Threatened Habitats of Carnivores: Identifying Conservation Areas in Michoacán, México
Abstract
1. Introduction
1.1. Michoacán’s Mammals
1.2. Carnivores and Biodiversity Indicators
1.3. Species Distribution Models
1.4. Conservation Areas
1.5. Statement of the Problem
2. Materials and Methods
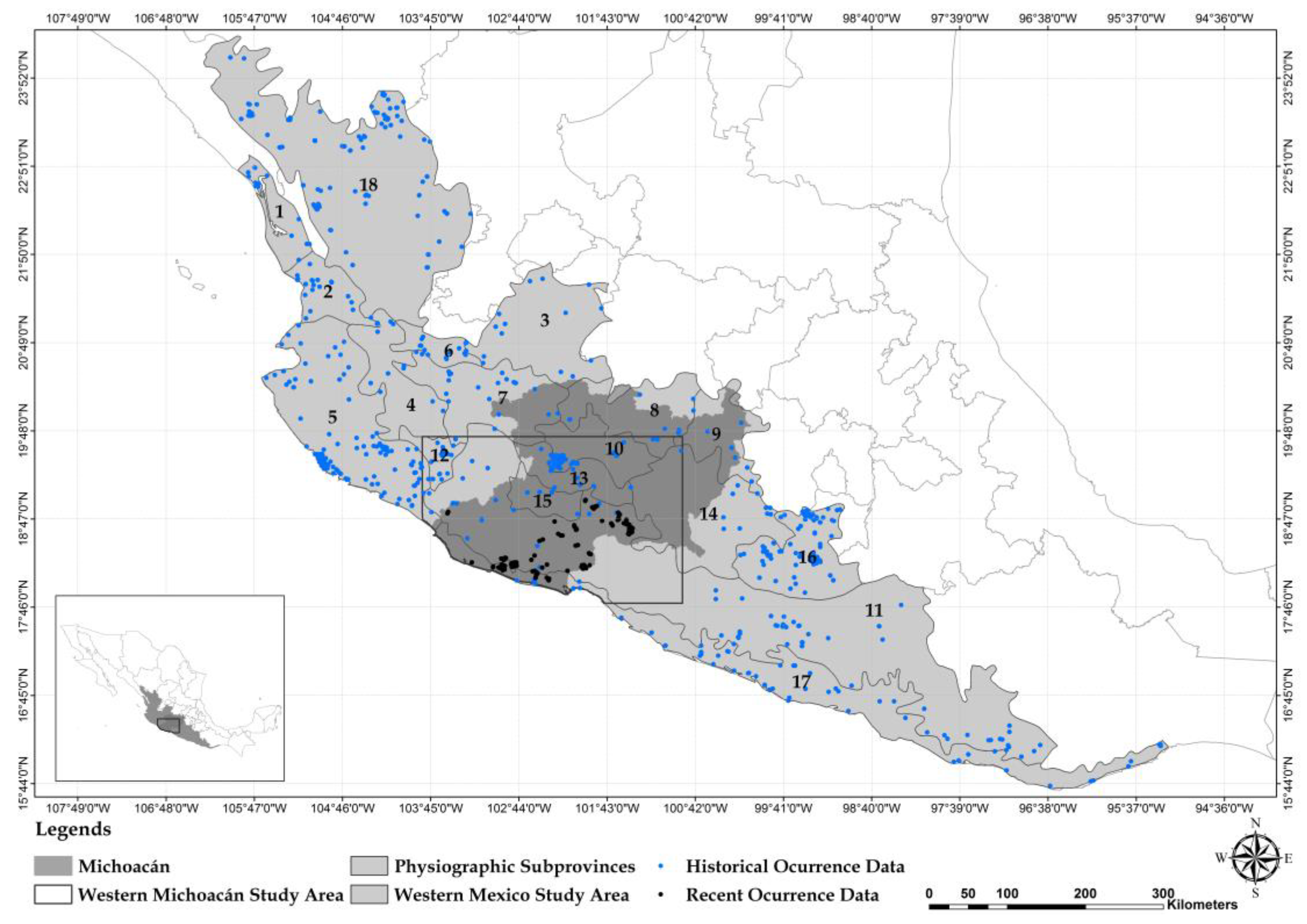
2.1. Carnivore Species Occurrence Data
2.2. Species Distribution Modeling
2.3. Prediction Variables
2.4. Bioclimatic Envelope Models
2.5. Habitat Suitability Models
2.6. Model Validation
2.7. Land-Use/Land-Cover Change
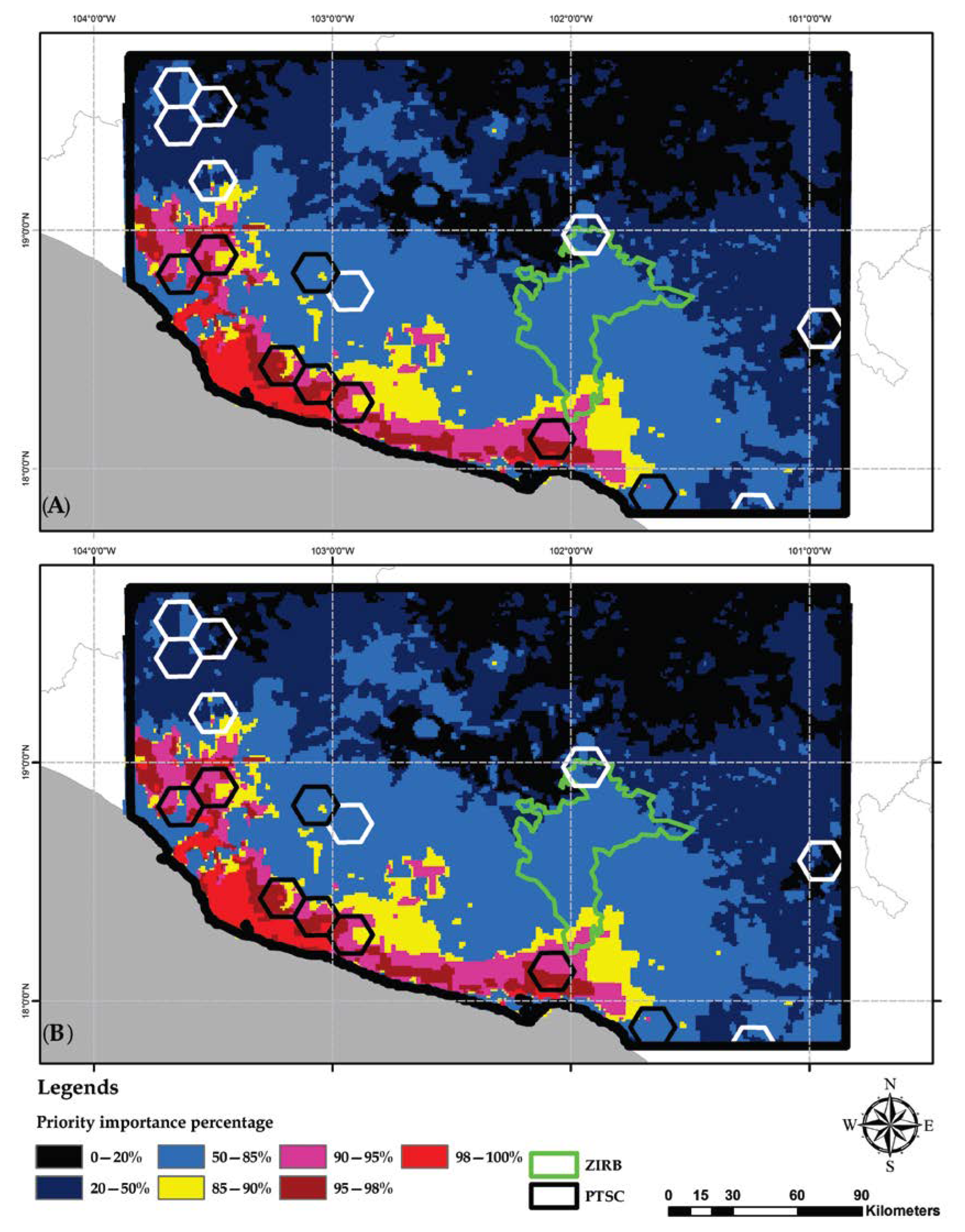
2.8. Identification of Priority Areas for Conservation
2.9. Comparing Identified and Already Proposed Priority Areas
3. Results
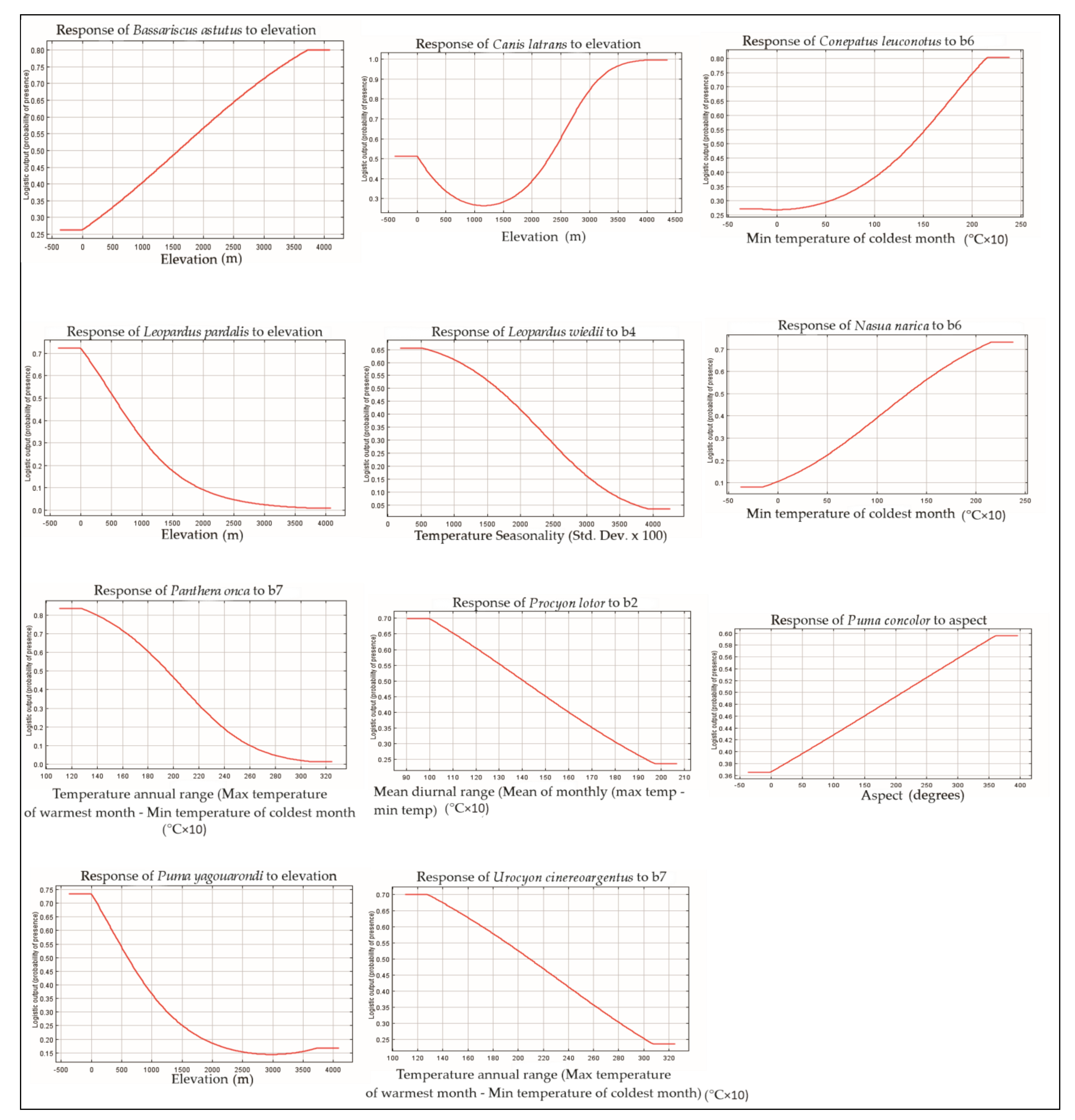
3.1. Variable Importance
3.2. Associations Species/Habitat Types
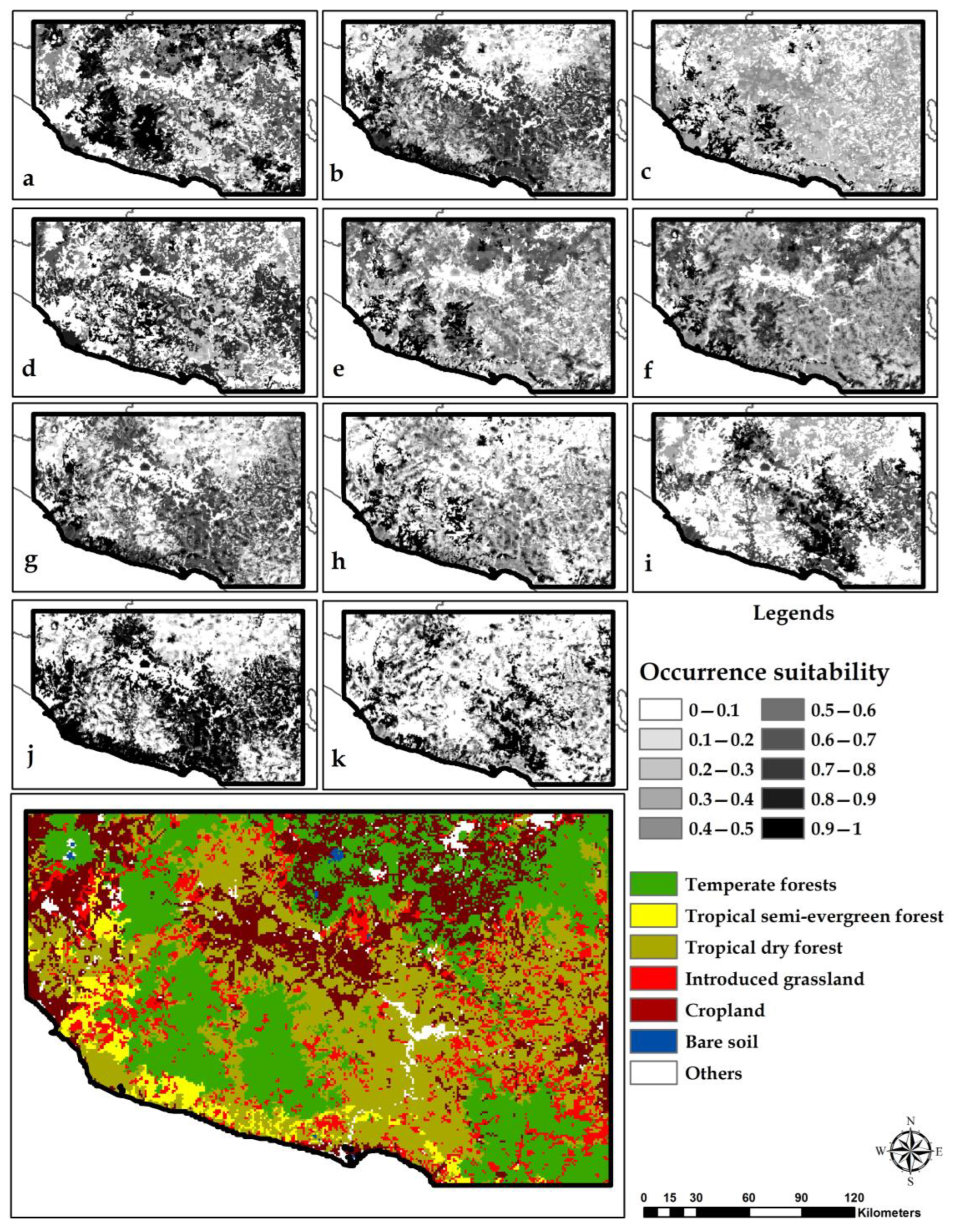
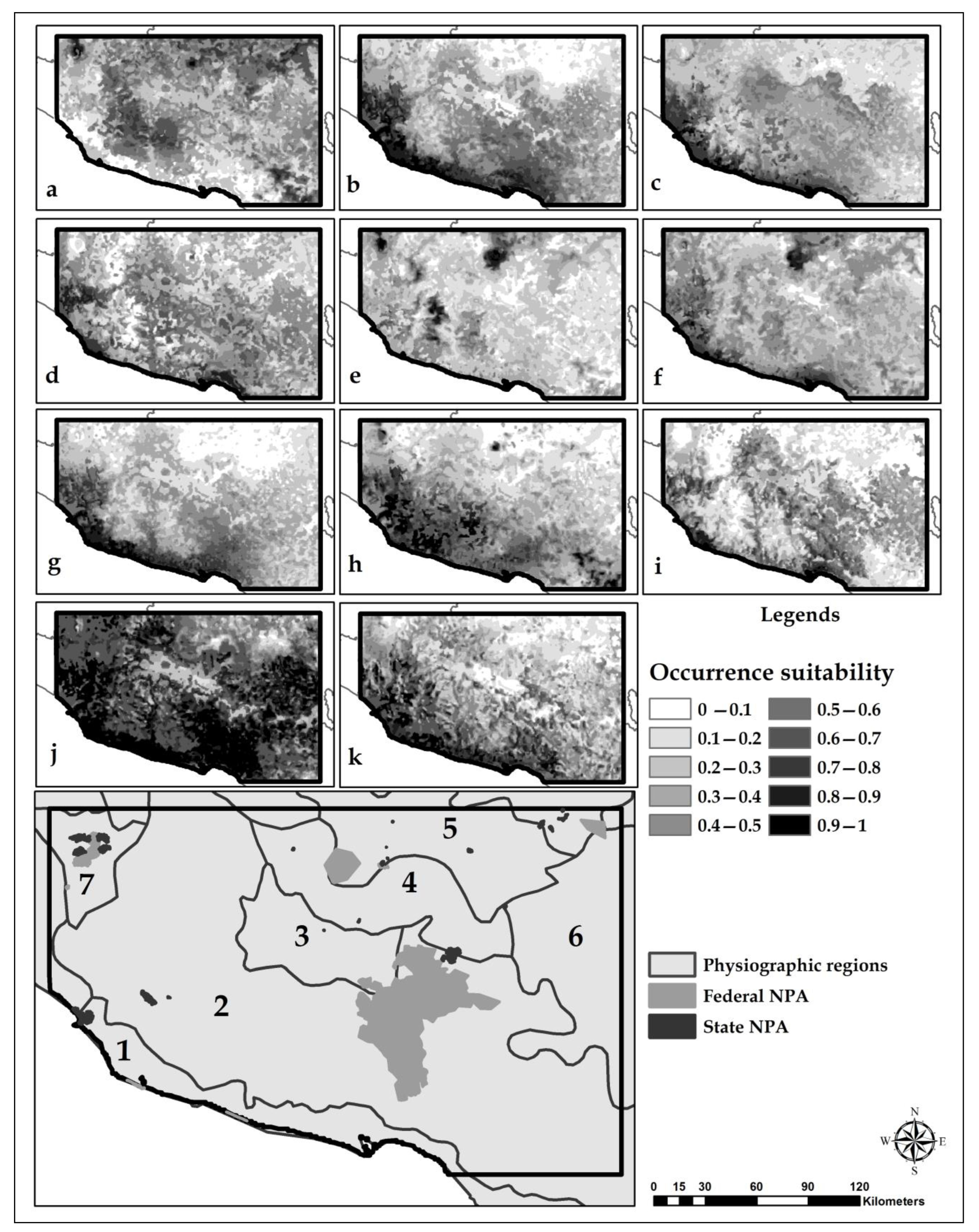
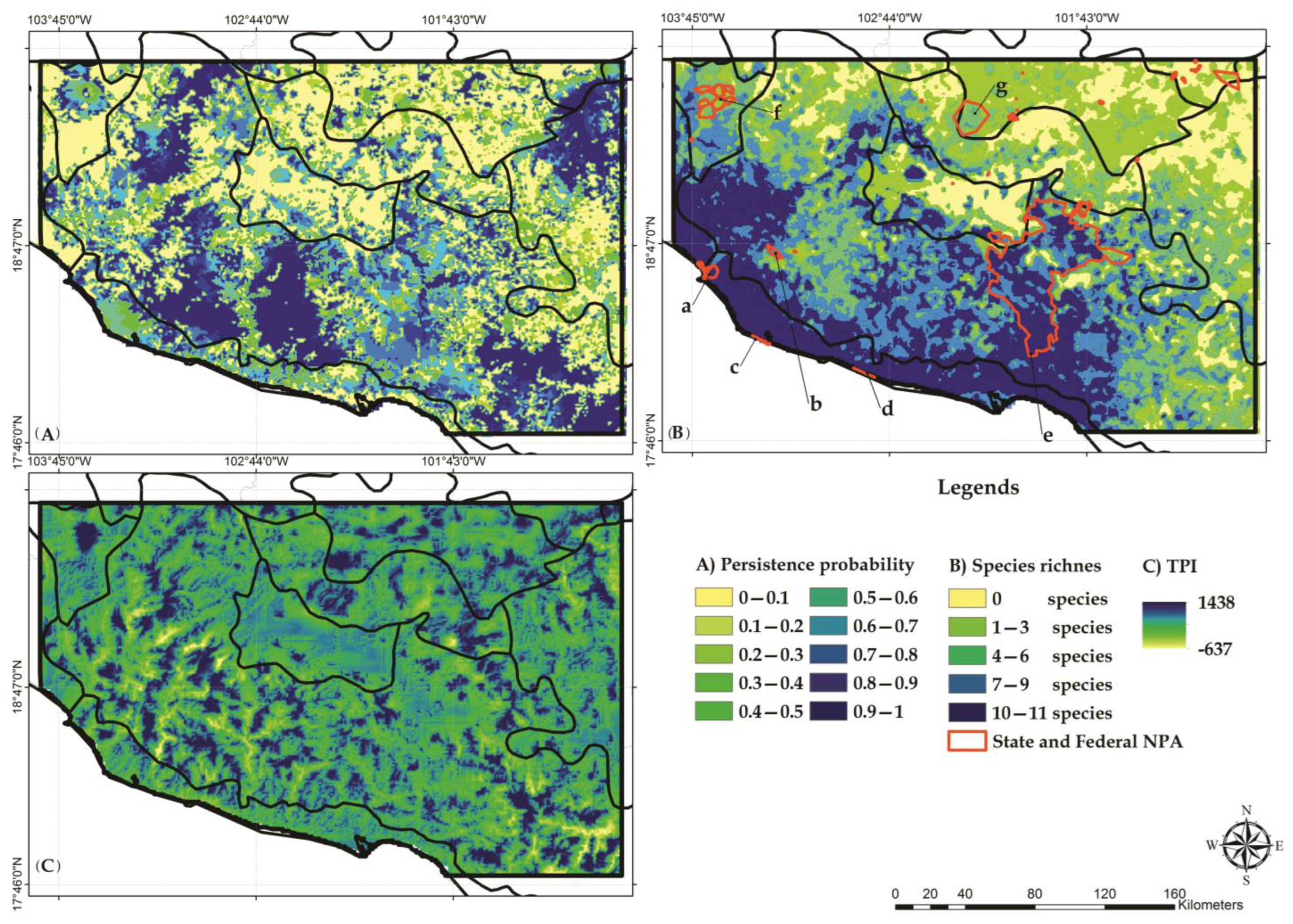
3.3. Habitat Suitability Models
3.4. Land-Use/Land-Cover Change
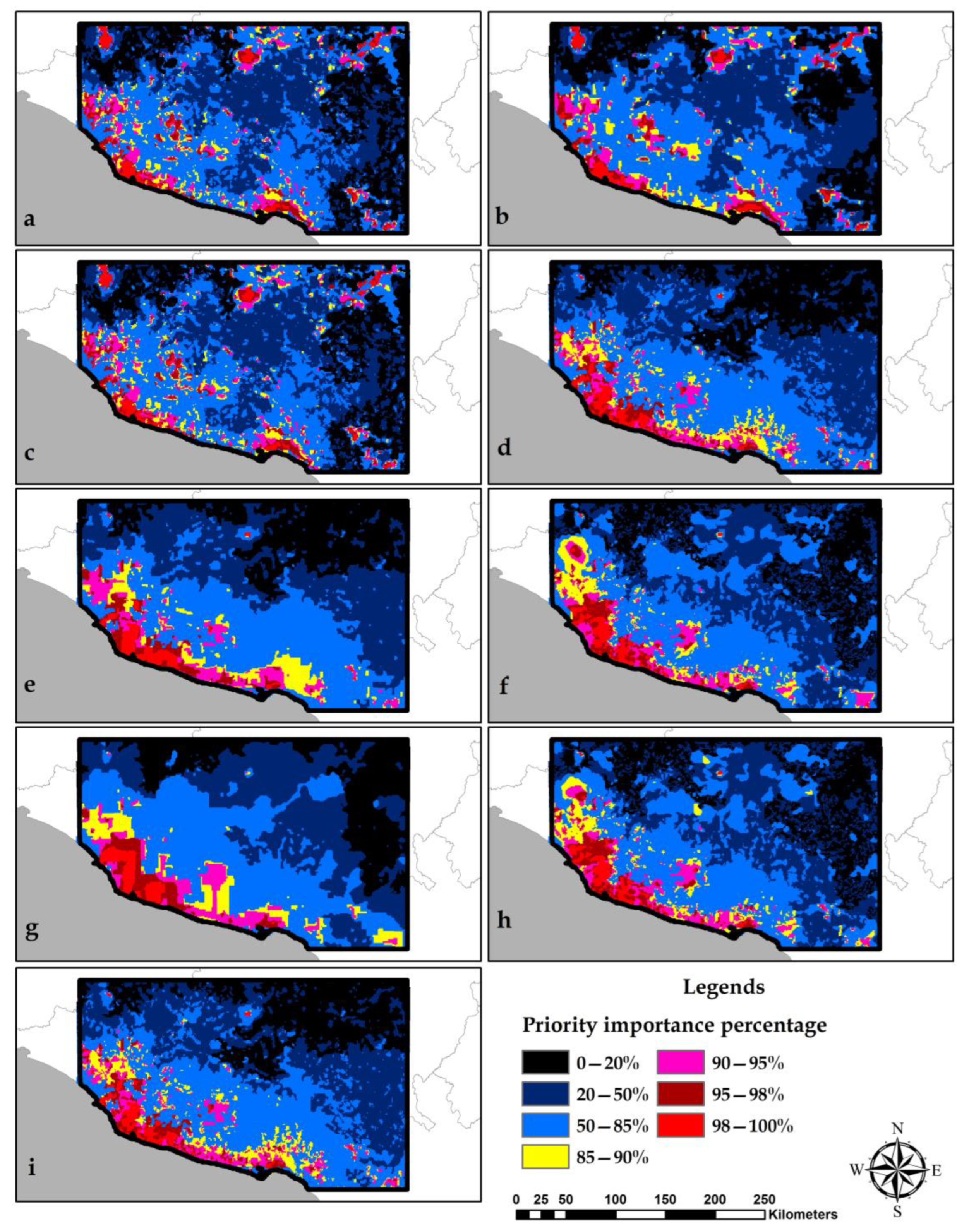
3.5. Identification of Conservation Priority Areas
4. Discussion
4.1. Modeling Scales
4.2. Suitable Species Habitat Models
4.3. Selection of Conservation Priority Areas
5. Study Limitations and Future Research
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Physiographic Province Name | English Translation | Dominant Elevation (m) | Dominant Slope (Degrees) |
|---|---|---|---|
| Delta del Río Grande de Santiago | Río Grande de Santiago Delta | 0–50 | 0–1 |
| Sierras Neovolcánicas Nayaritas | Neovolcanic mountain range in Nayarit state | 800–1000 | 4–8 |
| Altos de Jalisco | State of Jalisco’s highlands | 1800–2000 | 0–1 |
| Sierra de Jalisco | State of Jalisco’s mountain range | 1400–1600 | 8–16 |
| Sierras de la Costa de Jalisco y Colima | Coastal mountain range in the states of Jalisco and Colima | 200–400 | 8–16 |
| Guadalajara | Guadalajara | 1400–1600 | 0–1 |
| Chapala | Chapala | 1600–1800 | 0–1 |
| Sierras y Bajíos Michoacanos | Mountain range and lowlands in Michoacán state | 1800–2000 | 0–1 |
| Mil Cumbres | Thousand summits | 2200–2400 | 4–8 |
| Neovolcánica Tarasca | Neovolcanic Tarasca | 2200–2400 | 4–8 |
| Cordillera Costera del Sur | Southern coastal mountain range | 400–600 | 8–16 |
| Volcanes de Colima | State of Colima’s volcanos | 400–600 | 2–4 |
| Escarpa Limítrofe del Sur | Southern limiting escarpment | 1000–1200 | 4–8 |
| Depresión del Balsas | Balsas depression | 200–400 | 8–16 |
| Depresión de Tepalcatepec | Tepalcatepec depression | 200–400 | 0–1 |
| Sierras y Valles Guerrerenses | Mountain ranges and valleys in Guerrero state | 1000–1200 | 8–16 |
| Costas del Sur | Southern coasts | 50–100 | 0–20 |
| Mesetas y Cañadas del Sur | Southern plateau and ravines | 2000–2200 | 8–16 |
| Nuevo Atlas Nacional de México [117] | |||
| Family | Species | Common Name | Description |
|---|---|---|---|
| CANIDAE | Canis latrans Say, 1823 | Coyote | Medium-size canid. Social animals with twilight activity. They have a total length of between 1075 and 1150 mm and a weight between 8 and 16 kg. Generalist food habits with seasonal variations, including lagomorphs, rodents, ungulates, fruits, insects, reptiles, and birds. Their home range size is inverse to population density. For resident individuals, it is higher in adults than juveniles (5 vs. 2.4 km2); and it is even higher for transient individuals. They live in every vegetation type in México, particularly in plains with desert scrub and grassland. |
| Urocyon cinereoargenteus (Schreber, 1775) | Gray fox | Medium-size canid with a 500–600 mm total length and 3–5 kg weight. Opportunistic feeding habits, consuming rodents, lagomorphs, fruit, and insects. Home range varies from 1 to 8 km2, depending on the season, population density and habitat quality. They live mainly in forests and shrubland, but can also live in every vegetation type, from sea level to 3500 masl. | |
| FELIDAE | Herpailurus yagouaroundi (Lacépède, 1809) | Jaguarundi | As a felid, it is a small animal (888–1372 mm total length and 3.5–9 kg weight). It can be found in tropical evergreen and dry forests, but can also live in mangroves, cloud forests, desert scrubs, and occasionally in temperate forests, from sea level to 2000 masl. It is well adapted to arboreal environments and its food habits consist of a wide range of prey, including invertebrates, reptiles, birds, and small mammals. Its activity area is 88.8- 99.9 km2 for males and 20.1 km2 for females. |
| Leopardus pardalis (Linnaeus, 1758) | Ocelot | This is a medium-size felid (920–1367 mm total length and 6–15 kg weight). Lives mainly in tropical evergreen forests, tropical forests and mangroves, but also in cloud forests and desert scrubs occasionally. It can be found from sea level to 2000 masl. Prefers dense land-cover habitats and feeds on small and medium-size rodents, but also on invertebrates, reptiles, birds and other mammals, including the temazate deer. Its activity area varies from 3.5 to 17.7 km2 for males and 0.7–14.6 km2 for females. | |
| Leopartus wiedii (Schinz, 1821) | Margay | Small-size felid with 805–1300 mm total length and 3–5 kg weight. It is the most arboreal of México’s felids, because it sleeps, rests and hunts in trees. Lives in tropical evergreen and tropical dry forests, mangroves, and cloud forests, from sea level to 3000 masl. It feeds on invertebrates, birds, and small mammals, mainly rodents. | |
| Puma concolor (Linnaeus, 1771) | Mountain lion/cougar | This is a large-size felid with a 1100–2200 mm total length and 38–110 kg weight. They have solitary habits. They can be found in every vegetation formation of México, being the most abundant in temperate forests, from sea level to 3500 masl. This species is more tolerant to human presence than the jaguar and feeds on large rodents, armadillos, deer, peccaries and even rats and rabbits. Its activity area varies from 66 to 685 km2 for females and 152–826 km2 for males. | |
| Panthera onca (Linnaeus, 1758) | Jaguar | This is the largest felid in América, with a 1574 a 2419 mm total length and 36–158 kg weight. Lives mainly in tropical evergreen and tropical dry forests and mangroves, but also in cloud forests, thorny forests, desert scrubs, and temperate forests, from sea level to 2000 masl. It is considered as an opportunistic carnivore because its diet is broad and depends on prey availability and density. More than 85 species have been reported as part of its diet, including invertebrates, fish, reptiles, birds, and mammals. Its activity areas are 10–38 km2 for females and 28–90 km2 for males. | |
| MEPHITIDAE | Conepatus leuconotus (Lichtenstein, 1832) | White-backed skunk | These are the largest skunks with a size similar to a domestic cat. Its total length is 410–633 mm with a 900–4500 g. Lives in a variety of environments, from temperate to desert and tropical environments, including thorny forests, tropical dry forests, scrubland, grassland, temperate forests, and crop land, from sea level to 2500 masl. It feeds on invertebrates such as insects (mainly beetles), worms, fruits, small vertebrates, and occasionally carrion. They are solitary animals. In México, four individuals per square kilometer densities have been reported. |
| PROCYONIDAE | Bassariscus astutus (Lichtenstein, 1830) | Ringtail | This is a medium-size carnivore with a 616–811 mm total length and 870–1100 g weight. Lives in desert scrubs, temperate forests, tropical semi-desert, scrubland, and even urban parks, from sea level to 2880 masl. It is an omnivore and feeds mainly on small mammals, insects, fruits, birds, reptiles and occasionally nectar. They are solitary animals with nocturnal habits. Its activity area is variable and depends on habitat type, season, and sex. |
| Nasua narica (Linnaeus, 1766) | White-nosed coati | Procyonids with a size similar to a medium size dog (850–1.340 mm total length and 4–6 kg weight). It is a highly social species, gathering in groups of up to 20 individuals, made up of almost exclusively adult females and juveniles. It is an omnivore collector animal that feeds mainly on fruits and invertebrates such as insects, myriapods, arachnids, crustaceans, and annelids. To a lesser extent, they hunt terrestrial vertebrates, such as rodents, amphibians and reptiles. They can be found mainly in tropical evergreen and tropical dry forests, but they are also common in temperate forests and desert scrubs, from sea level to 2900 masl. | |
| Procyon lotor (Linnaeus, 1758) | Raccoon | Medium-size procyonid with a 603–950 mm total length and 3–9 kg weight. It has twilight and nocturnal habits. They have adapted to a wide variety of habitat types with permanent water bodies. They are more abundant in tropical evergreen and tropical dry forests, mangroves, and areas with aquatic and semi-aquatic vegetation associated with marshes, swamps, and wetlands, from the sea level to almost 3000 masl. It is an omnivore and feeds on a wide variety of plant and animal foods. It is considered as a solitary species and its activity area widely varies from 0.2 to 4946 ha, although a typical annual average is 65 ha for males and 39 ha for females. |
| Species | Sensibility/Specificity Maximization Threshold |
|---|---|
| Bassariscus astutus | 0.31 |
| Canis latrans | 0.24 |
| Conepatus leuconotus | 0.26 |
| Leopartus pardalis | 0.34 |
| Leopartus wiedii | 0.23 |
| Nasua narica | 0.44 |
| Puma concolor | 0.64 |
| Procyon lotor | 0.28 |
| Panthera onca | 0.32 |
| Herpailurus yagouaroundi | 0.33 |
| Urocyon cinereoargenteus | 0.39 |

References
- Cervantes-Zamora, Y.; Cornejo-Olgín, S.; Lucero-Márquez, R.; Espinoza-Rodríguez, J.; Miranda-Viquez, E.; Pineda-Velázquez, A. Provincias Fisiográficas de México; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad: Tlalpan, Mexico, 2001. [Google Scholar]
- Bocco, G.; Mendoza, M.E.; Velázquez, A.; Torres, A. La Regionalización Geomorfológica Como Una Alternativa de Regionalización Ecológica En México: El Caso de Michoacán de Ocampo. Investig. Geográficas 1999, 40, 7–22. [Google Scholar] [CrossRef]
- Rzedowski, J. Diversidad y Orígenes de La Flora Fanerogámica de México. Acta Botánica Mex. 1991, 14, 3–21. [Google Scholar] [CrossRef]
- Villaseñor Gómez, L.; Leal Nares, O.A. La Biodiversidad En Michoacán: Estudio de Estado; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad (CONABIO): Tlalpan, Mexico, 2005; ISBN 970-9000-28-4. [Google Scholar]
- Monterrubio-Rico, T.C.; Medellín, J.F.C.; Colín-Soto, C.Z.; Paniagua, L.L. Los Mamíferos de Michoacán. Rev. Mex. Mastozool. (Nueva Época) 2014, 4, 1–17. [Google Scholar]
- Gittleman, J.L.; Funk, S.M.; MacDonald, D.W.; Wayne, R.K. Carnivore Conservation; Cambridge University Press: Cambridge, UK, 2001; Volume 5. [Google Scholar]
- Ray, J.; Sunquist, M. Trophic Relations in a Community of African Rainforest Carnivores. Oecologia 2001, 127, 395–408. [Google Scholar] [CrossRef]
- Berger, J. Anthropogenic Extinction of Top Carnivores and Interspecific Animal Behaviour: Implications of the Rapid Decoupling of a Web Involving Wolves, Bears, Moose and Ravens. Proceedings of the Royal Society of London. Ser. B Biol. Sci. 1999, 266, 2261–2267. [Google Scholar] [CrossRef]
- Weber, W.; Rabinowitz, A. A Global Perspective on Large Carnivore Conservation. Conserv. Biol. 1996, 10, 1046–1054. [Google Scholar] [CrossRef]
- Wolf, C.; Ripple, W.J. Range Contractions of the World’s Large Carnivores. R. Soc. Open Sci. 2017, 4, 170052. [Google Scholar] [CrossRef]
- Ripple, W.J.; Estes, J.A.; Beschta, R.L.; Wilmers, C.C.; Ritchie, E.G.; Hebblewhite, M.; Berger, J.; Elmhagen, B.; Letnic, M.; Nelson, M.P.; et al. Status and Ecological Effects of the World’s Largest Carnivores. Science 2014, 343, 1241484. [Google Scholar] [CrossRef]
- Di Minin, E.; Slotow, R.; Hunter, L.T.B.; Montesino Pouzols, F.; Toivonen, T.; Verburg, P.H.; Leader-Williams, N.; Petracca, L.; Moilanen, A. Global Priorities for National Carnivore Conservation under Land Use Change. Sci. Rep. 2016, 6, 23814. [Google Scholar] [CrossRef]
- Ray, J.; Redford, K.H.; Steneck, R.; Berger, J. Large Carnivores and the Conservation of Biodiversity; Island Press: Washington, DC, USA, 2013; ISBN 978-1-59726-609-3. [Google Scholar]
- Terraube, J.; Van Doninck, J.; Helle, P.; Cabeza, M. Assessing the Effectiveness of a National Protected Area Network for Carnivore Conservation. Nat. Commun. 2020, 11, 2957. [Google Scholar] [CrossRef]
- Cardillo, M.; Purvis, A.; Sechrest, W.; Gittleman, J.L.; Bielby, J.; Mace, G.M. Human Population Density and Extinction Risk in the World’s Carnivores. PLOS Biology 2004, 2, e197. [Google Scholar] [CrossRef]
- Karanth, K.U.; Chellam, R. Carnivore Conservation at the Crossroads. Oryx 2009, 43, 1–2. [Google Scholar] [CrossRef]
- Franklin, J. Mapping Species Distributions: Spatial Inference and Prediction; Cambridge University Press: Cambridge, UK, 2010; ISBN 978-1-139-48529-6. [Google Scholar]
- Augustin, N.H.; Mugglestone, M.A.; Buckland, S.T. An Autologistic Model for the Spatial Distribution of Wildlife. J. Appl. Ecol. 1996, 33, 339–347. [Google Scholar] [CrossRef]
- Edwards, T.C., Jr.; Deshler, E.T.; Foster, D.; Moisen, G.G. Adequacy of Wildlife Habitat Relation Models for Estimating Spatial Distributions of Terrestrial Vertebrates. Conserv. Biol. 1996, 10, 263–270. [Google Scholar] [CrossRef]
- Brito, C.; Crespo, E.G.; Paulo, O.S. Modelling Wildlife Distributions: Logistic Multiple Regression vs. Overlap Analysis. Ecography 1999, 22, 251–260. [Google Scholar] [CrossRef]
- Manel, S.; Dias, J.M.; Buckton, S.T.; Ormerod, S.J. Alternative Methods for Predicting Species Distribution: An Illustration with Himalayan River Birds. J. Appl. Ecol. 1999, 36, 734–747. [Google Scholar] [CrossRef]
- Spitz, F.; Lek, S. Environmental Impact Prediction Using Neural Network Modelling. An Example in Wildlife Damage. J. Appl. Ecol. 1999, 36, 317–326. [Google Scholar] [CrossRef]
- Venier, L.A.; McKenney, D.W.; Wang, Y.; McKee, J. Models of Large-Scale Breeding-Bird Distribution as a Function of Macro-Climate in Ontario, Canada. J. Biogeogr. 1999, 26, 315–328. [Google Scholar] [CrossRef]
- Cowley, M.J.R.; Wilson, R.J.; León-Cortés, J.L.; Gutiérrez, D.; Bulman, C.R.; Thomas, C.D. Habitat-Based Statistical Models for Predicting the Spatial Distribution of Butterflies and Day-Flying Moths in a Fragmented Landscape. J. Appl. Ecol. 2000, 37, 60–72. [Google Scholar] [CrossRef]
- Jaberg, C.; Guisan, A. Modelling the Distribution of Bats in Relation to Landscape Structure in a Temperate Mountain Environment. J. Appl. Ecol. 2001, 38, 1169–1181. [Google Scholar] [CrossRef]
- Peterson, A.T. Predicting Species’ Geographic Distributions Based on Ecological Niche Modeling. Condor 2001, 103, 599–605. [Google Scholar] [CrossRef]
- Anderson, R.P.; Gómez-Laverde, M.; Peterson, A.T. Geographical Distributions of Spiny Pocket Mice in South America: Insights from Predictive Models. Glob. Ecol. Biogeogr. 2002, 11, 131–141. [Google Scholar] [CrossRef]
- Ball, L.; Peterson, A.; Cohoon, K. Predicting Distributions of Tropical Birds. Ibis 2002, 144, e27–e32. [Google Scholar]
- Vetaas, O.R. Realized and Potential Climate Niches: A Comparison of Four Rhododendron Tree Species. J. Biogeogr. 2002, 29, 545–554. [Google Scholar] [CrossRef]
- Guisan, A.; Hofer, U. Predicting Reptile Distributions at the Mesoscale: Relation to Climate and Topography. J. Biogeogr. 2003, 30, 1233–1243. [Google Scholar] [CrossRef]
- Elith, J.; Graham, C.H. Do They? How Do They? Why Do They Differ? On Finding Reasons for Differing Performances of Species Distribution Models. Ecography 2009, 32, 66–77. [Google Scholar] [CrossRef]
- Araújo, M.B.; New, M. Ensemble Forecasting of Species Distributions. Trends Ecol. Evol. 2007, 22, 42–47. [Google Scholar] [CrossRef]
- Peterson, A.T.; Soberón, J.; Pearson, R.G.; Anderson, R.P.; Martínez-Meyer, E.; Nakamura, M.; Araújo, M.B. Ecological Niches and Geographic Distributions (MPB-49); Princeton University Press: Princeton, NJ, USA, 2011; ISBN 978-0-691-13688-2. [Google Scholar]
- Peterson, A.T.; Soberón, J. Species Distribution Modeling and Ecological Niche Modeling: Getting the Concepts Right. Nat. Conserv. 2012, 10, 102–107. [Google Scholar] [CrossRef]
- Grinell, J. The Niche Relationship of California Thrsher. Auk 1917, 1, 64–82. [Google Scholar]
- Pressey, R.L. Ad Hoc Reservations: Forward or Backward Steps in Developing Representative Reserve Systems? Conserv. Biol. 1994, 8, 662–668. [Google Scholar] [CrossRef]
- Pressey, R.L.; Humphries, C.J.; Margules, C.R.; Vane-Wright, R.I.; Williams, P.H. Beyond Opportunism: Key Principles for Systematic Reserve Selection. Trends Ecol. Evol. 1993, 8, 124–128. [Google Scholar] [CrossRef]
- Cabeza, M.; Moilanen, A. Design of Reserve Networks and the Persistence of Biodiversity. Trends Ecol. Evol. 2001, 16, 242–248. [Google Scholar] [CrossRef]
- Prendergast, J.R.; Quinn, R.M.; Lawton, J.H. The Gaps between Theory and Practice in Selecting Nature Reserves. Conserv. Biol. 1999, 13, 484–492. [Google Scholar] [CrossRef]
- Stokland, J.N. Representativeness and Efficiency of Bird and Insect Conservation in Norwegian Boreal Forest Reserves. Conserv. Biol. 1997, 11, 101–111. [Google Scholar] [CrossRef]
- Pressey, R.L.; Possingham, H.P.; Day, J.R. Effectiveness of Alternative Heuristic Algorithms for Identifying Indicative Minimum Requirements for Conservation Reserves. Biol. Conserv. 1997, 80, 207–219. [Google Scholar] [CrossRef]
- Hopkinson, P.; Travis, J.M.J.; Prendergast, J.R.; Evans, J.; Gregory, R.D.; Telfer, M.G.; Williams, P.H. A Preliminary Assessment of the Contribution of Nature Reserves to Biodiversity Conservation in Great Britain. Anim. Conserv. Forum 2000, 3, 311–320. [Google Scholar] [CrossRef]
- Williams, P.; Gibbons, D.; Margules, C.; Rebelo, A.; Humphries, C.; Pressey, R. A Comparison of Richness Hotspots, Rarity Hotspots, and Complementary Areas for Conserving Diversity of British Birds. Conserv. Biol. 1996, 10, 155–174. [Google Scholar] [CrossRef]
- Margules, C.R.; Sarkar, S. Planeación Sistemática de La Conservación; UNAM-CONANP-CONABIO: Cuidad de México, México, 2009; ISBN 607-7607-12-6. [Google Scholar]
- Possingham, H.; Wilson, K.; Andelman, S.A.; Vynne, C.H. Protected Areas: Goals, Limitations, and Design. In Principles of Conservation Biology, 3rd ed.; Groom, M.J., Meffe, G.K., Carroll, R.C., Eds.; Sinauer Associates: Sunderland, MA, USA, 2006; pp. 507–549. ISBN 978-0-87893-518-5. [Google Scholar]
- Comisión Nacional de Áreas Naturales Protegidas (CONANP); Comisión Nacional de Áreas Naturales Protegidas (SIMEC). 2022. Available online: https://simec.conanp.gob.mx/ficha.php?anp=73®=11 (accessed on 21 March 2021).
- Arriaga Cabrera, L.; Espinoza Rodríguez, J.M.; Aguilar Zuñiga, C.; Martínez Romero, E.; Gómez Mendoza, L.; Loa Loza, E. Regiones Terrestres Prioritarias de México; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad (CONABIO): Tlalpan, Mexico, 2000; ISBN 970-9000-16-0. [Google Scholar]
- Arizmendi, C.; Márquez-Valdemar, L. Áreas de Importancia Para La Conservación de Las Aves En México (AICA); CIPAMEX-CONABIO-CCN-FMCN: Mexico City, México, 2000. [Google Scholar]
- CONABIO (Comisión Nacional para el Conocimiento y Uso de la Biodiversidad); CONANP (Comisión Nacional de Áreas Naturales Protegidas); TNC (The Nature Conservancy Program México); PRONATURA (Pronatura, A.C.); FCF (Facultad de Ciencias Forestales); UANL (Universidad Autónoma de Nuevo León, México). Análisis de Vacíos y Omisiones En Conservación de La Biodiversidad Terrestre de México: Espacios y Especies. 2007. Ciudad de México, México. Available online: https://simec.conanp.gob.mx/pdf_evaluacion/terrestre.pdf (accessed on 23 April 2021).
- UMSNH-SEDUE Catálogo Selecto de La Biodiversidad de Michoacán; Gobierno del Estado–Secretaría de Desarrollo Urbano y Ecología: Chihuahua, Mexico, 1999.
- Velázquez, A.; Sosa, N.; Navarrete, J.A.; Torres, A. Bases Para La Conformación Del Sistema de Áreas de Conservación Del Estado de Michoacán; Secretaría de Urbanismo y Medio Ambiente, Universidad Nacional Autónoma de México: Mexico City, México, 2005; ISBN 978-970-703-329-0. [Google Scholar]
- Vázquez, L.; Bustamante–Rodríguez, C.; Arce, D.B. Area Selection for Conservation of Mexican Mammals. Anim. Biodivers. Conserv. 2009, 32, 29–39. [Google Scholar] [CrossRef]
- Álvarez-Solórzano, T.; López-Vidal, J. Biodiversidad de Los Mamíferos En El Estado de Michoacán; Instituto Politécnico Nacional. Escuela Nacional de Ciencias Biológicas. Base de Datos SNIB2010-CONABIO Proyecto: Ciudad de México, Mexico, 1998. [Google Scholar]
- Trejo, C.O.; Campillo, A.C.; Pulido, J.R. Mammals from the Tarascan Plateau, Michoacán, México. Rev. Mex. Mastozool. (Nueva Época) 1999, 4, 53–68. [Google Scholar] [CrossRef]
- Charre-Medellín, J.F. Uso de Manantiales Por Los Mamíferos Silvestres En Bosques Tropicales de Michoacán; Maestría Institucional en Ciencias Biológicas, Universidad Michoacana de San Nicolás de Hidalgo: Morelia, Michoacán, 2012. [Google Scholar]
- Monterrubio-Rico, T.C.; Charre-Medellín, J.F.; Villanueva-Hernández, A.I.; León-Paniagua, L. Nuevos registros de la martucha (Potos flavus) para Michoacán, México, que establecen su límite de distribución al norte por el Pacífico. Rev. Mex. Biodivers. 2013, 84, 1002–1006. [Google Scholar] [CrossRef]
- Charre-Medellín, J.F.; Monterrubio-Rico, T.C.; Botello, F.J.; León-Paniagua, L.; Núñez, R. FIRST RECORDS OF JAGUAR (Panthera onca) From the State of Michoacán, México. Southwest. Nat. 2013, 58, 264–268. [Google Scholar] [CrossRef]
- Urrea-Galeano, L.A.; Rojas-López, M.; Sánchez-Sánchez, L.; Ibarra-Manríquez, G. Registro de Puma yagouaroundi En La Reserva de La Biosfera Zicuirán-Infiernillo, Michoacán. Rev. Mex. Biodivers. 2016, 87, 548–551. [Google Scholar] [CrossRef]
- Ramamoorthy, T.; Bye, R.; Lot, A.; Fa, J. Biological Diversity of México: Origins and Distribution; Oxford University Press: Oxford, UK, 1993. [Google Scholar]
- Ramírez-Pulido, J.; Arroyo-Cabrales, J.; Castro-Campillo, A. Estado Actual y Relación Nomenclatural de Los Mamíferos Terrestres de México. Acta Zool. Mex. 2005, 21, 21–82. [Google Scholar] [CrossRef]
- Ramırez-Pulido, J.; González-Ruiz, N.; Gardner, A.; Arroyo-Cabrales, J. List of Recent Land Mammals of México. Special Publications of the Museum of Texas Tech University; Texas Tech University: Lubbock, TX, USA, 2014. [Google Scholar]
- Ceballos, G.; Arroyo-Cabrales, J.; Medellin, R.A.; Domínguez-Castellanos, Y. Lista Actualizada de Los Mamíferos de México. Rev. Mex. Mastozool. 2005, 9, 21–71. [Google Scholar]
- Ceballos, G.; Arroyo-Cabrales, J. Lista Actualizada de Los Mamíferos de México. Rev. Mex. Mastozool. Nueva Época 2013, 2, 27–80. [Google Scholar] [CrossRef]
- Guo, Q.; Liu, Y. ModEco: An Integrated Software Package for Ecological Niche Modeling. Ecography 2010, 33, 637–642. [Google Scholar] [CrossRef]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-Km Spatial Resolution Climate Surfaces for Global Land Areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M. Modeling of Species Distributions with Maxent: New Extensions and a Comprehensive Evaluation. Ecography 2008, 31, 161–175. [Google Scholar] [CrossRef]
- Elith, J.; Graham, C.H.; Anderson, R.P.; Dudík, M.; Ferrier, S.; Guisan, A.; Hijmans, R.J.; Huettmann, F.; Leathwick, J.R.; Lehmann, A.; et al. Novel Methods Improve Prediction of Species’ Distributions from Occurrence Data. Ecography 2006, 29, 129–151. [Google Scholar] [CrossRef]
- Phillips, S.J.; Dudík, M.; Elith, J.; Graham, C.H.; Lehmann, A.; Leathwick, J.; Ferrier, S. Sample Selection Bias and Presence-Only Distribution Models: Implications for Background and Pseudo-Absence Data. Ecol. Appl. 2009, 19, 181–197. [Google Scholar] [CrossRef]
- Hirzel, A.H.; Helfer, V.; Metral, F. Assessing Habitat-Suitability Models with a Virtual Species. Ecol. Model. 2001, 145, 111–121. [Google Scholar] [CrossRef]
- Zaniewski, A.E.; Lehmann, A.; Overton, J.M. Predicting Species Spatial Distributions Using Presence-Only Data: A Case Study of Native New Zealand Ferns. Ecol. Model. 2002, 157, 261–280. [Google Scholar] [CrossRef]
- Engler, R.; Guisan, A.; Rechsteiner, L. An Improved Approach for Predicting the Distribution of Rare and Endangered Species from Occurrence and Pseudo-Absence Data. J. Appl. Ecol. 2004, 41, 263–274. [Google Scholar] [CrossRef]
- Instituto Nacional de Estadística y Geografía, (INEGI). Carta de Uso Del Suelo y Vegetación, Serie V, Escala 1: 250,000; Instituto Nacional de Estadística y Geografía, (INEGI): Aguascalientes, Mexico, 2014. [Google Scholar]
- Instituto Nacional de Estadística y Geografía, (INEGI) Guía Para La Interpretación de Cartografía de Uso de Suelo y Vegetación: Escala 1:250,000: Serie V. 2014. Available online: https://www.inegi.org.mx/contenidos/temas/mapas/usosuelo/metadatos/guia_interusosuelov.pdf (accessed on 15 April 2020).
- Jenness, J. Topographic Position Index (Tpi_jen. Avx_extension for Arcview 3. x, v. 1.3 a, Jenness Enterprises [EB/OL]. 2006. Available online: http://www.jennessent.com/arcview/tpi.htm (accessed on 16 June 2021).
- Peterson, A.T.; Papeş, M.; Soberón, J. Rethinking Receiver Operating Characteristic Analysis Applications in Ecological Niche Modeling. Ecol. Model. 2008, 213, 63–72. [Google Scholar] [CrossRef]
- Osorio-Olvera, L. NicheToolbox: A Web Tool for Exploratory Data Analysis and Niche Modeling. 2016. Available online: http://shiny.conabio.gob.mx:3838/nichetoolb2/ (accessed on 3 March 2021).
- Instituto Nacional de Estadística y Geografía, (INEGI). Conjunto de Datos Vectoriales de La Carta de Uso Del Suelo y Vegetación. Escala 1:250 000. Serie II. Continuo Nacional 2001. Available online: https://www.inegi.org.mx/app/biblioteca/ficha.html?upc=702825007021 (accessed on 5 May 2020).
- Instituto Nacional de Estadística y Geografía, (INEGI). Conjunto de Datos Vectoriales de La Carta de Uso Del Suelo y Vegetación. Escala 1:250 000. Serie V. Conjunto Nacional 2013. Available online: https://www.inegi.org.mx/app/biblioteca/ficha.html?upc=702825007024 (accessed on 5 May 2020).
- Jiménez-Valverde, A.; Lobo, J.M. Threshold Criteria for Conversion of Probability of Species Presence to Either–or Presence–Absence. Acta Oecologica 2007, 31, 361–369. [Google Scholar] [CrossRef]
- Moilanen, A. Landscape Zonation, Benefit Functions and Target-Based Planning: Unifying Reserve Selection Strategies. Biol. Conserv. 2007, 134, 571–579. [Google Scholar] [CrossRef]
- Moilanen, A.; Anderson, B.J.; Eigenbrod, F.; Heinemeyer, A.; Roy, D.B.; Gillings, S.; Armsworth, P.R.; Gaston, K.J.; Thomas, C.D. Balancing Alternative Land Uses in Conservation Prioritization. Ecol. Appl. 2011, 21, 1419–1426. [Google Scholar] [CrossRef]
- Laitila, J.; Moilanen, A. Use of Many Low-Level Conservation Targets Reduces High-Level Conservation Performance. Ecol. Model. 2012, 247, 40–47. [Google Scholar] [CrossRef]
- Secretaría de Medio Ambiente y Recursos Naturales, (SEMARNAT). Norma Oficial Mexicana NOM-059-SEMARNAT-2010; Secretaría de Medio Ambiente y Recursos Naturales, Diario Oficial 30 Diciembre 2010: Distrito Federal, México, 2010. [Google Scholar]
- Soberón, J. Grinnellian and Eltonian Niches and Geographic Distributions of Species. Ecol. Lett. 2007, 10, 1115–1123. [Google Scholar] [CrossRef]
- Soberón, J.M. Niche and Area of Distribution Modeling: A Population Ecology Perspective. Ecography 2010, 33, 159–167. [Google Scholar] [CrossRef]
- Soberón, J.; Peterson, A.T. Interpretation of Models of Fundamental Ecological Niches and Species’ Distributional Areas. Biodivers. Inform. 2005, 2, 1–10. [Google Scholar] [CrossRef]
- Peterson, A.T. Uses and Requirements of Ecological Niche Models and Related Distributional Models. Biodivers. Inform. 2006, 3, 59–72. [Google Scholar] [CrossRef]
- Guisan, A.; Thuiller, W. Predicting Species Distribution: Offering More than Simple Habitat Models. Ecol. Lett. 2005, 8, 993–1009. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Cordero, V.; Munguia, M.; Peterson, A.T. GIS-Based Predictive Biogeography in the Context of Conservation. In Frontiers of Biogeography: New Directions in the Geography of the Nature; Lomolino, M.V., Heany, L.R., Eds.; Sinauer: Sunderland, MA, USA, 2004; pp. 311–324. [Google Scholar]
- Sánchez-Cordero, V.; Illoldi-Rangel, P.; Linaje, M.; Sarkar, S.; Peterson, A.T. Deforestation and Extant Distributions of Mexican Endemic Mammals. Biol. Conserv. 2005, 126, 465–473. [Google Scholar] [CrossRef]
- Sánchez-Cordero, V.; Illoldi-Rangel, P.; Escalante, T.; Figueroa, F.; Rodríguez, G.; Linaje, M. Deforestation and Biodiversity Conservation in México. In Endangered Species: New Research; Columbus, A., y Kuznetsov, L., Eds.; Nova Science Publishers: New Haven, CT, USA, 2009; pp. 279–298. [Google Scholar]
- Ortega-Huerta, M.A.; Peterson, A.T. Modelling Spatial Patterns of Biodiversity for Conservation Prioritization in North-Eastern México. Divers. Distrib. 2004, 10, 39–54. [Google Scholar] [CrossRef]
- Botello, F.; Sánchez-Cordero, V.; Ortega-Huerta, M.A. Disponibilidad de hábitats adecuados para especies de mamíferos a escalas regional (estado de Guerrero) y nacional (México). Rev. Mex. Biodivers. 2015, 86, 226–237. [Google Scholar] [CrossRef]
- Gillingham, P.K.; Palmer, S.C.F.; Huntley, B.; Kunin, W.E.; Chipperfield, J.D.; Thomas, C.D. The Relative Importance of Climate and Habitat in Determining the Distributions of Species at Different Spatial Scales: A Case Study with Ground Beetles in Great Britain. Ecography 2012, 35, 831–838. [Google Scholar] [CrossRef]
- Rodas-Trejo, J.; Rebolledo, G.; Rau, J. Uso y Selección de Hábitat Por Mamíferos Carnívoros y Herbívoros En Bosque Nativo y Plantaciones Forestales Del Sur de Chile. Gestión Ambient. 2010, 19, 33–46. [Google Scholar]
- Briceño-Méndez, M.; Reyna-Hurtado, R.; Calmé, S.; García-Gil, G. Preferenciasde hábitat y abundancia relativa de Tayassu pecari en un área con cacería en la región de Calakmul, Campeche, México. Rev. Mex. Biodivers. 2014, 85, 242–250. [Google Scholar] [CrossRef]
- Carrillo-Reyna, N.; Reyna-Hurtado, R.; Schmook, B. Abundancia relativa y selección de hábitat de Tapirus bairdii en las reservas de Calakmul y Balam Kú, Campeche, México. Rev. Mex. Biodivers. 2015, 86, 202–207. [Google Scholar] [CrossRef]
- Pinto-Ledezma, J.N.; Sandoval, X.V.; Pérez, V.N.; Caballero, T.J.; Mano, K.; Pinto Viveros, M.A.; Sosa, R. Desarrollo de un modelo espacial explícito de hábitat para la paraba jacinta (Anodorhynchus hyacinthinus) en el Pantanal boliviano (Santa Cruz, Bolivia). Ecol. Boliv. 2014, 49, 51–64. [Google Scholar]
- Hirzel, A.; Guisan, A. Which Is the Optimal Sampling Strategy for Habitat Suitability Modelling. Ecol. Model. 2002, 157, 331–341. [Google Scholar] [CrossRef]
- Reese, G.C.; Wilson, K.R.; Hoeting, J.A.; Flather, C.H. Factors Affecting Species Distribution Predictions: A Simulation Modeling Experiment. Ecol. Appl. 2005, 15, 554–564. [Google Scholar] [CrossRef]
- Wisz, M.S.; Hijmans, R.J.; Li, J.; Peterson, A.T.; Graham, C.H.; Guisan, A.; Group, N.P.S.D.W. Effects of Sample Size on the Performance of Species Distribution Models. Divers. Distrib. 2008, 14, 763–773. [Google Scholar] [CrossRef]
- Anderson, R.P.; Martínez-Meyer, E. Modeling Species’ Geographic Distributions for Preliminary Conservation Assessments: An Implementation with the Spiny Pocket Mice (Heteromys) of Ecuador. Biol. Conserv. 2004, 116, 167–179. [Google Scholar] [CrossRef]
- de la Torre, J.A.; Torres-Knoop, L. DISTRIBUCIÓN POTENCIAL DEL PUMA (Puma concolor) EN EL ESTADO DE AGUASCALIENTES, MÉXICO. Rev. Mex. Mastozool. (Nueva Época) 2014, 4, 45–56. [Google Scholar] [CrossRef]
- Torres, R.; Jayat, J.P. Modelos Predictivos de Distribución Para Cuatro Especies de Mamíferos (Cingulata, Artiodactyla y Rodentia) Típicas Del Chaco En Argentina. Mastozool. Neotrop. 2010, 17, 335–352. [Google Scholar]
- Cuervo-Robayo, A.P.; Monroy-Vilchis, O. Distribución potencial del jaguar Panthera onca (Carnivora: Felidae) en Guerrero, México: Persistencia de zonas para su conservación. Rev. Biol. Trop. 2012, 60, 1357–1367. [Google Scholar] [CrossRef]
- Charre-Medellín, J.F.; Monterrubio-Rico, T.C.; Guido-Lemus, D.; Mendoza, E.; Charre-Medellín, J.F.; Monterrubio-Rico, T.C.; Guido-Lemus, D.; Mendoza, E. Patrones de distribución de felinos silvestres (Carnívora: Felidae) en el trópico seco del Centro-Occidente de México. Rev. Biol. Trop. 2015, 63, 783–797. [Google Scholar] [CrossRef]
- Yañez-Arenas, C.; Peterson, A.T.; Mokondoko, P.; Rojas-Soto, O.; Martínez-Meyer, E. The Use of Ecological Niche Modeling to Infer Potential Risk Areas of Snakebite in the Mexican State of Veracruz. PLoS ONE 2014, 9, e100957. [Google Scholar] [CrossRef]
- Ceballos, G.; Blanco, S.; González, C.; Martínez, E. Modelado de La Distribución de Las Especies de Mamíferos de México Para Un Análisis GAP; Informe final SNIB-CONABIO proyecto DS006; EcoCiencia SC: Ciudad de México, México, 2008. [Google Scholar]
- Ceballos, G.; Oliva, G. Los Mamíferos Silvestres de México; CONABIO/Fondo de Cultura Económica: Mexico City, México, 2005. [Google Scholar]
- Nelson, E.W.; Goldman, E.A. Two New Cacomistles from México, with Remarks on the Genus Jentinkia. J. Wash. Acad. Sci. 1932, 22, 484–488. [Google Scholar]
- Burt, W.H. Some Effects of Volcan Paricutin on Vertebrates. Occasional Papers of the Museum of Zoology. Univ. Mich. 1961, 620, 1–24. [Google Scholar]
- Guido-Lemus, D. Riqueza de La Comunidad de Los Mamíferos Silvestres de La Cuenca Del Lago de Cuitzeo, Michoacán, Una Comparación Utilizando Métodos de Muestreo. Tesis de Licenciatura; Facultad de Biología, Universidad Michoacana de San Nicolás de Hidalgo: Morelia, Michoacán, México, 2012. [Google Scholar]
- Bocco, G.; Mendoza, M.; Masera, O. La Dinamica Del Cambio Del Uso de Suelo En Michoacán. Una Propuesta Metodológica Para el Estudio de Los Procesos de Deforestación; Instituto de Geografía-UNAM: Mexico City, México, 2001. [Google Scholar]
- Moilanen, A.; Franco, A.M.A.; Early, R.I.; Fox, R.; Wintle, B.; Thomas, C.D. Prioritizing Multiple-Use Landscapes for Conservation: Methods for Large Multi-Species Planning Problems. Proc. R. Soc. B Biol. Sci. 2005, 272, 1885–1891. [Google Scholar] [CrossRef] [PubMed]
- Comisión Nacional de Áreas Naturales Protegidas, (CONANP) CONANP 2018. Available online: https://simec.conanp.gob.mx/ficha.php?anp=47®=11 (accessed on 23 May 2021).
- Comisión Nacional de Áreas Naturales Protegidas, (CONANP) CONANP 2018. Available online: https://simec.conanp.gob.mx/ficha.php?anp=52®=11 (accessed on 23 May 2021).
- Coll-Hurtado, A. Nuevo Atlas Nacional de México; Universidad Nacional Autónoma de México: Mexico City, Mexico, 2007; ISBN 978-970-32-5047-9. [Google Scholar]


| Family | Species | Conservation Status | Number of Records | |||
|---|---|---|---|---|---|---|
| SEMARNAT a | CITES b | IUCN c | wMex | wMich | ||
| CANIDAE | Canis latrans (Say, 1823) | 216 | 25 | |||
| Urocyon cinereoargenteus (Schreber, 1775) | 366 | 20 | ||||
| FELIDAE | Herpailurus yagouaroundi (Lacépède, 1809) | A | I | EN | 91 | 6 |
| Leopardus pardalis (Linnaeus, 1758) | P | I | EN | 127 | 43 | |
| Leopartus wiedii (Schinz, 1821) | P | I | NT | 77 | 11 | |
| Puma concolor (Linnaeus, 1771) | 97 | 33 | ||||
| Panthera onca (Linnaeus, 1758) | P | NT | 84 | 18 | ||
| MEPHITIDAE | Conepatus leuconotus (Lichtenstein, 1832) | 231 | 6 | |||
| PROCYONIDAE | Bassariscus astutus (Lichtenstein, 1830) | 239 | 6 | |||
| Nasua narica (Linnaeus, 1766) | III | 376 | 81 | |||
| Procyon lotor (Linnaeus, 1758) | 242 | 14 | ||||
| TOTAL RECORDS | 2146 | 263 | ||||
| Species (Order Carnivora) | Prediction Variables | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioclimatic Variables | Topo | ||||||||||||||||||||
| b1 | b2 | b3 | b4 | b5 | b6 | b7 | b8 | b9 | b10 | b11 | b12 | b13 | b14 | b15 | b16 | b17 | b18 | b19 | asp | ele | |
| Canis latrans (Say, 1823) | X | X | X | X | X | X | X | X | X | X | X | ||||||||||
| Urocyon cinereoargenteus (Schreber, 1775) | X | X | X | X | X | X | X | X | X | X | |||||||||||
| Herpailurus yagouaroundi (Lacépède, 1809) | X | X | X | X | X | X | X | X | X | ||||||||||||
| Leopardus pardalis (Linnaeus, 1758) | X | X | X | X | X | X | X | X | |||||||||||||
| Leopartus wiedii (Schinz, 1821) | X | X | X | X | X | X | |||||||||||||||
| Puma concolor (Linnaeus, 1771) | X | X | X | X | X | X | X | ||||||||||||||
| Panthera onca (Linnaeus, 1758) | X | X | X | X | X | X | X | ||||||||||||||
| Conepatus leuconotus (Lichtenstein, 1832) | X | X | X | X | X | X | X | X | X | ||||||||||||
| Bassariscus astutus (Lichtenstein, 1830) | X | X | X | X | X | X | X | X | X | X | |||||||||||
| Nasua narica (Linnaeus, 1766) | X | X | X | X | X | X | X | X | X | X | X | X | |||||||||
| Procyon lotor (Linnaeus, 1758) | X | X | X | X | X | X | X | ||||||||||||||
| SPECIES | Conservation Status (NOM059) | Weight |
|---|---|---|
| Canis latrans (Say, 1823 | - | 1 |
| Urocyon cinereoargenteus (Schreber, 1775) | - | 1 |
| Herpailurus yagouaroundi (Lacépède, 1809) | T | 2 |
| Leopardus pardalis (Linnaeus, 1758) | EN | 3 |
| Leopartus wiedii (Schinz, 1821) | EN | 3 |
| Puma concolor (Linnaeus, 1771) | - | 1 |
| Panthera onca (Linnaeus, 1758) | EN | 3 |
| Conepatus leuconotus (Lichtenstein, 1832) | - | 1 |
| Bassariscus astutus (Lichtenstein, 1830) | - | 1 |
| Nasua narica (Linnaeus, 1766) | - | 1 |
| Procyon lotor (Linnaeus, 1758) | - | 1 |
| SPECIES (ORDER CARNIVORA) | Dispersion Parameters | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Presence/Absence Records | Significant Variables | Deviance Null | Deviance Residual | Deviance (D2) | Estimate | AIC | Z Value | p Value | |
| Canis latrans | 25/25 | TPI | 205.1 | 143.2 | 30.1% | 0.002014 | 171.2 | 1.841 | 0.0656 |
Urocyon cinereoargenteus | 20/20 | TPI/ Oak Forest | 213.4 | 164.9 | 22.7% | 0.00322/ −2.747 | 190.9 | 3.142 | 0.00168/ 0.092 |
| Herpailurus yagouaroundi | 6/6 | (-) | 30.4 | 22.7 | 25.5% | - | 38.7 | (-) | (-) |
| Leopardus pardalis | 43/43 | TPI | 155.2 | 105.7 | 31.8% | 0.003406 | 137.7 | 2.365 | 0.018 |
| Leopartus wiedii | 11/11 | TPI | 72.0 | 34.3 | 52.3% | 0.004993 | 60.3 | 1.942 | 0.0522 |
| Puma concolor | 33/33 | TPI | 130.3 | 44.8 | 65.5% | 0.008745 | 78.8 | 3.231 | 0.00123 |
| Panthera onca | 18/18 | TPI | 55.4 | 20.6 | 62.6% | 0.00834 | 42.6 | 1.719 | 0.0855 |
| Conepatus leuconotus | 6/6 | (-) | 63.7 | 36.7 | 42.4% | - | 60.7 | (-) | (-) |
| Bassariscus astutus | 6/6 | (-) | 77.6 | 39.9 | 48.5% | - | 65.9 | (-) | (-) |
| Nasua narica | 81/81 | TPI | 291.1 | 179.2 | 38.4% | 0.003061 | 215.2 | 2.942 | 0.00327 |
| Procyon lotor | 14/14 | (-) | 88.7 | 57.4 | 35.2% | - | 85.4 | (-) | (-) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Del Moral-Alvarez, M.; Ortega-Huerta, M.A.; Nuñez, R. Threatened Habitats of Carnivores: Identifying Conservation Areas in Michoacán, México. Conservation 2023, 3, 247-276. https://doi.org/10.3390/conservation3010018
Del Moral-Alvarez M, Ortega-Huerta MA, Nuñez R. Threatened Habitats of Carnivores: Identifying Conservation Areas in Michoacán, México. Conservation. 2023; 3(1):247-276. https://doi.org/10.3390/conservation3010018
Chicago/Turabian StyleDel Moral-Alvarez, Marisol, Miguel A. Ortega-Huerta, and Rodrigo Nuñez. 2023. "Threatened Habitats of Carnivores: Identifying Conservation Areas in Michoacán, México" Conservation 3, no. 1: 247-276. https://doi.org/10.3390/conservation3010018
APA StyleDel Moral-Alvarez, M., Ortega-Huerta, M. A., & Nuñez, R. (2023). Threatened Habitats of Carnivores: Identifying Conservation Areas in Michoacán, México. Conservation, 3(1), 247-276. https://doi.org/10.3390/conservation3010018






