Pseudomonas fluorescens. Environ. Microbiol. 2008, 10, 2368–2386. [Google Scholar] [CrossRef]
- Vesga, P.; Flury, P.; Vacheron, J.; Keel, C.; Croll, D.; Maurhofer, M. Transcriptome plasticity underlying plant root colonization and insect invasion by Pseudomonas protegens. ISME J. 2020, 14, 2766–2782. [Google Scholar] [CrossRef]
- Kupferschmied, P.; Péchy-Tarr, M.; Imperiali, N.; Maurhofer, M.; Keel, C. Domain Shuffling in a Sensor Protein Contributed to the Evolution of Insect Pathogenicity in Plant-Beneficial Pseudomonas protegens. PLoS Pathog. 2014, 10, e1003964. [Google Scholar] [CrossRef][Green Version]
- Kupferschmied, P.; Maurhofer, M.; Keel, C. Promise for plant pest control: Root-associated pseudomonads with insecticidal activities. Front. Plant Sci. 2013, 4, 287. [Google Scholar] [CrossRef]
- Vesga, P.; Augustiny, E.; Keel, C.; Maurhofer, M.; Vacheron, J. Phylogenetically closely related pseudomonads isolated from arthropods exhibit differential insect-killing abilities and genetic variations in insecticidal factors. Environ. Microbiol. 2021, 23, 5378–5394. [Google Scholar] [CrossRef] [PubMed]
- Aujoulat, F.; Pagès, S.; Masnou, A.; Emboulé, L.; Teyssier, C.; Marchandin, H.; Gaudriault, S.; Givaudan, A.; Jumas-Bilak, E. The population structure of Ochrobactrum isolated from Entomopathogenic nematodes indicates interactions with the symbiotic system. Infect. Genet. Evol. 2019, 70, 131–139. [Google Scholar] [CrossRef]
- Flury, P.; Vesga, P.; Dominguez-Ferreras, A.; Tinguely, C.; Ullrich, C.I.; Kleespies, R.G.; Keel, C.; Maurhofer, M. Persistence of root-colonizing Pseudomonas protegens in herbivorous insects throughout different developmental stages and dispersal to new host plants. ISME J. 2019, 13, 860–872. [Google Scholar] [CrossRef] [PubMed]
- Loper, J.E.; Hassan, K.A.; Mavrodi, D.V.; Davis, E.W.; Lim, C.K.; Shaffer, B.T.; Elbourne, L.D.H.; Stockwell, V.O.; Hartney, S.L.; Breakwell, K.; et al. Comparative Genomics of Plant-Associated Pseudomonas spp.: Insights into Diversity and Inheritance of Traits Involved in Multitrophic Interactions. PLoS Genet. 2012, 8, e1002784. [Google Scholar] [CrossRef]
- Paulsen, I.T.; Press, C.M.; Ravel, J.; Kobayashi, D.Y.; Myers, G.S.A.; Mavrodi, D.V.; DeBoy, R.T.; Seshadri, R.; Ren, Q.; Madupu, R.; et al. Complete genome sequence of the plant commensal Pseudomonas fluorescens Pf-5. Nat. Biotechnol. 2005, 23, 873–878. [Google Scholar] [CrossRef]
- Dominelli, N. Adaptation and Regulation of the Alternative Lifestyle of Insect Pathogenic Photorhabdus luminescens in the Soil and its Potential as Biocontrol Agent. 2022. Available online: https://openscience.ub.uni-mainz.de/handle/20.500.12030/7270 (accessed on 12 June 2025).
- Regaiolo, A.; Dominelli, N.; Andresen, K.; Heermann, R. The Biocontrol Agent and Insect Pathogen Photorhabdus luminescens Interacts with Plant Roots. Appl. Environ. Microbiol. 2020, 86, e00891-20. [Google Scholar] [CrossRef] [PubMed]
- Helmberger, M.S.; Shields, E.J.; Wickings, K.G. Ecology of belowground biological control: Entomopathogenic nematode interactions with soil biota. Appl. Soil Ecol. 2017, 121, 201–213. [Google Scholar] [CrossRef]
- Park, Y.-S.; Ryu, C.-M. Understanding Plant Social Networking System: Avoiding Deleterious Microbiota but Calling Beneficials. Int. J. Mol. Sci. 2021, 22, 3319. [Google Scholar] [CrossRef]
- Afridi, M.S.; Fakhar, A.; Kumar, A.; Ali, S.; Medeiros, F.H.V.; Muneer, M.A.; Ali, H.; Saleem, M. Harnessing microbial multitrophic interactions for rhizosphere microbiome engineering. Microbiol. Res. 2022, 265, 127199. [Google Scholar] [CrossRef]
- Ansari, M.; Devi, B.M.; Sarkar, A.; Chattopadhyay, A.; Satnami, L.; Balu, P.; Choudhary, M.; Shahid, M.A.; Jailani, A.A.K. Microbial Exudates as Biostimulants: Role in Plant Growth Promotion and Stress Mitigation. J. Xenobiotics 2023, 13, 572–603. [Google Scholar] [CrossRef]
- Martins, D.P.; Barros, M.T.; Pierobon, M.; Kandhavelu, M.; Lio’, P.; Balasubramaniam, S. Computational Models for Trapping Ebola Virus Using Engineered Bacteria. IEEE/ACM Trans. Comput. Biol. Bioinform. 2018, 15, 2017–2027. [Google Scholar] [CrossRef]
- Awan, H.; Adve, R.S.; Wallbridge, N.; Plummer, C.; Eckford, A.W. Communication and Information Theory of Single Action Potential Signals in Plants. IEEE Trans. NanoBiosci. 2019, 18, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Convertino, M. Inferring ecosystem networks as information flows. Sci. Rep. 2021, 11, 7094. [Google Scholar] [CrossRef] [PubMed]
- Bi, D.; Almpanis, A.; Noel, A.; Deng, Y.; Schober, R. A Survey of Molecular Communication in Cell Biology: Establishing a New Hierarchy for Interdisciplinary Applications. IEEE Commun. Surv. Tutor. 2021, 23, 1494–1545. [Google Scholar] [CrossRef]
- Unluturk, B.D.; Akyildiz, I.F. An End-to-End Model of Plant Pheromone Channel for Long Range Molecular Communication. IEEE Trans. NanoBiosci. 2017, 16, 11–20. [Google Scholar] [CrossRef]
- Soheilypour, M.; Mofrad, M.R.K. Agent-Based Modeling in Molecular Systems Biology. BioEssays 2018, 40, 1800020. [Google Scholar] [CrossRef] [PubMed]
- Le, D.-H.; Kwon, Y.-K. A coherent feedforward loop design principle to sustain robustness of biological networks. Bioinformatics 2013, 29, 630–637. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Azeloglu, E.U.; Iyengar, R. Good practices for building dynamical models in systems biology. Sci. Signal. 2015, 8, fs8. [Google Scholar] [CrossRef] [PubMed]
- Pineda, A.; Zheng, S.-J.; van Loon, J.J.; Pieterse, C.M.J.; Dicke, M. Helping plants to deal with insects: The role of beneficial soil-borne microbes. Trends Plant Sci. 2010, 15, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Zhan, J.; Gurung, S.; Parsa, S.P.K. Identification of top-K nodes in large networks using Katz centrality. J. Big Data 2017, 4, 16. [Google Scholar] [CrossRef]
- Dray, K.E.; Edelstein, H.I.; Dreyer, K.S.; Leonard, J.N. Control of mammalian cell-based devices with genetic programming. Curr. Opin. Syst. Biol. 2021, 28, 100372. [Google Scholar] [CrossRef]
- Ortiz-Romero, N.; Ochoa-Martínez, L.A.; González-Herrera, S.M.; Rutiaga-Quiñones, O.M.; Gallegos-Infante, J.A. Avances en las investigaciones sobre la encapsulación mediante gelación iónica: Una revisión sistemática. TecnoLógicas 2021, 24, e1962. [Google Scholar] [CrossRef]
- Thakur, S.; Sharma, B.; Verma, A.; Chaudhary, J.; Tamulevicius, S.; Thakur, V.K. Recent progress in sodium alginate based sustainable hydrogels for environmental applications. J. Clean. Prod. 2018, 198, 143–159. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed]
- Riseh, R.S.; Tamanadar, E.; Pour, M.M.; Thakur, V.K. Novel Approaches for Encapsulation of Plant Probiotic Bacteria with Sustainable Polymer Gums: Application in the Management of Pests and Diseases. Adv. Polym. Technol. 2022, 2022, 4419409. [Google Scholar] [CrossRef]
- Rojas-Sánchez, B.; Guzmán-Guzmán, P.; Morales-Cedeño, L.R.; Orozco-Mosqueda, M.d.C.; Saucedo-Martínez, B.C.; Sánchez-Yáñez, J.M.; Fadiji, A.E.; Babalola, O.O.; Glick, B.R.; Santoyo, G. Bioencapsulation of Microbial Inoculants: Mechanisms, Formulation Types and Application Techniques. Appl. Biosci. 2022, 1, 198–220. [Google Scholar] [CrossRef]
- Saberi Riseh, R.; Skorik, Y.A.; Thakur, V.K.; Moradi Pour, M.; Tamanadar, E.; Noghabi, S.S. Encapsulation of Plant Biocontrol Bacteria with Alginate as a Main Polymer Material. Int. J. Mol. Sci. 2021, 22, 11165. [Google Scholar] [CrossRef]
- Balla, A.; Silini, A.; Cherif-Silini, H.; Chenari Bouket, A.; Alenezi, F.N.; Belbahri, L. Recent Advances in Encapsulation Techniques of Plant Growth-Promoting Microorganisms and Their Prospects in the Sustainable Agriculture. Appl. Sci. 2022, 12, 9020. [Google Scholar] [CrossRef]
- Vargas, P.O.; Pereira, N.R.; Guimarães, A.O.; Waldman, W.R.; Pereira, V.R. Shrinkage and deformation during convective drying of calcium alginate. LWT 2018, 97, 213–222. [Google Scholar] [CrossRef]
- Saberi-Riseh, R.; Moradi-Pour, M.; Mohammadinejad, R.; Thakur, V.K. Biopolymers for Biological Control of Plant Pathogens: Advances in Microencapsulation of Beneficial Microorganisms. Polymers 2021, 13, 1938. [Google Scholar] [CrossRef]
- Saberi Riseh, R.; Hassanisaadi, M.; Vatankhah, M.; Kennedy, J.F. Encapsulating biocontrol bacteria with starch as a safe and edible biopolymer to alleviate plant diseases: A review. Carbohydr. Polym. 2023, 302, 120384. [Google Scholar] [CrossRef]
- Kim, J.; Hiltpold, I.; Jaffuel, G.; Sbaiti, I.; Hibbard, B.E.; Turlings, T.C.J. Calcium-alginate beads as a formulation for the application of Entomopathogenic nematodes to control rootworms. J. Pest Sci. 2021, 94, 1197–1208. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Martínez, H.; Ruiz-Vega, J.; Matadamas-Ortíz, P.T.; Cortés-Martínez, C.I.; Rosas-Diaz, J. Formulation of Entomopathogenic nematodes for crop pest control—A review. Plant Prot. Sci. 2017, 53, 15–24. [Google Scholar] [CrossRef]
- Gaugler, R.; Lewis, E.; Stuart, R.J. Ecology in the service of biological control: The case of Entomopathogenic nematodes. Oecologia 1997, 109, 483–489. [Google Scholar] [CrossRef]
- Pour, M.M.; Saberi-Riseh, R.; Mohammadinejad, R.; Hosseini, A. Investigating the formulation of alginate-gelatin encapsulated Pseudomonas fluorescens (VUPF5 and T17-4 strains) for controlling Fusarium solani on potato. Int. J. Biol. Macromol. 2019, 133, 603–613. [Google Scholar] [CrossRef] [PubMed]
- Panichikkal, J.; Prathap, G.; Nair, R.A.; Krishnankutty, R.E. Evaluation of plant probiotic performance of Pseudomonas sp. encapsulated in alginate supplemented with salicylic acid and zinc oxide nanoparticles. Int. J. Biol. Macromol. 2021, 166, 138–143. [Google Scholar] [CrossRef]
- Vejan, P.; Khadiran, T.; Abdullah, R.; Ismail, S.; Dadrasnia, A. Encapsulation of plant growth promoting Rhizobacteria—Prospects and potential in agricultural sector: A review. J. Plant Nutr. 2019, 42, 2600–2623. [Google Scholar] [CrossRef]
- Kaya, H.K.; Nelsen, C.E. Encapsulation of Steinernematid and Heterorhabditid nematodes with Calcium Alginate: A New Approach for Insect Control and Other Applications. Environ. Entomol. 1985, 14, 572–574. [Google Scholar] [CrossRef]
- Hiltpold, I.; Hibbard, B.E.; French, B.W.; Turlings, T.C.J. Capsules containing Entomopathogenic nematodes as a Trojan horse approach to control the western corn rootworm. Plant Soil 2012, 358, 11–25. [Google Scholar] [CrossRef]
- Kim, J.; Jaffuel, G.; Turlings, T.C.J. Enhanced alginate capsule properties as a formulation of Entomopathogenic nematodes. BioControl 2015, 60, 527–535. [Google Scholar] [CrossRef]
- Jaffuel, G.; Sbaiti, I.; Turlings, T.C.J. Encapsulated Entomopathogenic nematodes Can Protect Maize Plants from Diabrotica balteata Larvae. Insects 2019, 11, 27. [Google Scholar] [CrossRef]
- van Elsas, J.D.; Trevors, J.T.; Jain, D.; Wolters, A.C.; Heijnen, C.E.; van Overbeek, L.S. Survival of, and root colonization by, alginate-encapsulated Pseudomonas fluorescens cells following introduction into soil. Biol. Fertil. Soils 1992, 14, 14–22. [Google Scholar] [CrossRef]
- Russo, A.; Basaglia, M.; Tola, E.; Casella, S. Survival, root colonisation and biocontrol capacities of Pseudomonas fluorescens F113 LacZY in dry alginate microbeads. J. Ind. Microbiol. Biotechnol. 2001, 27, 337–342. [Google Scholar] [CrossRef]
- Heo, K.R.; Lee, K.Y.; Lee, S.H.; Jung, S.J.; Lee, S.W.; Moon, B.J. Control of Crisphead Lettuce Damping-off and Bottom Rot by Seed Coating with Alginate and Pseudomonas aeruginosa LY-11. Plant Pathol. J. 2008, 24, 67–73. [Google Scholar] [CrossRef][Green Version]
- Power, B.; Liu, X.; Germaine, K.J.; Ryan, D.; Brazil, D.; Dowling, D.N. Alginate beads as a storage, delivery and containment system for genetically modified PCB degrader and PCB biosensor derivatives of Pseudomonas fluorescens F113: Alginate beads; a delivery system for GM PCB degrading Pseudomonas. J. Appl. Microbiol. 2011, 110, 1351–1358. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Darwish, I.A.A.; Martins, D.P.; Ryan, D.; Kakouli-Duarte, T. State of the Art on the Interaction of Entomopathogenic Nematodes and Plant Growth-Promoting Rhizobacteria to Innovate a Sustainable Plant Health Product. Crops 2025, 5, 52. https://doi.org/10.3390/crops5040052
Darwish IAA, Martins DP, Ryan D, Kakouli-Duarte T. State of the Art on the Interaction of Entomopathogenic Nematodes and Plant Growth-Promoting Rhizobacteria to Innovate a Sustainable Plant Health Product. Crops. 2025; 5(4):52. https://doi.org/10.3390/crops5040052
Chicago/Turabian StyleDarwish, Islam Ahmed Abdelalim, Daniel P. Martins, David Ryan, and Thomais Kakouli-Duarte. 2025. "State of the Art on the Interaction of Entomopathogenic Nematodes and Plant Growth-Promoting Rhizobacteria to Innovate a Sustainable Plant Health Product" Crops 5, no. 4: 52. https://doi.org/10.3390/crops5040052
APA StyleDarwish, I. A. A., Martins, D. P., Ryan, D., & Kakouli-Duarte, T. (2025). State of the Art on the Interaction of Entomopathogenic Nematodes and Plant Growth-Promoting Rhizobacteria to Innovate a Sustainable Plant Health Product. Crops, 5(4), 52. https://doi.org/10.3390/crops5040052








